Amphiphilic Peptides for Efficient siRNA Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Lines
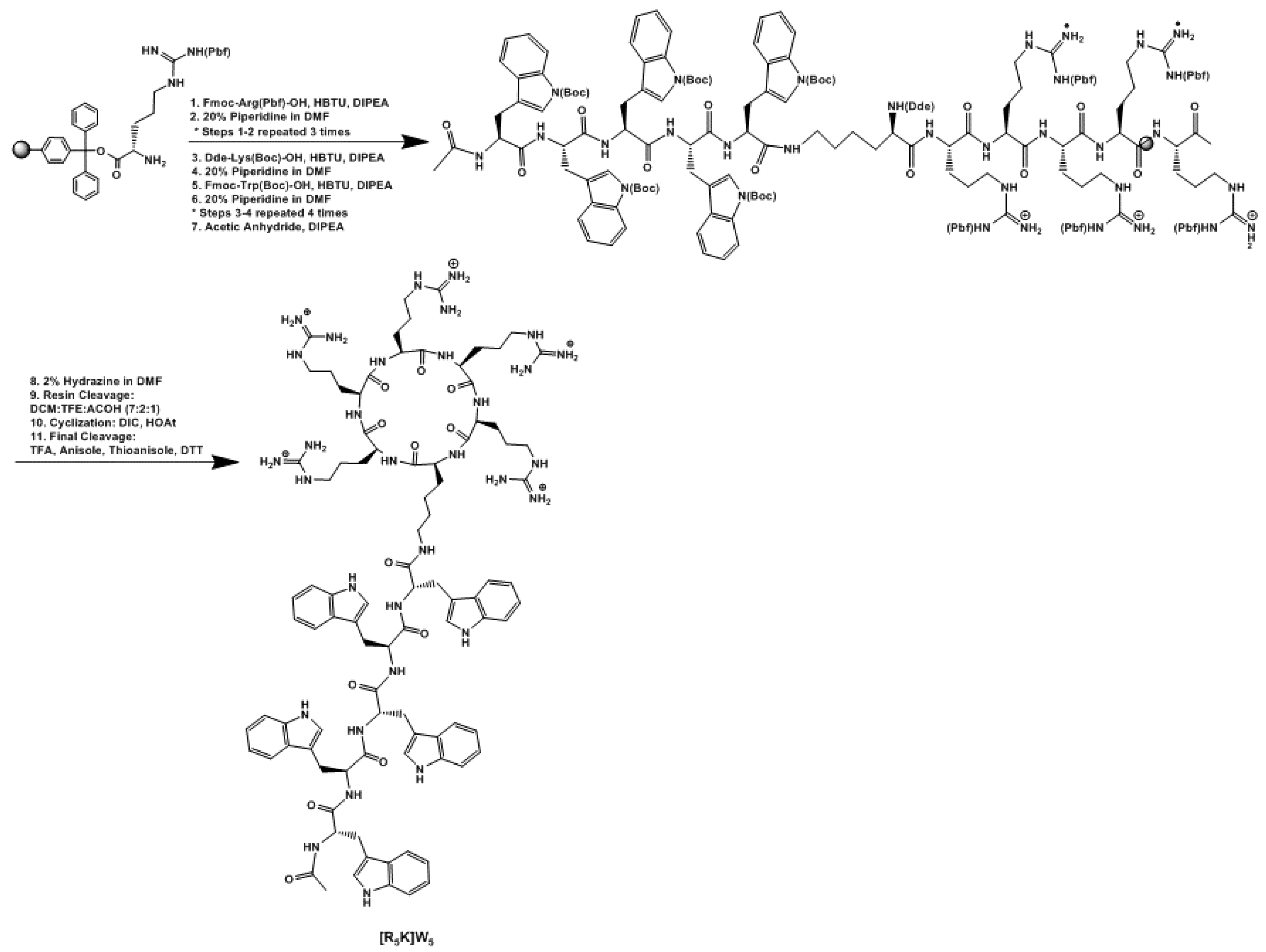
2.3. Synthesis and Purification
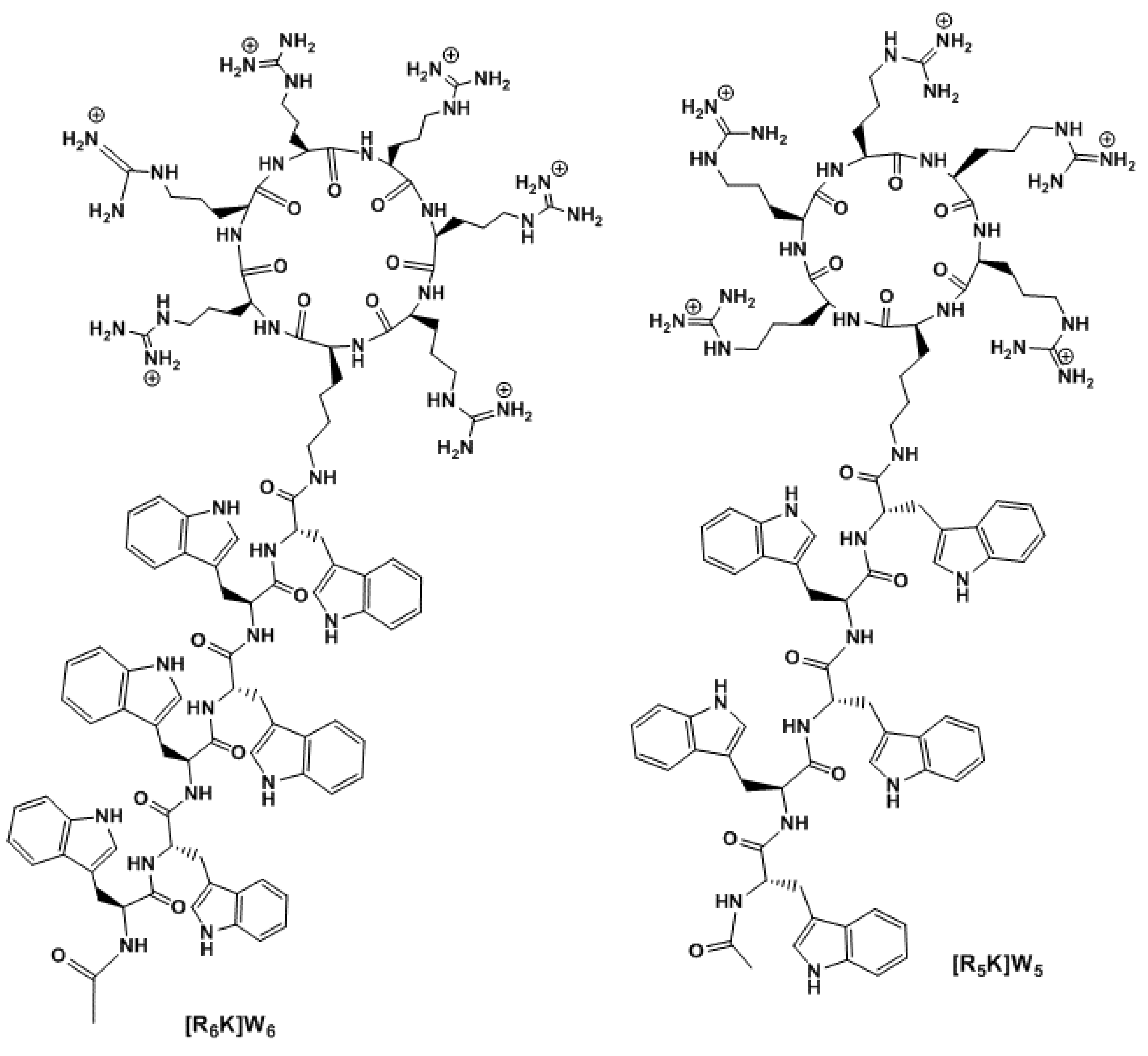
- [R5K]W5: MALDI-TOF (m/z): C93H124N32O12, calculated: 1881.0076, found: 1883.8647 [M+2H]+;
- [R6K]W6: MALDI-TOF (m/z): C110H146N38O14, calculated: 2223.1881, found: 2225.0676 [M+2H]+
2.4. Complex Formation and Cytotoxicity of Peptide/siRNA Complexes
2.5. Binding Affinity
2.6. Size and ξ-potential of Peptide/siRNA Complexes
2.7. Cellular Internalization (Flow Cytometry)
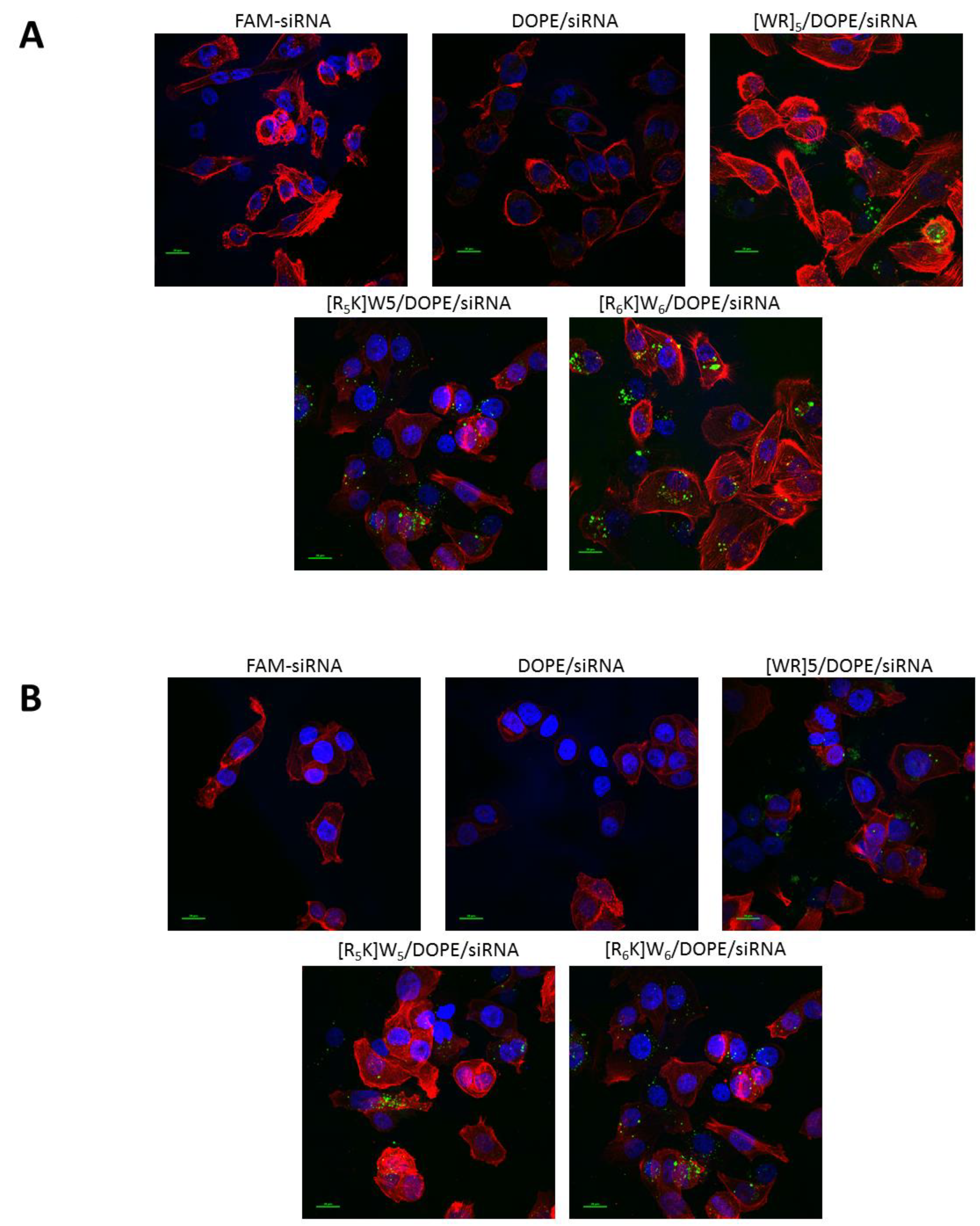
2.8. Confocal Microscopy
2.9. mRNA Interference (Real-Time Polymerase Chain Reaction; RT-PCR)
2.10. Protein Quantification (Western Blot)
3. Results and Discussion
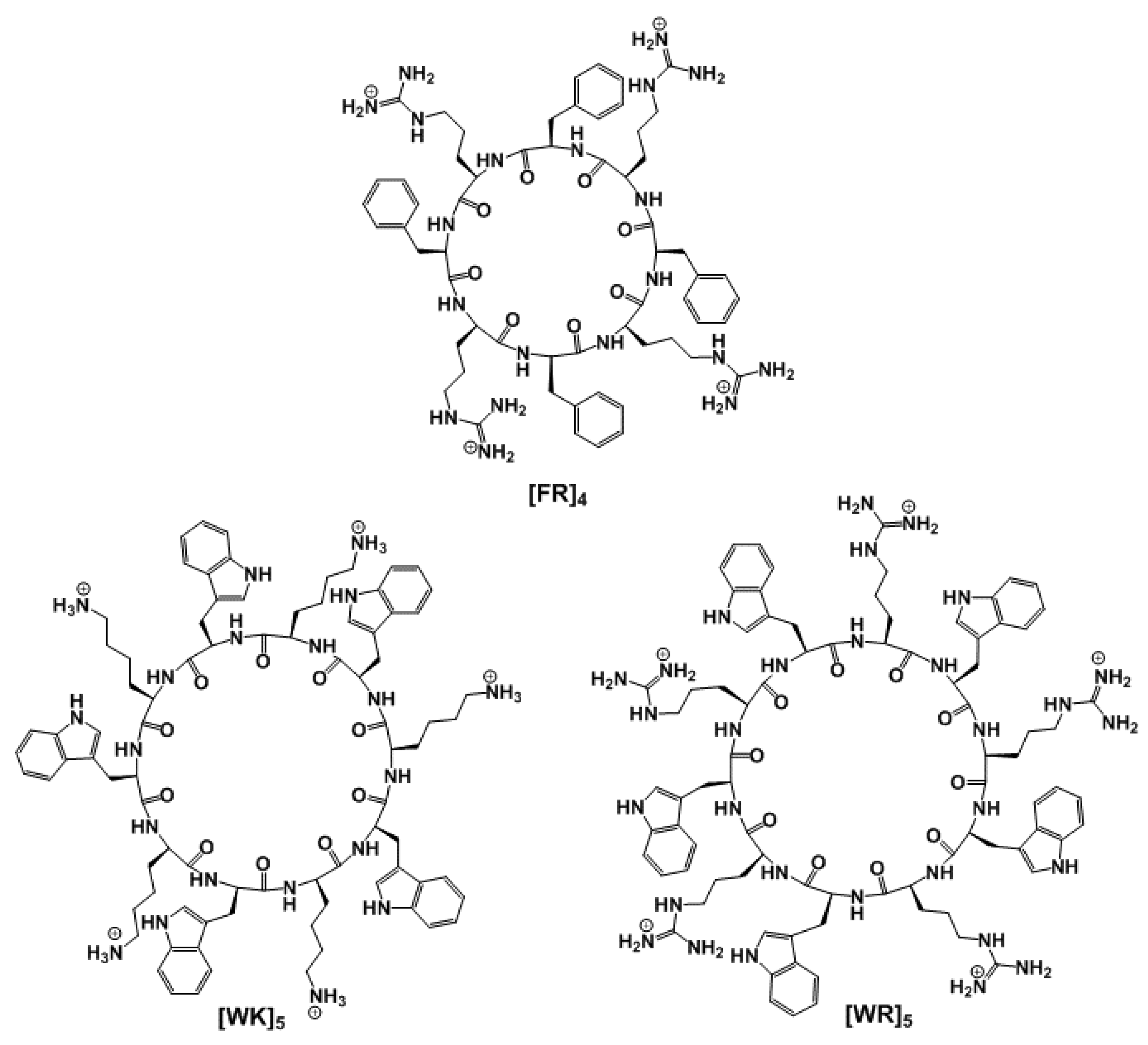
3.1. Chemistry
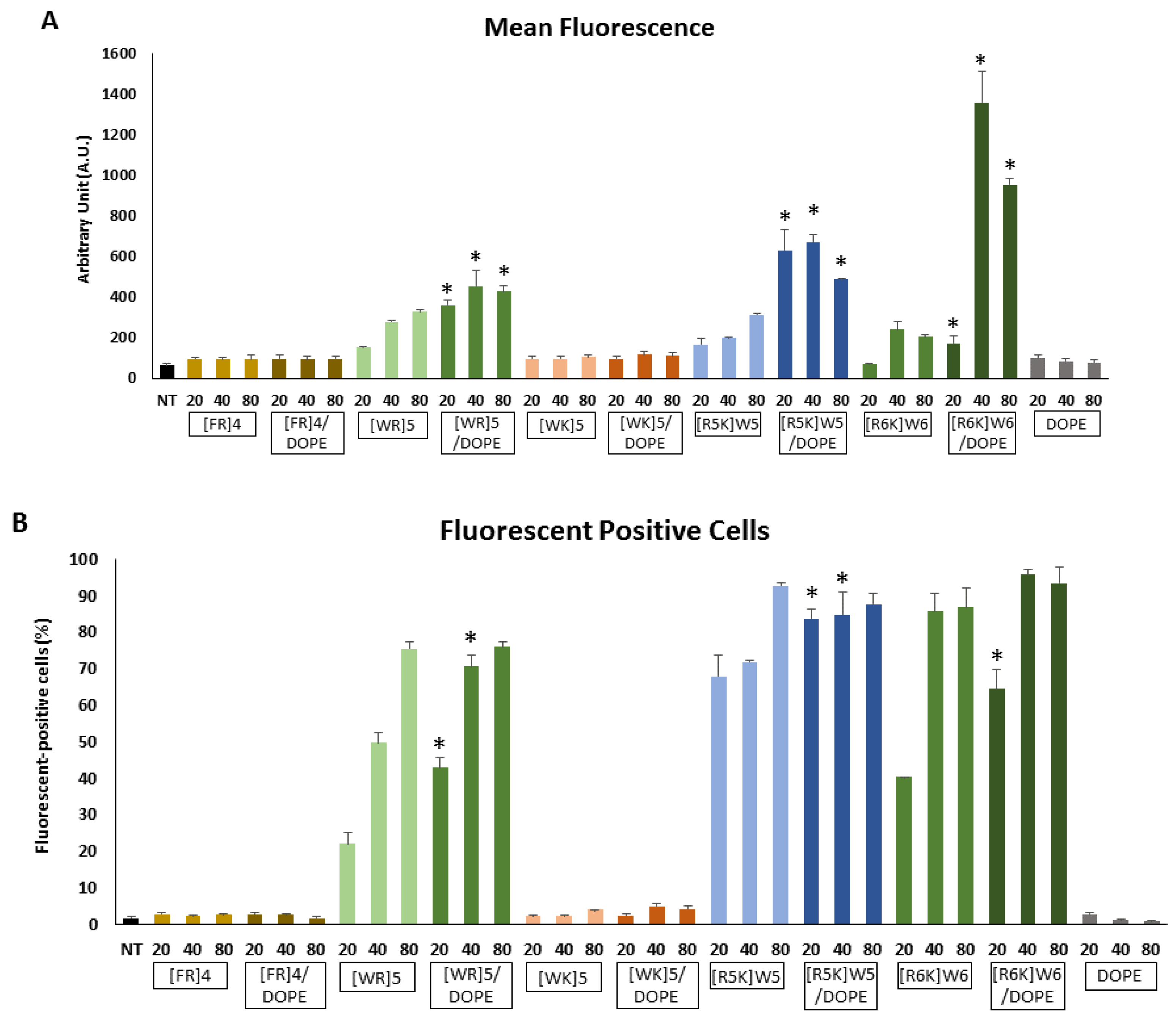
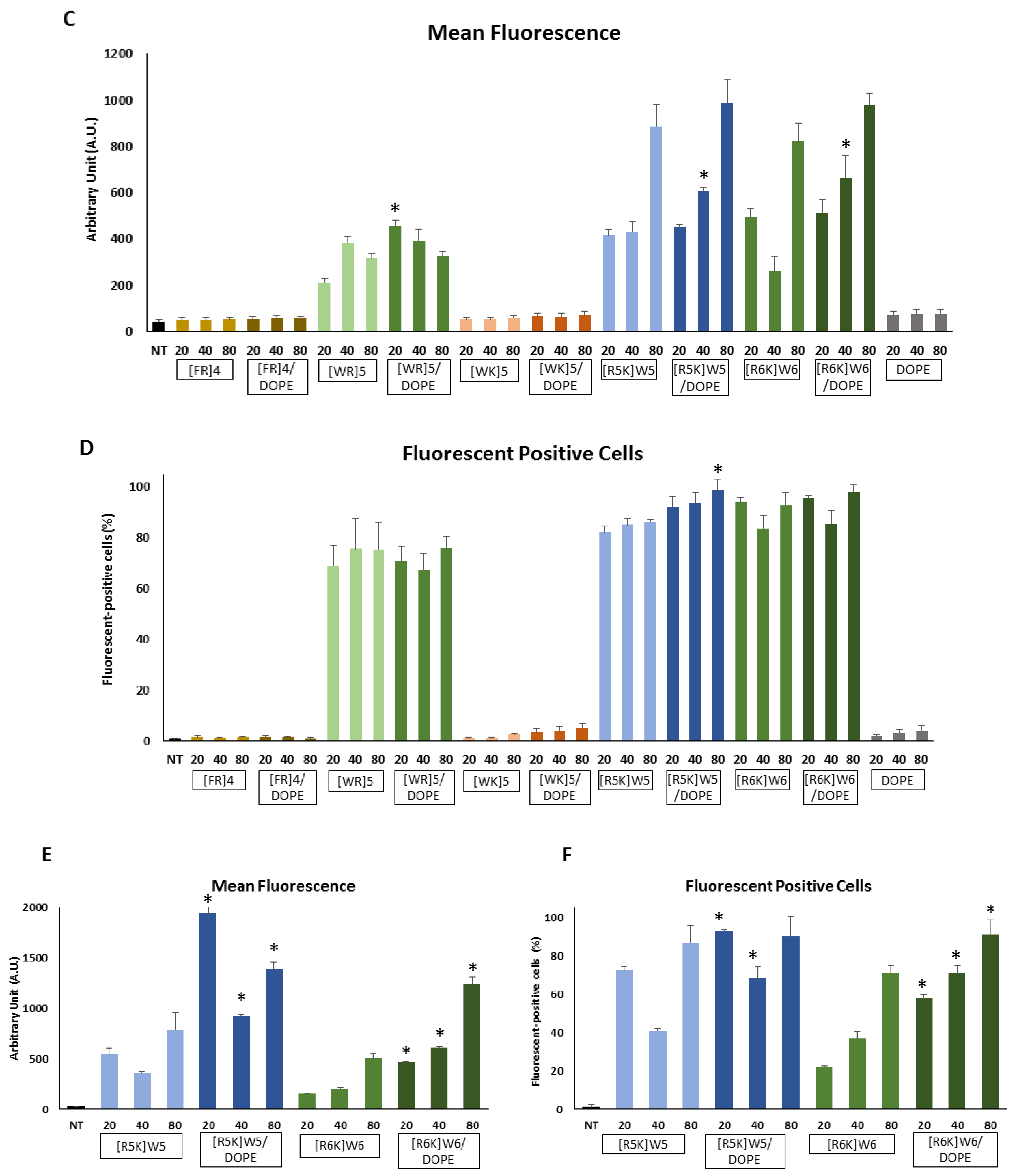
3.2. Cellular Internalization
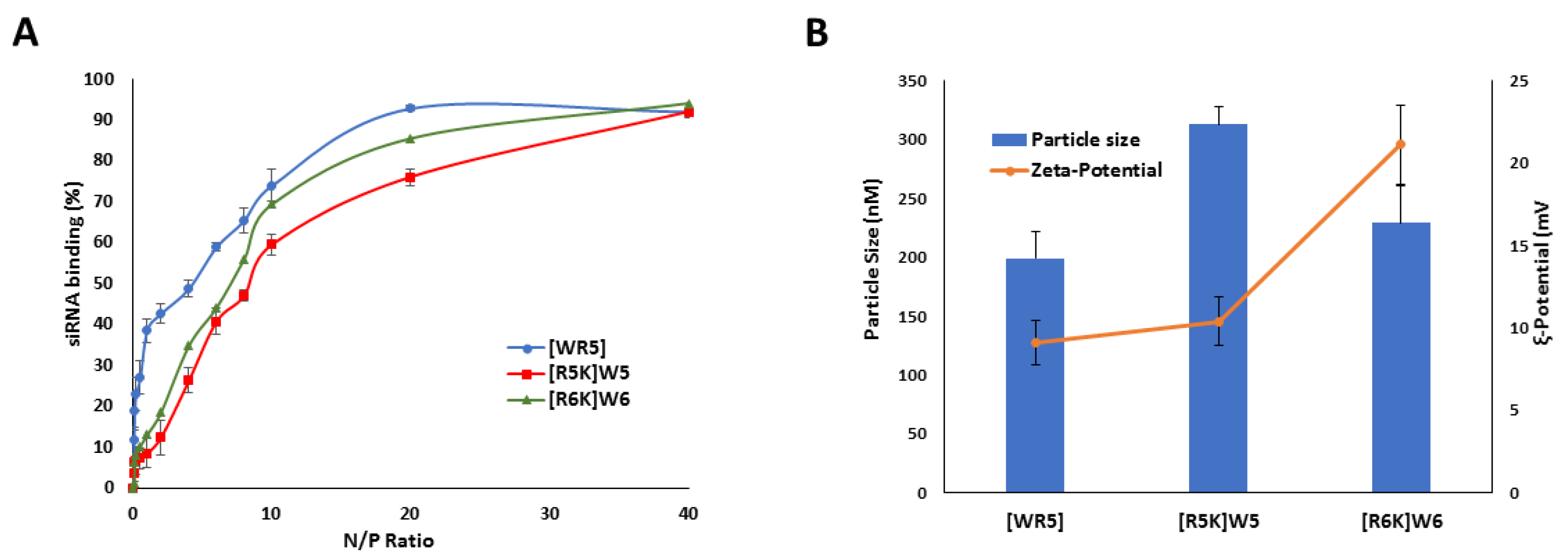
3.3. Binding Affinity
3.4. Particle Size and Surface Charge
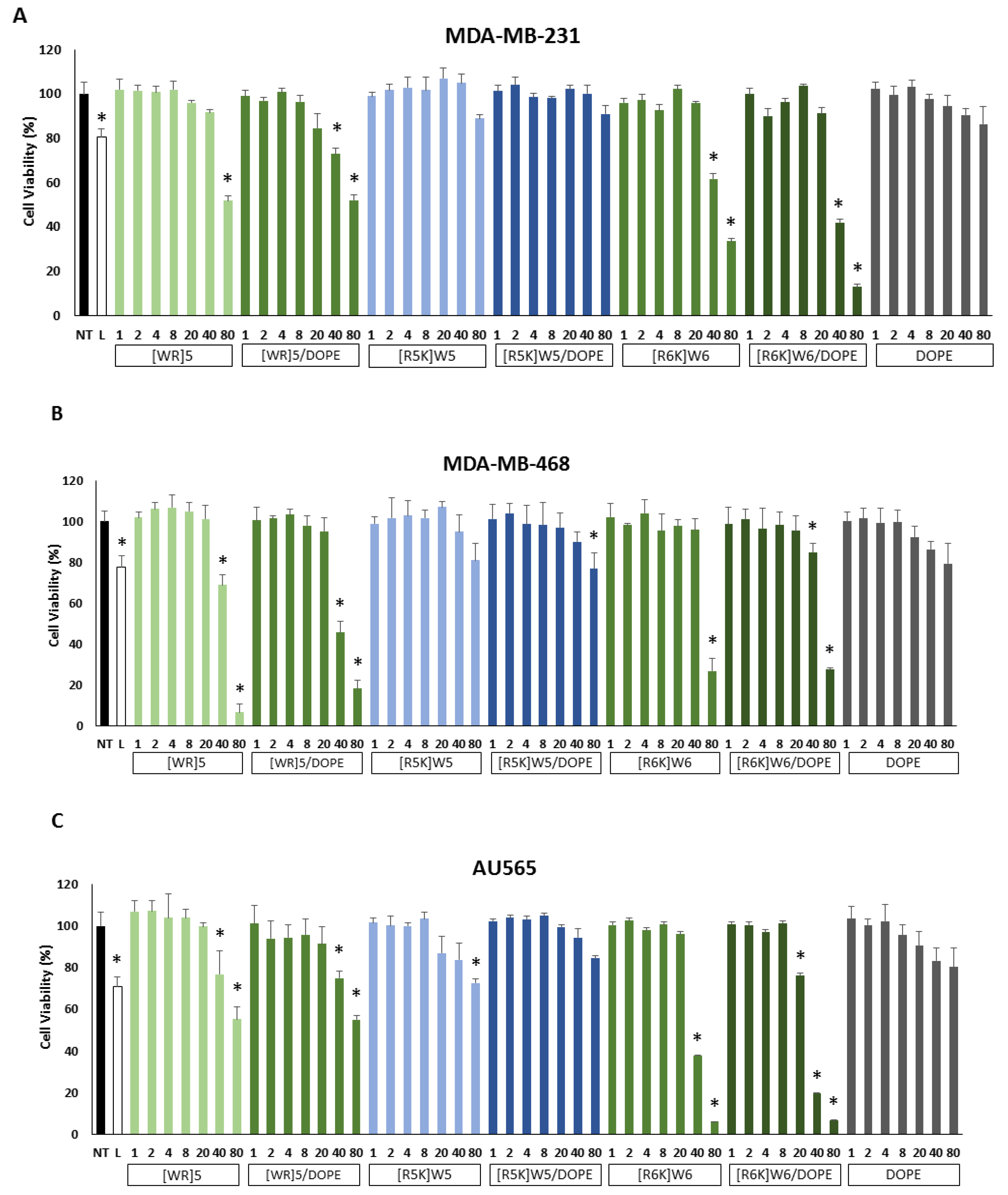
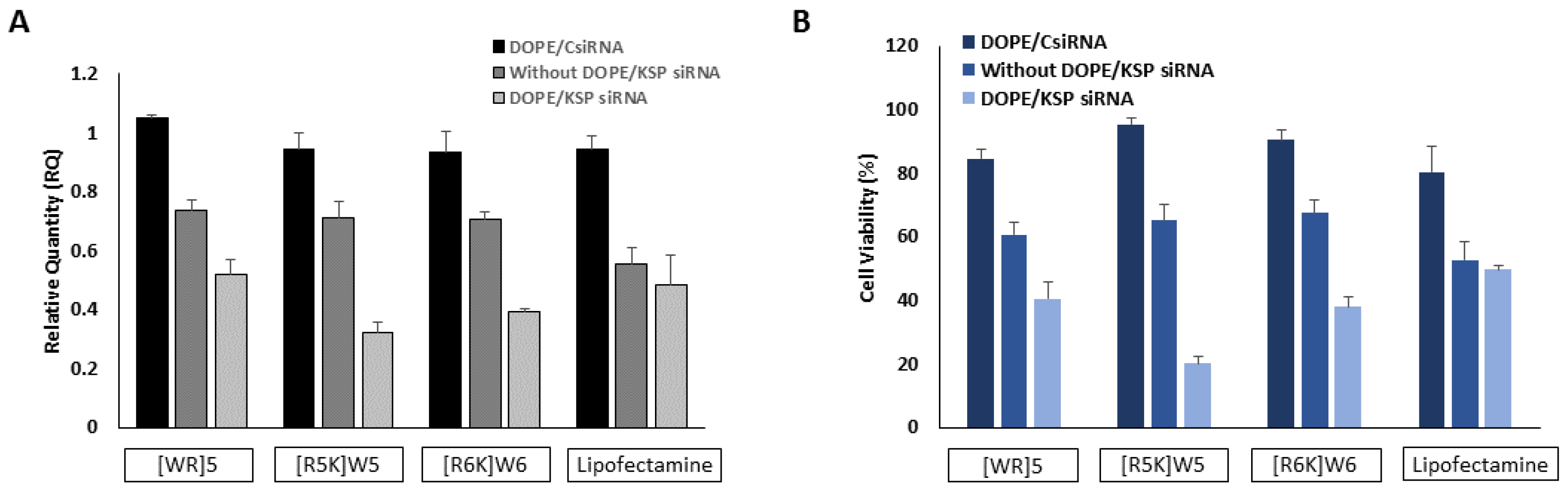
3.5. Cytotoxicity
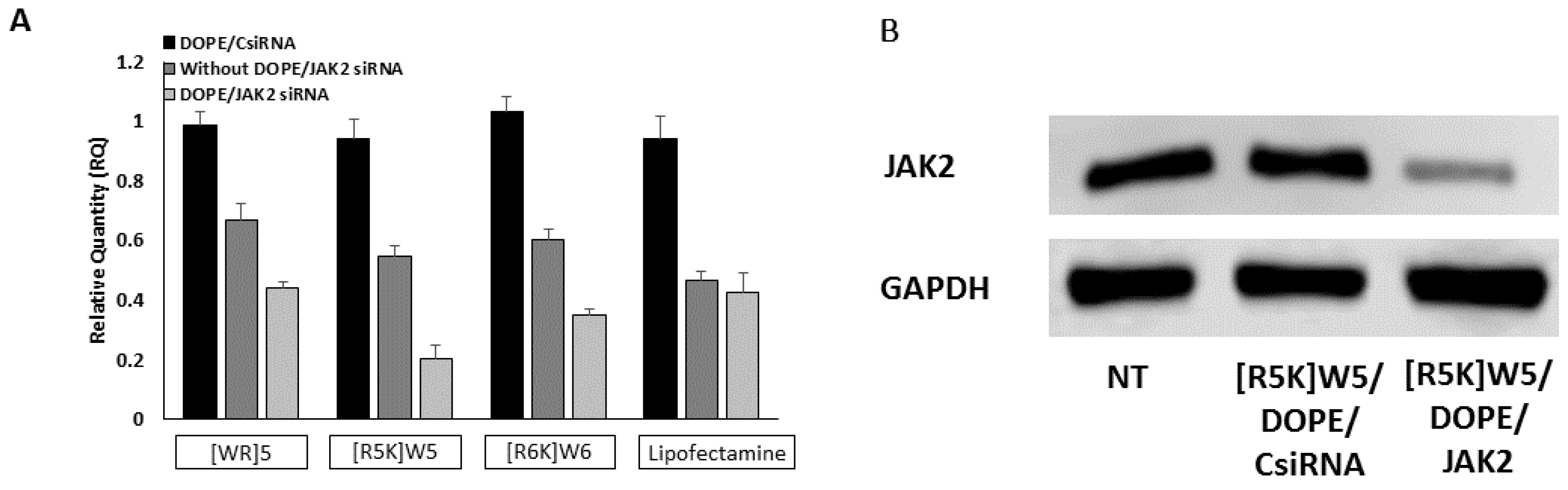
3.6. Protein Silencing
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ghildiyal, M.; Zamore, P.D. Small silencing RNAs: An expanding universe. Nat. Rev. Genet. 2009, 10, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Ender, C.; Meister, G. Argonaute proteins at a glance. J. Cell Sci. 2010, 123, 1819–1823. [Google Scholar] [CrossRef] [PubMed]
