Contrasting Photo-Switching Rates in Azobenzene Derivatives: How the Nature of the Substituent Plays a Role
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
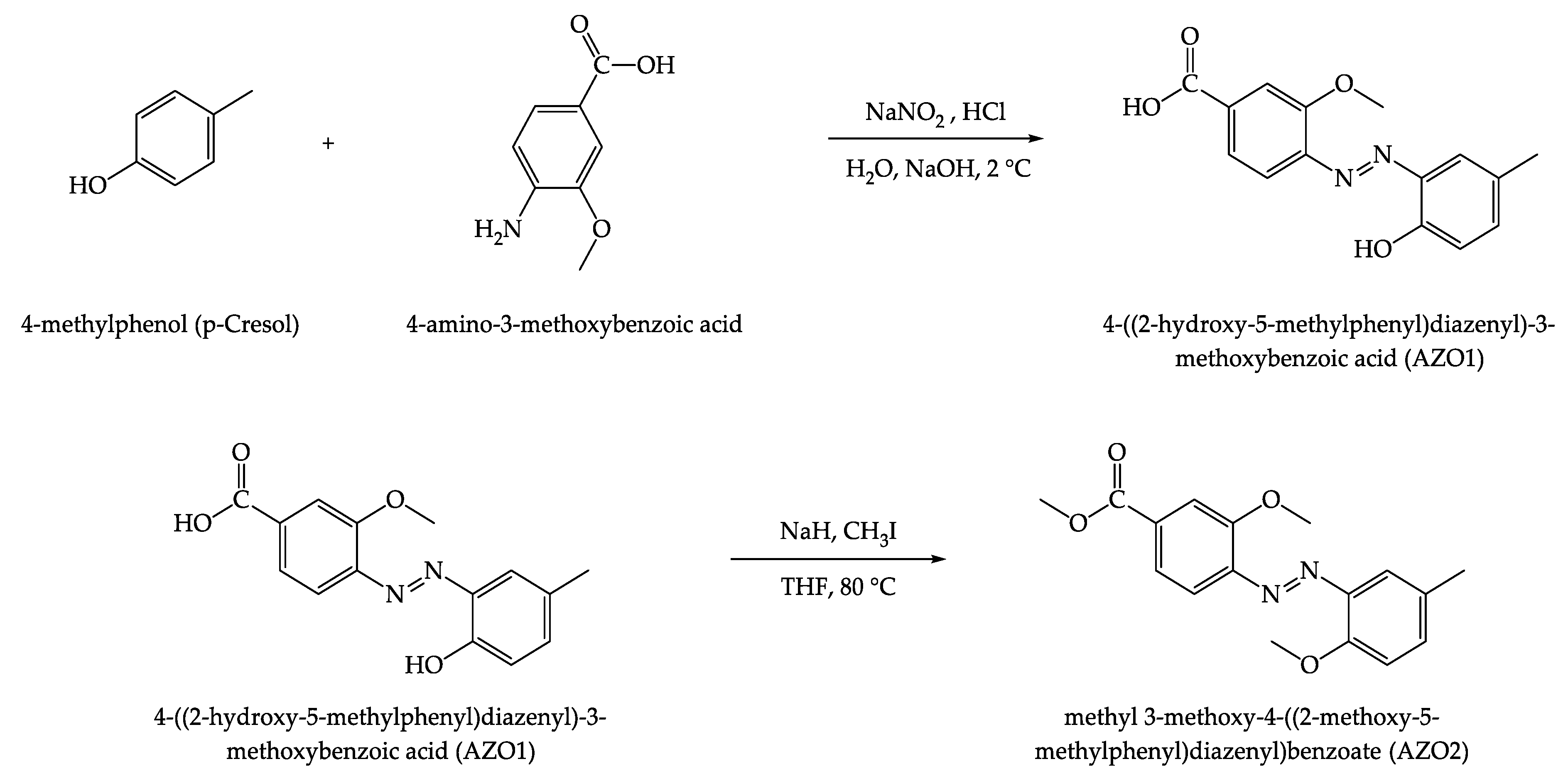
2.3. Synthesis of 4-((2-hydroxy-5-methylphenyl) diazenyl)-3-methoxybenzoic acid (AZO1) and Methyl-3-methoxy-4-((2-methoxy-5-methylphenyl)diazinyl)benzoate (AZO2)
2.4. Computational Methods
3. Results & Discussions
3.1. Molecular Design of the Azo-derivatives:
- Produce a molecule with only one reactive site for further modifications and be used as a side-chain group.
- Move the excitation wavelength of the E-Z isomerization from the UV region of the absorbance spectrum to the visible light one.
- Enhance the stability of the Z form once obtained after irradiation with white light.
