In our previous work, it was shown that PDMS patches have unique features that can be useful for the design of dermal drug delivery carriers [
6,
7]. The present study explored the possibility of modification of PDMS films to enhance the release of a model drug—IND. Since many physicochemical features can affect the drug release mechanism, a multiple approach was considered essential. Firstly, the study focused on increasing the release rate by drug solubility enhancement in the matrix. PEG, PG and low-viscosity SO were chosen as solvents, hence dissolved fraction of IND in both liquid and solid PDMS component was assessed. Additionally, the liquid additives were supposed to alter the inner structure of a solid PDMS matrix. Estimation of the relevance of each of these factors for transdermal patch formulation performance was conducted.
The combination of three microscopic techniques was used to characterize in details the microstructure of PDMS films. SEM was used to provide a general overview of the film morphology, whereas fluorescent microscopy was employed to determine the distribution of IND within the polymeric film and assess the size of IND particles. AFM was used to more closely investigate the two-phase microstructure created by the liquid additives. Apart from the drug release experiments, water transport in the PMDS films modified with the additives was evaluated in order to characterize diffusion dynamics.
3.1. Fluorescent Microscope Imaging
Fluorescence microscopy with digital image analysis allowed a clear overview of the suspended IND particles without interference from the PDMS background, since the liquid additives caused no visible changes in PDMS fluorescence microscopic image.
Figure 1, in the left column, shows microscopic images of the cross-section of the additive-free films and the films with one of the liquid additives, all loaded with IND (5%).
Preliminary examination at 10× magnitude confirmed the uniform distribution of IND particles across the full-thickness films in all PDMS blends. SO, PEG and PG added at the first stage of film preparation allowed elimination of particle aggregates visible in the images of the additive-free formulation (up to maximum size of 10 μm). Therefore, the liquid additives allow a homogenous distribution of the suspended particles within the matrix if the proposed compounding protocol is applied.
Table 1 presents a comparison of IND particle size in the powder used for film preparation and in PDMS films. No significant differences in the mean particle size was found for the examined blends.
In case of PEG and PG films an increased light intensity from a matrix was considered to be the effect of IND, which dissolved in the matrix (
Table 1). Further analysis of the acquired 3D images (
Figure 1, right column) allowed assessment of the particle volume fraction within the film and estimation of whether the liquid additives affect the size and number of IND particles in the PDMS films. A clear difference, depending on the additive type, was observed (
Table 1). Approximately 30% of the analyzed volume of PDMS and PDMS–SO films was identified as IND particles. Significant decrease of the solid particles volume (down to 7%) was noted for both hydrophilic excipients (PEG and PG), which explained the previous observation of the amplified matrix luminescence. To further investigate the observed phenomena, experimental data were correlated with the theoretically estimated fraction of IND dissolved in the films (DF), where calculation was based on IND solubility in the additives and in PDMS. DF value for the SO containing film was found comparable to the additive-free film, which agrees with the particle volume fraction and confirms similarity of those formulations in terms of IND distribution. Theoretical estimation of IND fraction dissolved in the additive (DF) suggested, that a significant amount of the active substance can be present in the molecular state as dissolved in a hydrophilic phase of the films. This assumption is consistent with much lower fraction of the solid particles observed in the films with PG and PEG.
