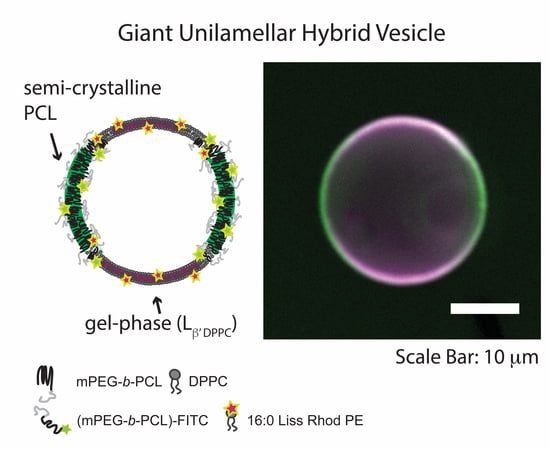
Hybrid Unilamellar Vesicles of Phospholipids and Block Copolymers with Crystalline Domains
Abstract
1. Introduction
2. Materials and Methods
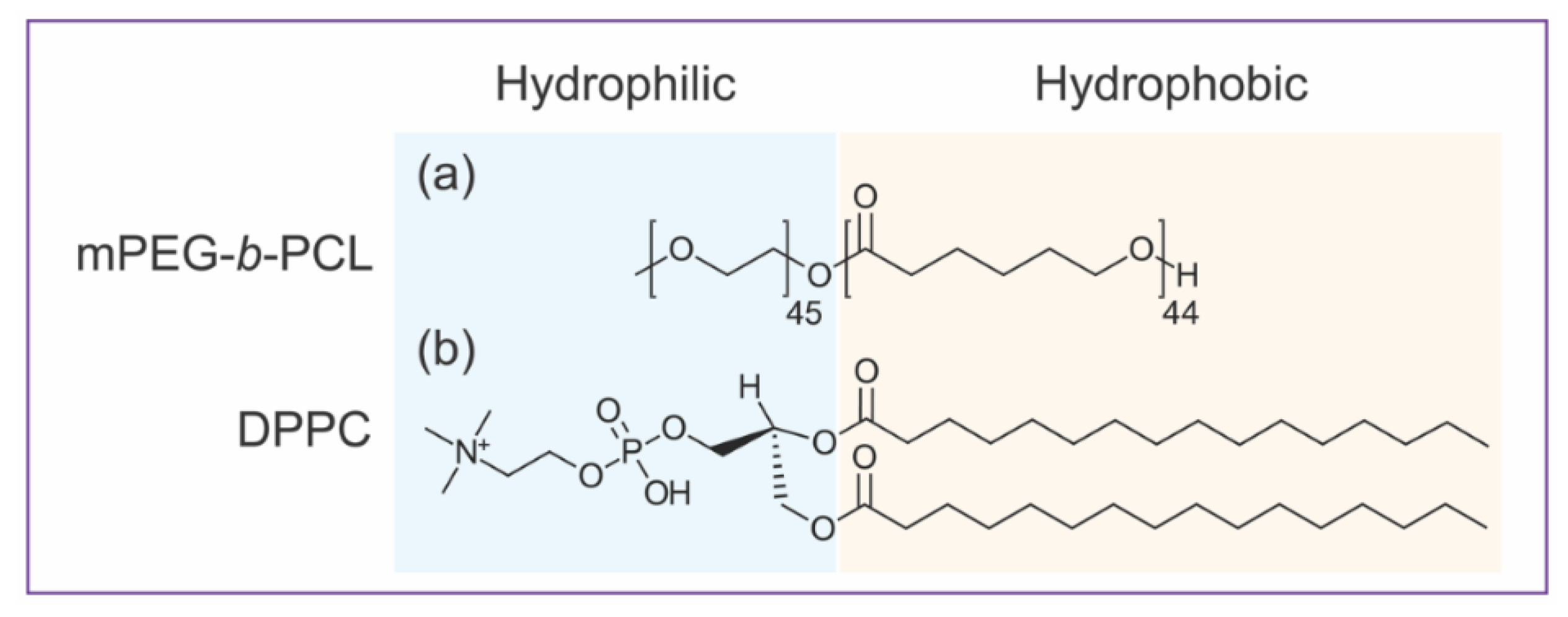
2.1. Materials
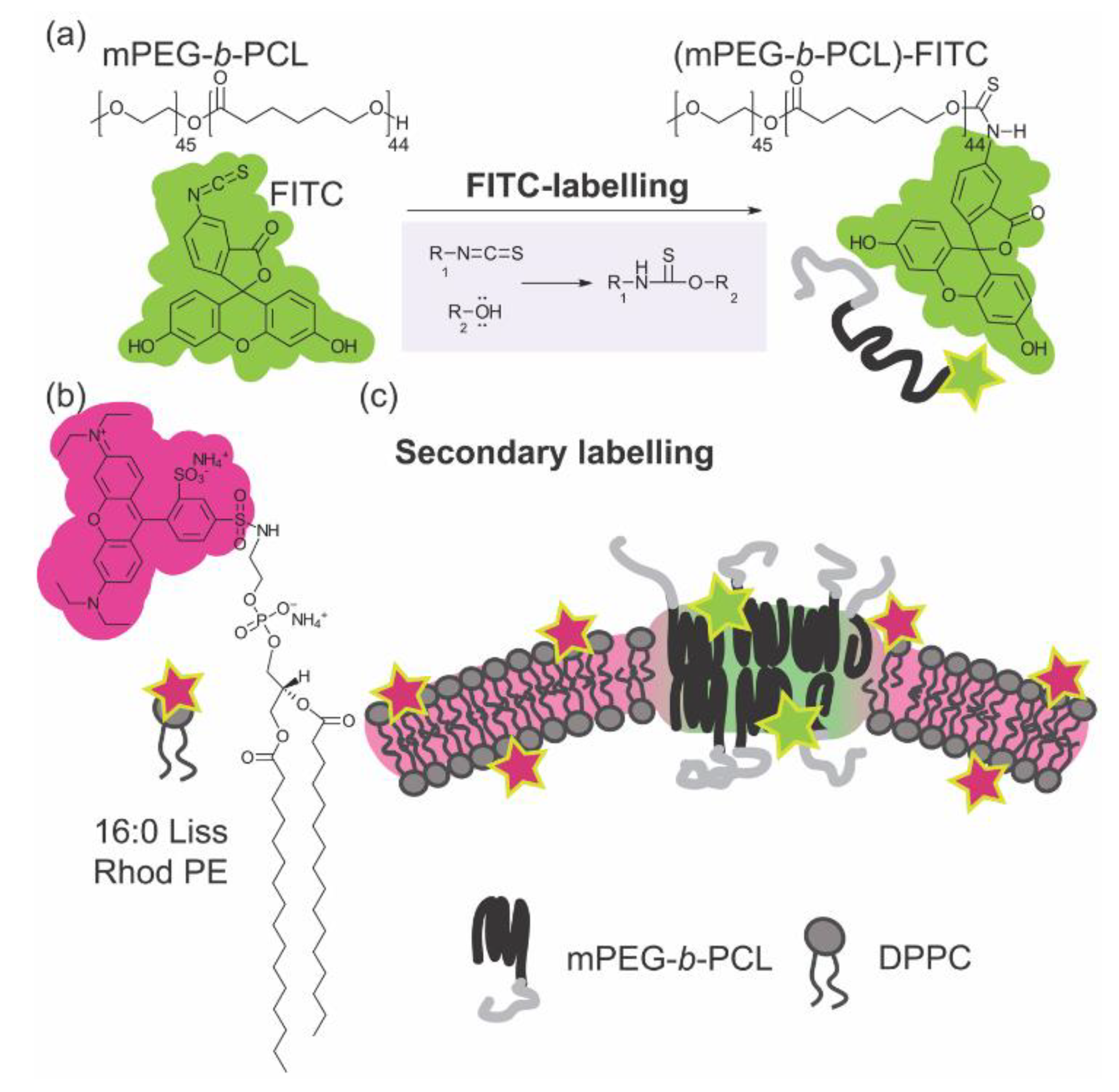
2.2. FITC-labelling of mPEG-b-PCL
2.3. Giant Unilamellar Vesicle (GUV) formation by Electroformation
2.4. Giant Hybrid Unilamellar Vesicle (GHUV) formation by the PAPYRUS Method
2.5. Optical Microscopy Experiments
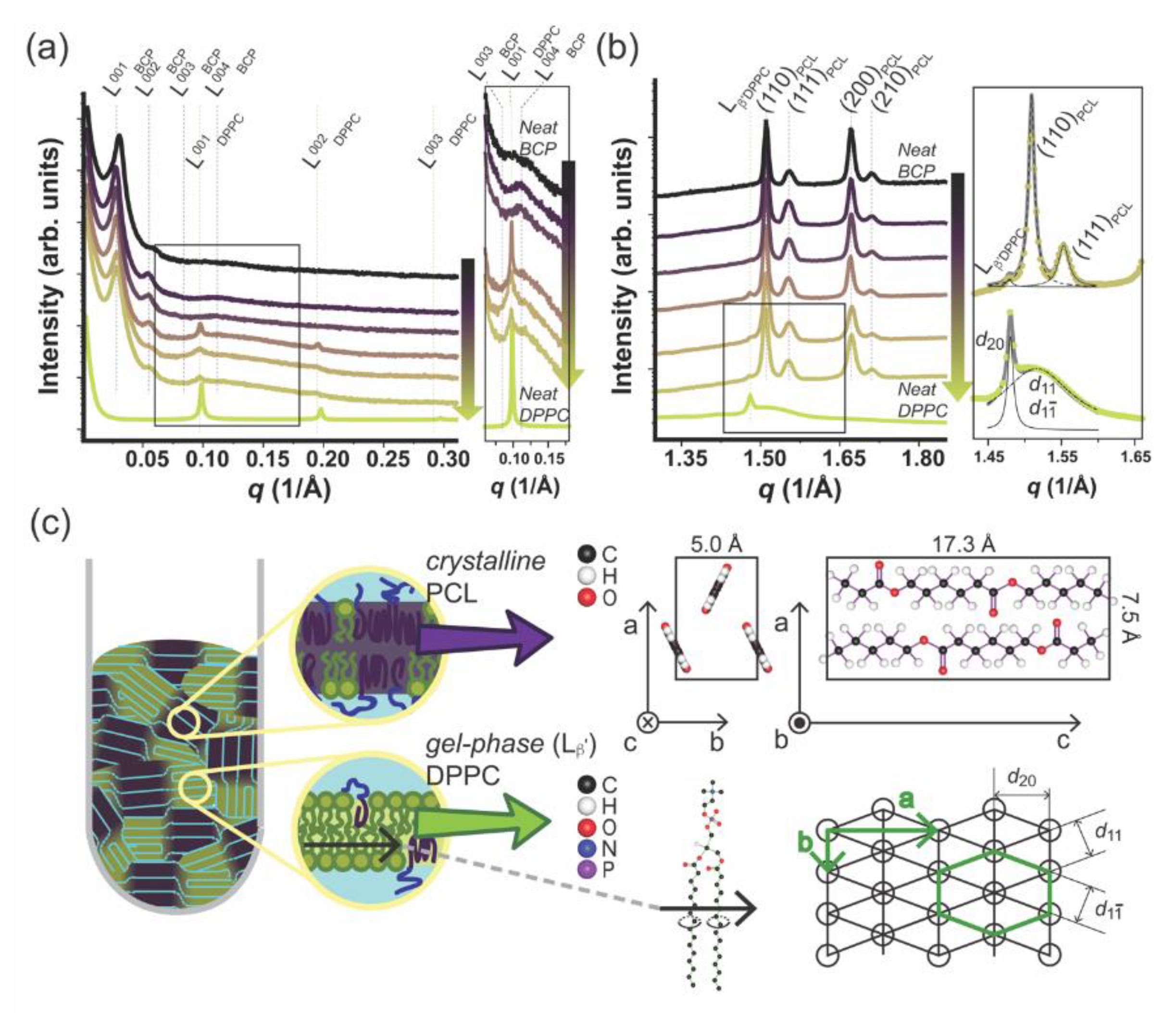
2.6. Small Angle X-ray Scattering (SAXS) and Wide Angle X-ray Scattering (WAXS)
3. Results and Discussion
3.1. Pure mPEG-b-PCL BCPs and DPPC PLs form Giant Unilamellar Vesicles
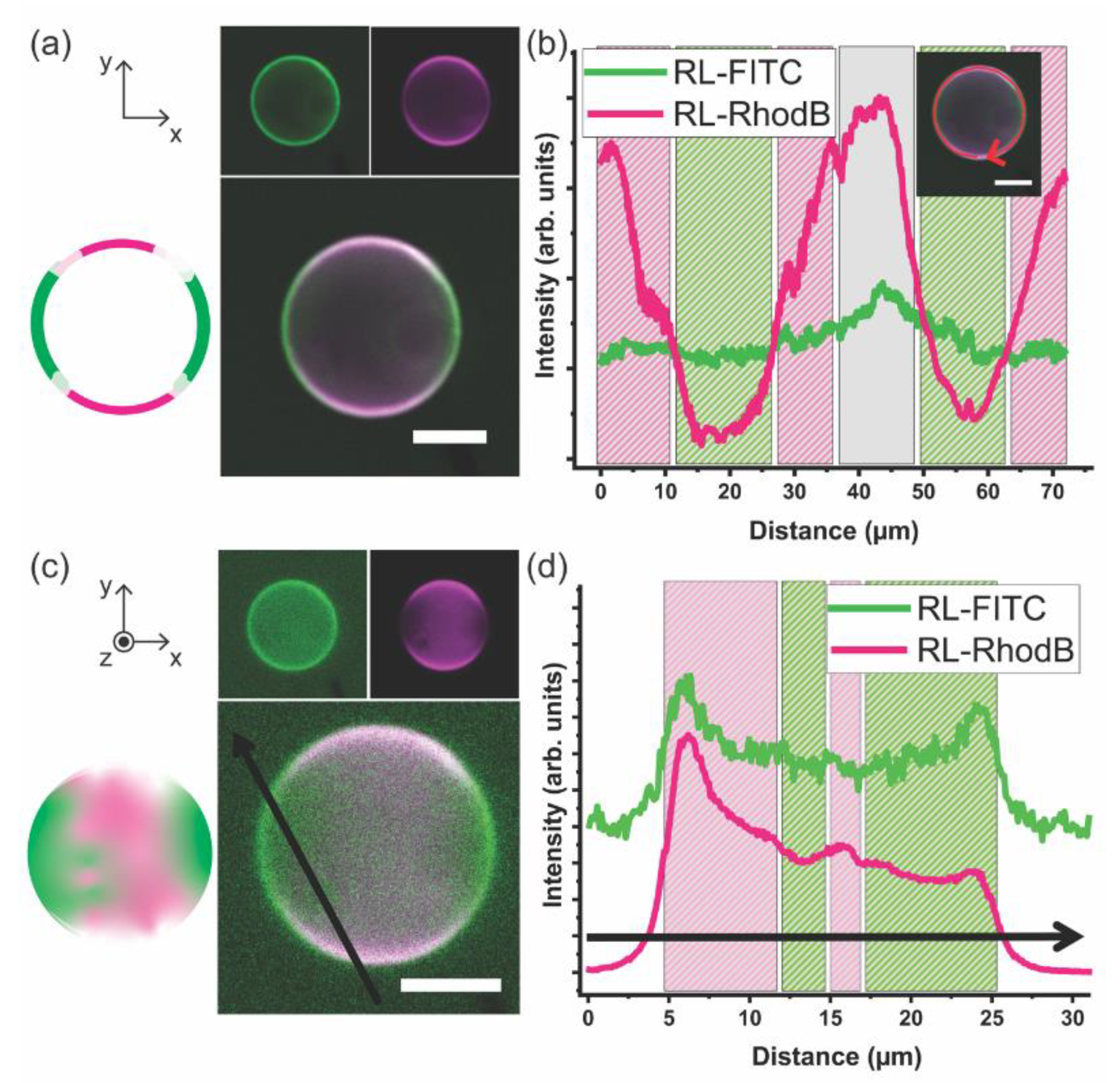
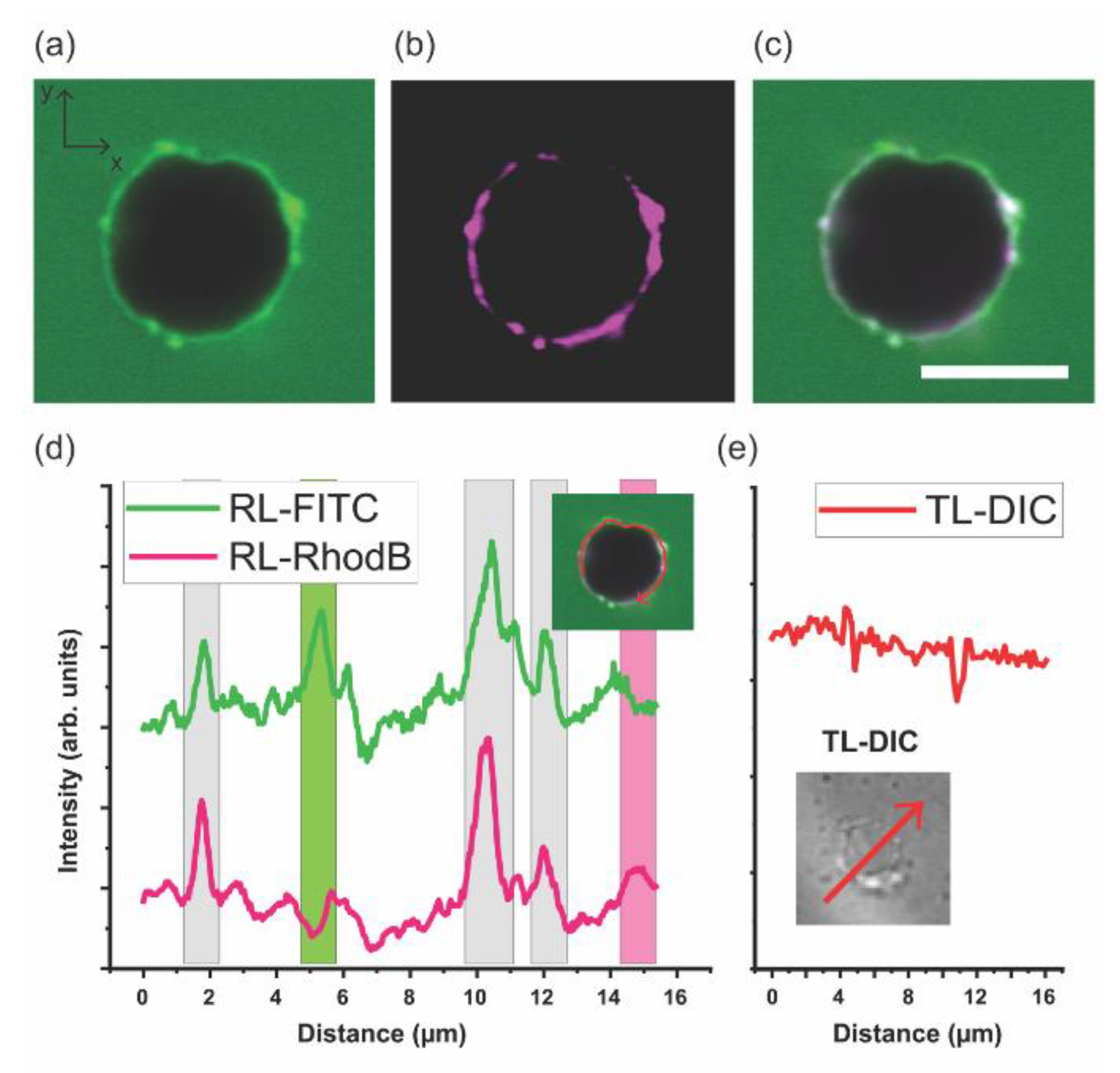
3.2. Giant Hybrid Unilamellar Vesicles (GHUVs) of mPEG-b-PCL and DPPC
3.3. DPPC–mPEG-b-PCL Hybrid Membrane Structure and Molecular Order
3.4. DPPC–mPEG-b-PCL GHUVs in Hypertonic Media
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Israelachvili, J.N.; Mitchell, D.J.; Ninham, B.W. Theory of self-assembly of hydrocarbon amphiphiles into micelles and bilayers. J. Chem. Soc. Faraday Trans. 2 Mol. Chem. Phys. 1976, 72, 1525–1568. [Google Scholar] [CrossRef]
- Bates, C.M.; Bates, F.S. 50th anniversary perspective: Block polymers-pure potential. Macromolecules 2017, 50, 3–22. [Google Scholar] [CrossRef]
- Discher, B.M.; Won, Y.Y.; Ege, D.S.; Lee, J.C.M.; Bates, F.S.; Discher, D.E.; Hammer, D.A. Polymersomes: Tough vesicles made from diblock copolymers. Science 1999, 284, 1143–1146. [Google Scholar] [CrossRef]
- Lee, J.S.; Feijen, J. Polymersomes for drug delivery: Design, formation and characterization. J. Control. Release 2012, 161, 473–483. [Google Scholar]
