Preparation and Characterization of Starch/Empty Fruit Bunch-Based Bioplastic Composites Reinforced with Epoxidized Oils
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Epoxidation of Palm Oil (PO) and Soybean Oil (SO)
2.2.2. Preparation of Starch/Empty Fruit Bunch-Based Bioplastic Composites
2.3. Fourier Transform Infrared Spectroscopy (FTIR)
2.4. Scanning Electron Microscopy (SEM)
2.5. Diffraction Scanning Calorimetry (DSC)
2.6. Thermogravimetric Analysis (TGA)
2.7. Tensile Properties
2.8. Water Vapor Permeability (WVP)
2.9. Water Uptake and Solubility
2.10. Statistical Analysis
3. Results and Discussion
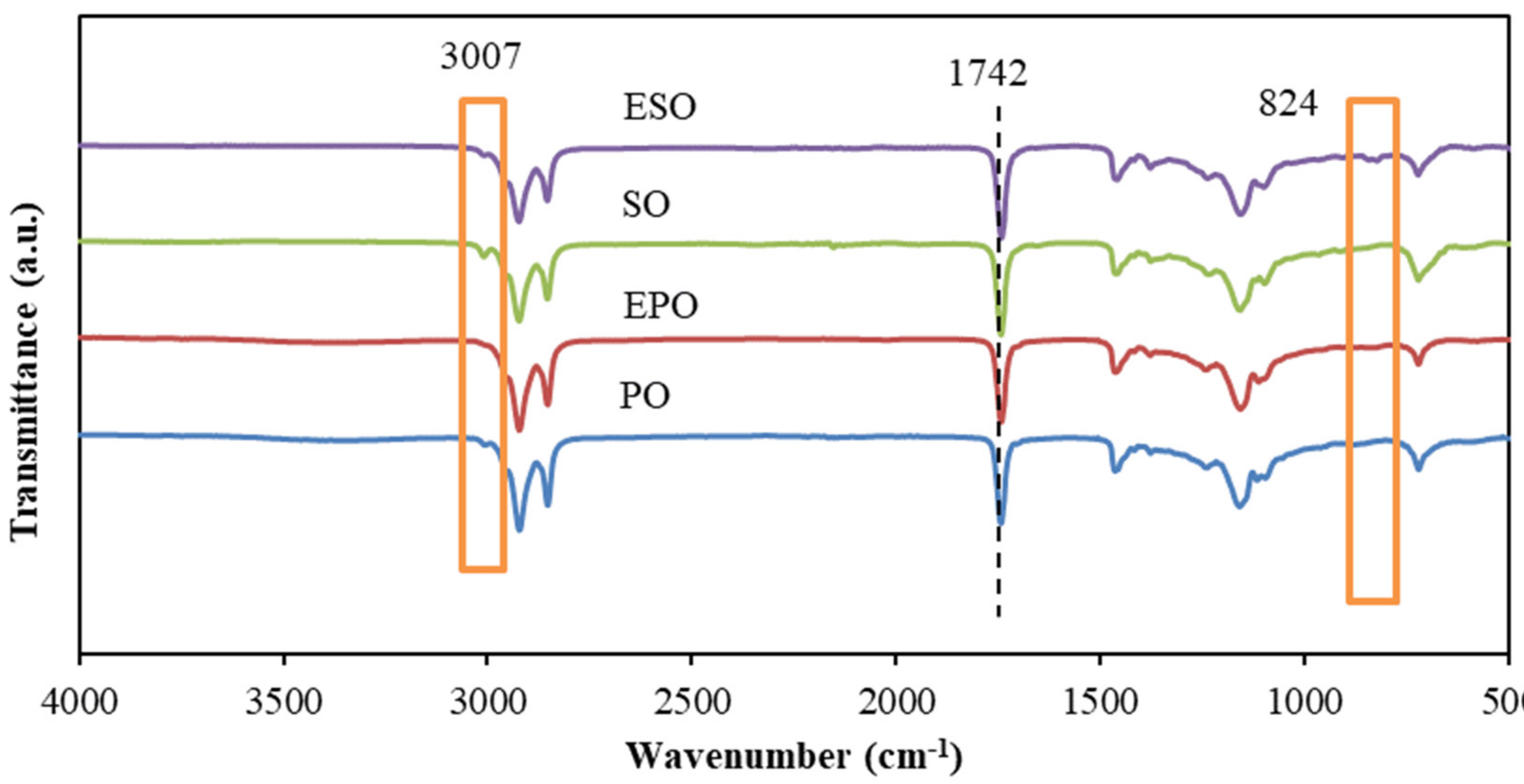
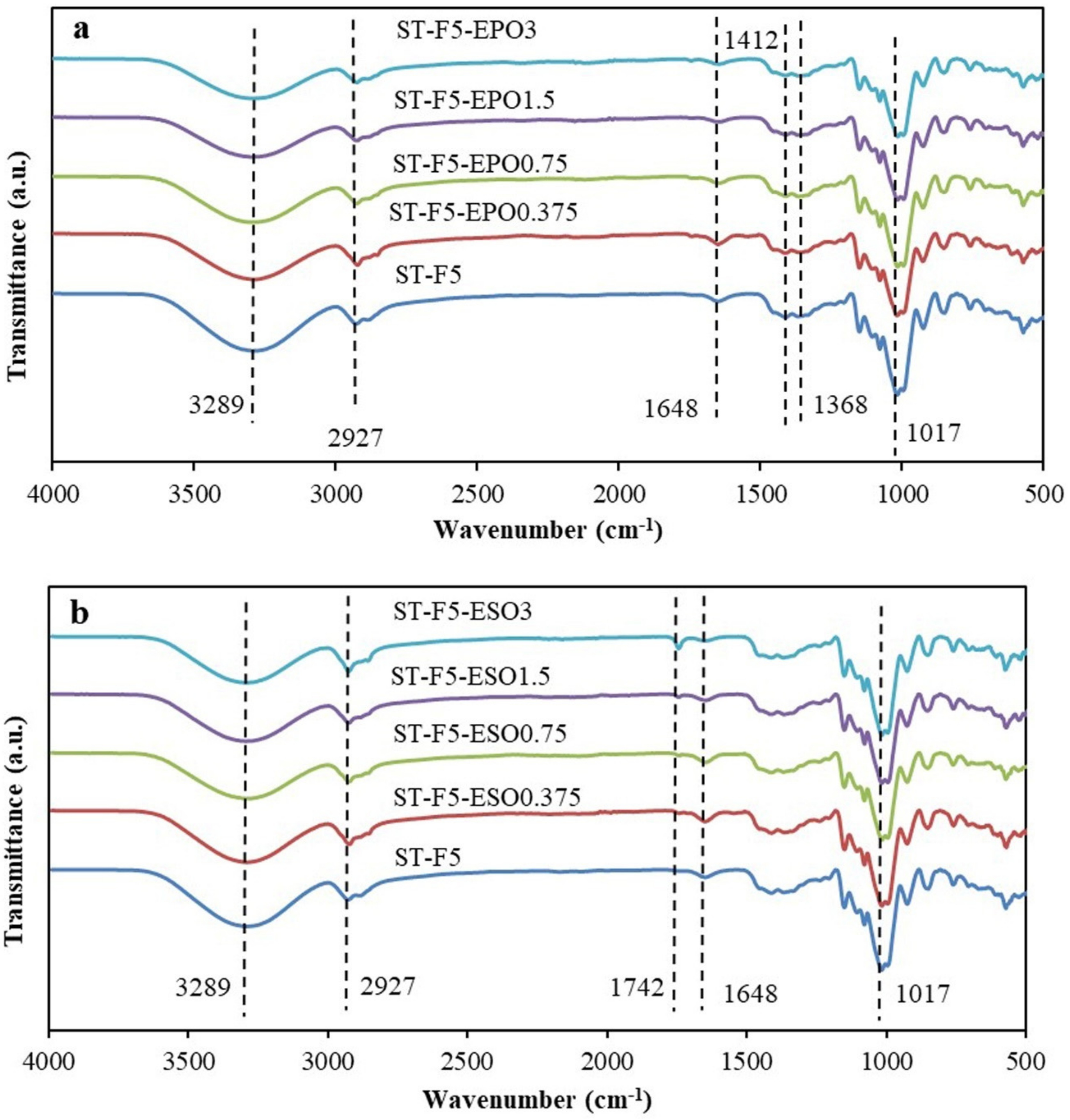
3.1. FTIR
3.2. SEM
3.3. DSC
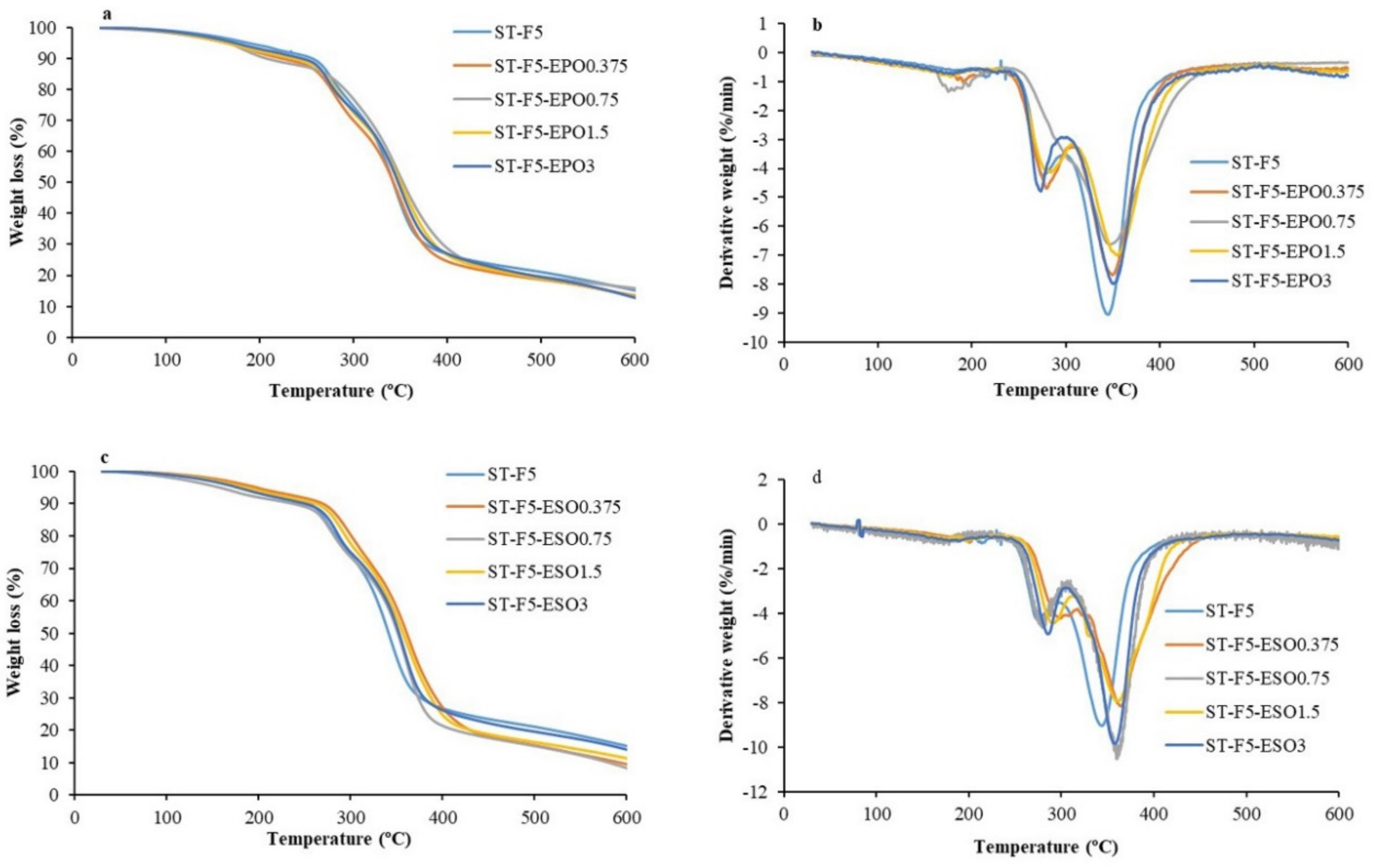
3.4. TGA
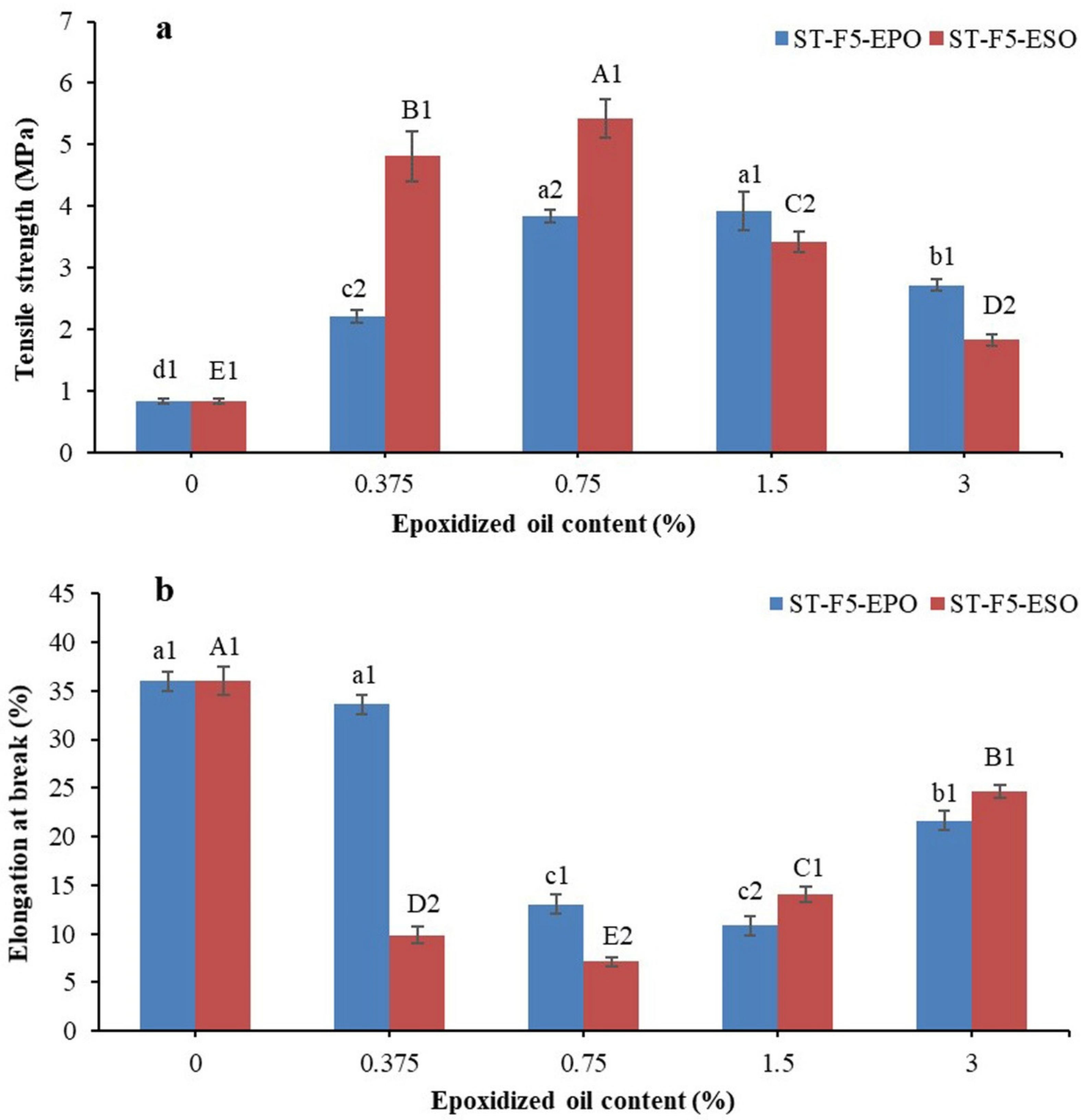
3.5. Mechanical Properties
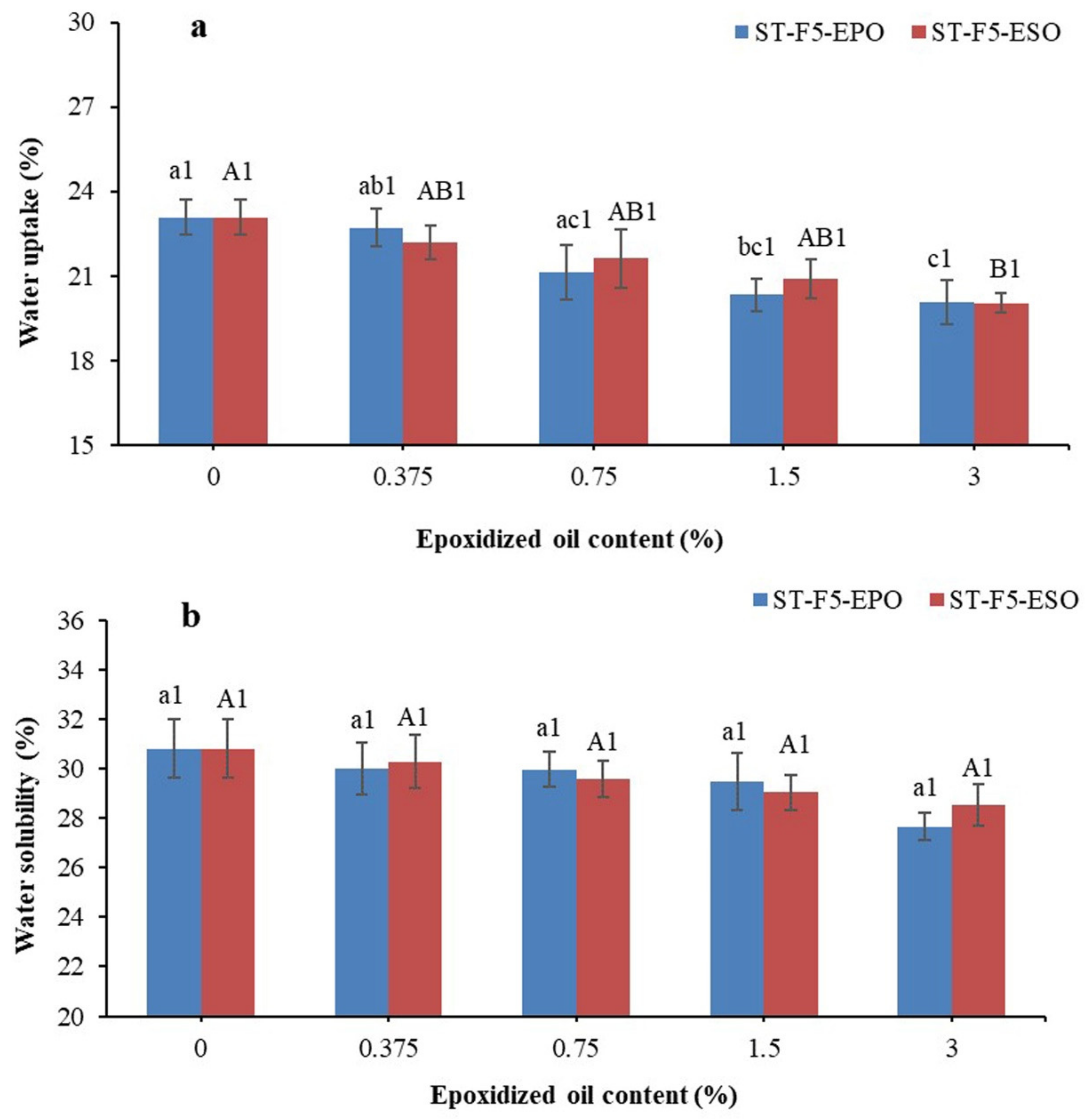
3.6. Water Uptake and Solubility
3.7. WVP
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Belhassen, R.; Vilaseca, F.; Mutjé, P.; Boufi, S. Thermoplasticized starch modified by reactive blending with epoxidized soybean oil. Ind. Crops Prod. 2014, 53, 261–267. [Google Scholar] [CrossRef]
- Ghanbari, A.; Tabarsa, T.; Ashori, A.; Shakeri, A.; Mashkour, M. Preparation and characterization of thermoplastic starch and cellulose nanofibers as green nanocomposites: Extrusion processing. Int. J. Biol. Macromol. 2018, 112, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Montero, B.; Rico, M.; Rodriguez-Llamazares, S.; Barral, L.; Bouza, R. Effect of nanocellulose as a filler on biodegradable thermoplastic starch films from tuber, cereal and legume. Carbohydr. Polym. 2017, 157, 1094–1104. [Google Scholar] [CrossRef] [PubMed]
- Muller, J.; Gonzalez-Martinez, C.; Chiralt, A. Combination of Poly(lactic) Acid and Starch for Biodegradable Food Packaging. Materials 2017, 10, 952. [Google Scholar] [CrossRef] [PubMed]
- Umar, S.; Kamarudin, M.S.; Ramezani-Fard, E. Physical properties of extruded aquafeed with a combination of sago and tapioca starches at different moisture contents. Anim. Feed Sci. Technol. 2013, 183, 51–55. [Google Scholar] [CrossRef]
