Polymeric Carriers for Delivery Systems in the Treatment of Chronic Periodontal Disease
Abstract
1. Introduction
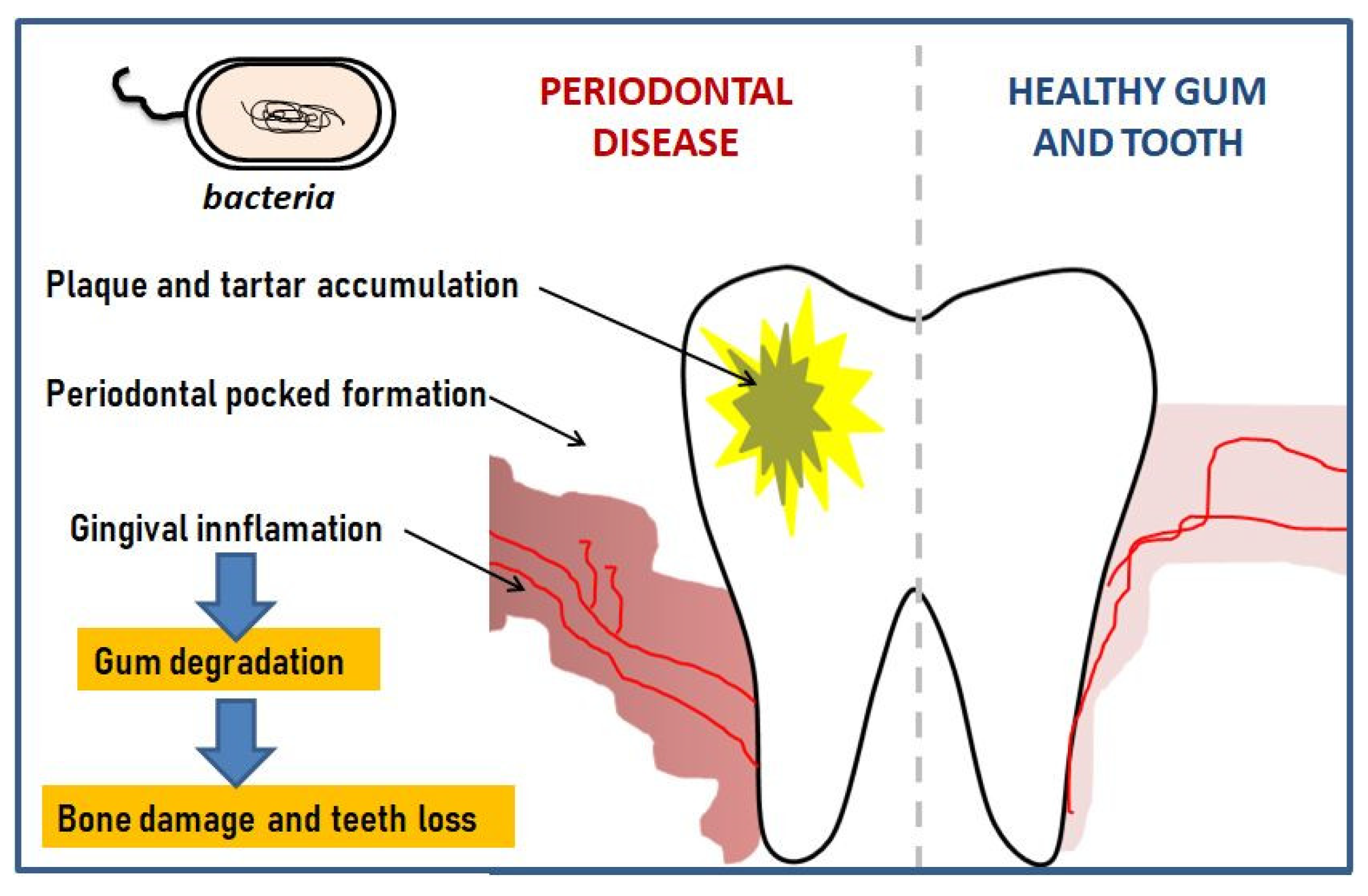
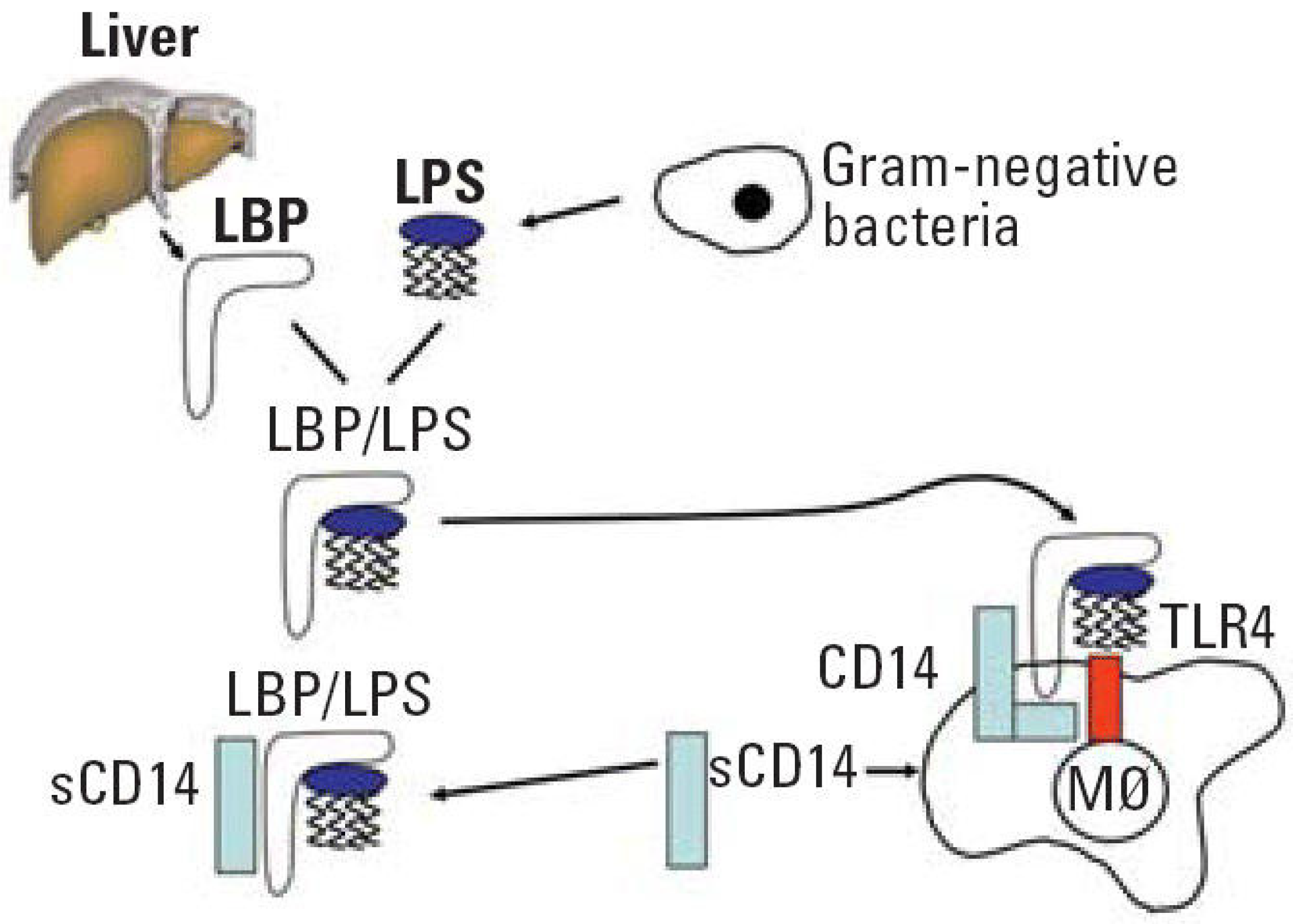
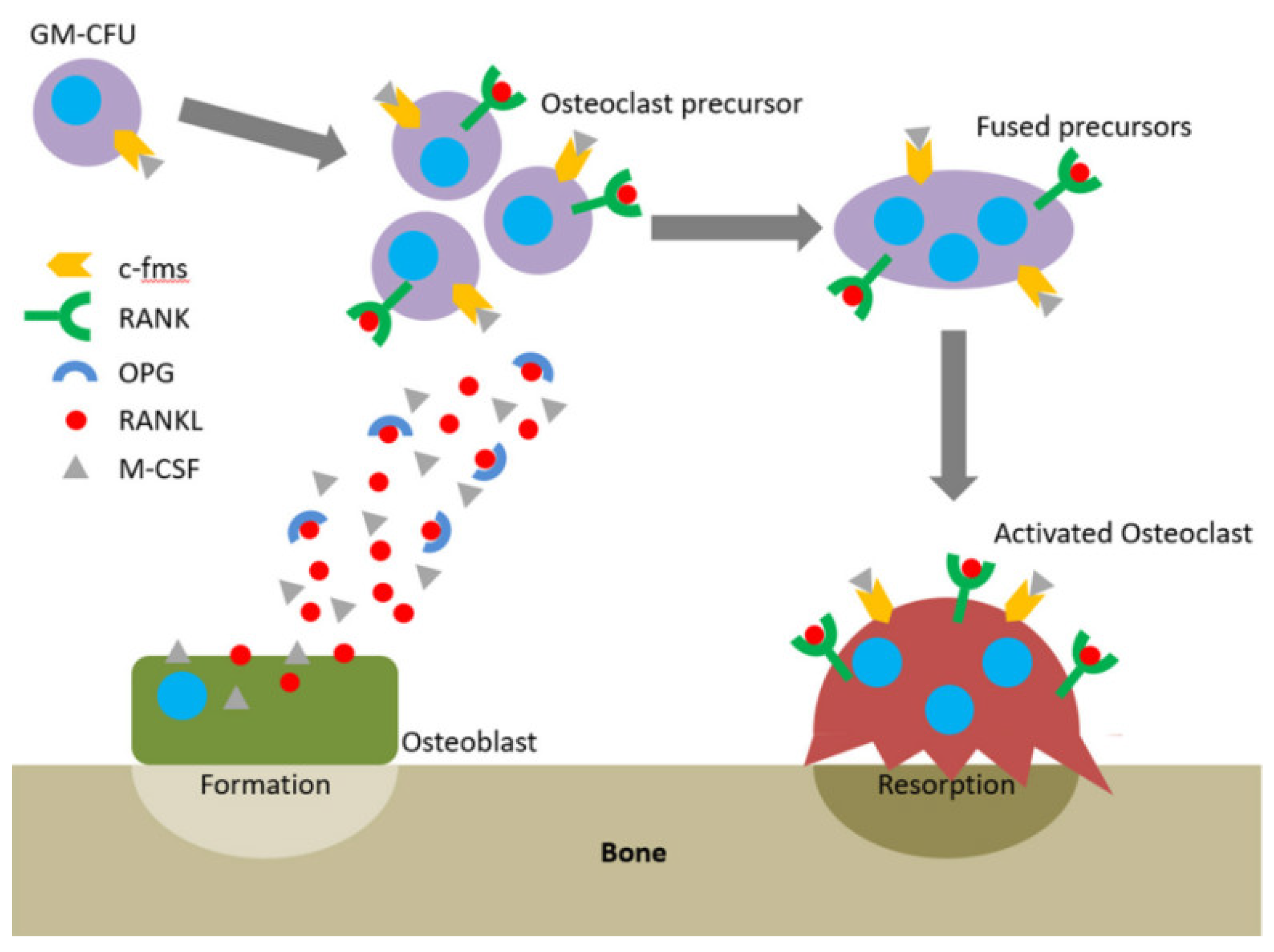
2. The Pathogenesis of Periodontitis Diseases
3. Drug Delivery Devices
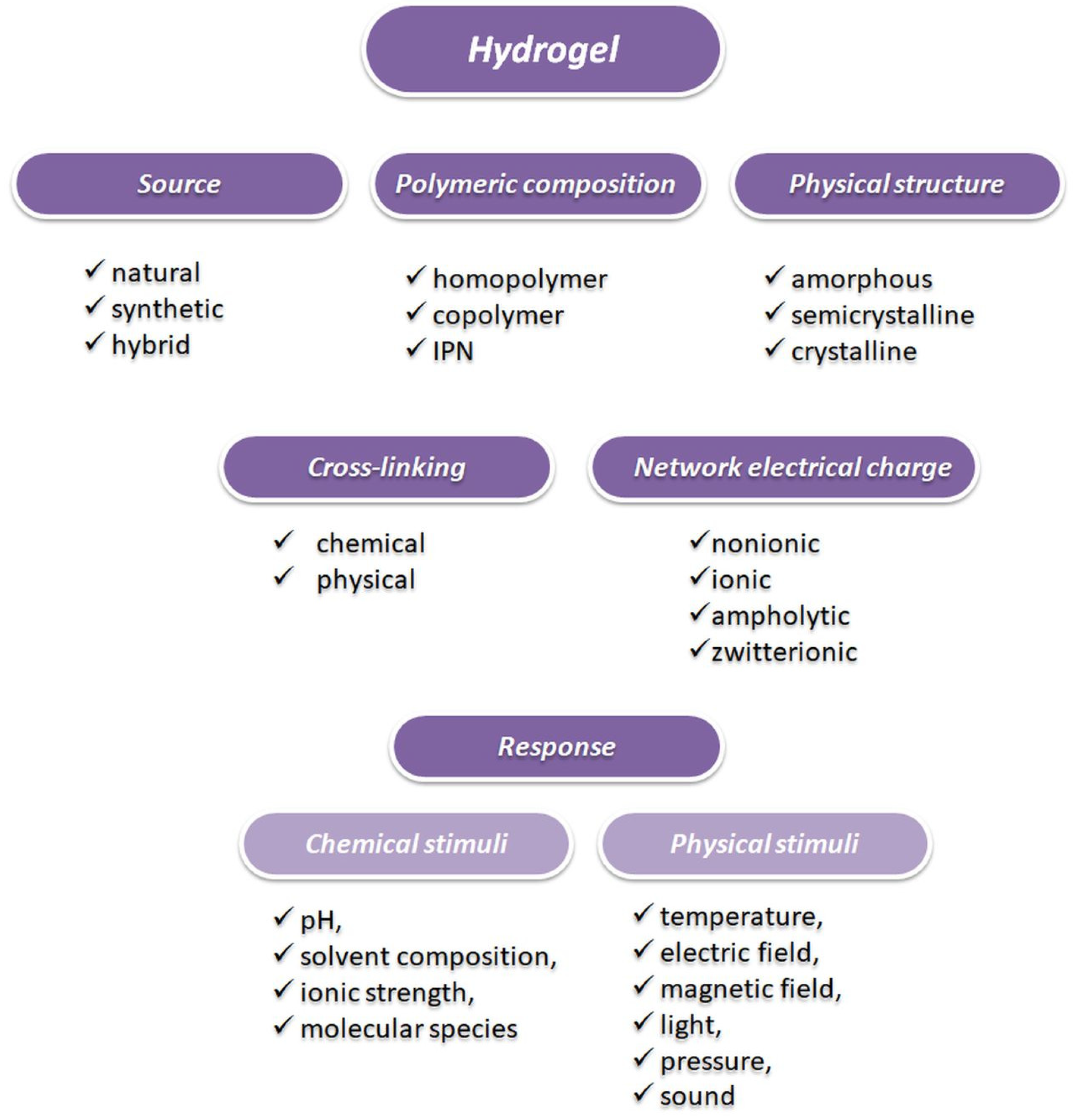
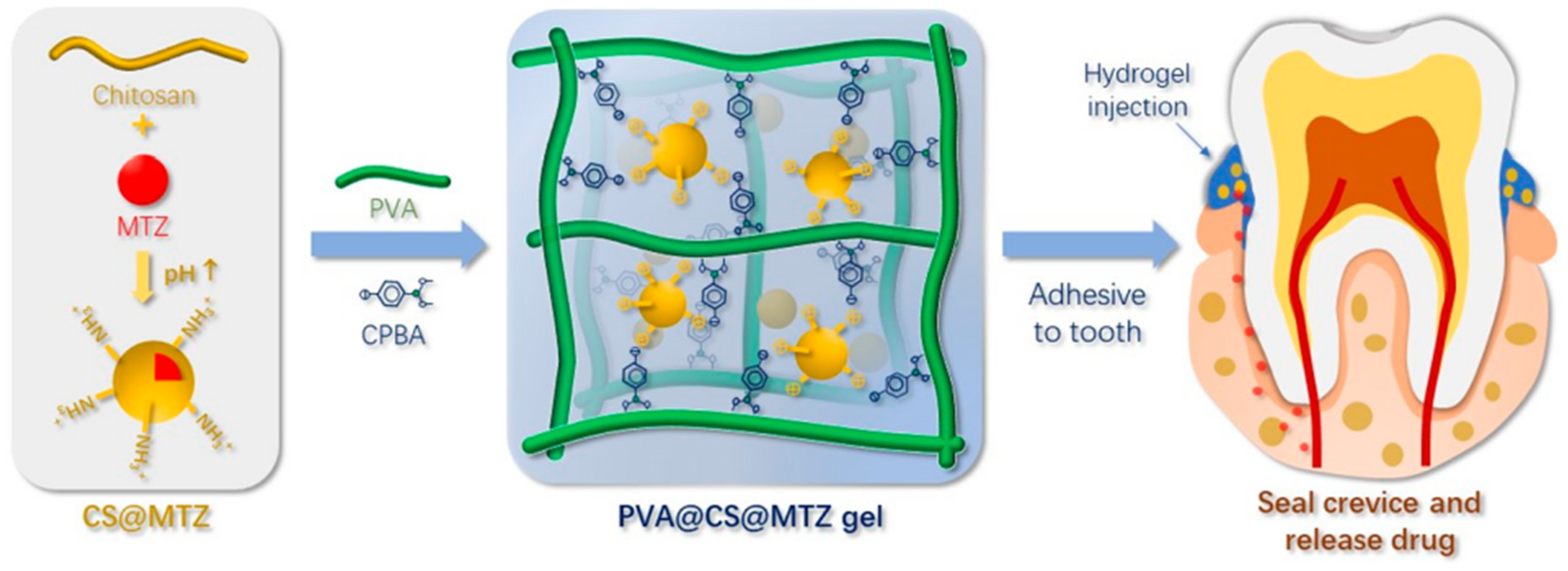
4. Hydrogels
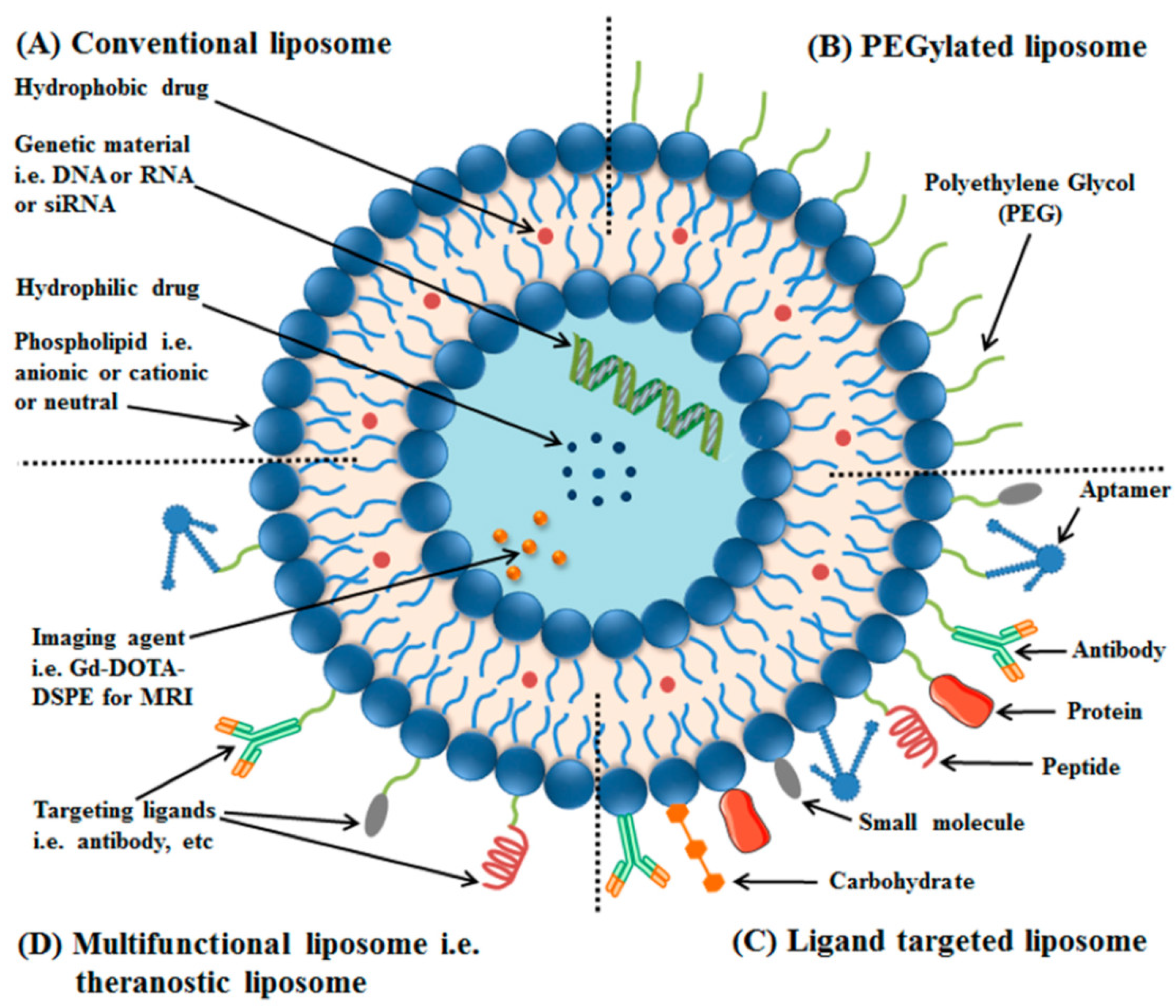
5. Liposomes
6. Micro- and Nanoparticles
7. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Khan, G.; Patel, R.R.; Yadav, S.K.; Kumar, N.; Chaurasia, S.; Ajmal, G.; Mishra, P.K.; Mishra, B. Development, optimization and evaluation of tinidazole functionalized electrospun poly(ε-caprolactone) nanofiber membranes for the treatment of periodontitis. RSC Adv. 2016, 6, 100214–100229. [Google Scholar] [CrossRef]
- Petersen, P.E.; Ogawa, H. Strengthening the prevention of periodontal disease: The WHO approach. J. Periodontol. 2005, 76, 2187–2193. [Google Scholar] [CrossRef]
- Dhamecha, H.R.; Jagwani, D.; Rao, S.; Jadhav, M.; Shaikh, K.; Puzhankara, S.; Jalalpure, S. Local drug delivery systems in the management of periodontitis: A scientific review. J. Control. Release 2019, 307, 393–409. [Google Scholar]
- Lim, S.Y.; Dafydd, M.; Ong, J.; Ord-McDermott, L.; Board-Davies, E.; Sands, K.; Williams, D.; Sloan, A.; Heard, C.M. Mucoadhesive thin films for the simultaneous delivery of microbicide and anti-inflammatory drugs in the treatment of periodontal diseases. Int. J. Pharm. 2020, 573, 118860. [Google Scholar] [CrossRef]
- Joshi, D.; Garg, T.; Goyal, A.K.; Rath, G. Advanced drug delivery approaches against periodontitis. Drug Deliv. 2016, 23, 363–377. [Google Scholar] [CrossRef] [PubMed]
- Zamani, F.; Jahanmard, F.; Ghasemkhah, F.; Amjad-Iranagh, S.; Bagherzadeh, R.; Amani-Tehran, M.; Latifi, M. Chapter 7 Nanofibrous and Nanoparticle Materials as Drug-Delivery Systems; Nanostructures for Drug Deliver; Andronescu, E., Grumezescu, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 239–270. [Google Scholar]
- Gaurav, N.J.; Shamama, K.J.; Zeenat, J.; Sushama, I.; Farhan, T.; Roop, J.A.; Khar, K. Recent approaches for the treatment of periodontitis. Drug Discov. Today 2008, 13, 932–943. [Google Scholar]
- Hamed, R.; AbuRezeq, A.; Tarawneh, O. Development of hydrogels, oleogels, and bigels as local drug delivery systems for periodontitis. Drug Dev. Ind. Pharm. 2018, 44, 1488–1497. [Google Scholar] [CrossRef]
- Greenstein, G.; Polson, A. The role of local drug delivery in the management of periodontal diseases: A comprehensive review. J. Periodontol. 1998, 69, 507–520. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. An overview of clinical and commercial impact of drug delivery systems. J. Controll. Release 2014, 190, 15–28. [Google Scholar] [CrossRef]
- Stewart, S.A.; Domínguez-Robles, J.; Donnelly, R.F.; Larrañeta, E. Implantable polymeric drug delivery devices: Classification, manufacture, materials, and clinical applications. Polymers 2018, 10, 1379. [Google Scholar] [CrossRef]
- Wang, B.; Wang, S.; Zhang, Q.; Deng, Y.; Li, X.; Peng, L.; Zuo, X.; Piao, M.; Kuang, X.; Sheng, S.; et al. Recent advances in polymer-based drug delivery systems for local anesthetics. Acta Biomater. 2019, 15, 55–67. [Google Scholar] [CrossRef] [PubMed]
- de la Portilla, F.; Pereira, S.; Molero, M.; De Marco, F.; Perez-Puyana, V.; Guerrero, A.; Romero, A. Microstructural, mechanical, and histological evaluation of modified alginate-based scaffolds. J. Biomed. Mater. Res. A 2016, 104, 3107–3114. [Google Scholar] [CrossRef] [PubMed]
