Investigations of the Optical and Thermal Properties of the Pyrazoloquinoline Derivatives and Their Application for OLED Design
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Quantum Chemical Calculations
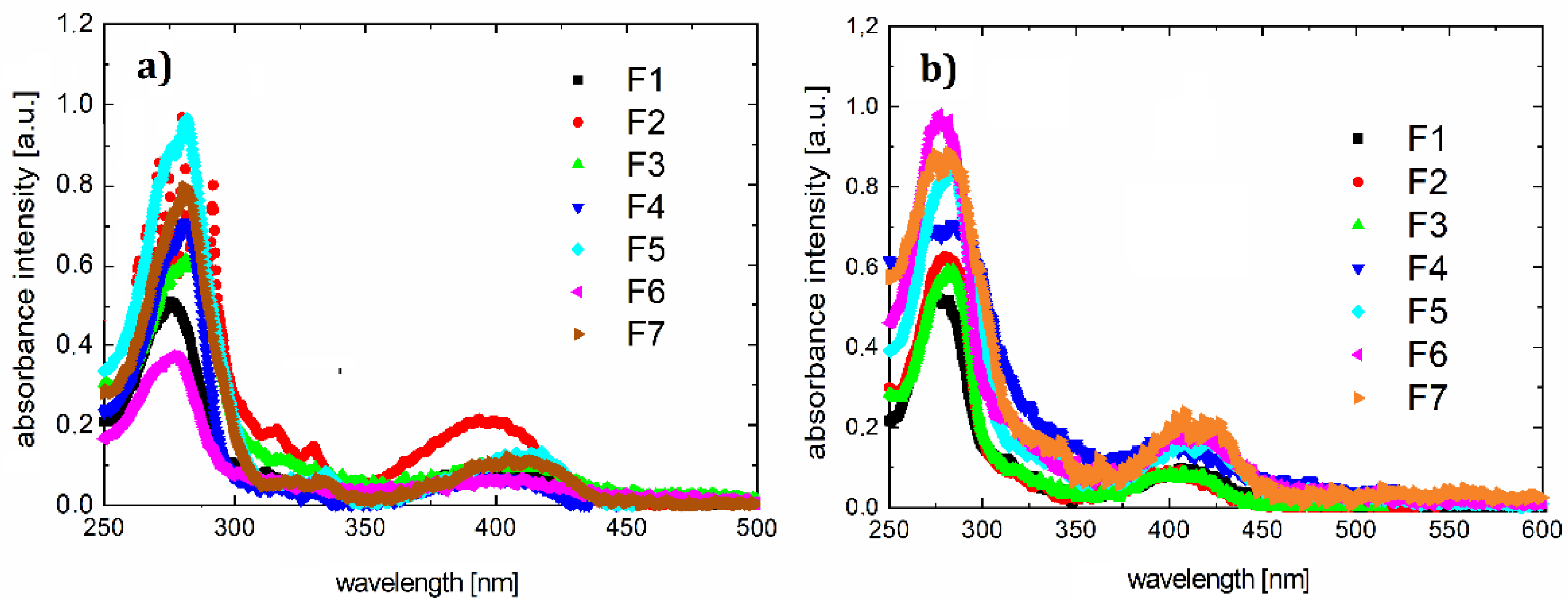
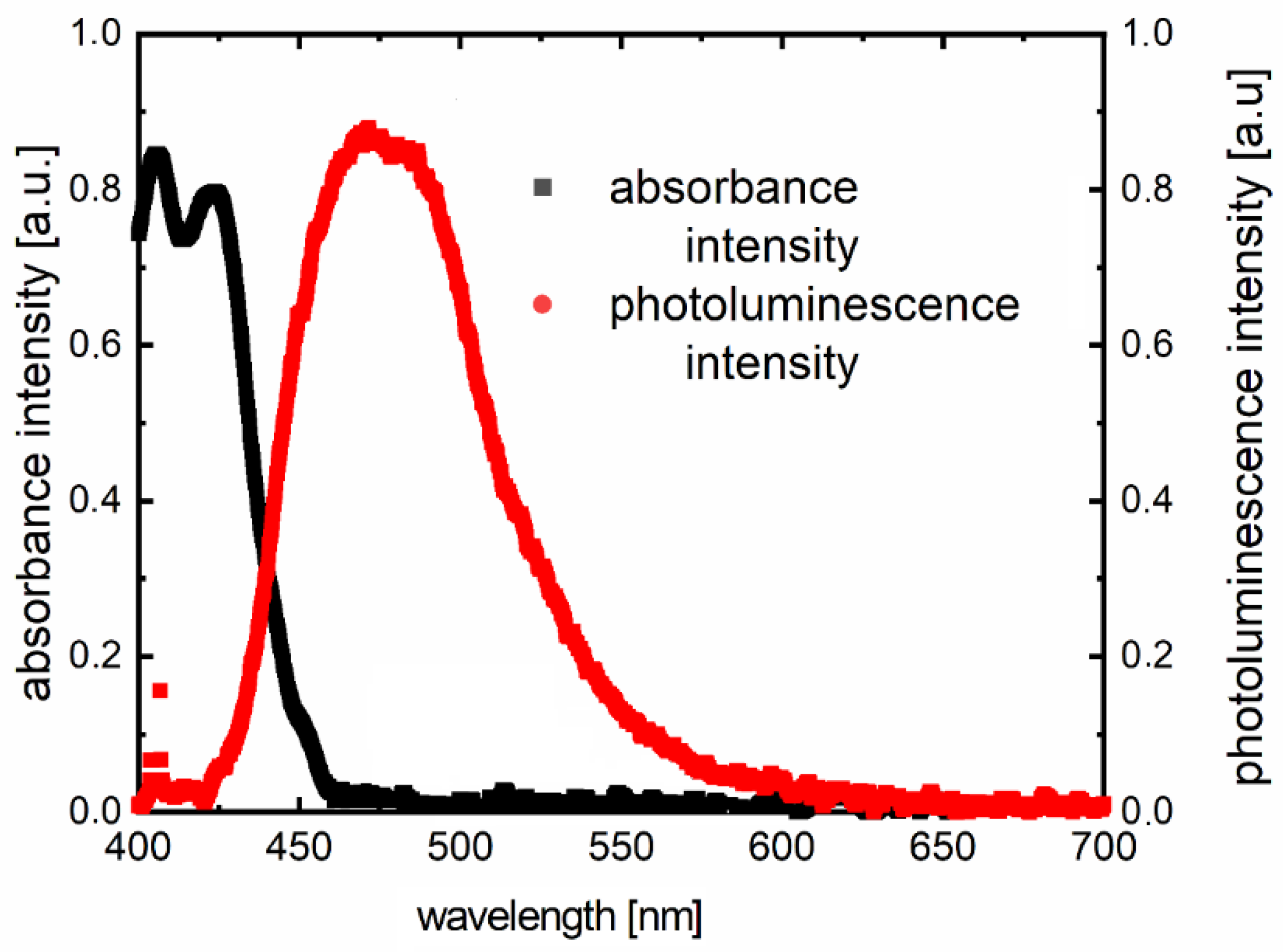
3.2. Optical Properties
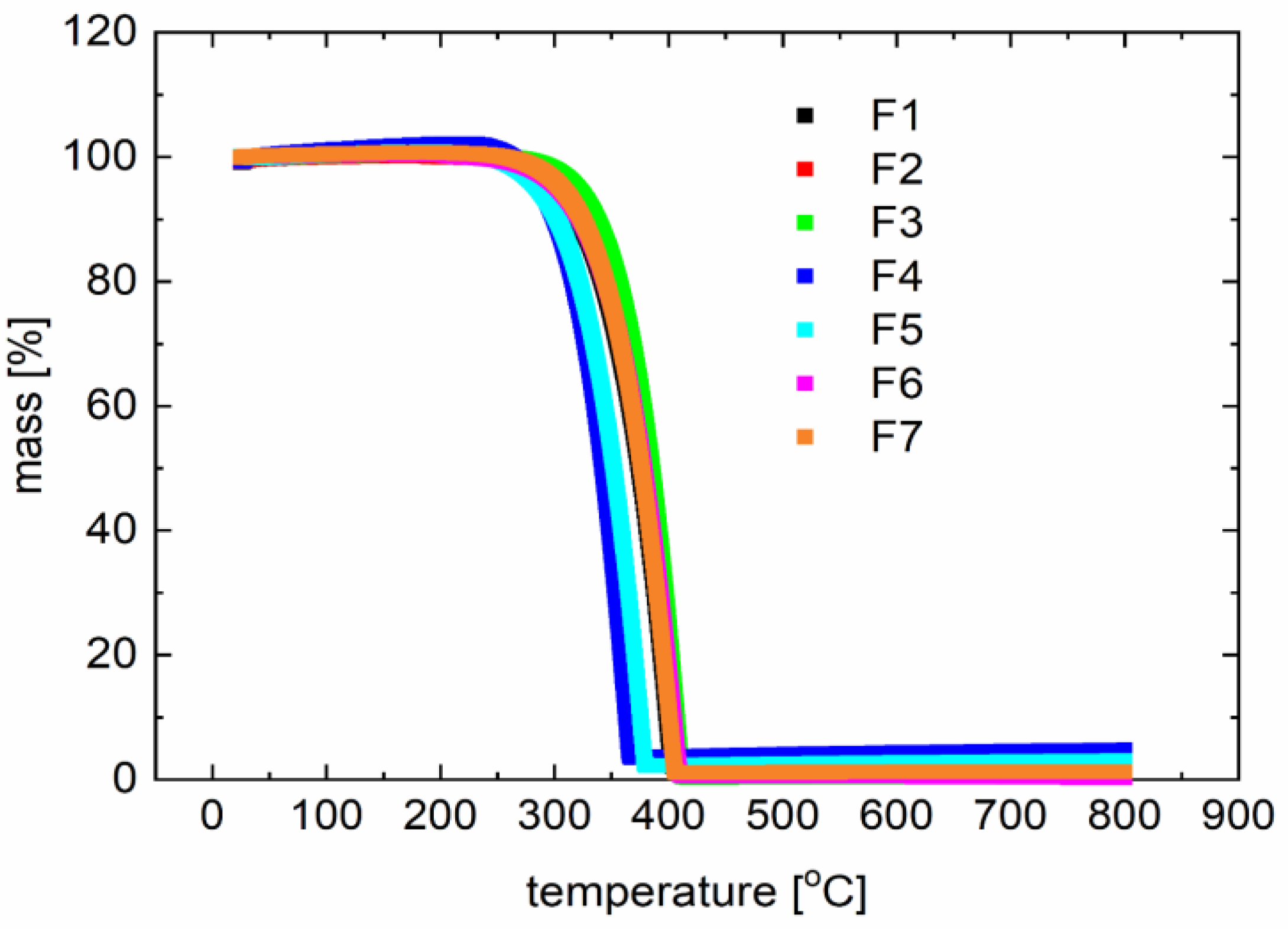
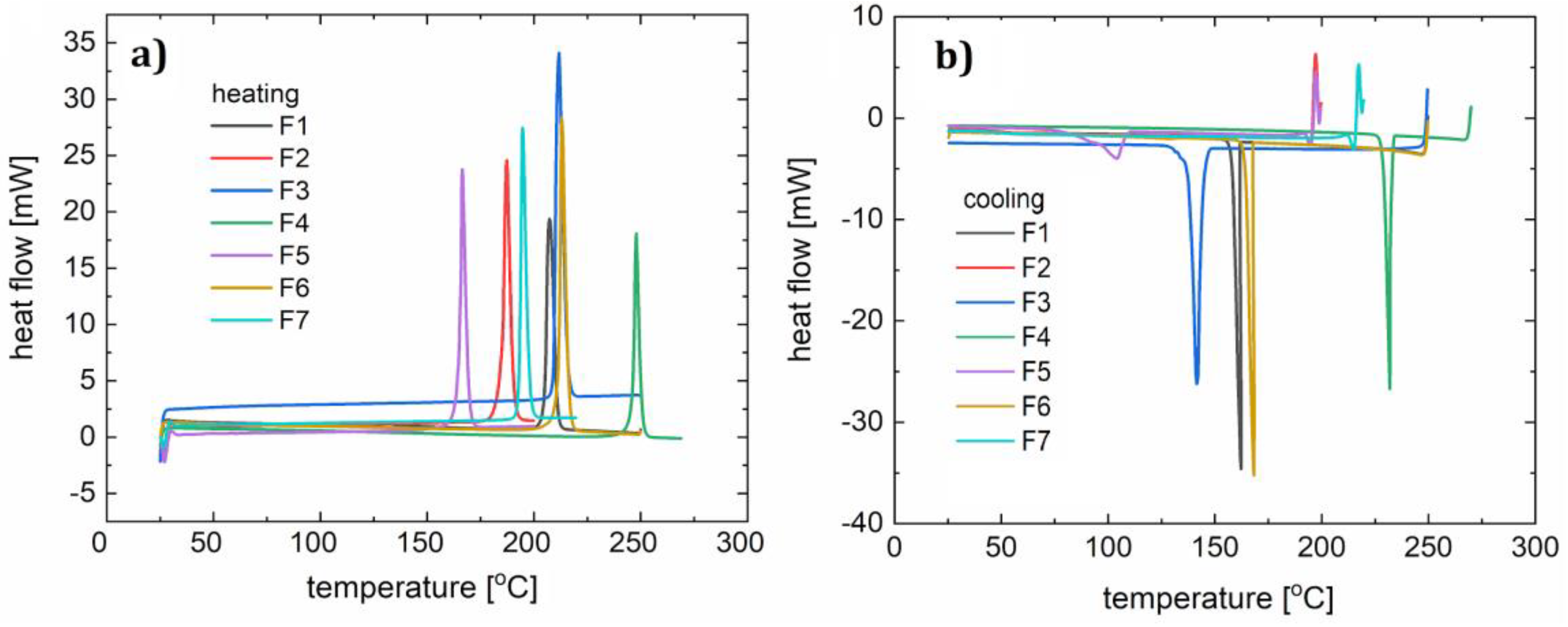
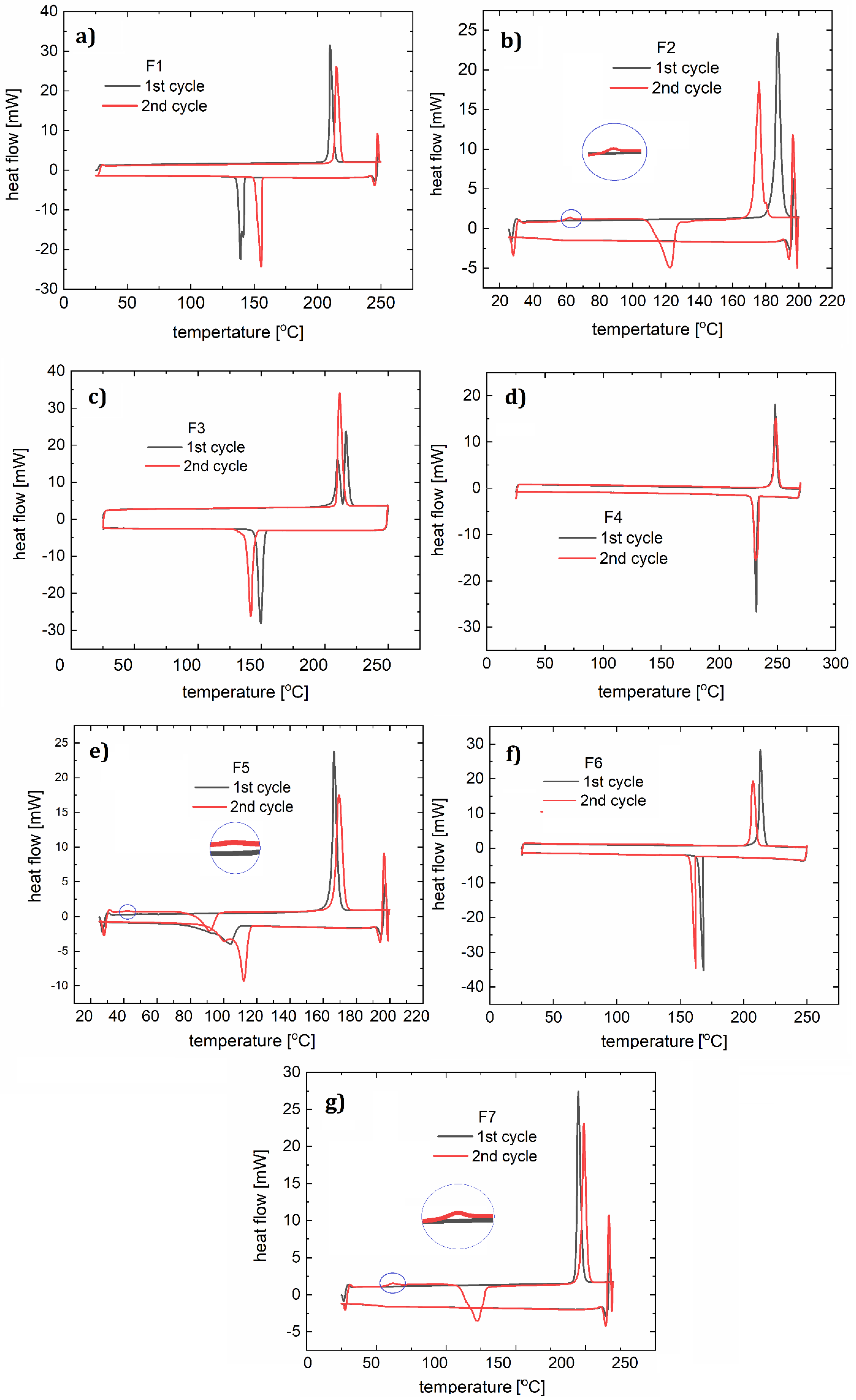
3.3. Thermogravimetry (TG) and Differential Scanning Calorimetry (DSC) Study
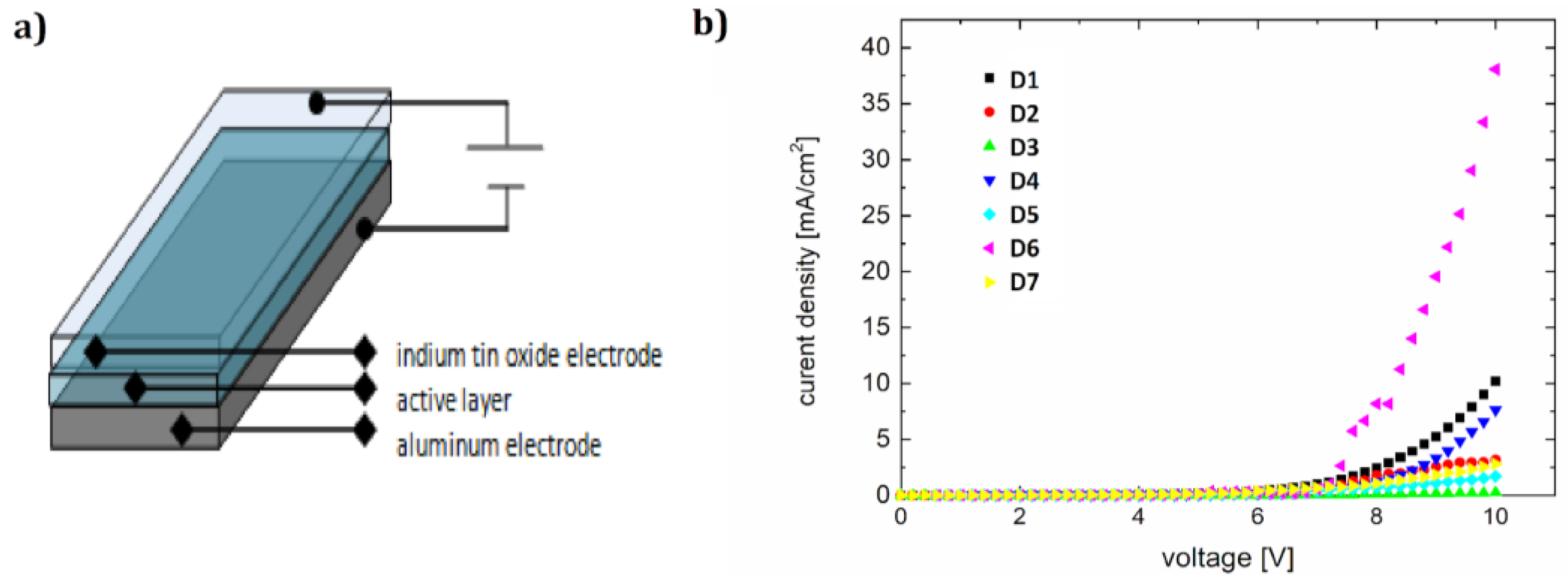
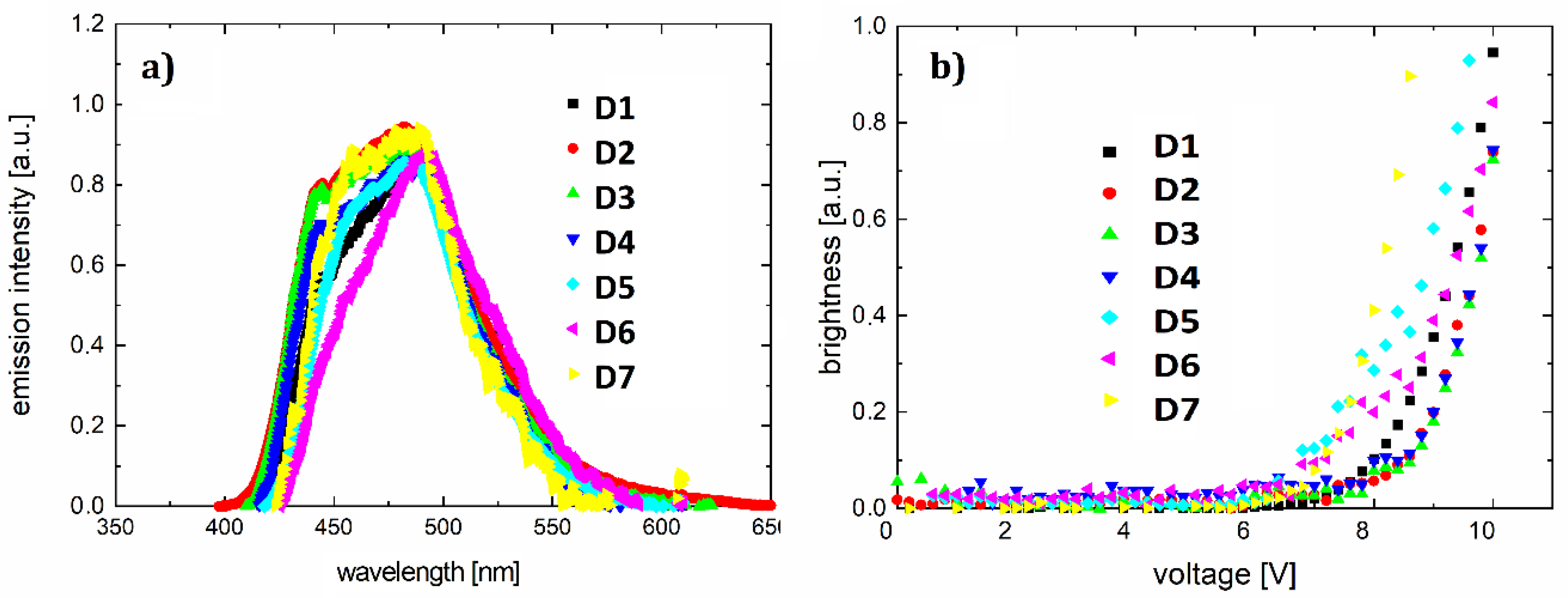
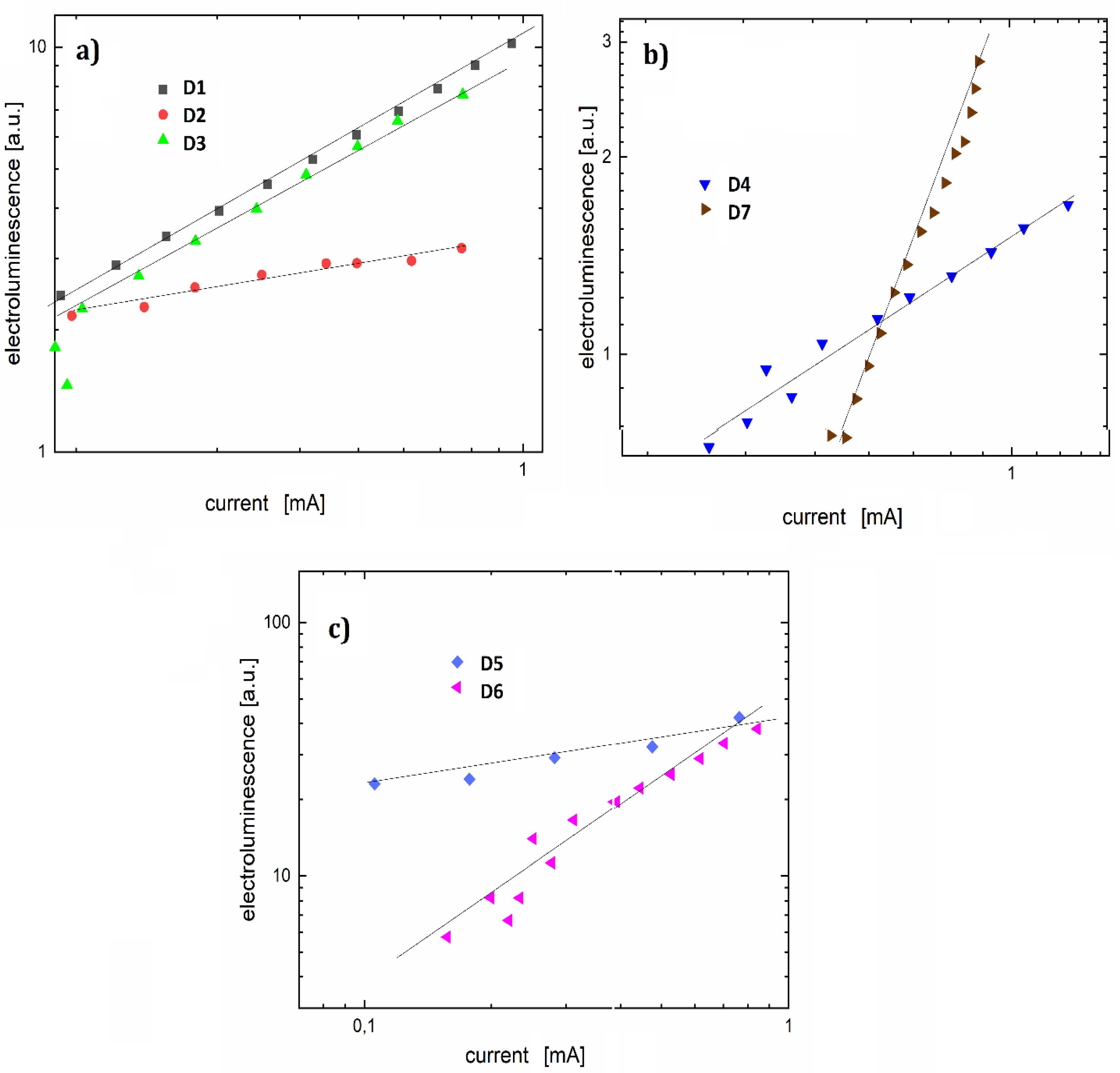
3.4. OLEDs
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tang, C.W.; Vanslyke, S.A. Organic electroluminescent diodes. Appl. Phys. Lett. 1987, 51, 913–915. [Google Scholar] [CrossRef]
- Tang, C.W.; Vanslyke, S.A.; Chen, C.H. Electroluminescence of doped organic thin films. J. Appl. Phys. 1989, 65, 3610–3616. [Google Scholar] [CrossRef]
- Grabka, D.; Danel, A.; Kolbus, A.; Szary, K. Photophysical properties of 6-N,N-dimethylpyrazolo[3,4-b]quinoline substituted with pyridyl in the 3-position. Opt. Mater. 2017, 66, 527–533. [Google Scholar] [CrossRef]
- Slodek, A.; Zych, D.; Maroń, A.; Golba, S.; Schab-Balcerzak, E.; Janeczek, H.; Siwy, M.; Maćkowski, S. Fluorene vs. carbazole substituent at quinoline core toward organic electronics. Dye. Pigment. 2019, 166, 98–106. [Google Scholar] [CrossRef]
- Thejo Kalyani, N.; Dhoble, S.J. Novel materials for fabrication and encapsulation of OLEDs. Renew. Sustain. Energy Rev. 2015, 44, 319–347. [Google Scholar] [CrossRef]
- Bizzarri, C.; Spuling, E.; Knoll, D.M.; Volz, D.; Bräse, S. Sustainable metal complexes for organic light-emitting diodes (OLEDs). Coord. Chem. Rev. 2018, 373, 49–82. [Google Scholar] [CrossRef]
- Ledwon, P. Recent advances of donor-acceptor type carbazole-based molecules for light emitting applications. Org. Electron. 2019, 75, 105422. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, H.; Zhang, B.; Xie, Z.; Wong, W.Y. Towards high-power-efficiency solution-processed OLEDs: Material and device perspectives. Mater. Sci. Eng. R Rep. 2020, 140, 100547. [Google Scholar] [CrossRef]