- Kim, V.N.; Han, J.; Siomi, M.C. Biogenesis of small RNAs in animals. Nat. Rev. Mol. Cell Biol. 2009, 10, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Aliabadi, H.M.; Landry, B.; Sun, C.; Tang, T.; Uludag, H. Supramolecular assemblies in functional siRNA delivery: Where do we stand? Biomaterials 2012, 33, 2546–2569. [Google Scholar] [CrossRef]
- De Fougerolles, A.; Vornlocher, H.P.; Maraganore, J.; Lieberman, J. Interfering with disease: A progress report on siRNA-based therapeutics. Nat. Rev. Drug Discov. 2007, 6, 443–453. [Google Scholar] [CrossRef]
- Chabot, S.; Teissie, J.; Golzio, M. Targeted electro-delivery of oligonucleotides for RNA interference: SiRNA and antimir. Adv. Drug Deliv. Rev. 2015, 81, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Kanasty, R.L.; Eltoukhy, A.A.; Vegas, A.J.; Dorkin, J.R.; Anderson, D.G. Non-viral vectors for gene-based therapy. Nat. Rev. Genet. 2014, 15, 541–555. [Google Scholar] [CrossRef]
- Morrissey, D.V.; Lockridge, J.A.; Shaw, L.; Blanchard, K.; Jensen, K.; Breen, W.; Hartsough, K.; Machemer, L.; Radka, S.; Jadhav, V.; et al. Potent and persistent in vivo anti-hbv activity of chemically modified siRNAs. Nat. Biotechnol. 2005, 23, 1002–1007. [Google Scholar] [CrossRef]
- Titze-de-Almeida, R.; David, C.; Titze-de-Almeida, S.S. The race of 10 synthetic RNAi-based drugs to the pharmaceutical market. Pharm. Res. 2017, 34, 1339–1363. [Google Scholar] [CrossRef]
