3.1. Characterization of the PDMS Matrix Material
The wetting of a solid surface is highly influenced by its roughness [
24,
25]. In the case of magnetic grass composites, we manipulate the surface roughness through directing the strands in an external magnetic field: the grass stiffens when the field, perpendicular to the substrate is turned on, resulting in a rough surface, decreased contact area, and adhesion between the droplet and the grass, which means higher contact angle values [
17]. To provide reversible shaping and wetting transitions, the elasticity is a key property of these composites [
15,
16,
17].
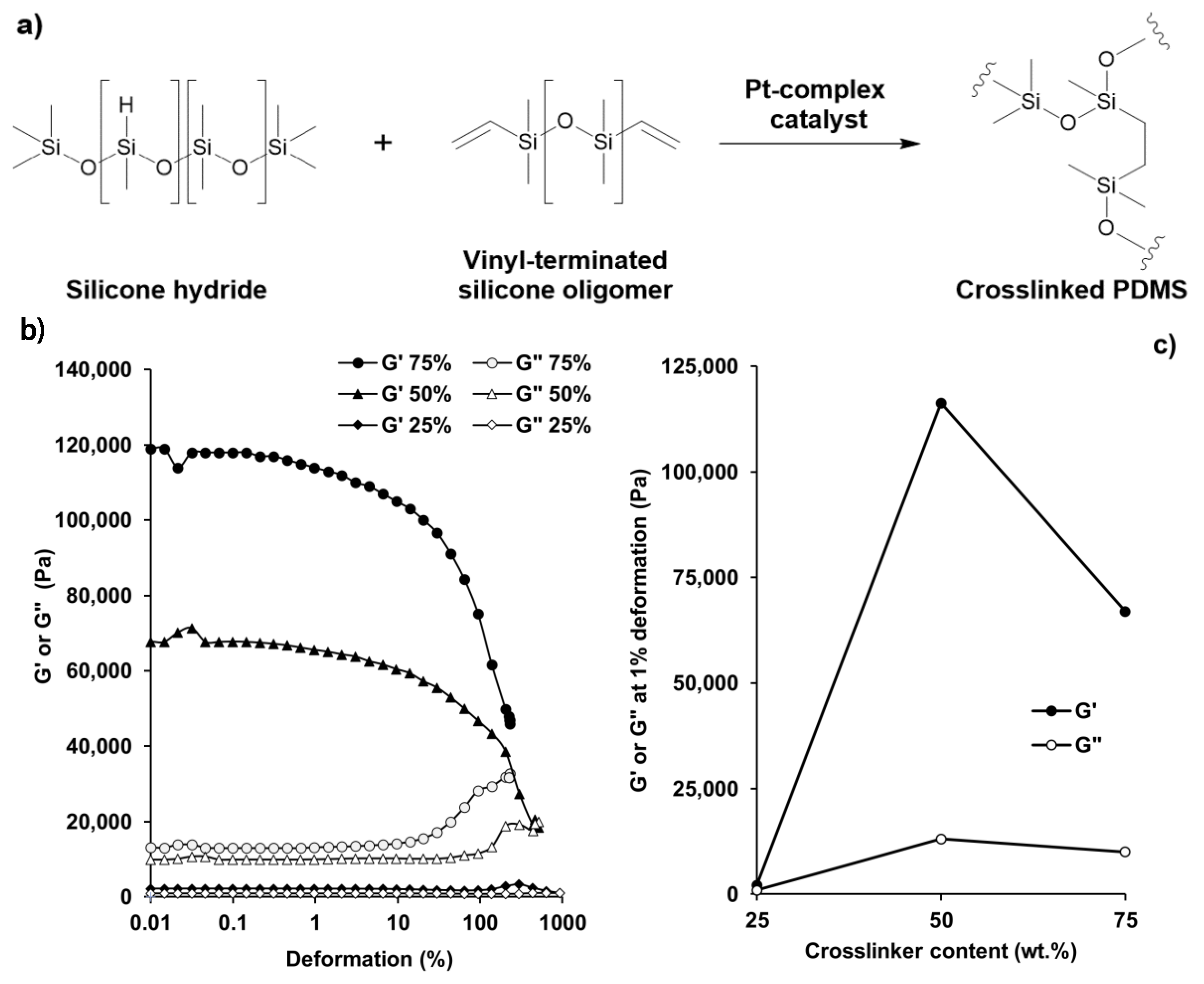
Due to its easily tunable elasticity and hydrophobic nature, PDMS was used as a matrix material during the preparation of magnetoresponsive composite coatings with tunable wetting properties. Upon mixing the two components of the PDMS, the curing took place according to the scheme of
Figure 1a [
26] for 3 h at room temperature and in ambient air. The crosslinking reaction took place between the silicone hydride and the vinyl-terminated silicone oligomer components through coordinative-polymerization by a Pt-complex catalyst [
27]. We examined the elastic (storage moduli;
G′) and viscous (loss moduli;
G″) properties of evolving PDMS samples with different amounts of crosslinker during oscillatory viscosity measurements:
Figure 1b shows the characteristic modulus vs. deformation curves of PDMS-samples with different crosslinking densities. As
Figure 1c shows, the elasticity maximum was reached at 1:1 (mass-to-mass) ratio, and by increasing either the crosslinker (component A) or the catalyst (component B) of the PDMS, the measured values drastically decreased; i.e., excess of the catalyst or the crosslinker resulted in more liquid-like characteristics.