- Mahesh, K.; Karpagam, S.; Goubard, F. Conductive and photoactive nature of conjugated polymer based on thiophene functionalized thiazole or benzothiadiazole. Express Polym. Lett. 2018, 12, 238–255. [Google Scholar] [CrossRef]
- Li, J.; Qiao, B.; Zhao, S.; Song, D.; Zhang, C.; Xu, Z. Investigation on OLEDs efficiency roll-off with interfacial charge storage and their time-resolved emission spectra. Org. Electron. 2020, 83, 105756. [Google Scholar] [CrossRef]
- Steudel, S.; Myny, K.; Schols, S.; Vicca, P.; Smout, S.; Tripathi, A.; Van Der Putten, B.; Van Der Steen, J.L.; Van Neer, M.; Schütze, F.; et al. Design and realization of a flexible QQVGA AMOLED display with organic TFTs. Org. Electron. 2012, 13, 1729–1735. [Google Scholar] [CrossRef]
- Jia, Y.; Liu, Z.; Wu, D.; Chen, J.; Meng, H. Mechanical simulation of foldable AMOLED panel with a module structure. Org. Electron. 2019, 65, 185–192. [Google Scholar] [CrossRef]
- Tokito, S. High-Performance Organic Thin-Film Transistors for Flexible AM-OLED Displays. In ECS Transactions; The Electrochemical Society: Pennington, NJ, USA, 2008; Volume 16, pp. 23–31. [Google Scholar]
- Tsai, T.C.; Hung, W.Y.; Chi, L.C.; Wong, K.T.; Hsieh, C.C.; Chou, P.T. A new ambipolar blue emitter for NTSC standard blue organic light-emitting device. Org. Electron. 2009, 10, 158–162. [Google Scholar] [CrossRef]
- Yin, M.; Pan, T.; Yu, Z.; Peng, X.; Zhang, X.; Xie, W.; Liu, S.; Zhang, L. Color-stable WRGB emission from blue OLEDs with quantum dots-based patterned down-conversion layer. Org. Electron. 2018, 62, 407–411. [Google Scholar] [CrossRef]
- Liu, H.; Bala, H.; Zhang, B.; Zong, B.; Huang, L.; Fu, W.; Sun, G.; Cao, J.; Zhan, Z. Thickness-dependent photovoltaic performance of TiO2 blocking layer for perovskite solar cells. J. Alloys Compd. 2018, 736, 87–92. [Google Scholar] [CrossRef]
- Rosales-Gallegos, I.; Ávila-Niño, J.A.; Hérnandez-Arriaga, D.; Reyes-Reyes, M.; López-Sandoval, R. Flexible rewritable organic memory devices using nitrogen-doped CNTs/PEDOT: PSS composites. Org. Electron. 2017, 45, 159–168. [Google Scholar] [CrossRef]
- Jiang, C.; Huang, X.; Sun, B.; Li, Y.; Gao, M.; Ye, L.; Ade, H.; Forrest, S.R.; Fan, J. A 3D nonfullerene electron acceptor with a 9,9′-bicarbazole backbone for high-efficiency organic solar cells. Org. Electron. 2020, 84, 105784. [Google Scholar] [CrossRef]
