Polyetherimide Foams Filled with Low Content of Graphene Nanoplatelets Prepared by scCO2 Dissolution
Abstract
1. Introduction
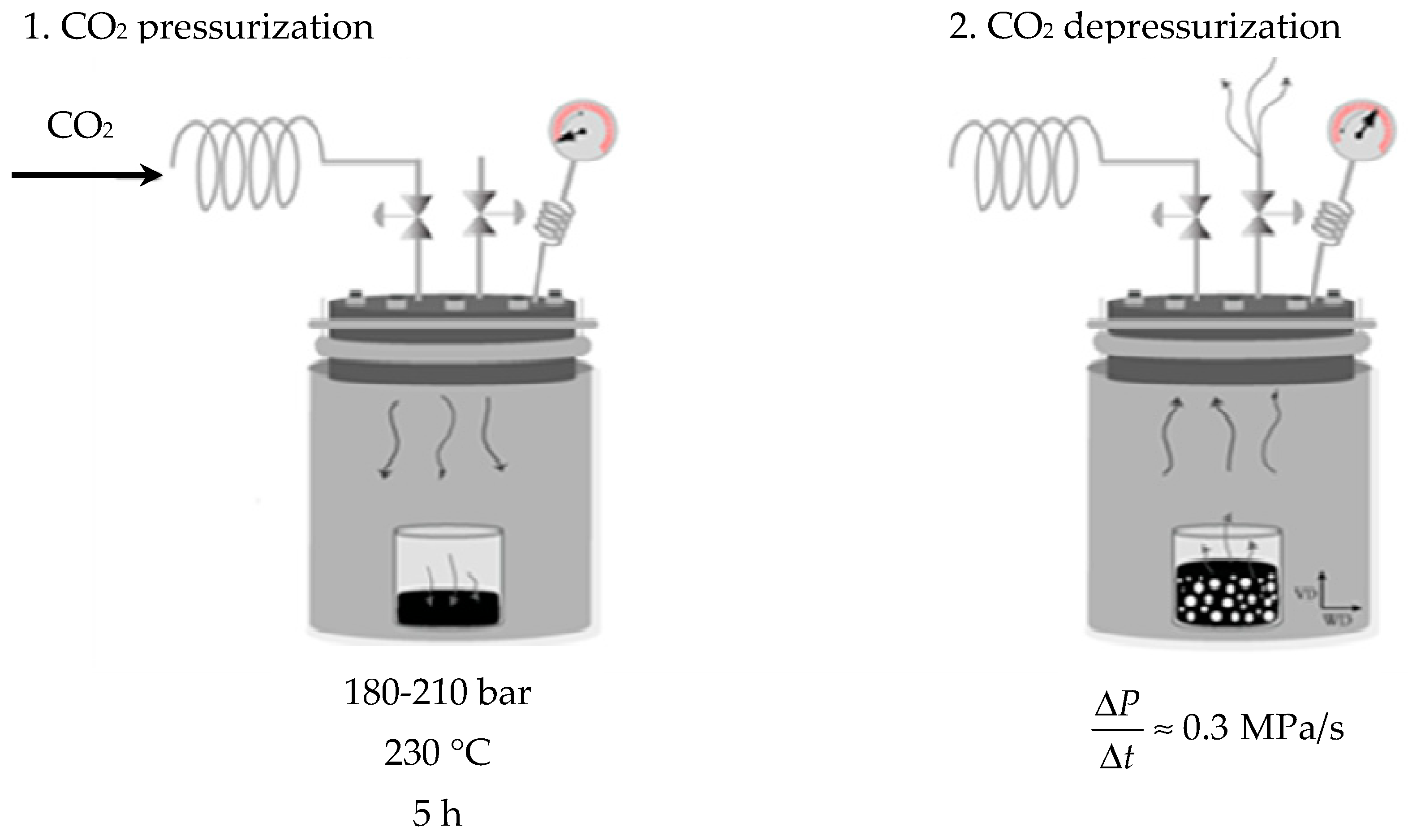
2. Materials and Methods
3. Results
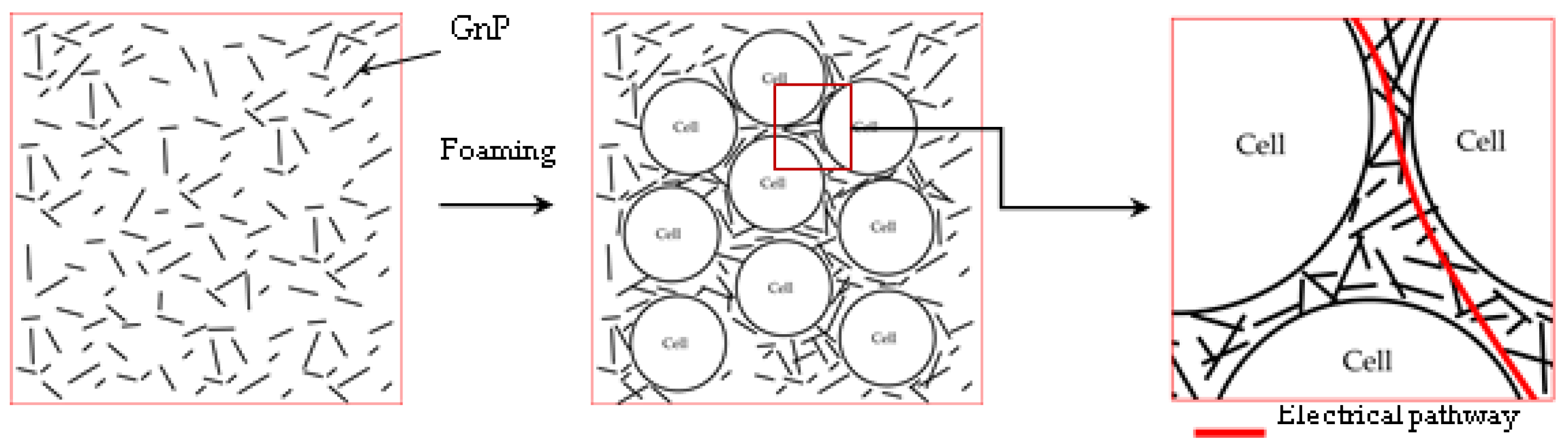
3.1. Cellular Structure of the Foams
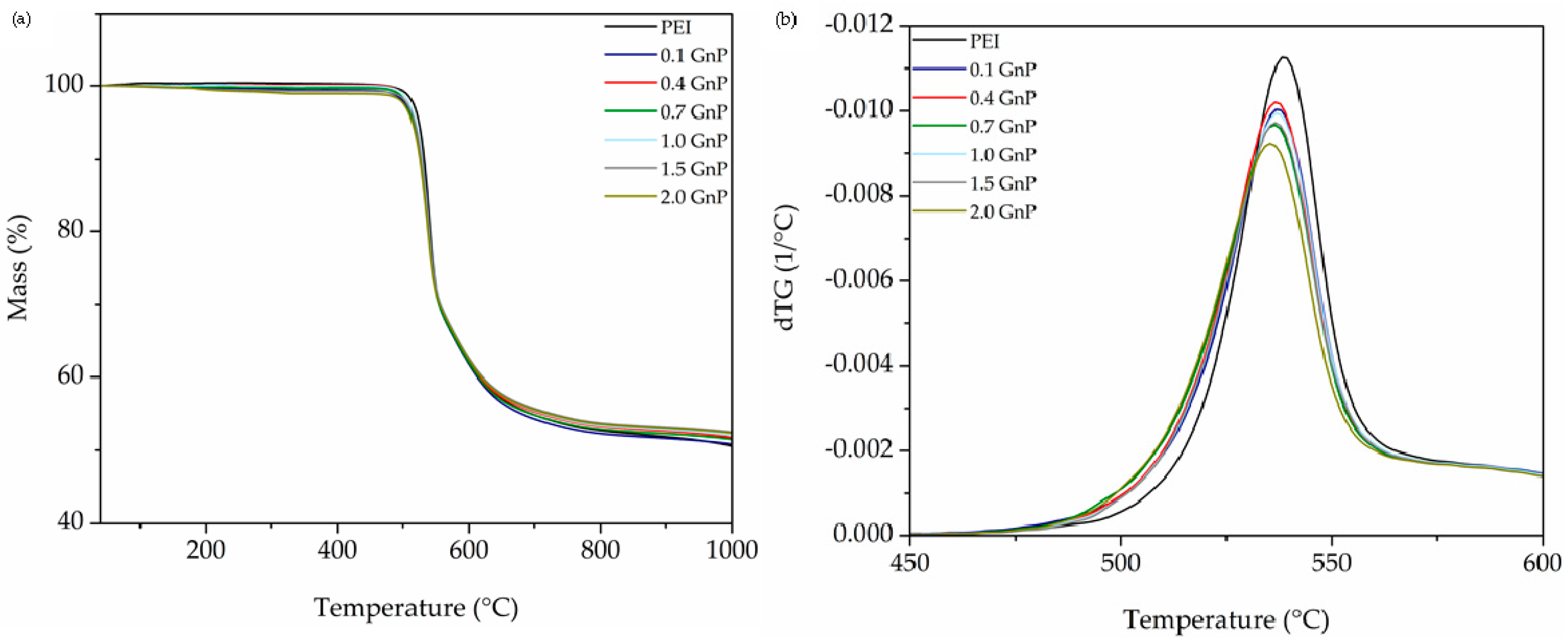
3.2. Thermal Analysis
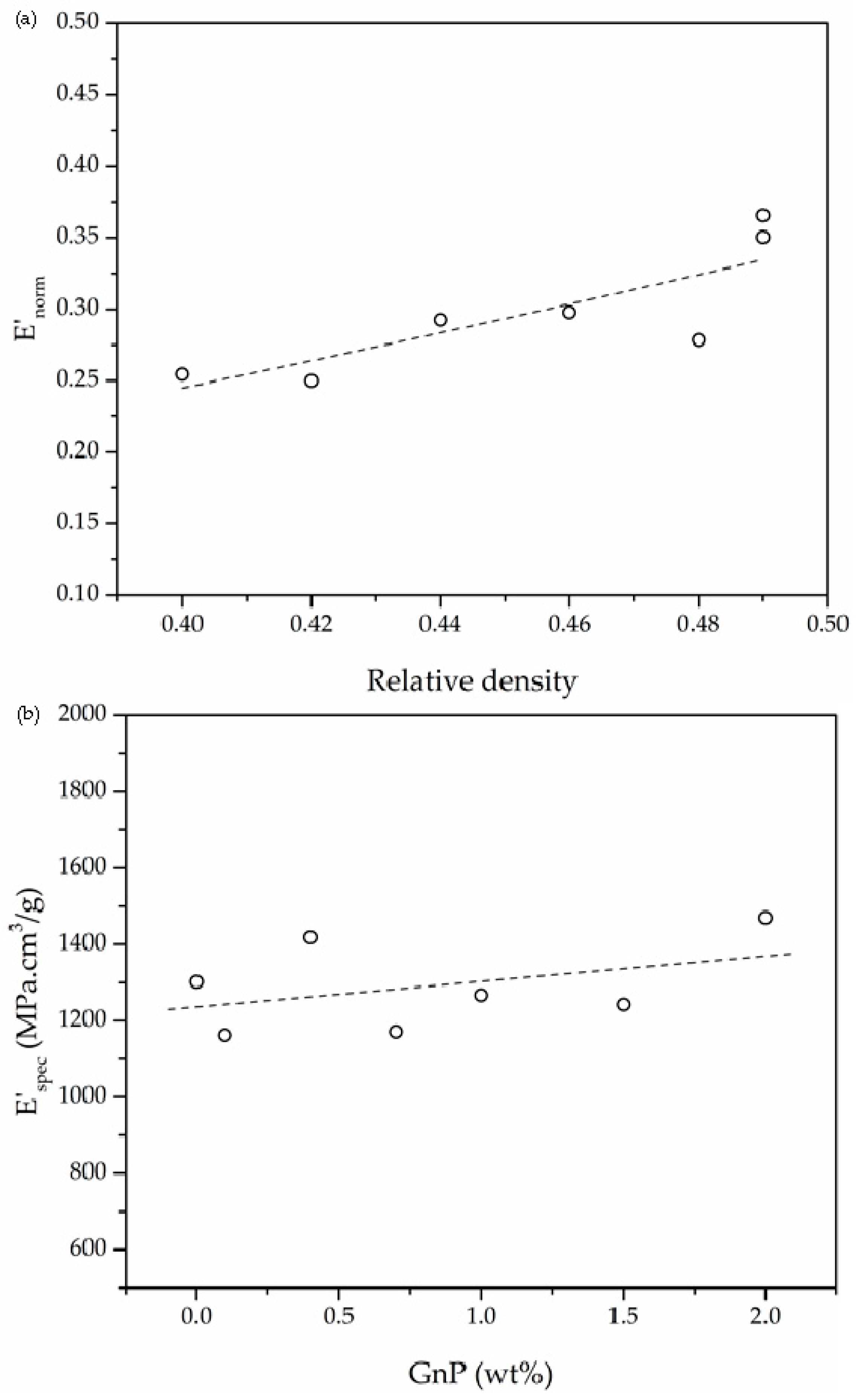
3.3. Dynamic-Mechanical-Thermal Behavior
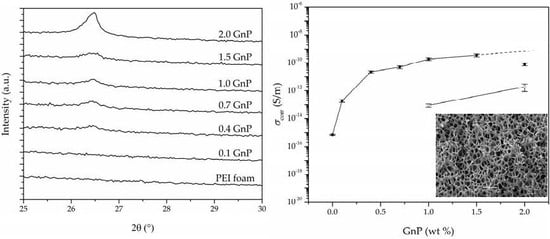
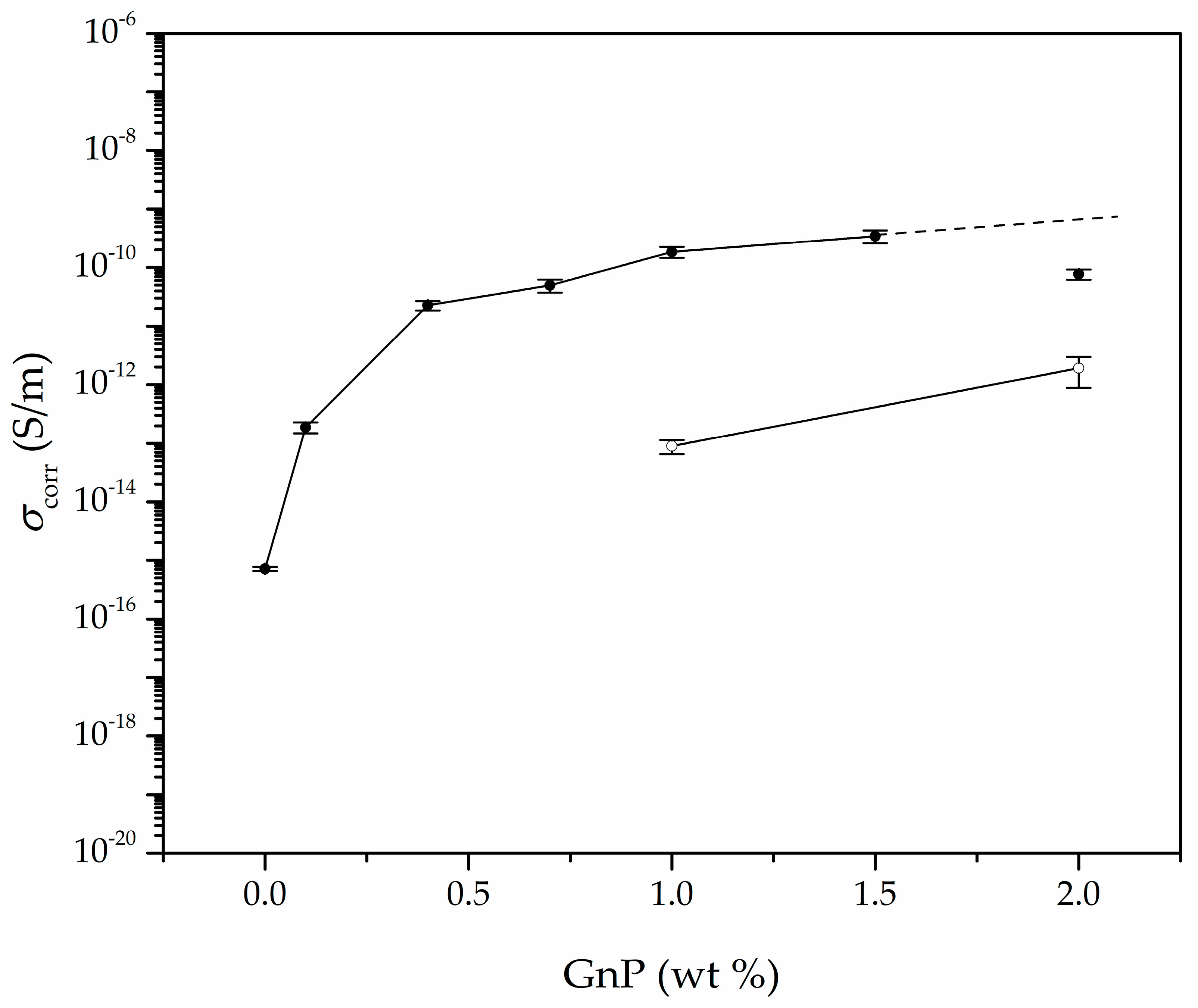
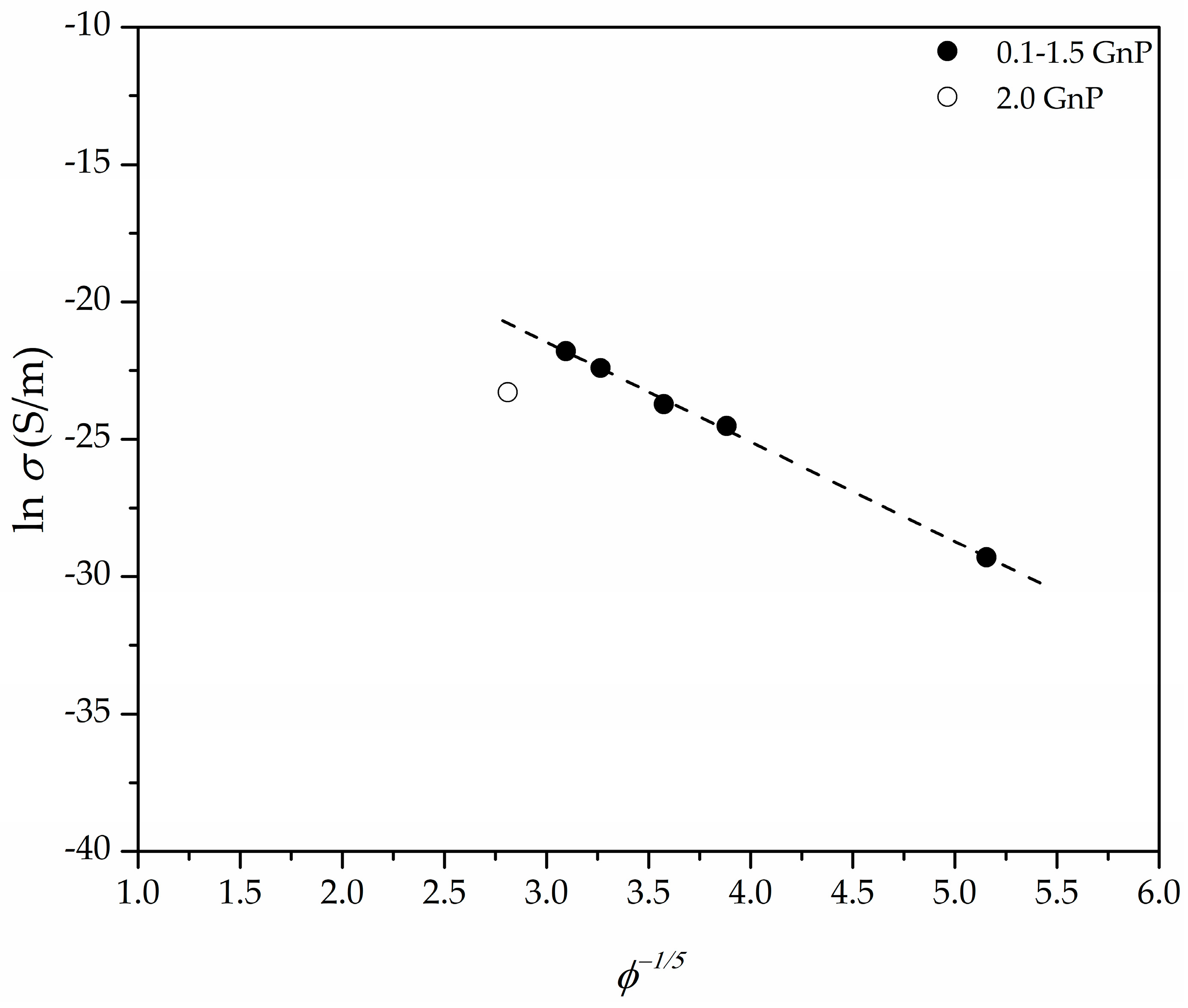
3.4. Electrical Conductivity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- McKeen, L.W. Polyimides. In Permeability Properties of Plastics and Elastomers; William Andrew Publishing: Oxford, UK, 2012; pp. 107–120. ISBN 978-1-4377-3469-0. [Google Scholar]
