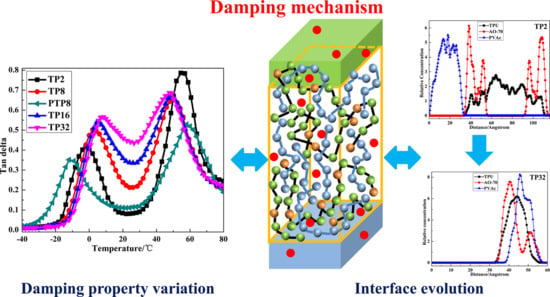
Designing a Polymer-Based Hybrid with Simultaneously Improved Mechanical and Damping Properties via a Multilayer Structure Construction: Structure Evolution and a Damping Mechanism
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
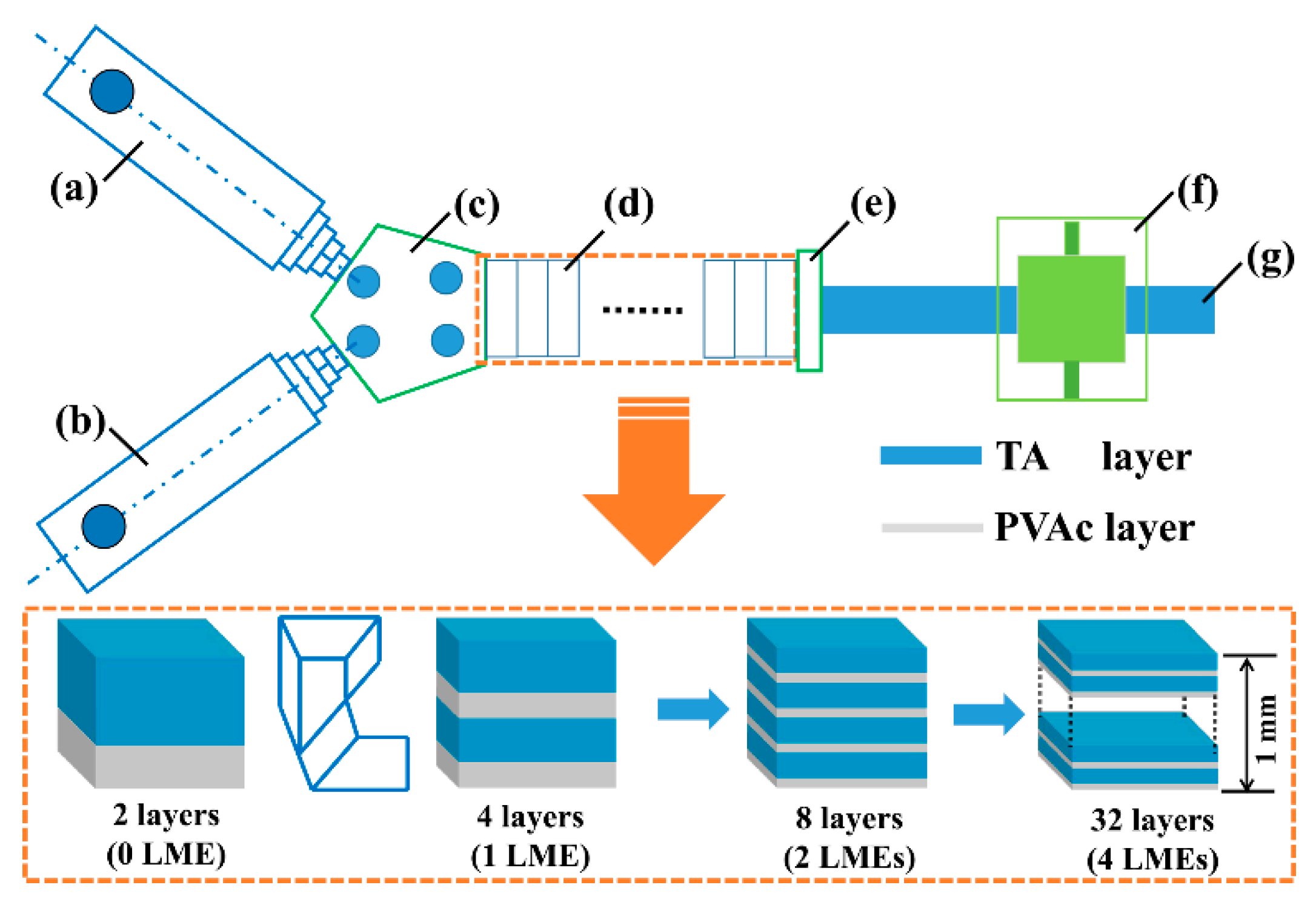
2.2. Sample Preparation
2.3. Characterization
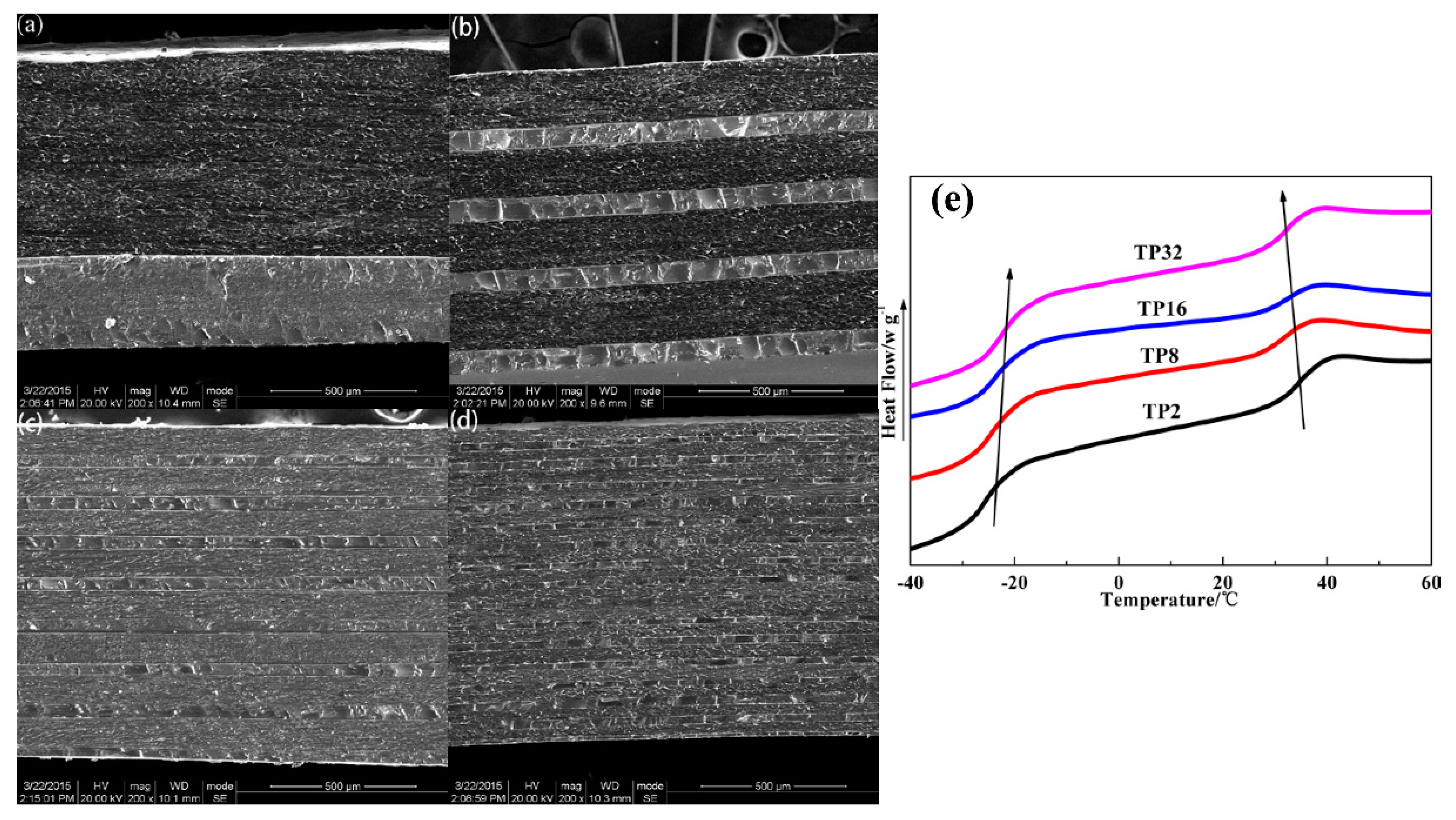
2.3.1. Morphological Observation
2.3.2. Differential Scanning Calorimetry (DSC)
2.3.3. Mechanical Property Testing
2.3.4. Dynamic Mechanical Analysis (DMA)
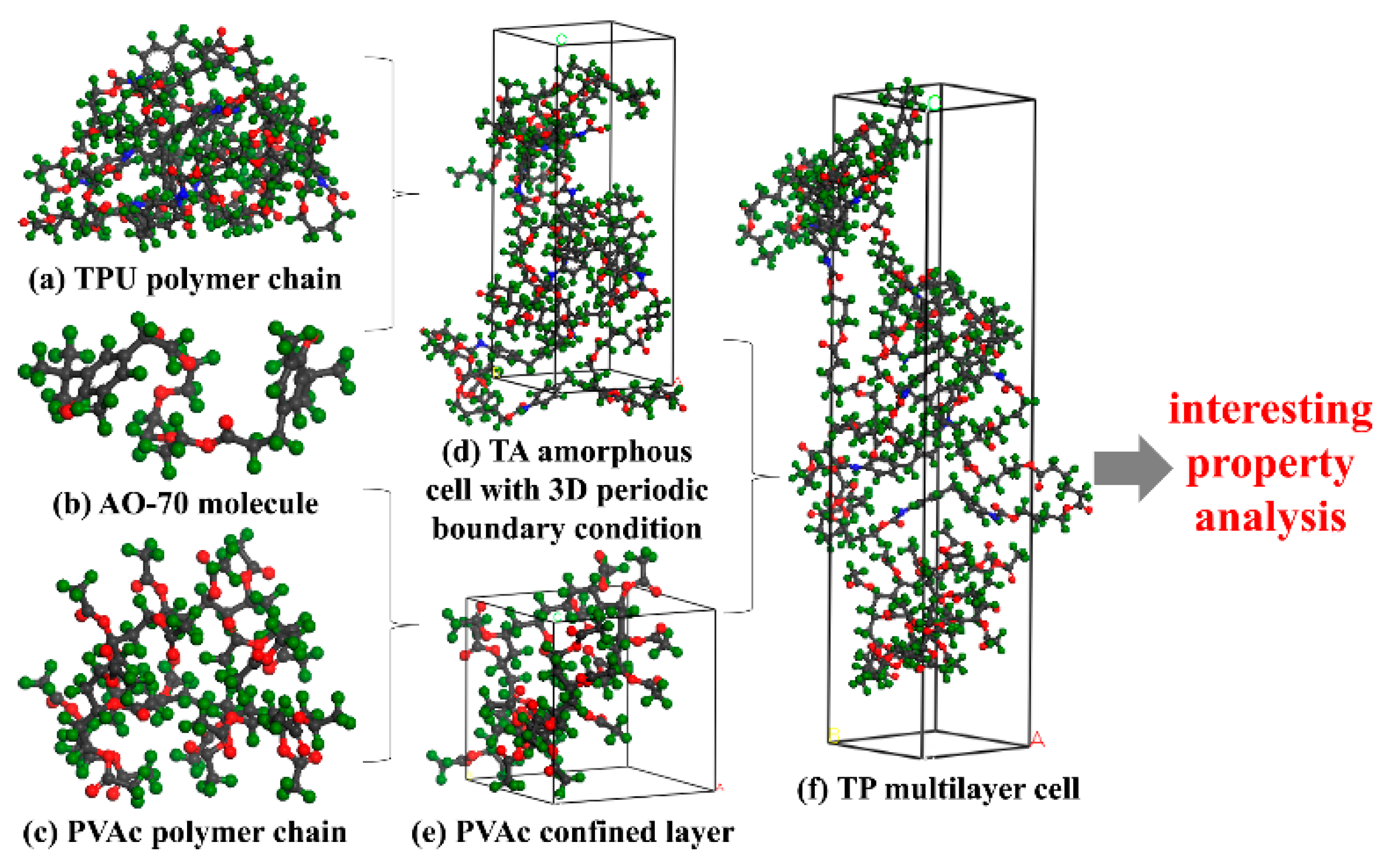
2.4. Simulation Strategies
3. Results and Discussion
3.1. Microstructure and Compatibility
3.2. MD Simulation Analysis
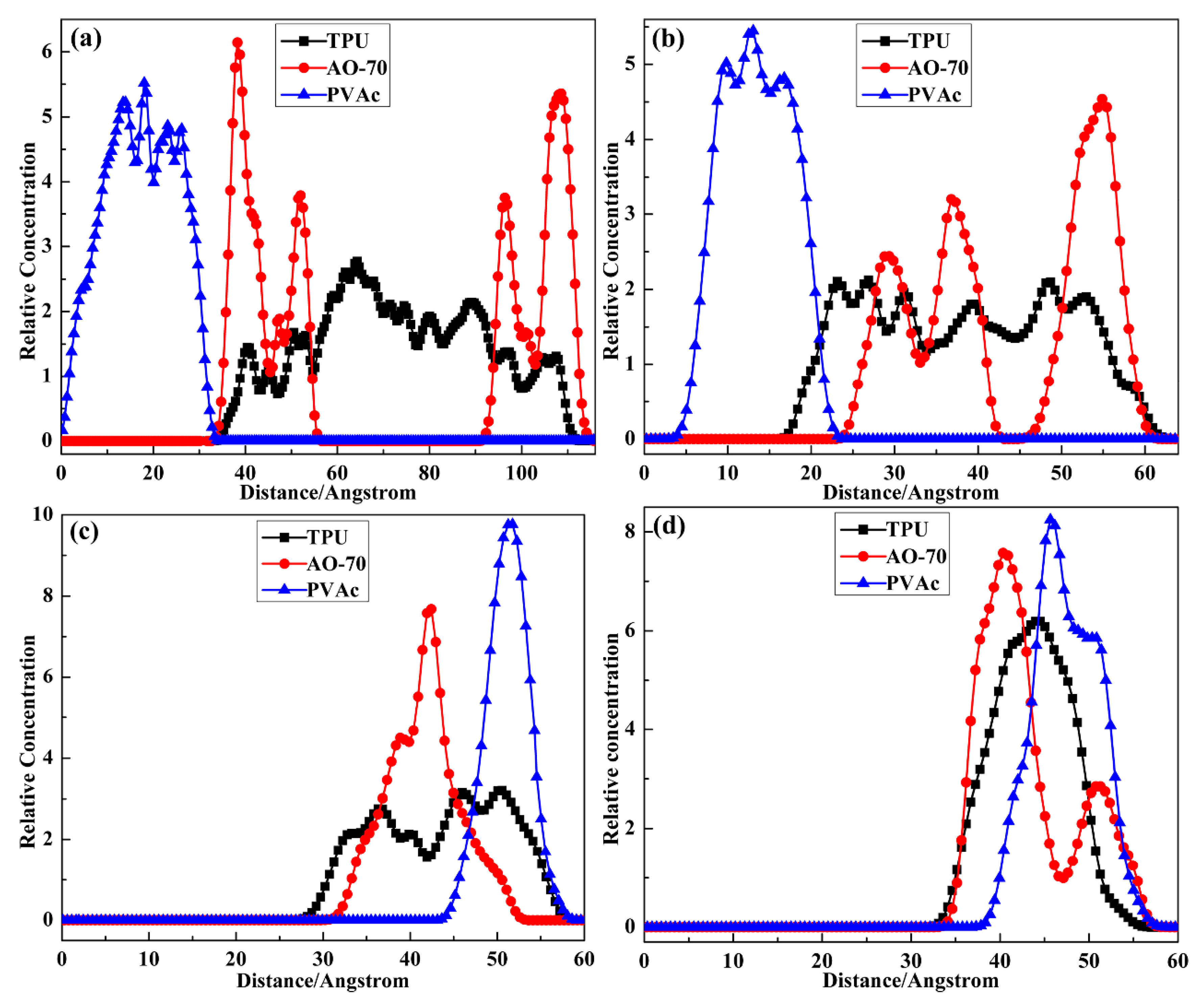
3.2.1. Possible Location of Chains
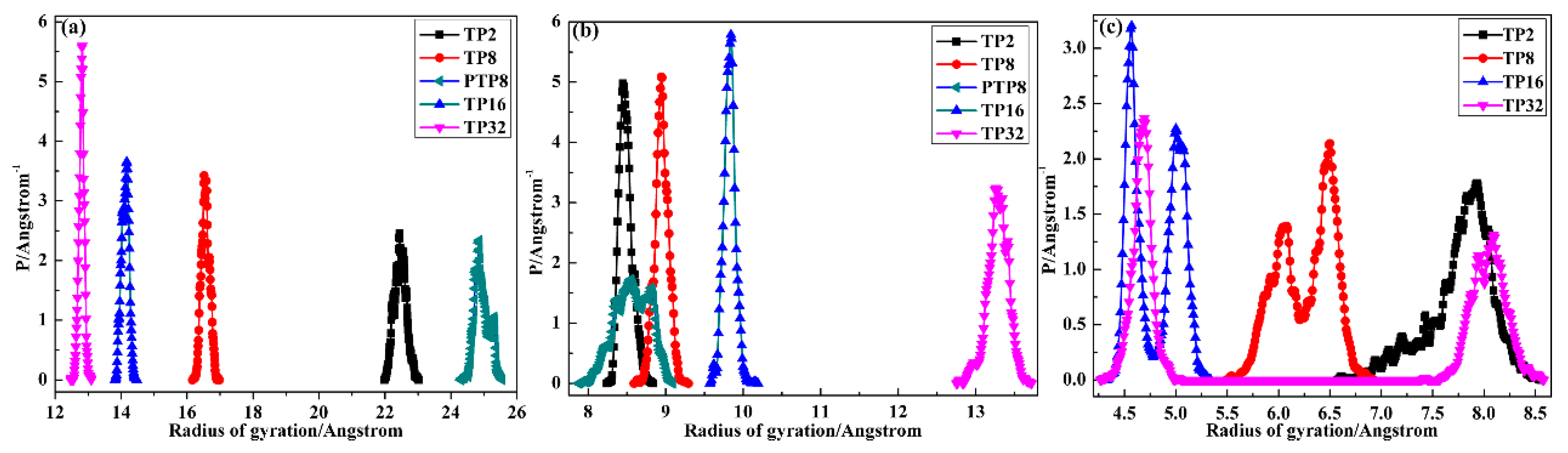
3.2.2. Agglomeration Degree of Chains
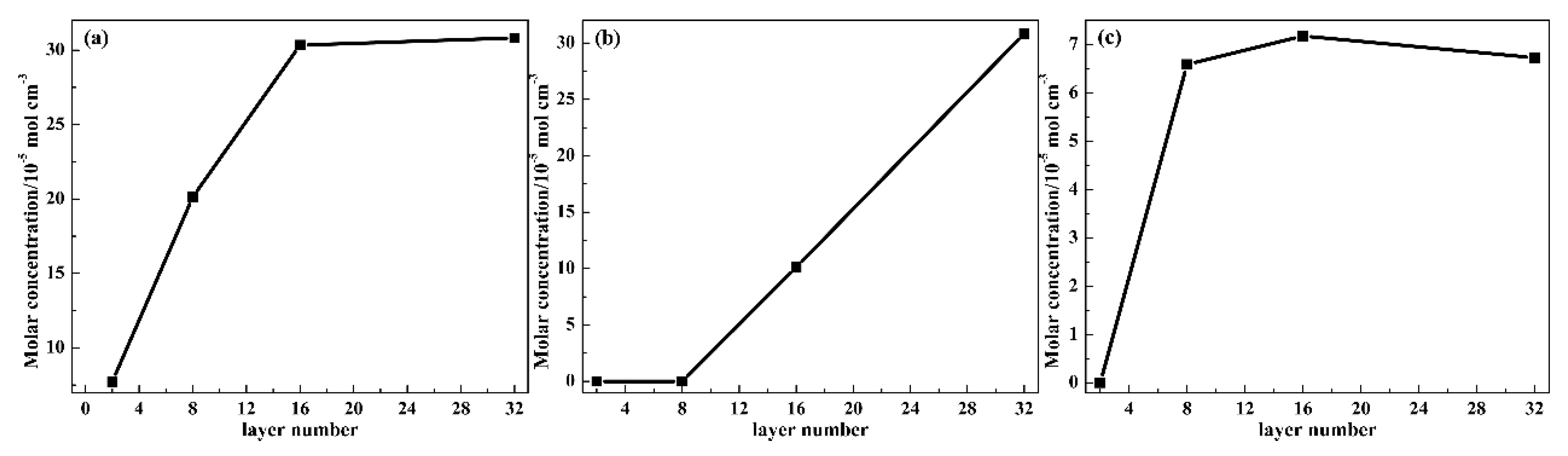
3.2.3. Hydrogen Bond Evolution
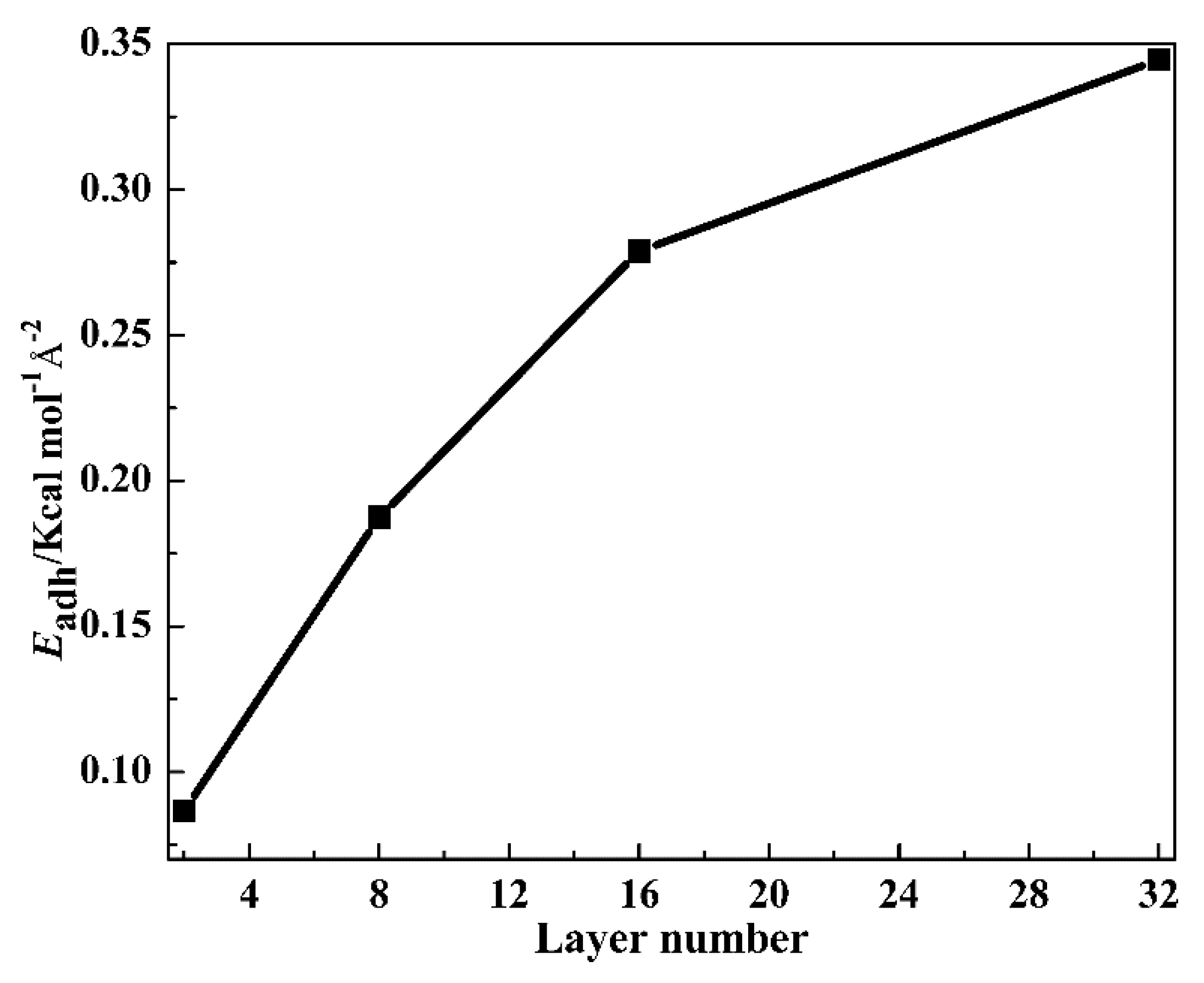
3.2.4. Adhesion Strength of Layers
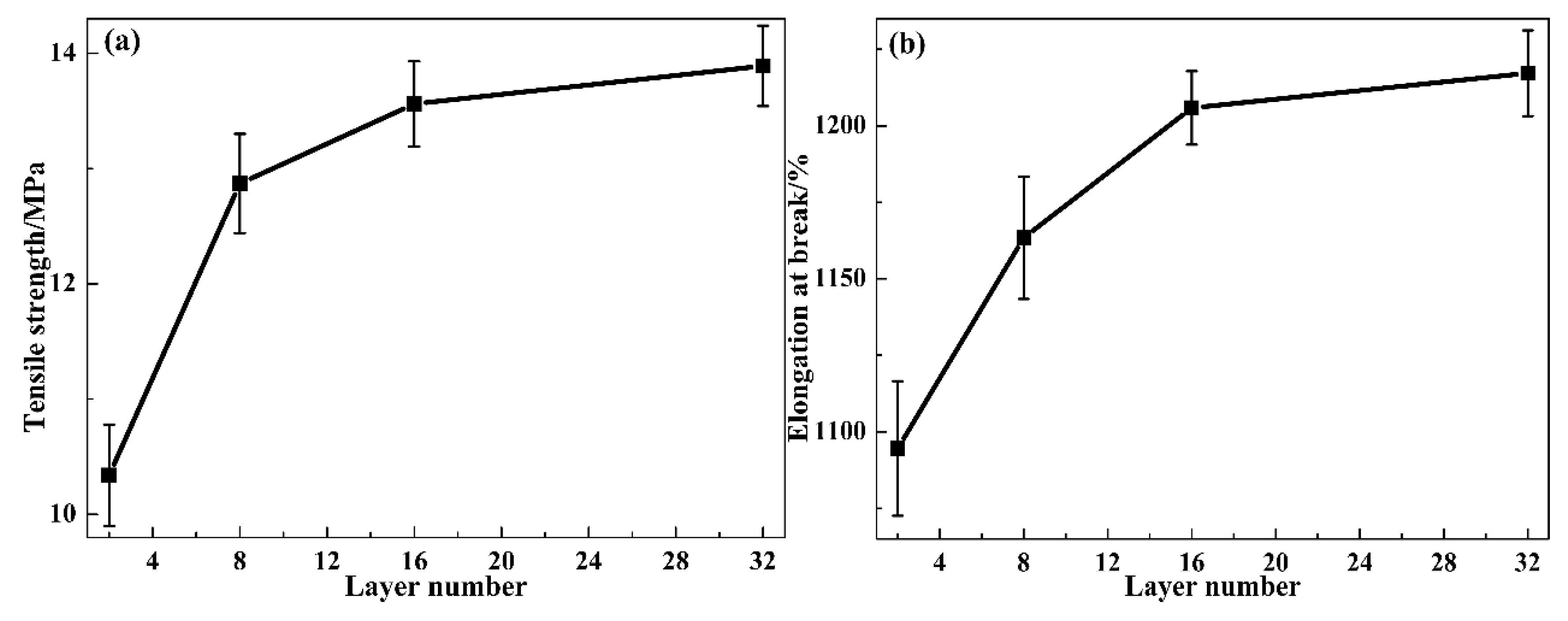
3.3. Mechanical and Dynamic Mechanical Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Latimer, W.-M.; Rodebush, W.-H. Polarity and ionization from the standpoint of the Lewis theory of valence. J. Am. Chem. Soc. 1920, 42, 1419–1433. [Google Scholar] [CrossRef]
- Aakeröy, C.-B.; Seddon, K.-R. The hydrogen bond and crystal engineering. Chem. Soc. Rev. 1993, 22, 397–407. [Google Scholar] [CrossRef]
