Thermal Degradation of Polystyrene (PS) Nanocomposites Loaded with Sol Gel-Synthesized ZnO Nanorods
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of PS/ZnO Nanocomposites
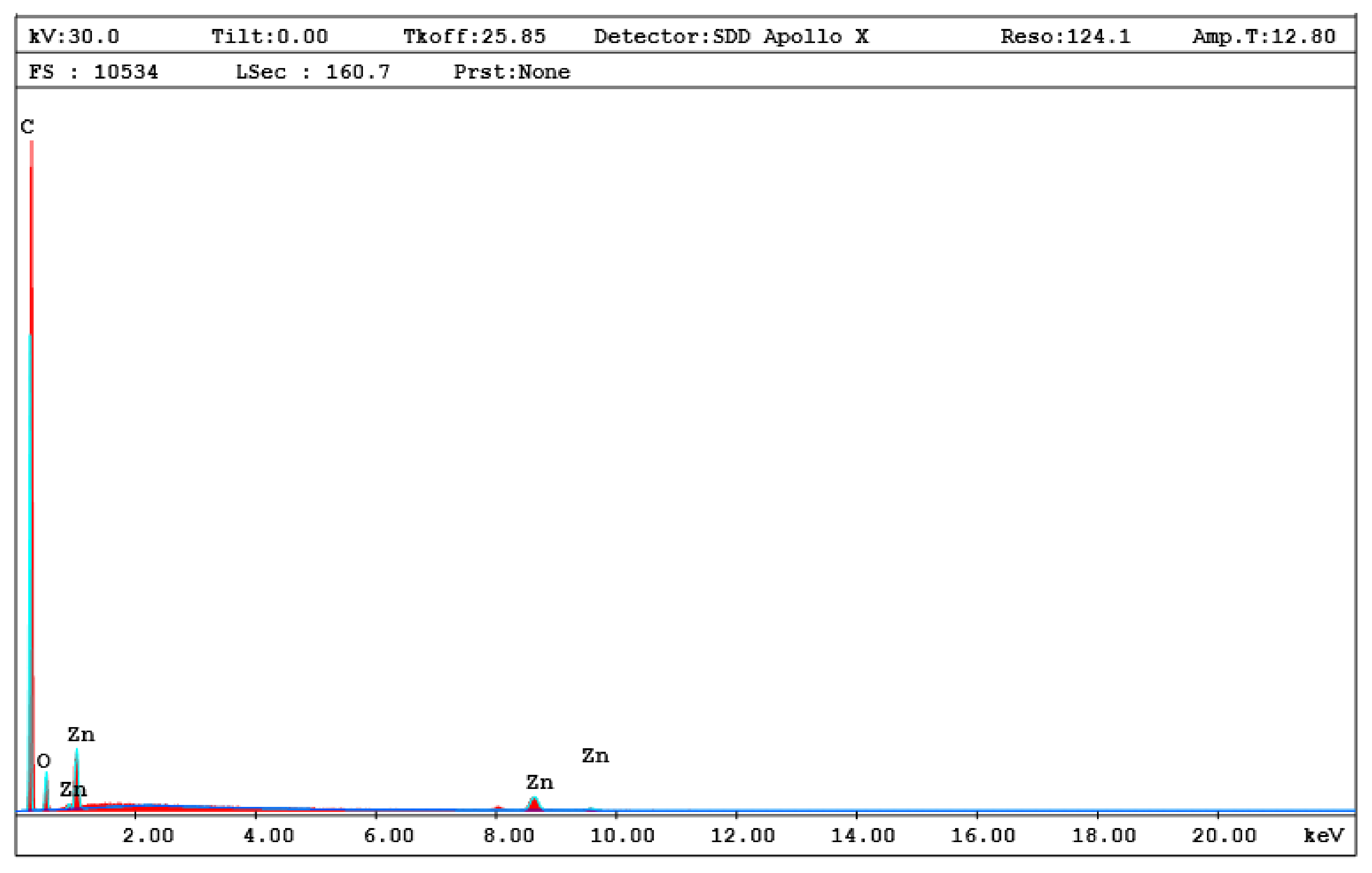
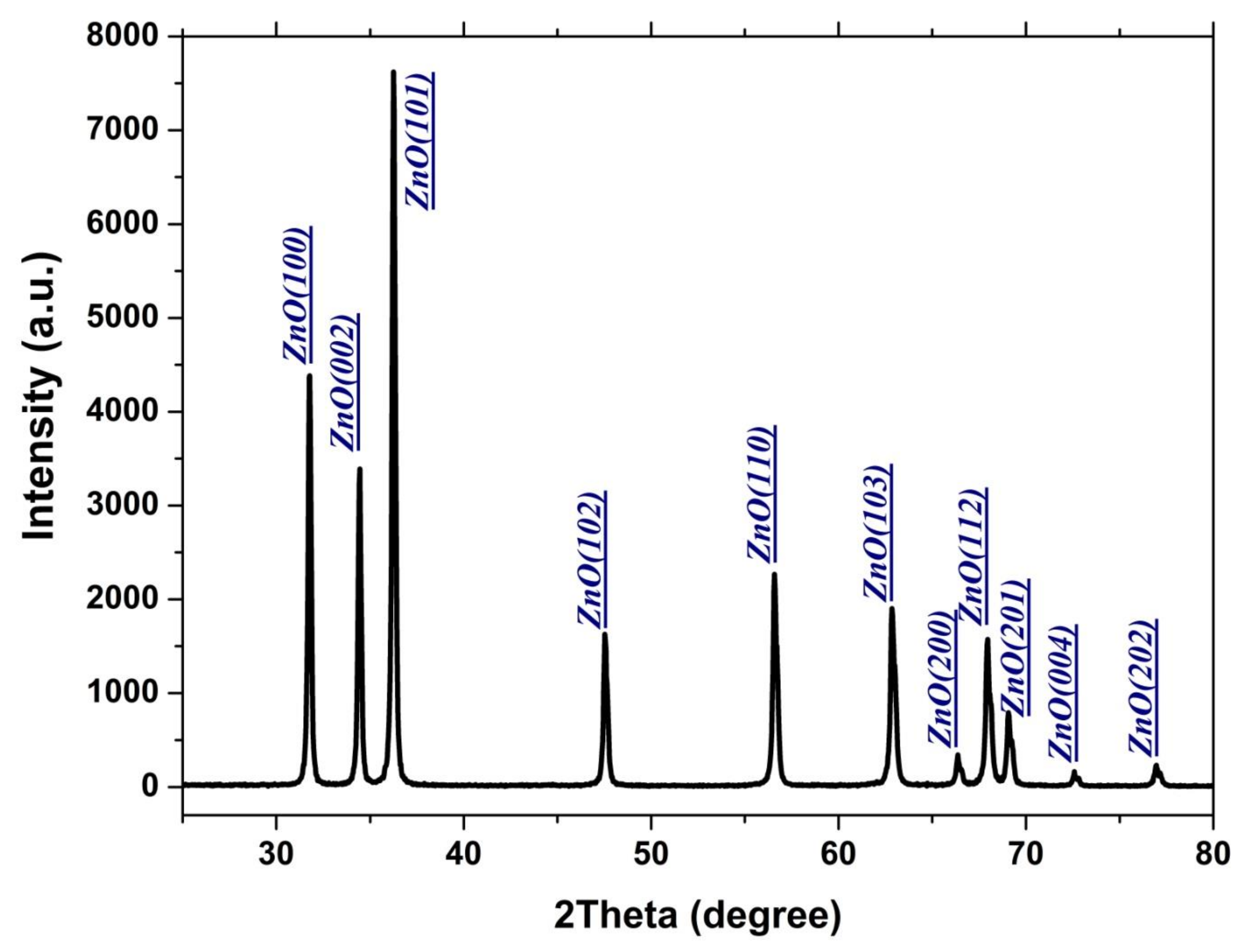
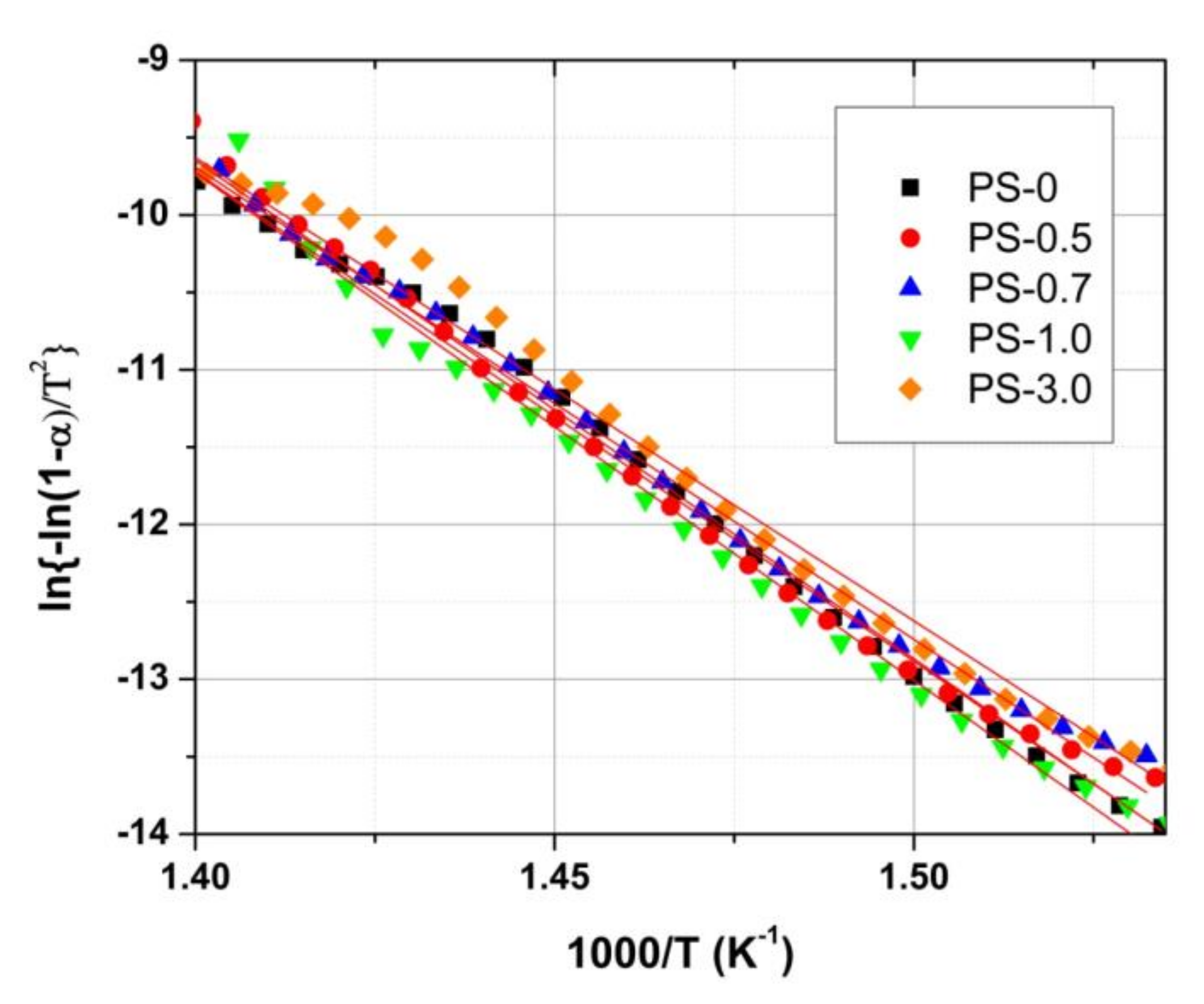
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lan, T.; Kaviratna, P.D.; Pinnavaia, T.J. On the Nature of Polyimide-Clay Hybrid Composites. Chem. Mater. 1994, 6, 573–575. [Google Scholar] [CrossRef]
- Yano, K.; Usuki, A.; Okada, A.; Kurauchi, T.; Kamigaito, O. Synthesis and properties of polyimide–clay hybrid. J. Polym. Sci. Part A Polym. Chem. 1993, 31, 2493–2498. [Google Scholar] [CrossRef]
- Saujanya, C.; Radhakrishnan, S. Structure development and crystallization behaviour of PP/nanoparticulate composite. Polymer 2001, 42, 6723–6731. [Google Scholar] [CrossRef]
- Rong, M.Z.; Zhang, M.Q.; Zheng, Y.X.; Zeng, H.M.; Walter, R.; Friedrich, K. Structure–property relationships of irradiation grafted nano-inorganic particle filled polypropylene composites. Polymer 2001, 42, 167–183. [Google Scholar] [CrossRef]
- Mansour, S. Study of thermal stabilization for polystyrene/carbon nanocomposites via TG/DSC techniques. J. Therm. Anal. Calorim. 2013, 11, 579. [Google Scholar] [CrossRef]
