Structure–Property Relationships of Pure Cellulose and GO/CEL Membranes Regenerated from Ionic Liquid Solutions
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Cellulose Solution
2.3. GO Synthesis and Preparation of GO/DMF Dispersion
2.4. Pure Cellulose Membrane and GO/CEL Membrane Formation
2.5. Measurements of Water Flux
2.6. Measurements of Rejection
2.7. Analytical Methods
3. Results and Discussion
3.1. Pure Cellulose (CEL) Membranes
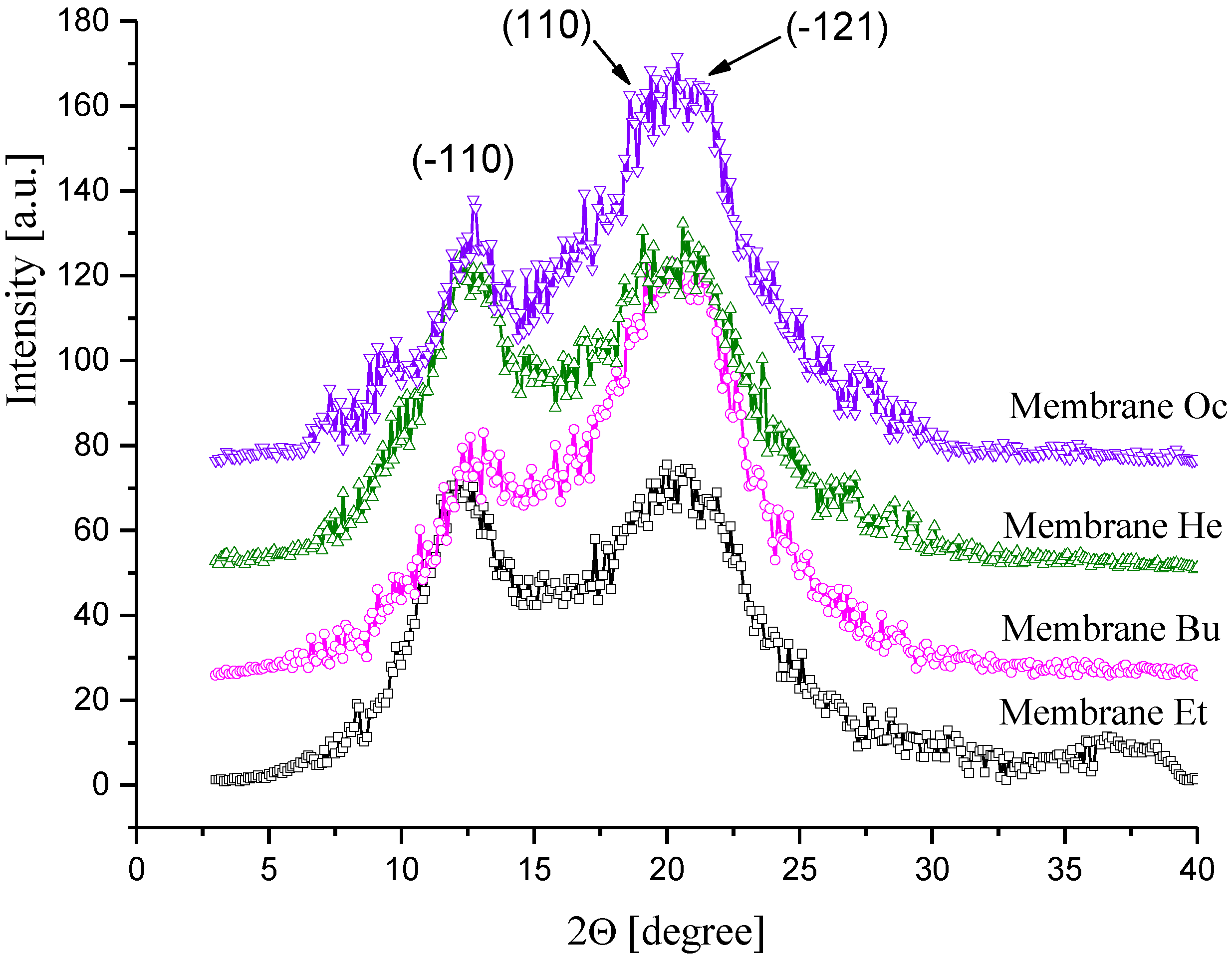
3.1.1. Porous Structure of CEL Membranes
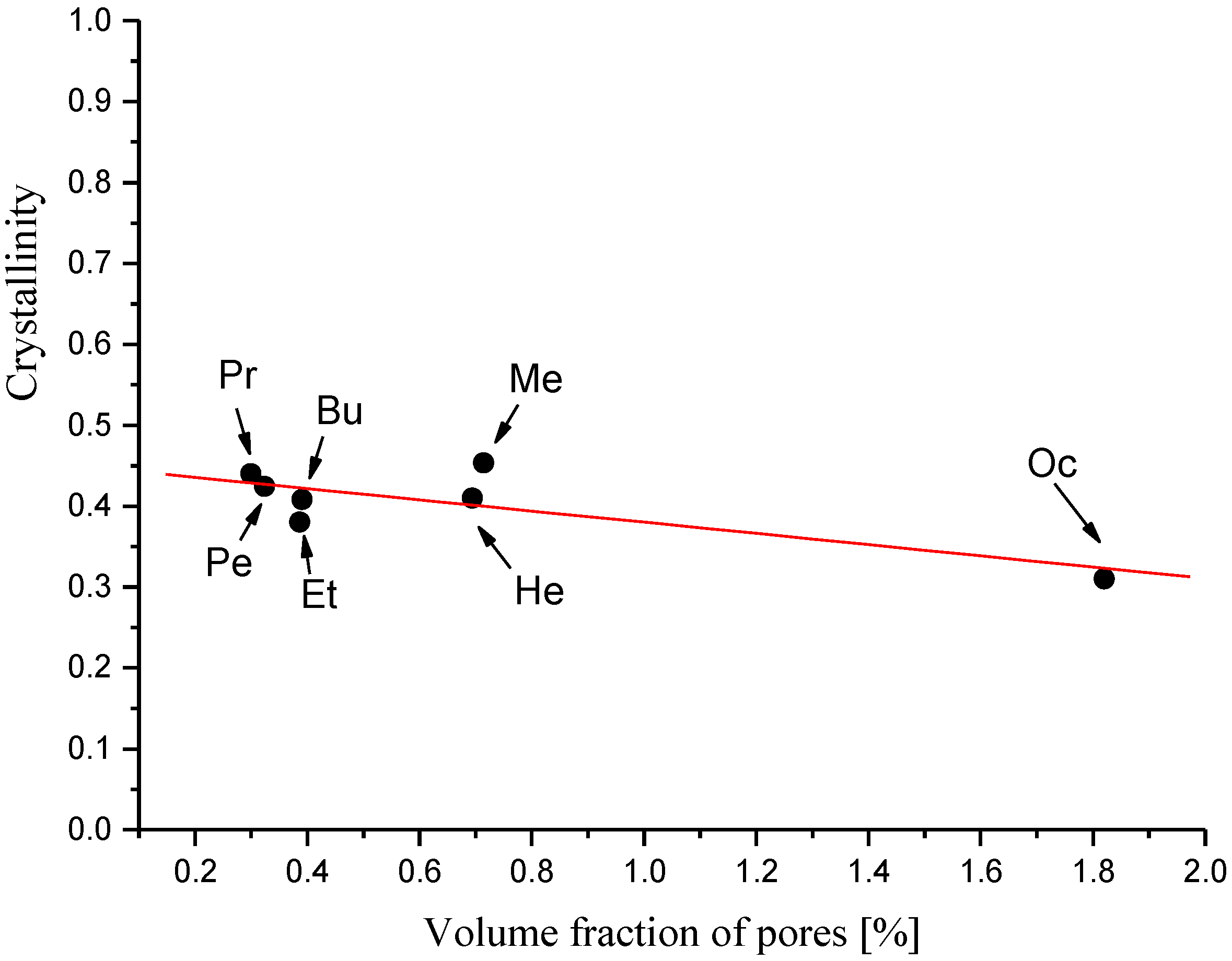
3.1.2. Morphology and Crystallinity of CEL Membranes
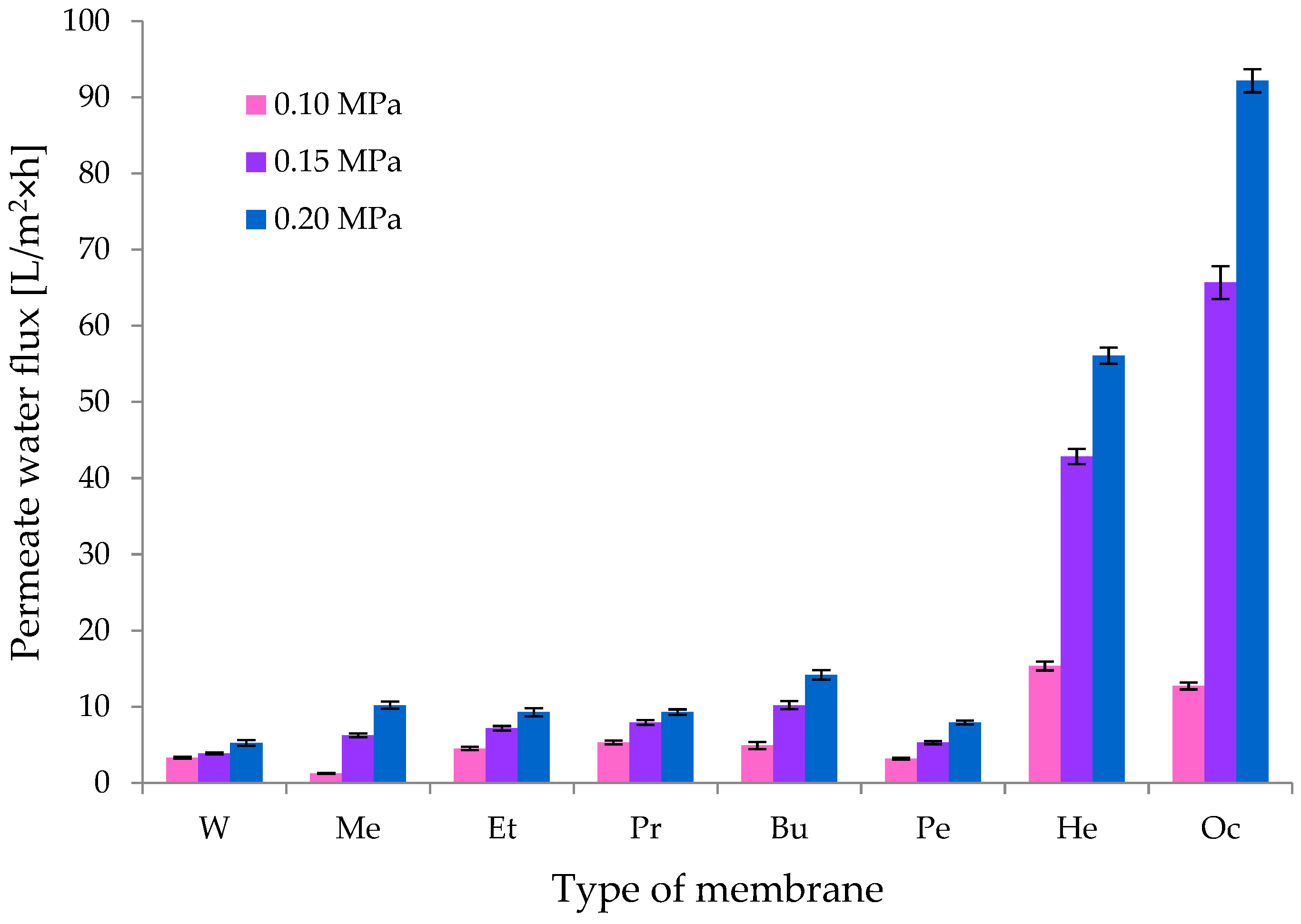
3.1.3. Transport Properties of CEL Membranes
3.1.4. Separation Properties of CEL Membranes
3.2. Graphene Oxide/Cellulose (GO/CEL) Membranes
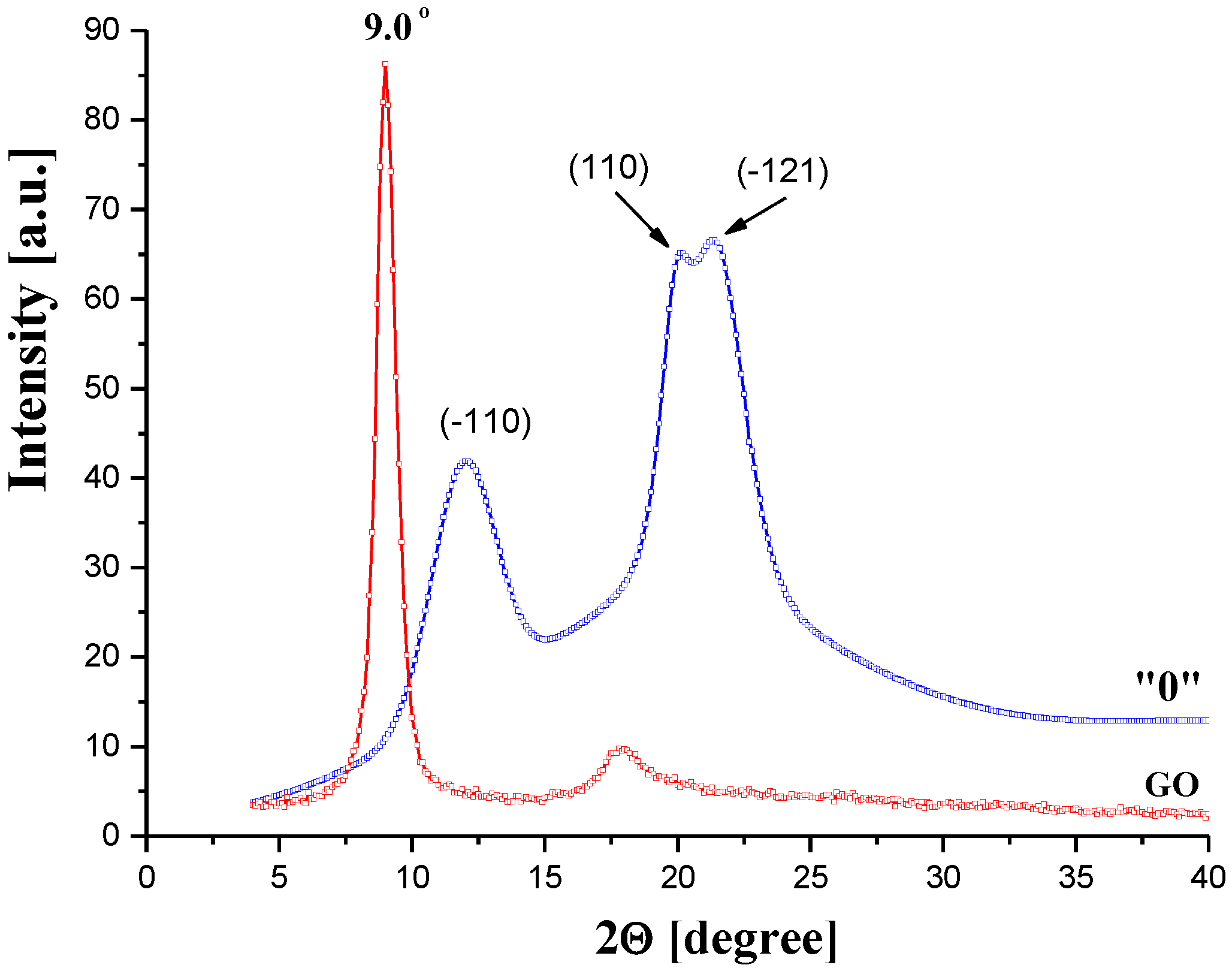
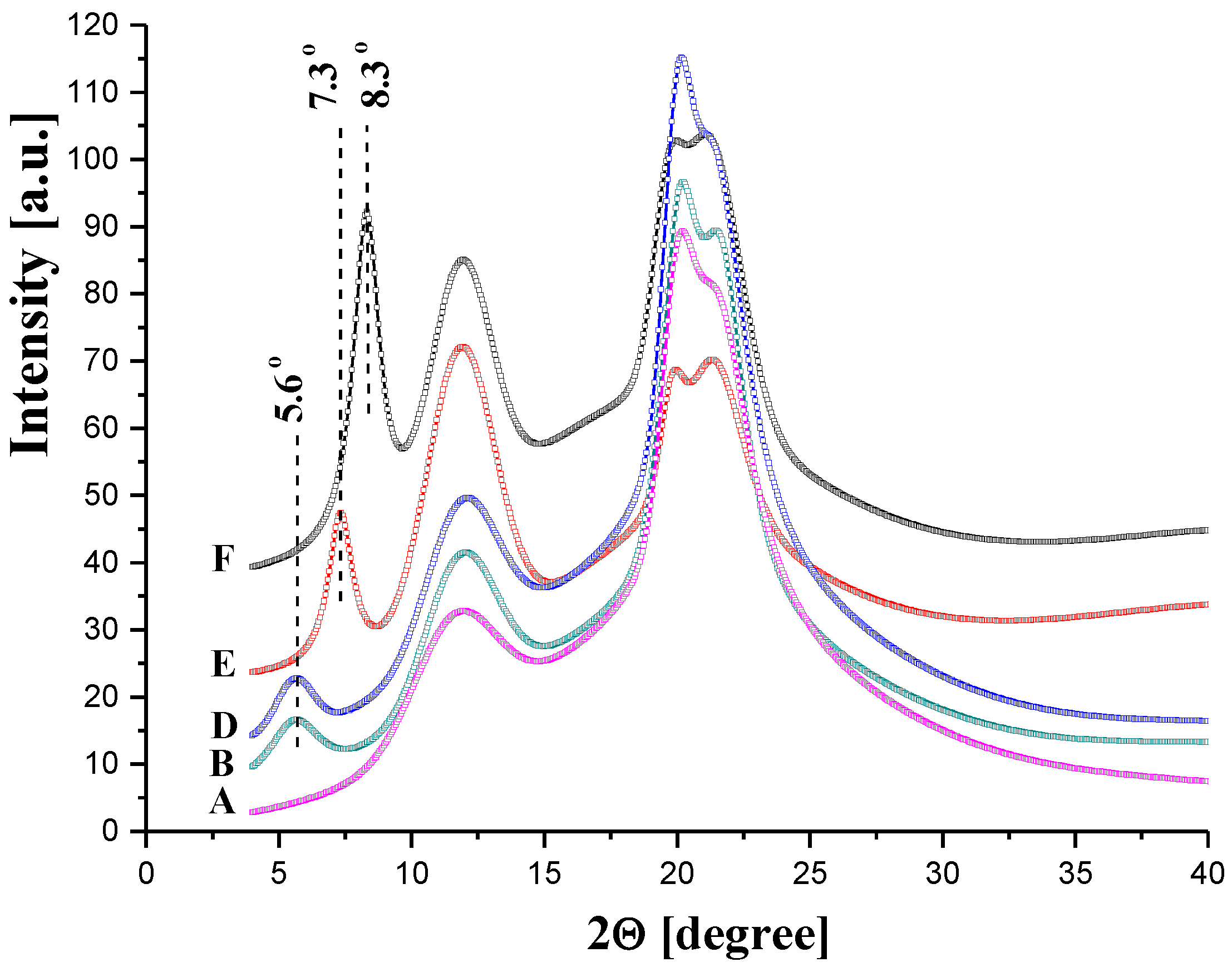
3.2.1. Morphology and Structure of GO/CEL Membranes
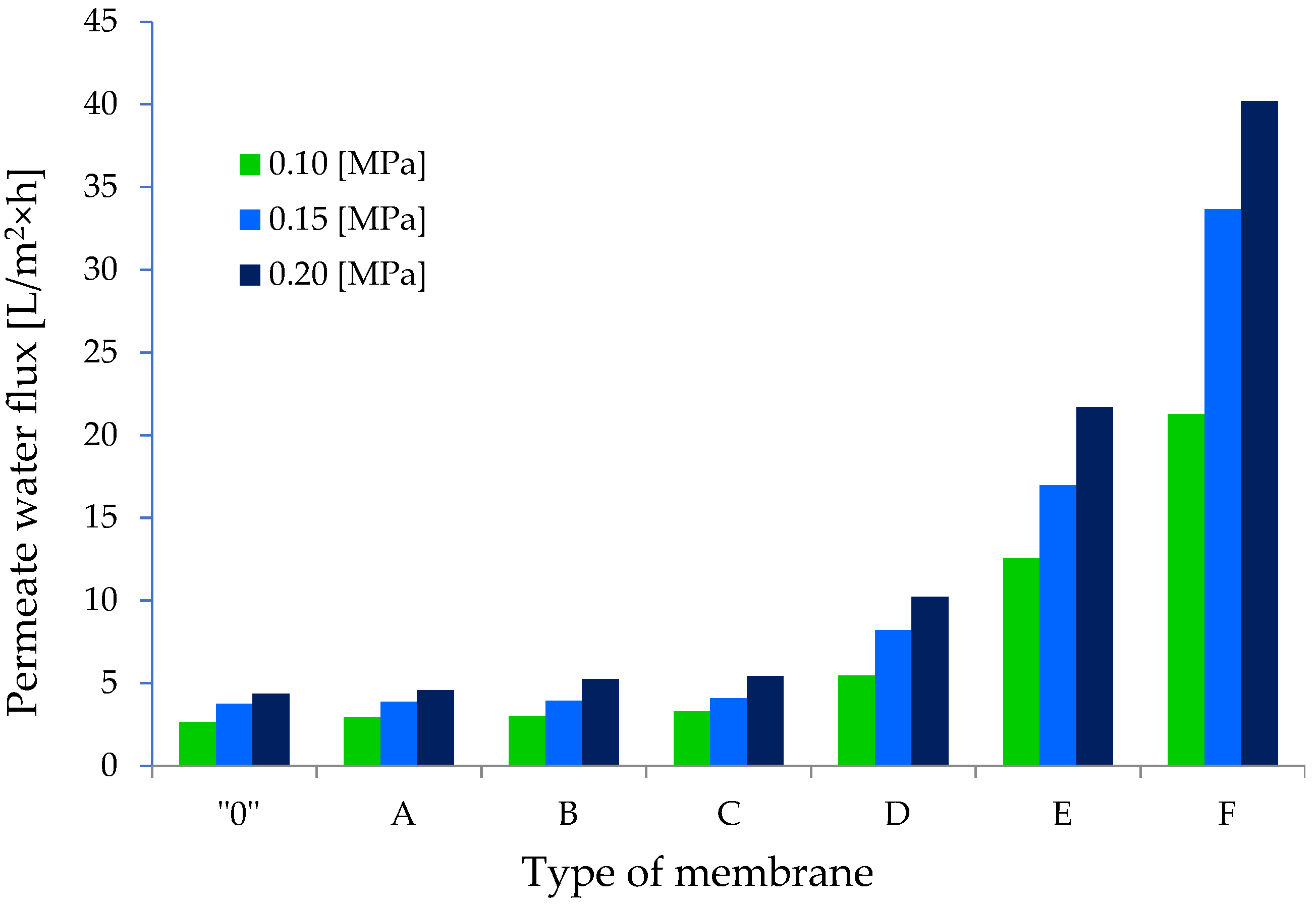
3.2.2. Transport Properties of GO/CEL Membranes
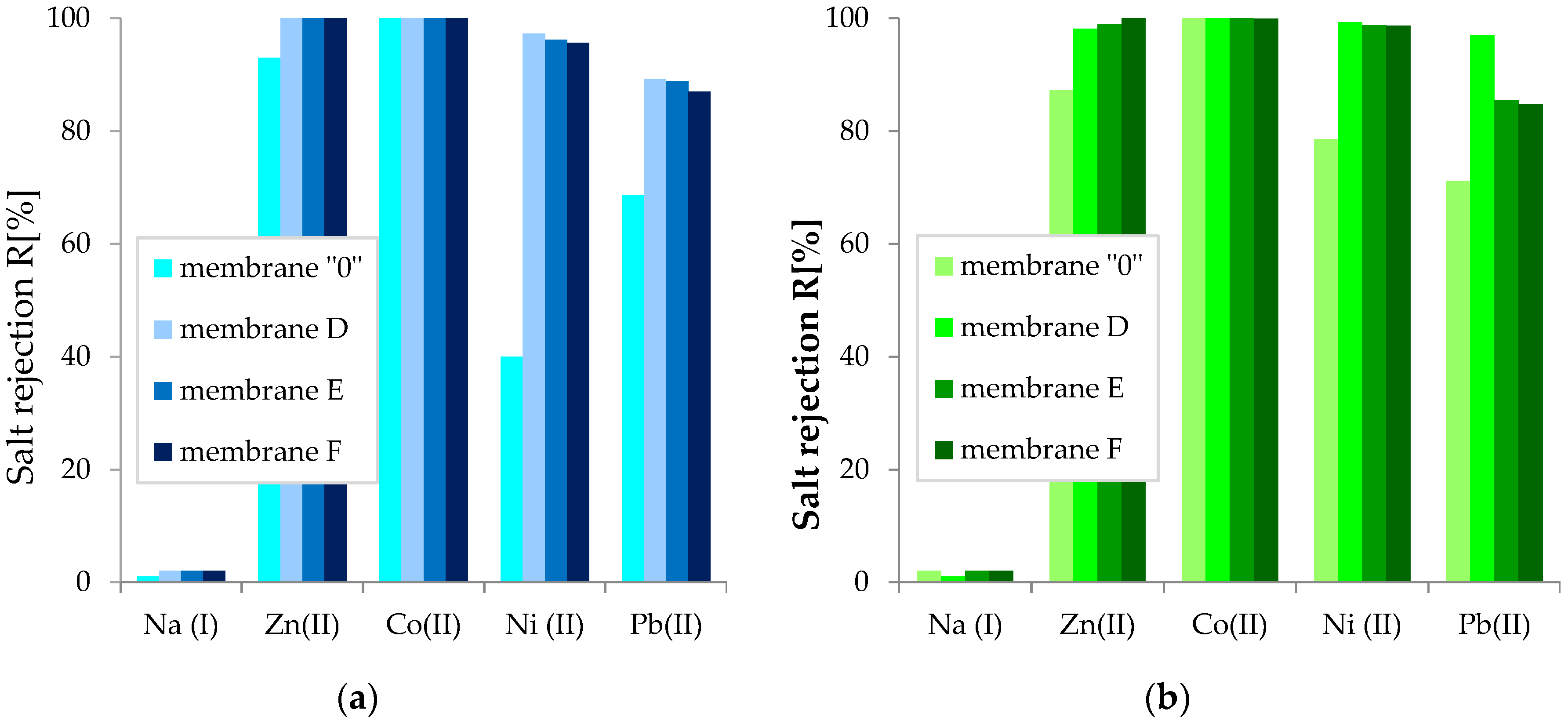
3.2.3. Separation Properties of GO/CEL Membranes
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ramamoorthy, S.K.; Skrifvars, M.; Persson, A. A review of natural fibers used in biocomposites: Plant, animal and regenerated cellulose fibers. Polym. Rev. 2015, 55, 107–162. [Google Scholar] [CrossRef]
- Klemm, D.; Heublein, B.; Fink, H.P.; Bohn, A. Cellulose: Fascinating biopolymer and sustainable raw material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Duarte, H.; Alves, L.; Antures, F.; Le Moigne, N.; Dormanns, J.; Duchemi, B.; Staiger, M.P. From cellulose dissolution and regeneration to added value applications—Synergism between molecular understanding and material development. In Cellulose—Fundamental Aspects and Current Trends. Cellulose—Fundamental Aspects and Current Trends; SE1 9SG; Poletto, M., Ed.; IntechOpen Limited: London, UK, 2015; pp. 237–261. [Google Scholar]
- Edgar, K.J.; Buchanan, C.M.; Debenham, J.S.; Rundquist, P.A.; Seiler, B.D.; Shelton, M.C.; Tindall, D. Advances in cellulose ester performance and application. Prog. Polym. Sci. 2001, 26, 1605–1688. [Google Scholar] [CrossRef]