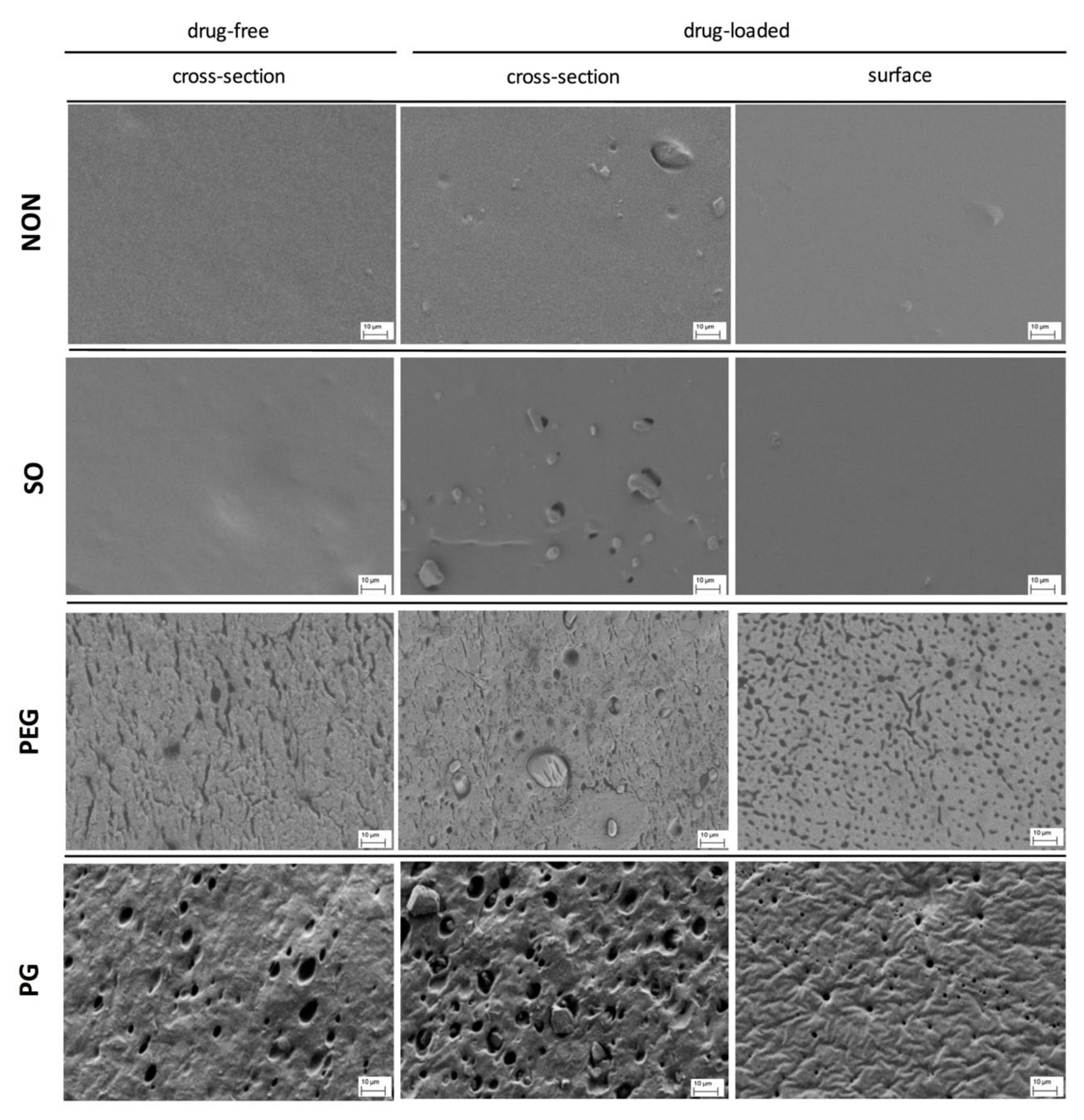
3.2. SEM Imaging
Scanning electron microscopy was used to study the films morphology. Generally, it allowed observation of the spatial distribution of the separated components and changes caused by the additives in the inner structure of the films.
The most pronounced differences in the film structure were observed for the samples containing PEG or PG, where the pore-resembling structures were observed, whilst SO had no visible effect on the basic PDMS structure (
Figure 2). IND particles were visible in drug-loaded films, with size resembling the mean size of the particles given in
Table 1.
In the case of both hydrophilic liquid additives, the excipient-rich domains can be clearly distinguished. For PG film, a pin-hole-like (max. 2 μm in diameter) topography of the surface can be noticed, whereas the cross-section revealed a homogenous distribution of droplet-like structures of 5–10 μm in diameter. The microstructure formed within the network can be described as highly uniform and regular in the pore arrangement. In contrast, PEG-loaded film differed in both shape and size of the observed structures: more irregular, channel-shaped formations can be seen with only single droplets present (up to 5 μm). Further examination of the film surface has shown multiple droplets with a considerable number of particles with elongated shapes.
As expected, for the two examined hydrophilic excipients a phase separation in the patch was clearly visible. However, PEG was found to be the most pronounced pore-former regarding the pore shape and distribution. Since the additives generated diverse networks in PDMS, different properties of these films may be expected.