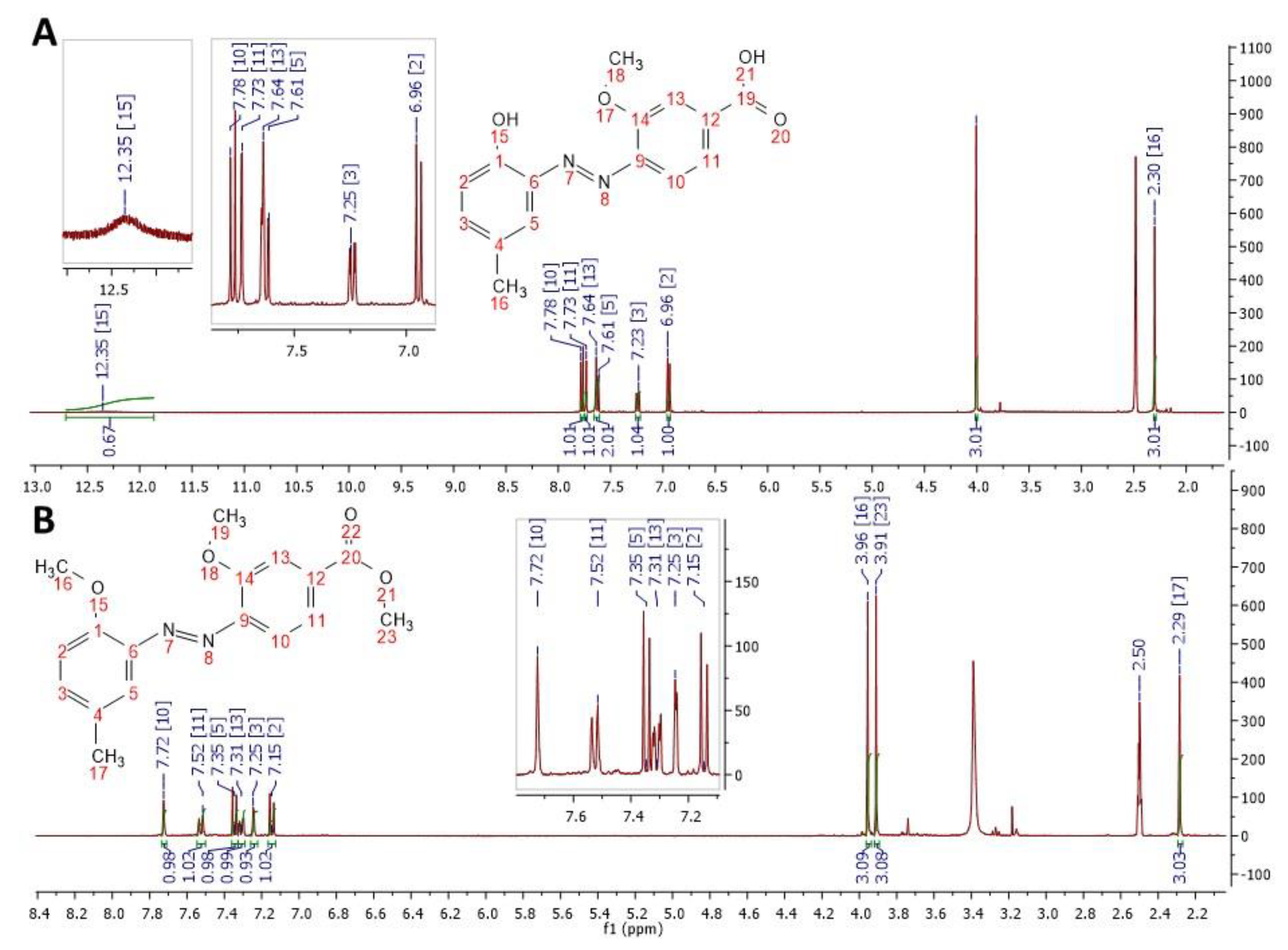
3.2. H–NMR and 13C–NMR Characterizations:
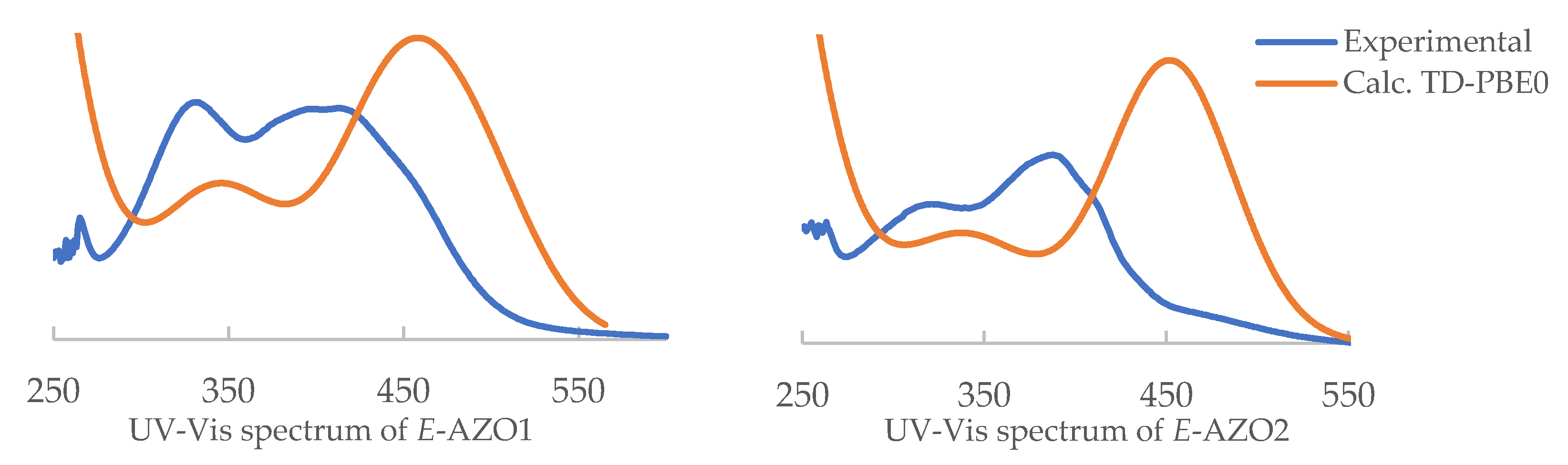
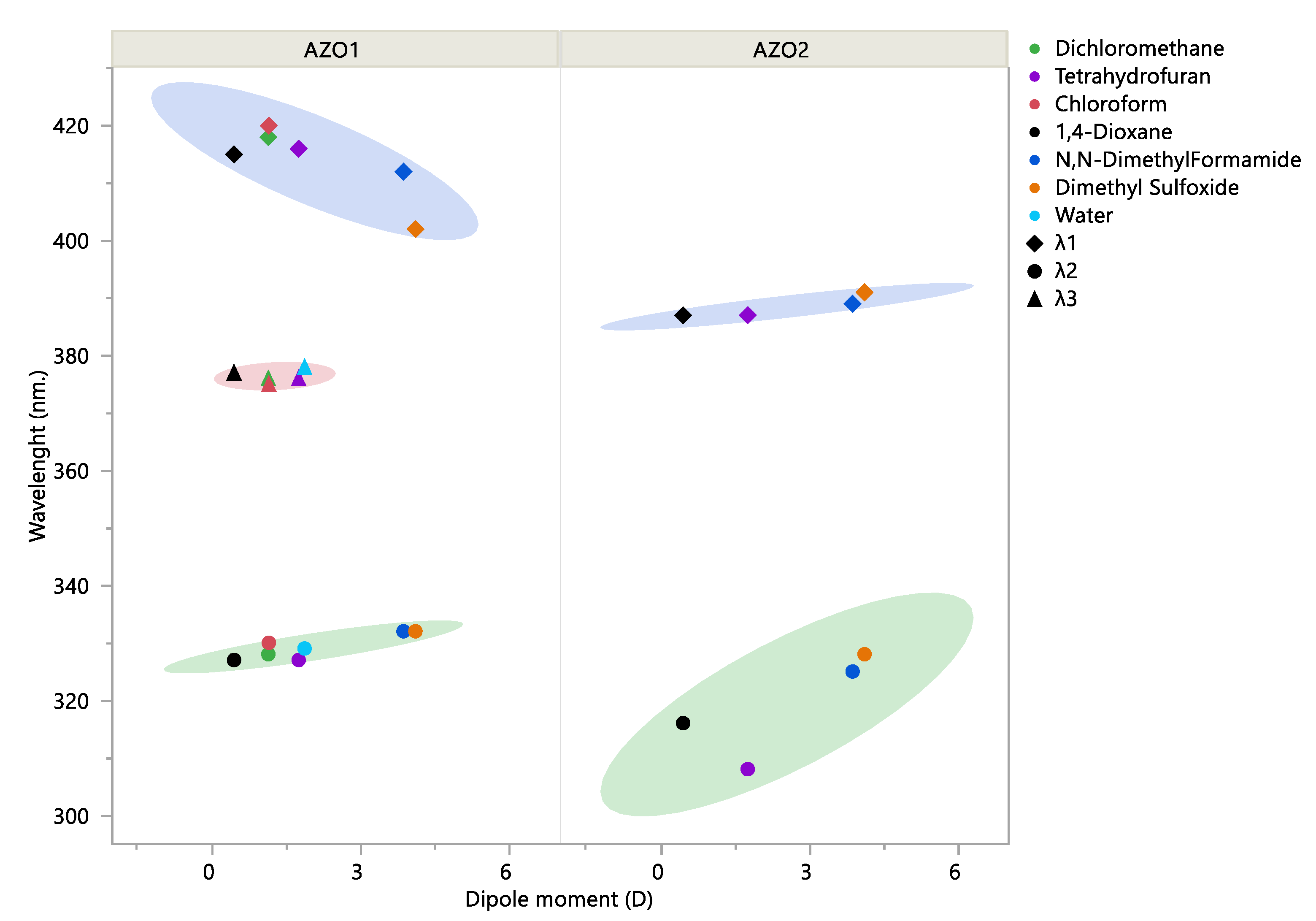
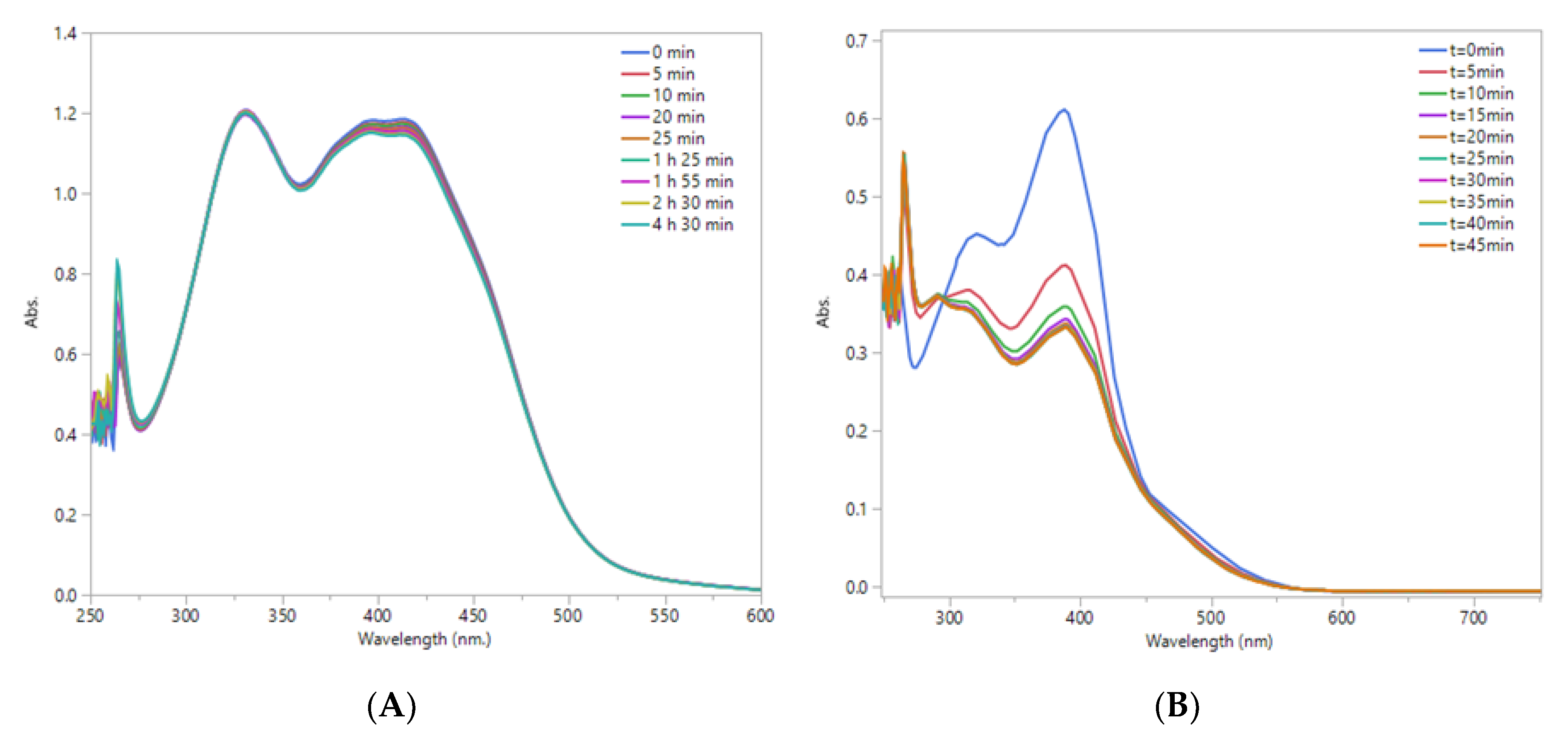
3.3. UV/Vis Spectra Interpretation:
3.4. E–Z Photoisomerization
3.5. Z–E Thermal Relaxation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
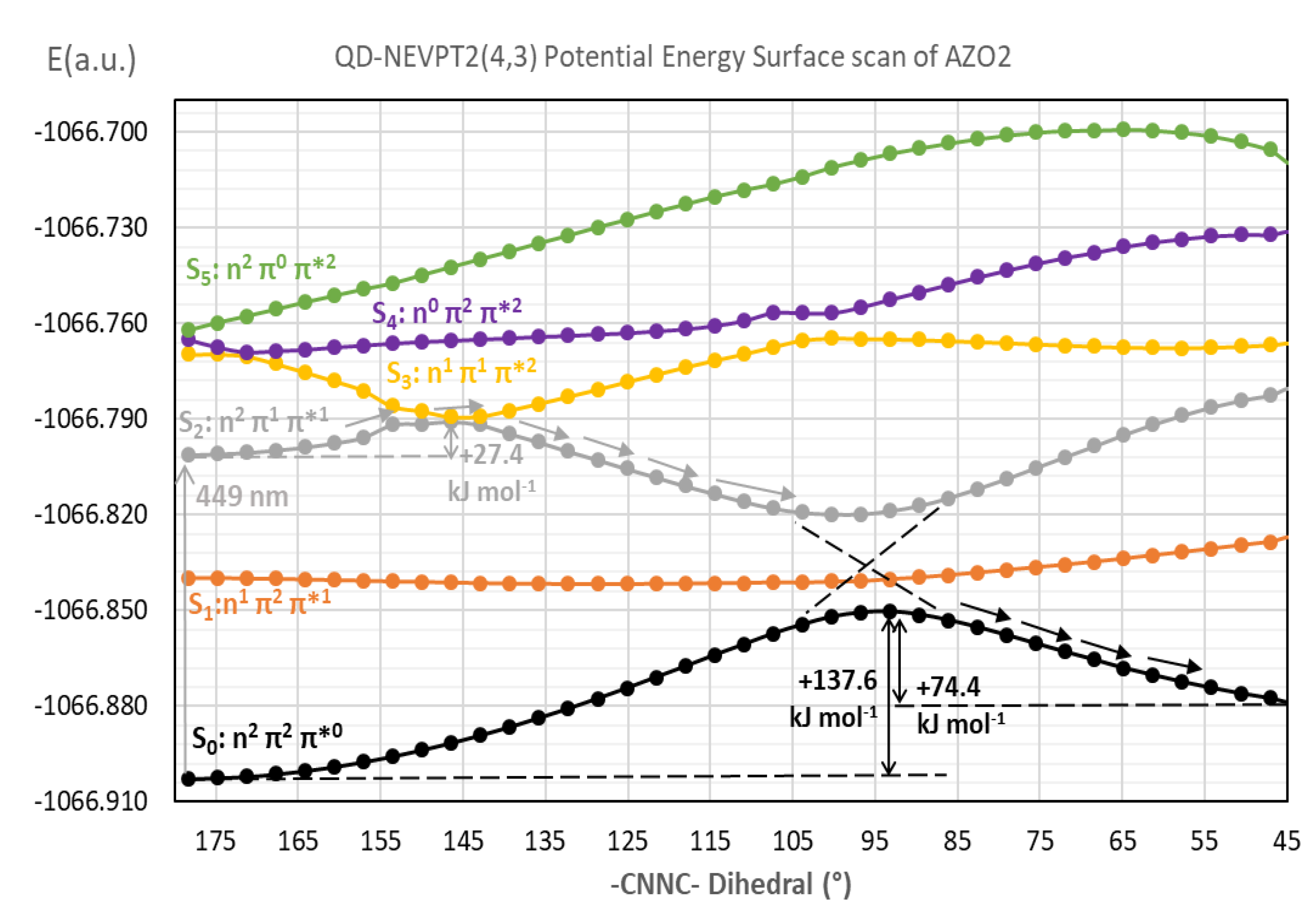
Appendix A.1. Theory and Calculations
References
- Rau, H. Photoisomerization of azobenzenes. Photochem. Photophysics 1990, 2, 119–141. [Google Scholar]
- Wang, L.; Dong, H.; Li, Y.; Xue, C.; Sun, L.-D.; Yan, C.-H.; Li, Q. Reversible Near-Infrared Light Directed Reflection in a Self-Organized Helical Superstructure Loaded with Upconversion Nanoparticles. J. Am. Chem. Soc. 2014, 136, 4480–4483. [Google Scholar] [CrossRef]
- Yamada, M.; Kondo, M.; Mamiya, J.; Yu, Y.; Kinoshita, M.; Barrett, C.J.; Ikeda, T. Photomobile Polymer Materials: Towards Light-Driven Plastic Motors. Angew. Chem. Int. Ed. 2008, 47, 4986–4988. [Google Scholar] [CrossRef] [PubMed]
- Yin, R.; Xu, W.; Kondo, M.; Yen, C.; Mamiya, J.; Ikeda, T.; Yu, Y. Can sunlight drive the photoinduced bending of polymer films? J. Mater. Chem. 2009, 19, 3141–3143. [Google Scholar] [CrossRef]
- Deng, W.; Li, M.-H.; Wang, X.; Keller, P. Light-responsive wires from side-on liquid crystalline azo polymers. Liq. Cryst. 2009, 36, 1023–1029. [Google Scholar] [CrossRef]
- Dong, L.; Feng, Y.; Wang, L.; Feng, W. Azobenzene-based solar thermal fuels: Design, properties, and applications. Chem. Soc. Rev. 2018, 47, 7339–7368. [Google Scholar] [CrossRef] [PubMed]
- Silong, S.; Lutfor, M.R.; Rahman, M.Z.A.; Yunus, W.M.Z.W.; Haron, M.J.; Ahmad, M.B.; Yusoff, W.M.D.W. Synthesis and characterization of side-chain liquid-crystalline polyacrylates containing azobenzene moieties. J. Appl. Polym. Sci. 2002, 86, 2653–2661. [Google Scholar] [CrossRef]
- Baba, K.; Ono, H.; Itoh, E.; Itoh, S.; Noda, K. Kinetic study of thermal Z to e isomerization reactions of azobenzene and 4-dimethvlaniino-4′-nitroazobenzene in Ionic Liquids [1-R-3- methyliniidazolium bis(trifluoromethylsulfonyl)imide with R = Butyl, Pentyl, and Hexyl]. Chemistry 2006, 12, 5328–5333. [Google Scholar] [CrossRef]
