Lignin-Based Nanoparticles: A Review on Their Preparations and Applications
Abstract
1. Introduction
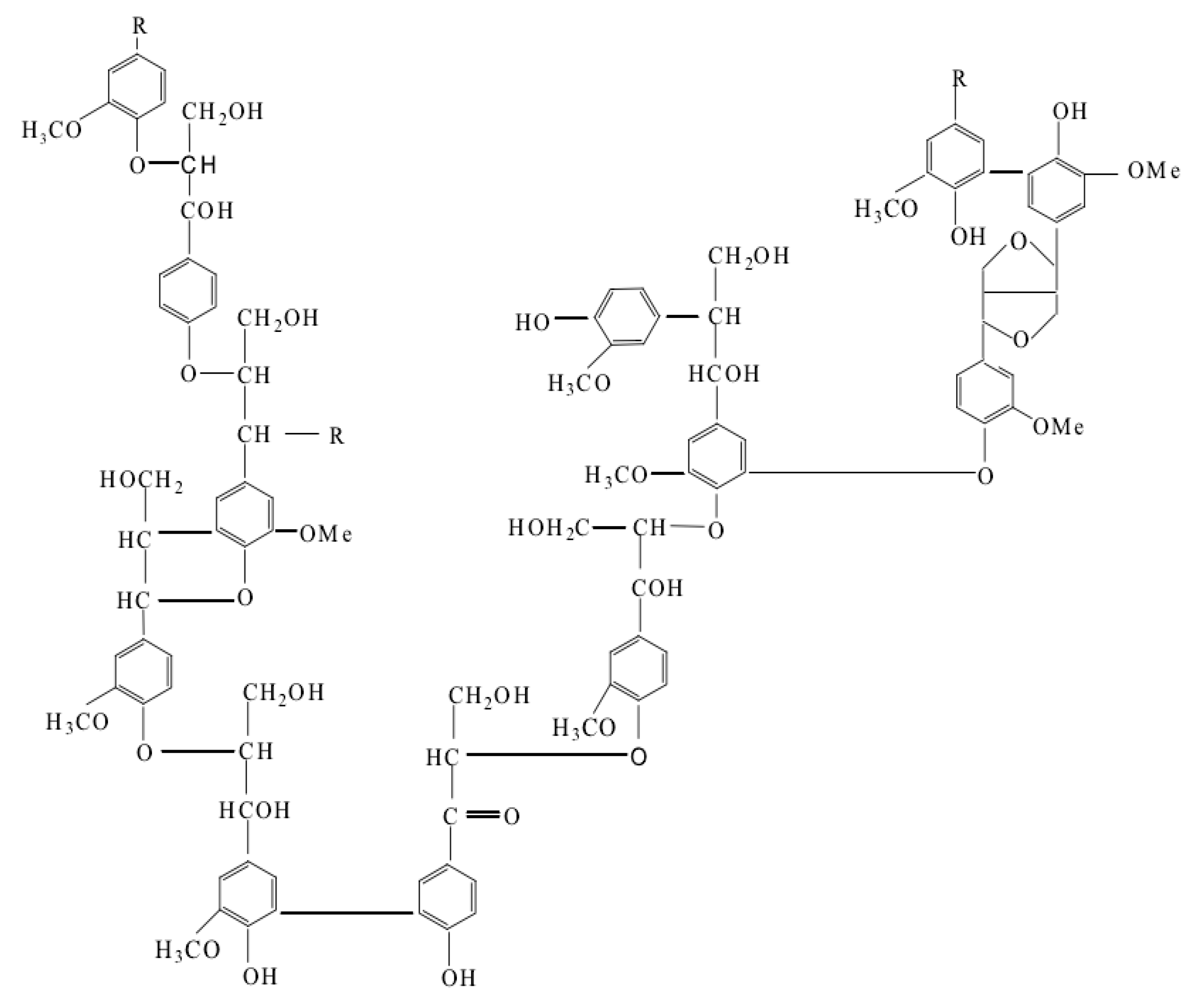
2. Main Types of Lignin
2.1. Lignosulfonate
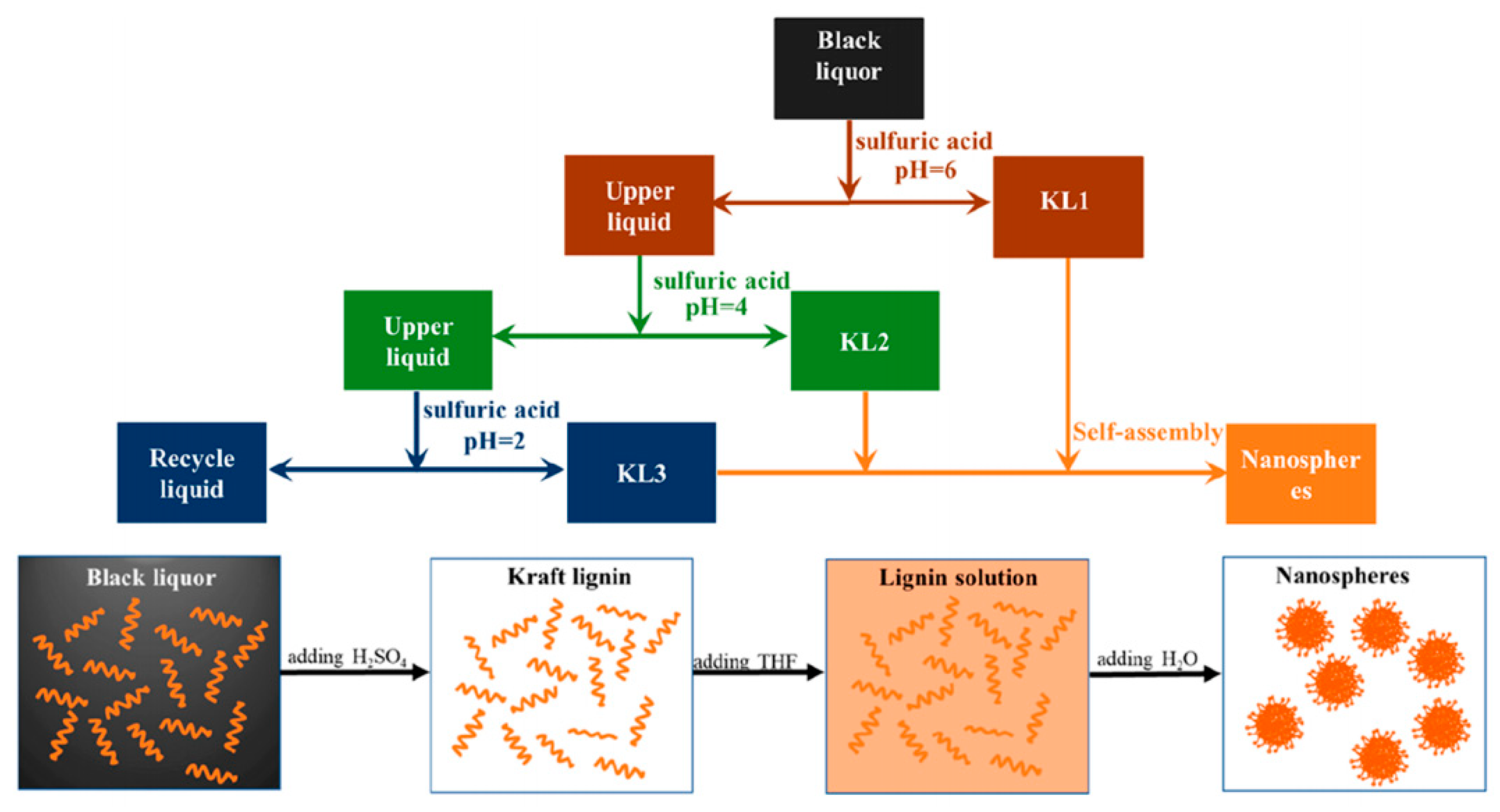
2.2. Kraft Lignin
2.3. Organosolv Lignin
2.4. Soda Lignin
2.5. Enzymatic Hydrolysis Lignin
3. Different Preparations Methods for Lignin-Based Nanoparticles
3.1. Self-assembly Method
3.2. Solvent Exchange Method
3.3. Acid Precipitation Method
3.4. Polymerization Method
3.5. Ultrasonication Method
3.6. Crosslinking Method
3.7. CO2 Antisolvent Method
4. Industrial Application of Lignin-Based Nanoparticles
4.1. Drug Delivery
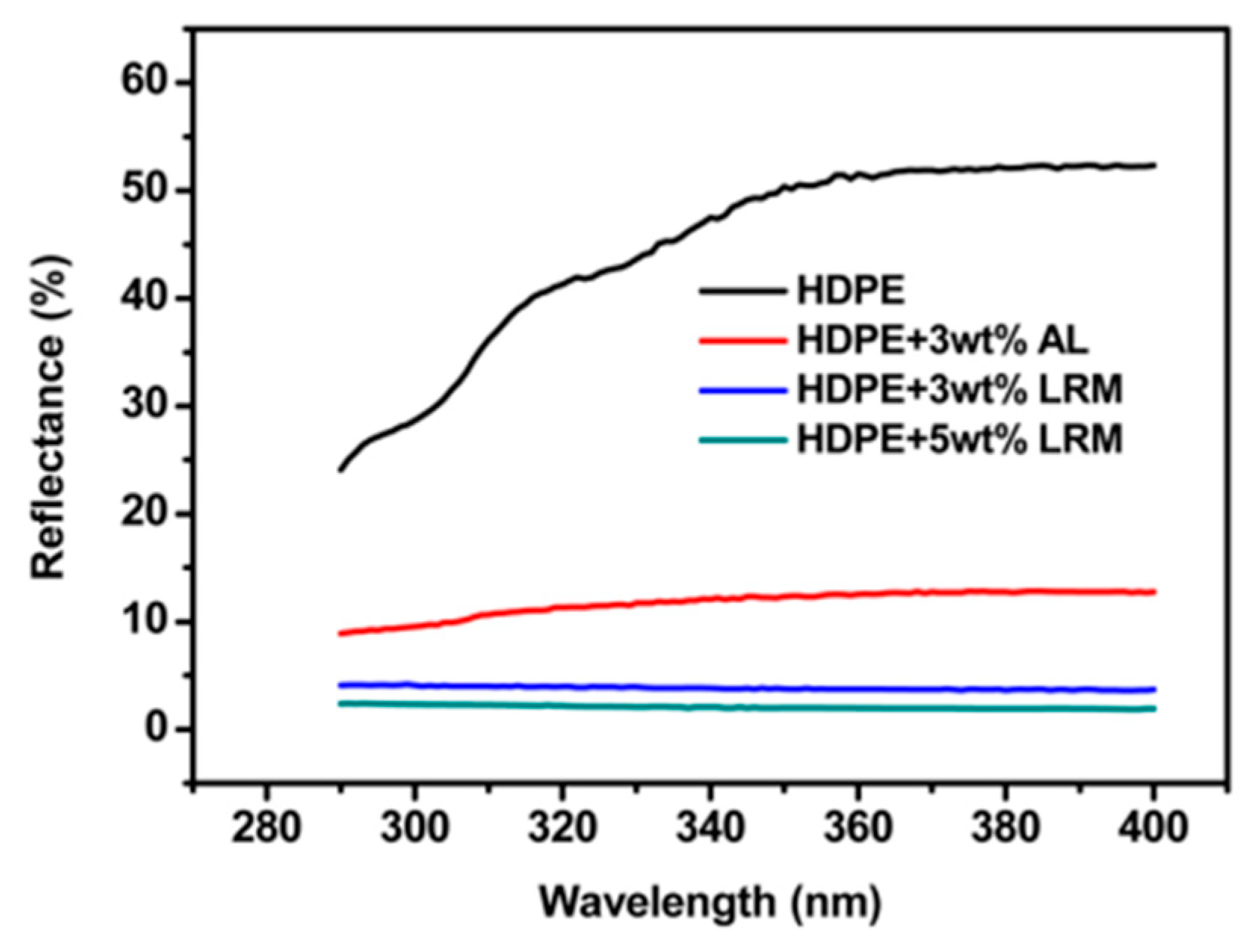
4.2. UV Absorbents
4.3. Hybrid Nanocomposites
4.4. Antioxidant Agents
4.5. Other Applications
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liao, J.J.; Latif, N.H.A.; Trache, D.; Brosse, N.; Hussin, M.H. Current advancement on the isolation, characterization and application of lignin. Int. J. Biol. Macromol. 2020, 162, 985–1024. [Google Scholar] [CrossRef]
- Cao, L.C.; Yu, I.K.M.; Liu, Y.Y.; Ruan, X.X.; Tsang, D.C.W.; Hunt, A.J.; Sik Ok, Y.; Song, H.; Zhang, S.C. Lignin valorization for the production of renewable chemicals: State-of-the-art review and future prospects. Bioresour. Technol. 2018, 269, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Sagues, W.J.; Bao, H.X.; Nemenyi, J.L.; Tong, Z.H. Lignin-first approach to biorefining: Utilizing Fenton’s reagent and supercritical ethanol for the production of phenolics and sugars. ACS Sustain. Chem. Eng. 2018, 6, 4958–4965. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K.; Raghavan, P.; Kessler, M.R. Progress in green polymer composites from lignin for multifunctional applications: A review. ACS Sustain. Chem. Eng. 2014, 2, 1072–1092. [Google Scholar] [CrossRef]
- Figueiredo, P.; Lintinen, K.; Hirvonen, J.T.; Kostiainen, M.A.; Santos, H.A. Properties and chemical modifications of lignin: Towards lignin-based nanomaterials for biomedical applications. Prog. Mater. Sci. 2018, 93, 233–269. [Google Scholar] [CrossRef]
- Ragauskas, A.J.; Beckham, G.T.; Biddy, M.J.; Chandra, R.; Chen, F.; Davis, M.F.; Davison, B.H.; Dixon, R.A.; Gilna, P.; Keller, M.; et al. Lignin valorization: Improving lignin processing in the biorefinery. Science 2014, 344, 1246843. [Google Scholar] [CrossRef]
- Tang, Q.Q.; Zhou, M.S.; Li, Y.X.; Qiu, X.Q.; Yang, D.J. Formation of uniform colloidal spheres based on lignosulfonate, a renewable biomass resource recovered from pulping spent liquor. ACS Sustain. Chem. Eng. 2018, 6, 1379–1386. [Google Scholar] [CrossRef]
- Tang, Q.Q.; Zhou, M.S.; Qiu, X.Q.; Yang, D.J. Effects of cationic cetyltrimethylammonium bromide on aggregation behaviors of sodium lignosulfonate (NaLS) in concentrated solutions and preparation of uniform lignosulfonate-based colloidal spheres. J. Agric. Food Chem. 2020. [Google Scholar] [CrossRef]
- Haq, I.; Mazumder, P.; Kalamdhad, A.S. Recent advances in removal of lignin from paper industry wastewater and its industrial applications-A review. Bioresour. Technol. 2020, 312, 123636. [Google Scholar] [CrossRef]
- Rico-García, D.; Ruiz-Rubio, L.; Pérez-Alvarez, L.; Hernández-Olmos, S.L.; Guerrero-Ramírez, G.L.; Vilas-Vilela, J.L. Lignin-based hydrogels: Synthesis and applications. Polymers 2020, 12, 81. [Google Scholar] [CrossRef]
- Wang, J.Y.; Deng, Y.H.; Qian, Y.; Qiu, X.Q.; Ren, Y.; Yang, D.J. Reduction of lignin color via one-step UV irradiation. Green Chem. 2016, 18, 695–699. [Google Scholar] [CrossRef]
- Gosselink, R.J.A.; Abächerli, A.; Semke, H.; Malherbe, R.; Käuper, P.; Nadif, A.; Van Dam, J.E.G. Analytical protocols for characterisation of sulphur-free lignin. Ind. Crop. Prod. 2004, 19, 271–281. [Google Scholar] [CrossRef]
