Gamma-Irradiation-Prepared Low Molecular Weight Hyaluronic Acid Promotes Skin Wound Healing
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Low-Molecular-Weight Hyaluronic Acid
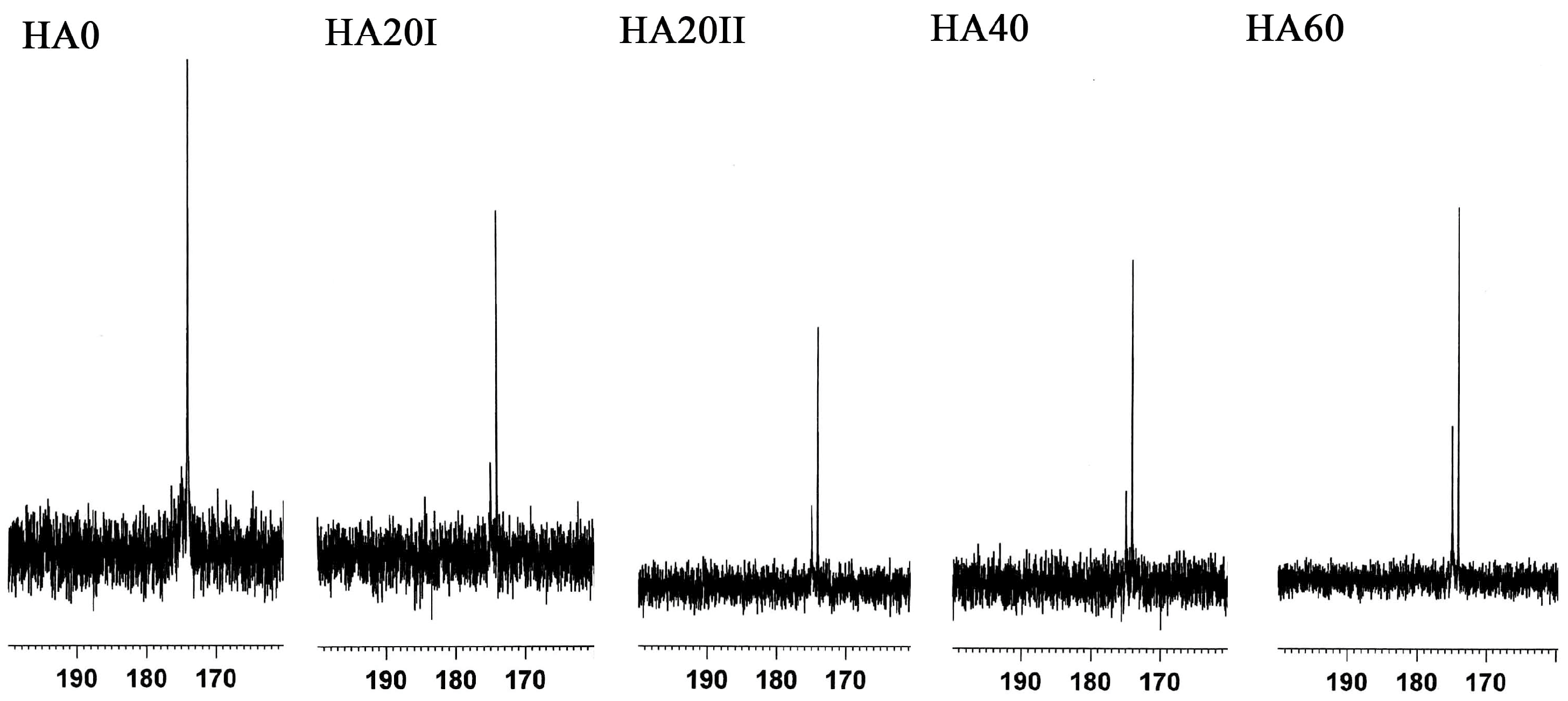
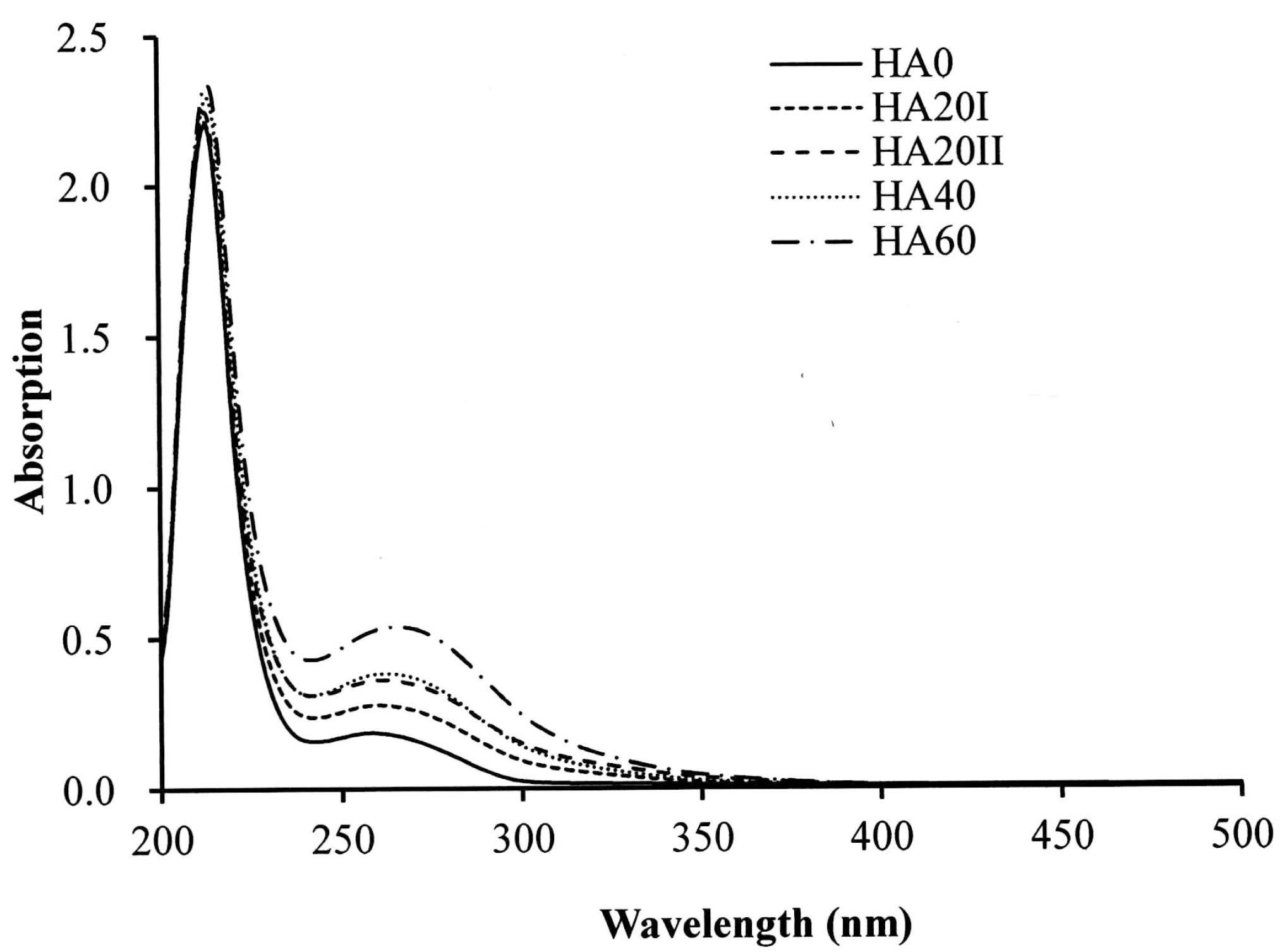
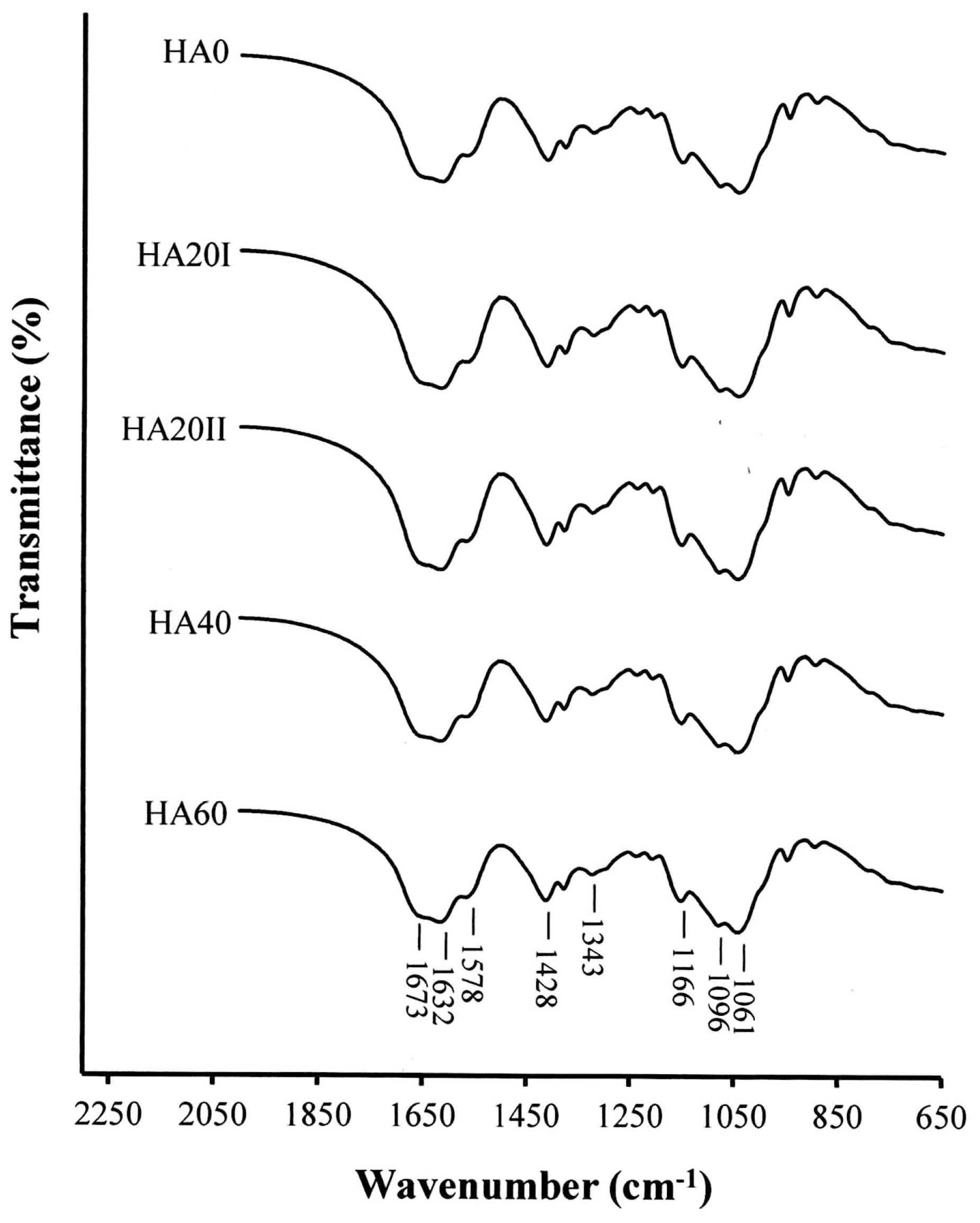
2.2. Chemical Property Analysis
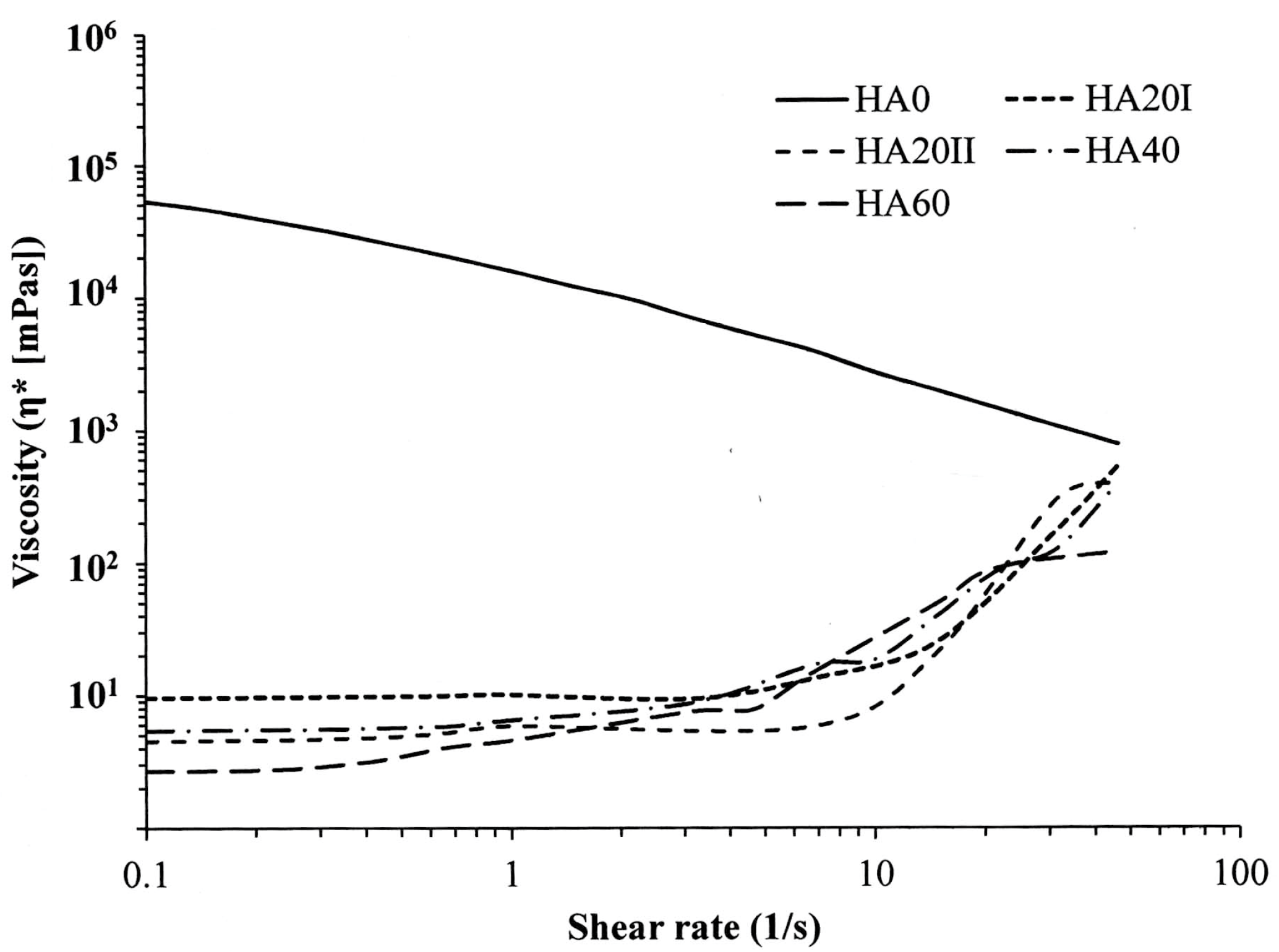
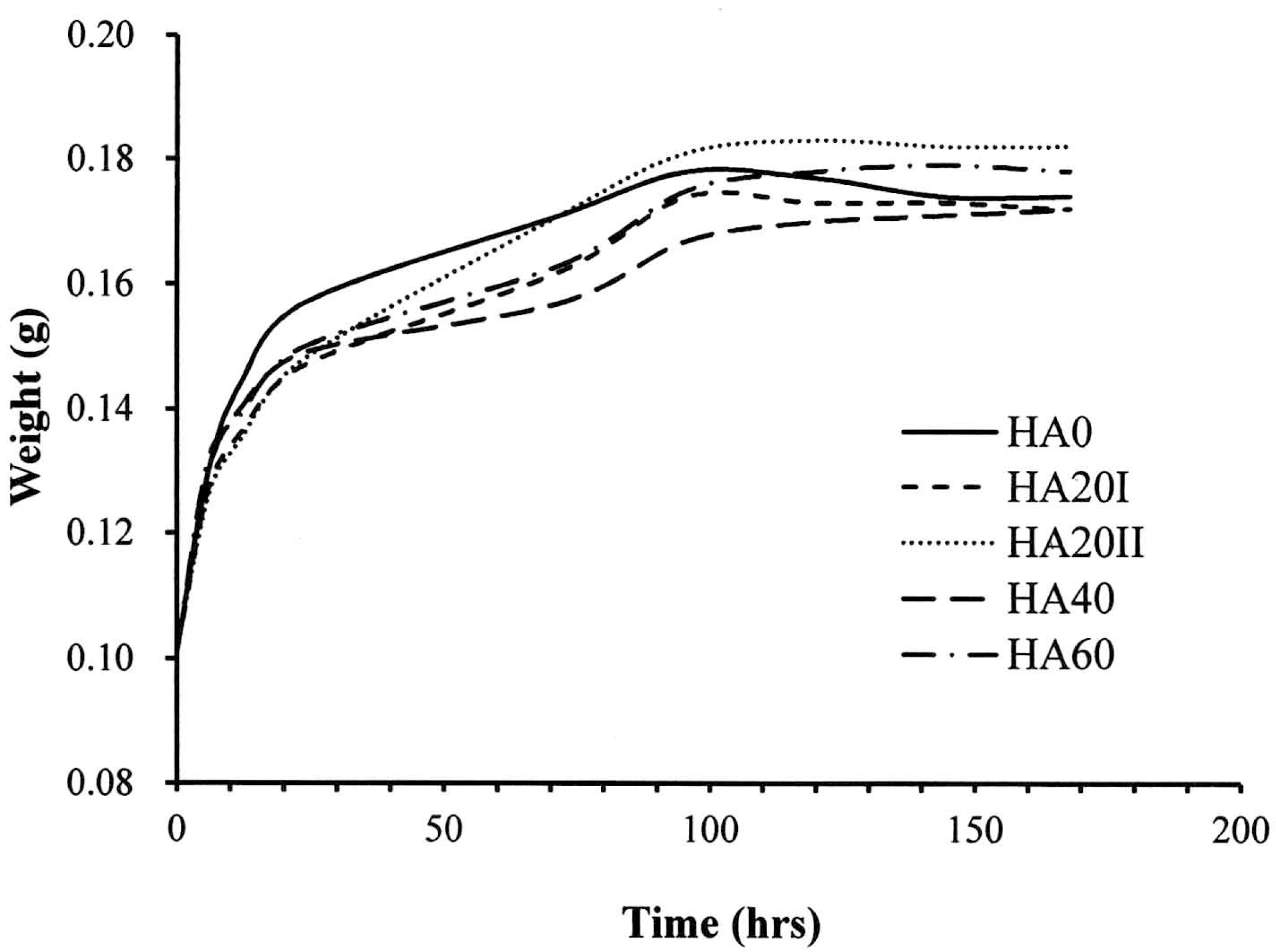
2.3. Physical Property Detection
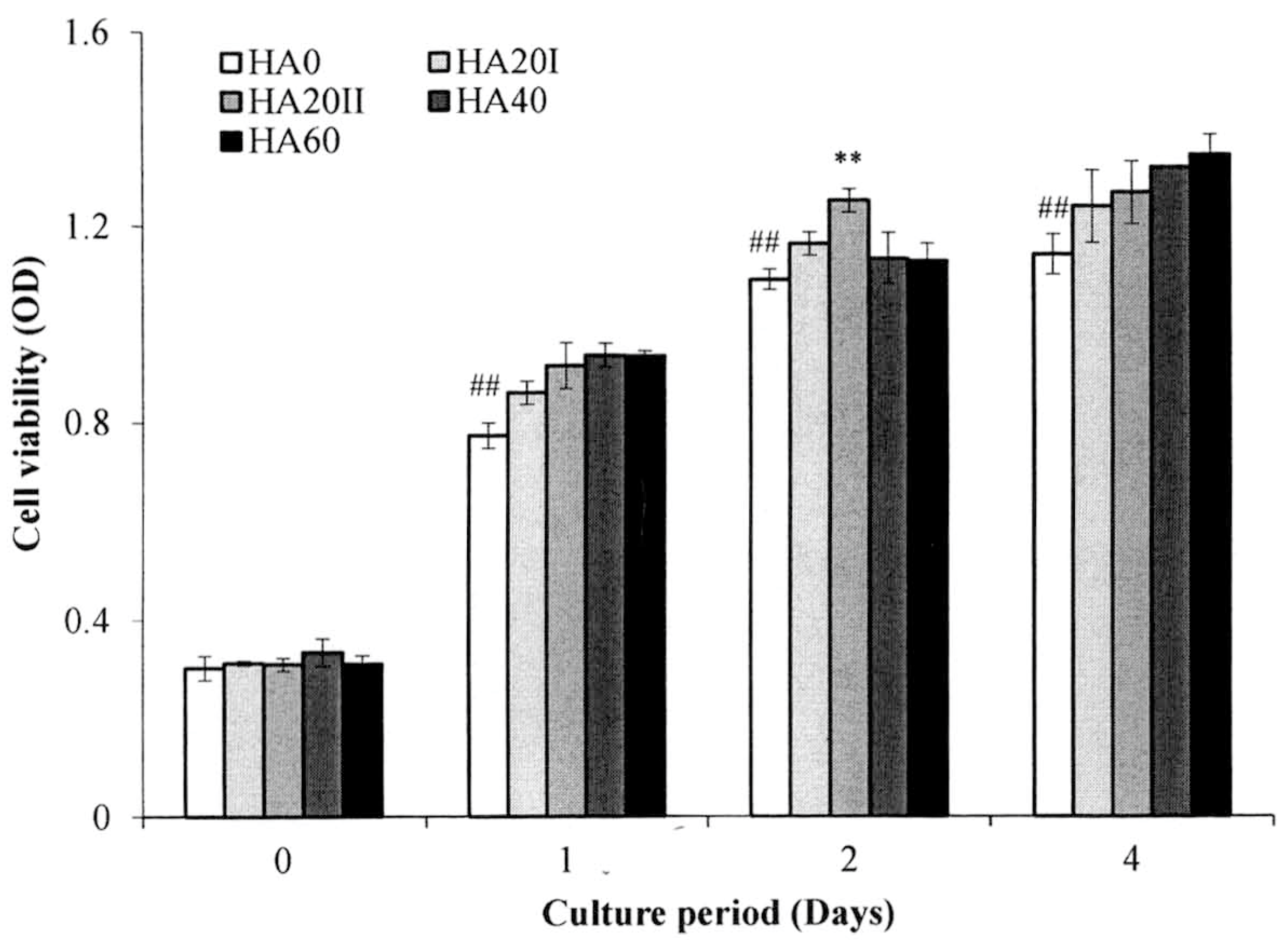
2.4. In Vitro Cell Viability Experiments
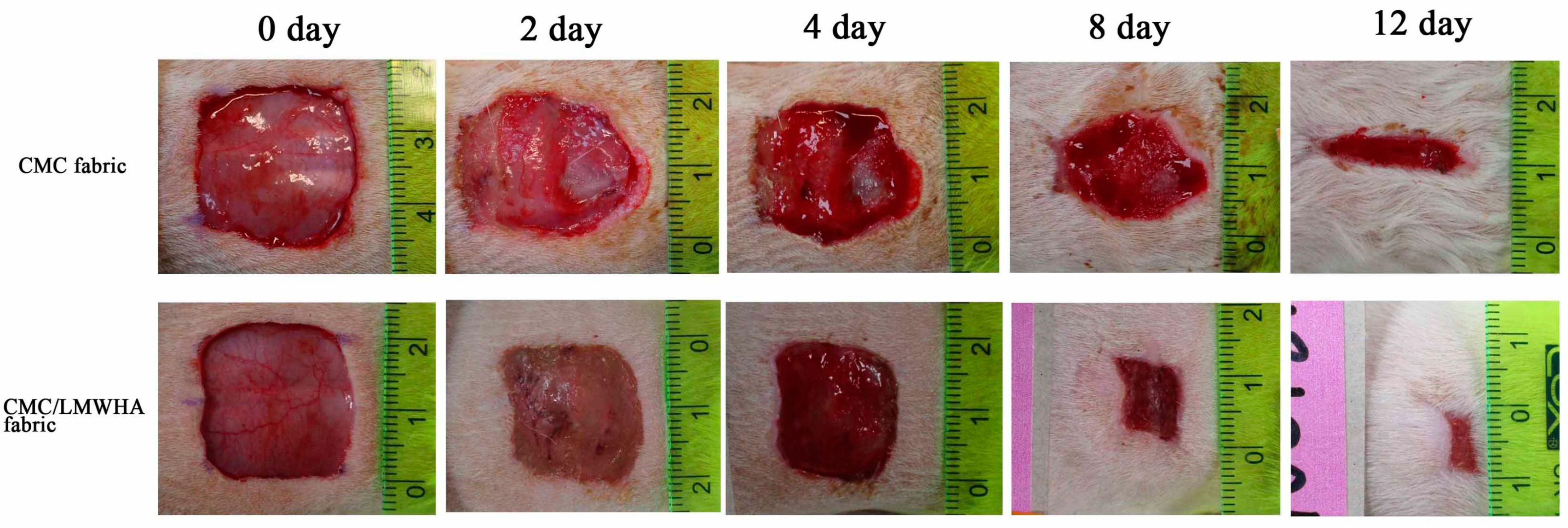
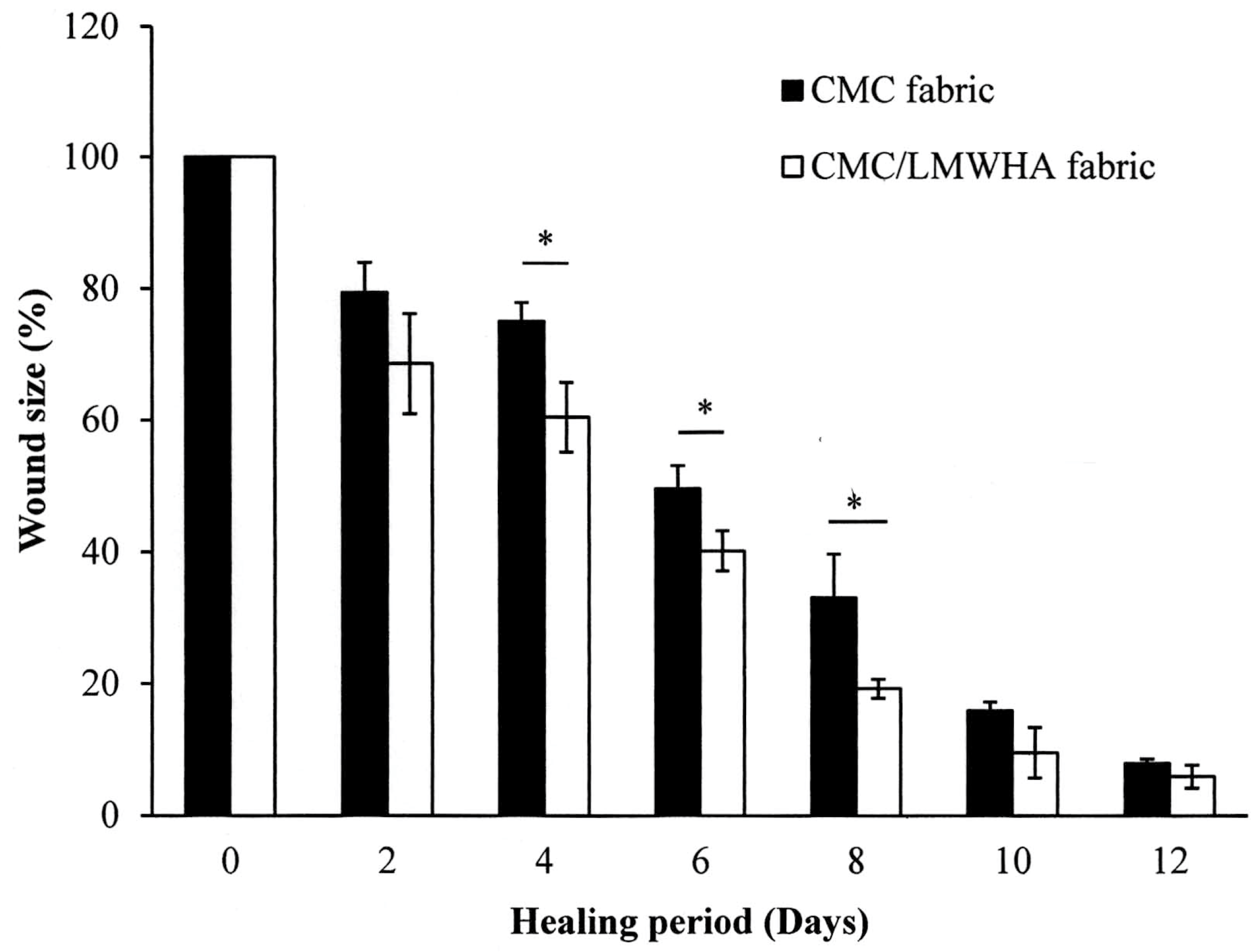
2.5. In Vivo Wound Healing Tests
2.5.1. Carboxymethyl Cellulose (CMC)/LMWHA Dressing Fabrication
2.5.2. In Vivo Wound Healing Experiment
2.6. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Monsuur, H.N.; Boink, M.A.; Weijers, E.M.; Roffel, S.; Breetveld, M.; Gefen, A.; van den Broek, L.J.; Gibbs, S. Methods to study differences in cell mobility during skin wound healing in vitro. J. Biomech. 2016, 49, 1381–1387. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.Z.; Zhong, L.; Santiago, J.L. Anti-inflammatory and skin barrier repair effects of topical application of some plant oils. Int. J. Mol. Sci. 2018, 19, 70. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.F.; Que, H.F.; Wang, Y.J.; Cui, X.J. Chinese herbal medicines for treating skin and soft-tissue infections. Cochrane Database Syst. Rev. 2014, 25, CD010619. [Google Scholar] [CrossRef] [PubMed]