- Garcia-Amorós, J.; Martínez, M.; Finkelmann, H.; Velasco, D. Kinetico-Mechanistic Study of the Thermal Cis-to-Trans Isomerization of 4,4′-Dialkoxyazoderivatives in Nematic Liquid Crystals. J. Phys. Chem. B 2010, 114, 1287–1293. [Google Scholar] [CrossRef]
- Dhammika Bandara, H.M.; Burdette, S.C. Photoisomerization in different classes of azobenzene. Chem. Soc. Rev. 2012, 41, 1809–1825. [Google Scholar] [CrossRef]
- Spiridon, M.C.; Jerca, F.A.; Jerca, V.V.; Vasilescu, D.S.; Vuluga, D.M. 2-Oxazoline based photo-responsive azo-polymers. Synthesis, characterization and isomerization kinetics. Eur. Polym. J. 2013, 49, 452–463. [Google Scholar] [CrossRef]
- Jerca, F.A.; Jerca, V.V.; Kajzar, F.; Manea, A.M.; Rau, I.; Vuluga, D.M. Simultaneous two and three photon resonant enhancement of third-order NLO susceptibility in an azo-dye functionalized polymer film. Phys. Chem. Chem. Phys. 2013, 15, 7060–7063. [Google Scholar] [CrossRef]
- Tamai, N.; Miyasaka, H. Ultrafast Dynamics of Photochromic Systems. Chem. Rev. 2000, 100, 1875–1890. [Google Scholar] [CrossRef] [PubMed]
- Dürr, H.; Bouas-Laurent, H. (Eds.) Photochromism: Molecules and Systems; Elsevier: Amsterdam, The Netherlands, 1990. [Google Scholar]
- El’tśòv, A.V.; Sviridov, Y.E.; Whittall, J.W. Organic Photochromes; Consultants Bureau: New York, NY, USA, 1990. [Google Scholar]
- Bang, C.-U.; Shishido, A.; Ikeda, T. Azobenzene Liquid-Crystalline Polymer for Optical Switching of Grating Waveguide Couplers with a Flat Surface. Macromol. Rapid Commun. 2007, 28, 1040–1044. [Google Scholar] [CrossRef]
- Gibbons, W.M.; Shannon, P.J.; Sun, S.-T.; Swetlin, B.J. Surface-mediated alignment of nematic liquid crystals with polarized laser light. Nature 1991, 351, 49–50. [Google Scholar] [CrossRef]
- Tylkowski, B.; Giamberini, M.; Underiner, T.; Prieto, S.F.; Smets, J. Photo-Triggered Microcapsules. Macromol. Symp. 2016, 360, 192–198. [Google Scholar] [CrossRef]
- Tylkowski, B.; Peris, S.; Giamberini, M.; Garcia-Valls, R.; Reina, J.A.; Ronda, J.C. Light-Induced Switching of the Wettability of Novel Asymmetrical Poly(vinyl alcohol)-co-ethylene Membranes Blended with Azobenzene Polymers. Langmuir 2010, 26, 14821–14829. [Google Scholar] [CrossRef] [PubMed]
- Trojanowska, A.; Marturano, V.; Bandeira, N.A.G.; Giamberini, M.; Tylkowski, B. Smart microcapsules for precise delivery systems. Funct. Mater. Lett. 2018, 11, 1850041. [Google Scholar] [CrossRef]