- Bermúdez, H.; Hammer, D.A.; Discher, D.E. Effect of bilayer thickness on membrane bending rigidity. Langmuir 2004, 20, 540–543. [Google Scholar] [CrossRef]
- Kang, M.; Tuteja, M.; Centrone, A.; Topgaard, D.; Leal, C. Nanostructured lipid-based films for substrate-mediated applications in biotechnology. Adv. Funct. Mater. 2018, 28, 1704356. [Google Scholar] [CrossRef]
- Le Meins, J.F.; Schatz, C.; Lecommandoux, S.; Sandre, O. Hybrid polymer/lipid vesicles: State of the art and future perspectives. Mater. Today 2013, 16, 397–402. [Google Scholar] [CrossRef]
- Schulz, M.; Binder, W.H. Mixed hybrid lipid/polymer vesicles as a novel membrane platform. Macromol. Rapid Commun. 2015, 36, 2031–2041. [Google Scholar]
- Chemin, M.; Brun, P.-M.; Lecommandoux, S.; Sandre, O.; Le Meins, J.-F. Hybrid polymer/lipid vesicles: Fine control of the lipid and polymer distribution in the binary membrane. Soft Matter 2012, 8, 2867–2874. [Google Scholar]
- Lim, S.K.; De Hoog, H.P.; Parikh, A.N.; Nallani, M.; Liedberg, B. Hybrid, nanoscale phospholipid/block copolymer vesicles. Polymers (Basel) 2013, 5, 1102–1114. [Google Scholar] [CrossRef]
- Chen, D.; Santore, M.M. Hybrid copolymer-phospholipid vesicles: Phase separation resembling mixed phospholipid lamellae, but with mechanical stability and control. Soft Matter 2015, 11, 2617–2626. [Google Scholar] [CrossRef]
- Kang, M.; Lee, B.; Leal, C. Three-dimensional microphase separation and synergistic permeability in stacked lipid-polymer hybrid membranes. Chem. Mater. 2017, 29, 9120–9132. [Google Scholar]
- Magnani, C.; Montis, C.; Mangiapia, G.; Mingotaud, A.F.; Mingotaud, C.; Roux, C.; Joseph, P.; Berti, D.; Lonetti, B. Hybrid vesicles from lipids and block copolymers: Phase behavior from the micro- to the nano-scale. Colloids Surfaces B Biointerfaces 2018, 168, 18–28. [Google Scholar]