- Korn, P.; Schulz, M.C.; Hintze, V.; Range, U.; Mai, R.; Eckelt, U.; Schnabelrauch, M.; Möller, S.; Becher, J.; Scharnweber, D.; et al. Chondroitin sulfate and sulfated hyaluronan-containing collagen coatings of titanium implants influence peri-implant bone formation in a minipig model. J. Biomed. Mater. Res. A 2014, 102, 2334–2344. [Google Scholar] [CrossRef] [PubMed]
- Correia, C.R.; Moreira-Teixeira, L.S.; Moroni, L.; Reis, R.L.; van Blitterswijk, C.A.; Karperien, M.; Mano, J.F. Chitosan scaffolds containing hyaluronic acid for cartilage tissue engineering. Tissue Eng. Part C Methods 2011, 17, 717–730. [Google Scholar] [CrossRef] [PubMed]
- Dahiya, P.; Kamal, R. Hyaluronic acid: A boon in periodontal therapy. N. Am. J. Med. Sci. 2013, 5, 309–315. [Google Scholar] [CrossRef]
- Zhang, W.; Mu, H.; Zhang, A.; Cui, G.; Chen, H.; Duan, J.; Wang, S. A decrease in moisture absorption–retention capacity of N-deacetylation of hyaluronic acid. Glycoconj. J. 2013, 30, 577–583. [Google Scholar] [CrossRef]
- Kablik, J.; Monheit, G.D.; Yu, L.; Chang, G.; Gershkovich, J. Comparative physical properties of hyaluronic acid dermal fillers. Dermatol. Surg. 2009, 35 (Suppl. S1), 302–312. [Google Scholar] [CrossRef]
- Jentsch, H.; Pomowski, R.; Kundt, G.; Göcke, R. Treatment of gingivitis with hyaluronan. J. Clin. Periodontol. 2003, 30, 159–164. [Google Scholar] [CrossRef]
