Electrosynthesis and Electrochromism of a New Crosslinked Polydithienylpyrrole with Diphenylpyrenylamine Subunits
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Electrochemical Polymerization
2.3. Measurements and Methods
2.4. Fabrication of Electrochromic Devices
3. Results and Discussion
3.1. Synthesis and Structural Characterization
3.2. UV-Vis Absorption and Photoluminescence of DPPA-2SNS Monomer
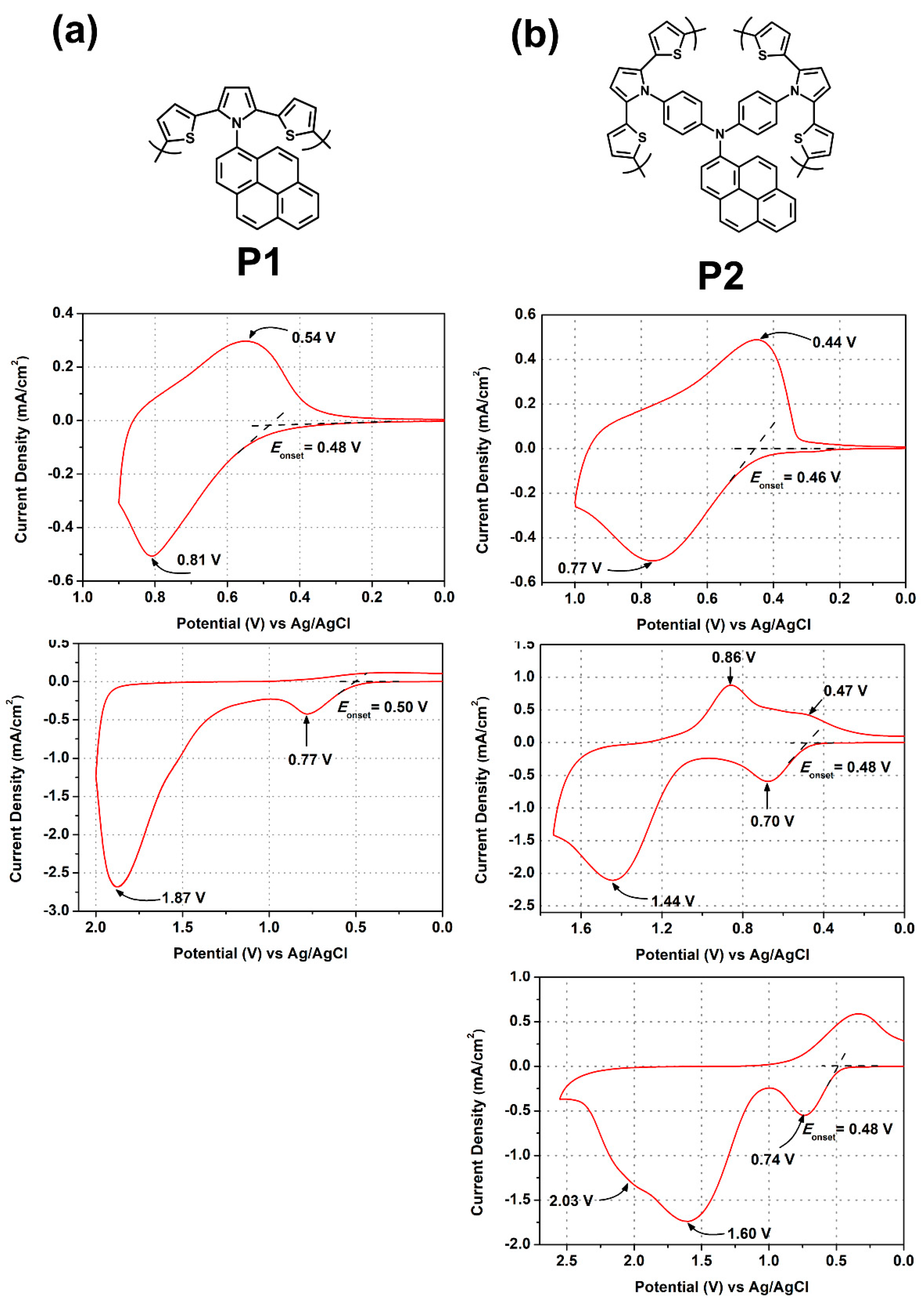
3.3. Electrochemical Activity and Polymerization of SNS Monomers
3.4. UV-vis Absorption of the PSNS Films
3.5. Redox Response of Polymers
3.6. Spectroelectrochemical Properties of Polymers
3.7. Electrochromic Switching
3.8. Electrochromic Devices
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rosseinsky, D.R.; Mortimer, R.J. Electrochromic systems and the prospects for devices. Adv. Mater. 2001, 13, 783–793. [Google Scholar] [CrossRef]
- Granqvist, C.G. Electrochromics for smart windows: Oxide-based thin films and devices. Thin Solid Films 2014, 564, 1–38. [Google Scholar] [CrossRef]
- Beaujuge, P.M.; Ellinger, S.; Reynolds, J.R. The donor-acceptor approach allows a black-to-transmissive switching polymeric electrochrome. Nat. Mater. 2008, 7, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Malti, A.; Brooke, R.; Liu, X.; Zhao, D.; Ersman, P.A.; Fahlman, M.; Jonsson, M.P.; Berggren, M.; Crispin, X. Freestanding electrochromic paper. J. Mater. Chem. C 2016, 4, 9680–9686. [Google Scholar] [CrossRef]
- Osterholm, A.M.; Shen, D.E.; Kerszulis, J.A.; Bulloch, R.H.; Kuepfert, M.; Dyer, A.L.; Reynolds, J.R. Four shades of brown: Tuning of electrochromic polymer blends toward high-contrast eyewear. ACS Appl. Mater. Interfaces 2015, 7, 1413–1421. [Google Scholar] [CrossRef] [PubMed]
- Koyuncu, S.; Koyuncu, F.B. A new ITO-compatible side chain-functionalized multielectrochromic polymer for use in adaptive camouflage-like electrochromic devices. React. Funct. Polym. 2018, 131, 174–180. [Google Scholar] [CrossRef]
- Yang, P.; Sun, P.; Mai, W. Electrochromic energy storage devices. Mater. Today 2016, 19, 394–402. [Google Scholar] [CrossRef]
- Mortimer, R.J. Electrochromic materials. Annu. Rev. Mater. Res. 2011, 41, 241–268. [Google Scholar] [CrossRef]
- Amb, C.M.; Dyer, A.L.; Reynolds, J.R. Navigating the color palette of solution-processable electrochromic polymers. Chem. Mater. 2011, 23, 397–415. [Google Scholar] [CrossRef]
- Gunbas, G.; Toppare, L. Electrochromic conjugated polyheterocycles and derivatives—highlights from the last decade towards realization of long lived aspirations. Chem. Commun. 2012, 48, 1083–1101. [Google Scholar] [CrossRef]
- Neo, W.; Ye, Q.; Chua, S.-J.; Xu, J. Conjugated polymer-based electrochromics: Materials, device fabrication and application prospects. J. Mater. Chem. C 2016, 4, 7364–7376. [Google Scholar] [CrossRef]
- Lv, X.; Li, W.; Ouyang, M.; Zhang, Y.; Wright, D.S.; Zhang, C. Polymeric electrochromic materials with donor-acceptor structures. J. Mater. Chem. C 2017, 5, 12–28. [Google Scholar] [CrossRef]
