Acemannan Gels and Aerogels
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. AC Isolation
2.3. AC Characterization
2.4. AC Gelation
2.5. AC Aerogel Formation
2.6. Gels and Aerogels Characterization
2.6.1. Syneresis and Shrinkage
2.6.2. Structural Analysis
2.6.3. Chemical Identity
3. Results and Discussion
3.1. AC Characterization
3.2. AC Gelation and Aerogels Characterization
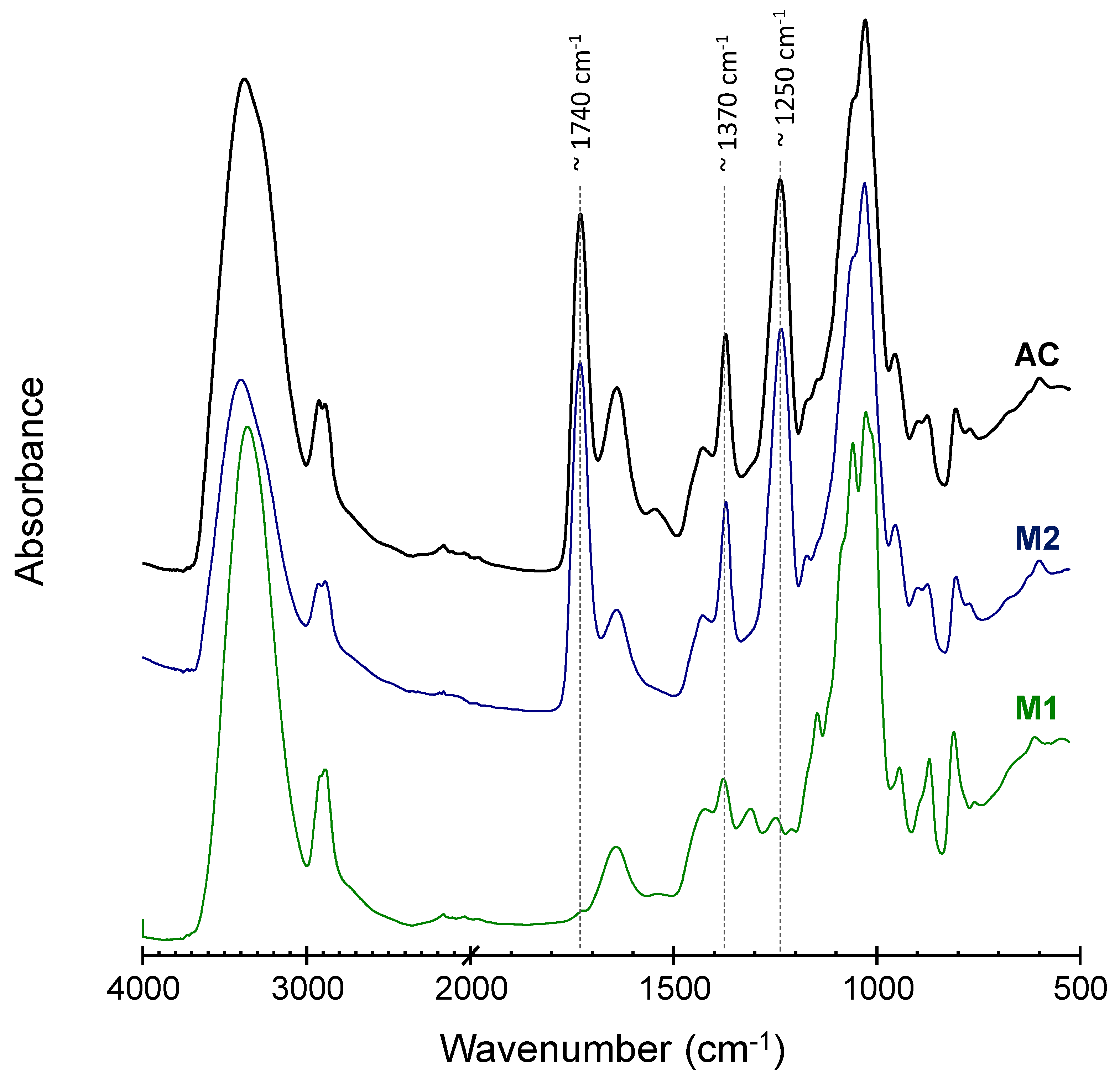
3.3. AC and Aerogels Chemical Identity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kumar, S.; Tiku, A.B. Immunomodulatory potential of acemannan (polysaccharide from Aloe vera) against radiation induced mortality in Swiss albino mice. Food Agric. Immunol. 2016, 27, 72–86. [Google Scholar] [CrossRef]
- Silva, S.S.; Caridade, S.G.; Mano, J.F.; Reis, R.L. Effect of crosslinking in chitosan/aloe vera-based membranes for biomedical applications. Carbohydr. Polym. 2013, 98, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.S.; Oliveira, M.B.; Mano, J.F.; Reis, R.L. Bio-inspired Aloe vera sponges for biomedical applications. Carbohydr. Polym. 2014, 112, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Berger, J.; Reist, M.; Mayer, J.M.; Felt, O.; Peppas, N.A.; Gurny, R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur. J. Pharm. Biopharm. 2004, 57, 19–34. [Google Scholar] [CrossRef]
- Zhang, C. Interplay between long-range and short-range interactions in polymer self-assembly and cell adhesion. Ph.D. Thesis, California Institute of Technology, Pasadena, CA, USA, 2008. [Google Scholar]
- García-González, C.A.; Alnaief, M.; Smirnova, I. Polysaccharide-based aerogels - Promising biodegradable carriers for drug delivery systems. Carbohydr. Polym. 2011, 86, 1425–1438. [Google Scholar] [CrossRef]
- Mehling, T.; Smirnova, I.; Guenther, U.; Neubert, R.H.H. Polysaccharide-based aerogels as drug carriers. J. Non-Cryst. Solids 2009, 355, 2472–2479. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Y.; Jiang, H.; Song, Y.; Zhou, Z.; Zhao, H. Fabrication and characterization of nano-cellulose aerogels via supercritical CO2 drying technology. Mater. Lett. 2016, 183, 179–182. [Google Scholar] [CrossRef]