- Fernandes, S.S.M.; Raposo, M.M.M.; Mesquita, I.; Andrade, L.; Mendes, A.; Justino, L.L.G.; Burrows, H.D. Synthesis and characterization of push-pull bithiophene and thieno[3,2-b]thiophene derivatives bearing an ethyne linker as sensitizers for dye-sensitized solar cells. Org. Electron. 2017, 49, 194–205. [Google Scholar] [CrossRef]
- Wang, P.H.; Lee, H.F.; Huang, Y.C.; Jung, Y.J.; Gong, F.L.; Huang, W.Y. The proton dissociation constant of additive effect on self-assembly of poly(3-hexyl-thiophene) for organic solar cells. Electron. Mater. Lett. 2014, 10, 767–773. [Google Scholar] [CrossRef]
- Wang, L.; Hu, M.; Zhang, Y.; Yuan, Z.; Hu, Y.; Zhao, X.; Chen, Y. Single-strand and ladder-type polymeric acceptors based on regioisomerically-pure perylene diimides towards all-polymer solar cells. Polymer 2019, 162, 108–115. [Google Scholar] [CrossRef]
- Feng, L.; Xu, X.; Guo, X. Comparative Analysis of Organic Thin Film Transistor Structures for Flexible E-Paper and AMOLED Displays. In ECS Transactions; ECS: Bristol, UK, 2011; Volume 37, pp. 105–112. [Google Scholar]
- Wang, Z.; He, M.; Chen, B.; Hu, B. Azo-linked porous organic polymers/polydimethylsiloxane coated stir bar for extraction of benzotriazole ultraviolet absorbers from environmental water and soil samples followed by high performance liquid chromatography-diode array detection. J. Chromatogr. A 2020, 1616, 460793. [Google Scholar] [CrossRef]
- Li, F.; Chen, W.; Yuan, K.; Chen, Y. Photovoltaic performance enhancement in P3HT/ZnO hybrid bulk-heterojunction solar cells induced by semiconducting liquid crystal ligands. Org. Electron. 2012, 13, 2757–2762. [Google Scholar] [CrossRef]
- Xu, X.; Sun, L.; Shen, K.; Zhang, S. Organic and hybrid organic-inorganic flexible optoelectronics: Recent advances and perspectives. Synth. Met. 2019, 256, 116137. [Google Scholar] [CrossRef]
- An, Q.; Zhang, F.; Sun, Q.; Zhang, M.; Zhang, J.; Tang, W.; Yin, X.; Deng, Z. Efficient organic ternary solar cells with the third component as energy acceptor. Nano Energy 2016, 26, 180–191. [Google Scholar] [CrossRef]