- Thelakkat, M. Star-shaped, dendrimeric and polymeric triarylamines as photoconductors and hole transport materials for electro-optical applications. Macromol. Mater. Eng. 2002, 287, 442–461. [Google Scholar] [CrossRef]
- Shirota, Y. Photo- and electroactive amorphous molecular materials—molecular design, syntheses, reactions, properties, and applications. J. Mater. Chem. 2005, 15, 75–93. [Google Scholar] [CrossRef]
- Shirota, Y.; Kageyama, H. Charge carrier transporting molecular materials and their applications in devices. Chem. Rev. 2007, 107, 953–1010. [Google Scholar] [CrossRef]
- Ning, Z.; Tian, H. Triarylamine: A promising core unit for efficient photovoltaic materials. Chem. Commun. 2009, 5483–5495. [Google Scholar] [CrossRef]
- Iwan, A.; Sek, D. Polymers with triphenylamine units: Photonic and electroactive materials. Prog. Polym. Sci. 2011, 36, 1277–1325. [Google Scholar] [CrossRef]
- Liang, M.; Chen, J. Arylamine organic dyes for dye-sensitized solar cells. Chem. Soc. Rev. 2013, 42, 3453–3488. [Google Scholar] [CrossRef]
- Yen, H.-J.; Liou, G.-S. Recent advances in triphenylamine-based electrochromic derivatives and polymers. Polym. Chem. 2018, 9, 3001–3018. [Google Scholar] [CrossRef]
- Yen, H.-J.; Liou, G.-S. Design and preparation of triphenylamine-based polymeric materials towards emergent optoelectronic applications. Prog. Polym. Sci. 2019, 89, 250–287. [Google Scholar] [CrossRef]
- Park, S.Y.; Yoon, J.H.; Hong, C.S.; Souane, R.; Kim, J.S.; Mattews, S.E.; Vicens, J. A pyrenyl-appended triazole-based calix[4]arene as a fluorescent sensor for Cd2+ and Zn2+. J. Org. Chem. 2008, 73, 8212–8218. [Google Scholar] [CrossRef] [PubMed]
- Conlon, P.; Yang, C.J.; Wu, Y.; Chan, Y.; Martinez, K.; Kim, Y.; Stevens, N.; Marti, A.A.; Jockusch, S.; Turro, N.J.; et al. Pyrene excimer signaling molecular beacons for probing nucleic acids. J. Am. Chem. Soc. 2008, 130, 336–342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Figueira-Duarte, T.M.; Mullen, K. Pyrene-based materials for organic electronics. Chem. Rev. 2011, 111, 7260–7314. [Google Scholar] [CrossRef] [PubMed]
- Kung, Y.-C.; Hsiao, S.-H. Fluorescent and electrochromic polyamides with pyrenylamine chromophore. J. Mater. Chem. 2010, 20, 5481–5492. [Google Scholar] [CrossRef]
- Kung, Y.-C.; Hsiao, S.-H. Solution-processable, high-Tg, ambipolar polyimide electrochromics bearing pyrenylamine units. J. Mater. Chem. 2011, 21, 1746–1754. [Google Scholar] [CrossRef]
- Hsiao, S.-H.; Huang, Y.-P. Redox-active and fluorescent pyrene-based triarylamine dyes and their derived electrochromic polymers. Dye. Pigment. 2018, 158, 368–381. [Google Scholar] [CrossRef]
- Kumar, A.; Welsh, D.M.; Morvant, M.C.; Piroux, F.; Abboud, K.A.; Reynolds, J.R. Conducting poly(3,4-alkylenedioxythiophene) derivatives as fast electrochromics with high-contrast ratios. Chem. Mater. 1998, 10, 896–902. [Google Scholar] [CrossRef]