- Hobza, P.; Havlas, Z. Blue-shifting hydrogen bonds. Chem. Rev. 2000, 100, 4253–4264. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.-S.; Dong, V.-M. Catalytic dehydrogenative cross-coupling: Forming carbon-carbon bonds by oxidizing two carbon-hydrogen bonds. Chem. Rev. 2011, 111, 1215–1292. [Google Scholar] [CrossRef] [PubMed]
- Voth, A.-R.; Khuu, P.; Oishi, K. Halogen bonds as orthogonal molecular interactions to hydrogen bonds. Nat. Chem. 2009, 1, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Uranov, E.-D.; Chapman, D.-R.; Isaacs, A. The Penguin Dictionary of Science; Penguin: London, UK, 1971. [Google Scholar]
- Saenger, W.; Jeffrey, G.-A. Hydrogen Bonding in Biological Structures; Springer: Berlin, Germany, 1991. [Google Scholar]
- Dong, J.; Ozaki, Y. FTIR and FT-Raman studies of partially miscible poly (methyl methacrylate)/poly (4-vinylphenol) blends in solid states. Macromolecules 1997, 30, 286–292. [Google Scholar] [CrossRef]
- Luo, X.; Goh, S.-H.; Lee, S.-Y. Miscibility and interpolymer complexation of poly (1-vinyl-imidazole) with hydroxyl-and carboxyl-containing polymers. Macromol. Chem. Phys. 1999, 200, 399–404. [Google Scholar] [CrossRef]
- Hsu, W.-P. Phase behavior of hydrogen-bonded ternary polymer blends. J. Appl. Polym. Sci. 2003, 89, 2088–2094. [Google Scholar] [CrossRef]
- Cesteros, L.-C.; Meaurio, E.; Katime, I. Miscibility and specific interactions in blends of poly(hydroxy methacry-lates) with poly(vinylpyridines). Macromolecules 1993, 26, 2323–2330. [Google Scholar] [CrossRef]
