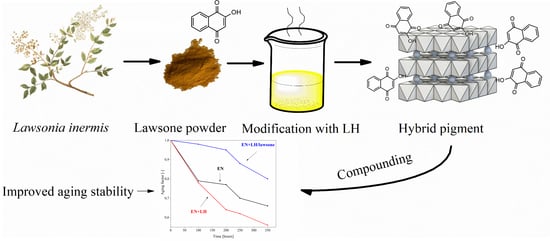
Improved Aging Stability of Ethylene-Norbornene Composites Filled with Lawsone-Based Hybrid Pigment
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Pigment Preparation
2.3. Composite Preparation
2.4. Pigment Characterization Techniques
2.5. Composite Characterization Techniques
3. Results
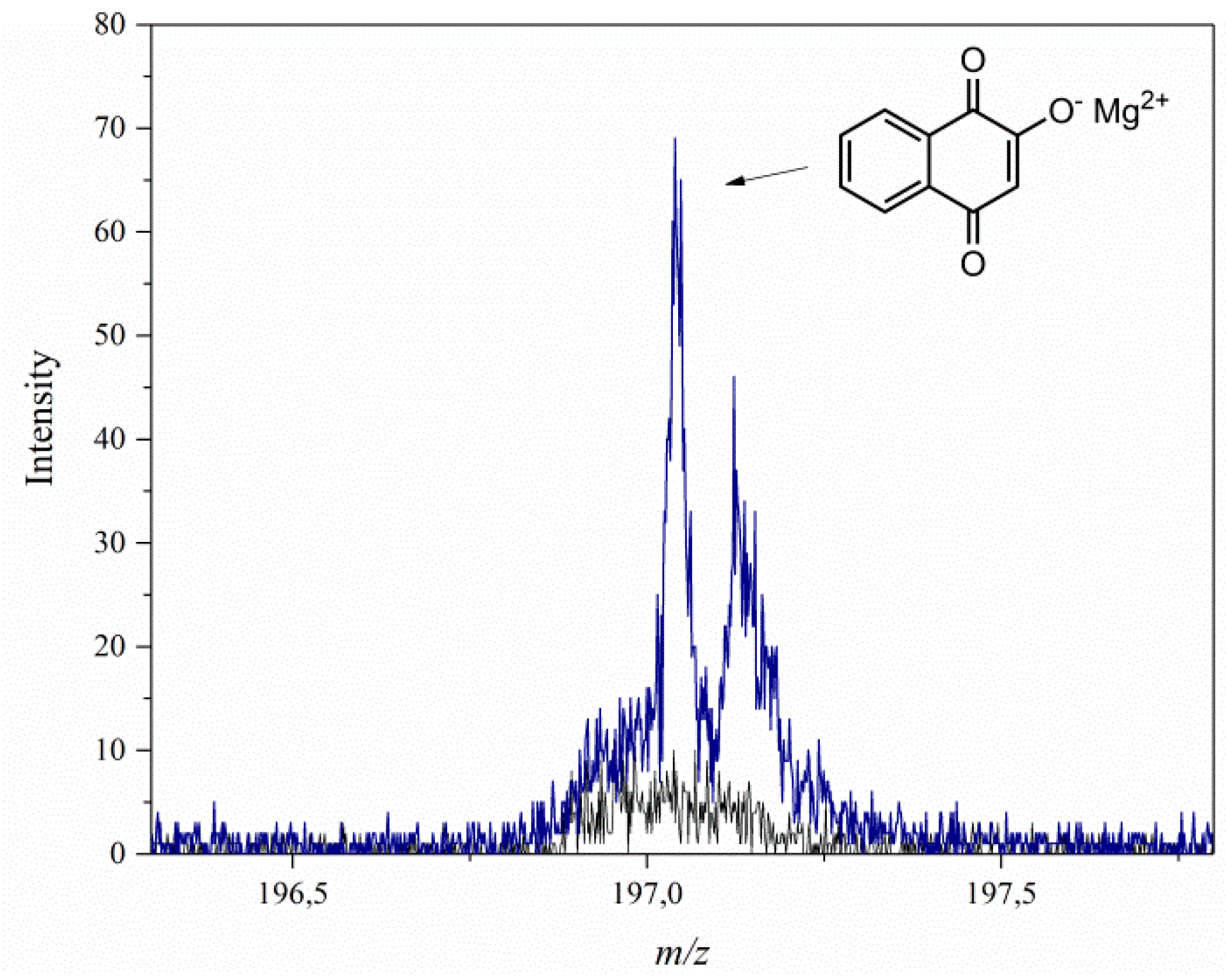
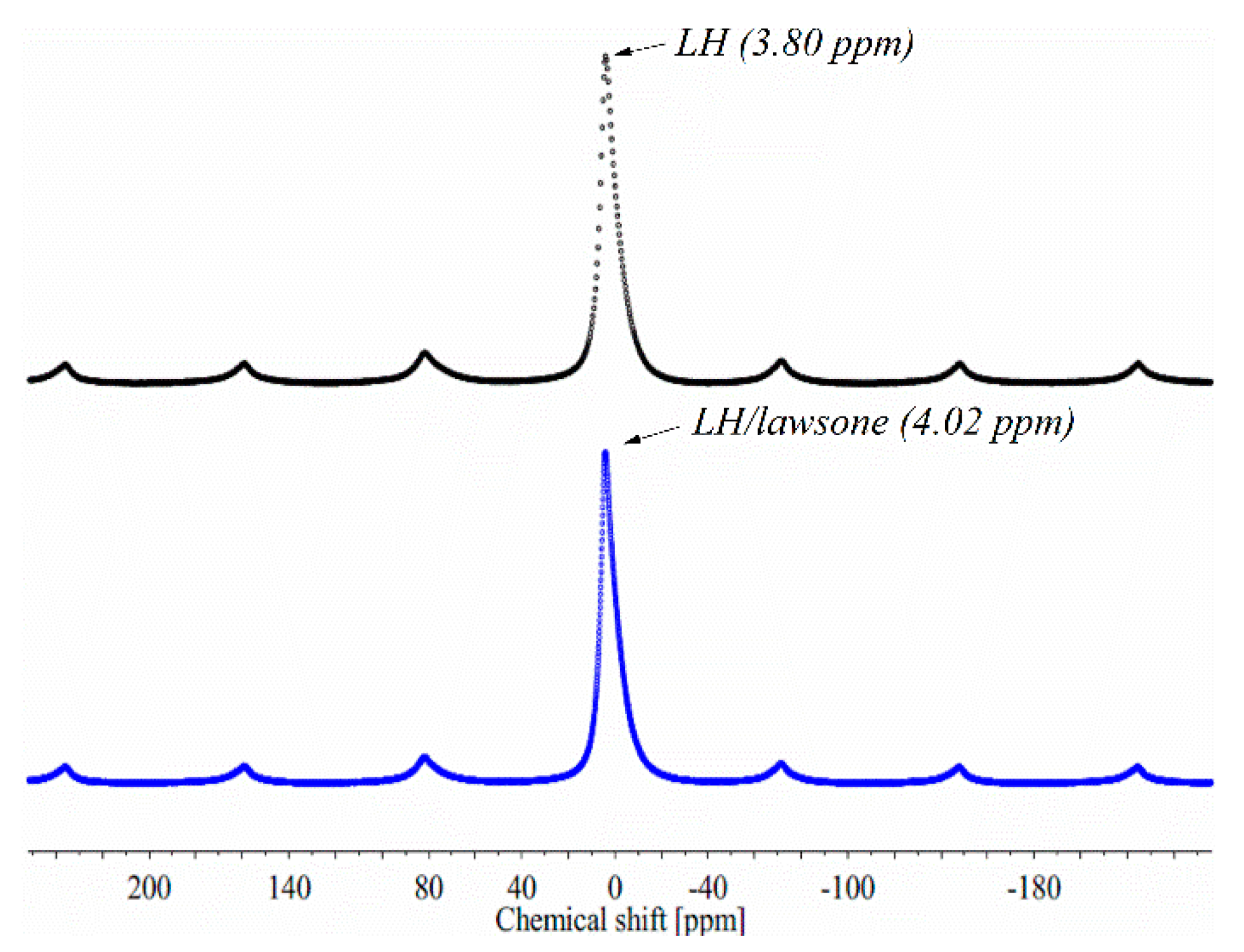
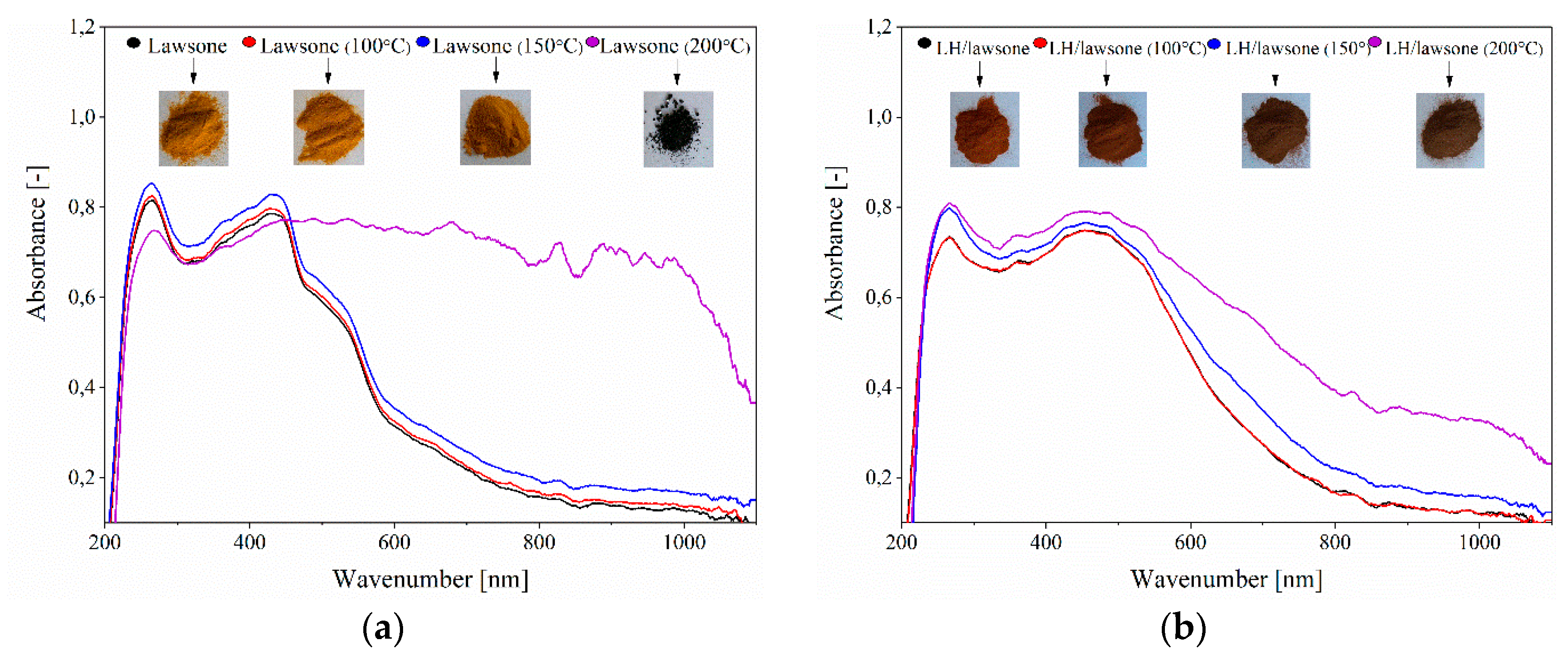
3.1. Secondary Ion Mass Spectrometry (TOF-SIMS) and 27Al NMR Spectroscopy
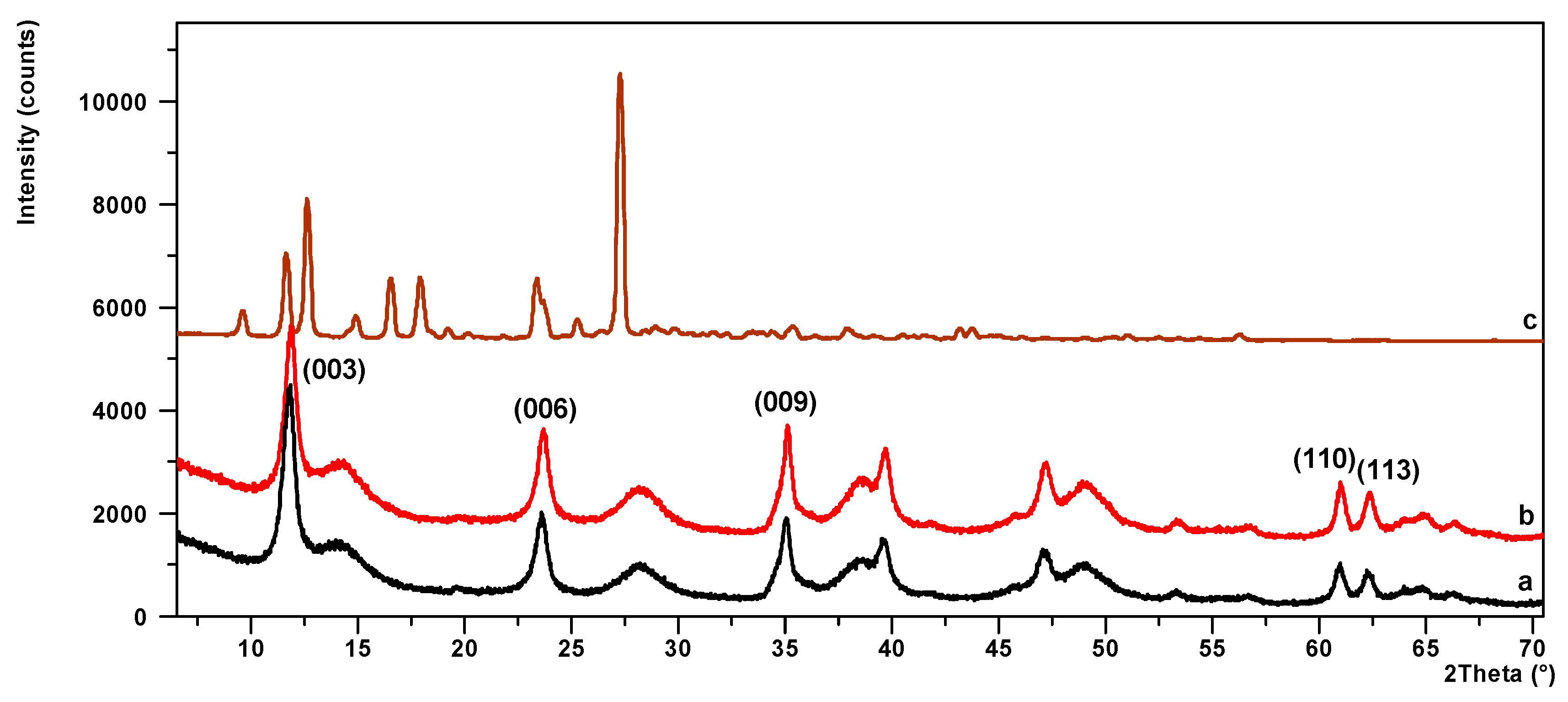
3.2. Powder X-ray Diffraction (PXRD) and Scanning Electron Microscopy (SEM)
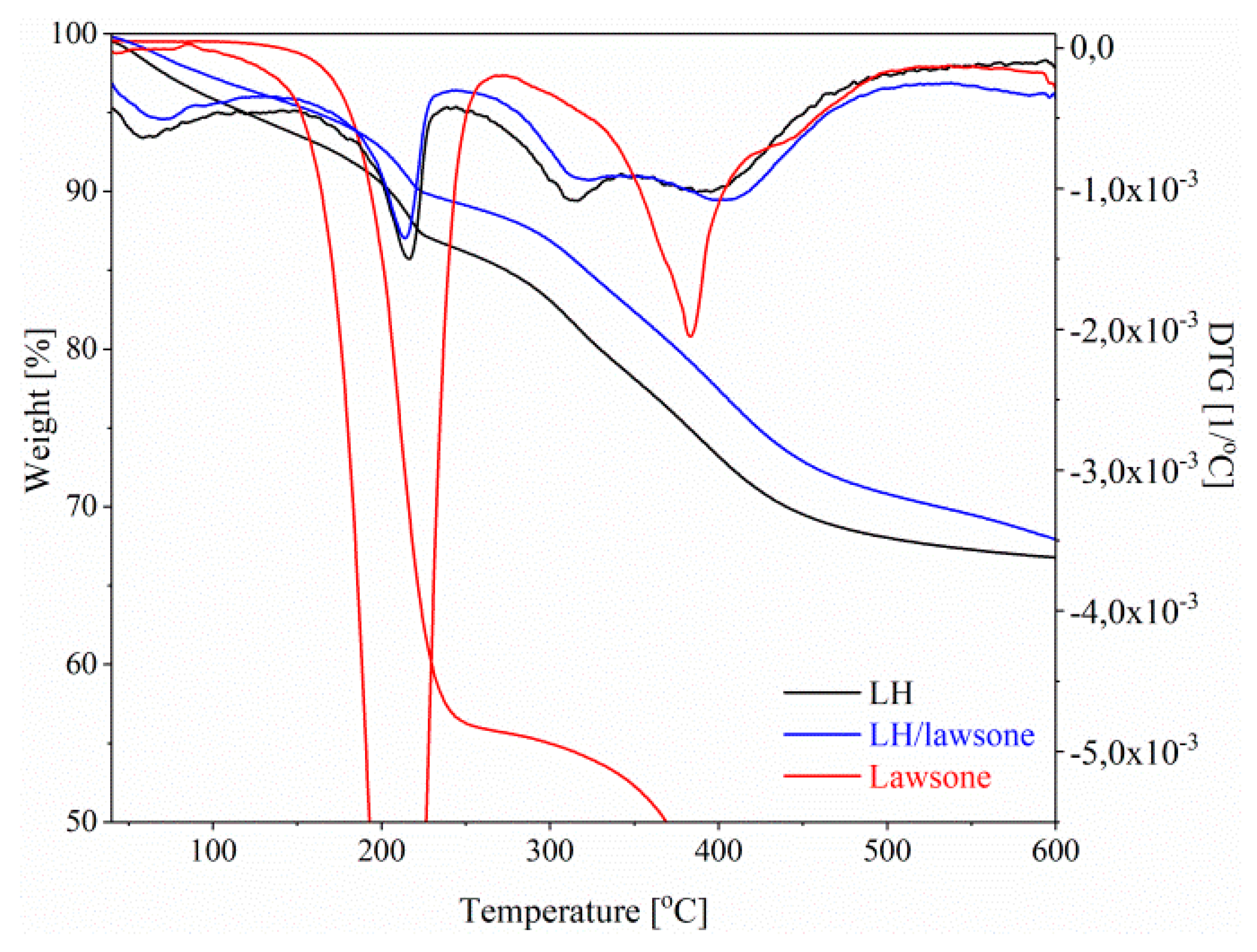
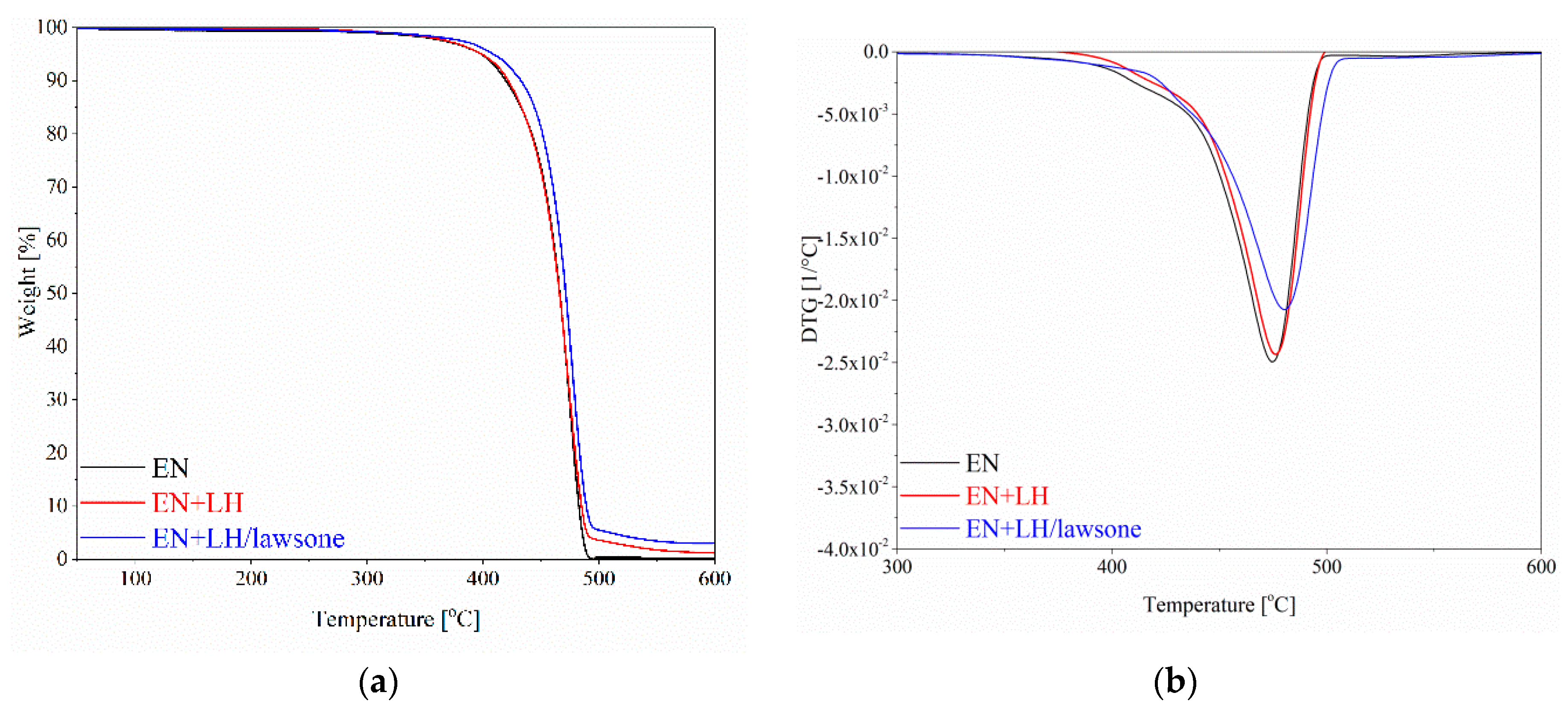
3.3. Thermogravimetric Analysis (TGA)
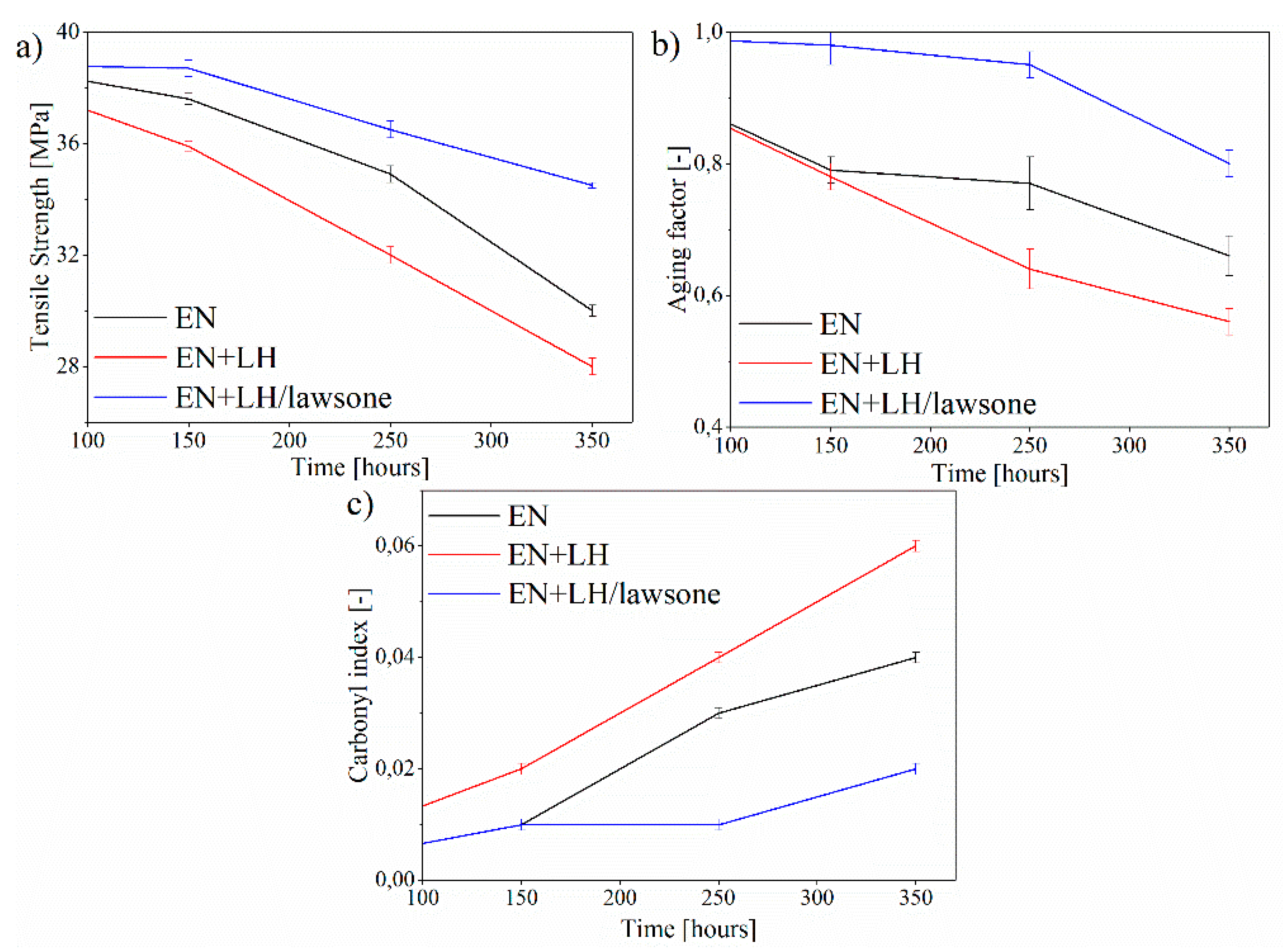
3.4. Mechanical Properties and Carbonyl Index of EN Composites
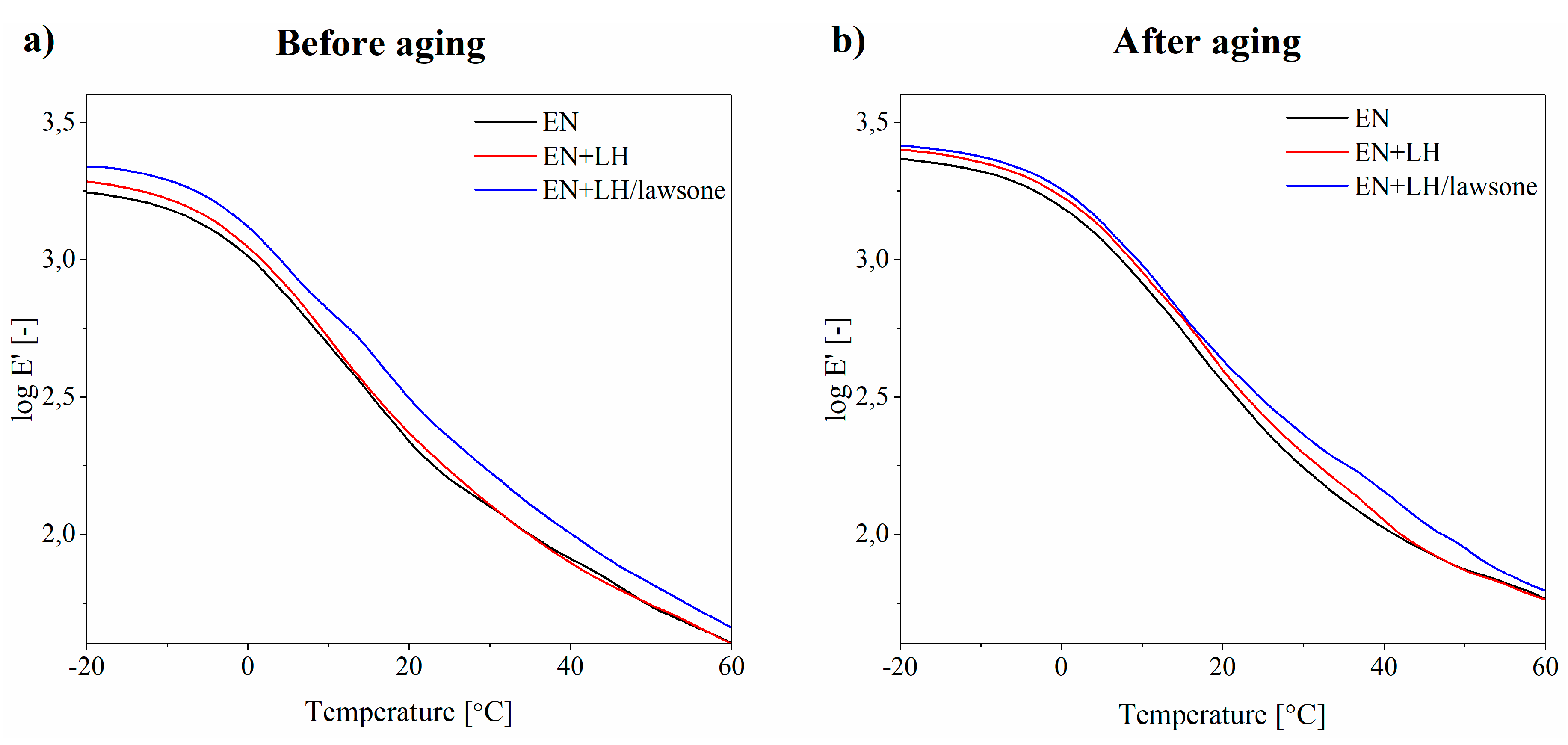
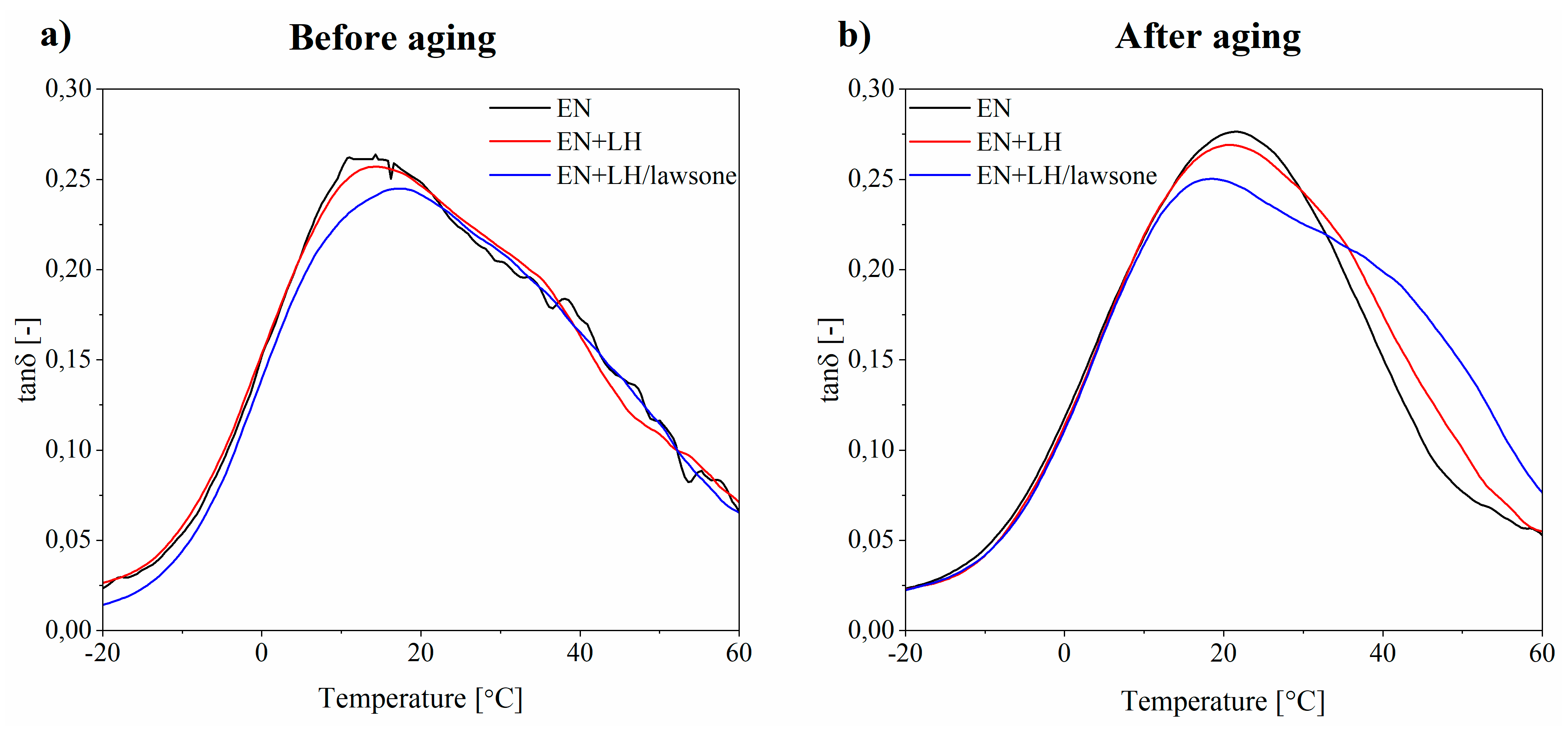
3.5. Thermal Stability of EN Composites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mohammad, S.; ul-Islam, S.; Mohammad, F. Recent Advancements in Natural Dye Applications: A Review. J. Clean. Prod. 2013, 53, 310–331. [Google Scholar]