- Frenkel, J.S. The role of hyaluronan in wound healing. Int. Wound J. 2014, 11, 159–163. [Google Scholar] [CrossRef]
- D’Agostino, A.; Stellavato, A.; Busico, T.; Papa, T.; Tirino, V.; Papaccio, G.; La Gatta, A.; De Rosa, M.; Schiraldi, C. In vitro analysis of the effects on wound healing of high- and low-molecular weight chains of hyaluronan and their hybrid H-HA/L-HA complexes. BMC Cell Biol. 2015, 16, 19. [Google Scholar] [CrossRef] [PubMed]
- Voigt, J.; Driver, V.R. Hyaluronic acid derivatives and their healing effect on burns, epithelial surgical wounds, and chronic wounds: A systematic review and meta-analysis of randomized controlled trials. Wound Repair Regen. 2012, 20, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Neuman, M.G.; Nanau, R.M.; Oruña-Sanchez, L.; Coto, G. Hyaluronic acid and wound healing. J. Pharm. Pharm. Sci. 2015, 18, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Cowman, M.K.; Hittner, D.M.; Feder-Davis, J. 13C-NMR studies of hyaluronan: Conformational sensitivity to varied environments. Macromolecules 1996, 29, 2894–2902. [Google Scholar] [CrossRef]
- Ke, C.; Sun, L.; Qiao, D.; Wang, D.; Zeng, X. Antioxidant activity of low molecular weight hyaluronic acid. Food Chem. Toxicol. 2011, 49, 2670–2675. [Google Scholar] [CrossRef] [PubMed]