3.3. AFM Analysis
To further investigate the two-phase structures created by the addition of hydrophilic liquid additives, atomic force microscopy was employed.
Figure 3 presents an example of 3D images of cross-sections of the IND-loaded films. In each of the examined films, single IND particles were observed with sizes consistent with previous findings.
Roughness of the surface for the additive-free film was noted, which was contributed by the amorphous silica particles that are commonly added to silicone biomaterials as a reinforcing filler [
4]. It can be seen that a slightly smoother plane was achieved with the addition of SO to the film. The texture of PG-loaded films was regular, and the presence of spherical droplets, already noticed in the corresponding SEM images, was confirmed. Since this dot-like structure was surrounded by smooth PDMS regions, this leads to the conclusion that PG droplets do not form a network but are homogenously distributed within the film. In contrast, within examined cross-sections PEG created rough and elongated irregular structures. Moreover, due to the presence of PEG, complex channel-like domains make a larger contribution to the highly heterogeneous structure, that can be seen in AFM (
Figure 3), which might have not been concluded from SEM images alone.
3.4. Mechanical Characteristics of PDMS Films
Mechanical properties of the films were characterized by tensile strength (TS), Young modulus (Module E) and elongation at break (% EB) values presented in
Table 2, together with the calculated density of the films. As suggested before, it was hypothesized that the matrix inner structure may influence the mechanical properties of the films profoundly. When the pure PDMS film was used as a reference, multiple effects were observed for composed films, with statistical significance of the differences confirmed, which varied depending on the additive type and the presence of active substance.
In the IND-free films, all of the examined liquid additives significantly lowered the tensile strength of the PDMS films. The most pronounced decrease was observed in the case of SO, in spite of the fact that no additional structural contribution of the additive was noted, as was observed for PG and PEG. A similar mechanism of interference in polymeric films that could confirm this assumption was found in literature [
23,
24]. The Young modulus parameter of PDMS film significantly increased in the presence of PEG (i.e., the film was stiffer). GP caused an opposite effect and more flexible material was obtained, comparable to SO films. Moreover, considering the opposite effects in the presence of PEG or GP and higher density of the PEG films, it was concluded that the unique shape of the microstructures within the PEG film is the main cause of the observed changes.
Further changes in the film performance were noticed when the active substance was present. In general, simultaneous addition of IND and one of the excipients enhanced the strength of the films, but only in case of SO was the increase statistically significant, yet still not as pronounced as in the additive-free film. It should be noted that a simultaneous decrease of % EB value in the presence of GP indicates that the material is more fragile, due to liquid droplets present in the matrix. This might be due to IND occurring in a dissolved form in the excipients phase, where fewer particles can strengthen the material and alter the shear transfer, hence acting as a filler reinforcing the material (DF approx. 20%,
Table 1). Thus, the basic mechanical analysis confirmed the pronounced structure differences within the films observed by imaging analysis.
3.5. Water Transport—Permeability, Swelling and Erosion of PDMS Films
A detailed characterization of the structures in PDMS films was made with SEM and AFM, while the contribution of the pore microstructure to the permeability of the films was assessed based on the results of the tritiated water permeation through the films and their ability to swell and erode.