- Jin, H.; Zha, C.; Gu, L. Direct dissolution of cellulose in NaOH/thiourea/urea aqueous solution. Carbohydr. Res. 2007, 342, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Fink, H.P.; Weigel, P.; Purz, H.J.; Ganster, J. Structure formation of regenerated cellulose materials from NMMO-solutions. Prog. Polym. Sci. 2001, 26, 1473–1524. [Google Scholar] [CrossRef]
- Gathergood, N.; Garcia, M.T.; Scammells, P.J. Biodegradable ionic liquids: Part I. Concept, preliminary targets and evaluation. Green Chem. 2004, 6, 166. [Google Scholar] [CrossRef]
- Jastorff, B.; Störmann, R.; Ranke, J.; Mölter, K.; Stock, F.; Oberheitmann, B.; Hoffmann, W.; Hoffmann, J.; Nüchter, M.; Ondruschka, B.; et al. How hazardous are ionic liquids? Structure-activity relationships and biological testing as important elements for sustainability evaluation. Green Chem. 2003, 5, 136–142. [Google Scholar] [CrossRef]
- Earle, M.J.; Seddon, K.R. Ionic liquids. Green solvents for the future. Pure Appl. Chem. 2000, 72, 1391–1398. [Google Scholar] [CrossRef]
- Novoselov, N.P.; Sashina, E.S.; Kuz’mina, O.G.; Troshenkova, S.V. Ionic liquids and their use for the dissolution of natural polymers. Russ. J. Gen. Chem. 2007, 77, 1395–1405. [Google Scholar] [CrossRef]
- Kosan, B.; Michels, C.; Meister, F. Dissolution and forming of cellulose with ionic liquids. Cellulose 2008, 15, 59–66. [Google Scholar] [CrossRef]
- Greaves, T.L.; Drummond, C.J. Solvent nanostructure, the solvophobic effect and amphiphile self-assembly in ionic liquids. Chem. Soc. Rev. 2013, 42, 1096–1120. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.M.; Xiao, Y.B.; Niu, K.; Zhao, Y.; Zhang, B.X.; Hu, B.Z. Functional ionic liquids for hydrolysis of lignocellulose. Carbohydr. Polym. 2013, 97, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.D.; Wu, Y.X.; Chen, Q.M.; Yu, Z.N.; Wang, C.W.; Jin, S.W. Dissolution of cellulose with ionic liquids and its application: A mini-review. Green Chem. 2006, 8, 325–327. [Google Scholar] [CrossRef]
- Hermanutz, F.; Gähr, F.; Uerdingen, E.; Meister, F.; Kosan, B. New developments in dissolving and processing of cellulose in ionic liquids. Macromol. Symp. 2008, 262, 23–27. [Google Scholar] [CrossRef]