- Huang, Y. Preclinical and clinical advances of galnac-decorated nucleic acid therapeutics. Mol. Ther. Nucleic Acids 2017, 6, 116–132. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, A.; Garg, S. SiRNA delivery using polyelectrolyte-gold nanoassemblies in neuronal cells for bace1 gene silencing. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 80, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Sun, X.; Shao, R.; Xu, Y.; Gao, J.; Liang, W. Vegf siRNA delivered by polycation liposome-encapsulated calcium phosphate nanoparticles for tumor angiogenesis inhibition in breast cancer. Int. J. Nanomed. 2017, 12, 6075–6088. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G.; Shen, Y.; Zhao, R.; Chen, F.; Zhang, Y.; Xu, A.; Shao, J. Dual-targeting multifuntional mesoporous silica nanocarrier for codelivery of siRNA and ursolic acid to folate receptor overexpressing cancer cells. J. Agric. Food Chem. 2017, 65, 6904–6911. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, A.; Higashi, T.; Motoyama, K.; Arima, H. In vitro and in vivo tumor-targeting siRNA delivery using folate-peg-appended dendrimer (g4)/alpha-cyclodextrin conjugates. Bioconjug Chem 2016, 27, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, A.; Higashi, T.; Motoyama, K.; Arima, H. Ternary complexes of folate-peg-appended dendrimer (g4)/alpha-cyclodextrin conjugate, siRNA and low-molecular-weight polysaccharide sacran as a novel tumor-selective siRNA delivery system. Int. J. Biol. Macromol. 2017, 99, 21–28. [Google Scholar] [CrossRef]
