Improving the Reactivity of Sugarcane Bagasse Kraft Lignin by a Combination of Fractionation and Phenolation for Phenol–Formaldehyde Adhesive Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Phenolation of Lignin
2.3. Preparation of Adhesive
2.3.1. PF Adhesives Preparation
2.3.2. LPF Adhesives Preparation
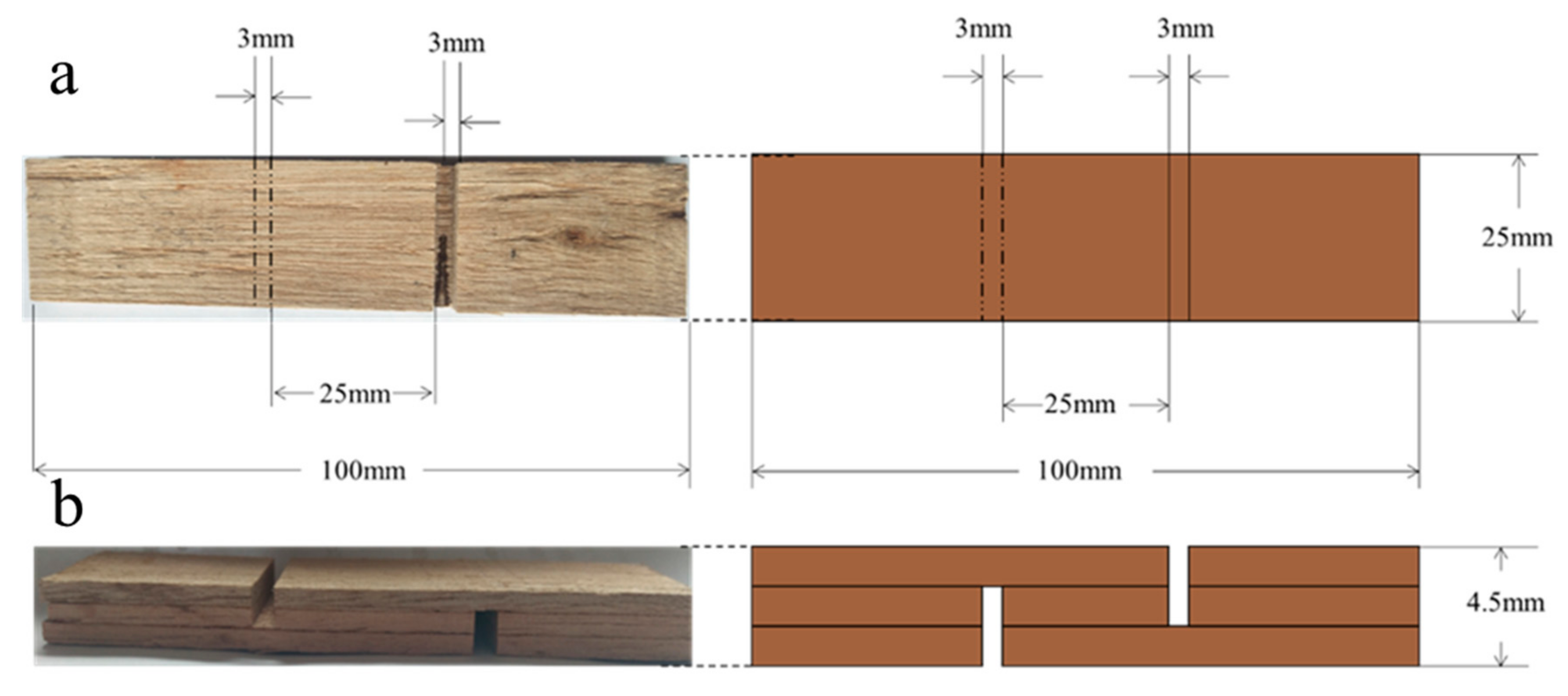
2.4. Plywood Preparation
2.5. Characterization
2.5.1. Fourier Transform Infrared Spectrometer Analysis (FT-IR)
2.5.2. GPC Analysis
2.5.3. 1H-13C HSQC NMR Analysis
2.5.4. 31P NMR Analysis
2.5.5. Properties of Adhesive
2.5.6. Properties of Plywood
3. Results and Discussion
3.1. Adhesive Properties and Plywood Performance
3.2. Characterization of Phenolated Lignin
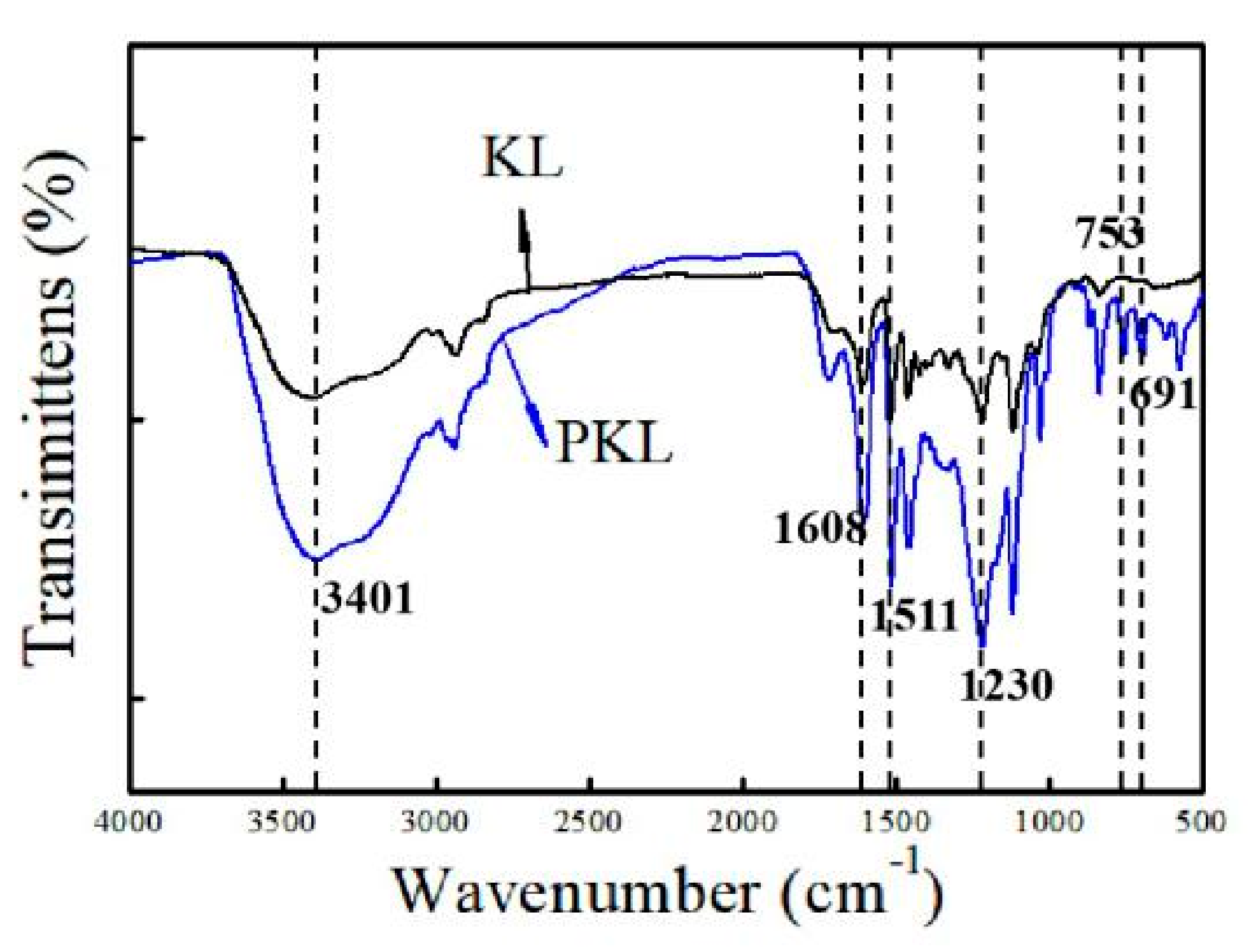
3.2.1. FT-IR Analysis
3.2.2. GPC Analysis
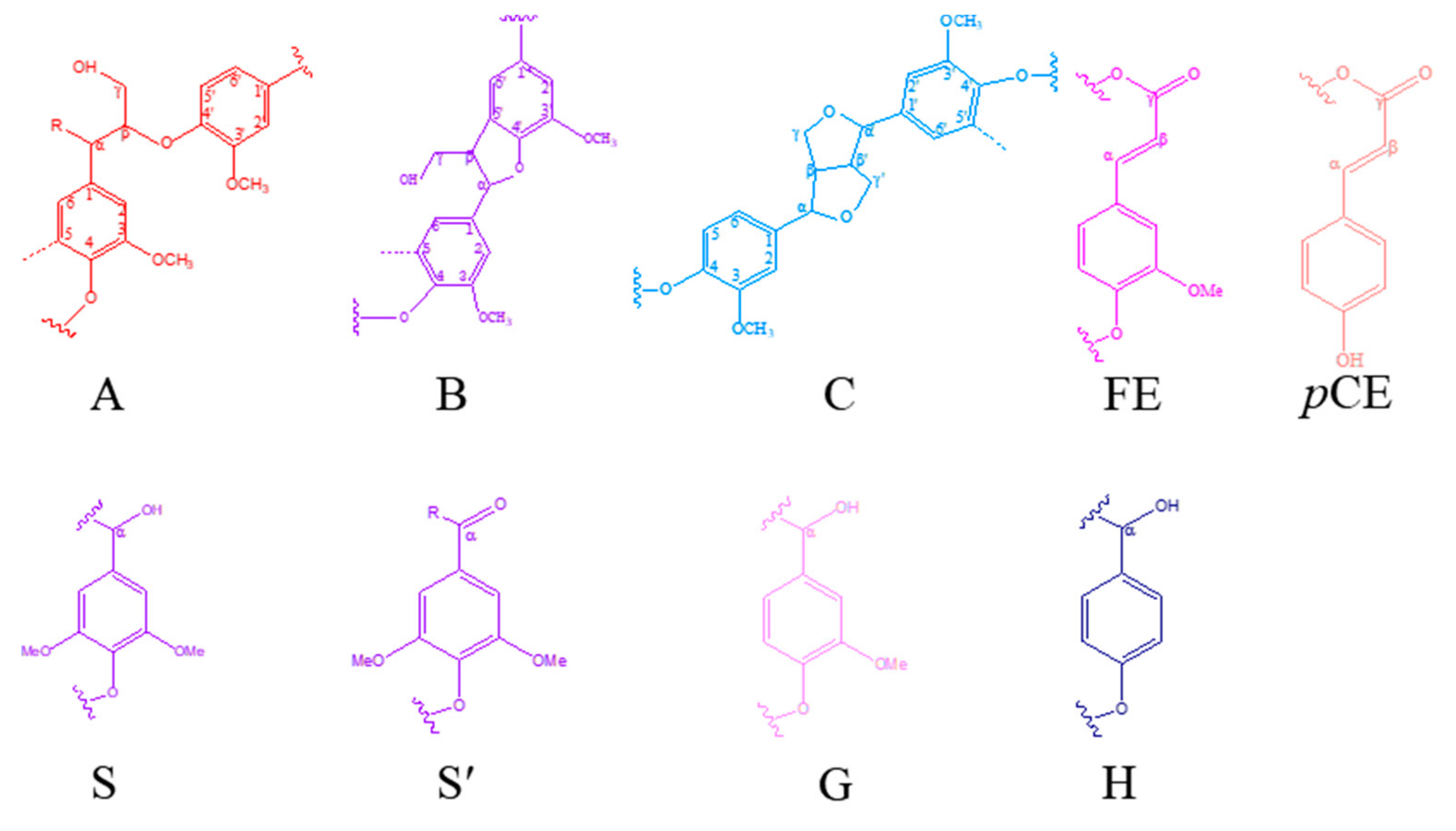
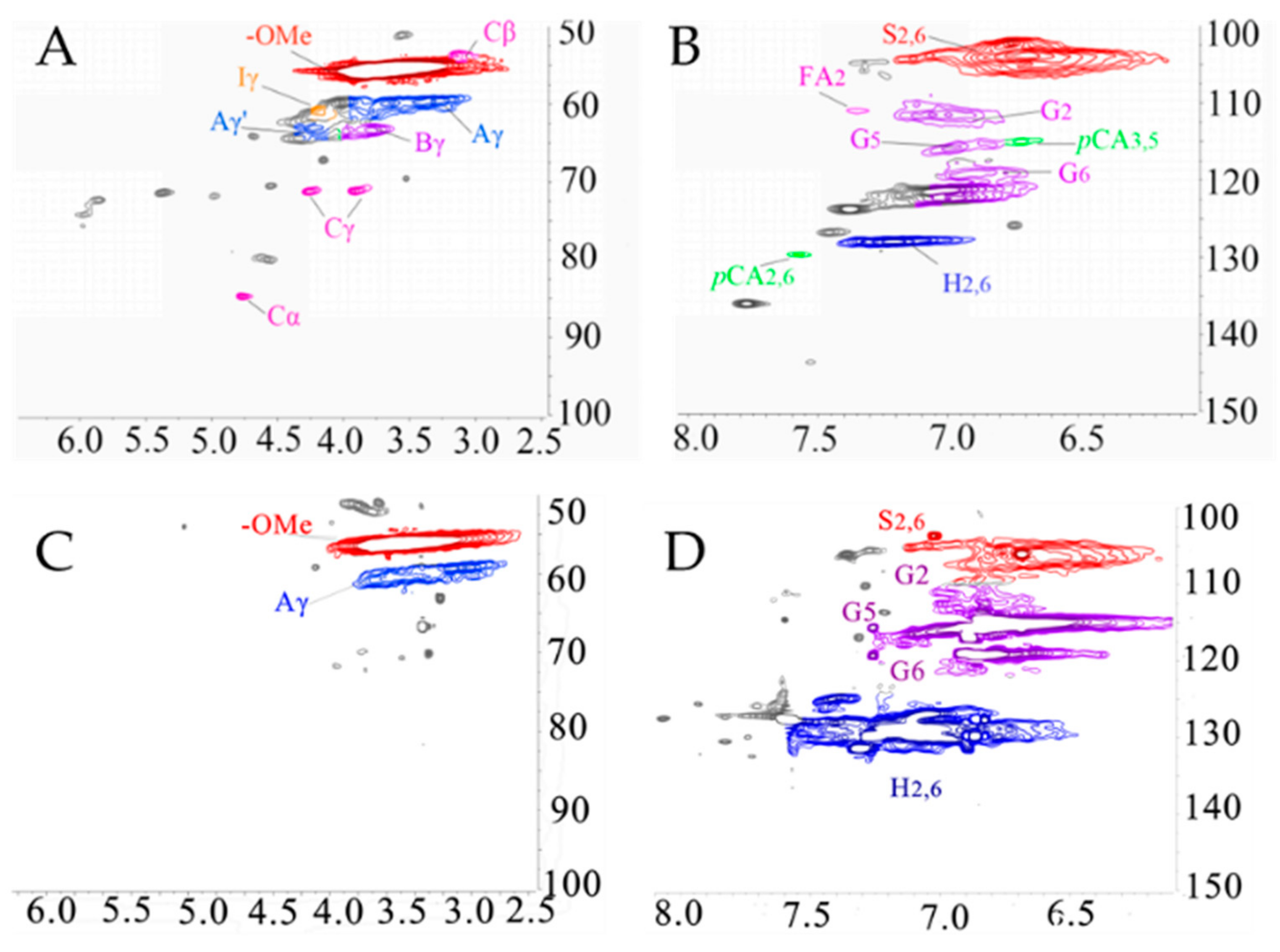
3.2.3. 1H-13C HSQC NMR Analysis
3.2.4. 31P NMR Analysis
3.3. PLPF Adhesive Properties and Plywood Performances
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lee, S.H.; Yoshioka, M.; Shiraishi, N. Preparation and properties of phenolated corn bran (CB)/phenol/formaldehyde cocondensed resin. J. Appl. Polym. Sci. 2000, 77, 2901–2907. [Google Scholar] [CrossRef]