- Gedler, G.; Antunes, M.; Velasco, J.I.; Ozisik, R. Enhanced electromagnetic interference shielding effectiveness of polycarbonate/graphene nanocomposites foamed via 1-step supercritical carbon dioxide process. Mater. Des. 2016, 90, 906–914. [Google Scholar] [CrossRef]
- Ling, J.; Zhai, W.; Feng, W.; Shen, B.; Zhang, J.; Zheng, W. Ge Facile Preparation of Lightweight Microcellular Polyetherimide/Graphene Composite Foams for Electromagnetic Interference Shielding. ACS Appl. Mater. Interfaces 2013, 5, 2677–2684. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, H.; Antunes, M.; Velasco, J.I. Graphene nanoplatelets-reinforced polyetherimide foams prepared by water vapor-induced phase separation. eXPRESS Polym. Lett. 2015, 9, 412–423. [Google Scholar] [CrossRef]
- Abbasi, H.; Antunes, M.; Velasco, J. Effects of Carbon Nanotubes/Graphene Nanoplatelets Hybrid Systems on the Structure and Properties of Polyetherimide-Based Foams. Polymers 2018, 10, 348. [Google Scholar] [CrossRef]
- Abbasi, H.; Antunes, M.; Velasco, J.I. Enhancing the electrical conductivity of polyetherimide-based foams by simultaneously increasing the porosity and graphene nanoplatelets dispersion. Polym. Compos. 2018. [Google Scholar] [CrossRef]
- Shen, B.; Zhai, W.; Tao, M.; Ling, J.; Zheng, W. Lightweight, Multifunctional Polyetherimide/Graphene@Fe3O4 Composite Foams for Shielding of Electromagnetic Pollution. ACS Appl. Mater. Interfaces 2013, 5, 11383–11391. [Google Scholar] [CrossRef]
- Nadella, K.; Kumar, V. Tensile and flexural properties of solid-state microcellular ABS panels. In Experimental Analysis of Nano and Engineering Materials and Structures; Springer: Berlin/Heidelberg, Germany, 2007; pp. 765–766. [Google Scholar]
- Martín-de León, J.; Bernardo, V.; Rodríguez-Pérez, M.Á. Low Density Nanocellular Polymers Based on PMMA Produced by Gas Dissolution Foaming: Fabrication and Cellular Structure Characterization. Polymers 2016, 8, 265. [Google Scholar] [CrossRef]