- Chugh, A.; Eudes, F.; Shim, Y.S. Cell-penetrating peptides: Nanocarrier for macromolecule delivery in living cells. IUBMB Life 2010, 62, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P. Cell penetrating peptide-modified pharmaceutical nanocarriers for intracellular drug and gene delivery. Biopolymers 2008, 90, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Mandal, D.; Nasrolahi Shirazi, A.; Parang, K. Cell-penetrating homochiral cyclic peptides as nuclear-targeting molecular transporters. Angew. Chem. Int. Ed. Engl. 2011, 50, 9633–9637. [Google Scholar] [CrossRef] [PubMed]
- Mandal, D.; Tiwari, R.K.; Shirazi, A.N.; Oh, D.; Ye, G.; Banerjee, A.; Yadav, A.; Parang, K. Self-assembled surfactant cyclic peptide nanostructures as stabilizing agents. Soft Matter 2013, 9. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Tiwari, R.K.; Oh, D.; Banerjee, A.; Yadav, A.; Parang, K. Efficient delivery of cell impermeable phosphopeptides by a cyclic peptide amphiphile containing tryptophan and arginine. Mol. Pharm. 2013, 10, 2008–2020. [Google Scholar] [CrossRef]
- Oh, D.; Darwish, S.A.; Shirazi, A.N.; Tiwari, R.K.; Parang, K. Amphiphilic bicyclic peptides as cellular delivery agents. ChemMedChem 2014, 9, 2449–2453. [Google Scholar] [CrossRef] [PubMed]
- Shirazi, A.N.; Paquin, K.L.; Howlett, N.G.; Mandal, D.; Parang, K. Cyclic peptide-capped gold nanoparticles for enhanced siRNA delivery. Molecules 2014, 19, 13319–13331. [Google Scholar] [CrossRef]
- Do, H.; Sharma, M.; El-Sayed, N.S.; Mahdipoor, P.; Bousoik, E.; Parang, K.; Montazeri Aliabadi, H. Difatty acyl-conjugated linear and cyclic peptides for siRNA delivery. ACS Omega 2017, 2, 6939–6957. [Google Scholar] [CrossRef] [PubMed]