- Sjöström, E. Wood Chemistry: Fundamentals and Applications; Gulf Professional Publishing: Houston, TX, USA, 1981. [Google Scholar]
- Falkehag, S.I.; Braddon, D.V.; Dougherty, W.K. Lignin polymer applications. In Renewable Resources for Plastics: Growth and Change in Adhesives, Chemical Requirements of the Tobacco Industry; American Chemical Society: Brooklyn, NY, USA, 1975; pp. 68–72. [Google Scholar]
- Yang, S.; Wen, J.L.; Yuan, T.Q.; Sun, R.C. Characterization and phenolation of biorefinery technical lignin for lignin-phenol-formaldehyde resin adhesive synthesis. RSC Adv. 2014, 4, 57996–58004. [Google Scholar] [CrossRef]
- Li, M.F.; Sun, S.N.; Xu, F.; Sun, R.C. Sequential solvent fractionation of heterogeneous bamboo organosolv lignin for value-added application. Sep. Purif. Technol. 2012, 101, 18–25. [Google Scholar] [CrossRef]
- Cui, C.; Sun, R.; Argyropoulos, D.S. Fractional Precipitation of Softwood Kraft Lignin: Isolation of Narrow Fractions Common to a Variety of Lignins. ACS Sustain. Chem. Eng. 2014, 2, 959–968. [Google Scholar] [CrossRef]
- dos Santos, P.S.; Erdocia, X.; Gatto, D.A.; Labidi, J. Characterisation of Kraft lignin separated by gradient acid precipitation. Ind. Crops Prod. 2014, 55, 149–154. [Google Scholar] [CrossRef]