- Ngwuluka, N.C.; Ochekpe, N.A.; Aruoma, O.I. Naturapolyceutics: The science of utilizing natural polymers for drug delivery. Polymers 2014, 6, 1312–1332. [Google Scholar] [CrossRef]
- Garg, V.; Chawla, K.; Pawar, S.K. Nanotechnology controlled local drug delivery system for the treatment of periodontitisc. J. Adv. Med. Med. Res. 2018, 26, 1–17. [Google Scholar] [CrossRef]
- Agrawal, P. Significance of polymers in drug delivery system. J. Pharmacovigilance. 2014, 3, e127. [Google Scholar] [CrossRef]
- Dong, Z.; Sun, Y.; Chen, Y.; Liu, Y.; Tang, C.; Qu, X. Injectable adhesive hydrogel through a microcapsule cross-link for periodontitis treatment. ACS Appl. Biol. Mater. 2019, 2, 5985–5994. [Google Scholar] [CrossRef]
- Paster, B.J.; Boches, S.K.; Galvin, J.L.; Ericson, R.E.; Lau, C.N.; Levanos, V.A.; Sahasrabudhe, A.; Dewhirst, F.E. Bacterial diversity in human subgingival plaque. J. Bacteriol. 2001, 183, 3770–3783. [Google Scholar] [CrossRef]
- Darveau, R.P. Periodontitis: A polymicrobial disruption of host homeostasis. Nat. Rev. Microbiol. 2010, 8, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.S.; Griffen, A.L.; Moeschberger, M.L.; Leys, E.J. Identification of candidate periodontal pathogens and beneficial species by quantitative 16S clonal analysis. J. Clin. Microbiol. 2005, 43, 3944–3955. [Google Scholar] [CrossRef]
- Kumar, P.S.; Leys, E.J.; Bryk, J.M.; Martinez, F.J.; Moeschberger, M.L.; Griffen, A.L. Changes in periodontal health status are associated with bacterial community shifts as assessed by quantitative 16S cloning and sequencing. J. Clin. Microbiol. 2006, 44, 3665–3673. [Google Scholar] [CrossRef]
- Lu, Q.; Jin, L.; Darveau, R.P.; Samaranayake, L.P. Expression of human β-defensins-1 and -2 peptides in unresolved chronic periodontitis. J. Periodontal Res. 2004, 39, 221–227. [Google Scholar] [CrossRef]
- Zeng, H.; Wu, H.; Sloane, V.; Jones, R.; Yu, Y.; Lin, P.; Gewirtz, A.T.; Neish, A.S. Flagellin/TLR5 responses in epithelia reveal intertwined activation of inflammatory and apoptotic pathways. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Eskan, M.A.; Hajishengallis, G.; Kinane, D.F. Differential activation of human gingival epithelial cells and monocytes by Porphyromonas gingivalis fimbriae. Infect Immun. 2007, 75, 892–898. [Google Scholar] [CrossRef]
- Lu, Y.C.; Yeh, W.C.; Ohashi, P.S. LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.K.; Kim, S.J.; Rah, S.H.; Kang, J.I.; Jung, H.E.; Lee, D.; Lee, H.K.; Lee, J.O.; Park, B.S.; Yoon, T.Y.; et al. Reconstruction of LPS transfer cascade reveals structural determinants within LBP, CD14, and TLR4-MD2 for efficient LPS recognition and transfer. Immunity 2017, 46, 38–50. [Google Scholar] [CrossRef]
- Raymond, S.L.; Holden, D.C.; Mira, J.C.; Stortz, J.A.; Loftus, T.J.; Mohr, A.M.; Moldawer, L.L.; Moore, F.A.; Larson, S.D.; Efron, P.A. Microbial recognition and danger signals in sepsis and trauma. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 2564–2573. [Google Scholar] [CrossRef] [PubMed]
- Nylander, K.; Danielsen, B.; Fejerskov, O.; Dabelsteen, E. Expression of the endothelial leukocyte adhesion Molecule-1 (ELAM-1) on endothelial cells in experimental gingivitis in humans. J. Periodontol. 1993, 64, 355–357. [Google Scholar] [CrossRef]