- Aliabadi, H.M.; Landry, B.; Bahadur, R.K.; Neamnark, A.; Suwantong, O.; Uludag, H. Impact of lipid substitution on assembly and delivery of siRNA by cationic polymers. Macromol. Biosci. 2011, 11, 662–672. [Google Scholar] [CrossRef]
- Capel, V.; Vllasaliu, D.; Watts, P.; Stolnik, S. Insight into the relationship between the cell culture model, cell trafficking and siRNA silencing efficiency. Biochem. Biophys. Res. Commun. 2016, 477, 260–265. [Google Scholar] [CrossRef]
- Nakamura, T.; Kuroi, M.; Fujiwara, Y.; Warashina, S.; Sato, Y.; Harashima, H. Small-sized, stable lipid nanoparticle for the efficient delivery of siRNA to human immune cell lines. Sci. Rep. 2016, 6, 37849. [Google Scholar] [CrossRef] [PubMed]
- Cao, N.; Cheng, D.; Zou, S.; Ai, H.; Gao, J.; Shuai, X. The synergistic effect of hierarchical assemblies of siRNA and chemotherapeutic drugs co-delivered into hepatic cancer cells. Biomaterials 2011, 32, 2222–2232. [Google Scholar] [CrossRef] [PubMed]
- Convertine, A.J.; Benoit, D.S.; Duvall, C.L.; Hoffman, A.S.; Stayton, P.S. Development of a novel endosomolytic diblock copolymer for siRNA delivery. J. Control. Release 2009, 133, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Fan, H.; Levorse, D.A.; Crocker, L.S. Ionization behavior of amino lipids for siRNA delivery: Determination of ionization constants, SAR, and the impact of lipid pKa on cationic lipid-biomembrane interactions. Langmuir 2011, 27, 1907–1914. [Google Scholar] [CrossRef]