- Velho, S.R.K.; Brum, L.F.W.; Petter, C.O.; dos Santos, J.H.Z.; Šimunić, Š.; Kappa, W.H. Development of Structured Natural Dyes for Use into Plastics. Dyes Pigments 2017, 136, 248–254. [Google Scholar] [CrossRef]
- Bakowska-Barczak, A. Acylated anthocyanins as stable, natural food colorants—A review. Pol. J. Food Nutr. Sci. 2005, 41, 107–116. [Google Scholar]
- Trigueiro, P.; Pereira, F.A.R.; Guillermin, D.; Rigaud, B.; Balme, S.; Janot, J.M.; Santos, I.M.G.; Fonseca, M.G.; Walter, P.; Jaber, M. When anthraquinone dyes meet pillared montmorillonite: Stability or fading upon exposure to light? Dyes Pigments 2018, 159, 384–394. [Google Scholar] [CrossRef]
- Kohno, Y.; Kinoshita, R.; Ikoma, S.; Yoda, K.; Shibata, M.; Matsushima, R.; Tomita, Y.; Maeda, Y.; Kobayashi, K. Stabilization of natural anthocyanin by intercalation into montmorillonite. Appl. Clay Sci. 2009, 42, 519–523. [Google Scholar]
- Mahmoodi, A.; Ebrahimi, M.; Khosravi, A.; Mohammadloo, H.E. A hybrid dye-clay nano-pigment: Synthesis, characterization and application in organic coatings. Dyes Pigments 2017, 147, 234–240. [Google Scholar] [CrossRef]