- European-Bioplastics. Bioplastics Market Data. Available online: https://www.european-bioplastics.org/market/ (accessed on 1 December 2020).
- Li, J.; Ye, F.; Liu, J.; Zhao, G. Effects of octenylsuccination on physical, mechanical and moisture-proof properties of stretchable sweet potato starch film. Food Hydrocoll. 2015, 46, 226–232. [Google Scholar] [CrossRef]
- Gironès, J.; López, J.P.; Mutjé, P.; Carvalho, A.J.F.; Curvelo, A.A.S.; Vilaseca, F. Natural fiber-reinforced thermoplastic starch composites obtained by melt processing. Compos. Sci. Technol. 2012, 72, 858–863. [Google Scholar] [CrossRef]
- Ge, X.; Yu, L.; Liu, Z.; Liu, H.; Chen, Y.; Chen, L. Developing acrylated epoxidized soybean oil coating for improving moisture sensitivity and permeability of starch-based film. Int. J. Biol. Macromol. 2019, 125, 370–375. [Google Scholar] [CrossRef]
- Teaca, C.A.; Bodirlau, R.; Spiridon, I. Effect of cellulose reinforcement on the properties of organic acid modified starch microparticles/plasticized starch bio-composite films. Carbohydr. Polym. 2013, 93, 307–315. [Google Scholar] [CrossRef]
- Chen, J.; Chen, F.; Meng, Y.; Wang, S.; Long, Z. Oxidized microcrystalline cellulose improve thermoplastic starch-based composite films: Thermal, mechanical and water-solubility properties. Polymer 2019, 168, 228–235. [Google Scholar] [CrossRef]
- Woggum, T.; Sirivongpaisal, P.; Wittaya, T. Characteristics and properties of hydroxypropylated rice starch based biodegradable films. Food Hydrocoll. 2015, 50, 54–64. [Google Scholar] [CrossRef]
- Cieśla, K.; Sartowska, B. Modification of the microstructure of the films formed by gamma irradiated starch examined by SEM. Radiat. Phys. Chem. 2016, 118, 87–95. [Google Scholar] [CrossRef]
- Liu, P.; Sun, S.; Hou, H.; Dong, H. Effects of fatty acids with different degree of unsaturation on properties of sweet potato starch-based films. Food Hydrocoll. 2016, 61, 351–357. [Google Scholar] [CrossRef]