- Godbey, W.T.; Wu, K.K.; Mikos, A.G. Tracking the intracellular path of poly(ethylenimine)/DNA complexes for gene delivery. Proc. Natl. Acad. Sci. USA 1999, 96, 5177–5181. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Tiwari, R.K.; Oh, D.; Sullivan, B.; McCaffrey, K.; Mandal, D.; Parang, K. Surface decorated gold nanoparticles by linear and cyclic peptides as molecular transporters. Mol. Pharm. 2013, 10, 3137–3151. [Google Scholar] [CrossRef]
- Hoyer, J.; Neundorf, I. Knockdown of a g protein-coupled receptor through efficient peptide-mediated siRNA delivery. J. Control. Release 2012, 161, 826–834. [Google Scholar] [CrossRef]
- Kim, W.J.; Chang, C.W.; Lee, M.; Kim, S.W. Efficient siRNA delivery using water soluble lipopolymer for anti-angiogenic gene therapy. J. Control. Release 2007, 118, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Sarett, S.M.; Kilchrist, K.V.; Miteva, M.; Duvall, C.L. Conjugation of palmitic acid improves potency and longevity of siRNA delivered via endosomolytic polymer nanoparticles. J. Biomed. Mater. Res. A 2015, 103, 3107–3116. [Google Scholar] [CrossRef]
- Lee, J.; Ahn, H.J. PEGylated DC-Chol/DOPE cationic liposomes containing KSP siRNA as a systemic siRNA delivery carrier for ovarian cancer therapy. Biochem. Biophys. Res. Commun. 2018, 503, 1716–1722. [Google Scholar] [CrossRef] [PubMed]
- Klein, P.M.; Kern, S.; Lee, D.J.; Schmaus, J.; Hohn, M.; Gorges, J.; Kazmaier, U.; Wagner, E. Folate receptor-directed orthogonal click-functionalization of siRNA lipopolyplexes for tumor cell killing in vivo. Biomaterials 2018, 178, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Saw, P.E.; Gujrati, V.; Lee, Y.; Kim, H.; Kang, S.; Choi, M.; Kim, J.I.; Jon, S. Mono-arginine