- Gettel, D.L.; Sanborn, J.; Patel, M.A.; De Hoog, H.P.; Liedberg, B.; Nallani, M.; Parikh, A.N. Mixing, diffusion, and percolation in binary supported membranes containing mixtures of lipids and amphiphilic block copolymers. J. Am. Chem. Soc. 2014, 136, 10186–10189. [Google Scholar] [CrossRef]
- Schulz, M.; Olubummo, A.; Bacia, K.; Binder, W.H. Lateral surface engineering of hybrid lipid-BCP vesicles and selective nanoparticle embedding. Soft Matter 2014, 10, 831–839. [Google Scholar] [CrossRef]
- Dao, T.P.T.; Fernandes, F.; Ibarboure, E.; Ferji, K.; Prieto, M.; Sandre, O.; Le Meins, J.F. Modulation of phase separation at the micron scale and nanoscale in giant polymer/lipid hybrid unilamellar vesicles (GHUVs). Soft Matter 2017, 13, 627–637. [Google Scholar] [CrossRef]
- Rideau, E.; Dimova, R.; Schwille, P.; Wurm, F.R.; Landfester, K. Liposomes and polymersomes: A comparative review towards cell mimicking. Chem. Soc. Rev. 2018, 47, 8572–8610. [Google Scholar] [CrossRef]
- Wang, W.; Qi, H.; Zhou, T.; Mei, S.; Han, L.; Higuchi, T.; Jinnai, H.; Li, C.Y. Highly robust crystalsome via directed polymer crystallization at curved liquid/liquid interface. Nat. Commun. 2016, 7, 3–8. [Google Scholar] [CrossRef]
- Qi, H.; Zhou, H.; Tang, Q.; Lee, J.Y.; Fan, Z.; Kim, S.; Staub, M.C.; Zhou, T.; Mei, S.; Han, L.; et al. Block copolymer crystalsomes with an ultrathin shell to extend blood circulation time. Nat. Commun. 2018, 9, 3005. [Google Scholar] [CrossRef]
- Ahmed, F.; Discher, D.E. Self-porating polymersomes of PEG-PLA and PEG-PCL: Hydrolysis-triggered controlled release vesicles. J. Control. Release 2004, 96, 37–53. [Google Scholar] [CrossRef]