- Tongnuanchan, P.; Benjakul, S.; Prodpran, T.; Pisuchpen, S.; Osako, K. Mechanical, thermal and heat sealing properties of fish skin gelatin film containing palm oil and basil essential oil with different surfactants. Food Hydrocoll. 2016, 56, 93–107. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Citric acid cross-linking of starch films. Food Chem. 2010, 118, 702–711. [Google Scholar] [CrossRef]
- Sun, S.; Liu, P.; Ji, N.; Hou, H.; Dong, H. Effects of various cross-linking agents on the physicochemical properties of starch/PHA composite films produced by extrusion blowing. Food Hydrocoll. 2018, 77, 964–975. [Google Scholar] [CrossRef]
- Yildirim-Yalcin, M.; Seker, M.; Sadikoglu, H. Development and characterization of edible films based on modified corn starch and grape juice. Food Chem. 2019, 292, 6–13. [Google Scholar] [CrossRef]
- Bergel, B.F.; Dias Osorio, S.; da Luz, L.M.; Santana, R.M.C. Effects of hydrophobized starches on thermoplastic starch foams made from potato starch. Carbohydr. Polym. 2018, 200, 106–114. [Google Scholar] [CrossRef]
- Basiak, E.; Debeaufort, F.; Lenart, A. Effect of oil lamination between plasticized starch layers on film properties. Food Chem. 2016, 195, 56–63. [Google Scholar] [CrossRef]
- Thakur, R.; Pristijono, P.; Golding, J.B.; Stathopoulos, C.E.; Scarlett, C.J.; Bowyer, M.; Singh, S.P.; Vuong, Q.V. Amylose-lipid complex as a measure of variations in physical, mechanical and barrier attributes of rice starch-ι-carrageenan biodegradable edible film. Food Packag. Shelf Life 2017, 14, 108–115. [Google Scholar] [CrossRef]
- Xiong, Z.; Yang, Y.; Feng, J.; Zhang, X.; Zhang, C.; Tang, Z.; Zhu, J. Preparation and characterization of poly(lactic acid)/starch composites toughened with epoxidized soybean oil. Carbohydr. Polym. 2013, 92, 810–816. [Google Scholar] [CrossRef] [PubMed]
- Uzoh, C.F.; Onukwuli, O.D.; Odera, R.S.; Ofochebe, S. Optimization of polyesterification process for production of palm oil modified alkyd resin using response surface methodology. J. Environ. Chem. Eng. 2013, 1, 777–785. [Google Scholar] [CrossRef]