- Walczak, R.M.; Reynolds, J.R. Poly(3,4-alkylenedioxypyrrole)s: The PXDOPs as versatile yet underutilized electroactive and conducting polymers. Adv. Mater. 2006, 18, 1121–1131. [Google Scholar] [CrossRef]
- Mert, O.; Demir, A.S.; Cihaner, A. Pyrrole coupling chemistry: Investigation of electroanalytic, spectroscopic and thermal properties of N-substituted poly(bis-pyrrole) films. RSC Adv. 2013, 3, 2035–2042. [Google Scholar] [CrossRef]
- Witker, D.; Reynolds, J.R. Soluble variable color carbazole-containing electrochromic polymers. Macromolecules 2005, 38, 7636–7644. [Google Scholar] [CrossRef]
- Shah, K.W.; Wang, S.-X.; Soo, X.Y.; Xu, J. Viologen-based electrochromic materials: From small molecules, polymers and composites to their applications. Polymers 2019, 11, 1839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beaupre, S.; Dumas, J.; Leclerc, M. Toward the development of new textile/plastic electrochromic cells using triphenylamine-based copolymers. Chem. Mater. 2006, 18, 4011–4018. [Google Scholar] [CrossRef]
- Mcleod, G.G.; Mahboubian-Jones, M.G.B.; Pethrick, R.A.; Watson, S.D.; Truong, N.D.; Galin, J.C.; Francois, J. Synthesis, electrochemical polymerization and properties of poly(2,5-di(2-thienyl)pyrrole). Polymer 1986, 27, 455–458. [Google Scholar] [CrossRef]
- Ferraris, J.P.; Skiles, G.D. ‘Substitutional alloys’ of organic conductors. Polymer 1987, 28, 179–182. [Google Scholar] [CrossRef]
- Ferraris, J.P.; Hanlon, T.R. Optical, electrical and electrochemical properties of heteroaromatic copolymers. Polymer 1989, 30, 1319–1327. [Google Scholar] [CrossRef]
- Cihaner, A.; Algi, F. Processable electrochromic and fluorescent polymers based on N-substituted thienylpyrrole. Electrochim. Acta 2008, 54, 665–670. [Google Scholar] [CrossRef]
- Camurlu, P.; Karagoren, N. Clickable, versatile poly(2,5-dithienylpyrrole) derivatives. React. Funct. Polym. 2013, 73, 847–853. [Google Scholar] [CrossRef]
- Camurlu, P. Polypyrrole derivatives for electrochromic applications. RSC Adv. 2014, 4, 55832–55845. [Google Scholar] [CrossRef]
- Pandule, S.; Oprea, A.; Barsan, N.; Weimar, U.; Persaud, K. Syntheses of poly(2,5-di(thiophen-2-yl)-1H-pyrrole) derivatives and the effects of the substituents on their properties. Synth. Met. 2014, 196, 158–165. [Google Scholar] [CrossRef]
- Sefer, E.; Bilgili, H.; Gultekin, B.; Tonga, M.; Koyuncu, S. A narrow range multielectrochromism from 2,5-di-(2-thienyl)-1H-pyrrole polymer bearing pendent perylenediimide moiety. Dye. Pigment. 2015, 113, 121–128. [Google Scholar] [CrossRef]
- Soganci, T.; Soyleyici, H.C.; Giziroglu, E.; Ak, M. Processable amide substituted 2,5-bis(2-thienyl)pyrrole based conducting polymer and its fluorescent and electrochemical properties. J. Electrochem. Soc. 2016, 163, H1096–H1103. [Google Scholar] [CrossRef]