To achieve stimuli-responsive surfaces with reversible shape and wetting transitions, high elasticity is essential: as the 1:1 composition possessed the highest G′ and G″ values and hence the most elastic behavior among all samples, this polymer mixture was applied in our further experiments.
3.2. Preparation and Surface Morphologies of Magnetoresponsive Grass Coatings
To form elastic coatings with dual magnetoresponsivity and photoreactivity, the elastic PDMS matrix was loaded with 59.2–50.0% carbonyl Fe microparticles and 6.3–16.7% plasmonic Ag-TiO
2 nanoparticles, respectively. As the TEM image of
Figure 2 shows, the nearly spherical photocatalyst particles are nanosized (d~50 nm) and contain Ag nanodots on their surface (0.5 wt.% Ag content), which makes their excitation with visible light possible (λ
Ag;max ~ 450 nm) [
28]. However, the spherical carbonyl Fe particles are microsized with relatively broad (
d = 0.5–4 µm) size distribution.
The preparation process of the magnetic grass composites is a combination of spray-coating and magnetic field-directed self-assembly on glass substrates, which is based on the fact that magnetic particles also direct each other by their own magnetostatic forces in external magnetic field: such systems can reach their free-energy minimum upon the formation of ordered structures [
17,
29], which in this case are the magnetic Fe/PDMS pillars.
Spray-coating techniques in general have many parameters that influence product quality [
30]. The size of the nebulized droplets is dependent on the viscosity and the surface tension of the spraying dispersion [
31], but nozzle geometry, the applied gas pressure, and the sprayed amounts are also determining factors [
31,
32]. As in the case of magnetoresponsive grass coatings, the provision of adequate strand density and geometry (thickness and height) is vital to achieving reversible wetting transitions; in other words, the distances between the single strands have to be small enough to produce the desired non-wetted rough surface, which can result in pinning contact between the grass and the droplets. These vital parameters are mostly affected by the magnetic flux density, the distance between the substrate and the permanent magnet, and the droplet size. Higher flux density generally leads to denser grass [
17], but as the self-assembly is also driven by the magnetostatic interactions between the single particles, the size of the nebulized droplets is more important as thinner strands attract less nebulized particles than the ones formed upon the drying of bigger droplets.
Yang et al. extensively studied the effects of these parameters on product quality and found their proper ranges [
17]. According to their study, the suitable spraying composition to achieve reversible wetting transitions requires carbonyl Fe, PDMS and toluene in a (4.5:3:7.8) mass-to-mass ratio, respectively, and the appropriate amount of sprayed dispersion to reach optimal grass height (~2 mm) was 3–4 mL. Reference [
17] specifies that the provision of similar heights is vital in order to achieve contact areas large enough to increase the adhesion of the randomly-oriented grass (0 T magnetic field), although—according to our observations—even higher grasses may suffer from the inability to reversible wetting transitions.
Before enhancing these grass coatings with photocatalytic properties, one must consider that as the features of these systems already depend on many parameters, the addition of another particulate (photocatalyst) component may imply further optimization on the preparation process.