- Kwong, C.Y.; Djurišić, A.B.; Choy, W.C.H.; Li, D.; Xie, M.H.; Chan, W.K.; Cheah, K.W.; Lai, P.T.; Chui, P.C. Efficiency and stability of different tris(8-hydroxyquinoline) aluminium (Alq3) derivatives in OLED applications. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2005, 116, 75–81. [Google Scholar] [CrossRef]
- Xiao, P.; Dong, T.; Xie, J.; Luo, D.; Yuan, J.; Liu, B. Emergence of White Organic Light-Emitting Diodes Based on Thermally Activated Delayed Fluorescence. Appl. Sci. 2018, 8, 299. [Google Scholar] [CrossRef]
- Ciric-Marjanovic, G. Recent advances in polyaniline research: Polymerization mechanisms, structural aspects, properties and applications. Synth. Met. 2013, 177, 1–47. [Google Scholar] [CrossRef]
- Khanna, V.K. Displays and light-emission devices. In Flexible Electronics; Institute of Physics Publishing: Bristol, UK, 2019; Volume 3, pp. 1–62. ISBN 978-0-7503-2456-4. [Google Scholar]
- Mekheimer, R.A.; Ahmed, E.A.; Sadek, K.U. Recent developments in the chemistry of pyrazolo[4,3-c]quinolines. Tetrahedron 2012, 68, 1637–1667. [Google Scholar] [CrossRef]
- Wainwright, M.; Kristiansen, J.E. Quinoline and cyanine dyes—Putative anti-MRSA drugs. Int. J. Antimicrob. Agents 2003, 22, 479–486. [Google Scholar] [CrossRef]
- Stephens, D.E.; Larionov, O.V. Recent advances in the C-H-functionalization of the distal positions in pyridines and quinolines. Tetrahedron 2015, 71, 8683–8716. [Google Scholar] [CrossRef]
- Elie, M.; Renaud, J.L.; Gaillard, S. N-Heterocyclic carbene transition metal complexes in light emitting devices. Polyhedron 2018, 140, 158–168. [Google Scholar] [CrossRef]
- Hung, L.S.; Chen, C.H. Recent progress of molecular organic electroluminescent materials and devices. Mater. Sci. Eng. R Rep. 2002, 39, 143–222. [Google Scholar] [CrossRef]
- Li, A.; Luo, C.; Liu, Y.; Li, L.; Lin, Y.; Liu, K.; Zhou, C. Zn-promoted Hβ zeolite for gas-phase catalyzed aza-heterocyclic-aromatization of acrolein dimethyl acetal and aniline to quinolines. Mol. Catal. 2020, 486, 110833. [Google Scholar] [CrossRef]
- Alemi Tameh, F.; Safaei-Ghomi, J. Synthesis of spiro[pyrazoloquinoline-oxindoles] and spiro[chromenopyrazolo-oxindoles] promoted by guanidine-functionalized magnetic Fe3O4 nanoparticles. J. Iran. Chem. Soc. 2018, 15, 1633–1637. [Google Scholar] [CrossRef]