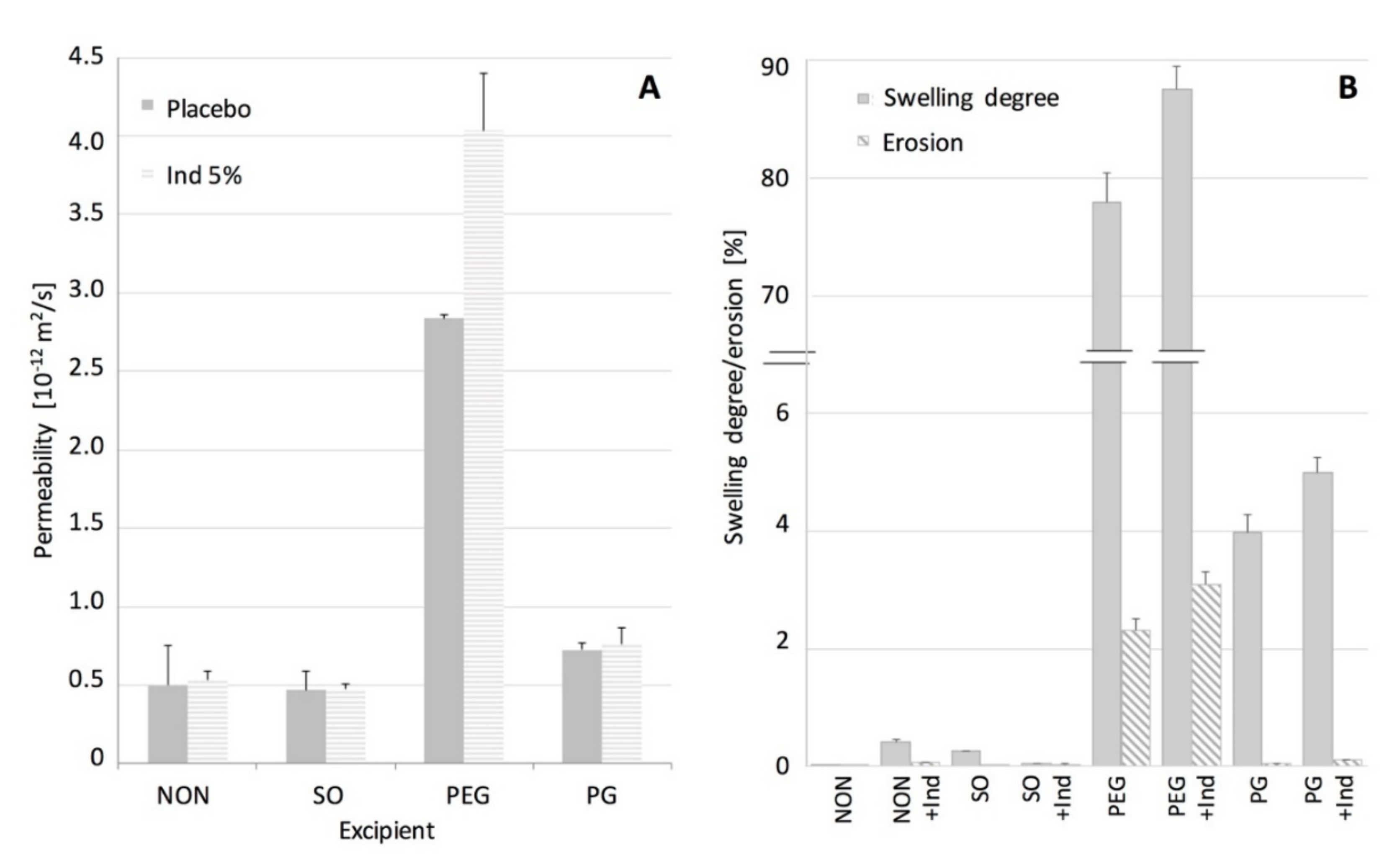
In
Figure 4. permeability of the examined films to water is shown, expressed as a steady state flux. The swelling and erosion data are also shown. When SO or IND were present in the films no changes in their permeability was noted, in contrast to PEG and GP films. It can be stated that in this case the water permeation will depend only on diffusivity of a hydrophobic PDMS material. Many authors stated that solid particles suspended in the matrix can increase the diffusivity of the polymeric materials [
18,
25], but in our case the phenomenon seemed unpronounced, probably due to relatively low IND particles load (5%
w/
w). This assumption was supported by similar effect described in the literature for a silicone elastomer [
22]. Lack of swelling and erosion (below 0.5%) for films containing SO and IND indicated that the silicone polymer chains remained in the initial state, as noted for the pure PDMS. Thus it can be concluded that the water uptake and potential additive leakage do not interfere with the water flux.
