Covalently Crosslinked Nanogels: An NMR Study of the Effect of Monomer Reactivity on Composition and Structure
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Isolation of Imatinib Free Base (Im) from Imatinib Mesylate
2.3. General Procedure for the Preparation of Nanogels
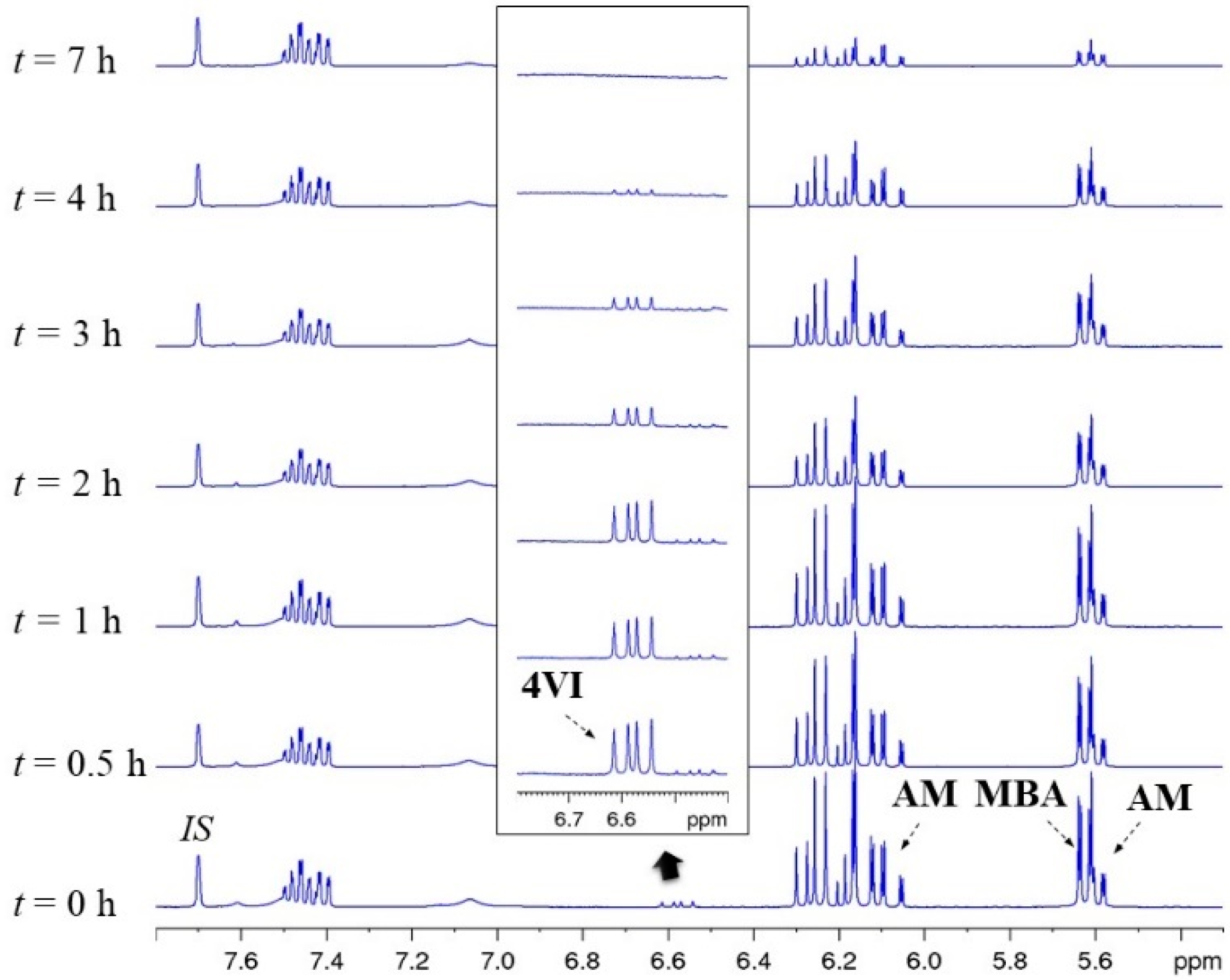
2.4. General Procedures for the Determination of Monomer Conversions by 1H NMR
2.5. 1H NMR Kinetics
2.6. Dynamic Light Scattering
2.7. Procedure for the Determination of the Volume Phase Transition Temperatures
3. Results and Discussion
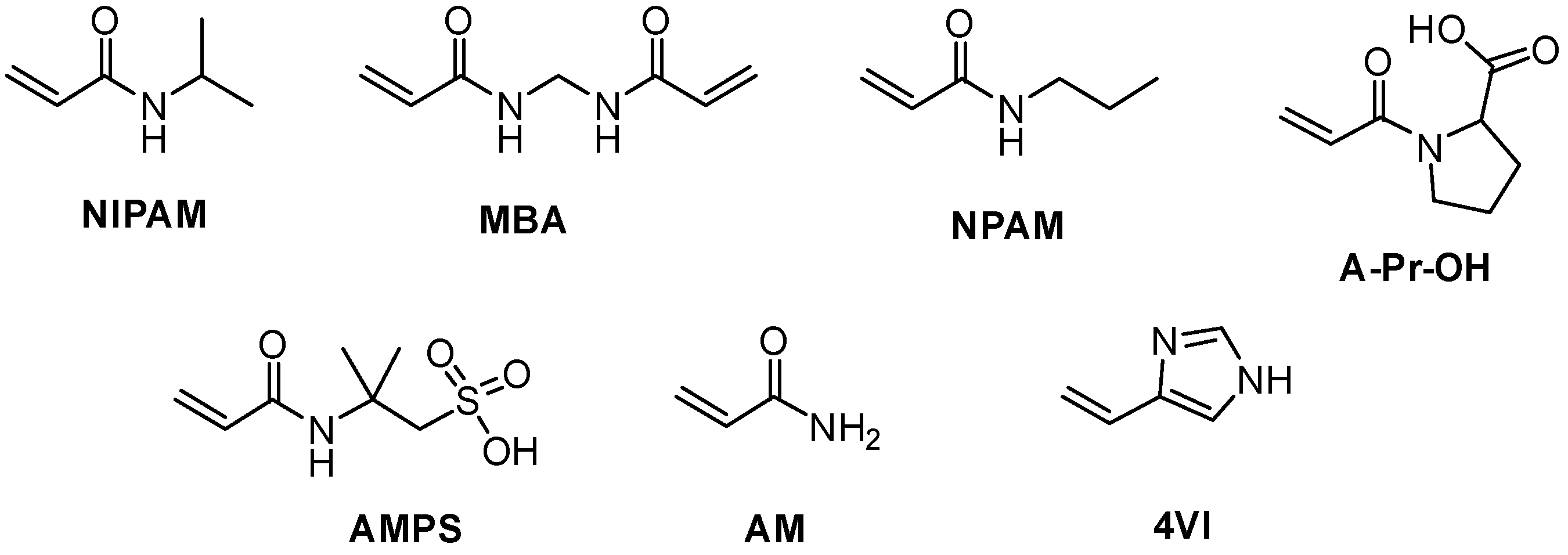
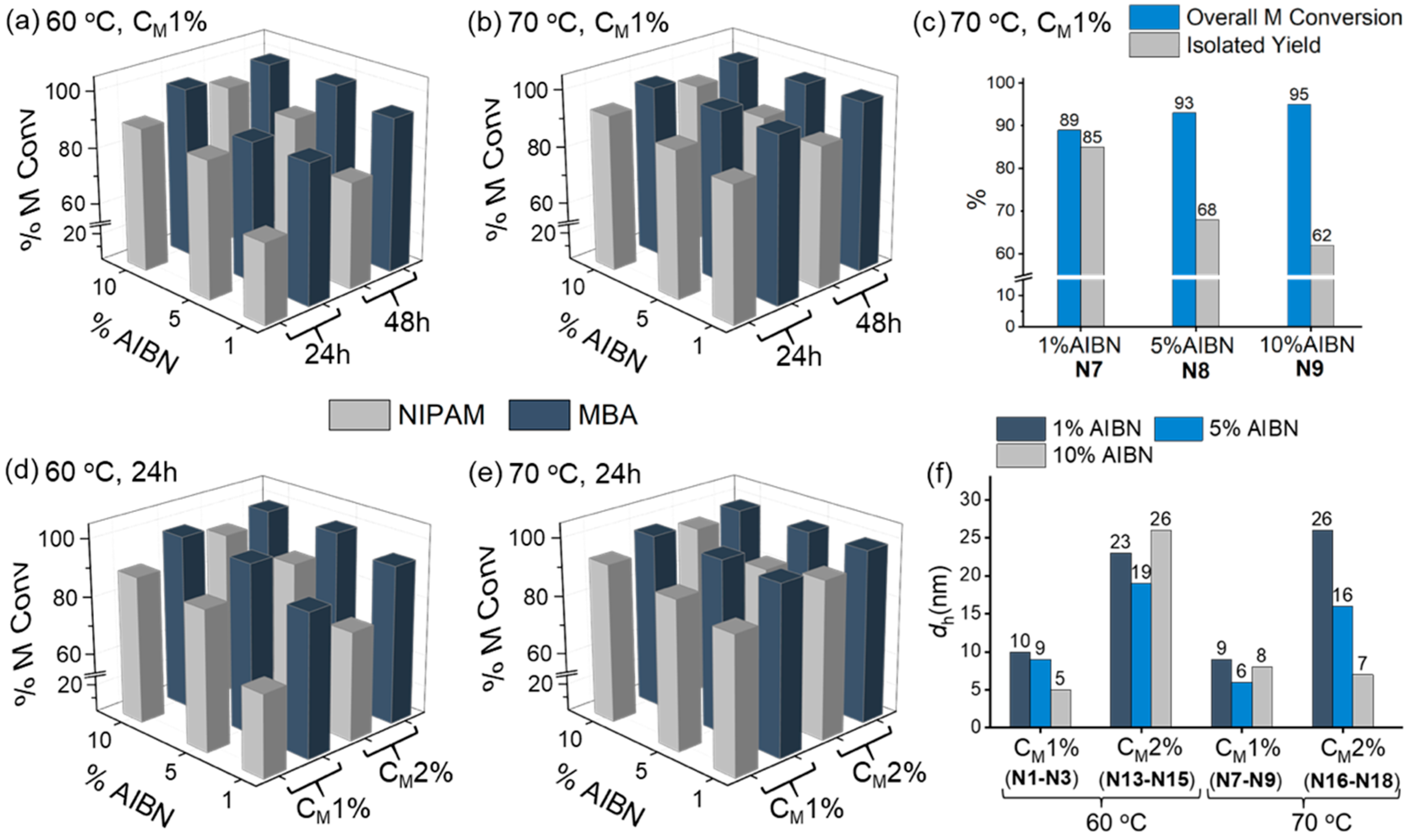
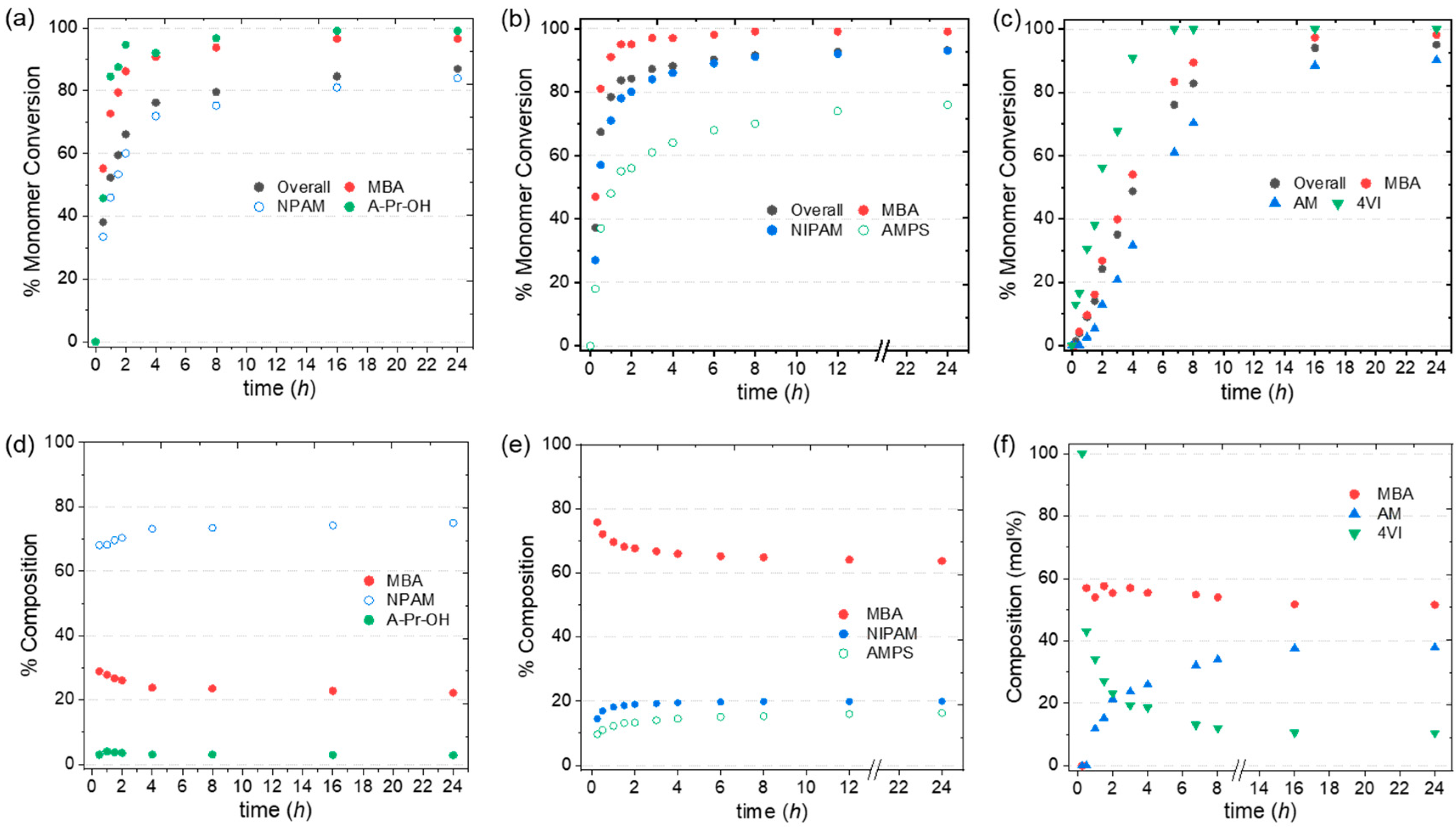
3.1. Two-Monomer Nanogels
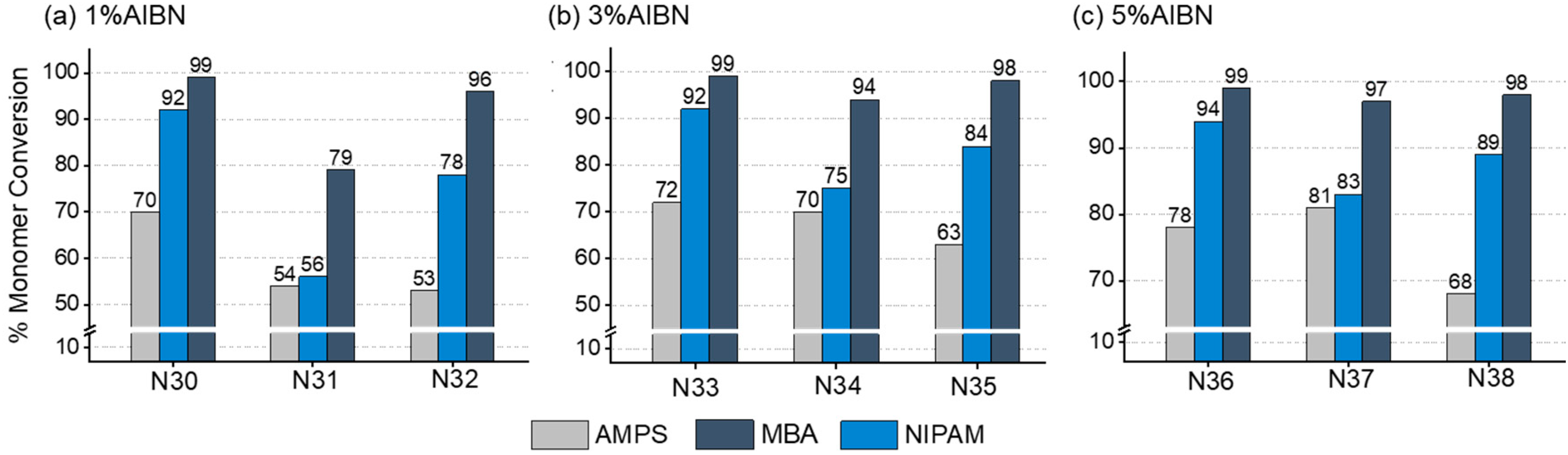
3.2. Three-Monomer System: Chemical Composition of pH and Temperature-Responsive Nanogels
3.3. Three-Monomer Polymers in Combination with a Template Molecule
The Effect of Introducing a Vinyl Monomer
3.4. Kinetic Study of Selected Two- and Three-Monomer systems
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Oh, J.K.; Drumright, R.; Siegwart, D.J.; Matyjaszewski, K. The development of microgels/nanogels for drug delivery applications. Prog. Polym. Sci. 2008, 33, 448–477. [Google Scholar] [CrossRef]
- Chan, M.; Almutairi, A. Nanogels as imaging agents for modalities spanning the electromagnetic spectrum. Mater. Horiz. 2016, 3, 21–40. [Google Scholar] [CrossRef] [PubMed]