- Wang, G.; Chen, H. Fractionation of alkali-extracted lignin from steam-exploded stalk by gradient acid precipitation. Sep. Purif. Technol. 2013, 105, 98–105. [Google Scholar] [CrossRef]
- Jamin, M.; Adam, M.; Damblon, C.; Christiaens, L.; Frère, J.M. Lignin separation and fractionation by ultrafiltration. Biochem. J. 1991, 71, 38–43. [Google Scholar]
- Jiang, X.; Liu, J.; Du, X.; Hu, Z.; Chang, H.; Jameel, H. Phenolation to improve lignin reactivity towards thermosets application. ACS Sustain. Chem. Eng. 2018, 4, 5504–5512. [Google Scholar] [CrossRef]
- Hong, S.; Lian, H.; Sun, X.; Pan, D.; Carranza, A.; Pojman, J.A.; Mota-Morales, J.D. Zinc-based deep eutectic solvent-mediated hydroxylation and demethoxylation of lignin for the production of wood adhesive. RSC Adv. 2016, 6, 89599–89608. [Google Scholar] [CrossRef]
- Zhou, W.; Chen, F.; Zhang, H.; Wang, J. Preparation of a polyhydric aminated lignin and its use in the preparation of polyurethane film. J. Wood Chem. Technol. 2017, 37, 323–333. [Google Scholar] [CrossRef]
- Nagieb, Z.A. Demethylation of thiolignin by reaction with potassium dichromate-a kinetic study. Wood Sci. Technol. 1985, 19, 233–242. [Google Scholar] [CrossRef]
- Xia, C.L.; Xu, Y.Z.; Liu, X.H.; Wang, C.P. Preparation and characterization of demethylated lignin assisted by microwave irradiation. Chem. Ind. For. Prod. 2016, 36, 57–63. [Google Scholar]