The effect of the investigated hydrophilic excipients on the water transport through the films differed considerably (
Figure 4). Although, in both cases, a statistically significant increase in the permeability to water was noted, PEG-containing films showed the more-pronounced improvement in water transport, with simultaneous substantial ability to swell (up to 90%). It was observed by Choi et al. [
26] that water penetration into a hydrophobic material can be controlled by material porosity design. Regarding the microstructure of PEG films, it can be assumed that not only the size, but more the channel-like shape of the pores contributes to the water transport as additional paths, easily accessible for water, are created in the silicone structure. Since even higher permeability and swelling degree was noted in the presence of both PEG and IND, the solid particles in this case are likely to create additional regions or branching points in the pore structure, easily accessible for larger quantities of the medium to penetrate. Moreover, approximately 2.5% erosion of the film, further increased with simultaneous presence of IND, suggests the PEG leakage from the films, and seems to support observations on the branching of the PEG domains, where water has an access to deeper layers of the film and to the IND particles. Besides, it can be presumed that PEG is partially trapped in the pores it created, therefore not all of the regions are equally accessible to water, which explains a relatively low erosion of these films (lower than initially expected). Presumably, due to high molecular weight of PEG and differences in chemical structure of the surrounding silicone, PEG chains can be partially immobilized by PDMS surface free energy, as suggested by other authors [
19].
Even though PG-containing films showed a porous inner structure, its effect on the permeability and material ability to swell, was much less manifested than in the case of PEG. Considering the low erosion of PG films, even those with IND, one can assume that water access to the inner part of the film seems to be limited by more isolated PG regions in the inner structure of the PG–PDMS films.
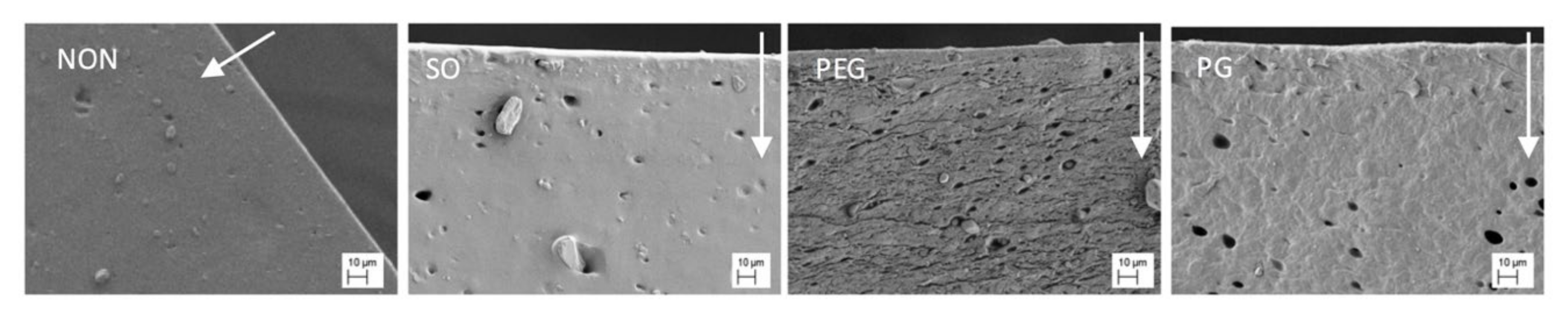
To provide additional data, SEM study of the films exposed to water was conducted and the images are presented in
Figure 5. The films soaked in water were freeze-dried before SEM examination of the cross-section. The pore structure in these films was compared with the corresponding untreated samples (
Figure 2). For pure PDMS films and the films with SO no changes in the morphology were noted. After exposure to water the inner structure of PG-containing films revealed a lower concentration of PG droplets near the surface, undoubtedly due to being superficially washed out. The low water penetration into this film explains its minimal erosion and the swelling occurring only on the surface. Since the water uptake in PEG-loaded films was the highest one noted, in consequence the most pronounced changes in the microstructure were observed. In this case, the matrix seemed to shrink and vertical layers can be distinguished across the film, indicating where the film swelled and then eroded most significantly, with a clearly noticeable decrease of the characteristic channel structure.