cholesterol-based small lipid nanoparticles as a systemic siRNA delivery platform for effective cancer therapy. Theranostics 2016, 6, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Doan, C.C.; Le, L.T.; Hoang, S.N.; Do, S.M.; Le, D.V. Simultaneous silencing of VEGF and KSP by siRNA cocktail inhibits proliferation and induces apoptosis of hepatocellular carcinoma Hep3B cells. Biol. Res. 2014, 47, 70. [Google Scholar] [CrossRef]
- Ying, B.; Campbell, R.B. Delivery of kinesin spindle protein targeting siRNA in solid lipid nanoparticles to cellular models of tumor vasculature. Biochem. Biophys. Res. Commun. 2014, 446, 441–447. [Google Scholar] [CrossRef]
- Marra, E.; Palombo, F.; Ciliberto, G.; Aurisicchio, L. Kinesin spindle protein siRNA slows tumor progression. J. Cell Physiol. 2013, 228, 58–64. [Google Scholar] [CrossRef]
- Brower, V. Rna interference advances to early-stage clinical trials. J. Natl. Cancer Inst. 2010, 102, 1459–1461. [Google Scholar] [CrossRef] [PubMed]
- Tabernero, J.; Shapiro, G.I.; LoRusso, P.M.; Cervantes, A.; Schwartz, G.K.; Weiss, G.J.; Paz-Ares, L.; Cho, D.C.; Infante, J.R.; Alsina, M.; et al. First-in-humans trial of an RNA interference therapeutic targeting vegf and ksp in cancer patients with liver involvement. Cancer Discov. 2013, 3, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Bousoik, E.; Montazeri Aliabadi, H. “Do we know jack” about JAK? A closer look at JAK/STAT signaling pathway. Front. Oncol. 2018, 8, 287. [Google Scholar] [CrossRef] [PubMed]