- Gu, Z.; Guo, J.; Hao, R.; Lin, Z.; Qian, Y.; Ma, C.; Fan, Y.; Deng, X.; Zhang, G.; Peng, W.; et al. An efficient strategy to supervise absorption, mobility, morphology of photovoltaic molecule by inserting a D-A unit. Dye. Pigment. 2019, 166, 515–522. [Google Scholar] [CrossRef]
- Matuszek, M.; Filapek, M.; Gancarz, P.; Krompiec, S.; Grzegorz Małecki, J.; Kotowicz, S.; Siwy, M.; Maćkowski, S.; Chrobok, A.; Schab-Balcerzak, E.; et al. Synthesis and photophysical properties of new perylene bisimide derivatives for application as emitting materials in OLEDs. Dye. Pigment. 2018, 159, 590–599. [Google Scholar] [CrossRef]
- Ulahannan, R.T.; Panicker, C.Y.; Varghese, H.T.; Musiol, R.; Jampilek, J.; Alsenoy, C.V.; War, J.A.; Manojkumar, T.K. Vibrational spectroscopic studies and molecular docking study of 2-[(E)-2-phenylethenyl]quinoline-5-carboxylic acid. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 150, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Szlachcic, P.; Uchacz, T. Influence of fluorine on the photophysical, electrochemical properties and basicity of 1,3-diphenylpyrazolo[3,4-b]quinoline derivatives. J. Lumin. 2018, 194, 579–587. [Google Scholar] [CrossRef]
- Rechthaler, K.; Rotkiewicz, K.; Danel, A.; Tomasik, P.; Khatchatryan, K.; Köhler, G. Emissive Properties and Intramolecular Charge Transfer of Pyrazoloquinoline Derivatives. J. Fluoresc. 1997, 7, 301–309. [Google Scholar] [CrossRef]
- Hu, Y.Q.; Gao, C.; Zhang, S.; Xu, L.; Xu, Z.; Feng, L.S.; Wu, X.; Zhao, F. Quinoline hybrids and their antiplasmodial and antimalarial activities. Eur. J. Med. Chem. 2017, 139, 22–47. [Google Scholar] [CrossRef]
- Chen, N.; Zhang, C.; Dong, X.; Liu, Y.; Sun, Y. Activation and stabilization of lipase by grafting copolymer of hydrophobic and zwitterionic monomers onto the enzyme. Biochem. Eng. J. 2020, 158, 107557. [Google Scholar] [CrossRef]
- Ghate, M.; Dahule, H.K.; Kalyani, N.T.; Dhoble, S.J. Deep blue light emitting Cyno-DPQ phosphor with large stokes shift and high thermal stability for OLEDs and display applications. Optik 2017, 149, 198–205. [Google Scholar] [CrossRef]
- Luszczynska, B.; Dobruchowska, E.; Glowacki, I.; Danel, A.; Ulanski, J. Thermoluminescence of the blue light-emitting system based on poly(9-vinylcarbazole) doped with a pyrazoloquinoline dye. J. Lumin. 2009, 129, 1215–1218. [Google Scholar] [CrossRef]