- Maharjan, A.S.; Pilling, D.; Gomer, R.H. High and low molecular weight hyaluronic acid differentially regulate human fibrocyte differentiation. PLoS ONE 2011, 6, e26078. [Google Scholar] [CrossRef] [PubMed]
- Rayahin, J.E.; Buhrman, R.S.; Zhang, Y.; Koh, T.J.; Gemeinhart, R.A. High and low molecular weight hyaluronic acid differentially influence macrophage activation. ACS Biomater. Sci. Eng. 2015, 13, 481–493. [Google Scholar] [CrossRef]
- Kavasi, R.M.; Berdiaki, A.; Spyridaki, I.; Corsini, E.; Tsatsakis, A.; Tzanakakis, G.; Nikitovic, D. HA metabolism in skin homeostasis and inflammatory disease. Food Chem. Toxicol. 2017, 101, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Trabucchi, E.; Pallotta, S.; Morini, M.; Corsi, F.; Franceschini, R.; Casiraghi, A.; Pravettoni, A.; Foschi, D.; Minghetti, P. Low molecular weight hyaluronic acid prevents oxygen free radical damage to granulation tissue during wound healing. Int. J. Tissue React. 2002, 24, 65–71. [Google Scholar]
- Gariboldi, S.; Palazzo, M.; Zanobbio, L.; Selleri, S.; Sommariva, M.; Sfondrini, L.; Cavicchini, S.; Balsari, A.; Rumio, C. Low molecular weight hyaluronic acid increases the self-defense of skin epithelium by induction of-defensin 2 via TLR2 and TLR4. J. Immunol. 2008, 181, 2103–2110. [Google Scholar] [CrossRef]
- Chen, H.; Qin, J.; Hu, Y. Efficient degradation of high-molecular-weight hyaluronic acid by a combination of ultrasound, hydrogen peroxide, and copper ion. Molecules 2019, 24, 617. [Google Scholar] [CrossRef] [PubMed]