- Chile, L.; Kaser, S.J.; Hatzikiriakos, S.G.; Mehrkhodavandi, P. Synthesis and Thermorheological Analysis of Biobased Lignin-graft-poly (lactide) Copolymers and Their Blends. ACS Sustain. Chem. Eng. 2018, 2, 1650–1661. [Google Scholar] [CrossRef]
- Du, X.; Li, J.; Lindström, M.E. Modification of industrial softwood kraft lignin using Mannich reaction with and without phenolation pretreatment. Ind. Crops Prod. 2014, 52, 729–735. [Google Scholar] [CrossRef]
- Podschun, J.; Stücker, A.; Saake, B.; Lehnen, R. Structure–Function Relationships in the Phenolation of Lignins from Different Sources. ACS Sustain. Chem. Eng. 2015, 3, 2526–2532. [Google Scholar] [CrossRef]
- Zhang, Y.; Yuanauthor, Z.; Xuauthor, C. Sustainable bio-phenol-hydroxymethylfurfural resins using phenolated de-polymerized hydrolysis lignin and their application in bio-composites. Ind. Crops Prod. 2016, 79, 84–90. [Google Scholar] [CrossRef]
- Jia, Z.; Li, M.; Wan, G.; Luo, B.; Guo, C.; Wang, S.; Min, D. Improving the homogeneity of sugarcane bagasse kraft lignin through sequential solvents. RSC Adv. 2018, 8, 42269–42279. [Google Scholar] [CrossRef]
- Yang, S.; Wu, J.; Zhang, Y.; Yuan, T.; Sun, R. Preparation of lignin-phenol-formaldehyde resin adhesive based on active sites of technical lignin. J. Biobased Mater. Bio. 2015, 9, 266–272. [Google Scholar] [CrossRef]