- Aung, M.M.; Yaakob, Z.; Abdullah, L.C.; Rayung, M.; Li, W.J. A comparative study of acrylate oligomer on Jatropha and Palm oil-based UV-curable surface coating. Ind. Crops Prod. 2015, 77, 1047–1052. [Google Scholar] [CrossRef]
- Kim, J.R.; Sharma, S. The development and comparison of bio-thermoset plastics from epoxidized plant oils. Ind. Crops Prod. 2012, 36, 485–499. [Google Scholar] [CrossRef]
- Orue, A.; Eceiza, A.; Arbelaiz, A. Preparation and characterization of poly(lactic acid) plasticized with vegetable oils and reinforced with sisal fibers. Ind. Crops Prod. 2018, 112, 170–180. [Google Scholar] [CrossRef]
- Tanrattanakul, V.; Saithai, P. Mechanical properties of bioplastics and bioplastic-organoclay nanocomposites prepared from epoxidized soybean oil with different epoxide contents. J. Appl. Polym. Sci. 2009, 114, 3057–3067. [Google Scholar] [CrossRef]
- Khoshnava, S.M.; Rostami, R.; Ismail, M.; Rahmat, A.R.; Ogunbode, B.E. Woven hybrid Biocomposite: Mechanical properties of woven kenaf bast fibre/oil palm empty fruit bunches hybrid reinforced poly hydroxybutyrate biocomposite as non-structural building materials. Constr. Build. Mater. 2017, 154, 155–166. [Google Scholar] [CrossRef]
- Ching, Y.C.; Ershad, A.; Luqman, C.A.; Choo, K.W.; Yong, C.K.; Sabariah, J.J.; Chuah, C.H.; Liou, N.S. Rheological properties of cellulose nanocrystal-embedded polymer composites: A review. Cellulose 2016, 23, 1011–1030. [Google Scholar] [CrossRef]
- Balart, J.F.; Fombuena, V.; Fenollar, O.; Boronat, T.; Sánchez-Nacher, L. Processing and characterization of high environmental efficiency composites based on PLA and hazelnut shell flour (HSF) with biobased plasticizers derived from epoxidized linseed oil (ELO). Compos. Part B Eng. 2016, 86, 168–177. [Google Scholar] [CrossRef]
- Meng, X.T.; Bocharova, V.; Tekinalp, H.; Cheng, S.W.; Kisliuk, A.; Sokolov, A.P.; Kunc, V.; Peter, W.H.; Ozcan, S. Toughening of nanocelluose/PLA composites via bio-epoxy interaction: Mechanistic study. Mater. Des. 2018, 139, 188–197. [Google Scholar] [CrossRef]