- Calvo-Flores, F.G.; Dobado, J.A. Lignin as renewable raw material. ChemSusChem 2010, 3, 1227–1235. [Google Scholar] [CrossRef]
- Chatel, G.; Rogers, R.D. Review: Oxidation of lignin using ionic liquids-an innovative strategy to produce renewable chemicals. ACS Sustain. Chem. Eng. 2014, 2, 322–339. [Google Scholar] [CrossRef]
- Sadeghifar, H.; Ragauskas, A. Lignin as a UV light blocker-A review. Polymer 2020, 12, 1134. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D. Lignin nanocomposites. J. Macromol. Sci. A 2016, 53, 382–387. [Google Scholar] [CrossRef]
- Upton, B.M.; Kasko, A.M. Strategies for the conversion of lignin to high-value polymeric materials: Review and perspective. Chem. Rev. 2016, 116, 2275–2306. [Google Scholar] [CrossRef]
- Ahvazi, B.; Cloutier, É.; Wojciechowicz, O.; Ngo, T.D. Lignin profiling: A guide for selecting appropriate lignins as precursors in biomaterials development. ACS Sustain. Chem. Eng. 2016, 4, 5090–5105. [Google Scholar] [CrossRef]
- Gao, W.J.; Fatehi, P. Lignin for polymer and nanoparticle production: Current status and challenges. Can. J. Chem. Eng. 2019, 97, 2827–2842. [Google Scholar] [CrossRef]
- Ciolacu, D.; Cazacu, G. New green hydrogels based on lignin. J. Nanosci. Nanotechnol. 2018, 18, 2811–2822. [Google Scholar] [CrossRef]
- Naseem, A.; Tabasum, S.; Zia, K.M.; Zuber, M.; Noreen, A. Lignin-derivatives based polymers, blends and composites: A review. Int. J. Biol. Macromol. 2016, 93, 296–313. [Google Scholar] [CrossRef]
- Meng, Y.; Lu, J.; Cheng, Y.; Li, Q.; Wang, H.S. Lignin-based hydrogels: A review of preparation, properties, and application. Int. J. Biol. Macromol. 2020, 35, 1006–1019. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.F.; Yang, D.J.; Deng, Y.H.; Chen, P.; Zhou, H.F.; Qiu, X.Q. Influence of pH on the behavior of lignosulfonate macromolecules in aqueous solution. Colloids Surf. A 2010, 371, 50–58. [Google Scholar] [CrossRef]
- Tang, Q.Q. Researches on Concentrated Solution Behaviors of Lignosulfonate and Preparation of Lignin Nanoparticles. Ph.D. Thesis, South China University of Technology, Guangzhou, China, 2015. [Google Scholar]
- Chakar, F.S.; Ragauskas, A.J. Review of current and future softwood kraft lignin process chemistry. Ind. Crop. Prod. 2004, 20, 131–141. [Google Scholar] [CrossRef]
- Zhou, M.S.; Qiu, X.Q.; Yang, D.J.; Lou, H.M.; Ouyang, X.P. High-performance dispersant of coal-water slurry synthesized from wheat straw alkali lignin. Fuel Process. Technol. 2007, 88, 375–382. [Google Scholar] [CrossRef]
- Ouyang, X.P.; Ke, L.X.; Qiu, X.Q.; Guo, Y.X.; Pang, Y.X. Sulfonation of alkali lignin and its potential use in dispersant for cement. J. Disper. Sci. Technol. 2009, 30, 1–6. [Google Scholar] [CrossRef]
- Ouyang, X.P.; Lin, Z.X.; Deng, Y.H.; Yang, D.J.; Qiu, X.Q. Oxidative degradation of soda lignin assisted by microwave irradiation. Chin. J. Chem. Eng. 2010, 18, 695–702. [Google Scholar] [CrossRef]
- Mai, C.; Majcherczyk, A.; Hüttermann, A. Chemo-enzymatic synthesis and characterization of graft copolymers from lignin and acrylic compounds. Enzym. Microb. Technol. 2000, 27, 167–175. [Google Scholar] [CrossRef]
- Ouyang, X.P.; Lin, Z.X.; Yang, D.J.; Qiu, X.Q. Chemical modification of lignin assisted by microwave irradiation. Holzforschung 2011, 65, 697–701. [Google Scholar] [CrossRef]
- Cerrutti, B.M.; de Souza, C.S.; Castellan, A.; Ruggiero, R.; Frollini, E. Carboxymethyl lignin as stabilizing agent in aqueous ceramic suspensions. Ind. Crop. Prod. 2012, 36, 108–115. [Google Scholar] [CrossRef]
- Kalliola, A.; Vehmas, T.; Liitia, T.; Tamminen, T. Alkali-O2 oxidized lignin-a bio-based concrete plasticizer. Ind. Crop. Prod. 2015, 74, 150–157. [Google Scholar] [CrossRef]
- Wu, H.R.; Chen, F.G.; Feng, Q.H.; Yue, X.P. Oxidation and sulfomethylation of alkali-extracted lignin from corn stalk. BioResources 2012, 7, 2742–2751. [Google Scholar]