- Tonetti, M.S.; Imboden, M.A.; Lang, N.P. Neutrophil migration into the gingival sulcus is associated with Transepithelial gradients of interleukin-8 and ICAM-1. J. Periodontol. 1998, 69, 1139–1147. [Google Scholar] [CrossRef]
- Zeldin, D.C.; Eggleston, P.; Chapman, M.; Piedimonte, G.; Renz, H.; Peden, D. How exposures to biologics influence the induction and incidence of asthma. Environ. Health Perspect. 2006, 114, 4. [Google Scholar] [CrossRef]
- Ren, L.; Jin, L.; Leung, W.K. Local expression of lipopolysaccharide-binding protein in human gingival tissues. J. Periodontal. Res. 2004, 39, 242–248. [Google Scholar] [CrossRef]
- Burns, E.; Bachrach, G.; Shapira, L.; Nussbaum, G. Cutting edge: TLR2 is required for the innate response to Porphyromonas Gingivalis: Activation leads to bacterial persistence and TLR2 deficiency attenuates induced alveolar bone resorption. J. Immunol. 2006, 177, 8296–8300. [Google Scholar] [CrossRef] [PubMed]
- Graves, D.T.; Cochran, D. The contribution of Interleukin-1 and tumor necrosis factor to periodontal tissue destruction. J. Periodontol. 2003, 74, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Boyle Ocl. Rev. 2003, 423, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Owen, R.; Reilly, G.C. In vitro models of bone remodelling and associated disorders. Front. Bioeng. Biotechnol. 2018, 6, 134. [Google Scholar] [CrossRef] [PubMed]
- Cochran, D.L. Inflammation and bone loss in periodontal disease. J. Periodontol. 2008, 79, 1569–1576. [Google Scholar] [CrossRef]
- Crotti, T.; Smith, M.D.; Hirsch, R.; Soukoulis, S.; Weedon, H.; Capone, M.; Ahern, M.J.; Haynes, D. Receptor activator NF κB ligand (RANKL) and osteoprotegerin (OPG) protein expression in periodontitis. J. Periodontal Res. 2003, 38, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Wara-aswapati, N.; Surarit, R.; Chayasadom, A.; Boch, J.A.; Pitiphat, W. RANKL Upregulation associated with periodontitis and Porphyromonas gingivalis. J. Periodontol. 2007, 78, 1062–1069. [Google Scholar] [CrossRef]
- Assuma, R.; Oates, T.; Cochran, D.; Amar, S.; Graves, D.T. IL-1 and TNF antagonists inhibit the inflammatory response and bone loss in experimental periodontitis. J. Immunol. 1998, 160, 403–409. [Google Scholar]
- Baker, P.J.; Dixon, M.; Evans, R.T.; Dufour, L.; Johnson, E.; Roopenian, D.C. CD4+ T cells and the proinflammatory cytokines gamma interferon and Interleukin-6 contribute to alveolar bone loss in mice. Infect Immun. 1999, 67, 2804–2809. [Google Scholar] [CrossRef]
- Franco, C.; Patricia, H.R.; Timo, S.; Claudia, B.; Marcela, H. Matrix metalloproteinases as regulators of periodontal inflammation. Int. J. Mol. Sci. 2017, 18, 440. [Google Scholar] [CrossRef]
- Mäntylä, P.; Stenman, M.; Kinane, D.; Salo, T.; Suomalainen, K.; Tikanoja, S.; Sorsa, T. Monitoring periodontal disease status in smokers and nonsmokers using a gingival crevicular fluid matrix metalloproteinase-8-specific chair-side test. J. Periodontal Res. 2006, 41, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Hernández Ríos, M.; Sorsa, T.; Obregón, F.; Tervahartiala, T.; Valenzuela, M.A.; Pozo, P.; Dutzan, N.; Lesaffre, E.; Molas, M.; Gamonal, J. Proteolytic roles of matrix metalloproteinase (MMP)-13 during progression of chronic periodontitis: Initial evidence for MMP-13/MMP-9 activation cascade. J. Clin. Periodontol. 2009, 36, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, R.; Asopa Swati, J.; Joseph, M.D.; Singh, B.; Rajguru Jagadish, P.; Saidath, K.; Sharma, U. Red complex: Polymicrobial conglomerate in oral flora: A review. J. Family Med. Prim. Care 2019, 8, 3480–3486. [Google Scholar]