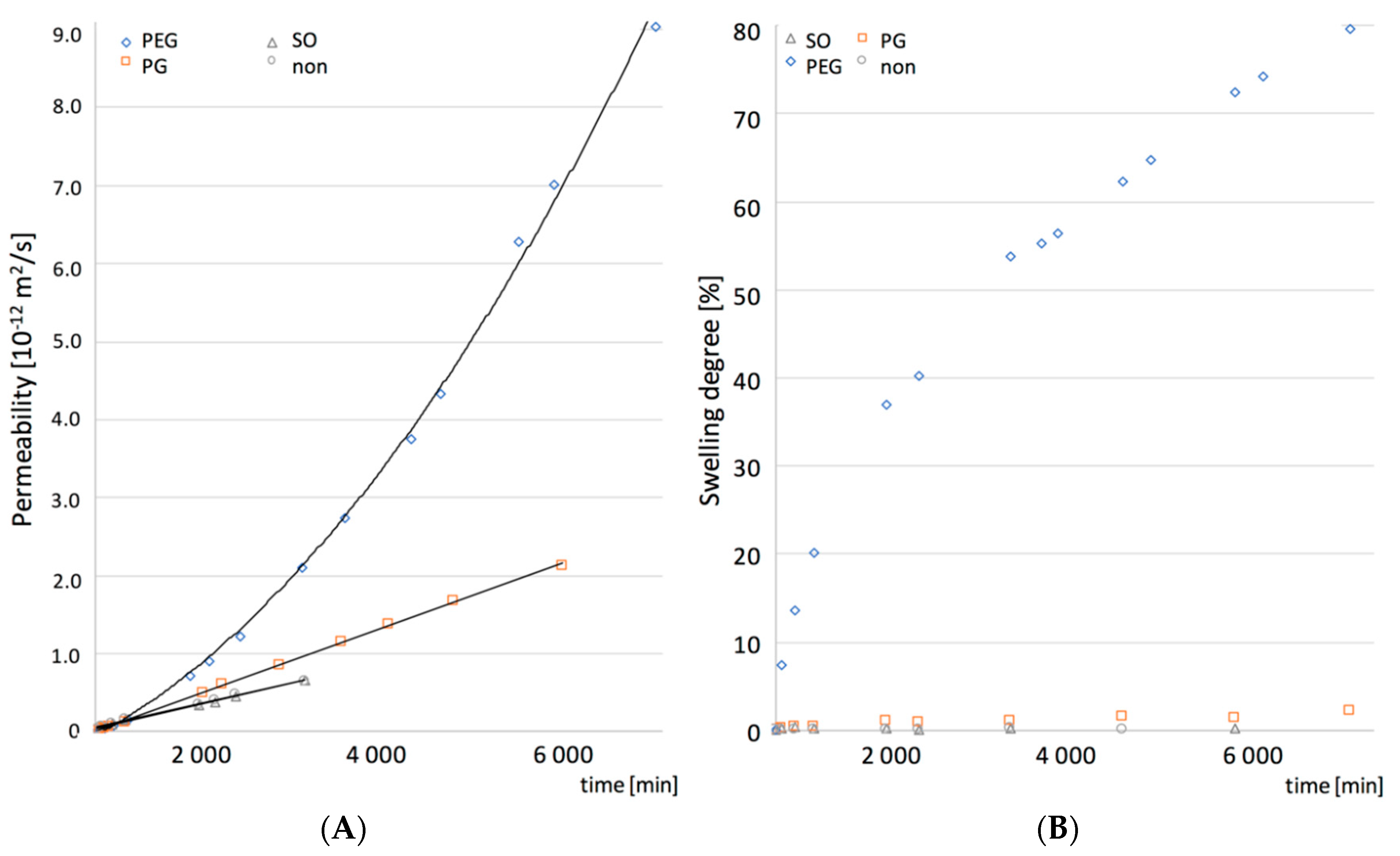
To further explain the observed structural phenomena and assess whether the water transport kinetics correlate with the swelling, more detailed analysis of the water permeability and the swelling kinetics was conducted and the results are presented in
Figure 6. The additive-free PDMS, SO and PG films showed linearity in both the water permeation and the swelling slopes, resulting in a steady state flux observed during the course of experiment. In the case of water permeation through PEG-containing PDMS, two regions of the curve with the steady-state flux could be distinguished, which suggested a more complex mechanism of the water uptake. Moreover, a dual stage swelling rate was obtained, which seemed consistent with the film permeability.