- Meister, J.J. Modification of lignin. J. Macromol. Sci. C 2002, 42, 235–289. [Google Scholar] [CrossRef]
- Liu, Y.; Li, K. Preparation and characterization of demethylated lignin-polyethylenimine adhesives. J. Adhes. 2006, 82, 593–605. [Google Scholar] [CrossRef]
- Qian, Y.; Wang, T.; Qiu, X.Q.; Zhao, D.C.; Liu, D.; Deng, Y.H. Conductivity enhancement of poly(3,4-ethylenedioxythiophene)/lignosulfonate acid complexes via pickering emulsion polymerization. ACS Sustain. Chem. Eng. 2016, 4, 7193–7199. [Google Scholar] [CrossRef]
- Graca, M.P.F.; Rudnitskaya, A.; Faria, F.A.C.; Evtuguin, D.V.; Gomes, M.T.S.R.; Oliveira, J.A.B.P.; Costa, L.C. Electrochemical impedance study of the lignin-derived conducting polymer. Electrochim. Acta 2012, 76, 69–76. [Google Scholar] [CrossRef]
- Rozite, L.; Varna, J.; Joffe, R.; Pupurs, A. Nonlinear behavior of PLA and lignin-based flax composites subjected to tensile loading. J. Thermoplast. Compos. Mater. 2013, 26, 476–496. [Google Scholar] [CrossRef]
- Agarwal, A.; Rana, M.; Park, J.M. Advancement in technologies for the depolymerization of lignin. Fuel Process. Technol. 2018, 181, 115–132. [Google Scholar] [CrossRef]
- Gosselink, R.J.A.; Abacherli, A.; de Jong, E.; Guran, B. Co-ordination network for lignin-standardisation, production and applications adapted to market requirements (EUROLIGNIN). Ind. Crop. Prod. 2004, 20, 121–129. [Google Scholar] [CrossRef]
- Iravani, S.; Varma, R.S. Greener synthesis of lignin nanoparticles and their applications. Green Chem. 2020, 22, 612–636. [Google Scholar] [CrossRef]
- Lou, H.M.; Lai, H.R.; Wang, M.X.; Pang, Y.X.; Yang, D.J.; Qiu, X.Q.; Wang, B.; Zhang, H.B. Preparation of lignin-based superplasticizer by graft sulfonation and investigation of the dispersive performance and mechanism in a cementitious system. Ind. Eng. Chem. Res. 2013, 52, 16101–16109. [Google Scholar] [CrossRef]
- Lin, X.L.; Zhou, M.S.; Wang, S.Y.; Lou, H.M.; Yang, D.J.; Qiu, X.Q. Synthesis, structure and dispersion property of a novel lignin-based polyoxyethylene ether from kraft lignin and polyethylene glycol. ACS Sustain. Chem. Eng. 2014, 2, 1902–1909. [Google Scholar] [CrossRef]
- Zhou, M.S.; Kong, Q.; Pan, B.; Qiu, X.Q.; Yang, D.J.; Lou, H.M. Evaluation of treated black liquor used as dispersant of concentrated coal-water slurry. Fuel 2010, 89, 716–723. [Google Scholar] [CrossRef]
- Zhou, H.F.; Chang, Y.Q.; Wu, X.L.; Yang, D.J.; Qiu, X.Q. Horseradish peroxidase modification of sulfomethylated wheat straw alkali lignin to improve its dispersion performance. ACS Sustain. Chem. Eng. 2015, 3, 518–523. [Google Scholar] [CrossRef]
- Chiwetelu, C.I.; Hornof, V.; Neale, G.H.; George, A.E. Use of mixed surfactants to improve the transient interfacial tension behavior of heavy oil/alkaline systems. Can. J. Chem. Eng. 1994, 72, 534–540. [Google Scholar] [CrossRef]
- Zhou, H.F.; Yang, D.J.; Wu, X.L.; Deng, Y.H.; Qiu, X.Q. Physicochemical properties of sodium lignosulfonates (NaLS) modified by laccase. Holzforschung 2012, 66, 825–832. [Google Scholar] [CrossRef]
- Deng, Y.H.; Liu, Y.F.; Qian, Y.; Zhang, W.J.; Qiu, X.Q. Preparation of photoresponsive azo polymers based on lignin, a renewable biomass resources. ACS Sustain. Chem. Eng. 2015, 3, 1111–1116. [Google Scholar] [CrossRef]
- Qin, Y.L.; Yang, D.J.; Qiu, X.Q. Hydroxypropyl sulfonated lignin as dye dispersant: Effect of average molecular weight. ACS Sustain. Chem. Eng. 2015, 3, 3239–3244. [Google Scholar] [CrossRef]
- Qin, Y.L.; Yu, L.X.; Wu, R.C.; Yang, D.J.; Qiu, X.Q.; Zhu, J.Y. Biorefinery lignosulfonates from sulfite-pretreated softwoods as dispersant for graphite. ACS Sustain. Chem. Eng. 2016, 4, 2200–2205. [Google Scholar] [CrossRef]
- James, A. Composition and Process for Cleaning Metal Surfaces. U.S. Patent 3247120, 19 May 1966. [Google Scholar]