- Swatloski, R.P.; Spear, S.K.; Holbrey, J.D.; Rogers, R.D. Dissolution of cellulose with ionic liquids. J. Am. Chem. Soc. 2002, 124, 4974–4975. [Google Scholar] [CrossRef]
- Wendler, F.; Todi, L.N.; Meister, F. Thermostability of imidazolium ionic liquids as direct solvents for cellulose. Thermochim. Acta 2012, 528, 76–84. [Google Scholar] [CrossRef]
- Mahadeva, S.K.; Kim, J. Influence of residual ionic liquid on the thermal stability and electromechanical behavior of cellulose regenerated from 1-ethyl-3-methylimidazolium acetate. Fibers Polym. 2012, 13, 289–294. [Google Scholar] [CrossRef]
- Sun, N.; Rahman, M.; Qin, Y.; Maxim, M.L.; Rodríguez, H.; Rogers, R.D. Complete dissolution and partial delignification of wood in the ionic liquid 1-ethyl-3-methylimidazolium acetate. Green Chem. 2009, 11, 646. [Google Scholar] [CrossRef]
- Livazovic, S.; Li, Z.; Behzad, A.R.; Peinemann, K.V.; Nunes, S.P. Cellulose multilayer membranes manufacture with ionic liquid. J. Membr. Sci. 2015, 490, 282–293. [Google Scholar] [CrossRef]
- Pinkert, A.; Marsh, K.N.; Pang, S.; Staiger, M.P. Ionic liquids and their interaction with cellulose. Chem. Rev. 2009, 109, 6712–6728. [Google Scholar] [CrossRef]
- Ruan, D.; Zhang, L.; Mao, Y.; Zeng, M.; Li, X. Microporous membranes prepared from cellulose in NaOH/thiourea aqueous solution. J. Membr. Sci. 2004, 241, 265–274. [Google Scholar] [CrossRef]
- Rambo, C.R.; Recouvreux, D.O.S.; Carminatti, C.A.; Pitlovanciv, A.K.; Antônio, R.V.; Porto, L.M. Template assisted synthesis of porous nanofibrous cellulose membranes for tissue engineering. Mater. Sci. Eng. C 2008, 28, 549–554. [Google Scholar] [CrossRef]
- Yadav, M. Study on thermal and mechanical properties of cellulose/iron oxide bionanocomposites film. Compos. Commun. 2018, 10, 1–5. [Google Scholar] [CrossRef]
- Rotaru, R.; Savin, M.; Tudorachi, N.; Peptu, C.; Samoila, P.; Sacarescu, L.; Harabagiu, V. Ferromagnetic iron oxide–cellulose nanocomposites prepared by ultrasonication. Polym. Chem. 2018, 9, 860–868. [Google Scholar] [CrossRef]
- Rac-Rumijowska, O.; Fiedot, M.; Karbownik, I.; Suchorska-Woźniak, P.; Teterycz, H. Synthesis of silver nanoparticles in NMMO and their in situ doping into cellulose fiber. Cellulose 2017, 24, 1355–1370. [Google Scholar] [CrossRef]