- Sarwono, A.; Man, Z.; Bustam, M.A. Blending of epoxidised palm oil with epoxy resin: The effect on morphology, thermal and mechanical properties. J. Polym. Environ. 2012, 20, 540–549. [Google Scholar] [CrossRef]
- Sahari, J.; Sapuan, S.M.; Zainudin, E.S.; Maleque, M.A. Thermo-mechanical behaviors of thermoplastic starch derived from sugar palm tree (Arenga pinnata). Carbohydr. Polym. 2013, 92, 1711–1716. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.; Jiménez, A.; Cháfer, M.; González-Martínez, C.; Chiralt, A. Physical properties and stability of starch-gelatin based films as affected by the addition of esters of fatty acids. Food Hydrocoll. 2015, 49, 135–143. [Google Scholar] [CrossRef]
- Xiong, Z.; Ma, S.Q.; Fan, L.B.; Tang, Z.B.; Zhang, R.Y.; Na, H.N.; Zhu, J. Surface hydrophobic modification of starch with bio-based epoxy resins to fabricate high-performance polylactide composite materials. Compos. Sci. Technol. 2014, 94, 16–22. [Google Scholar] [CrossRef]
- Javidi, Z.; Hosseini, S.F.; Rezaei, M. Development of flexible bactericidal films based on poly(lactic acid) and essential oil and its effectiveness to reduce microbial growth of refrigerated rainbow trout. LWT Food Sci. Technol. 2016, 72, 251–260. [Google Scholar] [CrossRef]
- Rodríguez, M.; Osés, J.; Ziani, K.; Maté, J.I. Combined effect of plasticizers and surfactants on the physical properties of starch based edible films. Food Res. Int. 2006, 39, 840–846. [Google Scholar] [CrossRef]
- Volpe, V.; De Feo, G.; De Marco, I.; Pantani, R. Use of sunflower seed fried oil as an ecofriendly plasticizer for starch and application of this thermoplastic starch as a filler for PLA. Ind. Crops Prod. 2018, 122, 545–552. [Google Scholar] [CrossRef]
- Otoni, C.G.; Avena-Bustillos, R.J.; Olsen, C.W.; Bilbao-Sáinz, C.; McHugh, T.H. Mechanical and water barrier properties of isolated soy protein composite edible films as affected by carvacrol and cinnamaldehyde micro and nanoemulsions. Food Hydrocoll. 2016, 57, 72–79. [Google Scholar] [CrossRef]
- Mohd, A.C.M.; Ching, Y.C.; Luqman, C.A.; Poh, S.C.; Chuah, C.H. Review of bionanocomposite coating films and their applications. Polymers 2016, 8, 246. [Google Scholar]