- Kai, D.; Tan, M.J.; Chee, P.L.; Chu, Y.K.; Yap, Y.L.; Loh, X.J. Towards lignin-based functional materials in a sustainable world. Green Chem. 2016, 18, 1175–1200. [Google Scholar] [CrossRef]
- Tuck, C.O.; Pérez, E.; Horváth, I.T.; Sheldon, R.A.; Poliakoff, M. Valorization of biomass: Deriving more value from waste. Science 2012, 337, 695. [Google Scholar] [CrossRef] [PubMed]
- Norgren, M.; Edlund, H. Lignin: Recent advances and emerging applications. Curr. Opin. Colloid Interface Sci. 2014, 19, 409–416. [Google Scholar] [CrossRef]
- Li, H.; Deng, Y.H.; Liu, B.; Ren, Y.; Liang, J.Q.; Qian, Y.; Qiu, X.Q.; Li, C.L.; Zheng, D.F. Preparation of nanocapsules via the self-assembly of kraft lignin: A totally green process with renewable resources. ACS Sustain. Chem. Eng. 2016, 4, 1946–1953. [Google Scholar] [CrossRef]
- Doherty, W.O.S.; Mousavioun, P.; Fellows, C.M. Value-adding to cellulosic ethanol: Lignin polymers. Ind. Crop. Prod. 2011, 33, 259–276. [Google Scholar] [CrossRef]
- Zhao, W.W.; Simmons, B.; Singh, S.; Ragauskas, A.J.; Cheng, G. From lignin association to nano-/micro-particle preparation: Extracting higher value of lignin. Green Chem. 2016, 18, 5693–5700. [Google Scholar] [CrossRef]
- Duval, A.; Lawoko, M. A review on lignin-based polymeric, micro- and nano-structured materials. React. Funct. Polym. 2014, 85, 78–96. [Google Scholar] [CrossRef]
- Kumar, A.; Anushree, K.J.; Bhaskar, T. Utilization of lignin: A sustainable and eco-friendly approach. J. Energy Inst. 2020, 93, 235–271. [Google Scholar] [CrossRef]
- Azadi, P.; Inderwildi, O.R.; Farnood, R.; King, D.A. Liquid fuels, hydrogen and chemicals from lignin: A critical review. Renew. Sustain. Energy Rev. 2013, 21, 506–523. [Google Scholar] [CrossRef]
- Lora, J.H. Quality Living through Chemurgy and Green Chemistry, 1st ed.; Springer: Berlin, Germany, 2016; pp. 223–226. [Google Scholar]
- Tejado, A.; Pena, C.; Labidi, J.; Echeverria, J.M.; Mondragon, I. Physico-chemical characterization of lignins from different sources for use in phenol formaldehyde resin synthesis. Bioresour. Technol. 2007, 98, 1655–1663. [Google Scholar] [CrossRef]
- Zakzeski, J.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The catalytic valorization of lignin for the production of renewable chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef]
- Galkin, M.V.; Samec, J.S.M. Lignin valorization through catalytic lignocellulose fractionation: A fundamental platform for the future biorefinery. ChemSusChem 2016, 9, 1544–1558. [Google Scholar] [CrossRef] [PubMed]
- Liitiä, T.M.; Maunu, S.L.; Hortling, B.; Toikka, M.; Kilpeläinen, I. Analysis of technical lignins by two- and three-dimensional NMR spectroscopy. J. Agric. Food Chem. 2003, 51, 2136–2143. [Google Scholar] [CrossRef]
- Chio, C.; Sain, M.; Qin, W.S. Lignin utilization: A review of lignin depolymerization from various aspects. Renew. Sustain. Energ. Rev. 2019, 107, 232–249. [Google Scholar] [CrossRef]
- De la Torre, M.J.; Moral, A.; Hernández, M.D.; Cabeza, E.; Tijero, A. Organosolv lignin for biofuel. Ind. Eng. Chem. Res. 2013, 45, 58–63. [Google Scholar] [CrossRef]
- El Hage, R.; Brosse, N.; Sannigrahi, P.; Ragauskas, A. Effects of process severity on the chemical structure of Miscanthus ethanol organosolv lignin. Polym. Degrad. Stab. 2010, 95, 997–1003. [Google Scholar] [CrossRef]
- Lancefield, C.S.; Panovic, I.; Deuss, P.J.; Barta, K.; Westwood, N.J. Pre-treatment of lignocellulosic feedstocks using biorenewable alcohols: Towards complete biomass valorisation. Green Chem. 2017, 19, 202–214. [Google Scholar] [CrossRef]
- Prinsen, P.; Rencoret, J.; Gutiérrez, A.; Liitiä, T.; Tamminen, T.; Colodette, J.L.; Berbis, M.A.; Barbero, J.J.; Martinez, T.A.; Del Rio, J.C. Modification of the lignin structure during alkaline delignification of eucalyptus wood by kraft, soda-AQ, and soda-O2 cooking. Ind. Eng. Chem. Res. 2013, 52, 15702–15712. [Google Scholar] [CrossRef]