- Aonso, M.V.; Oliet, M.; Rodrıguez, F.; Garcıa, J.; Gilarranz, M.A.; Rodr Guez, J.J. Modification of ammonium lignosulfonate by phenolation for use in phenolic resins. Bioresour. Technol. 2005, 96, 1013–1018. [Google Scholar] [CrossRef]
- Glasser, W.G.; Davé, V.; Frazier, C.E. Molecular weight distribution of (semi-) commercial lignin derivatives. J. Wood Chem. Technol. 1993, 13, 545–559. [Google Scholar] [CrossRef]
- Argyropoulos, D.S. Dimitris quantitative phosphorus-31 NMR analysis of six soluble lignin. J. Wood Chem. Technol. 1994, 14, 65–82. [Google Scholar] [CrossRef]
- Granata, A.; Argyropoulos, D.S. 2-Chloro-4,4,5,5-tetramethyl-1,3,2-dioxaphospholane, a reagent for the accurate determination of the uncondensed and condensed phenolic moieties in lignins. J. Agric. Food Chem. 1995, 43, 1538–1544. [Google Scholar] [CrossRef]
- Guerra, A.; Filpponen, I.; Lucia, L.A.; Saquing, C.; Baumberger, S.; Argyropoulos, D.S. Toward a better understanding of the lignin isolation process from wood. J. Agric. Food Chem. 2006, 54, 5939–5947. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Wan, G.; Zhang, Q.; Li, M.; Luo, B.; Guo, C.; Wang, S.; Min, D. Phenolation of bagasse kraft lignin for application in lignin-based phenol formaldehyde adhesives. China Pulp. Paper 2018, 37, 7–14. [Google Scholar]