- Zenobia, C.; Hajishengallis, G. Porphyromonas gingivalis virulence factors involved in subversion of leukocytes and microbial dysbiosis. Virulence 2015, 6, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Coats, S.R.; Jones, J.W.; Do, C.T.; Braham, P.H.; Bainbridge, B.W.; To, T.T.; Goodlett, D.R.; Ernst, R.K.; Darveau, R.P. Human Toll-like receptor 4 responses to P. gingivalis are regulated by lipid A 1-and 4’-phosphatase activities. Cell. Microbiol. 2009, 11, 1587–1599. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Wang, M.; Liang, S.; Triantafilou, M.; Triantafilou, K. Pathogen induction of CXCR4/TLR2 cross-talk impairs host defense function. Proc. Natl. Acad. Sci. USA 2008, 105, 13532–13537. [Google Scholar] [CrossRef]
- Naito, M.; Sakai, E.; Shi, Y.; Ideguchi, H.; Shoji, M.; Ohara, N.; Yamamoto, K.; Nakayama, K. Porphyromonas gingivalis-induced platelet aggregation in plasma depends on Hgp44 adhesin but not Rgp proteinase. Mol. Microbiol. 2006, 59, 152–167. [Google Scholar] [CrossRef]
- Yun, Y.H.; Lee, B.K.; Park, K. Controlled drug delivery: Historical perspective for the next generation. J. Control. Release 2015, 219, 2–7. [Google Scholar] [CrossRef]
- Goodson, J.; Haffajee, A.; Socransky, S. Periodontal therapy by local delivery of tetracycline. J. Clin. Periodontol. 1979, 6, 83–92. [Google Scholar] [CrossRef]
- Kohane, D.S.; Langer, R. Biocompatibility and drug delivery systems. Chem. Sci. 2010, 1, 441–446. [Google Scholar] [CrossRef]
- Nair, S.C.; Anoop, K. Intraperiodontal pocket: An ideal route for local antimicrobial drug delivery. J. Adv. Pharm. Technol. Res. 2012, 3, 9. [Google Scholar]
- Fu, Y.; Kao, W.J. Drug release kinetics and transport mechanisms from semiinterpenetrating networks of gelatin and poly(ethylene glycol) diacrylate. Pharm. Res. 2009, 26, 2115–2124. [Google Scholar] [CrossRef] [PubMed]
- Langer, R.; Peppas, N. Chemical and physical structure of polymers as carriers for controlled release of bioactive agents: A review. J. Macromol. Sci. Rev. Macromol. Chem. Phys. 1983, 23, 61–126. [Google Scholar] [CrossRef]
- Arifin, D.Y.; Lee, L.Y.; Wang, C.-H. Mathematical modeling and simulation of drug release from microspheres: Implications to drug delivery systems. Adv. Drug Deliv. Rev. 2006, 58, 1274–1325. [Google Scholar] [CrossRef]
- Fu, Y.; Kao, W.J. Drug release kinetics and transport mechanisms of non-degradable and degradable polymeric delivery systems. Expert Opin. Drug Deliv. 2010, 7, 429–444. [Google Scholar] [CrossRef] [PubMed]
- Acharya, G.; Park, K. Mechanisms of controlled drug release from drug-eluting stents. Adv. Drug Deliv. Rev. 2006, 58, 387–401. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, D.M.; Chagas, P.A.; Leite, I.S.; Inada, N.M.; de Annunzio, S.R.; Fontana, C.R.; Campana-Filho, S.P.; Correa, D.S. Core-sheath nanostructured chitosan-based nonwovens as a potential drug delivery system for periodontitis treatment. Int. J. Biol. Macromol. 2020, 142, 521–534. [Google Scholar] [CrossRef]
- Parhi, R. Drug delivery applications of chitin and chitosan: A review. Environ. Chem. Lett. 2020, 18, 577–594. [Google Scholar] [CrossRef]
- Gerstel, M.S.; Place, V.A. Drug Delivery Device. U.S. Patent 3,964,482A, 22 June 1976. [Google Scholar]
- Greenstein, G.; Tonetti, M. The role of controlled drug delivery for periodontitis. The research, science and therapy committee of the American academy of periodontology. J. Periodontol. 2000, 71, 125–140. [Google Scholar]