- Wacharawichanant, S.; Thongbunyoung, N.; Churdchoo, P.; Sookjai, T.; Thongyai, S. Morphology and properties of poly(styrene-co-acrylonitrile)/poly(methyl methacrylate)/zinc oxide composites. J. Reinf. Plast. Compos. 2013, 32, 1112–1121. [Google Scholar] [CrossRef]
- Zhang, Y.; Wan, Y.; Shi, Y.; Pan, G.; Yan, H.; Xu, J.; Guo, M.; Qin, L.; Liu, Y. Facile modification of thin-film composite nanofiltration membrane with silver nanoparticles for anti-biofouling. J. Polym. Res. 2016, 23, 105. [Google Scholar] [CrossRef]
- Mansour, S.; Elsad, R.; Izzularab, M. Dielectric investigation of high density polyethylene loaded by ZnO nanoparticles synthesized by sol–gel route. J. Sol-Gel Sci. Technol. 2016, 80, 333–341. [Google Scholar] [CrossRef]
- Chrissafis, K.; Bikiaris, D. Can nanoparticles really enhance thermal stability of polymers? Part I: An overview on thermal decomposition of addition polymers. Thermochim. Acta 2011, 523, 1–24. [Google Scholar] [CrossRef]
- Chae, D.; Kim, B. Characterization on polystyrene/zinc oxide nanocomposites prepared from solution mixing. Polym. Adv. Technol. 2005, 16, 846–850. [Google Scholar] [CrossRef]
- Ahmed, L.; Zhang, B.; Hawkins, S.; Mannan, M.S.; Cheng, Z. Study of thermal and mechanical behaviors of flame retardant polystyrene-based nanocomposites prepared via in-situ polymerization method. J. Loss Prev. Process Ind. 2017, 49, 228–239. [Google Scholar] [CrossRef]