- Moujahid, E.M.; Lahkale, R.; Ouassif, H.; Bouragba, F.Z.; Elhatimi, W. New organic dye/anionic clay hybrid pigments: Preparation, optical properties and structural stability. Dyes Pigments 2019, 162, 998–1004. [Google Scholar] [CrossRef]
- Szadkowski, B.; Marzec, A.; Rybiński, P.; Maniukiewicz, W.; Zaborski, M. Aluminum-magnesium hydroxycarbonate/azo dye hybrids as novel multifunctional colorants for elastomer composites. Polymers 2019, 11, 43. [Google Scholar] [CrossRef]
- Marzec, A.; Szadkowski, B.; Rogowski, J.; Maniukiewicz, W.; Moszyński, D.; Rybiński, P.; Zaborski, M. Carminic acid stabilized with aluminum-magnesium hydroxycarbonate as new colorant reducing flammability of polymer composites. Molecules 2019, 24, 560. [Google Scholar] [CrossRef]
- Kutlu, B.; Leuteritz, A.; Häußler, L.; Oertel, U.; Heinrich, G. Stabilization of polypropylene using dye modified layered double hydroxides. Polym. Degrad. Stab. 2014, 102, 9–14. [Google Scholar] [CrossRef]
- Zimmermann, A.; Jaerger, S.; Zawadzki, S.F.; Wypych, F. Synthetic zinc layered hydroxide salts intercalated with anionic azo dyes as fillers into high-density polyethylene composites: First insights. J. Polym. Res. 2013, 20, 224. [Google Scholar] [CrossRef]
- Rahman Bhuiyan, M.A.; Islam, A.; Ali, A.; Islam, M.N. Color and chemical constitution of natural dye henna (Lawsonia inermis L.) and its application in the coloration of textiles. J. Clean. Prod. 2017, 167, 14–22. [Google Scholar] [CrossRef]
- Rehman, F.-U.; Adeel, S.; Qaiser, S.; Bhatti, I.A.; Shahid, M.; Zuber, M. Dyeing behaviour of gamma irradiated cotton fabric using Lawson dye extracted from henna leaves (Lawsonia inermis). Radiat. Phys. Chem. 2012, 81, 1752–1756. [Google Scholar] [CrossRef]
- Gulrajani, M.; Gupta, D.; Aggarwal, V.; Jain, M. Some studies on natural yellow dyes, Part III: Quinones: Henna, dolu. Indian Text. J. 1992, 102, 76–83. [Google Scholar]
- Yusuf, M.; Ahmad, A.; Shahid, M.; Khan, M.I.; Khan, S.A.; Manzoor, N.; Mohammad, F. Assessment of colorimetric, antibacterial and antifungal properties of woollen yarn dyed with the extract of the leaves of henna (Lawsonia inermis). J. Clean. Prod. 2012, 27, 42–50. [Google Scholar] [CrossRef]