- Soni, K.S.; Desale, S.S.; Bronich, T.K. Nanogels: An overview of properties, biomedical applications and obstacles to clinical translation. J. Control. Release 2016, 240, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhai, Y.; Wang, J.; Zhai, G. New progress and prospects: The application of nanogel in drug delivery. Mater. Sci. Eng. C 2016, 60, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Resmini, M.; Flavin, K.; Carboni, D. Microgels and nanogels with catalytic activity. Top. Curr. Chem. 2012, 325, 307–342. [Google Scholar] [CrossRef] [PubMed]
- Salinas, Y.; Castilla, A.M.; Resmini, M. An l-proline based thermoresponsive and pH-switchable nanogel as a drug delivery vehicle. Polym. Chem. 2018, 9, 2271–2280. [Google Scholar] [CrossRef]
- Lux, J.; White, A.G.; Chan, M.; Anderson, C.J.; Almutairi, A. Nanogels from metal-chelating crosslinkers as versatile platforms applied to copper-64 PET imaging of tumors and metastases. Theranostics 2015, 5, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, A.; Martínez, F.; Economopoulos, V.; Foster, P.J.; Scholl, T.J.; Gillies, E.R. Polymer cross-linking: A nanogel approach to enhancing the relaxivity of MRI contrast agents. J. Mater. Chem. B 2013, 1, 1027–1034. [Google Scholar] [CrossRef]
- Wedel, B.; Hertle, Y.; Wrede, O.; Bookhold, J.; Hellweg, T. Smart Homopolymer Microgels: Influence of the Monomer Structure on the Particle Properties. Polymers 2016, 8, 162. [Google Scholar] [CrossRef]
- Vicario-de-la-Torre, M.; Forcada, J. The Potential of Stimuli-Responsive Nanogels in Drug and Active Molecule Delivery for Targeted Therapy. Gels 2017, 3, 16. [Google Scholar] [CrossRef]