- Ghoroghchian, P.P.; Li, G.; Levine, D.H.; Davis, K.P.; Bates, F.S.; Hammer, D.A.; Therien, M.J. Bioresorbable vesicles formed through spontaneous self-assembly of amphiphilic poly(ethylene oxide)-block-polycaprolactone. Macromolecules 2006, 39, 1673–1675. [Google Scholar] [CrossRef]
- Rajagopal, K.; Mahmud, A.; Christian, D.A.; Pajerowski, J.D.; Brown, A.E.X.; Loverde, S.M.; Discher, D.E. Curvature-coupled hydration of semicrystalline polymer amphiphiles yields flexible worm micelles but favors rigid vesicles: Polycaprolactone-based block copolymers. Macromolecules 2010, 43, 9736–9746. [Google Scholar] [CrossRef]
- Qi, W.; Ghoroghchian, P.P.; Li, G.; Hammer, D.A.; Therien, M.J. Aqueous self-assembly of poly(ethylene oxide)-block-poly(ε-caprolactone) (PEO-b-PCL) copolymers: Disparate diblock copolymer compositions give rise to nano- and meso-scale bilayered vesicles. Nanoscale 2013, 5, 10908–10915. [Google Scholar] [CrossRef]
- Pippa, N.; Kaditi, E.; Pispas, S.; Demetzos, C. PEO-b-PCL-DPPC chimeric nanocarriers: Self-assembly aspects in aqueous and biological media and drug incorporation. Soft Matter 2013, 9, 4073–4082. [Google Scholar] [CrossRef]
- Pippa, N.; Deli, E.; Mentzali, E.; Pispas, S.; Demetzos, C. PEO-b-PCL grafted DPPC liposomes: Physicochemical characterization and stability studies of novel bio-inspired advanced drug delivery nano systems (aDDnSs). J. Nanosci. Nanotechnol. 2014, 14, 5676–5681. [Google Scholar]
- Kang, J.Y.; Choi, I.; Seo, M.; Lee, J.Y.; Hong, S.; Gong, G.; Shin, S.S.; Lee, Y.; Kim, J.W. Enhancing membrane modulus of giant unilamellar lipid vesicles by lateral co-assembly of amphiphilic triblock copolymers. J. Colloid Interface Sci. 2020, 561, 318–326. [Google Scholar] [CrossRef]