- Del Pezzo, R.; Bandeira, N.A.G.; Trojanowska, A.; Prieto, S.F.; Underiner, T.; Giamberini, M.; Tylkowski, B. Ortho-substituted azobenzene: Shedding light on new benefits. Pure Appl. Chem. 2018, 91, 1533–1547. [Google Scholar] [CrossRef]
- Pirone, D.; Marturano, V.; del Pezzo, R.; Prieto, S.F.; Underiner, T.; Giamberini, M.; Tylkowski, B. Molecular Design of Microcapsule Shells for Visible Light-Triggered Release. Polymers 2019, 11, 904. [Google Scholar] [CrossRef]
- Nicolescu, F.A.; Jerca, V.V.; Stancu, I.C.; Vasilescu, D.S.; Vuluga, D.M. New Organic–Inorganic Hybrids with Azo-dye Content. Des. Monomers Polym. 2010, 13, 437–444. [Google Scholar] [CrossRef][Green Version]
- Whitten, D.G.; Wildes, P.D.; Pacifici, J.G.; Irick, G. Solvent and substituent on the thermal isomerization of substituted azobenzenes. Flash spectroscopic study. J. Am. Chem. Soc. 1971, 93, 2004–2008. [Google Scholar] [CrossRef]
- Merino, E.; Ribagorda, M. Control over molecular motion using the cis–trans photoisomerization of the azo group. Beilstein J. Org. Chem. 2012, 8, 1071–1090. [Google Scholar] [CrossRef] [PubMed]
- Marcandalli, B.; Liddo, L.P.-D.; Fede, C.D.; Bellobono, I.R. Solvent and substituent effects on thermal cis–trans-isomerization of some 4-diethylaminoazobenzenes. J. Chem. Soc. Perkin Trans. 1984, 2, 589–593. [Google Scholar] [CrossRef]
- Tanino, T.; Yoshikawa, S.; Ujike, T.; Nagahama, D.; Moriwaki, K.; Takahashi, T.; Kotani, Y.; Nakano, H.; Shirota, Y. Creation of azobenzene-based photochromic amorphous molecular materials—synthesis, glass-forming properties, and photochromic response. J. Mater. Chem. 2007, 17, 4953–4963. [Google Scholar] [CrossRef]
- Rau, H.; Rötger, D. Photochromic Azobenzenes Which are Stable in the Trans and cis Forms. Mol. Cryst. Liq. Cryst. Sci. Technol. Sect. A 1994, 246, 143–146. [Google Scholar] [CrossRef]
- Jerca, F.A.; Jerca, V.V.; Anghel, D.F.; Stinga, G.; Marton, G.; Vasilescu, D.S.; Vuluga, D.M. Novel Aspects Regarding the Photochemistry of Azo-Derivatives Substituted with Strong Acceptor Groups. J. Phys. Chem. C 2015, 119, 10538–10549. [Google Scholar] [CrossRef]
- Velde, G.T.; Bickelhaupt, F.M.; Baerends, E.J.; Guerra, C.F.; van Gisbergen, S.J.A.; Snijders, J.G.; Ziegler, T. Chemistry with ADF. J. Comput. Chem. 2001, 22, 931–967. [Google Scholar] [CrossRef]