The observed correlation between the water permeability and the swelling kinetics of the PDMS films with additives was further analyzed for its significance with the Pearson’s correlation coefficients factor (r
p,
Table 3). In the presence of hydrophilic excipients (PEG and PG) a strong positive correlation was found (r
p > 0.9,
p < 0.05). However, for PDMS and SO-PDMS films, a poor correlation was found (r
p < 0.21,
p < 0.05). These observations are considered as a support of the hypothesis that the increased permeability of PDMS films is attributed to the unique pore shape created by PEG or PG within the PDMS film.
3.6. IND Release Rate
The rate at which the active substance is released from a drug delivery matrix system can be controlled by a number of phenomena, among which most commonly mentioned in the literature are: water penetration into the system, polymer swelling and dissolution, drug diffusion through polymeric networks and the process of drug dissolution [
27,
28], all considered in this research so far. As proposed by Gehrke et al. [
19], the importance of drug solubility in the matrix for reaching the optimal release kinetics should be taken into account. Thus, the most reasonable approach to optimize the release rate of an active substance in a transdermal patch is to increase the amount of the drug dissolved in the dosage form, which was achieved in this study by the addition of the liquid additives, PG and PEG. To evaluate this approach, the release of a model drug from PDMS films was examined. In vitro dissolution testing of the films containing 5%
w/
w of IND was performed in sink conditions, with an excess of the substance suspended in the formulation to sustain an infinite dose, both essential to obtain the insight into the mechanism of the release process [
9]. The results expressed as a cumulative amount of IND released (µg/cm
2) in a function of squared root of time are given in
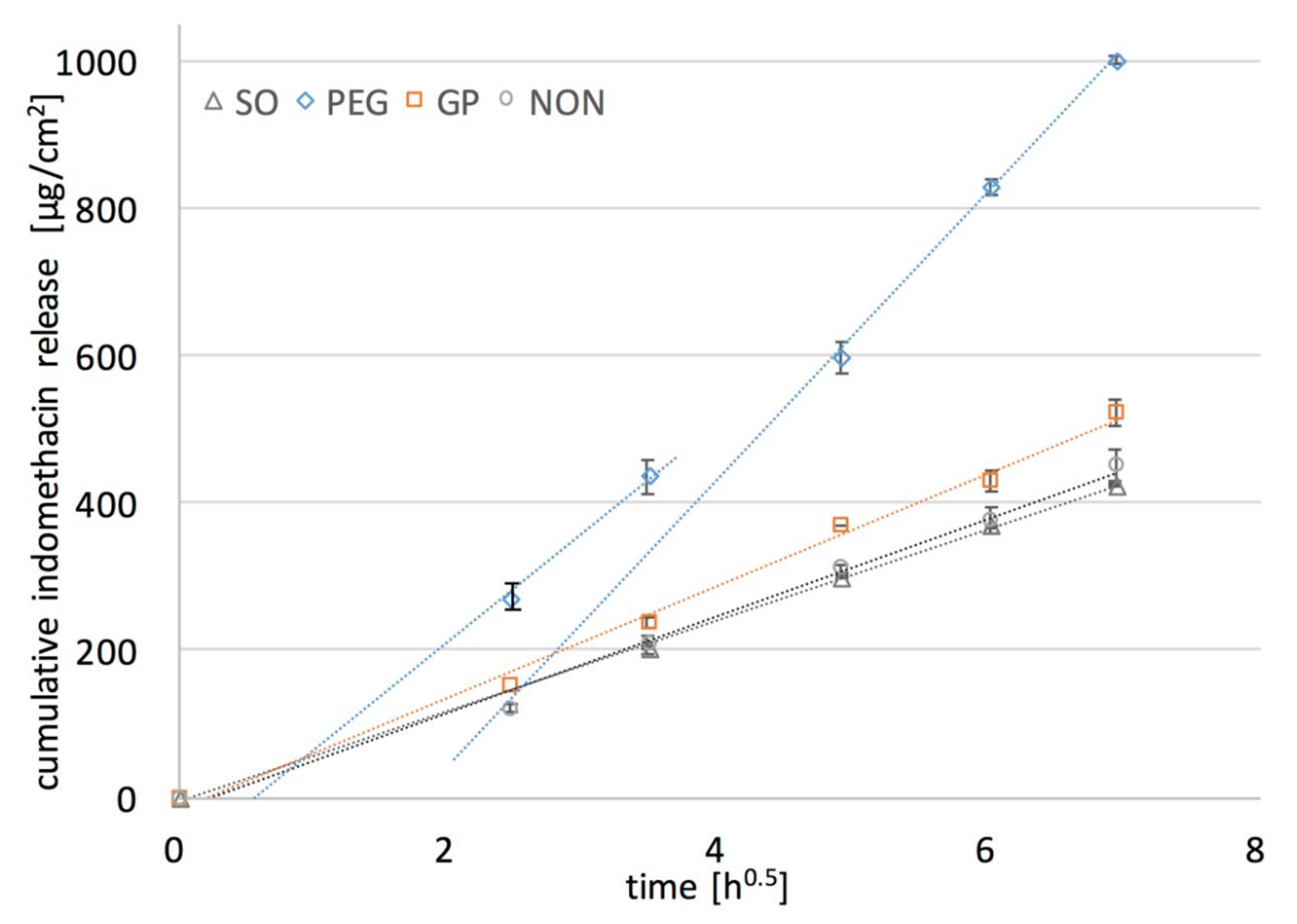
Figure 7.