- Painter, P.-C.; Shenoy, S.-L.; Bhagwagar, D.-E.; Fishburn, J.; Coleman, M.-M. Effect of hydrogen bonding on the melting point depression in polymer blends where one component crystallizes. Macromolecules 1991, 24, 5623–5629. [Google Scholar] [CrossRef]
- He, Y.; Zhu, B.; Inoue, Y. Hydrogen bonds in polymer blends. Prog. Polym. Sci. 2004, 29, 1021–1051. [Google Scholar] [CrossRef]
- Wu, C.; Yamagishi, T.-A.; Nakamoto, Y.; Ishida, S.-I.; Nitta, K.-H.; Kubota, S. Viscoelastic properties of an organic hybrid of chlorinated polyethylene and a small molecule. J. Polym. Sci. Pol. Phys. 2000, 38, 1341–1347. [Google Scholar] [CrossRef]
- Wu, C.; Yamagishi, T.-A.; Nakamoto, Y.; Ishida, S.; Nitta, K.-H.; Kubota, S. Organic hybrid of chlorinated polyethylene and hindered phenol. I. Dynamic mechanical properties. J. Polym. Sci. Pol. Phys. 2000, 38, 2285–2295. [Google Scholar] [CrossRef]
- Cao, Y.; Shen, F.; Mou, H.; Cao, D.; Xu, H.; Wu, C. Hydrogenated nitrile butadiene rubber and hindered phenol composite. I. Miscibility and dynamic mechanical property. Polym. Eng. Sci. 2010, 50, 2375–2381. [Google Scholar] [CrossRef]
- Cao, Y.; Mou, H.; Shen, F.; Xu, H.; Hu, G.-H.; Wu, C. Hydrogenated nitrile butadiene rubber and hindered phenol composite. II. Characterization of hydrogen bonding. Polym. Eng. Sci. 2011, 51, 201–208. [Google Scholar] [CrossRef]
- Zhao, X.-Y.; Xiang, P.; Tian, M.; Hao, F.; Jin, R.; Zhang, L.-Q. Nitrile butadiene rubber/hindered phenol nanocomposites with improved strength and high damping performance. Polymer 2007, 48, 6056–6063. [Google Scholar] [CrossRef]