- Gunathilake, T.M.S.U.; Ching, Y.C.; Chuah, C.H. Synthesis and characterization of nanocellulose reinforced semi-interpenetrating polymer network of chitosan hydrogel. Cellulose 2017, 24, 2215–2228. [Google Scholar]
- Rubentheren, V.; Thomas, A.W.; Ching, Y.C.; Praveena, N.; Erfan, S.; Christopher, F. Effects of heat treatment on chitosan nanocomposite film reinforced with nanocrystalline cellulose and tannic acid. Carbohydr. Polym. 2016, 140, 202. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, V.C.R.; Porto, L.M.; Laurindo, J.B.; Menegalli, F.C. Water vapor barrier and mechanical properties of starch films containing stearic acid. Ind. Crops Prod. 2013, 41, 227–234. [Google Scholar] [CrossRef]
- Brandelero, R.P.; Grossmann, M.V.; Yamashita, F. Films of starch and poly(butylene adipate co-terephthalate) added of soybean oil (SO) and Tween 80. Carbohydr. Polym. 2012, 90, 1452–1460. [Google Scholar] [CrossRef] [PubMed]
- Choo, K.W.; Ching, Y.C.; Chuah, C.H.; Sabariah, J.; Liou, N.S. Preparation and characterization of polyvinyl alcohol-chitosan composite films reinforced with cellulose nanofiber. Materials 2016, 9, 644. [Google Scholar] [CrossRef]
- Jianlei, Y.; Ching, Y.C.; Chuah, C.H. Applications of Lignocellulosic Fibers and Lignin in Bioplastics: A Review. Polymers 2019, 11, 751. [Google Scholar] [CrossRef]
- Atarés, L.; De Jesús, C.; Talens, P.; Chiralt, A. Characterization of SPI-based edible films incorporated with cinnamon or ginger essential oils. J. Food Eng. 2010, 99, 384–391. [Google Scholar] [CrossRef]
- Gunathilake, T.M.S.U.; Ching, Y.C.; Chuah, C.H.; Illias, H.A.; Ching, K.Y.; Singh, R.; Nai-Shang, L. Influence of a nonionic surfactant on curcumin delivery of nanocellulose reinforced chitosan hydrogel. Int. J. Biol. Macromol. 2018, 118, 1055–1064. [Google Scholar] [CrossRef]
- Udeni Gunathilake, T.M.S.; Ching, Y.C.; Chuah, C.H.; Sabariah, J.; Lin, P.-C. Fabrication of porousmaterials from natural/synthetic biopolymers and their composites. Materials 2016, 9, 991. [Google Scholar]