- Hosseinnezhad, M.; Rouhani, S.; Gharanjig, K. Extraction and application of natural pigments for fabrication of green dye-sensitized solar cells. Opto-Electron. Rev. 2018, 26, 165–171. [Google Scholar] [CrossRef]
- Iwan, A.; Boharewicz, B.; Parafiniuk, K.; Tazbir, I.; Gorecki, L.; Sikora, A.; Filapek, M.; Schab-Balcerzak, E. New air-stable aromatic polyazomethines with triphenylamine or phenylenevinylene moieties towards photovoltaic application. Synth. Met. 2014, 195, 341–349. [Google Scholar] [CrossRef]
- Iwan, A. Thermotropic and opto(electrical) properties of liquid crystalline imine with two fluorinated chains. J. Mol. Liq. 2010, 157, 67–72. [Google Scholar] [CrossRef]
- Kirsch, P. Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications; Wiley Blackwell: Hoboken, NJ, USA, 2005; ISBN 9783527603930. [Google Scholar]
- Tao, Y.T.; Balasubramaniam, E.; Danel, A.; Jarosz, B.; Tomasik, P. Sharp green electroluminescence from 1H-pyrazolo[3,4-b]quinoline-based light-emitting diodes. Appl. Phys. Lett. 2000, 77, 1575–1577. [Google Scholar] [CrossRef]
- Andersson, T.G.; Andreasson, M.; Klement, U.; Lee, C.B.; Pujilaksono, B.; Uddin, A. Preparation and characterization of the ITO surface and the Al/Alq3/ITO heterostructure for OLEDs. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2007, 145, 48–56. [Google Scholar] [CrossRef]
- Nalwa, H.S.; Rohwer, L.S. Handbook of Luminescence, Display Materials, and Devices; American Scientific Publishers: Valencia, CA, USA, 2003; ISBN 9781588830104. [Google Scholar]
- Liu, B.; Altintas, Y.; Wang, L.; Shendre, S.; Sharma, M.; Sun, H.; Mutlugun, E.; Demir, H.V. Light-Emitting Diodes: Record High External Quantum Efficiency of 19.2% Achieved in Light-Emitting Diodes of Colloidal Quantum Wells Enabled by Hot-Injection Shell Growth. Adv. Mater. 2020, 32, 2070054. [Google Scholar] [CrossRef]
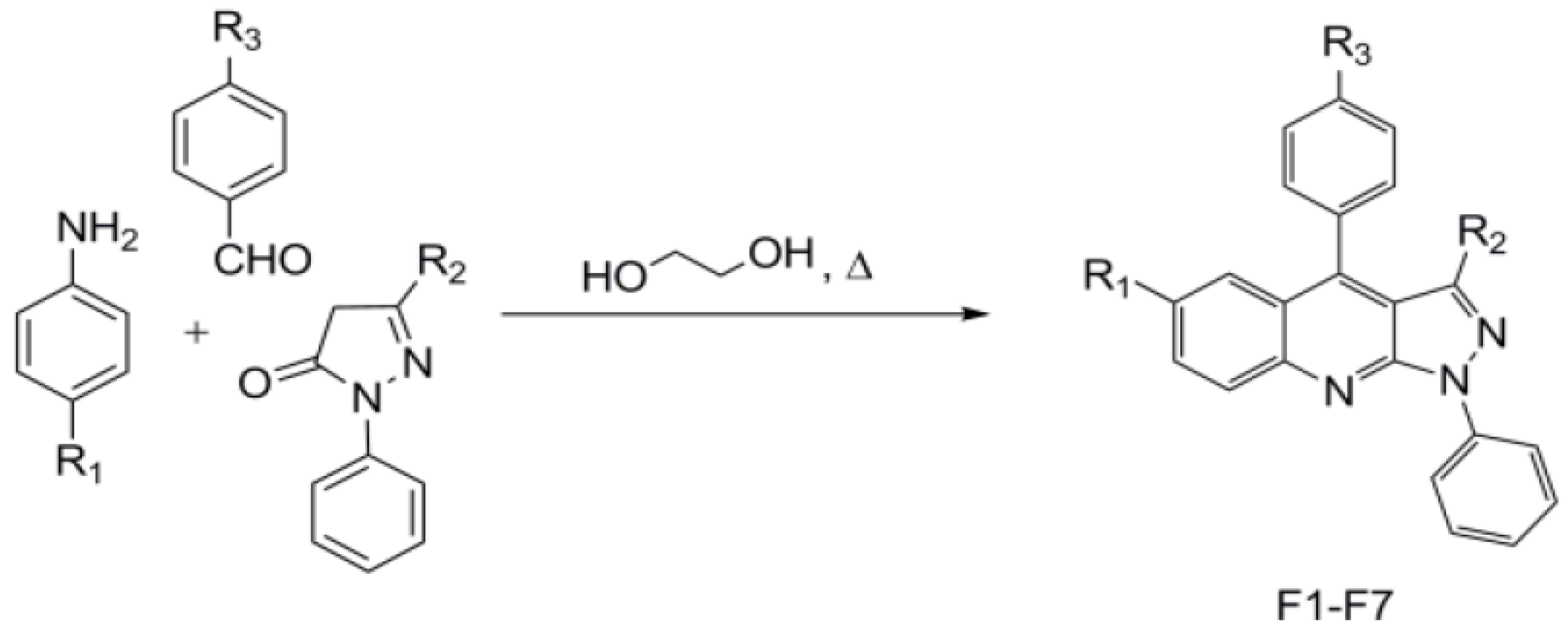

| Symbol | Compound Name | R1 | R2 | R3 |
|---|---|---|---|---|
| F1 | 4-(4-tert-butylphenyl)-6-fluoro-3-methyl-1-phenyl-1H-pyrazolo[3,4-b]quinoline | F | t-Bu | Me |
| (fluorine) | (t-butyl) | (methyl) | ||
| F2 | 6-tert-butyl-4-(4-fluorophenyl)-3-methyl-1-phenyl-1H-pyrazolo[3,4-b]quinoline | t-Bu | F | Me |