- Hokputsa, S.; Jumel, K.; Alexander, C.; Harding, S.E. A comparison of molecular mass determination of hyaluronic acid using SEC/MALLS and sedimentation equilibrium. Eur. Biophys. J. 2003, 32, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Kim, J.K.; Kim, J.H.; Kweon, D.K.; Lee, J.W. Degradation of hyaluronic acid powder by electron beam irradiation, gamma ray irradiation, microwave irradiation and thermal treatment: A comparative study. Carbohydr. Polym. 2010, 79, 1080–1085. [Google Scholar] [CrossRef]
- Kim, J.K.; Sung, N.Y.; Srinivasan, P.; Choi, J.I.; Kim, S.K.; Oh, J.M.; Kim, J.H.; Song, B.S.; Park, H.J.; Byun, M.W.; et al. Effect of gamma irradiated hyaluronic acid on acetaminophen induced acute hepatotoxicity. Chem. Biol. Interact. 2008, 172, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Yue, W. Preparation of low-molecular-weight hyaluronic acid by ozone treatment. Carbohydr. Polym. 2012, 89, 709–712. [Google Scholar]
- Huang, Y.C.; Huang, K.U.; Yang, B.Y.; Ko, C.H.; Huang, H.M. Fabrication of novel hydrogel with berberine-enriched carboxymethylcellulose and hyaluronic acid as an anti-inflammatory barrier membrane. BioMed Res. Int. 2016, 3640182. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.E.; Heatley, F. Hyaluronan forms specific stable tertiary structures in aqueous solution: A 13C NMR study. Proc. Natl. Acad. Sci. USA 1999, 96, 4850–4855. [Google Scholar] [CrossRef]