- Zhu, Z.; Li, Y.; Li, X.; Li, R.; Jia, Z.; Liu, B.; Guo, W.; Wu, W.; Jiang, X. Paclitaxel-loaded poly(N-vinylpyrrolidone)-b-poly(ε-caprolactone) nanoparticles: Preparation and antitumor activity in vivo. J. Control. Release 2010, 142, 438–446. [Google Scholar] [CrossRef]
- Stein, H.; Spindler, S.; Bonakdar, N.; Wang, C.; Sandoghdar, V. Production of isolated giant unilamellar vesicles under high salt concentrations. Front. Physiol. 2017, 8, 63. [Google Scholar]
- Kresse, K.M.; Xu, M.; Pazzi, J.; García-Ojeda, M.; Subramaniam, A.B. Novel application of cellulose paper as a platform for the macromolecular self-assembly of biomimetic giant liposomes. ACS Appl. Mater. Interfaces 2016, 8, 32102–32107. [Google Scholar] [CrossRef]
- Steer, D.; Leung, S.S.W.; Meiselman, H.; Topgaard, D.; Leal, C. Structure of lung-mimetic multilamellar bodies with lipid compositions relevant in pneumonia. Langmuir 2018, 34, 7561–7574. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Dao, T.T.; Fernandes, F.; Er-Rafik, M.; Salva, R.; Schmutz, M.; Brûlet, A.; Prieto, M.; Sandre, O.; Le Meins, J.F. Phase separation and nanodomain formation in hybrid polymer/lipid vesicles. ACS Macro Lett. 2015, 4, 182–186. [Google Scholar] [CrossRef]
- Nagarajan, R. Molecular packing parameter and surfactant self-assembly: The neglected role of the surfactant tail. Langmuir 2002, 18, 31–38. [Google Scholar] [CrossRef]
- Wnȩtrzak, A.; Ła̧tka, K.; Dynarowicz-ła̧tka, P. Interactions of alkylphosphocholines with model membranes—The langmuir monolayer study. J. Membr. Biol. 2013, 246, 453–466. [Google Scholar]
- Discher, D.E.; Eisenberg, A. Polymer vesicles. Science 2002, 297, 967–973. [Google Scholar]
- Bates, F.S.; Fredrickson, G.H. Block copolymers-designer soft materials. Phys. Today 1999, 52, 32–38. [Google Scholar]