- Zamani, M.; Morshed, M.; Varshosaz, J.; Jannesari, M. Controlled release of metronidazole benzoate from poly ε-caprolactone electrospun nanofibers for periodontal diseases. Eur. J. Pharm. Biopharm. 2010, 75, 179–185. [Google Scholar] [CrossRef]
- Esposito, E.; Cortesi, R.; Cervellati, F.; Menegatti, E.; Nastruzzi, C. Biodegradable microparticles for sustained delivery of tetracycline to the periodontal pocket: Formulatory and drug release studies. J. Microencapsul. 1997, 14, 175–187. [Google Scholar] [CrossRef] [PubMed]
- El-Kamel, A.H.; Ashri, L.Y.; Alsarra, I.A. Micromatricial metronidazole benzoate film as a local mucoadhesive delivery system for treatment of periodontal diseases. AAPS Pharmscitech. 2007, 8, E184–E194. [Google Scholar] [CrossRef] [PubMed]
- Llindhe, J.; Heijl, L.; Goodson, J.M.; Socransky, S. Local tetracycline delivery using hollow fiber devices in periodontal therapy. J. Clin. Periodontol. 1979, 6, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Goodson, J.; Holborow, D.; Dunn, R.; Hogan, P.; Dunham, S. Monolithic tetracycline-containing fibers for controlled delivery to periodontal pockets. J. Periodontol. 1983, 54, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Goodson, J.; Offenbacher, S.; Farr, D.; Hogan, P. Periodontal disease treatment by local drug delivery. J. Periodontol. 1985, 56, 265–272. [Google Scholar] [CrossRef]
- Hasnain, M.S.; Rishishwar, P.; Ali, S.; Nayak, A.K. Preparation and evaluation of aceclofenac dental pastes using dillenia fruit gum for periodontitis treatment. SN Appl. Sci. 2020, 2, 1–8. [Google Scholar] [CrossRef]
- Ashri, L.Y.; Amal El Sayeh, F.; Ibrahim, M.A.; Alshora, D.H. Optimization and evaluation of chitosan buccal films containing tenoxicam for treating chronic periodontitis: In vitro and in vivo studies. J. Drug Deliv. Sci. Tech. 2020, 57, 101720. [Google Scholar] [CrossRef]
- Osorio, R.; Alfonso-Rodrı´guez, C.A.; Medina Castillo, A.L.; Alaminos, M.; Toledano, M. Bioactive polymeric nanoparticles for periodontal therapy. PLoS ONE 2016, 11, e0166217. [Google Scholar] [CrossRef]
- Beg, S.; Dhiman, S.; Sharma, T.; Jain, A.; Sharma, R.K.; Jain, A.; Singh, B. Stimuli responsive in situ gelling systems loaded with PLGA nanoparticles of moxifloxacin hydrochloride for effective treatment of periodontitis. AAPS PharmSciTech 2020, 21, 76. [Google Scholar] [CrossRef]
- Takahashi, C.; Hattori, Y.; Yagi, S.; Murai, T.; Tanemura, M.; Kawashima, Y.; Yamamoto, H. Ionic liquid-incorporated polymeric nanoparticles as carriers for prevention and at an earlier stage of periodontal disease. Materialia 2019, 8, 100395. [Google Scholar] [CrossRef]
- Gjoseva, S.; Geskovski, N.; Dimchevska Sazdovska, S.; Popeski-Dimovski, R.; Petruševski, G.; Mladenovska, K.; Goracinova, K. Design and biological response of doxycycline loaded chitosan microparticles for periodontal disease treatment. Carbohydr. Polym. 2018, 186, 260–272. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.G.; Harish, N.M.; Charyulu, R.N.; Prabhu, P. Formulation of chitosan-based ciprofloxacin and diclofenac film for periodontitis therapy. Trop. J. Pharm. Res. 2009, 8, 33–41. [Google Scholar] [CrossRef][Green Version]
- Khan, G.; Yadav, S.K.; Patel, R.R.; Kumar, N.; Bansal, M.; Mishra, B. Tinidazole functionalized homogeneous electrospun chitosan/poly (ε-caprolactone) hybrid nanofiber membrane: Development, optimization and its clinical implications. Int. J. Biol. Macromol. 2017, 103, 1311–1326. [Google Scholar] [CrossRef] [PubMed]