- Karimi, M.; Ghasemi, A.; Sahandi Zangabad, P.; Rahighi, R.; Moosavi Basri, S.M.; Mirshekari, H.; Amiri, M.; Shafaei Pishabad, Z.; Aslani, A.; Bozorgomid, M.; et al. Smart micro/nanoparticles in stimulus-responsive drug/gene delivery systems. Chem. Soc. Rev. 2016, 45, 1457–1501. [Google Scholar] [CrossRef] [PubMed]
- Molina, M.; Asadian-Birjand, M.; Balach, J.; Bergueiro, J.; Miceli, E.; Calderón, M. Stimuli-responsive nanogel composites and their application in nanomedicine. Chem. Soc. Rev. 2015, 44, 6161–6186. [Google Scholar] [CrossRef] [PubMed]
- Mura, S.; Nicolas, J.; Couvreur, P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2013, 12, 991. [Google Scholar] [CrossRef] [PubMed]
- Oishi, M.; Nagasaki, Y. Stimuli-responsive smart nanogels for cancer diagnostics and therapy. Nanomedicine 2010, 5, 451–468. [Google Scholar] [CrossRef] [PubMed]
- Neamtu, I.; Rusu, A.G.; Diaconu, A.; Nita, L.E.; Chiriac, A.P. Basic concepts and recent advances in nanogels as carriers for medical applications. Drug Deliv. 2017, 24, 539–557. [Google Scholar] [CrossRef] [PubMed]
- Sanson, N.; Rieger, J. Synthesis of nanogels/microgels by conventional and controlled radical crosslinking copolymerization. Polym. Chem. 2010, 1, 965–977. [Google Scholar] [CrossRef]
- An, Z.; Qiu, Q.; Liu, G. Synthesis of architecturally well-defined nanogels viaRAFT polymerization for potential bioapplications. Chem. Commun. 2011, 47, 12424–12440. [Google Scholar] [CrossRef] [PubMed]
- Moad, G. RAFT polymerization to form stimuli-responsive polymers. Polym. Chem. 2017, 8, 177–219. [Google Scholar] [CrossRef]