- Qiao, B.; Zhao, X.; Yue, D.; Zhang, L.; Wu, S. A combined experiment and molecular dynamics simulation study of hydrogen bonds and free volume in nitrile-butadiene rubber/hindered phenol damping mixtures. J. Mater. Chem. 2012, 22, 12339–12348. [Google Scholar] [CrossRef]
- Song, M.; Zhao, X.; Li, Y.; Hu, S.; Zhang, L.; Wu, S. Molecular dynamics simulations and microscopic analysis of the damping performance of hindered phenol AO-60/nitrile-butadiene rubber composites. RSC Adv. 2014, 4, 6719–6729. [Google Scholar] [CrossRef]
- Song, M.; Zhao, X.; Li, Y.; Chan, T.; Zhang, L.; Wu, S. Effect of acrylonitrile content on compatibility and damping properties of hindered phenol AO-60/nitrile-butadiene rubber composites: Molecular dynamics simulation. RSC Adv. 2014, 4, 48472–48479. [Google Scholar] [CrossRef]
- Yang, D.; Zhao, X.; Chan, T.; Zhang, L.; Wu, S. Investigation of the damping properties of hindered phenol AO-80/polyacrylate hybrids using molecular dynamics simulations in combination with experimental methods. J. Mater. Sci. 2016, 51, 5760–5774. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, G.; Lu, F.; Zhang, L.; Wu, S. Molecular-level insight of hindered phenol AO-70/nitrile-butadiene rubber damping composites through a combination of a molecular dynamics simulation and experimental method. RSC Adv. 2016, 6, 85994–86005. [Google Scholar] [CrossRef]
- Xiao, D.; Zhao, X.; Feng, Y. The structure and dynamic properties of thermoplastic polyurethane elastomer/hindered phenol hybrids. J. Appl. Polym. Sci. 2010, 116, 2143–2150. [Google Scholar]