- Yue, X.; Zhang, T.; Yang, D.; Qiu, F.; Li, Z.; Wei, G.; Qiao, Y. Ag nanoparticles coated cellulose membrane with high infrared reflection, breathability and antibacterial property for human thermal insulation. J. Colloid Interface Sci. 2019, 535, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Suna, Z.; Yanga, L.; Zhangc, D.; Songa, W. High performance, flexible and renewable nano-biocomposite artificial muscle based on mesoporous cellulose/ionic liquid electrolyte membrane. Sens. Actuators B Chem. 2019, 283, 579–589. [Google Scholar] [CrossRef]
- Liu, P.; Zhu, C.; Mathew, A.P. Mechanically robust high flux graphene oxide—Nanocellulose membranes for dye removal from water. J. Hazard. Mater. 2019, 371, 484–493. [Google Scholar] [CrossRef]
- Lv, J.; Zhang, G.; Zhang, H.; Yang, F. Graphene oxide-cellulose nanocrystal (GO-CNC) composite functionalized PVDF membrane with improved antifouling performance in MBR: Behavior and mechanism. Chem. Eng. J. 2018, 352, 765–773. [Google Scholar] [CrossRef]
- Lyu, Y.; Zhang, Q.; Wang, Z.; Pu, J. A graphene oxide nanofiltration membrane intercalated with cellulose nano-crystals. BioResources 2018, 13, 9116–9131. [Google Scholar] [CrossRef]
- Yadav, M.; Rhee, K.Y.; Jung, I.H.; Park, S.J. Eco-friendly synthesis, characterization and properties of a sodium carboxymethyl cellulose/graphene oxide nanocomposite film. Cellulose 2013, 20, 687–698. [Google Scholar] [CrossRef]
- Yadav, M.; Rhee, K.Y.; Park, S.J. Synthesis and characterization of grapheneoxidecarboxymethylcellulose/alginate composite blend films. Carbohydr. Polym. 2014, 110, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.M.; Hu, Z.; Jiang, J. Mechanistic understanding of interactions between cellulose and ionic liquids: A molecular simulation study. Polymer 2011, 52, 5904–5911. [Google Scholar] [CrossRef]
- Tan, H.T.; Lee, K.T.; Mohamed, A.R. Pretreatment of lignocellulosic palm biomass using a solvent-ionic liquid [BMIM]Cl for glucose recovery: An optimisation study using response surface methodology. Carbohydr. Polym. 2011, 83, 1862–1868. [Google Scholar] [CrossRef]