- Liu, J.; Rad, I.Y.; Sun, F.; Stansbury, J.W. Photo-reactive nanogels as a means to tune properties during polymer network formation. Polym. Chem. 2014, 5, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Graham, N.B.; Cameron, A. Nanogels and microgels: The new polymeric materials playground. Pure Appl. Chem. 1998, 70, 1271–1275. [Google Scholar] [CrossRef]
- Maddock, S.C.; Pasetto, P.; Resmini, M. Novel imprinted soluble microgels with hydrolytic catalytic activity. Chem. Commun. 2004, 536–537. [Google Scholar] [CrossRef] [PubMed]
- Rey, M.; Hou, X.; Tang, J.S.J.; Vogel, N. Interfacial arrangement and phase transitions of PNiPAm microgels with different crosslinking densities. Soft Matter 2017, 13, 8717–8727. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Tian, Y.; Peng, S.; Zhang, L.; Men, Y.; Yang, W. Poly(2-methacryloyloxyethyl phosphorylcholine)-based biodegradable nanogels for controlled drug release. Polym. Chem. 2018, 9, 4556–4565. [Google Scholar] [CrossRef]
- Cenci, L.; Tatti, R.; Tognato, R.; Ambrosi, E.; Piotto, C.; Bossi, A.M. Synthesis and characterization of peptide-imprinted nanogels of controllable size and affinity. Eur. Polym. J. 2018, 109, 453–459. [Google Scholar] [CrossRef]
- Papadimitriou, S.A.; Robin, M.P.; Ceric, D.; O’Reilly, R.K.; Marino, S.; Resmini, M. Fluorescent polymeric nanovehicles for neural stem cell modulation. Nanoscale 2016, 8, 17340–17349. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, K.; Sun, H.; Campbell, R.A.; Zarbakhsh, A.; Resmini, M. Smart nanogels at the air/water interface: Structural studies by neutron reflectivity. Nanoscale 2016, 8, 4951–4960. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, K.; Campbell, R.A.; Zarbakhsh, A.; Resmini, M. Adsorption versus aggregation of NIPAM nanogels: New insight into their behavior at the air/water interface as a function of concentration. Phys. Chem. Chem. Phys. 2017, 19, 17173–17179. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zielinska, K.; Resmini, M.; Zarbakhsh, A. Interactions of NIPAM nanogels with model lipid multi-bilayers: A neutron reflectivity study. J. Colloid Interface Sci. 2019, 536, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.H.; Hemp, S.T.; Smith, A.E.; Long, T.E. Controlled radical polymerization of 4-vinylimidazole. Macromolecules 2012, 45, 3669–3676. [Google Scholar] [CrossRef]
- Miao, J.; Cheng, Z.; Zhou, M.; Pan, D. Kinetic studies on the copolymerization of acrylonitrile and itaconic acid in dimethylsulfoxide. J. Macromol. Sci. A 2012, 49, 869–875. [Google Scholar] [CrossRef]
- Odian, G. Principles of Polymerization, 4th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004. [Google Scholar]
- Antonietti, M. Microgels—Polymers with a special molecular architecture. Angew. Chem. Int. Ed. Eng. 1988, 27, 1743–1747. [Google Scholar] [CrossRef]
- Pelton, R. Temperature-sensitive aqueous microgels. Adv. Colloid Interface Sci. 2000, 85, 1–33. [Google Scholar] [CrossRef]