- Costa, A.F.D.S.; de Amorim, J.D.; Almeida, F.C.G.; de Lima, I.D.; de Paiva, S.C.; Rocha, M.A.V.; Vinhas, G.M.; Sarubbo, L.A. Dyeing of bacterial cellulose films using plant-based natural dyes. Int. J. Biol. Macromol. 2019, 121, 580–587. [Google Scholar] [CrossRef]
- Nayak, B.S.; Isitor, G.; Davis, E.M.; Pillai, G.K. The evidence based wound healing activity of Lawsonia inermis Linn. Phytother. Res. 2007, 21, 827–831. [Google Scholar] [CrossRef] [PubMed]
- White, J.M.; Kullavanijaya, P.; Dua-ngdeeden, I.; Zazzeroni, R.; Gilmour, N.J.; Basketter, D.A.; Mc Fadden, J.P. p-Phenylenediamine allergy: The role of Bandrowski’s base. Clin. Exp. Allergy 2006, 36, 1289–1293. [Google Scholar] [CrossRef]
- Yasin, Y.; Ismail, N.M. Controlled release of lawsone-intercalated Zn-Al-layered double hydroxide. J. Med. Sci. 2013, 13, 453–458. [Google Scholar] [CrossRef][Green Version]
- Marzec, A.; Szadkowski, B.; Rogowski, J.; Maniukiewicz, W.; Zaborski, M. Characterization and structure-property relationships of organic-inorganic hybrid composites based on aluminum-magnesium hydroxycarbonate and azo chromophore. Molecules 2019, 24, 880. [Google Scholar] [CrossRef]