- Alaee, M.; Arias, P.; Sjödin, A.; Bergman, Å. An overview of commercially used brominated flame retardants, their applications, their use patterns in different countries/regions and possible modes of release. Environ. Int. 2003, 29, 683–689. [Google Scholar] [CrossRef]
- Morgan, A.B.; Gilman, J.W. An overview of flame retardancy of polymeric materials: Application, technology, and future directions. Fire Mater. 2013, 37, 259–279. [Google Scholar] [CrossRef]
- Althues, H.; Henle, J.; Kaskel, S. Functional inorganic nanofillers for transparent polymers. Chem. Soc. Rev. 2007, 36, 1454–1465. [Google Scholar] [CrossRef]
- Barala, M.; Mohan, D.; Sanghi, S.; Siwach, B.; Kumari, S.; Yadav, S. Optical properties of PS/ZnO nanocomposites foils prepared by casting method. AIP Conf. Proc. 2019, 2142, 150009. [Google Scholar]
- Mostafaei, A.; Zolriasatein, A. Synthesis and characterization of conducting polyaniline nanocomposites containing ZnO nanorods. Prog. Nat. Sci. Mater. Int. 2012, 22, 273–280. [Google Scholar] [CrossRef]
- Bourbigot, J.S.; Gilman, W.; Wilkie, C.A. Kinetic analysis of the thermal degradation of polystyrene–montmorillonite nanocomposite. Polym. Degrad. Stab. 2004, 84, 483–492. [Google Scholar] [CrossRef]
- Walker, J.P.; Asher, S.A. Acetylcholinesterase-Based Organophosphate Nerve Agent Sensing Photonic Crystal. Anal. Chem. 2005, 77, 1596–1600. [Google Scholar] [CrossRef]