- Li, C.; Wang, W.; Mu, Y.; Zhang, J.; Zhang, S.; Li, J.; Zhang, W. Structural properties and copolycondensation mechanism of valonea tannin-modified phenol-formaldehyde resin. J. Polym. Environ. 2017, 26, 1297–1309. [Google Scholar] [CrossRef]
- Zhang, Q.; Wan, G.; Li, M.; Jiang, H.; Wang, S.; Min, D. Impact of bagasse lignin-carbohydrate complexes structural changes on cellulase adsorption behavior. Int. J. Biol. Macromol. 2020, 162, 236–245. [Google Scholar] [CrossRef]
- Song, Y.; Wang, Z.; Yan, N.; Zhang, R.; Li, J. Demethylation of wheat straw alkali lignin for application in phenol formaldehyde adhesives. Polymers 2016, 8, 209. [Google Scholar] [CrossRef]
| Sample | Adhesive Properties | Plywood Performance | ||||
|---|---|---|---|---|---|---|
| pH | Solid Content (%) | Free Formaldehyde (%) | Free Phenol (%) | Wet Bonding Strength (MPa) | Formaldehyde Release (mg/L) | |
| PF | 11.9 ± 0.1 | 38.1 ± 0.6 | 0.12 ± 0.02 | 0.24 ± 0.03 | 1.49 ± 0.06 | 0.19 ± 0.03 |
| KLPF | 11.2 ± 0.2 | 42.1 ± 0.5 | 1.04 ± 0.05 | 0.72 ± 0.01 | 0.68 ± 0.11 | 1.21 ± 0.07 |
| F1-LPF | 11.2 ± 0.1 | 42.3 ± 0.7 | 1.18 ± 0.07 | 0.65 ± 0.08 | 0.98 ± 0.17 | 1.18 ± 0.02 |
| F2-LPF | 11.2 ± 0.2 | 43.3 ± 0.8 | 0.91 ± 0.11 | 0.44 ± 0.04 | 0.83 ± 0.08 | 1.12 ± 0.03 |