- Qian, Y.; Deng, Y.H.; Qiu, X.Q.; Li, H.; Yang, D.J. Formation of uniform colloidal spheres from lignin, a renewable resource recovered from pulping spent liquor. Green Chem. 2014, 16, 2156–2163. [Google Scholar] [CrossRef]
- Qian, Y.; Qiu, X.Q.; Zhong, X.W.; Zheng, D.F.; Deng, Y.H.; Yang, D.J.; Zhu, S.P. Lignin reverse micelles for UV-absorbing and high mechanical performance thermoplastics. Ind. Eng. Chem. Res. 2015, 54, 12025–12030. [Google Scholar] [CrossRef]
- Deng, Y.H.; Zhao, H.J.; Qian, Y.; Lü, L.; Wang, B.B.; Qiu, X.Q. Hollow lignin azo colloids encapsulated avermectin with high anti-photolysis and controlled release performance. Ind. Crop. Prod. 2016, 87, 191–197. [Google Scholar] [CrossRef]
- Li, Y.L.; Wu, M.; Wang, B.; Wu, Y.Y.; Ma, M.G.; Zhang, X.M. Synthesis of magnetic lignin-based hollow microspheres: A highly adsorptive and reusable adsorbent derived from renewable resources. ACS Sustain. Chem. Eng. 2016, 4, 5523–5532. [Google Scholar] [CrossRef]
- Richter, A.P.; Bharti, B.; Armstrong, H.B.; Brown, J.S.; Plemmons, D.; Paunov, V.N.; Stoyanov, S.D.; Velev, O.D. Synthesis and characterization of biodegradable lignin nanoparticles with tunable surface properties. Langmuir 2016, 32, 6468–6477. [Google Scholar] [CrossRef]
- Qian, Y.; Zhong, X.W.; Li, Y.; Qiu, X.Q. Fabrication of uniform lignin colloidal spheres for developing natural broad-spectrum sunscreens with high sun protection factor. Ind. Crop. Prod. 2017, 101, 54–60. [Google Scholar] [CrossRef]
- Huang, J.B.; Wang, M.; Song, P.P.; Li, Y.L.; Xu, F.; Zhang, X.M. Directed 2D nanosheet assemblies of amphiphilic lignin derivatives: Formation of hollow spheres with tunable porous structure. Ind. Crop. Prod. 2019, 127, 16–25. [Google Scholar] [CrossRef]
- Yan, Z.Y.; Liao, G.F.; Zou, X.X.; Zhao, M.K.; Wu, T.; Chen, Y.H.; Fang, G.G. Size-Controlled and super long-term stable lignin nanospheres through a facile self-assembly strategy from kraft lignin. J. Agric. Food Chem. 2020, 68, 8341–8349. [Google Scholar] [CrossRef]
- Trevisan, H.; Rezende, C.A. Pure, stable and highly antioxidant lignin nanoparticles from elephant grass. Ind. Crop. Prod. 2020, 145, 112105. [Google Scholar] [CrossRef]
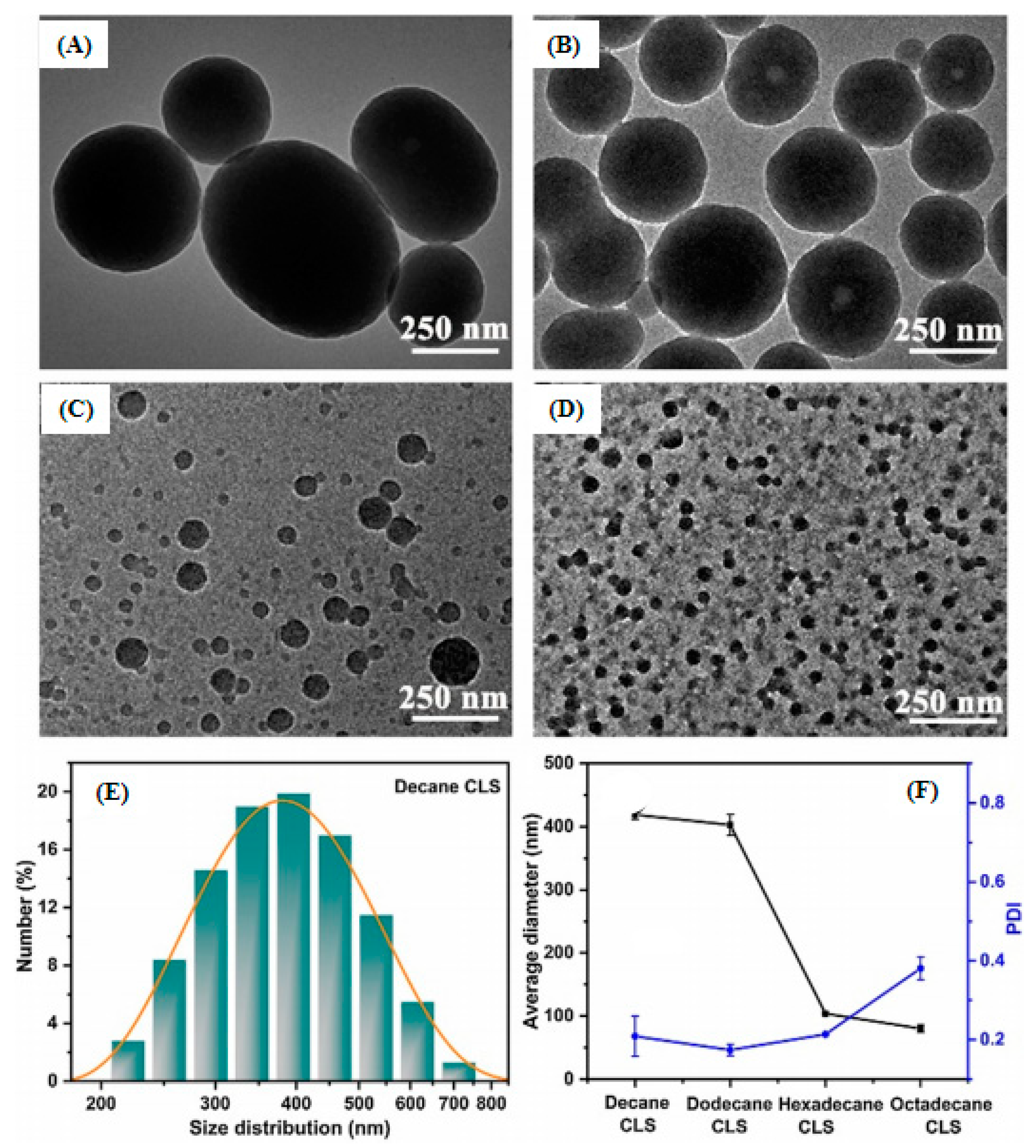
- Li, B.Q.; You, S.P.; Qi, W.; Wang, Y.F.; Su, R.X.; He, Z.M. Structure-tunable assembly of lignin sub-micro spheres by modifying the amphiphilic interfaces of lignin via n-alkane. Eur. Polym. J. 2020, 126, 109539. [Google Scholar] [CrossRef]
- Zhong, X.W.; Qian, Y.; Huang, J.H.; Yang, D.J.; Deng, Y.H.; Qiu, X.Q. Fabrication of lignosulfonate vesicular reverse micelles to immobilize horseradish peroxidase. Ind. Eng. Chem. Res. 2016, 55, 2731–2737. [Google Scholar] [CrossRef]
- Li, Y.Y.; Yang, D.J.; Lu, S.; Lao, S.L.; Qiu, X.Q. Modified lignin with anionic surfactant and its application in controlled release of avermectin. J. Agric. Food Chem. 2018, 66, 3457–3464. [Google Scholar] [CrossRef] [PubMed]
- Lievonen, M.; Valle-Delgado, J.J.; Mattinen, M.L.; Hult, E.L.; Lintinen, K.; Kostiainen, M.A.; Paananen, A.; Szilvay, G.R.; Setälä, H.; Österberg, M. Simple process for lignin nanoparticle preparation. Green Chem. 2016, 18, 1416–1422. [Google Scholar] [CrossRef]
- Lintinen, K.; Latikka, M.; Sipponen, M.H.; Ras, R.H.A.; Österberg, M.; Kostiainen, M.A. Structural diversity in metal-organic nanoparticles based on iron isopropoxide treated lignin. RSC Adv. 2016, 6, 31790–31796. [Google Scholar] [CrossRef]