- Feng, L.; Ian Chen, Z. Research progress on dissolution and functional modification of cellulose in ionic liquids. J. Mol. Liq. 2008, 142, 1–5. [Google Scholar] [CrossRef]
- Ding, Z.D.; Chi, Z.; Gu, W.X.; Gu, S.M.; Liu, J.H.; Wang, H.J. Theoretical and experimental investigation on dissolution and regeneration of cellulose in ionic liquid. Carbohydr. Polym. 2012, 89, 7–16. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Fryczkowska, B.; Sieradzka, M.; Sarna, E.; Fryczkowski, R.; Janicki, J. Influence of a graphene oxide additive and the conditions of membrane formation on the morphology and separative properties of poly(vinylidene fluoride) membranes. J. Appl. Polym. Sci. 2015, 132, 42789. [Google Scholar] [CrossRef]
- Moulik, S.; Vaishnavi, B.A.; Nagar, H.; Sridhar, S. Water competitive diffusion. In Encyclopedia of Membranes; Drioli, E., Giorno, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 1973–1983. [Google Scholar]
- Stabinger, H.; Kratky, O. A new technique for the measurement of absolute intensity of X-ray small-angle scattering. The moving slit method. Makromol. Chem. 1978, 179, 1655–1659. [Google Scholar] [CrossRef]
- Kratky, O.; Pilz, I.; Schmitz, P.J. Absolute intensity measurement of small angle X-ray scattering by means of standard sample. J. Colloid Interface Sci. 1966, 21, 24–34. [Google Scholar] [CrossRef]
- Crawshaw, J.; Cameron, R.E. A small angle X-ray scattering study of pore structure in Tencel cellulose fibres and the effects of physical treatments. Polymer 2000, 41, 4691–4698. [Google Scholar] [CrossRef]
- Rabiej, M. Application of the particle swarm optimization method for the analysis of wide-angle X-ray diffraction curves of semicrystalline polymers. J. Appl. Crystallogr. 2017, 50, 221–230. [Google Scholar] [CrossRef]
- Çifci, C.; Kaya, A. Preparation of poly(vinyl alcohol)/cellulose composite membranes for metal removal from aqueous solutions. Desalination 2010, 253, 175–179. [Google Scholar] [CrossRef]
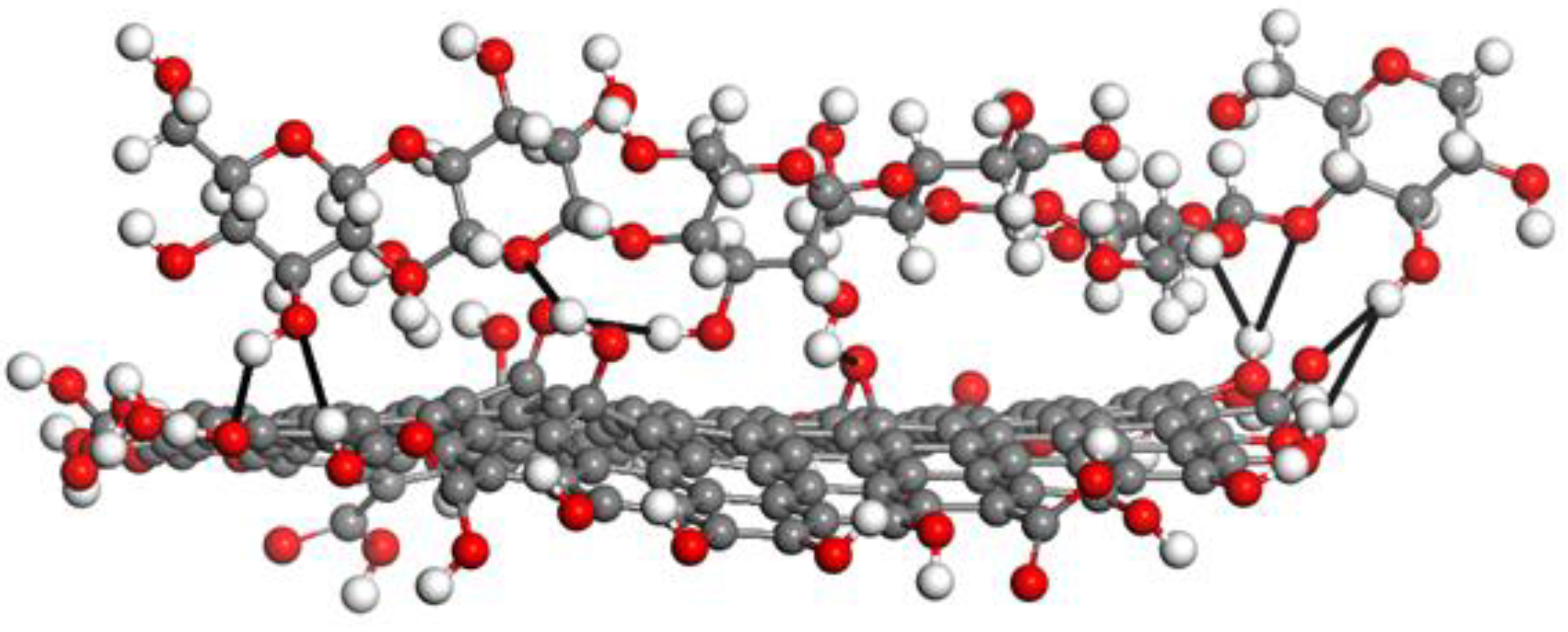
- Kongdee, A.; Bechtold, T. The complexation of Fe(III)-ions in cellulose fibres: A fundamental property. Carbohydr. Polym. 2004, 56, 47–53. [Google Scholar] [CrossRef]