- Tierney, K.J.; Block, D.E.; Longo, M.L. Elasticity and phase behavior of DPPC membrane modulated by cholesterol, ergosterol, and ethanol. Biophys. J. 2005, 89, 2481–2493. [Google Scholar] [CrossRef]
- Tanasescu, R.; Mettal, U.; Colom, A.; Roux, A.; Zumbuehl, A. Facile and rapid formation of giant vesicles from glass beads. Polymers (Basel) 2018, 10, 54. [Google Scholar] [CrossRef]
- Sercombe, L.; Veerati, T.; Moheimani, F.; Wu, S.Y.; Sood, A.K.; Hua, S. Advances and challenges of liposome assisted drug delivery. Front. Pharmacol. 2015, 6, 286. [Google Scholar] [CrossRef]
- Zylberberg, C.; Matosevic, S. Pharmaceutical liposomal drug delivery: A review of new delivery systems and a look at the regulatory landscape. Drug Deliv. 2016, 23, 3319–3329. [Google Scholar]
- Torchilin, V.P. Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug Discov. 2005, 4, 145–160. [Google Scholar] [CrossRef]
- Liu, G.Y.; Chen, C.J.; Ji, J. Biocompatible and biodegradable polymersomes as delivery vehicles in biomedical applications. Soft Matter 2012, 8, 8811–8821. [Google Scholar]
- Park, J.W. Sulfatide incorporation effect on mechanical properties of vesicles. Colloids Surfaces B Biointerfaces 2010, 80, 59–62. [Google Scholar]
- Jesorka, A.; Orwar, O. Liposomes: Technologies and analytical applications. Annu. Rev. Anal. Chem. 2008, 1, 801–832. [Google Scholar] [CrossRef]
- Nakagawa, S.; Marubayashi, H.; Nojima, S. Crystallization of polymer chains confined in nanodomains. Eur. Polym. J. 2015, 70, 262–275. [Google Scholar]
- Tuteja, M.; Kang, M.; Leal, C.; Centrone, A. Nanoscale partitioning of paclitaxel in hybrid lipid-polymer membranes. Analyst 2018, 143, 3808–3813. [Google Scholar] [CrossRef]
- Tayebi, L.; Ma, Y.; Vashaee, D.; Chen, G.; Sinha, S.K.; Parikh, A.N. Long-range interlayer alignment of intralayer domains in stacked lipid bilayers. Nat. Mater. 2012, 11, 1074–1080. [Google Scholar] [CrossRef]