- Su, Y.-S.; Chang, J.-C.; Wu, T.-Y. Applications of three dithienylpyrroles-based electrochromic polymers in high-contrast electrochromic devices. Polymers 2017, 9, 114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koyuncu, S.; Zafer, C.; Sefer, E.; Koyuncu, F.B.; Demic, S.; Kaya, I.; Ozdemir, E.; Icli, S. A new conduction polymer of 2,5-bis(2-thienyl)-1H-(pyrrole) (SNS) containing carbazole subunit: Electrochemical, optical and electrochromic properties. Synth. Met. 2009, 159, 2012–2021. [Google Scholar]
- Lai, J.-C.; Lu, X.-R.; Qu, B.-T.; Liu, F.; Li, C.-H.; You, X.-Z. A new multicolored and near-infrared electrochromic material based on triphenylamine-containing poly(3,4-dithienylpyrrole). Org. Electron. 2014, 15, 3735–3745. [Google Scholar] [CrossRef]
- Cai, S.; Wen, H.; Wang, S.; Niu, H.; Wang, C.; Jiang, X.; Bai, X.; Wang, W. Electrochromic polymers electrochemically polymerized from 2,5-dithienylpyrrole (DTP) with different triarylamine units: Synthesis, characterization and optoelectrochemical properties. Electrochim. Acta 2017, 228, 332–342. [Google Scholar] [CrossRef] [Green Version]
- Tirkeş, S.; Mersini, J.; Öztaş, Z.; Algi, M.P.; Algi, F.; Cihaner, A. A new processable and fluorescent polydithienylpyrrole electrochrome with pyrene appendages. Electrochim. Acta 2013, 90, 295–301. [Google Scholar]














| Polymers | Structures | UV-Vis Absorption (nm) a | Oxidation Potential (V) b | Optical Bandgap (Eg) (eV) c | HOMO (eV) d | LUMO (eV) d | |||
|---|---|---|---|---|---|---|---|---|---|
| λmax | λonset | Eonset | E1/2Ox1 | E1/2Ox2 | |||||
| P1 |  | 350 | 569 | 0.48 | 0.68 | – | 2.18 | −4.84 | −2.66 |
| P2 |  | 329 | 547 | 0.47 | 0.59 | 1.15 | 2.27 | −4.83 | −2.56 |
| Polymers | λmax a (nm) | Δ%T | Response Time b | ΔOD c | Qd d (mC/cm2) | CE e (cm2/C) | |
|---|---|---|---|---|---|---|---|
| tc (s) | tb (s) | ||||||
| P1 | 1000 | 32 | 3.3 | 1.0 | 0.30 | 2.76 | 108 |
| 460 | 16 | 1.3 | 2.9 | 0.17 | 2.76 | 62 | |
| P2 | 900 | 58 | 4.7 | 4.6 | 0.46 | 2.05 | 224 |
| 450 | 28 | 5.6 | 3.7 | 0.24 | 2.05 | 117 | |
| 650 | 45 | 8.1 | 15.3 | 0.59 | 9.09 | 65 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kung, Y.-R.; Cao, S.-Y.; Hsiao, S.-H. Electrosynthesis and Electrochromism of a New Crosslinked Polydithienylpyrrole with Diphenylpyrenylamine Subunits. Polymers 2020, 12, 2777. https://doi.org/10.3390/polym12122777
Kung Y-R, Cao S-Y, Hsiao S-H. Electrosynthesis and Electrochromism of a New Crosslinked Polydithienylpyrrole with Diphenylpyrenylamine Subunits. Polymers. 2020; 12(12):2777. https://doi.org/10.3390/polym12122777
Chicago/Turabian StyleKung, Yu-Ruei, Sin-Yun Cao, and Sheng-Huei Hsiao. 2020. "Electrosynthesis and Electrochromism of a New Crosslinked Polydithienylpyrrole with Diphenylpyrenylamine Subunits" Polymers 12, no. 12: 2777. https://doi.org/10.3390/polym12122777
APA StyleKung, Y.-R., Cao, S.-Y., & Hsiao, S.-H. (2020). Electrosynthesis and Electrochromism of a New Crosslinked Polydithienylpyrrole with Diphenylpyrenylamine Subunits. Polymers, 12(12), 2777. https://doi.org/10.3390/polym12122777