- Kaewiad, K.; Nakpheng, T.; Srichana, T. Dental floss impregnated with povidone-iodine coated with Eudragit L-100 as an antimicrobial delivery system against periodontal-associated pathogens. J. Med. Microbiol. 2020, 69, 298–308. [Google Scholar] [CrossRef]
- Available online: http://tchie.uni.opole.pl/freeECE/S_17_2/Zarzycki_17(S2).pdf (accessed on 25 May 2020).
- Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for biomedical applications: Their characteristics and the mechanisms behind them. Gels 2017, 3, 6. [Google Scholar] [CrossRef]
- Narayanaswamy, R.; Torchilin, V.P. Hydrogels and their applications in targeted drug delivery. Molecules 2019, 24, 603. [Google Scholar] [CrossRef]
- Li, J.; Mooney, D.J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 16071. [Google Scholar] [CrossRef]
- Liang, J.; Peng, X.; Zhou, X.; Zou, J.; Cheng, L. Emerging applications of drug delivery systems in oral infectious diseases prevention and treatment. Molecules 2020, 25, 516. [Google Scholar] [CrossRef]
- Croisfelt, F.M.; Tundisi, L.L.; Ataide, J.A.; Silveira, E.; Tambourgi, E.B.; Jozala, A.F.; Barbosa Souto, E.M.; Gava Mazzola, P. Modified-release topical hydrogels: A ten-year review. J. Mater. Sci. 2019, 54, 10963–10983. [Google Scholar] [CrossRef]
- Singha, R.; Gupta, K. A review: Tailor-made hydrogel structures (classifications and synthesis parameters). Polym.-Plast. Tech. Eng. 2016, 55, 54–70. [Google Scholar] [CrossRef]
- Dabhi, M.R.; Nagori, S.A.; Gohel, M.C.; Parikh, R.K.; Sheth, N.R. Formulation development of smart gel periodontal drug delivery system for local delivery of chemotherapeutic agents with application of experimental design. Drug Deliv. 2010, 17, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.S.; Woolfson, A.D.; Brown, A.F.; O’Neill, M.J. Mucoadhesive, syringeable drug delivery systems for controlled application of metronidazole to the periodontal pocket: In vitro release kinetics, syringeability, mechanical and mucoadhesive properties. J. Control. Release 1997, 49, 71–79. [Google Scholar] [CrossRef]
- Fujimoto, K.; Minami, N.; Goto, T.; Ishida, Y.; Watanabe, M.; Nagao, K.; Ichikawa, T. Hardness, cohesiveness, and adhesiveness of oral moisturizers and denture adhesives: Selection criteria for denture wearers. Dent. J. 2016, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, F.C.; Calixto, G.; Hatakeyama, I.N.; Luz, G.M.; Gremião, M.P.D.; Chorilli, M. Rheological, mechanical, and bioadhesive behavior of hydrogels to optimize skin delivery systems. Drug Dev. Ind. Pharm. 2012, 39, 1750–1757. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Torres, R.; López-Arellano, R.; Escobar-Chávez, J.J.; García-García, E.; Domínguez-Delgado, C.L.; Ramírez-Noguera, P. Effect of size and functionalization of pharmaceutical nanoparticles and their interaction with biological systems. In Handbook of Nanoparticles; Aliofkhazraei, M., Ed.; Springer: Cham, Switzerland, 2016; pp. 1041–1060. [Google Scholar]
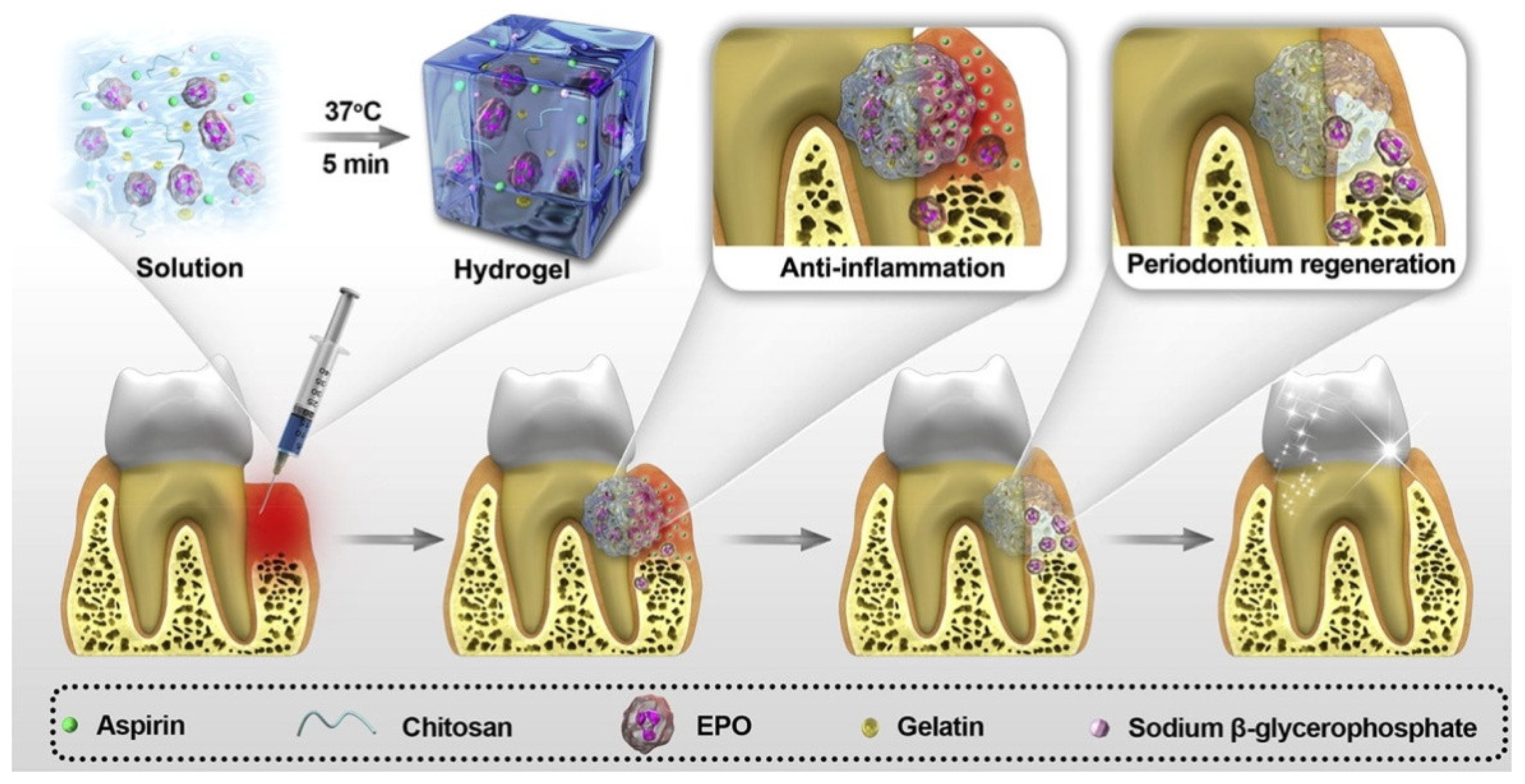
- Xu, X.; Gu, Z.; Chen, X.; Shi, C.; Liu, C.; Liu, M.; Wang, L.; Sunc, M.; Zhang, K.; Liu, Q.; et al. An injectable and thermosensitive hydrogel: Promoting periodontal regeneration by controlled-release of aspirin and erythropoietin. Acta Biomater. 2019, 86, 235–246. [Google Scholar] [CrossRef]
- Kumar, S.; Mukherjee, A.; Dutta, J. Chitosan based nanocomposite films and coatings: Emerging antimicrobial food packaging alternatives. Trends Food Sci. Technol. 2020, 97, 196–209. [Google Scholar] [CrossRef]