- Guerrero-Contreras, J.; Caballero-Briones, F. Graphene oxide powders with different oxidation degree, prepared by synthesis variations of the Hummers method. Mater. Chem. Phys. 2015, 153, 209–220. [Google Scholar] [CrossRef]
- Ghosh, T.; Biswas, C.; Oh, J.; Arabale, G.; Hwang, T.; Luong, N.D.; Jin, M.; Lee, Y.H.; Do Nam, J. Solution-processed graphite membrane from reassembled graphene oxide. Chem. Mater. 2012, 24, 594–599. [Google Scholar] [CrossRef]
- Yoon, K.Y.; An, S.J.; Chen, Y.; Lee, J.H.; Bryant, S.L.; Ruoff, R.S.; Huh, C.; Johnston, K.P. Graphene oxide nanoplatelet dispersions in concentrated NaCl and stabilization of oil/water emulsions. J. Colloid Interface Sci. 2013, 403, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Texter, J. Graphene dispersions. Curr. Opin. Colloid Interface Sci. 2014, 19, 163–174. [Google Scholar] [CrossRef]
- Konkena, B.; Vasudevan, S. Understanding aqueous dispersibility of graphene oxide and reduced graphene oxide through p K a measurements. J. Phys. Chem. Lett. 2012, 3, 867–872. [Google Scholar] [CrossRef]
- Goh, K.; Setiawan, L.; Wei, L.; Jiang, W.; Wang, R.; Chen, Y. Fabrication of novel functionalized multi-walled carbon nanotube immobilized hollow fiber membranes for enhanced performance in forward osmosis process. J. Membr. Sci. 2013, 446, 244–254. [Google Scholar] [CrossRef]
- Das, R.; Ali, M.E.; Hamid, S.B.A.; Ramakrishna, S.; Chowdhury, Z.Z. Carbon nanotube membranes for water purification: A bright future in water desalination. Desalination 2014, 336, 97–109. [Google Scholar] [CrossRef]
- Hinds, B.J.; Chopra, N.; Rantell, T.; Andrews, R.; Gavalas, V.; Bachas, L.G. Aligned multiwalled carbon nanotube membranes. Science 2004, 303, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Celik, E.; Park, H.; Choi, H.; Choi, H. Carbon nanotube blended polyethersulfone membranes for fouling control in water treatment. Water Res. 2011, 45, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, K.A.; Mansoor, B.; Mansour, A.; Khraisheh, M. Functional graphene nanosheets: The next generation membranes for water desalination. Desalination 2015, 356, 208–225. [Google Scholar] [CrossRef]