- Zhang, H.-C.; Yu, C.-N.; Liang, Y.; Lin, G.-X.; Meng, C. Foaming Behavior and Microcellular Morphologies of Incompatible SAN/CPE Blends with Supercritical Carbon Dioxide as a Physical Blowing Agent. Polymers 2019, 11, 89. [Google Scholar] [CrossRef]
- Kumar, V.; VanderWel, M.; Weller, J.; Seeler, K.A. Experimental Characterization of the Tensile Behavior of Microcellular Polycarbonate Foams. J. Eng. Mater. Technol. 1994, 116, 439–445. [Google Scholar] [CrossRef]
- Shimbo, M.; Higashitani, I.; Miyano, Y. Mechanism of Strength Improvement of Foamed Plastics Having Fine Cell. J. Cell. Plast. 2007, 43, 157–167. [Google Scholar] [CrossRef]
- Miller, D.; Chatchaisucha, P.; Kumar, V. Microcellular and nanocellular solid-state polyetherimide (PEI) foams using sub-critical carbon dioxide I. Processing and structure. Polymer 2009, 50, 5576–5584. [Google Scholar] [CrossRef]
- Miller, D.; Kumar, V. Microcellular and nanocellular solid-state polyetherimide (PEI) foams using sub-critical carbon dioxide II. Tensile and impact properties. Polymer 2011, 52, 2910–2919. [Google Scholar] [CrossRef]
- Badamshina, E.; Estrin, Y.; Gafurova, M. Nanocomposites based on polyurethanes and carbon nanoparticles: Preparation, properties and application. J. Mater. Chem. A 2013, 1, 6509–6529. [Google Scholar] [CrossRef]
- Kostopoulos, V.; Vavouliotis, A.; Karapappas, P.; Tsotra, P.; Paipetis, A. Damage Monitoring of Carbon Fiber Reinforced Laminates Using Resistance Measurements. Improving Sensitivity Using Carbon Nanotube Doped Epoxy Matrix System. J. Intell. Mater. Syst. Struct. 2009, 20, 1025–1034. [Google Scholar] [CrossRef]