| (t-butyl) | (fluorine) | (methyl) | ||
| F3 | 6-ethyl-4-(4-fluorophenyl)-3-methyl-1-phenyl-1H-pyrazolo[3,4-b]quinoline | Et- | F | Me |
| (ethylene) | (fluorine) | (methyl) | ||
| F4 | 4-(4-fluorophenyl)-3,6-dimethyl-1-phenyl-1H-pyrazolo[3,4-b]quinoline | Me | F | Me |
| (methyl-CH3) | (fluorine) | (methyl) | ||
| F5 | 4-(4-fluorophenyl)-6-methoxy-3-methyl-1-phenyl-1H-pyrazolo[3,4-b]quinoline | OMe | F | Me |
| (O-methyl) | (fluorine) | (methyl) | ||
| F6 | 4-(4-chlorophenyl)-6-fluoro-3-methyl-1-phenyl-1H-pyrazolo[3,4-b]quinoline | F | Cl | Me |
| (fluorine) | (chlorine) | (methyl) | ||
| F7 | 4-(4-fluorophenyl)-3-isopropyl-6-methoxy-1-phenyl-1H-pyrazolo[3,4-b]quinoline | OMe | F | i-Pr |
| (O-methyl) | (fluorine) | (isopropyl) |
| Level of Theory | B3LYP/6−31G(d,p) | B3LYP/6−31+G(d,p) | B3LYP/6−31G(d) | ||||
|---|---|---|---|---|---|---|---|
| Compound symbol | R1 | HOMO | LUMO | HOMO | LUMO | HOMO | LUMO |
| F1 | F (fluorine) | 5.42 | 1.96 | 5.73 | 2.28 | 5.57 | 2.05 |
| F2 | t-Bu (t-butyl) | 5.39 | 1.86 | 5.68 | 2.16 | 5.54 | 1.94 |
| F3 | Et (ethylene) | 5.39 | 1.88 | 5.68 | 2.19 | 5.53 | 1.96 |
| F4 | Me (methyl) | 5.39 | 1.88 | 5.69 | 2.19 | 5.54 | 1.95 |
| F5 | OMe (o-methyl) | 5.31 | 1.85 | 5.61 | 2.17 | 5.44 | 1.94 |
| F6 | F (fluorine) | 5.56 | 2.14 | 5.86 | 2.45 | 5.63 | 2.14 |
| F7 | OMe (o-methyl) | 5.32 | 1.84 | 5.62 | 2.16 | 5.45 | 1.93 |
| Compound | λmax (nm) | f. Oscillator Strength |
|---|---|---|
| F1 | 416.6 | 0.103 |
| 274.6 | 1.198 | |
| F2 | 405.8 | 3.055 |
| 279.9 | 4.429 | |
| F3 | 408.7 | 0.089 |
| 279.9 | 0.756 | |
| F4 | 406.7 | 0.094 |
| 279.3 | 0.653 | |
| F5 | 415.6 | 0.103 |
| 277.9 | 0.994 | |
| F6 | 420.3 | 0.091 |
| 278.7 | 0.577 | |
| F7 | 413.6 | 0.105 |
| 276.9 | 1.016 |
| Compounds | Absorption Maxima in Absorption Band 240–340 (nm) | Absorption Maxima in Absorption Band 400–650 (nm) | Maximum of Photoluminescence Spectra (λex = 405 nm) (nm) | Stokes Shift (nm) |
|---|---|---|---|---|
| F1 | 275 | 400 | 490 | 90 |
| F2 | 275 | 402 | 472 | 70 |
| F3 | 275 | 410 | 472 | 62 |
| F4 | 280 | 410 | 470 | 60 |
| F5 | 282 | 410 | 480 | 70 |
| F6 | 280 | 410 | 490 | 80 |
| F7 | 282 | 410 | 475 | 65 |