| F3-LPF | 11.3 ± 0.1 | 45.8 ± 0.5 | 0.98 ± 0.05 | 0.47 ± 0.01 | 0.93 ± 0.12 | 1.09 ± 0.05 |
| PKLPF | 11.5 ± 0.1 | 42.8 ± 0.8 | 0.19 ± 0.12 | 1.33 ± 0.03 | 1.27 ± 0.09 | 0.33 ± 0.06 |
| PF1PF | 11.4 ± 0.2 | 44.1 ± 0.7 | 0.25 ± 0.08 | 1.59 ± 0.07 | 1.19 ± 0.16 | 0.39 ± 0.07 |
| PF2PF | 11.4 ± 0.1 | 43.9 ± 0.5 | 0.27 ± 0.04 | 0.92 ± 0.02 | 1.15 ± 0.13 | 0.43 ± 0.05 |
| PF3PF | 11.9 ± 0.1 | 44.6 ± 0.7 | 0.16 ± 0.05 | 1.46 ± 0.02 | 1.36 ± 0.11 | 0.31 ± 0.06 |
| GB/T14732 | ≥7.0 | ≥35.0 | ≤0.30 | ≤6.00 | ≥0.70 | ≤0.50 a |
| Sample | Mw (g/mol) | PDI |
|---|---|---|
| KL | 3.2 × 103 | 3.01 |
| PKL | 1.9 × 103 | 2.13 |
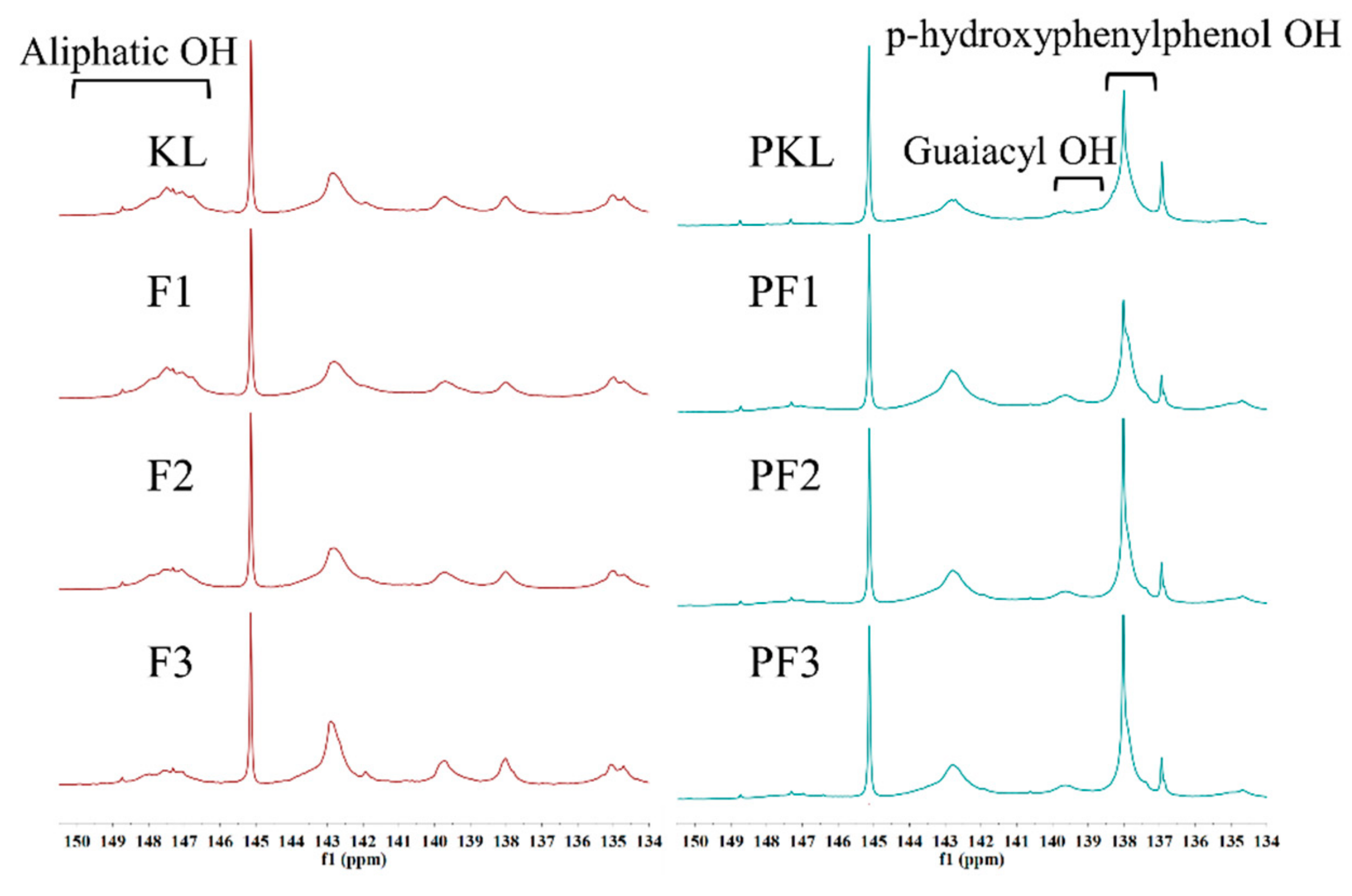
| Sample | –OH (mmol/g) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| S | G | NC–OH a | –COOH | Aliphatic–OH | Total –OH | Active Sites b | |||
| S | G | H | |||||||
| KL | 0.01 | 0.22 | 1.23 | 0.35 | 0.15 | 0.38 | 0.71 | 1.96 | 0.65 |
| PKL | 0.08 | 0.30 | 0.93 | 0.61 | 3.01 | 0.15 | 0.27 | 4.93 | 6.63 |
| F1 | 0.01 | 0.28 | 1.02 | 0.33 | 0.14 | 0.38 | 0.92 | 1.78 | 0.62 |
| PF1 | 0.09 | 0.43 | 0.94 | 0.55 | 2.77 | 0.25 | 0.13 | 4.78 | 6.08 |
| F2 | 0.02 | 0.24 | 1.37 | 0.37 | 0.16 | 0.34 | 0.44 | 2.16 | 0.69 |
| PF2 | 0.11 | 0.44 | 1.10 | 0.62 | 2.63 | 0.18 | 0.29 | 4.90 | 5.88 |