- Khaw, Y.Y.; Ching, Y.C.; Gan, S.N.; Ramesh, S.; Ghazali, N.N.N.; Liu, N.S. Poly(lactic acid) composite films reinforced with microcrystalline cellulose and keratin from chicken feather fiber in 1-butyl-3-methylimidazolium chloride. J. Appl. Polym. Sci. 2019, 136, 47642. [Google Scholar] [CrossRef]
- The, D.P.; Debeaufort, F.; Voilley, A.; Luu, D. Influence of hydrocolloid nature on the structure and functional properties of emulsified edible films. Food Hydrocoll. 2009, 23, 691–699. [Google Scholar] [CrossRef]
- Udeni Gunathilake, T.M.S.; Ching, Y.C.; Chuah, C.H. Enhancement of curcumin bioavailability using nanocellulose reinforced chitosan hydrogel. Polymers 2017, 9, 64. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Ching, Y.C.; Chuah, C.H. Synthesis of chitosan aerogels as promising carrierfor drug delivery: A review. Carbohydr. Polym. 2019, 231, 115744. [Google Scholar] [CrossRef] [PubMed]
- Udenni Gunathilake, T.; Ching, Y.; Ching, K.; Chuah, K.; Abdullah, L. Biomedical and microbiological applications of bio-based porous materials: A review. Polymers 2017, 9, 160. [Google Scholar] [CrossRef] [PubMed]
- Brandelero, R.P.H.; Yamashita, F.; Grossmann, M.V.E. The effect of surfactant Tween 80 on the hydrophilicity, water vapor permeation, and the mechanical properties of cassava starch and poly(butylene adipate-co-terephthalate) (PBAT) blend films. Carbohydr. Polym. 2010, 82, 1102–1109. [Google Scholar] [CrossRef]
- Coral Medina, J.D.; Woiciechowski, A.L.; Filho, A.Z.; Brar, S.K.; Magalhães, A.I., Jr.; Soccol, C.R. Energetic and economic analysis of ethanol, xylitol and lignin production using oil palm empty fruit bunches from a Brazilian factory. J. Clean. Prod. 2018, 195, 44–55. [Google Scholar] [CrossRef]



| Sample Names | Starch (wt%) | Glycerol (wt%) | EFB Fibers (wt%) | EPO (wt%) | ESO (wt%) | Tween 80 (wt%) |
|---|---|---|---|---|---|---|
| ST-F5 | 70 | 30 | 5 | - | - | - |
| ST-F5-EPO0.375 | 70 | 30 | 5 | 0.375 | - | 0.09375 |
| ST-F5-EPO0.75 | 70 | 30 | 5 | 0.75 | - | 0.1875 |
| ST-F5-EPO1.5 | 70 | 30 | 5 | 1.5 | - | 0.375 |