| Compound | Speed of the DSC Process (°C/min) | DSC Heating Peak(s) (°C) | DSC Cooling Peak(s) (°C) | Capillary Method m.p. (°C) | Tdeg (°C) |
|---|---|---|---|---|---|
| F1 | 10 | 210 | 141, 136 | 218–221 | 340 |
| 15 | 214 | 156 | |||
| F2 | 12 | 187 | - | 192–193 | 438 |
| 10 | 175, 180 | - | |||
| F3 | 15 | 209, 216 | 150 | 161–162 | 350 |
| 15 | 212 | 114 | |||
| F4 | 15 | 248 | 332 | 211–212 | 306 |
| 10 | 240 | 330 | |||
| F5 | 10 | 166 | 104 | 211–212 | 317 |
| 12 | 170 | 111 | |||
| F6 | 10 | 213 | 168 | 185 | 344 |
| 15 | 207 | 161 | |||
| F7 | 10 | 192 | - | 247–249 | 341 |
| 12 | 199 | - |
| Organic Electroluminescent Diode | Diode Active Layer Dye + PVK Matrix | Wavelength of Electroluminescence Maximum (nm) | Ignition Voltage (V) | Current-Electroluminescence Exponent n |
|---|---|---|---|---|
| D1 | F1 + PVK | 487 | 7.3 | 1.8 |
| D2 | F2 + PVK | 482 | 7.9 | 4.2 |
| D3 | F3 + PVK | 481 | 8.1 | 3.4 |
| D4 | F4 + PVK | 483 | 7.3 | 1.2 |
| D5 | F5 + PVK | 482 | 6.2 | 0.8 |
| D6 | F6 + PVK | 493 | 6.4 | 1.2 |
| D7 | F7 + PVK | 487 | 7.1 | 2.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewińska, G.; Khachatryan, K.; Danel, K.S.; Danel, Z.; Sanetra, J.; Marszałek, K.W. Investigations of the Optical and Thermal Properties of the Pyrazoloquinoline Derivatives and Their Application for OLED Design. Polymers 2020, 12, 2707. https://doi.org/10.3390/polym12112707
Lewińska G, Khachatryan K, Danel KS, Danel Z, Sanetra J, Marszałek KW. Investigations of the Optical and Thermal Properties of the Pyrazoloquinoline Derivatives and Their Application for OLED Design. Polymers. 2020; 12(11):2707. https://doi.org/10.3390/polym12112707
Chicago/Turabian StyleLewińska, Gabriela, Karen Khachatryan, Krzysztof S. Danel, Zoriana Danel, Jerzy Sanetra, and Konstanty W. Marszałek. 2020. "Investigations of the Optical and Thermal Properties of the Pyrazoloquinoline Derivatives and Their Application for OLED Design" Polymers 12, no. 11: 2707. https://doi.org/10.3390/polym12112707
APA StyleLewińska, G., Khachatryan, K., Danel, K. S., Danel, Z., Sanetra, J., & Marszałek, K. W. (2020). Investigations of the Optical and Thermal Properties of the Pyrazoloquinoline Derivatives and Their Application for OLED Design. Polymers, 12(11), 2707. https://doi.org/10.3390/polym12112707