- Marzec, A.; Szadkowski, B.; Rogowski, J.; Maniukiewicz, W.; Szynkowska, M.I.; Zaborski, M. Characteristics of hybrid pigments made from alizarin dye on mixed oxide host. Materials 2019, 12, 360. [Google Scholar] [CrossRef] [PubMed]
- Barbeş, L.; Rădulescu, C.; Stihi, C. ATR-FTIR spectrometry characterisation of polymeric materials. Rom. Rep. Phys. 2014, 66, 765–777. [Google Scholar]
- Masłowski, M.; Miedzianowska, J.; Strzelec, K. Natural rubber biocomposites containing corn, barley and wheat straw. Polym. Test. 2017, 63, 84–91. [Google Scholar] [CrossRef]
- Park, T.J.; Choi, S.S.; Kim, Y. 27Al solid-state NMR structural studies of hydrotalcite compounds calcined at different temperatures. Bull. Korean Chem. Soc. 2009, 30, 149–152. [Google Scholar]
- Christie, R.M.; Mackay, J.L. Metal salt azo pigments. Color. Technol. 2008, 124, 133–144. [Google Scholar] [CrossRef]
- Marzec, A.; Szadkowski, B.; Rogowski, J.; Maniukiewicz, W.; Moszyński, D.; Kozanecki, M.; Zaborski, M. Characterization and properties of new color-tunable hybrid pigments based on layered double hydroxides (LDH) and 1,2-dihydroxyanthraquinone dye. J. Ind. Eng. Chem. 2018, 70, 427–438. [Google Scholar] [CrossRef]