- Zhu, J.; Zhao, X.; Liu, L.; Yang, R.; Song, M.; Wu, S. Thermodynamic analyses of the hydrogen bond dissociation reaction and their effects on damping and compatibility capacities of polar small molecule/nitrile-butadiene rubber systems: Molecular simulation and experimental study. Polymer 2018, 155, 152–167. [Google Scholar] [CrossRef]
- Wu, C.; Kuriyama, T.; Inoue, T. Crystalline structure and morphology of a hindered phenol in a chlorinated polyethylene matrix. J. Mater. Sci. 2004, 39, 1249–1254. [Google Scholar] [CrossRef]
- Wu, C. Microstructural development of a vitrified hindered phenol compound during thermal annealing. Polymer 2003, 44, 1697–1703. [Google Scholar] [CrossRef]
- Shi, G.; Yin, X.; Wu, G. Thermodynamic phase analysis of acrylic polymer/hindered phenol hybrids: Effects of hydrogen bonding strength. Polymer 2018, 153, 317–324. [Google Scholar] [CrossRef]
- Chen, S.; Wang, Q.; Wang, T. Hydroxy-terminated liquid nitrile rubber modified castor oil based polyurethane/epoxy IPN composites: Damping, thermal and mechanical properties. Polym. Test. 2011, 30, 726–731. [Google Scholar] [CrossRef]
- Chen, B.; Gao, W.; Shen, J.; Guo, S.-Y. The multilayered distribution of intumescent flame retardants and its influence on the fire and mechanical properties of polypropylene. Compos. Sci. Technol. 2014, 93, 54–60. [Google Scholar] [CrossRef]
- Zhu, J.; Shen, J.; Guo, S.-Y.; Sue, H.-J. Confined distribution of conductive particles in polyvinylidene fluoride-based multilayered dielectrics: Toward high permittivity and breakdown strength. Carbon 2015, 84, 355–364. [Google Scholar] [CrossRef]