- Bikiaris, D. Can nanoparticles really enhance thermal stability of polymers? Part II: An overview on thermal decomposition of polycondensation polymers. Thermochim. Acta 2011, 523, 25–45. [Google Scholar] [CrossRef]
- Asaletha, R.; Kumaran, M.G.; Thomas, S. Thermal behaviour of natural rubber/polystyrene blends: Thermogravimetric and differential scanning calorimetric analysis. Polym. Degrad. Stab. 1998, 61, 431–439. [Google Scholar] [CrossRef]
- Hu, Y.-H.; Chen, C.-Y.; Wang, C.-C. Viscoelastic properties and thermal degradation kinetics of silica/PMMA nanocomposites. Polym. Degrad. Stab. 2004, 84, 545–553. [Google Scholar] [CrossRef]
- Alam, M.; Arif, S.; Shariq, M. Enhancement in Mechanical Properties of Polystyrene-ZnO Nanocomposites. Int. J. Innov. Res. Adv. Eng. 2015, 2, 122–129. [Google Scholar]
- Jassim, A.N.; Alwan, R.M.; Kadhim, Q.A.; Nsaif, A.A. Preparation and Characterization of ZnO/polystyrene Nanocomposite Films Using Ultrasound Irradiation. Nanosci. Nanotechnol. 2016, 6, 17–23. [Google Scholar]
- Mansour, S.A.; Elsad, R.A.; Izzularab, M.A. Dielectric properties enhancement of PVC nanodielectrics based on synthesized ZnO nanoparticles. J. Polym. Res. 2016, 23, 85. [Google Scholar] [CrossRef]