- Campestrini, L.H.; Silveira, J.L.M.; Duarte, M.E.R.; Koop, H.S.; Noseda, M.D. NMR and rheological study of Aloe barbadensis partially acetylated glucomannan. Carbohydr. Polym. 2013, 94, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Hemery, Y.; Holopainen, U.; Lampi, A.M.; Lehtinen, P.; Nurmi, T.; Piironen, V.; Edelmann, M.; Rouau, X. Potential of dry fractionation of wheat bran for the development of food ingredients, part II: Electrostatic separation of particles. J. Cereal Sci. 2011, 53, 9–18. [Google Scholar] [CrossRef]
- Escobedo-Lozano, A.Y.; Domard, A.; Velázquez, C.A.; Goycoolea, F.M.; Argüelles-Monal, W.M. Physical properties and antibacterial activity of chitosan/acemannan mixed systems. Carbohydr. Polym. 2015, 115, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Montembault, A.; Viton, C.; Domard, A. Rheometric Study of the Gelation of Chitosan in Aqueous Solution without Cross-Linking Agent. Biomacromolecules 2005, 6, 653–662. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Turner, D.; Yates, K.M.; Tizard, I. Isolation and characterization of structural components of Aloe vera L. leaf pulp. Int. Immunopharmacol. 2004, 4, 1745–1755. [Google Scholar] [CrossRef] [PubMed]
- Chokboribal, J.; Tachaboonyakiat, W.; Sangvanich, P.; Ruangpornvisuti, V.; Jettanacheawchankit, S.; Thunyakitpisal, P. Deacetylation affects the physical properties and bioactivity of acemannan, an extracted polysaccharide from Aloe vera. Carbohydr. Polym. 2015, 133, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Yates, K.M.; Tizard, I. Aloe polysaccharides. In Aloes: the genus Aloe; Reynolds, T., Ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 76–82. [Google Scholar]
- Wozniewski, T.; Wolfgang, B.; Franz, G. Isolation and structure analysis of a glucomannan from the leaves of Arborescens var. Miller. Carbohydr. Res. 1990, 198, 387–391. [Google Scholar] [CrossRef]