- Han, Y.; Xu, Z.; Gao, C. Ultrathin graphene nanofiltration membrane for water purification. Adv. Funct. Mater. 2013, 23, 3693–3700. [Google Scholar] [CrossRef]
- Joshi, R.K.; Alwarappan, S.; Yoshimura, M.; Sahajwalla, V.; Nishina, Y. Graphene oxide: The new membrane material. Appl. Mater. Today 2015, 1, 1–12. [Google Scholar] [CrossRef]
- Goh, P.S.; Ismail, A.F. Graphene-based nanomaterial: The state-of-the-art material for cutting edge desalination technology. Desalination 2015, 356, 115–128. [Google Scholar] [CrossRef]
- Nair, R.R.; Wu, H.A.; Jayaram, P.N.; Grigorieva, I.V.; Geim, A.K. Unimpeded permeation of water through helium-leak-tight graphene-based membranes. Science 2012, 335, 442–444. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, M.; Roy, S.; Mitra, S. Desalination across a graphene oxide membrane via direct contact membrane distillation. Desalination 2016, 378, 37–43. [Google Scholar] [CrossRef]
- Zhang, X.; Yu, H.; Yang, H.; Wan, Y.; Hu, H.; Zhai, Z.; Qin, J. Graphene oxide caged in cellulose microbeads for removal of malachite green dye from aqueous solution. J. Colloid Interface Sci. 2015, 437, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Phiria, J.; Johanssona, L.S.; Ganea, P.; Maloney, T. A comparative study of mechanical, thermal and electrical properties ofgraphene-, graphene oxide- and reduced graphene oxide-dopedmicrofibrillated cellulose nanocomposites. Compos. Part B 2018, 147, 104–113. [Google Scholar] [CrossRef]
- Fryczkowska, B.; Wiechniak, K. Preparation and properties of cellulose membranes with graphene oxide addition. Pol. J. Chem. Technol. 2017, 19, 41–49. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, F.; Wang, Y.; Lin, H.; Han, L. A tight nanofiltration membrane with multi-charged nanofilms for high rejection to concentrated salts. J. Membr. Sci. 2017, 537, 407–415. [Google Scholar] [CrossRef]
- Song, B.; Zhang, C.; Zeng, G.; Gong, J.; Chang, Y.; Jiang, Y. Antibacterial properties and mechanism of graphene oxide-silver nanocomposites as bactericidal agents for water disinfection. Arch. Biochem. Biophys. 2016, 604, 167–176. [Google Scholar] [CrossRef]
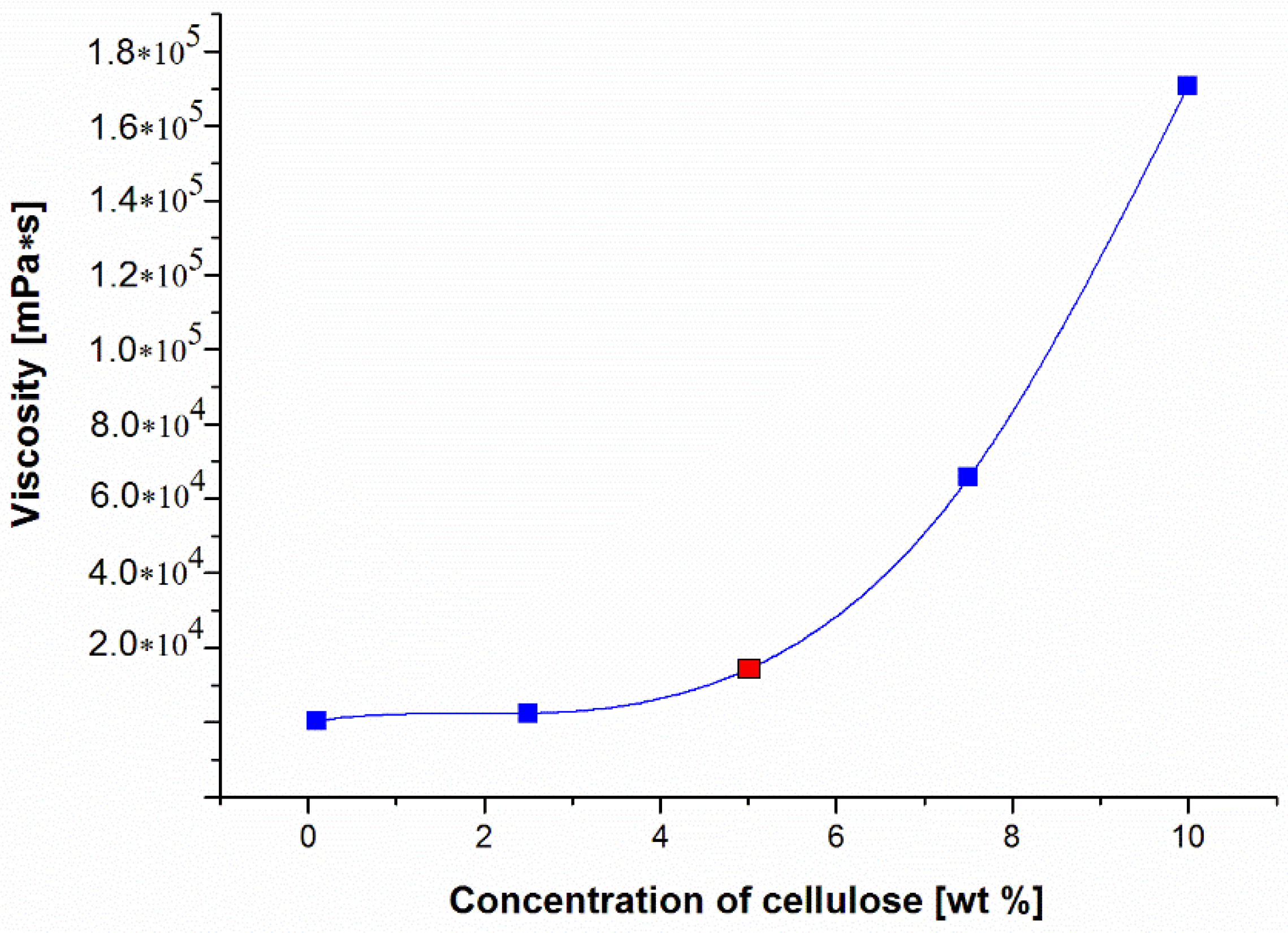


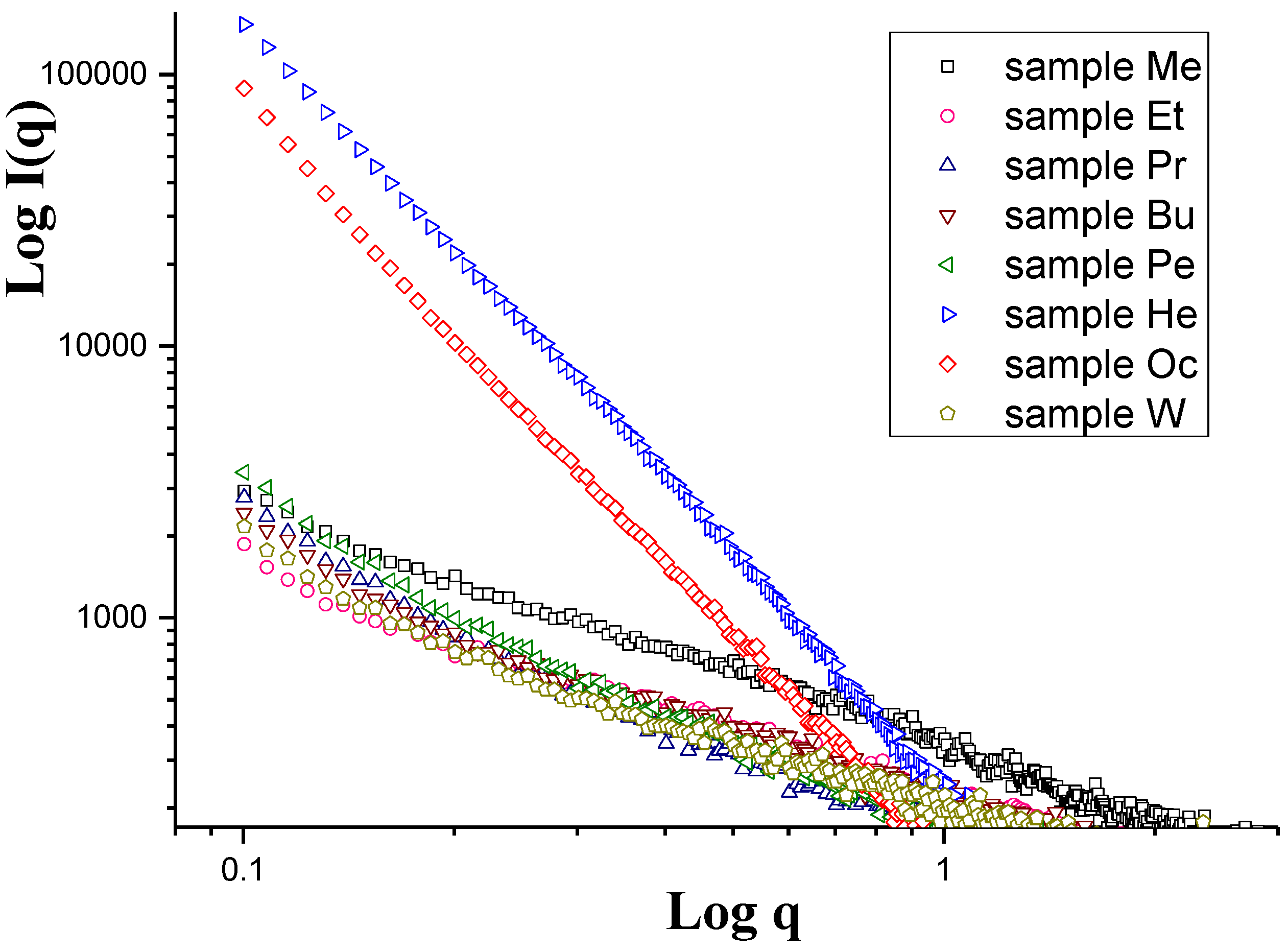
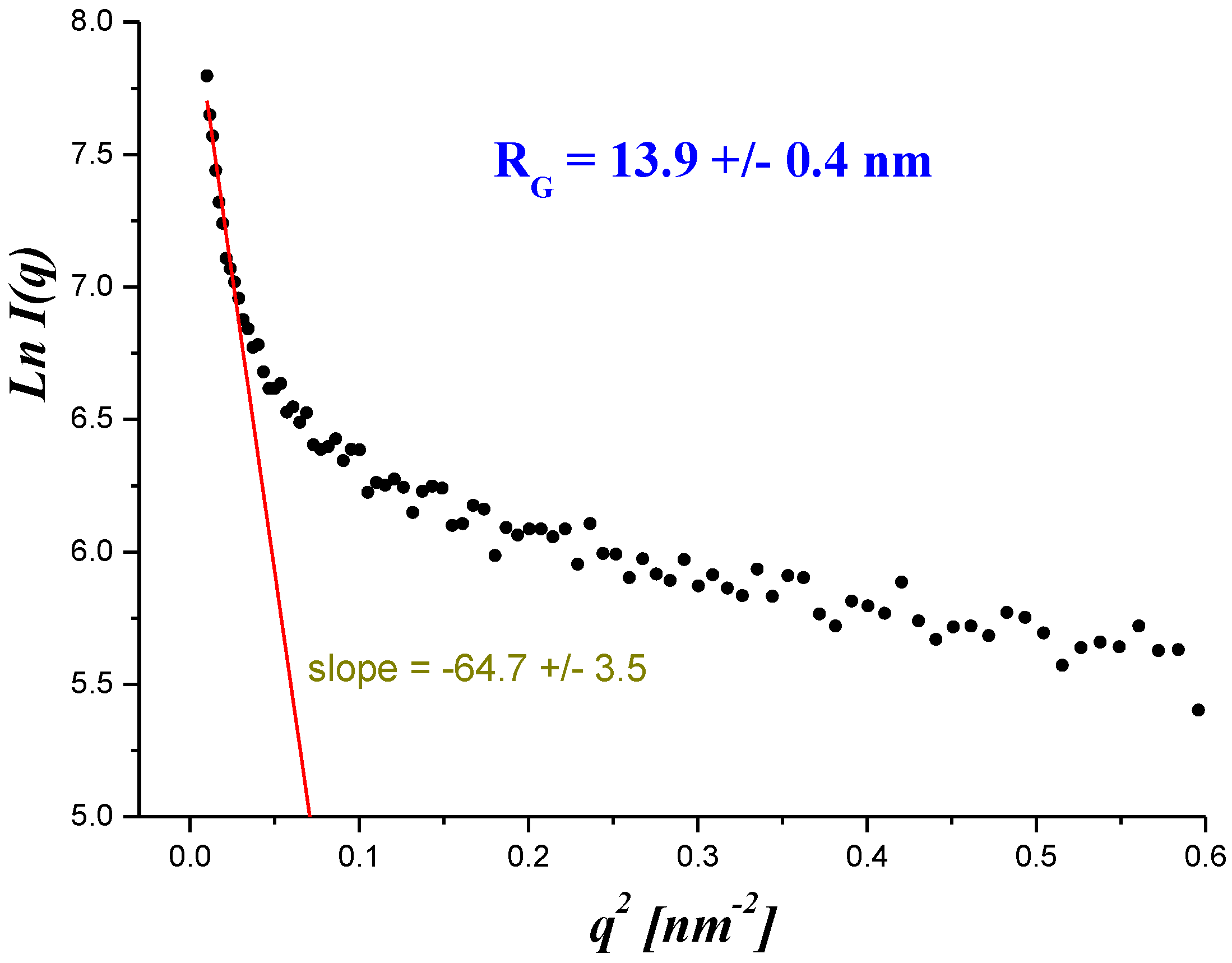