- He, G.; Li, J.; Zhang, F.; Lei, F.; Guo, S.-Y. A quantitative analysis of the effect of interface delamination on the fracture behavior and toughness of multilayered propylene–ethylene copolymer/low density polyethylene films by the essential work of fracture (EWF). Polymer 2014, 55, 1583–1592. [Google Scholar] [CrossRef]
- Gao, W.; Zheng, Y.; Shen, J.; Guo, S.-Y. Electrical Properties of Polypropylene-Based Composites Controlled by Multilayered Distribution of Conductive Particles. ACS Appl. Mater. Interfaces 2014, 7, 1541–1549. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xu, Y.; Zhang, X. Progress on the layer-by-layer assembly of multilayered polymer composites: Strategy, structural control and applications. Prog. Polym. Sci. 2018, 89, 76–107. [Google Scholar] [CrossRef]
- Xu, K.-M.; Zhang, F.; Zhang, X.; Hu, Q.-M.; Wu, H.; Guo, S.-Y. Molecular insights into hydrogen bonds in polyurethane/hindered phenol hybrids: Evolution and relationship with damping properties. J. Mater. Chem. A 2014, 2, 8545–8556. [Google Scholar] [CrossRef]
- Xu, K.-M.; Zhang, F.; Zhang, X.; Guo, J.; Wu, H.; Guo, S.-Y. Molecular insights into the damping mechanism of Poly(vinyl acetate)/hindered phenol hybrids by a combination of experiment and molecular dynamics simulation. RSC Adv. 2014, 5, 4200–4209. [Google Scholar] [CrossRef]