However, according to our experiments the grass coatings can be infused with the Ag-TiO
2 photocatalyst without losing their reversibly switchable wetting characteristics. During the preparation process, the proportions of Yang’s original composition formula were kept (4.5:3:7.8 = carbonyl Fe:PDMS:toluene; mass-to-mass ratio) [
17]—besides the addition of increasing amount of photocatalyst nanoparticles into the spraying dispersion. As the SEM images of grasses prepared in 0.30 T magnetic field in
Figure 3 show, the surface of the initially regular strands become rougher as the nanoparticle content is increased. As it was presented in our previous publications, the spherical-like surface structures that appear are characteristic of the spray-coated composite layers of the Ag-TiO
2 photocatalyst [
20,
21]. The dispersibility of the hydrophilic photocatalyst particles into the organic spraying medium (toluene) is low; hence, the limit of the nominal photocatalyst content was set to ~16.7 wt.% to avoid extreme aggregation, which would cause the instability and thus the undesired vulnerability of the composites. Above this photocatalyst content, the adhesion of the grass to the glass substrates was dissatisfactory and the switching between the wetting states could not be repeated as the grass was pulled off in the proximity of magnetic field. However, the SEM images in
Figure 3 show moderate aggregation, resulting in the formation of thicker strands and less dense grass alongside the increase in surface roughness. As the dispersibility of carbonyl Fe in toluene and PDMS is higher, the aggregation and the formation of surface irregularities are less considerable.
3.3. Wetting Properties of Magnetic Grass Coatings
To quantitatively measure the magnetic field-dependent wetting characteristics of the magnetic grass coatings, contact angle (sessile drop technique) and sliding angle measurements were applied in the presence or without a 0.30 T magnetic field, directed vertically or horizontally to the substrate. Both series of the prepared samples (sprayed in 0.30 T and 0.35 T magnetic fields) were studied: the detailed comparison can be seen in
Figure 4.
As we have already shown in the previous chapter, the addition of Ag-TiO
2 photocatalyst nanoparticles into the magnetic grass coatings can preferably increase the overall surface roughness of these systems. It is generally known, that lotus leaf-like hierarchical (micro- and nanoscale) surface roughness can amplify the initially hydrophobic or hydrophilic wetting character of a flat surface [
25]. As
Figure 4 shows, due to this increase in roughness, the vertically-stiffened pinning grasses (in the middle) show increasingly hydrophobic character, and even superhydrophobicity (150.9° < θ < 163.6°) can be achieved with higher amounts (>6.3 wt.%) of Ag-TiO
2. Without an external magnetic field, however, the strands are randomly oriented (on the left; see schematic representation and the corresponding droplet images of
Figure 4), and thus can be compressed by the sessile droplets. In this case the opposite trends were observed: the contact angles showed a little decrease as the photocatalyst content increased, which can be attributed to the roughness-induced increase of surface area in line contact mode and the decrease of grass density due to aggregation. However, during the evaluation of these data, it is important to note that the presented contact angle values are apparent as the droplets did not actually spread on the surface but penetrated the grass, which practically means that less dense and taller grasses allow deeper penetration for the droplets; therefore, they provide increased contact area and adhesion between the grass and the droplet [
17]. This phenomenon can also be obviously observed the X-ray computer tomography images of
Figure 5, in which the liquid drop shapes are visible thanks to the better contrast. The images clearly indicate that the randomly-oriented state of the surface allows larger wetting area than the vertically-stiffened state.
Grass density and thus indirectly the wetting characteristics can also be adjusted by changing magnetic flux density:
Figure 4 also shows that the achievable contact and sliding angle ranges of grass surfaces prepared in 0.35 T magnetic field are broader that the ones sprayed in 0.30 T with the same composition. In the vertically-stiffened state (middle), the increases in photocatalyst content and roughness generally resulted in increasing contact angle values, while the line contact mode of random orientation (left) had smaller contact angles due to the increased contact surface area and adhesion [
17].
Moreover, the randomly aligned grasses have good adhesion to water droplets with sliding angles of 180° (
Figure 4, left), independent from the photocatalyst content (in the 0–16.7 wt.% range), but in vertically aligned state (middle) the sliding angle values gradually decrease with increasing photocatalyst content; this also proves the influence of surface roughness over hydrophobicity.