- Hu, H.; Dorset, D.L. Crystal structure of poly(ϵ-caprolactone). Macromolecules 1990, 23, 4604–4607. [Google Scholar] [CrossRef]
- Leroux, F.; Montembault, V.; Piogé, S.; Pascual, S.; Brotons, G.; Fontaine, L. High molar mass poly(1,4-butadiene)-graft-poly(π-caprolactone) copolymers by ROMP: Synthesis via the grafting-from route and self-assembling properties. Macromolecules 2016, 49, 4739–4745. [Google Scholar]
- Wiener, M.C.; Suter, R.M.; Nagle, J.F. Structure of the fully hydrated gel phase of dipalmitoylphosphatidylcholine. Biophys. J. 1989, 55, 315–325. [Google Scholar]
- Et-Thakafy, O.; Delorme, N.; Gaillard, C.; Mériadec, C.; Artzner, F.; Lopez, C.; Guyomarch, F. Mechanical properties of membranes composed of gel-phase or fluid-phase phospholipids probed on liposomes by atomic force spectroscopy. Langmuir 2017, 33, 5117–5126. [Google Scholar]
- Tenchov, B.; Koynova, R.; Rapp, G. New ordered metastable phases between the gel and subgel phases in hydrated phospholipids. Biophys. J. 2001, 80, 1873–1890. [Google Scholar] [CrossRef]
- Sun, W.-J.; Suter, R.M.; Knewtson, M.A.; Worthington, C.R.; Tristram-Nagle, S.; Zhang, R.; Nagle, J.F. Order and disorder in fully hydrated unoriented bilayers of gel-phase dipalmitoylphosphatidylcholine. Phys. Rev. E 1994, 49, 4665–4676. [Google Scholar] [CrossRef]
- Pabst, G.; Rappolt, M.; Amenitsch, H.; Laggner, P. Structural information from multilamellar liposomes at full hydration: Full q-range fitting with high quality X-ray data. Phys. Rev. E Stat. Phys. Plasmas Fluids Relat. Interdiscip. Top. 2000, 62, 4000–4009. [Google Scholar] [CrossRef]