- Japić, D.; Marinšek, M.; Crnjak Orel, Z. Effect of ZnO on the thermal degradation behavior of poly(methyl methacrylate). Nanocomposites 2016, 63, 9. [Google Scholar] [CrossRef]
- Farha, A.H.; Mansour, S.A.; Kotkata, M.F. Structural, optical, and magnetic study of dilute magnetic semiconducting Co-doped ZnO nanocrystals synthesized using polymer-pyrolysis route. J. Mater. Sci. 2016, 51, 9855–9864. [Google Scholar] [CrossRef]
- Yang, F.; Nelson, G.L. PMMA/silica nanocomposite studies: Synthesis and properties. J. Appl. Polym. Sci. 2004, 91, 3844–3850. [Google Scholar] [CrossRef]
- Vaziri, H.S.; Omaraei, I.A.; Abadyan, M.; Mortezaei, M.; Yousefi, N. Thermophysical and rheological behavior of polystyrene/silica nanocomposites: Investigation of nanoparticle content. Mater. Des. 2011, 32, 4537–4542. [Google Scholar] [CrossRef]
- Bera, O.; Pilić, B.; Pavličević, J.; Jovičić, M.; Holló, B.; Szécsényi, K.M.; Špirkova, M. Preparation and thermal properties of polystyrene/silica nanocomposites. Thermochim. Acta 2011, 515, 1–5. [Google Scholar] [CrossRef]
- George, J.J.; Bhowmick, A.K. Ethylene vinyl acetate/expanded graphite nanocomposites by solution intercalation: Preparation, characterization and properties. J. Mater. Sci. 2008, 43, 702–708. [Google Scholar] [CrossRef]
- Tang, E.; Liu, H.; Sun, L.; Zheng, E.; Cheng, G. Fabrication of zinc oxide/poly(styrene) grafted nanocomposite latex and its dispersion. Eur. Polym. J. 2007, 43, 4210–4218. [Google Scholar] [CrossRef]