- Costa, F.R.; Leuteritz, A.; Wagenknecht, U.; Jehnichen, D.; Häußler, L.; Heinrich, G. Intercalation of Mg-Al layered double hydroxide by anionic surfactants: Preparation and characterization. Appl. Clay Sci. 2008, 38, 153–164. [Google Scholar] [CrossRef]
- Moreno, D.D.P.; Hirayama, D.; Saron, C. Accelerated aging of pine wood waste/recycled LDPE composites. Polym. Degrad. Stab. 2018, 149, 39–44. [Google Scholar] [CrossRef]
- Marzec, A.; Szadkowski, B.; Rogowski, J.; Maniukiewicz, W.; Zaborski, M. New organic-inorganic hybrids as multifunctional addditives to improve ethylene-norbornene (EN) composite stability. Polym. Degrad. Stab. 2019, 160, 110–119. [Google Scholar] [CrossRef]
- Gulminea, J.V.; Janissekb, P.R.; Heisec, H.M.; Akcelrud, L. The effect of accelerated aging on the surface mechanical properties of polyethylene. Polym. Degrad. Stab. 2003, 81, 367–373. [Google Scholar]
- Yang, R.; Li, Y.; Yu, J. Photo-stabilization of linear low density polyethylene by inorganic nano-particles. Polym. Degrad. Stab. 2005, 88, 168–174. [Google Scholar] [CrossRef]