- Figueiredo, P.; Lintinen, K.; Kiriazis, A.; Hynninen, V.; Liu, Z.H.; Bauleth-Ramos, T.; Rahikkala, A.; Correia, A.; Kohout, T.; Sarmento, B.; et al. In vitro evaluation of biodegradable lignin-based nanoparticles for drug delivery and enhanced antiproliferation effect in cancer cells. Biomaterials 2017, 121, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Zikeli, F.; Vinciguerra, V.; D’Annibale, A.; Capitani, D.; Romagnoli, M.; Mugnozza, G.S. Preparation of lignin nanoparticles from wood waste for wood surface treatment. Nanomaterials 2019, 9, 281. [Google Scholar] [CrossRef] [PubMed]
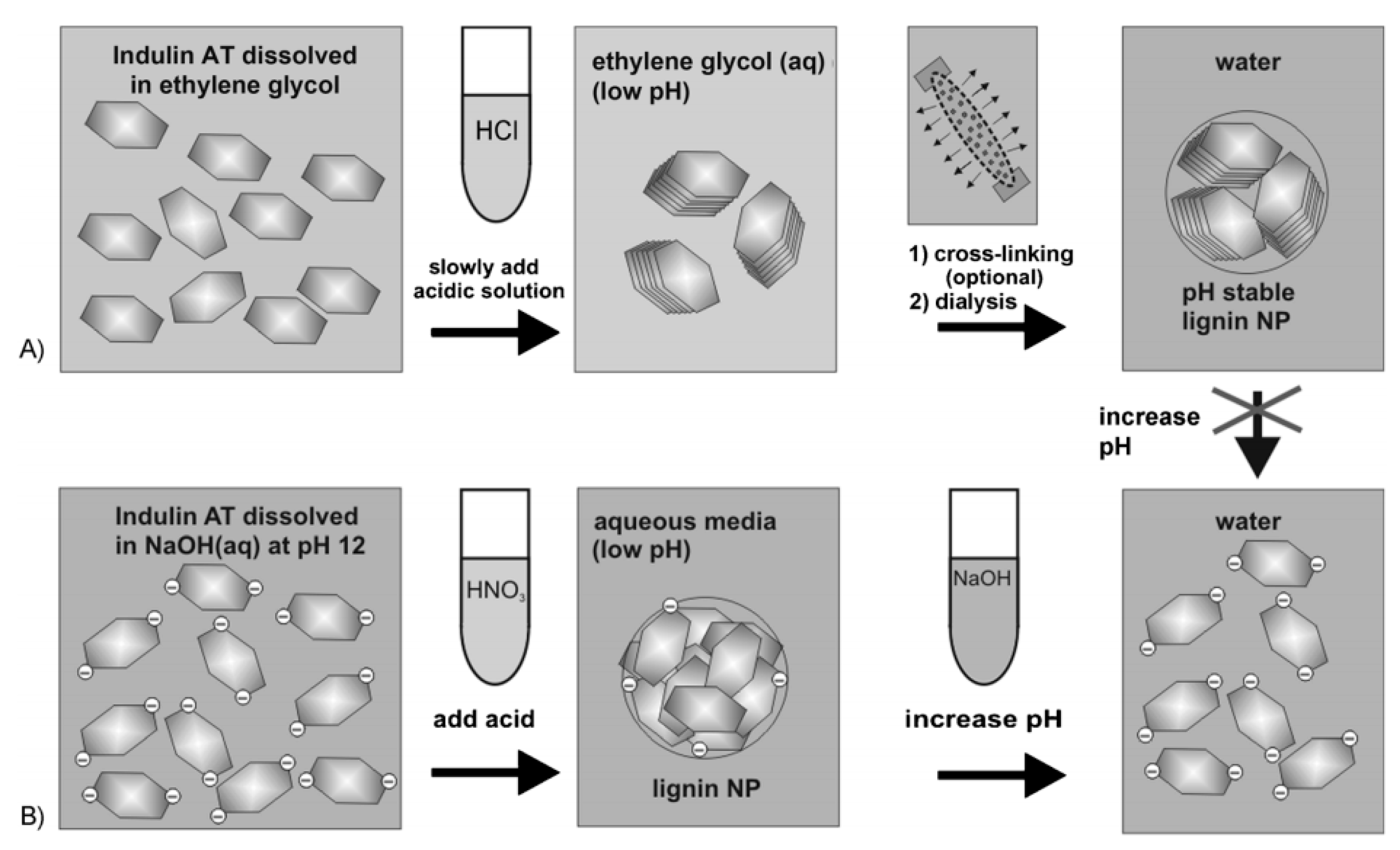
- Frangville, C.; Rutkevičius, M.; Richter, A.P.; Velev, O.D.; Stoyanov, S.D.; Paunov, V.N. Fabrication of environmentally biodegradable lignin nanoparticles. ChemPhysChem 2012, 13, 4235–4243. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Mohanty, S.; Nayak, S.K. Influence of addition of vapor grown carbon fibers on mechanical, thermal and biodegradation properties of lignin nanoparticle filled bio-poly(trimethylene terephthalate) hybrid nanocomposites. RSC Adv. 2015, 5, 56028–56036. [Google Scholar] [CrossRef]
- Pang, Y.X.; Wang, S.W.; Qiu, X.Q.; Luo, Y.L.; Lou, H.M.; Huang, J.H. Preparation of lignin/sodium dodecyl sulfate composite nanoparticles and their application in pickering emulsion template based microencapsulation. J. Agric. Food Chem. 2017, 65, 11011–11019. [Google Scholar] [CrossRef]
- Rahman, O.; Shi, S.B.; Ding, J.H.; Wang, D.L.; Ahmad, S.; Yu, H.B. Lignin nanoparticles: Synthesis, characterization and corrosion protection performance. New J. Chem. 2018, 42, 3415–3425. [Google Scholar] [CrossRef]
- Barakat, A.; Putaux, J.L.; Saulnier, L.; Chabbert, B.; Cathala, B. Characterization of arabinoxylan-dehydrogenation polymer (synthetic lignin polymer) nanoparticles. Biomacromolecules 2007, 8, 1236–1245. [Google Scholar] [CrossRef]
- Qian, Y.; Zhang, Q.; Qiu, X.Q.; Zhu, S.P. CO2-responsive diethylaminoethyl-modified lignin nanoparticles and their application as surfactants for CO2/N2-switchable Pickering emulsions. Green Chem. 2014, 16, 4963–4968. [Google Scholar] [CrossRef]
- Gilca, I.A.; Popa, V.I.; Crestini, C. Obtaining lignin nanoparticles by sonication. Ultrason. Sonochem. 2015, 23, 369–375. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Qian, Y.; Wang, J.Y.; Qiu, X.Q.; Zeng, H.B. Bioinspired lignin-polydopamine nanocapsules with strong bioadhesion for long-acting and high-performance natural sunscreens. Biomacromolecules 2020, 21, 3231–3241. [Google Scholar] [CrossRef] [PubMed]
- Yiamsawas, D.; Baier, G.; Thines, E.; Landfester, K.; Wurm, F.R. Biodegradable lignin nanocontainers. RSC Adv. 2014, 4, 11661–11663. [Google Scholar] [CrossRef]
- Tortora, M.; Cavalieri, F.; Mosesso, P.; Ciaffardini, F.; Melone, F.; Crestini, C. Ultrasound driven assembly of lignin into microcapsules for storage and delivery of hydrophobic molecules. Biomacromolecules 2014, 15, 1634–1643. [Google Scholar] [CrossRef]
- Li, Z.L.; Ge, Y.Y.; Wan, L. Fabrication of a green porous lignin-based sphere for the removal of lead ions from aqueous media. J. Hazard. Mater. 2015, 285, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Nypelö, T.E.; Carrillo, C.A.; Rojas, O.J. Lignin supracolloids synthesized from (W/O) microemulsions: Use in the interfacial stabilization of Pickering systems and organic carriers for silver metal. Soft Matter 2015, 11, 2046–2054. [Google Scholar] [CrossRef]
- Chen, N.S.; Dempere, L.A.; Tong, Z.H. Synthesis of pH-responsive lignin-based nanocapsules for controlled release of hydrophobic molecules. ACS Sustain. Chem. Eng. 2016, 4, 5204–5211. [Google Scholar] [CrossRef]
- Beisl, S.; Miltner, A.; Friedl, A. Lignin from micro- to nanosize: Production methods. Int. J. Mol. Sci. 2017, 18, 1244. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Zhu, M.H.; Zu, Y.G.; Liu, W.J.; Yang, L.; Zhang, Y.; Zhao, X.H.; Zhang, X.N.; Zhang, X.N.; Li, W.G. Comparative antioxidant activity of nanoscale lignin prepared by a supercritical antisolvent (SAS) process with non-nanoscale lignin. Food Chem. 2012, 135, 63–67. [Google Scholar] [CrossRef]