| F3 | 0.07 | 0.18 | 1.73 | 0.53 | 0.38 | 0.45 | 0.28 | 2.89 | 1.29 |
| PF3 | 0.08 | 0.30 | 1.24 | 0.61 | 3.12 | 0.11 | 0.67 | 5.35 | 6.86 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, B.; Jia, Z.; Jiang, H.; Wang, S.; Min, D. Improving the Reactivity of Sugarcane Bagasse Kraft Lignin by a Combination of Fractionation and Phenolation for Phenol–Formaldehyde Adhesive Applications. Polymers 2020, 12, 1825. https://doi.org/10.3390/polym12081825
Luo B, Jia Z, Jiang H, Wang S, Min D. Improving the Reactivity of Sugarcane Bagasse Kraft Lignin by a Combination of Fractionation and Phenolation for Phenol–Formaldehyde Adhesive Applications. Polymers. 2020; 12(8):1825. https://doi.org/10.3390/polym12081825
Chicago/Turabian StyleLuo, Bin, Zhuan Jia, Hongrui Jiang, Shuangfei Wang, and Douyong Min. 2020. "Improving the Reactivity of Sugarcane Bagasse Kraft Lignin by a Combination of Fractionation and Phenolation for Phenol–Formaldehyde Adhesive Applications" Polymers 12, no. 8: 1825. https://doi.org/10.3390/polym12081825
APA StyleLuo, B., Jia, Z., Jiang, H., Wang, S., & Min, D. (2020). Improving the Reactivity of Sugarcane Bagasse Kraft Lignin by a Combination of Fractionation and Phenolation for Phenol–Formaldehyde Adhesive Applications. Polymers, 12(8), 1825. https://doi.org/10.3390/polym12081825