- Qian, C.; Yao, J.; Wang, J.; Wang, L.; Xue, M.; Zhou, T.; Liu, W.; Si, J. ERK1/2 inhibition enhances apoptosis induced by JAK2 silencing in human gastric cancer SGC7901 cells. Mol. Cell. Biochem. 2014, 387, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, J.; Wu, J.; Zhong, S.; Li, H. Inhibition of JAK2 reverses paclitaxel resistance in human ovarian cancer cells. Int. J. Gynecol. Cancer 2015, 25, 1557–1564. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mozaffari, S.; Bousoik, E.; Amirrad, F.; Lamboy, R.; Coyle, M.; Hall, R.; Alasmari, A.; Mahdipoor, P.; Parang, K.; Montazeri Aliabadi, H. Amphiphilic Peptides for Efficient siRNA Delivery. Polymers 2019, 11, 703. https://doi.org/10.3390/polym11040703
Mozaffari S, Bousoik E, Amirrad F, Lamboy R, Coyle M, Hall R, Alasmari A, Mahdipoor P, Parang K, Montazeri Aliabadi H. Amphiphilic Peptides for Efficient siRNA Delivery. Polymers. 2019; 11(4):703. https://doi.org/10.3390/polym11040703
Chicago/Turabian StyleMozaffari, Saghar, Emira Bousoik, Farideh Amirrad, Robert Lamboy, Melissa Coyle, Ryley Hall, Abdulaziz Alasmari, Parvin Mahdipoor, Keykavous Parang, and Hamidreza Montazeri Aliabadi. 2019. "Amphiphilic Peptides for Efficient siRNA Delivery" Polymers 11, no. 4: 703. https://doi.org/10.3390/polym11040703
APA StyleMozaffari, S., Bousoik, E., Amirrad, F., Lamboy, R., Coyle, M., Hall, R., Alasmari, A., Mahdipoor, P., Parang, K., & Montazeri Aliabadi, H. (2019). Amphiphilic Peptides for Efficient siRNA Delivery. Polymers, 11(4), 703. https://doi.org/10.3390/polym11040703