In all of the examined formulations within the time of experiment (48 h) only a small fraction of the initial drug loading was released (up to 12% of IND). Comparable release rates were observed for PDMS films with SO and with no additives. The increase in the release rate of IND was noted only if PEG or PG was added to the film, but much higher values for PEG than for PG were found. This might be attributed to the more pronounced erosion and PEG leakage from the films upon exposure to the medium. It was very important that a considerable amount of IND was dissolved in PEG. Even though DF factor is much higher for PG than for PEG films (
Table 3), the dissolved IND is located in the PG droplets distributed in the inner part of the film, and inaccessible to the dissolution medium. Therefore, the relatively low increase in the dissolution rate is caused only by the fraction of IND located near the surface of the film, since water penetration into the film deeper layer is rather limited. This presumption is supported by the observations with SEM (
Figure 5).
Since the analyzed films were considered as a monolithic solid material with slab geometry (planar system), and IND solid particles uniformly dispersed in the PDMS, Higuchi kinetics behavior was expected [
29]. In the first step of the analysis, the experimental data were fitted to the Higuchi model. Values of the dissolution constant at steady state flux (
ks) and the determination coefficient (
R2) indicating the agreement with the model are presented in
Table 3, additionally shown graphically as dotted curves in
Figure 7.
Based on the calculation, the release curve was linear for the reference PDMS film and the films containing PG or SO (R2 > 0.99) and for these formulations the drug release as a diffusion process based on the Fick’s first law was suggested. Considering the previous findings on water penetration kinetics in the films it can be concluded that the basic drug transport is controlled by the diffusivity of the PDMS polymeric structure. Nonetheless, it should be pointed out, that the presence of PG slightly improved the film performance (higher ks value), which is probably due to the superficially located drug dissolved in PG domains where additional dose of IND is present, and causes the increase of drug release at the beginning of the experiment. Regarding the observed rather minor increase in the in-vitro release rate from PG-containing films, together with the low erosion of the films occurring only on the surface (SEM), and small penetration of water, it can be stated that in the case of PG–PDMS films the drug diffusion is dominated mostly by diffusivity of the PDMS matrix.
In contrast, PEG–PDMS films substantially deviated from the Higuchi model. Interestingly, further calculations revealed that only the second part of the steady state slope (after 24 h of the release) was in good agreement with the Higuchi model (R2 > 0.98), which was also found to be consistent with the steady state water flux through the films observed in the permeation and swelling experiments. Thus, the assumption on the dual mechanism of drug release was stated, where not only solubility of the drug in the films but also the inner microstructure may affect the diffusion substantially. The correlation observed between the swelling and permeability kinetics and the release rate suggests that in the first stage a unique porosity of the matrix caused the increased water penetration into the film. As a result, drug-rich PEG was leached through the channels upon exposure to the dissolution medium. This can further be supported by the significant erosion of the films. The steady state of the release process was reached when the channels were fully filled and the contact surface between the medium and PDMS swollen pore network was constant. On this stage, when the drug flux fitted the Higuchi equation, the release was determined mostly by PDMS diffusivity.