- Scott, J.E.; Heatley, F. Biological properties of hyaluronan in aqueous solution are controlled and sequestered by reversible tertiary structures, defined by NMR Spectroscopy. Biomacromolecules 2002, 3, 547–553. [Google Scholar] [CrossRef]
- Nagasawa, N.; Mitomo, M.; Yoshii, F.; Kume, T. Radiation-induced degradation of sodium alginate. Polym. Degrad. Stab. 2000, 69, 279–285. [Google Scholar] [CrossRef]
- Gura, E.; Huckel, M.; Muller, P.J. Specific degradation of hyaluronic acid and its rheological properties. Polym. Degrad. Stab. 1998, 59, 297–302. [Google Scholar] [CrossRef]
- Lapčík, L., Jr.; Benešová, K.; Lapčík, L.; De Smedt, S.; Lapčíková, B. Chemical modification of hyaluronic acid: Alkylation. Int. J. Polym. Anal. Charact. 2010, 15, 486–496. [Google Scholar] [CrossRef]
- Haward, S.J.; Jaishankar, A.; Oliveira, M.S.N.; Alves, M.A.; McKinley, G.H. Extensional flow of hyaluronic acid solutions in an optimized microfluidic cross-slot device. Biomicrofluidics 2013, 7, 044108. [Google Scholar] [CrossRef] [PubMed]
- Necas, J.; Bartosikova, L.; Brauner, P.; Kolar, J. Hyaluronic acid (hyaluronan): A review. Vet. Med. Czech 2008, 53, 397–411. [Google Scholar] [CrossRef]