- Adamo, C.; Barone, V. Toward reliable density functional methods without adjustable parameters: The PBE0 model. J. Chem. Phys. 1999, 110, 6158–6170. [Google Scholar] [CrossRef]
- Adamo, C.; Barone, V. Toward chemical accuracy in the computation of NMR shieldings: The PBE0 model. Chem. Phys. Lett. 1998, 298, 113–119. [Google Scholar] [CrossRef]
- Ernzerhof, M.; Scuseria, G.E. Assessment of the Perdew–Burke–Ernzerhof exchange-correlation functional. J. Chem. Phys. 1999, 110, 5029–5036. [Google Scholar] [CrossRef]
- Klamt, A.; Schüürmann, G. COSMO: A new approach to dielectric screening in solvents with explicit expressions for the screening energy and its gradient. J. Chem. Soc. Perkin Trans. 1993, 2, 799–805. [Google Scholar] [CrossRef]
- Casida, M.E. Time-Dependent Density Functional Response Theory for Molecules. In Recent Advances in Density Functional Methods; World Scientific: Singapore, 1995; Volume 1, pp. 155–192. [Google Scholar]
- Roos, B.O. The Complete Active Space Self-Consistent Field Method and its Applications in Electronic Structure Calculations. In Advances in Chemical Physics; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2007; pp. 399–445. [Google Scholar]
- Neese, F. Software update: The ORCA program system, version 4.0. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2018, 8, e1327. [Google Scholar] [CrossRef]
- Neese, F. The ORCA program system. WIREs Comput. Mol. Sci. 2012, 2, 73–78. [Google Scholar] [CrossRef]
- Angeli, C.; Borini, S.; Cestari, M.; Cimiraglia, R. A quasidegenerate formulation of the second order n-electron valence state perturbation theory approach. J. Chem. Phys. 2004, 121, 4043–4049. [Google Scholar] [CrossRef]
- Angeli, C.; Cimiraglia, R.; Evangelisti, S.; Leininger, T.; Malrieu, J.-P. Introduction of n-electron valence states for multireference perturbation theory. J. Chem. Phys. 2001, 114, 10252–10264. [Google Scholar] [CrossRef]
- Nakano, H. Quasidegenerate perturbation theory with multiconfigurational self-consistent-field reference functions. J. Chem. Phys. 1993, 99, 7983–7992. [Google Scholar] [CrossRef]
- Schäfer, A.; Horn, H.; Ahlrichs, R. Fully optimized contracted Gaussian basis sets for atoms Li to Kr. J. Chem. Phys. 1992, 97, 2571–2577. [Google Scholar] [CrossRef]