- Coats, A.W.; Redfern, J.P. Kinetic Parameters from Thermogravimetric Data. Nature 1964, 201, 68–69. [Google Scholar] [CrossRef]
- Suresh, K.; Kumar, M.; Pugazhenthi, G.; Uppaluri, R. Enhanced mechanical and thermal properties of polystyrene nanocomposites prepared using organo-functionalized NiAl layered double hydroxide via melt intercalation technique. J. Sci. Mater. Devices 2017, 2, 245–254. [Google Scholar]
- Paulik, F.; Paulik, J. Simultaneous thermogravimetric and thermo-gastitrimetric investigations under quasi-isothermal and quasi-isobaric conditions. Anal. Chim. Acta 1973, 67, 437–443. [Google Scholar] [CrossRef]
- Kokta, B.V.; Valade, J.L.; Martin, W.N. Dynamic thermogravimetric analysis of polystyrene: Effect of molecular weight on thermal decomposition. J. Appl. Polym. Sci. 1973, 17, 1–19. [Google Scholar] [CrossRef]
- Paulik, J.; Paulik, F. “Quasi-isothermal” thermogravimetry. Anal. Chim. Acta 1971, 56, 328–331. [Google Scholar] [CrossRef]
- Serenko, O.A.; Roldughin, V.I.; Askadskii, A.A.; Serkova, E.S.; Strashnov, P.V.; Shifrina, Z.B. The effect of size and concentration of nanoparticles on the glass transition temperature of polymer nanocomposites. RSC Adv. 2017, 7, 50113–50120. [Google Scholar] [CrossRef]



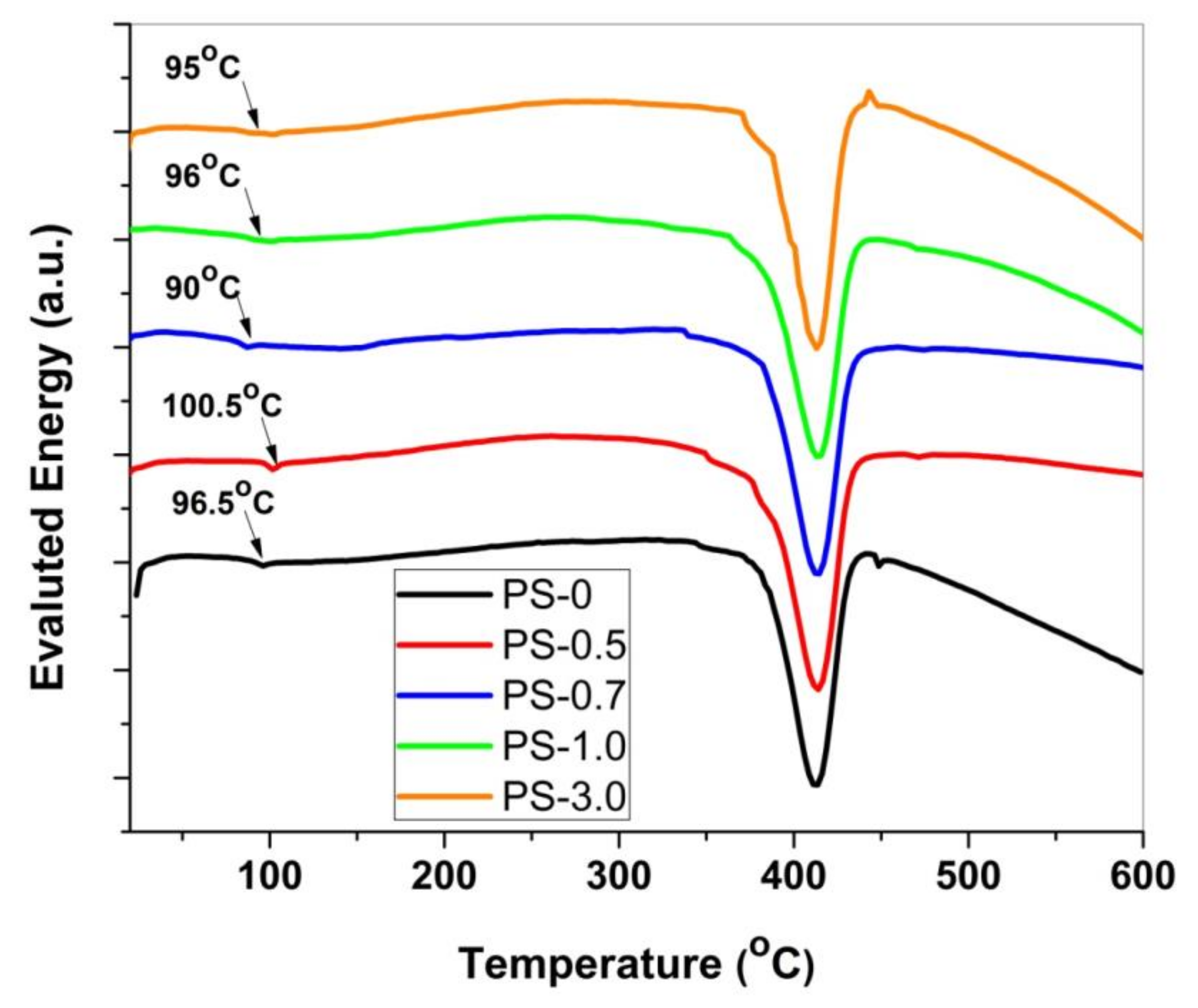
| Sample | T50 °C | T100 °C | TP °C | Tm °C |