- Myint, A.A.; Lee, H.W.; Seo, B.; Son, W.S.; Yoon, J.; Yoon, T.J.; Park, H.J.; Yu, J.; Yoon, J.; Lee, Y.W. One pot synthesis of environmentally friendly lignin nanoparticles with compressed liquid carbon dioxide as an antisolvent. Green Chem. 2016, 18, 2129–2146. [Google Scholar] [CrossRef]
- Dai, L.; Liu, R.; Hu, L.Q.; Zou, Z.F.; Si, C.L. Lignin nanoparticle as a novel green carrier for the efficient delivery of resveratrol. ACS Sustain. Chem. Eng. 2017, 5, 8241–8249. [Google Scholar] [CrossRef]
- Li, Y.X.; Zhou, M.S.; Pang, Y.X.; Qiu, X.Q. Lignin-based microsphere: Preparation and performance on encapsulating the pesticide avermectin. ACS Sustain. Chem. Eng. 2017, 5, 3321–3328. [Google Scholar] [CrossRef]
- Li, Y.Y.; Qiu, X.Q.; Qian, Y.; Xiong, W.L.; Yang, D.J. pH-responsive lignin-based complex micelles: Preparation, characterization and application in oral drug delivery. Chem. Eng. J. 2017, 327, 1176–1183. [Google Scholar] [CrossRef]
- Mishra, P.K.; Wimmer, R. Aerosol assisted self-assembly as a route to synthesize solid and hollow spherical lignin colloids and its utilization in layer by layer deposition. Ultrason. Sonochem. 2017, 35, 45–50. [Google Scholar] [CrossRef]
- Chen, L.H.; Zhou, X.Y.; Shi, Y.F.; Gao, B.; Wu, J.P.; Kirk, T.B.; Xu, J.K.; Xue, W. Green synthesis of lignin nanoparticle in aqueous hydrotropic solution toward broadening the window for its processing and application. Chem. Eng. J. 2018, 346, 217–225. [Google Scholar] [CrossRef]
- Wang, B.; Sun, D.; Wang, H.M.; Yuan, T.Q.; Sun, R.C. Green and facile preparation of regular lignin nanoparticles with high yield and their natural broad-spectrum sunscreens. ACS Sustain. Chem. Eng. 2019, 7, 2658–2666. [Google Scholar] [CrossRef]
- Ju, T.; Zhang, Z.L.; Li, Y.; Miao, X.F.; Ji, J.B. Continuous production of lignin nanoparticles using a microchannel reactor and its application in UV-shielding films. RSC Adv. 2019, 9, 24915–24921. [Google Scholar] [CrossRef]
- Chuang, Y.L.; Olsson, J.V.; Li, R.J.; Frank, C.W.; Waymouth, R.M.; Billington, S.L.; Sattely, E.S. A renewable lignin-lactide copolymer and application in biobased composites. ACS Sustain. Chem. Eng. 2013, 1, 1231–1238. [Google Scholar] [CrossRef]
- Jiang, C.; He, H.; Jiang, H.; Ma, L.; Jia, D.M. Nano-lignin filled natural rubber composites: Preparation and characterization. Express Polym. Lett. 2013, 7, 480–493. [Google Scholar] [CrossRef]
- Nair, S.S.; Sharma, S.; Pu, Y.; Sun, Q.; Pan, S.; Zhu, J.Y.; Deng, Y.; Ragauskas, A.J. High shear homogenization of lignin to nanolignin and thermal stability of nanolignin-polyvinyl alcohol blends. ChemSusChem 2014, 7, 3513–3520. [Google Scholar] [CrossRef]
- Kai, D.; Jiang, S.; Low, Z.W.; Loh, X.J. Engineering highly stretchable lignin-based electrospun nanofibers for potential biomedical Applications. J. Mater. Chem. B 2015, 3, 6194–6204. [Google Scholar] [CrossRef]
- Yang, W.J.; Rallini, M.; Wang, D.Y.; Gao, D.Q.; Dominici, F.; Torre, L.; Kenny, J.M.; Puglia, D. Role of lignin nanoparticles in UV resistance, thermal and mechanical performance of PMMA nanocomposites prepared by a combined free-radical graft polymerization/masterbatch procedure. Compos. Part A Appl. Sci. Manuf. 2018, 107, 61–69. [Google Scholar] [CrossRef]
- Ge, Y.Y.; Wei, Q.; Li, Z.L. Preparation and evaluation of the free radical scavenging activities of nanoscale lignin biomaterials. BioResources 2014, 9, 6699–6706. [Google Scholar] [CrossRef]
- Yang, W.; Owczarek, J.S.; Fortunati, E.; Kozanecki, M.; Mazzaglia, A.; Balestra, G.M.; Kenny, J.M.; Torre, L.; Puglia, D. Antioxidant and antibacterial lignin nanoparticles in polyvinyl alcohol/chitosan films for active packaging. Ind. Crop. Prod. 2016, 94, 800–811. [Google Scholar] [CrossRef]