- Neese, F. An improvement of the resolution of the identity approximation for the formation of the Coulomb matrix. J. Comput. Chem. 2003, 24, 1740–1747. [Google Scholar] [CrossRef]
- Glendening, E.D.; Landis, C.R.; Weinhold, F. NBO 6.0: Natural bond orbital analysis program. J. Comput. Chem. 2013, 34, 1429–1437. [Google Scholar] [CrossRef]
- Reed, A.E.; Schleyer, P.V.R. Chemical bonding in hypervalent molecules. The dominance of ionic bonding and negative hyperconjugation over d-orbital participation. J. Am. Chem. Soc. vol. 1990, 112, 1434–1445. [Google Scholar] [CrossRef]
- Merino, E. Synthesis of azobenzenes: The coloured pieces of molecular materials. Chem. Soc. Rev. 2011, 40, 3835–3853. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Babalhavaeji, A.; Samanta, S.; Beharry, A.A.; Woolley, G.A. Red-Shifting Azobenzene Photoswitches for in Vivo Use. Acc. Chem. Res. 2015, 48, 2662–2670. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, H.; Kashida, H.; Komiyama, M.; Liang, X.; Asanuma, H. Incorporation of methyl group on azobenzene for the effective photo-regulation of hybridization and suppression of thermal isomerization. Nucleic Acids Symp. Ser. 2006, 50, 85–86. [Google Scholar] [CrossRef]
- Djedaini-Pilard, F.; Barbot, F.; Len, C.; Hamon, F. Azobenzenes—synthesis and carbohydrate applications. ResearchGate 2009, 65, 10105–10123. [Google Scholar] [CrossRef]
- Bandara, H.M.D.; Friss, T.R.; Enriquez, M.M.; Isley, W.; Incarvito, C.; Frank, H.A.; Gascón, J.; Burdette, S.C. Proof for the Concerted Inversion Mechanism in the trans→cis Isomerization of Azobenzene Using Hydrogen Bonding To Induce Isomer Locking. J. Org. Chem. 2010, 75, 4817–4827. [Google Scholar] [CrossRef]
- Jerca, V.V.; Jerca, F.A.; Rau, I.; Manea, A.M.; Vuluga, D.M.; Kajzar, F. Advances in understanding the photoresponsive behavior of azobenzenes substituted with strong electron withdrawing groups. Opt. Mater. 2015, 48, 160–164. [Google Scholar] [CrossRef]
- Asano, T.; Okada, T. Thermal Z-E isomerization of azobenzenes. The pressure, solvent, and substituent effects. J. Org. Chem. 1984, 49, 4387–4391. [Google Scholar] [CrossRef]
- Wazzan, N.A.; Richardson, P.R.; Jones, A.C. Cis-Trans isomerisation of azobenzenes studied by laser-coupled NMR spectroscopy and DFT calculations. Photochem. Photobiol. Sci. 2010, 9, 968–974. [Google Scholar] [CrossRef]