- Zhang, F.; Guo, M.; Xu, K.-M.; He, G.; Wu, H.; Guo, S.-Y. Multilayered damping composites with damping layer/constraining layer prepared by a novel method. Compos. Sci. Technol. 2014, 101, 167–172. [Google Scholar] [CrossRef]
- Sun, H. COMPASS: An ab Initio Force-Field Optimized for Condensed-Phase Applications Overview with Details on Alkane and Benzene Compounds. J. Phys. Chem. B 1998, 102, 7338–7364. [Google Scholar] [CrossRef]
- Habasaki, J.; Ueda, A. Molecular dynamics study of one component soft-core system-Analytical expression of non-equilibrium relaxation in constant pressure conditions. J. Non-Cryst. Solids 2016, 447, 212–222. [Google Scholar] [CrossRef]
- Bian, C.; Wang, S.; Liu, Y.; Su, K.; Jing, X. Role of nonbond interactions in the glass transition of novolac-type phenolic resin: A molecular dynamics study. Ind. Eng. Chem. Res. 2016, 55, 9440–9451. [Google Scholar] [CrossRef]
- Prathab, B.; Subramanian, V.; Aminabhavi, T.-M. Molecular dynamics simulations to investigate polymer-polymer and polymer-metal oxide interactions. Polymer 2007, 48, 409–416. [Google Scholar] [CrossRef]
- Dedmon, M.-M.; Lindorff-Larsen, K.; Christodoulou, J.; Vendruscolo, M.; Dobson, C.-M. Mapping long-range interactions in alpha-synuclein using spin-label NMR and ensemble molecular dynamics simulations. J. Am. Chem. Soc. 2005, 127, 476–477. [Google Scholar] [CrossRef]
- Connolly, M.-L. Solvent-accessible surfaces of proteins and nucleic acids. Science 1983, 221, 709–713. [Google Scholar] [CrossRef] [PubMed]
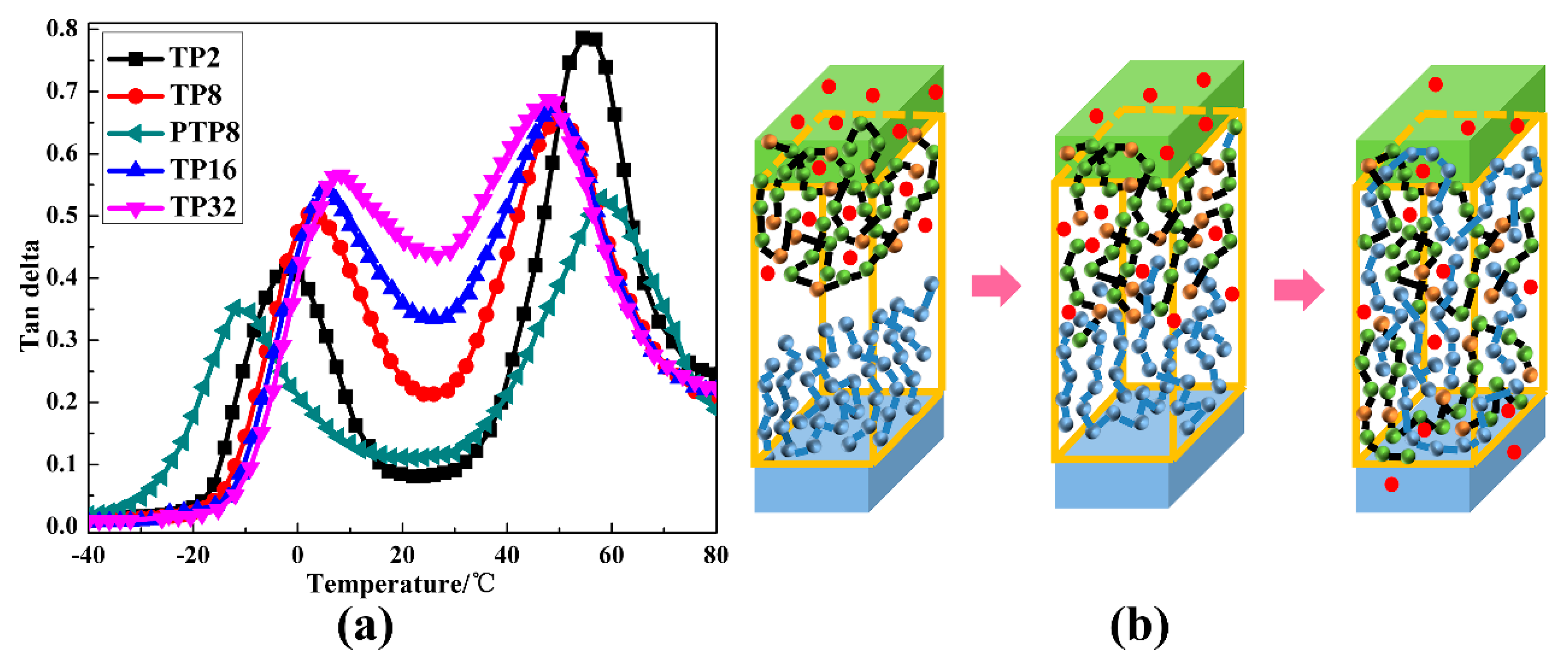
| Sample Code | Tan δmax (TPU) | Tg (TPU) | Tan δmax (PVAc) | Tg (PVAc) | Temperature Range of Tan δ > 0.3 |