In addition to these, we studied the wetting of a third possible grass alignment: the contact angles were also measured on lying grass surfaces with horizontally stiffened strands. To reach this state, we applied 0.30 T magnetic field with force field lines parallel to the surface of the substrate. As our results show (
Figure 4, on the right), this grass alignment is very similar to the one of the vertically-stiffened grass (middle) in terms of wetting properties: the photocatalyst-enhanced rough sides of the strands possessed contact angles resembling the values of the vertically-stiffened state (
Figure 4, in the middle). This behavior indicates similarities in the spacings between the single vertically-stiffened hydrophobic strands and between their surface microstructures.
After all, it is important to note that the achievable contact and sliding angle ranges were barely dependent on the magnetic field (0.30–0.35 T range), applied during sample preparation (except in the case of randomly-oriented grasses); however, there are examples of broader sliding angle ranges (e.g., 8–180°) in the literature where the applied magnetic flux density was slightly higher (4.5 T) [
17].
Thanks to the achievable broad sliding angle ranges and the fast-response reversible switching, the magnetic grass coatings with each composition can be used to pick up and release water droplets. As it can be seen in
Figure 6 and in the
supplementary video (Video S1), when no external magnetic field is present, the randomly aligned strands with sliding angle of 180° (
Figure 4) are able to catch and pick up a water droplet from a superhydrophobic surface, but when a 0.30 T permanent magnet is placed over the sample, the grass stiffens and the droplet falls off due to the suddenly increasing contact angles (163.9 ± 2.9°) and decreasing sliding angles (32.0 ± 7.4°) (
Figure 4). The process can be repeated many times, and as this behavior is not sensitive to the composition (0–16.7 wt.% Ag-TiO
2) and the self-assembly assisting magnetic field (0.30–0.35 T) in the studied ranges, these robust systems offer versatile liquid manipulation capabilities and the subsequently presented visible light photocatalytic activity, as well.
Besides the previously presented static contact angle measurements (
Figure 4.), the magnetic field-adjustable wetting of the composite layer caused by the controllable surface orientation of the strands was also presented in dynamic mode. Images presented in
Figure 7 captured from
supplementary video file (Video S2) show that the position of a non-wetting region of the composite surface can be arbitrarily changed by moving a 0.30 T magnet bar below the glass substrate.
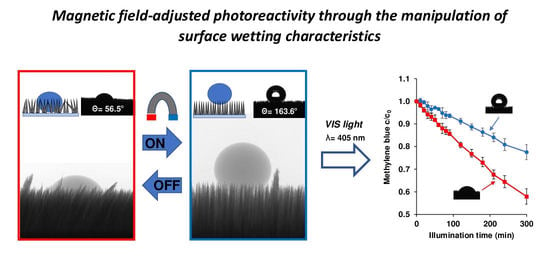
3.4. Magnetic Field-Tailored Photocatalytic Properties
In this chapter we study the effects of magnetic field tailored wetting properties on the photocatalytic behaviors of the composites. The photocatalytic efficiencies of the applied plasmonic Ag-TiO
2 semiconductor photocatalyst and its polymer based (PDMS, fluoropolyer, etc.) composites were proved in various scenarios, including the degradation of model organic pollutants at the S/G- and at the S/L-interfaces and antibacterial tests [
20,
21,
22,
23,
28]. In contrast to the UV-active TiO
2 (
Eg = 3.2 eV), the nanosized particles with a mean diameter of ~50 nm (
Figure 2) possess band gap energies of ~2.9 eV due to the presence of the
d = ~5 nm plasmonic Ag nanodots on their surface (0.5 wt.% Ag content), which makes their excitation with visible light possible (λ
Ag;maxn ≈ 450 nm) [
28]. At the semiconductor/liquid-interface, reactive oxygen species (e.g., hydroxyl radical) form during illumination [
33], which can completely mineralize organic compounds due to their high oxidative potential. The overall photocatalytic activity of these semiconductor composite surfaces also depends on many other factors: their surface roughness, porosity, wettability, and adsorption affinity towards the oxidizable molecules are equally important [
34,
35]. Methylene-blue (MB) is a widely applied model pollutant in the evaluation of the photocatalytic activity of photoreactive coatings, as its concentration can easily be determined through recording the VIS absorption spectra of the colorful MB solutions [
35]. If the pH is neutral and enough oxygen is present in the solution, only the mineralization of the MB molecules contributes to the decolorization, which allows a relatively exact determination of photocatalytic efficiency [
35]. The emission intensity of the exciting light source should also be low in the λ = 350–480 nm range to avoid direct MB photolysis [
35]: our LED-lamp fulfilled this requirement with an emission maximum of λ = 405 nm. As TiO
2 has a PZC value of pH ≈6, the adsorption of the positively charged MB molecules has good affinity to the negatively charged catalyst surface at pH ≈7 [
36].