- Deininger, M.; Buchdunger, E.; Druker, B.J. The development of imatinib as a therapeutic agent for chronic myeloid leukemia. Blood 2005, 105, 2640–2653. [Google Scholar] [CrossRef] [PubMed]
- Demetri, G.D.; von Mehren, M.; Blanke, C.D.; Van den Abbeele, A.D.; Eisenberg, B.; Roberts, P.J.; Heinrich, M.C.; Tuveson, D.A.; Singer, S.; Janicek, M.; et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N. Engl. J. Med. 2002, 347, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Barbero, A.; Fernández-Nieves, A.; Grillo, I.; López-Cabarcos, E. Structural modifications in the swelling of inhomogeneous microgels by light and neutron scattering. Phys. Rev. E 2002, 66, 051803. [Google Scholar] [CrossRef] [PubMed]

| NG No. | Feed Composition | Monomer Conversion 1 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| NIPAM | NPAM | APrOH | MBA | NIPAM | NPAM | APrOH | MBA | C2 | VPTT 3 | |
| mol % Monomer | mol % CL | % | % | % | °C | |||||
| N19 | 95 | 0 | 0 | 5 | 81 | 82 | 81 | 37 | ||
| N20 | 90 | 0 | 0 | 10 | 83 | 95 | 84 | 41 | ||
| N7 | 80 | 0 | 0 | 20 | 87 | 98 | 89 | 39 | ||
| N21 | 0 | 95 | 0 | 5 | 77 | 87 | 77 | 27 | ||
| N22 | 0 | 90 | 0 | 10 | 83 | 94 | 84 | 32 | ||
| N23 | 0 | 80 | 0 | 20 | 86 | 97 | 88 | 34 | ||
| N24 | 92.5 | 0 | 2.5 | 5 | 86 | >99 | 86 | 86 | 42 | |
| N25 | 87.5 | 0 | 2.5 | 10 | 88 | >99 | 94 | 89 | 48 | |
| N26 | 77.5 | 0 | 2.5 | 20 | 90 | >99 | 97 | 92 | 58 | |
| N27 | 0 | 92.5 | 2.5 | 5 | 71 | 90 | 85 | 72 | 29 | |
| N28 | 0 | 87.5 | 2.5 | 10 | 81 | 98 | 97 | 83 | 34 | |
| N29 | 0 | 77.5 | 2.5 | 20 | 85 | 98 | 98 | 88 | 40 | |