| ST-F5-EPO3 | 70 | 30 | 5 | 3 | - | 0.75 |
| ST-F5-ESO0.375 | 70 | 30 | 5 | - | 0.375 | 0.09375 |
| ST-F5-ESO0.75 | 70 | 30 | 5 | - | 0.75 | 0.1875 |
| ST-F5-ESO1.5 | 70 | 30 | 5 | - | 1.5 | 0.375 |
| ST-F5-ESO3 | 70 | 30 | 5 | - | 3 | 0.75 |
| Formulations | Tonset (°C) | Tm (°C) |
|---|---|---|
| ST-F5 | 171.0 | 182.3 |
| ST-F5-EPO0.375 | 215.4 | 220.1 |
| ST-F5-EPO0.75 | 152.2 | 172.9 |
| ST-F5-EPO1.5 | 208.8 | 219.1 |
| ST-F5-EPO3 | 202.6 | 213.7 |
| ST-F5-ESO0.375 | 210.5 | 216.3 |
| ST-F5-ESO0.75 | 162.6 | 174.7 |
| ST-F5-ESO1.5 | 188.6 | 199.7 |
| ST-F5-ESO3 | 195.4 | 206.8 |
| Formulations | T5%WL (°C) | Tmax1 (°C) | Tmax2 (°C) |
|---|---|---|---|
| ST-F5 | 189.4 | 276.0 | 346.6 |
| ST-F5-EPO0.375 | 166.3 | 279.5 | 349.4 |
| ST-F5-EPO0.75 | 168.7 | - | 345.8 |
| ST-F5-EPO1.5 | 167.8 | 284.1 | 354.1 |
| ST-F5-EPO3 | 175.8 | 273.1 | 350.1 |
| ST-F5-ESO0.375 | 200.4 | 298.5 | 364.3 |
| ST-F5-ESO0.75 | 159.6 | 279.6 | 359.5 |
| ST-F5-ESO1.5 | 188.3 | 290.3 | 359.6 |
| ST-F5-ESO3 | 179.4 | 285.2 | 357.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Ching, Y.C.; Chuah, C.H.; Liou, N.-S. Preparation and Characterization of Starch/Empty Fruit Bunch-Based Bioplastic Composites Reinforced with Epoxidized Oils. Polymers 2021, 13, 94. https://doi.org/10.3390/polym13010094
Yang J, Ching YC, Chuah CH, Liou N-S. Preparation and Characterization of Starch/Empty Fruit Bunch-Based Bioplastic Composites Reinforced with Epoxidized Oils. Polymers. 2021; 13(1):94. https://doi.org/10.3390/polym13010094
Chicago/Turabian StyleYang, Jianlei, Yern Chee Ching, Cheng Hock Chuah, and Nai-Shang Liou. 2021. "Preparation and Characterization of Starch/Empty Fruit Bunch-Based Bioplastic Composites Reinforced with Epoxidized Oils" Polymers 13, no. 1: 94. https://doi.org/10.3390/polym13010094
APA StyleYang, J., Ching, Y. C., Chuah, C. H., & Liou, N.-S. (2021). Preparation and Characterization of Starch/Empty Fruit Bunch-Based Bioplastic Composites Reinforced with Epoxidized Oils. Polymers, 13(1), 94. https://doi.org/10.3390/polym13010094