- Femenia, A.; Sánchez, E.S.; Simal, S.; Rosselló, C. Compositional features of polysaccharides from Aloe vera (Aloe barbadensis Miller) plant tissues. Carbohydr. Polym. 1999, 39, 109–117. [Google Scholar] [CrossRef]
- Turner, C.E.; Williamson, D.A.; Stroud, P.A.; Talley, D.J. Evaluation and comparison of commercially available Aloe vera L. products using size exclusion chromatography with refractive index and multi-angle laser light scattering detection. Int. Immunopharmacol. 2004, 4, 1727–1737. [Google Scholar] [CrossRef] [PubMed]
- Montembault, A.; Viton, C.; Domard, A. Rheometric study of the gelation of chitosan in a hydroalcoholic medium. Biomaterials 2005, 26, 1633–1643. [Google Scholar] [CrossRef] [PubMed]
- Subrahmanyam, R.; Gurikov, P.; Dieringer, P.; Sun, M.; Smirnova, I. On the Road to Biopolymer Aerogels—Dealing with the Solvent. Gels 2015, 1, 291–313. [Google Scholar] [CrossRef] [PubMed]
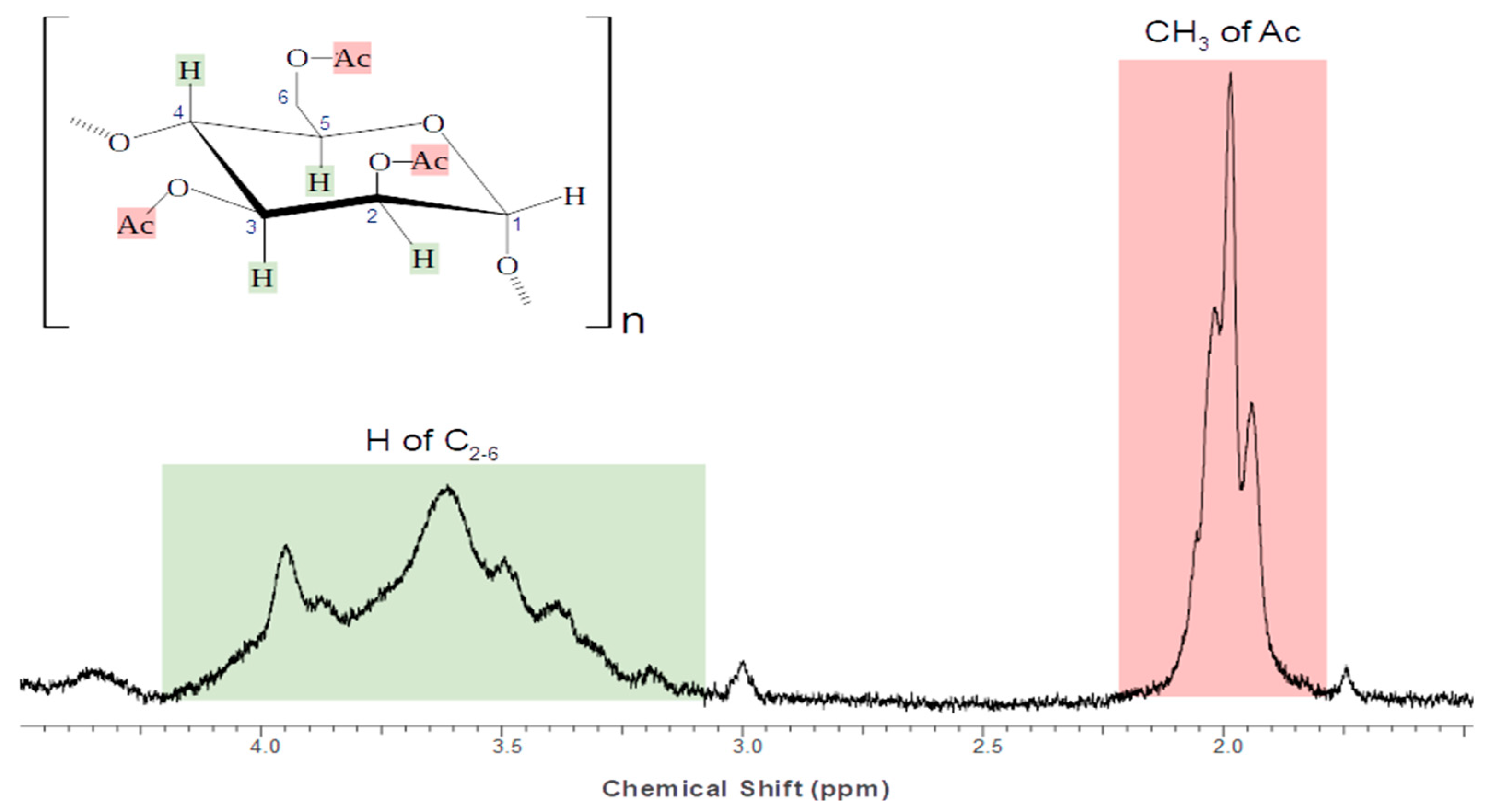




| Characteristic | Value | Unit |
|---|---|---|
| DA a | 1.31 ± 0.01 | |
| dn/dc b | 0.16 ± 0.00 | |
| Mw c | 1500 | kDa |
| Mn c | 870 | kDa |
| Ip c | 1.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miramon-Ortíz, D.A.; Argüelles-Monal, W.; Carvajal-Millan, E.; López-Franco, Y.L.; Goycoolea, F.M.; Lizardi-Mendoza, J. Acemannan Gels and Aerogels. Polymers 2019, 11, 330. https://doi.org/10.3390/polym11020330
Miramon-Ortíz DA, Argüelles-Monal W, Carvajal-Millan E, López-Franco YL, Goycoolea FM, Lizardi-Mendoza J. Acemannan Gels and Aerogels. Polymers. 2019; 11(2):330. https://doi.org/10.3390/polym11020330
Chicago/Turabian StyleMiramon-Ortíz, Daniel Alonso, Waldo Argüelles-Monal, Elizabeth Carvajal-Millan, Yolanda Leticia López-Franco, Francisco M. Goycoolea, and Jaime Lizardi-Mendoza. 2019. "Acemannan Gels and Aerogels" Polymers 11, no. 2: 330. https://doi.org/10.3390/polym11020330
APA StyleMiramon-Ortíz, D. A., Argüelles-Monal, W., Carvajal-Millan, E., López-Franco, Y. L., Goycoolea, F. M., & Lizardi-Mendoza, J. (2019). Acemannan Gels and Aerogels. Polymers, 11(2), 330. https://doi.org/10.3390/polym11020330