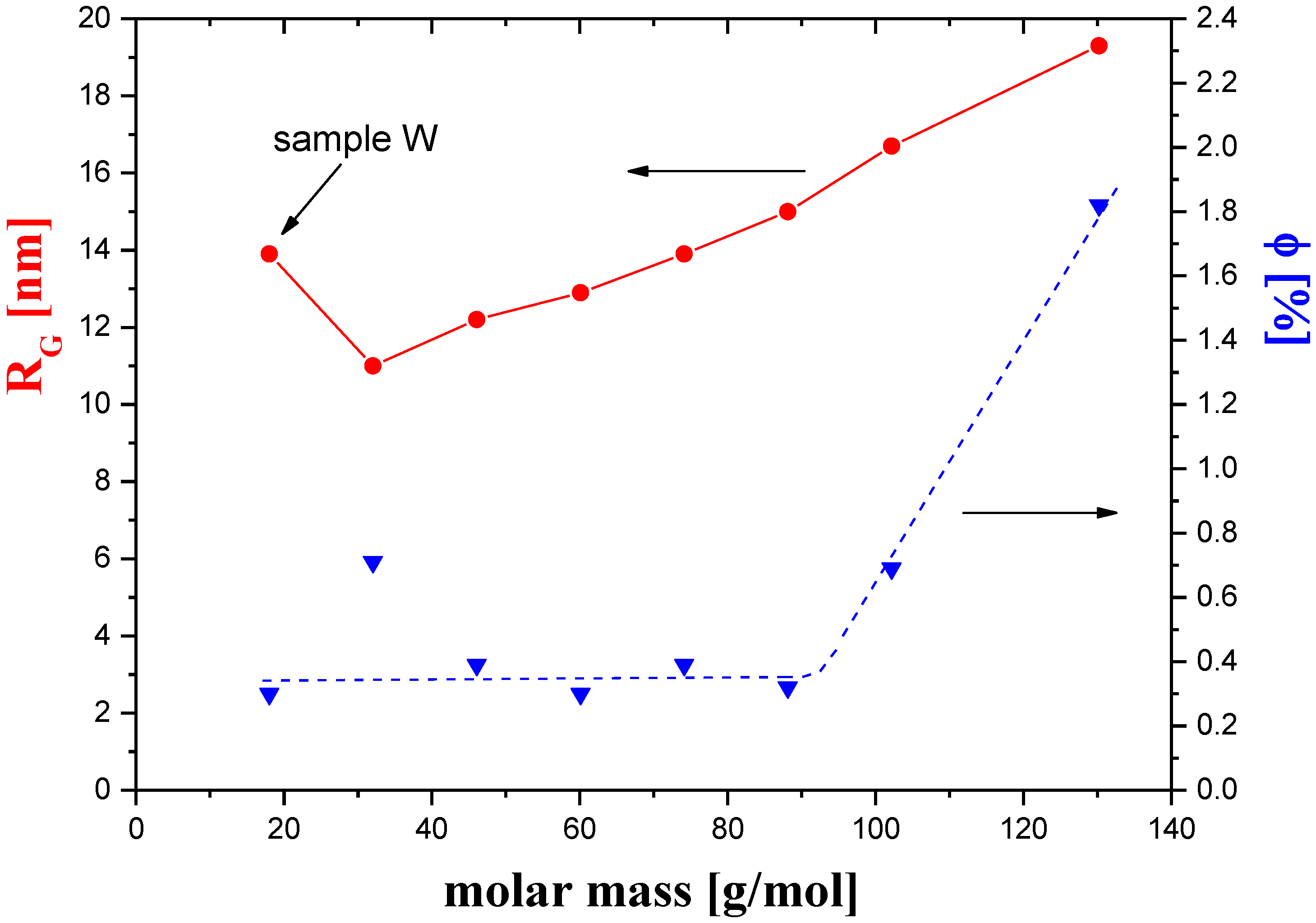




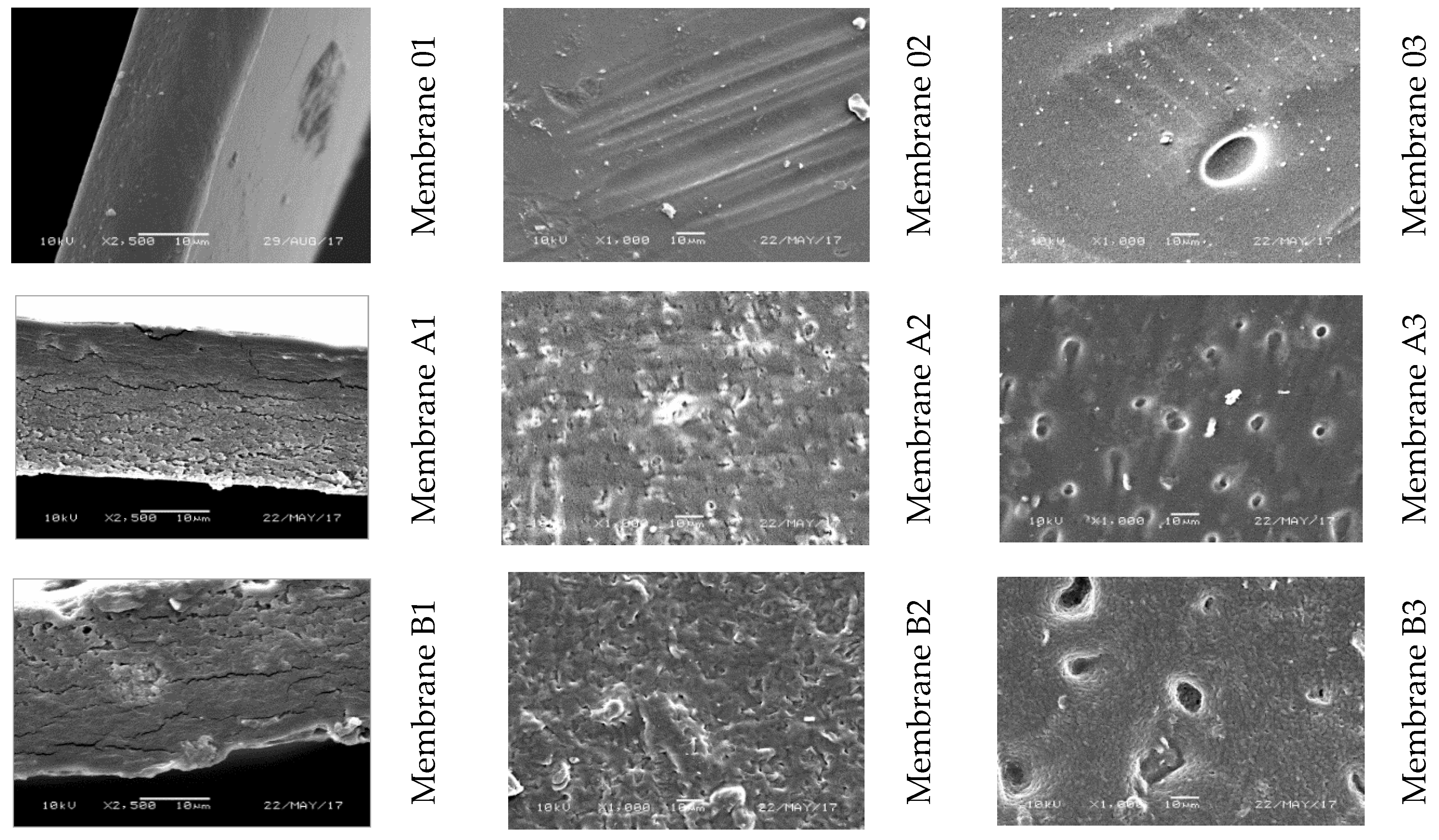
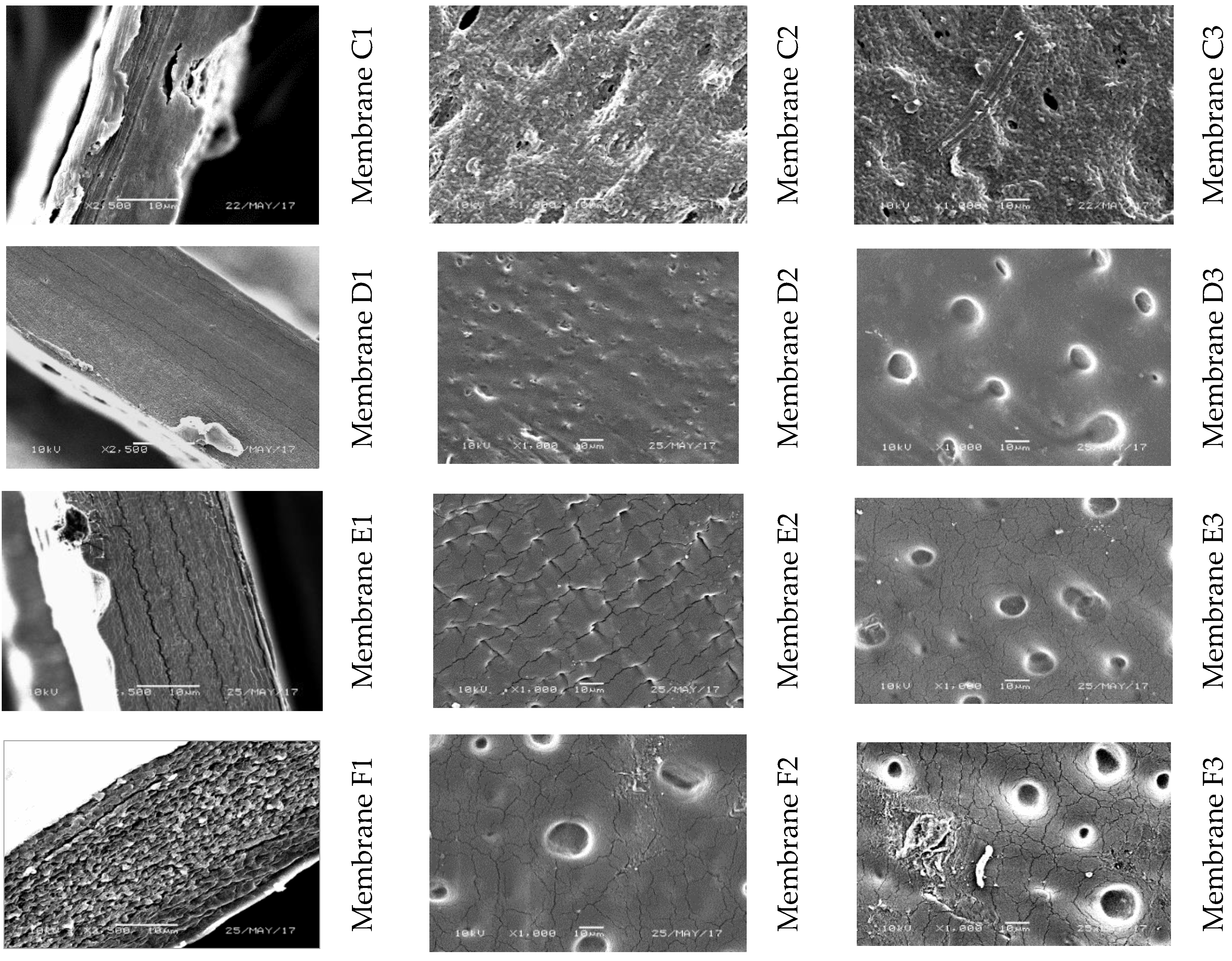
| Membrane Designation | “0” | A | B | C | D | E | F |
|---|---|---|---|---|---|---|---|
| Amount of 5.2% GO/DMF Solution (g) | 0 | 0.05 | 0.1 | 0.5 | 0.97 | 4.8 | 9.7 |
| Amount of CEL (g) | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 |
| Amount of EMIMAc (g) | 47.5 | 47.45 | 47.4 | 47.0 | 46.53 | 42.7 | 37.8 |
| Concentration of GO (% w/w) | 0 | 0.1 | 0.2 | 1 | 2 | 10 | 20 |
| Concentration of CEL (% w/w) | 100 | 99.9 | 99.8 | 99 | 98 | 90 | 80 |
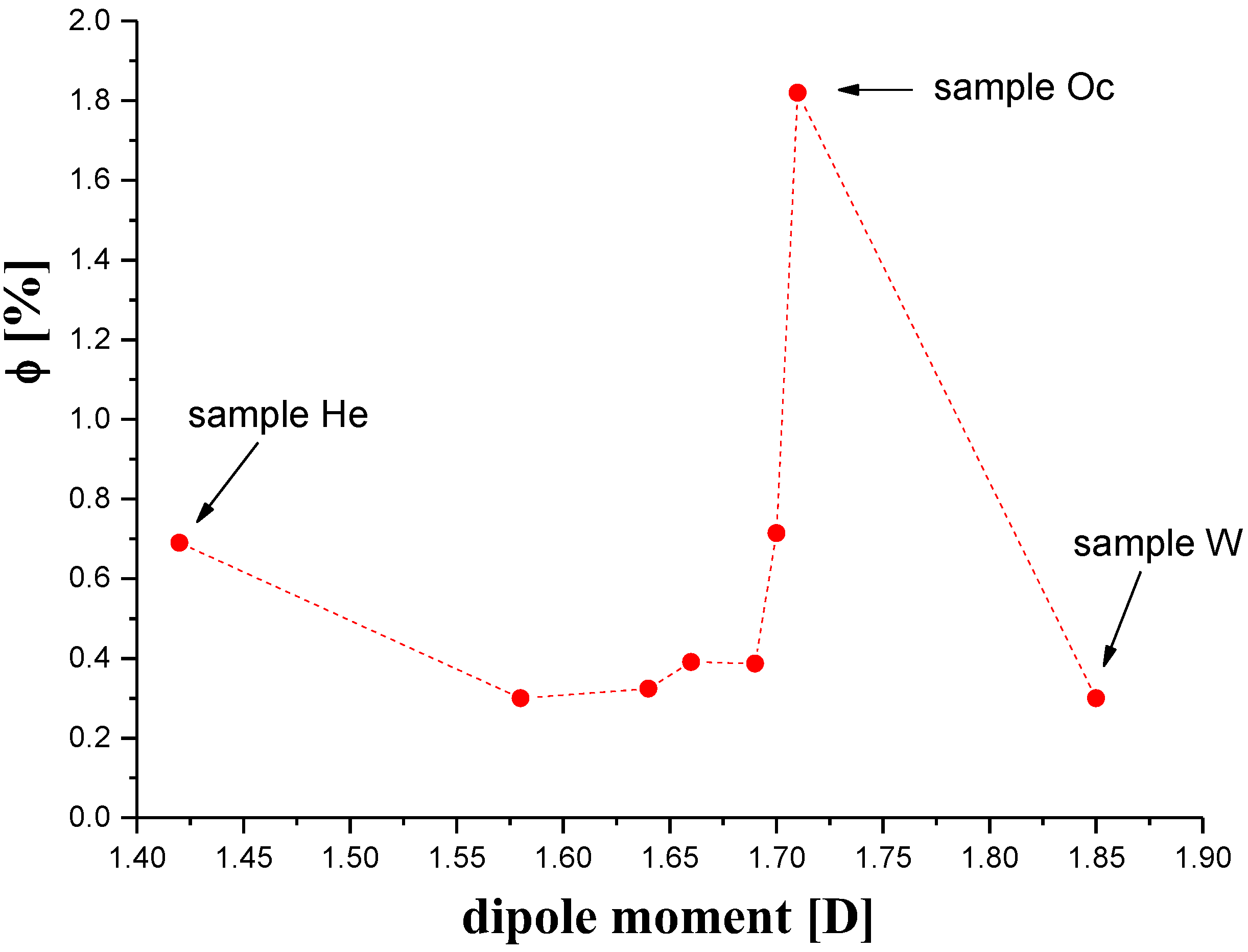
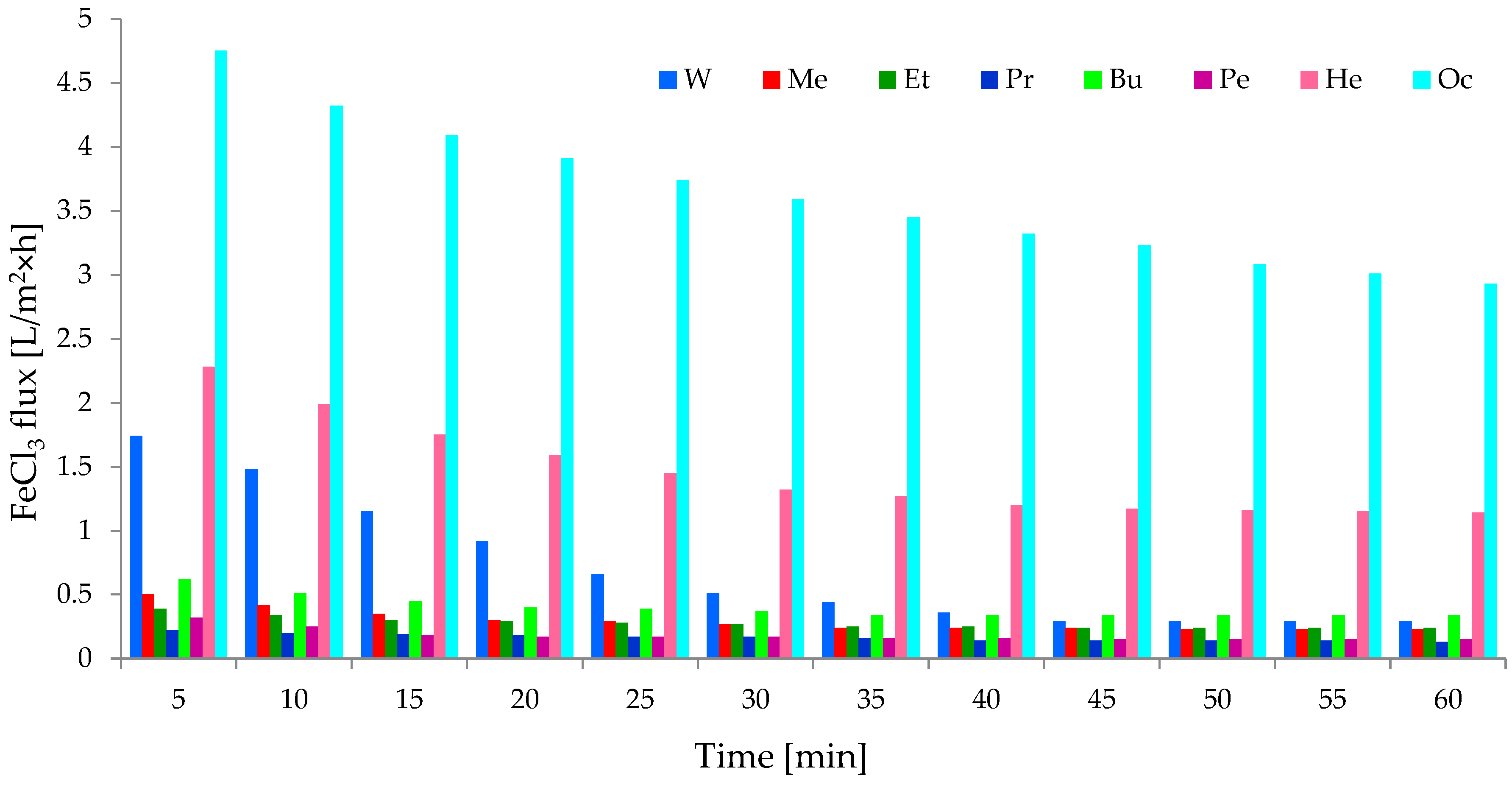
| Sample | Coagulant | SAXS Results | |||
|---|---|---|---|---|---|
| Type | Molar Mass (g/mol) | Dipole Moment (D) | RG (nm) | Φ (%) | |
| Me | methanol | 32.04 | 1.70 | 11.0 ± 0.3 | 0.71 |
| Et | ethanol | 46.07 | 1.69 | 12.2 ± 0.2 | 0.39 |
| Pr | 1-propanol | 60.1 | 1.58 | 12.9 ± 0.3 | 0.30 |
| Bu | 1-butanol | 74.12 | 1.66 | 13.9 ± 0.4 | 0.39 |
| Pe | 1-pentanol | 88.15 | 1.64 | 15.0 ± 0.4 | 0.32 |
| He | 1-hexanol | 102.17 | 1.42 | 16.7 ± 0.3 | 0.69 |
| Oc | 1-octanol | 130.23 | 1.71 | 19.3 ± 0.5 | 1.82 |
| W | water | 18.02 | 1.85 | 13.9 ± 0.3 | 0.30 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ślusarczyk, C.; Fryczkowska, B. Structure–Property Relationships of Pure Cellulose and GO/CEL Membranes Regenerated from Ionic Liquid Solutions. Polymers 2019, 11, 1178. https://doi.org/10.3390/polym11071178
Ślusarczyk C, Fryczkowska B. Structure–Property Relationships of Pure Cellulose and GO/CEL Membranes Regenerated from Ionic Liquid Solutions. Polymers. 2019; 11(7):1178. https://doi.org/10.3390/polym11071178
Chicago/Turabian StyleŚlusarczyk, Czesław, and Beata Fryczkowska. 2019. "Structure–Property Relationships of Pure Cellulose and GO/CEL Membranes Regenerated from Ionic Liquid Solutions" Polymers 11, no. 7: 1178. https://doi.org/10.3390/polym11071178
APA StyleŚlusarczyk, C., & Fryczkowska, B. (2019). Structure–Property Relationships of Pure Cellulose and GO/CEL Membranes Regenerated from Ionic Liquid Solutions. Polymers, 11(7), 1178. https://doi.org/10.3390/polym11071178