- Lönnroth, E.C. Toxicity of medical glove materials: A pilot study. Int. J. Occup. Saf. Ergon. 2005, 11, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Eskandarinia, A.; Kefayat, A.; Rafienia, M.; Agheb, M.; Navid, S.; Ebrahimpour, K. Cornstarch-based wound dressing incorporated with hyaluronic acid and propolis: In vitroand in vivo studies. Carbohydr. Polym. 2019, 15, 25–35. [Google Scholar] [CrossRef]
- Burns, J.W.; Colt, M.J.; Burgees, L.S.; Skinner, K.C. Preclinical evaluation of Seprafilm bioresorbable membrane. Eur. J. Surg. Suppl. 1997, 577, 40–48. [Google Scholar]
- Uchida, K.; Otake, M.; Inoue, Y.; Koike, K.; Matsushita, K.; Tanaka, K.; Inoue, Y.; Mohri, Y.; Kusunoki, M. Bacteriostatic effects of hyaluronan-based bioresorbable membrane. Surg. Sci. 2001, 2, 431–436. [Google Scholar] [CrossRef]
- Rekik, D.M.; Khedir, S.B.; Moalla, K.K.; Kammoun, N.G.; Rebai, T.; Sahnoun, Z. Evaluation of wound healing properties of grape seed, sesame, and fenugreek oils. Evid. Based Complement. Altern Med. 2016, 2016, 7965689. [Google Scholar]
- Saeidnia, S.; Manayi, A.; Gohari, A.R.; Abdollahi, M. The story of beta-sitosterol—A review. Eur. J. Med. Plants 2014, 4, 590–609. [Google Scholar] [CrossRef]
- Chen, C.C.; Nien, C.J.; Chen, L.G.; Huang, K.Y.; Chang, W.J.; Huang, H.M. Effects of Sapindus mukorossi seed oil on skin wound healing: In vivo and in vitro testing. Int. J. Mol. Sci. 2019, 20, 2579. [Google Scholar] [CrossRef]