- Yearla, S.R.; Padmasree, K. Preparation and characterisation of lignin nanoparticles: Evaluation of their potential as antioxidants and UV protectants. J. Exp. Nanosci. 2016, 11, 289–302. [Google Scholar] [CrossRef]
- Tian, D.; Hu, J.G.; Bao, J.; Chandra, R.P.; Saddler, J.N.; Lu, C.H. Lignin valorization: Lignin nanoparticles as high-value bio-additive for multifunctional nanocomposites. Biotechnol. Biofuels 2017, 10, 192. [Google Scholar] [CrossRef]
- He, X.Y.; Luzi, F.; Hao, X.L.; Yang, W.J.; Torre, L.; Xiao, Z.F.; Xie, Y.J.; Puglia, D. Thermal, antioxidant and swelling behaviour of transparent polyvinyl (alcohol) films in presence of hydrophobic citric acid-modified lignin nanoparticles. Int. J. Biol. Macromol. 2019, 127, 665–676. [Google Scholar] [CrossRef]
- Kim, S.; Fernandes, M.M.; Matamá, T.; Loureiro, A.; Gomes, A.C.; Cavaco-Paulo, A. Chitosan-lignosulfonates sono-chemically prepared nanoparticles: Characterisation and potential applications. Colloids Surf. B 2013, 103, 1–8. [Google Scholar] [CrossRef]
- Azimvand, J.; Didehban, K.; Mirshokraie, S.A. Safranin-O removal from aqueous solutions using lignin nanoparticle-g-polyacrylic acid adsorbent: Synthesis, properties, and application. Adsorpt. Sci. Technol. 2018, 36, 1422–1440. [Google Scholar] [CrossRef]


| Parameter | Lignosulfonate | Kraft Lignin | Organosolv Lignin | Soda Lignin | Enzymatic Hydrolysis Lignin |
|---|---|---|---|---|---|
| Treatment conditions | Metal sulfite + sulfur dioxide (Ca2+, Mg2+ or Na+) (pH = 2–12, T = 120–180 °C, for 1–5 h) | First: Sodium hydroxide and sodium sulfide (pH = 13–14, T ≈ 170 °C); Second: Sulfuric acid (pH = 5–7.5) | Organic solvents (such as acetone, ethanol, and methanol), usually mixed with water (T = 170–190 °C) | 13–16 wt % of sodium hydroxide solutions (T = 140–170 °C) + anthraquinone (catalyzer) | Cellulases and hemicellulases [10] |
| Solubility | Water | Alkali, organic solvents | Organic solvents | Alkali | Partially in organic solvents |
| Ash content (mass%) | 4.0–9.3 | 0.5–3.0 | 1.7 | 0.7–2.3 | 1.0–3.0 |
| Sulfur (%) | 3.5–8.0 | 1.0–3.0 | 0 | 0 | 0–1.0 |
| Carbohydrates (mass%) | ND | 1.0–2.3 | 1–3 | 1.5–3.0 | 10.0–22.4 |
| Molecular weight (Da) | 1000–50,000 | 1500–5000 | 500–5000 | 1000–3000 | 5000–10,000 |
| PDI | 4.2–8.0 | 2.5–3.5 | 1.5–2.5 | 2.5–3.5 | 4.0–11.0 |
| Advantages | A good aqueous solubility [5] | A higher purity [41] | A higher purity [9]; sulfur-free; basically preserves the native structure of lignin [5] | Sulfur-free [41] | Basically non-sulfur; possesses a closer structure to native lignin [19] |
| Disadvantages | A structural change of lignin and the low purity after isolation [5] | A worse solubility [19] | Difficult to produce on a large scale [1] | Difficult to recover through centrifugation or filtration [56] | Very low solubility in either water or some organic solvents [19] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, Q.; Qian, Y.; Yang, D.; Qiu, X.; Qin, Y.; Zhou, M. Lignin-Based Nanoparticles: A Review on Their Preparations and Applications. Polymers 2020, 12, 2471. https://doi.org/10.3390/polym12112471
Tang Q, Qian Y, Yang D, Qiu X, Qin Y, Zhou M. Lignin-Based Nanoparticles: A Review on Their Preparations and Applications. Polymers. 2020; 12(11):2471. https://doi.org/10.3390/polym12112471
Chicago/Turabian StyleTang, Qianqian, Yong Qian, Dongjie Yang, Xueqing Qiu, Yanlin Qin, and Mingsong Zhou. 2020. "Lignin-Based Nanoparticles: A Review on Their Preparations and Applications" Polymers 12, no. 11: 2471. https://doi.org/10.3390/polym12112471
APA StyleTang, Q., Qian, Y., Yang, D., Qiu, X., Qin, Y., & Zhou, M. (2020). Lignin-Based Nanoparticles: A Review on Their Preparations and Applications. Polymers, 12(11), 2471. https://doi.org/10.3390/polym12112471