| NG No. | Feed Composition | Monomer Conversion 1 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 4VI | AM | NIPAM | MBA | 4VI | AM | NIPAM | MBA | C2 | |
| mol % monomer | mol % CL | % | % | % | |||||
| N8 3 | 0 | 0 | 80 | 20 | - | - | 91 | 99 | 93 |
| N45 | 0 | 80 | 0 | 20 | - | 87 | - | 96 | 89 |
| N46 | 80 | 0 | 0 | 20 | 37 | - | - | 31 | 36 |
| N47 | 10 | 0 | 70 | 20 | 98 | - | 53 | 77 | 70 |
| N48 | 10 | 70 | 0 | 20 | 100 | 60 | - | 83 | 76 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, P.; Pearce, C.M.; Anastasiadi, R.-M.; Resmini, M.; Castilla, A.M. Covalently Crosslinked Nanogels: An NMR Study of the Effect of Monomer Reactivity on Composition and Structure. Polymers 2019, 11, 353. https://doi.org/10.3390/polym11020353
Liu P, Pearce CM, Anastasiadi R-M, Resmini M, Castilla AM. Covalently Crosslinked Nanogels: An NMR Study of the Effect of Monomer Reactivity on Composition and Structure. Polymers. 2019; 11(2):353. https://doi.org/10.3390/polym11020353
Chicago/Turabian StyleLiu, Pengfei, Charles M. Pearce, Rozalia-Maria Anastasiadi, Marina Resmini, and Ana M. Castilla. 2019. "Covalently Crosslinked Nanogels: An NMR Study of the Effect of Monomer Reactivity on Composition and Structure" Polymers 11, no. 2: 353. https://doi.org/10.3390/polym11020353
APA StyleLiu, P., Pearce, C. M., Anastasiadi, R.-M., Resmini, M., & Castilla, A. M. (2019). Covalently Crosslinked Nanogels: An NMR Study of the Effect of Monomer Reactivity on Composition and Structure. Polymers, 11(2), 353. https://doi.org/10.3390/polym11020353