- Shepard, R. The Multiconfiguration Self-Consistent Field Method. In Advances in Chemical Physics; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2007; pp. 63–200. [Google Scholar]
- Angeli, C.; Cimiraglia, R.; Malrieu, J.-P. N-electron valence state perturbation theory: A fast implementation of the strongly contracted variant. Chem. Phys. Lett. 2001, 350, 297–305. [Google Scholar] [CrossRef]
- Casellas, J.; Bearpark, M.J.; Reguero, M. Excited-State Decay in the Photoisomerisation of Azobenzene: A New Balance between Mechanisms. ChemPhysChem 2016, 17, 3068–3079. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, L.; Orlandi, G.; Bernardi, F.; Cembran, A.; Garavelli, M. A theoretical study of the lowest electronic states of azobenzene: The role of torsion coordinate in the cis–trans photoisomerization. Theor. Chem. Acc. 2004, 111, 363–372. [Google Scholar] [CrossRef]
- Persico, M.; Granucci, G. Photochemistry: A Modern Theoretical Perspective; Springer International Publishing: Berlin/Heidelberg, Germany, 2018. [Google Scholar]



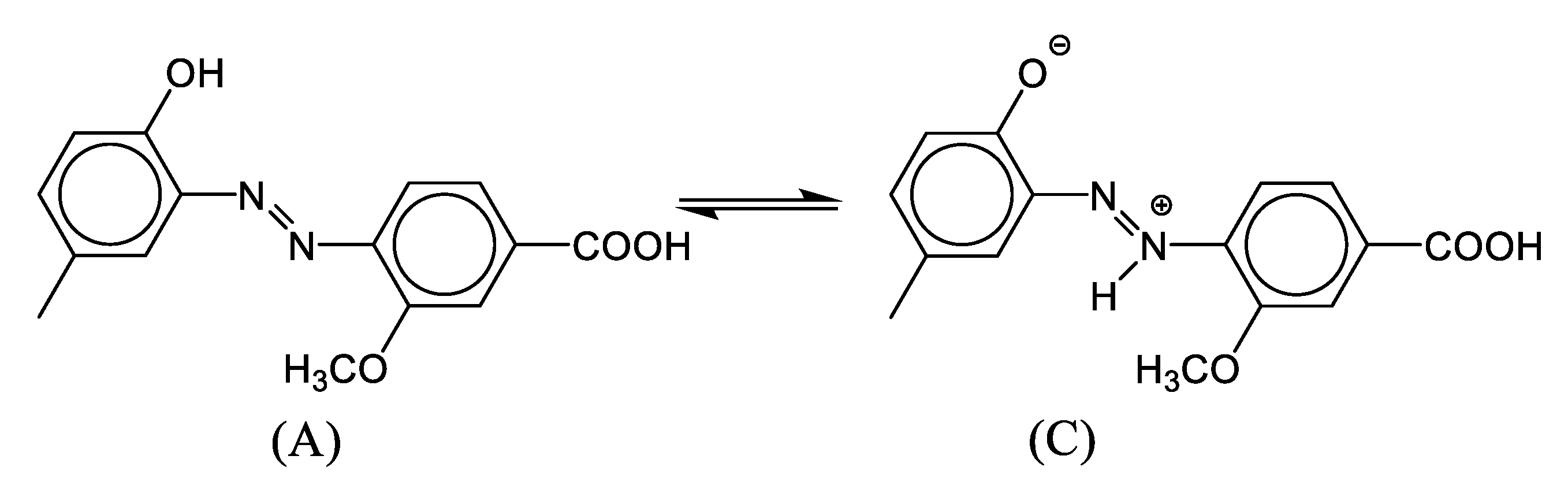
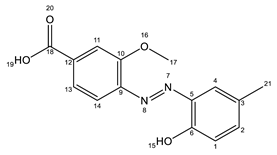 AZO1 AZO1 | 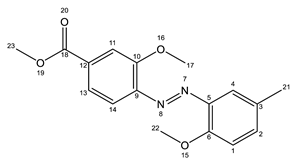 AZO2 AZO2 | |
|---|---|---|
| 1 | 118.2 | 113.3 |
| 2 | 135.1 | 133.0 |
| 3 | 134.2 | 129.4 |
| 4 | 128.1 | 113.6 |
| 5 | 141.7 | 142.2 |
| 6 | 151.7 | 144.5 |
| 9 | 138.1 | 141.7 |
| 10 | 155.5 | 154.8 |
| 11 | 113.7 | 116.6 |
| 12 | 128.9 | 156.0 |
| 13 | 122.0 | 121.4 |
| 14 | 116.1 | 115.4 |
| 17 | 56.3 | 55.8 |
| 18 | 166.7 | 169.1 |
| 21 | 19.9 | 20.1 |
| 22 | - | 56.1 |
| 23 | - | 55.8 |
| Sample | Solvent | λ1 (nm) | λ2 (nm) | ki (s−1) | kr (s−1) | αZ (%) |
|---|---|---|---|---|---|---|
| AZO1 | DMF | 415 | 330 | 1.5 × 10−4 | * | 3.2 |
| AZO2 | DMF | 389 | 320 | 3.08 × 10−3 | 3.77 × 10-6 | 45.7 |
| Excited State # | Main Character | Excitation (nm) | fosc |
|---|---|---|---|
| S1 | π2→ π* (48%) n1→ π* (37%) n2→ π* (12%) | 489 (415) | 0.34 |
| S2 | π2→ π* (51%) n1→ π* (36%) n2→ π* (11%) | 441 (399) | 0.57 |
| S3 | n2→ π* (76%) n1→ π* (23%) | 379 | 6.9 × 10−2 |
| S4 | π1→ π* (98%) | 338 (330) | 0.33 |
| Excited State # | Main Character | Excitation (nm) | fosc |
|---|---|---|---|