- Chung, C.H.; Golub, E.E.; Forbes, E.; Tokuoka, T.; Shapiro, I.M. Mechanism of action on β-glycerophosphate on bone cell mineralization. Calcif. Tissue Int. 1992, 51, 305–311. [Google Scholar] [CrossRef]
- Ke, X.; Li, M.; Wang, X.; Liang, J.; Wang, X.; Wu, S.; Long, M.; Hu, C. An injectable chitosan/dextran/β -glycerophosphate hydrogel as cell delivery carrier for therapy of myocardial infarction. Carbohydr. Polym. 2020, 229, 115516. [Google Scholar] [CrossRef]
- Ji, Q.X.; Chen, X.G.; Zhao, Q.S.; Liu, C.S.; Cheng, X.J.; Wang, L.C. Injectable thermosensitive hydrogel based on chitosan and quaternized chitosan and the biomedical properties. J. Mater. Sci. Mater. Med. 2009, 20, 1603–1610. [Google Scholar] [CrossRef]
- Ji, Q.X.; Zhao, Q.S.; Deng, J.; Lü, R. A novel injectable chlorhexidine thermosensitive hydrogel for periodontal application: Preparation, antibacterial activity and toxicity evaluation. J. Mater. Sci. Mater. Med. 2010, 21, 2435–2442. [Google Scholar]
- Swain, G.P.; Patel, S.; Gandhi, J.; Shah, P. Development of Moxifloxacin Hydrochloride loaded in-situ gel for the treatment of periodontitis: In-vitro drug release study and antibacterial activity. J. Oral. Biol. Craniofac. Res. 2019, 9, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, D.F.; Sant, S.V.; Shin, H.; Oliveira, J.T.; Gomes, M.E.; Neves, N.M.; Khademhosseini, A.; Reis, R.L. Modified Gellan gum hydrogels with tunable physical and mechanical properties. Biomaterials 2010, 31, 7494–7502. [Google Scholar] [CrossRef]
- Nakanishi, T.; Kaiho, F.; Hayashi, M. Improvement of drug release rate from carbopol 934P formulation. Chem. Pharm. Bull. (Tokyo) 1998, 46, 171–173. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hirun, N.; Tantishaiyakul, V.; Sangfai, T.; Ouiyangkul, P.; Li, L. In situ mucoadhesive hydrogel based on methylcellulose/xyloglucan for periodontitis. J. Sol-Gel Sci. Tech. 2020, 89, 531–542. [Google Scholar] [CrossRef]
- Mou, J.; Liu, Z.; Liu, J.; Lu, J.; Zhu, W.; Pei, D. Hydrogel containing minocycline and zinc oxide-loaded serum albumin nanopartical for periodontitis application: Preparation, characterization and evaluation. Drug Deliv. 2019, 26, 179–187. [Google Scholar] [CrossRef]
- Daraee, H.; Etemadi, A.; Kouhi, M.; Alimirzalu, S.; Akbarzadeh, A. Application of liposomes in medicine and drug delivery. Artif. Cells Nanomed. Biotechnol. 2016, 44, 381–391. [Google Scholar] [CrossRef]
- Alavi, M.; Karimi, N.; Safaei, M. Application of various types of liposomes in drug delivery systems. Adv. Pharm. Bull. 2017, 7, 3–9. [Google Scholar] [CrossRef]
- Perrie, Y.; Gregoriadis, G. Introducing liposomes to drug delivery. J. Drug Target. 2008, 16, 518–519. [Google Scholar] [CrossRef]
- Tayebi, L. Applications of Biomedical Engineering in Dentistry, Chapter 17: Injectable Gels for Dental and Craniofacial Applications; Springer International Publishing: Cham, Switzerland, 2020; pp. 359–375. [Google Scholar]
- Akbarzadeh, A.; Rezaei-Sadabady, R.; Davaran, S.; Joo, S.W.; Zarghami, N.; Hanifehpour, Y.; Samiei, M.; Kouhi, M.; Nejati-Koshki, K. Liposome: Classification, preparation, and applications. Nanoscale Res. Lett. 2013, 8, 102. [Google Scholar] [CrossRef]
- Available online: https://www.intechopen.com/books/application-of-nanotechnology-in-drug-delivery/liposomes-as-potential-drug-carrier-systems-for-drug-delivery (accessed on 16 June 2020).