|---|---|---|---|---|---|
| TP2 | 0.42 | −2.16 | 0.79 | 57.91 | 42.24 |
| TP8 | 0.50 | 2.44 | 0.66 | 52.34 | 55.62 |
| PTP8 | 0.35 | −12.01 | 0.53 | 57.98 | 32.17 |
| TP16 | 0.54 | 3.79 | 0.67 | 51.22 | 68.47 |
| TP32 | 0.56 | 4.47 | 0.68 | 50.18 | 68.55 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, K.; Hu, Q.; Wu, H.; Guo, S.; Zhang, F. Designing a Polymer-Based Hybrid with Simultaneously Improved Mechanical and Damping Properties via a Multilayer Structure Construction: Structure Evolution and a Damping Mechanism. Polymers 2020, 12, 446. https://doi.org/10.3390/polym12020446
Xu K, Hu Q, Wu H, Guo S, Zhang F. Designing a Polymer-Based Hybrid with Simultaneously Improved Mechanical and Damping Properties via a Multilayer Structure Construction: Structure Evolution and a Damping Mechanism. Polymers. 2020; 12(2):446. https://doi.org/10.3390/polym12020446
Chicago/Turabian StyleXu, Kangming, Qiaoman Hu, Hong Wu, Shaoyun Guo, and Fengshun Zhang. 2020. "Designing a Polymer-Based Hybrid with Simultaneously Improved Mechanical and Damping Properties via a Multilayer Structure Construction: Structure Evolution and a Damping Mechanism" Polymers 12, no. 2: 446. https://doi.org/10.3390/polym12020446
APA StyleXu, K., Hu, Q., Wu, H., Guo, S., & Zhang, F. (2020). Designing a Polymer-Based Hybrid with Simultaneously Improved Mechanical and Damping Properties via a Multilayer Structure Construction: Structure Evolution and a Damping Mechanism. Polymers, 12(2), 446. https://doi.org/10.3390/polym12020446