However, the wetting properties of a photoreactive layer can also significantly affect photoactivity. We previously presented that a rough, non-wetting superhydrophobic coating, composed of 80 wt.% Ag-TiO
2 and 20 wt.% fluoropolymer can degrade only ~20% of the Methylene Blue in aqueous solution after 90 min illumination with a blue-light LED lamp (λ
max = 405 nm) [
20]. According to our previous EDX-studies, the similar spray-coated Ag-TiO
2/polymer composite surfaces contain homogenously dispersed photocatalyst particles, which results in the reported considerable photocatalytic activity [
21].
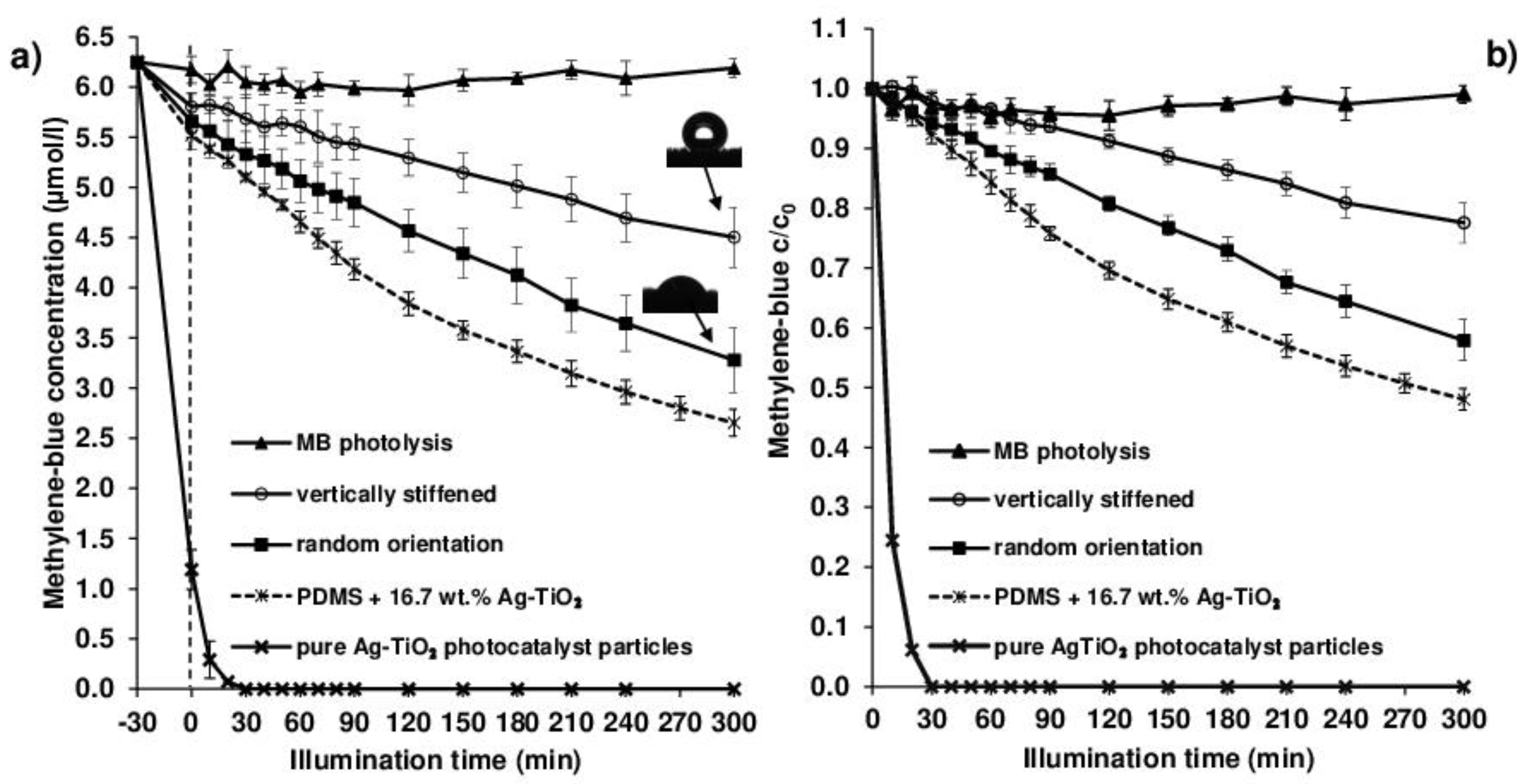
In the case of our photocatalyst-enhanced grass samples with 63.3 ± 1.6 mg/cm
2 specific mass and 16.7 wt.% nominal photocatalyst content, the hydrophobic, i.e., non- wetting nature, resulted in similarly moderate decomposition efficiencies. Moreover, as
Figure 8 shows, this photocatalytic efficiency was also influenced by the external magnetic field-assigned wetting properties: when no magnetic field is present, the randomly-oriented grass collapses under the liquid medium (
Figure 7), resulting in higher contact area and thus higher photocatalytic efficiency (the initial MB concentration decreased by 47.6 ± 5.1% after 300 min irradiation), and when a 0.30 T magnet was placed under the sample during illumination, the contact area between the vertically-stiffened grass and the liquid reduced due to the resulting pinning contact.
Figure 8a shows the absolute concentration changes with the corresponding adsorbed amount of MB, while
Figure 8b shows the relative (c/c
0) changes in MB solution concentration during LED light illumination.
The determined photocatalytic efficiencies of the grass coatings were 42.1 ± 3.5% (random orientation) and 22.4 ± 3.3% (vertically stiffened), respectively, after the 300 min illumination periods (
Figure 8b). According to the synthesis conditions, the nominal Ag-TiO
2 content of each sample was 0.208 g; thus, the photocatalytic measurement was repeated with this photocatalyst content but without PDMS for reference. It can be seen that the pure Ag-TiO