| S1 | n2→ π* (58%) n1→ π* (37%) | 494 | 9.6 × 10−3 |
| S2 | π2→ π* (98%) | 448 (389) | 0.91 |
| S3 | n1→ π* (59%) n2→ π* (39%) | 372 | 6.6 × 10−2 |
| S4 | π1→ π* (98%) | 333 (320) | 0.30 |
| Solvent | ki (s−1) | kr (s−1) | αZ (%) | τ1/2(s) |
|---|---|---|---|---|
| DMF | 3.08 × 10−3 | 3.77 × 10−6 | 45.7 | 1.84 × 105 |
| DMSO | 6.38 × 10−3 | 3.14 × 10−6 | 48.3 | 2.21 × 105 |
| DIOX | 8.51 × 10−4 | 2.14 × 10−6 | 16.7 | 3.24 × 105 |
| T (K) | DMF | DMSO | DIOX |
|---|---|---|---|
| 303.15 | 3.77 × 10−6 | 3.14 × 10−6 | 2.14 × 10−6 |
| 313.15 | 1.18 × 10−5 | 1.02 × 10−5 | 6.23 × 10−6 |
| 323.15 | 3.22 × 10−5 | 3.01 × 10−5 | 1.73 × 10−5 |
| 333.15 | 5.59 × 10−5 | 6.93 × 10−5 | 3.81 × 10−5 |
| Solvent | Dipole Moment (D) | A (s−1) | Ea (kJ mol−1) | ΔH‡ (kJ mol−1) | ΔS‡ (J mol−1 K−1) |
|---|---|---|---|---|---|
| DMF | 3.86 | 6.57 × 107 | 76.6 | 74.0 | −104.09 |
| DMSO | 4.1 | 3.41 × 109 | 87.2 | 84.5 | −71.25 |
| DIOX | 0.45 | 2.22 × 108 | 81.3 | 78.6 | −94.03 |
| NLMO π(N–N) Bond Order S0 | NLMO π(N–N) Bond Order S2 | |
|---|---|---|
| AZO1 | 0.951 | 7.84×10−3 |
| AZO2 | 0.934 | 4.43×10−3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pirone, D.; Bandeira, N.A.G.; Tylkowski, B.; Boswell, E.; Labeque, R.; Garcia Valls, R.; Giamberini, M. Contrasting Photo-Switching Rates in Azobenzene Derivatives: How the Nature of the Substituent Plays a Role. Polymers 2020, 12, 1019. https://doi.org/10.3390/polym12051019
Pirone D, Bandeira NAG, Tylkowski B, Boswell E, Labeque R, Garcia Valls R, Giamberini M. Contrasting Photo-Switching Rates in Azobenzene Derivatives: How the Nature of the Substituent Plays a Role. Polymers. 2020; 12(5):1019. https://doi.org/10.3390/polym12051019
Chicago/Turabian StylePirone, Domenico, Nuno A. G. Bandeira, Bartosz Tylkowski, Emily Boswell, Regine Labeque, Ricard Garcia Valls, and Marta Giamberini. 2020. "Contrasting Photo-Switching Rates in Azobenzene Derivatives: How the Nature of the Substituent Plays a Role" Polymers 12, no. 5: 1019. https://doi.org/10.3390/polym12051019
APA StylePirone, D., Bandeira, N. A. G., Tylkowski, B., Boswell, E., Labeque, R., Garcia Valls, R., & Giamberini, M. (2020). Contrasting Photo-Switching Rates in Azobenzene Derivatives: How the Nature of the Substituent Plays a Role. Polymers, 12(5), 1019. https://doi.org/10.3390/polym12051019