- Perera, M.C.S.; Ishiaku, U.S.; Ishak, Z.A.M. Thermal degradation of PVC/NBR and PVC/ENR50 binary blends and PVC/ENR50/NBR ternary blends studied by DMA and solid state NMR. Polym. Degrad. Stab. 2000, 68, 393. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marzec, A.; Szadkowski, B. Improved Aging Stability of Ethylene-Norbornene Composites Filled with Lawsone-Based Hybrid Pigment. Polymers 2019, 11, 723. https://doi.org/10.3390/polym11040723
Marzec A, Szadkowski B. Improved Aging Stability of Ethylene-Norbornene Composites Filled with Lawsone-Based Hybrid Pigment. Polymers. 2019; 11(4):723. https://doi.org/10.3390/polym11040723
Chicago/Turabian StyleMarzec, Anna, and Bolesław Szadkowski. 2019. "Improved Aging Stability of Ethylene-Norbornene Composites Filled with Lawsone-Based Hybrid Pigment" Polymers 11, no. 4: 723. https://doi.org/10.3390/polym11040723
APA StyleMarzec, A., & Szadkowski, B. (2019). Improved Aging Stability of Ethylene-Norbornene Composites Filled with Lawsone-Based Hybrid Pigment. Polymers, 11(4), 723. https://doi.org/10.3390/polym11040723