- Sun, W.J.; Tristram-Nagle, S.; Suter, R.M.; Nagle, J.F. Structure of gel phase saturated lecithin bilayers: Temperature and chain length dependence. Biophys. J. 1996, 71, 885–891. [Google Scholar] [CrossRef]
- Arias, J.M.; Tuttolomondo, M.E.; Díaz, S.B.; Ben Altabef, A. Reorganization of hydration water of DPPC multilamellar vesicles induced by l-cysteine Interaction. J. Phys. Chem. B 2018, 122, 5193–5204. [Google Scholar]
- Boroske, E.; Elwenspoek, M.; Helfrich, W. Osmotic shrinkage of giant egg-lecithin vesicles. Biophys. J. 1981, 34, 95–109. [Google Scholar] [CrossRef]
- Oglęcka, K.; Rangamani, P.; Liedberg, B.; Kraut, R.S.; Parikh, A.N. Oscillatory phase separation in giant lipid vesicles induced by transmembrane osmotic differentials. Elife 2014, 3, e03695. [Google Scholar] [CrossRef]
- Li, Y.; Lipowsky, R.; Dimova, R. Membrane nanotubes induced by aqueous phase separation and stabilized by spontaneous curvature. Proc. Natl. Acad. Sci. USA 2011, 108, 4731–4736. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Go, Y.K.; Kambar, N.; Leal, C. Hybrid Unilamellar Vesicles of Phospholipids and Block Copolymers with Crystalline Domains. Polymers 2020, 12, 1232. https://doi.org/10.3390/polym12061232
Go YK, Kambar N, Leal C. Hybrid Unilamellar Vesicles of Phospholipids and Block Copolymers with Crystalline Domains. Polymers. 2020; 12(6):1232. https://doi.org/10.3390/polym12061232
Chicago/Turabian StyleGo, Yoo Kyung, Nurila Kambar, and Cecilia Leal. 2020. "Hybrid Unilamellar Vesicles of Phospholipids and Block Copolymers with Crystalline Domains" Polymers 12, no. 6: 1232. https://doi.org/10.3390/polym12061232
APA StyleGo, Y. K., Kambar, N., & Leal, C. (2020). Hybrid Unilamellar Vesicles of Phospholipids and Block Copolymers with Crystalline Domains. Polymers, 12(6), 1232. https://doi.org/10.3390/polym12061232