2 photocatalyst nanopowder (without PDMS matrix) completely decomposed the MB-content of the solution under the same experimental conditions. This was due to the increased accessibility of the hydrophilic photocatalyst particles with BET surface area of 53.5 m
2/g; however, the measured data clearly indicate that the PDMS-based composite with much lower BET surface area (0.15 m
2/g) also showed obvious photoreactivity, which was also affected by the wetting properties, as was previously described. As it can be seen in
Figure 8a, the pure photocatalyst particles with higher surface area and more hydrophilic nature adsorbed more MB during the dark period of the experiments.
Figure 8 also shows that the photolysis of MB was insignificant, indicating that only the photocatalytic process on the grass surface caused the decolorization of the MB solutions.
As a comparison with the magnetic composites, photoreactive layers without magnetic particles were also prepared, consisting only 16.7 wt.% Ag-TiO
2 and PDMS. The morphological, wetting, and photocatalytic behaviors of similar layers were discussed in our previous publications [
20,
21]. Unsurprisingly, the layers showed significant photoreactivity in this case as well, with 52.0 ± 1.8% MB decomposition-efficiency (
Figure 8).
Thanks to the presented magnetic field-responsive wetting and photocatalytic characteristics, the prepared grass coatings may seek further applications, not only as sophisticated liquid manipulation tools but utilizable solutions when it comes to the mineralization of organic pollutants.
In our previous publications, we described other Ag-TiO
2-containing composite surfaces with systematically changing, composition-dependent wettability and adsorption affinity towards model pollutants with different polarities [
20,
21]. The now-presented composites with real-time tunable wetting properties potentially offer the same behavior, and they are, for instance, good candidates for such water-treatment and cleaning applications, in which the polarities of the contaminating chemical species change on a wider scale. To prove this theory, further investigations on the photocatalytic behavior of these magnetic composites against different kinds of model pollutants are planned as well.