|---|---|---|---|---|
| PS-0 | 401 | 440 | 403.3 | 412.3 |
| PS-0.5 | 409.3 | 441 | 410.8 | 413.5 |
| PS-0.7 | 409 | 445 | 410.7 | 413.5 |
| PS-1.0 | 412 | 451 | 413.2 | 414.4 |
| PS-3.0 | 405.5 | 445 | 408.3 | 412.8 |
| Sample | Ea (kJ/mol) | R2 |
|---|---|---|
| PS-0 | 263.0 | 0.997 |
| PS-0.5 | 267.6 | 0.996 |
| PS-0.7 | 253.7 | 0.998 |
| PS-1.0 | 272.6 | 0.995 |
| PS-3.0 | 248.8 | 0.992 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farha, A.H.; Al Naim, A.F.; Mansour, S.A. Thermal Degradation of Polystyrene (PS) Nanocomposites Loaded with Sol Gel-Synthesized ZnO Nanorods. Polymers 2020, 12, 1935. https://doi.org/10.3390/polym12091935
Farha AH, Al Naim AF, Mansour SA. Thermal Degradation of Polystyrene (PS) Nanocomposites Loaded with Sol Gel-Synthesized ZnO Nanorods. Polymers. 2020; 12(9):1935. https://doi.org/10.3390/polym12091935
Chicago/Turabian StyleFarha, Ashraf H., Abdullah F. Al Naim, and Shehab A. Mansour. 2020. "Thermal Degradation of Polystyrene (PS) Nanocomposites Loaded with Sol Gel-Synthesized ZnO Nanorods" Polymers 12, no. 9: 1935. https://doi.org/10.3390/polym12091935
APA StyleFarha, A. H., Al Naim, A. F., & Mansour, S. A. (2020). Thermal Degradation of Polystyrene (PS) Nanocomposites Loaded with Sol Gel-Synthesized ZnO Nanorods. Polymers, 12(9), 1935. https://doi.org/10.3390/polym12091935