- Mwipatayi, B.P.; Angel, D.; Norrish, J.; Hamilton, M.J.; Scott, A.; Sieunarine, K. The use of honey in chronic leg ulcers: A literature review. Prim. Intent. 2004, 12, 107–112. [Google Scholar]
| Sample | Molecular Weight (kDa) | Dispersity (Mw/Mn) | pH |
|---|---|---|---|
| HA0 | 2983.7 | 386.2 | 6.76 |
| HA20I | 232.4 | 3.7 | 5.95 |
| HA20II | 99.2 | 3.4 | 5.49 |
| HA40 | 141.8 | 3.2 | 5.54 |
| HA60 | 59.5 | 2.5 | 5.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.-C.; Huang, K.-Y.; Lew, W.-Z.; Fan, K.-H.; Chang, W.-J.; Huang, H.-M. Gamma-Irradiation-Prepared Low Molecular Weight Hyaluronic Acid Promotes Skin Wound Healing. Polymers 2019, 11, 1214. https://doi.org/10.3390/polym11071214
Huang Y-C, Huang K-Y, Lew W-Z, Fan K-H, Chang W-J, Huang H-M. Gamma-Irradiation-Prepared Low Molecular Weight Hyaluronic Acid Promotes Skin Wound Healing. Polymers. 2019; 11(7):1214. https://doi.org/10.3390/polym11071214
Chicago/Turabian StyleHuang, Yu-Chih, Kuen-Yu Huang, Wei-Zhen Lew, Kang-Hsin Fan, Wei-Jen Chang, and Haw-Ming Huang. 2019. "Gamma-Irradiation-Prepared Low Molecular Weight Hyaluronic Acid Promotes Skin Wound Healing" Polymers 11, no. 7: 1214. https://doi.org/10.3390/polym11071214
APA StyleHuang, Y.-C., Huang, K.-Y., Lew, W.-Z., Fan, K.-H., Chang, W.-J., & Huang, H.-M. (2019). Gamma-Irradiation-Prepared Low Molecular Weight Hyaluronic Acid Promotes Skin Wound Healing. Polymers, 11(7), 1214. https://doi.org/10.3390/polym11071214