- Riaz, M.K.; Riaz, M.A.; Zhang, X.; Lin, C.; Wong, K.H.; Chen, X.; Zhang, G.; Lu, A.; Yang, Z. Surface functionalization and targeting strategies of liposomes in solid tumor therapy: A review. Int. J. Mol. Sci. 2018, 19, 195. [Google Scholar] [CrossRef]
- Di Turp, G.; Riggi, C.; Vittorio, O.; Marconcin, S.; Brigugli, F.; Funev, N.; Campanp, D.; Barone, A.; Raffa, V.U. Sub-Micrometric liposomes as drug delivery systems in the treatment of periodontitis. Int. J. Immunopathol. Pharmacol. 2012, 25, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yang, P.S. Minocycline hydrochloride nanoliposomes inhibit the production of TNF-α in LPS-stimulated macrophages. Int. J. Nanomed. 2012, 7, 4769–4775. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Noguez Méndez, N.A.; Quirino Barreda, C.T.; Vega, A.F.; Miranda Calderon, J.E.; Urioste, C.G.; Palomec, X.C.; Martínez, A.R.; Díaz, M.P. Design and development of pharmaceutical microprocesses in the production of nanomedicine. In Nanostructures for Oral Medicine, 1st ed.; Andronescu, E., Grumezescu, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 669–697. [Google Scholar]
- Pathak, C.; Vaidya, F.U.; Pandey, S.M. Chapter 3—Mechanism for Development of Nanobased Drug Delivery System. In Micro and Nano Technologies, Applications of Targeted Nano Drugs and Delivery Systems; Mohapatra, S.S., Ranjan, S., Dasgupta, N., Mishra, R.K., Thomas, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 35–67. [Google Scholar]
- Powers, K.W.; Palazuelos, M.; Moudgil, B.M.; Roberts, S.M. Characterization of the size, shape, and state of dispersion of nanoparticles for toxicological studies. Nanotoxicology 2007, 1, 42–51. [Google Scholar] [CrossRef]
- Campos, E.; Branquinho, J.; Carreira, A.S.; Carvalho, A.; Coimbra, P.; Ferreira, P.; Gil, M.H. Designing polymeric microparticles for biomedical and industrial applications. Eur. Polym. J. 2013, 49, 2005–2021. [Google Scholar] [CrossRef]
- Teekamp, N.; Duque, L.F.; Frijlink, H.W.; Hinrichs, W.L.J.; Olinga, P. Production methods and stabilization strategies for polymer-based nanoparticles and microparticles for parenteral delivery of peptides and proteins. Expert Opin. Drug Deliv. 2015, 12, 1311–1331. [Google Scholar] [CrossRef]
- Lu, J.; Ren, B.; Wang, L.; Li, M.; Liu, Y. Preparation and evaluation of IL-1ra-Loaded Dextran/PLGA microspheres for inhibiting periodontal inflammation in vitro. Inflammation 2020, 43, 168–178. [Google Scholar] [CrossRef]
- Bakshi, P.S.; Selvakumar, D.; Kadirvelu, K.; Kumar, N.S. Chitosan as an environment friendly biomaterial—a review on recent modifications and applications. Int. J. Biol. Macromol. 2020, 150, 1072–1083. [Google Scholar] [CrossRef]
- Wang, B.; Wang, J.; Shao, J.; Kouwer, P.H.J.; Bronkhorst, E.M.; Jansen, J.A.; Walboomers, F.; Yang, F. A tunable and injectable local drug delivery system for personalized periodontal application. J. Control. Release 2020, 324, 134–145. [Google Scholar] [CrossRef]
- Novac, O.; Lisa, G.; Profire, L.; Tuchilus, C.; Popa, M.I. Antibacterial quaternized gellan gum based particles for controlled release of ciprofloxacin with potential dermal applications. Mater. Sci. Eng. C 2014, 35, 291–299. [Google Scholar] [CrossRef]
- Piñón-Segundo, E.; Ganem-Quintanar, A.; Alonso-Pérez, V.; Quintanar-Guerrero, D. Preparation and characterization of triclosan nanoparticles for periodontal treatment. Int. J. Pharm. 2005, 294, 217–232. [Google Scholar] [CrossRef]
- Gad, H.A.; El-Nabarawi, M.A.; El-Hady, S.S.A. Formulation and evaluation of PLA and PLGA in situ implants containing secnidazole and/or doxycycline for treatment of periodontitis. AAPS Pharm. Sci. Tech. 2008, 9, 878–884. [Google Scholar] [CrossRef] [PubMed]
- Chi, M.; Qi, M.; Wang, P.; Weir, M.D.; Melo, M.A.; Sun, X.; Dong, B.; Li, C.; Wu, J.; Wang, L.; et al. Novel bioactive and therapeutic dental polymeric materials to inhibit periodontal pathogens and biofilms. Int. J. Mol. Sci. 2019, 20, 278. [Google Scholar] [CrossRef] [PubMed]
- Schwach-Abdellaoui, K.; Vivien-Castioni, N.; Gurny, R. Local delivery of antimicrobial agents for the treatment of periodontal diseases. Eur. J. Pharm. Biopharm. 2000, 50, 83–99. [Google Scholar] [CrossRef]
- Mota, J.; Yu, N.; Caridade, S.G.; Luz, G.M.; Gomes, M.E.; Reis, R.L.; Jansen, J.A.; Walboomers, X.F.; Mano, J.F. Chitosan/bioactive glass nanoparticle composite membranes for periodontal regeneration. Acta Biomater. 2012, 8, 4173–4180. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zięba, M.; Chaber, P.; Duale, K.; Martinka Maksymiak, M.; Basczok, M.; Kowalczuk, M.; Adamus, G. Polymeric Carriers for Delivery Systems in the Treatment of Chronic Periodontal Disease. Polymers 2020, 12, 1574. https://doi.org/10.3390/polym12071574
Zięba M, Chaber P, Duale K, Martinka Maksymiak M, Basczok M, Kowalczuk M, Adamus G. Polymeric Carriers for Delivery Systems in the Treatment of Chronic Periodontal Disease. Polymers. 2020; 12(7):1574. https://doi.org/10.3390/polym12071574
Chicago/Turabian StyleZięba, Magdalena, Paweł Chaber, Khadar Duale, Magdalena Martinka Maksymiak, Maciej Basczok, Marek Kowalczuk, and Grazyna Adamus. 2020. "Polymeric Carriers for Delivery Systems in the Treatment of Chronic Periodontal Disease" Polymers 12, no. 7: 1574. https://doi.org/10.3390/polym12071574
APA StyleZięba, M., Chaber, P., Duale, K., Martinka Maksymiak, M., Basczok, M., Kowalczuk, M., & Adamus, G. (2020). Polymeric Carriers for Delivery Systems in the Treatment of Chronic Periodontal Disease. Polymers, 12(7), 1574. https://doi.org/10.3390/polym12071574