- Potts, J.R.; Dreyer, D.R.; Bielawski, C.W.; Ruoff, R.S. Graphene-based polymer nanocomposites. Polymer 2011, 52, 5–25. [Google Scholar] [CrossRef]
- Wang, H.; Robinson, J.T.; Diankov, G.; Dai, H. Nanocrystal growth on graphene with various degrees of oxidation. J. Am. Chem. Soc. 2010, 132, 3270–3271. [Google Scholar] [CrossRef]
- Kuilla, T.; Bhadra, S.; Yao, D.; Kim, N.H.; Bose, S.; Lee, J.H. Recent advances in graphene based polymer composites. Prog. Polym. Sci. 2010, 35, 1350–1375. [Google Scholar] [CrossRef]
- Zhou, C.; Vaccaro, N.; Sundarram, S.S.; Li, W. Fabrication and characterization of polyetherimide nanofoams using supercritical CO2. J. Cell. Plast. 2012, 48, 239–255. [Google Scholar] [CrossRef]
- Gedler, G.; Antunes, M.; Velasco, J.I. Enhanced electrical conductivity in graphene-filled polycarbonate nanocomposites by microcellular foaming with sc-CO2. J. Adhes. Sci. Technol. 2016, 30, 1017–1029. [Google Scholar] [CrossRef]
- Ma, H.-L.; Zhang, H.-B.; Li, X.; Zhi, X.; Liao, Y.-F.; Yu, Z.-Z. The effect of surface chemistry of graphene on cellular structures and electrical properties of polycarbonate nanocomposite foams. Ind. Eng. Chem. Res. 2014, 53, 4697–4703. [Google Scholar] [CrossRef]
- Yang, C.; Zhao, Q.; Xing, Z.; Zhang, W.; Zhang, M.; Tan, H.; Wang, J.; Wu, G. Improving the Supercritical CO2 Foaming of Polypropylene by the Addition of Fluoroelastomer as a Nucleation Agent. Polymers 2019, 11, 226. [Google Scholar] [CrossRef]
- Ventura, H.; Sorrentino, L.; Laguna-Gutierrez, E.; Rodriguez-Perez, M.A.; Ardanuy, M. Gas Dissolution Foaming as a Novel Approach for the Production of Lightweight Biocomposites of PHB/Natural Fibre Fabrics. Polymers 2018, 10, 249. [Google Scholar] [CrossRef]
- Sims, G.L.A.; Khunniteekool, C. Cell-size measurement of polymeric foams. Cell. Polym. 1994, 13, 137–146. [Google Scholar]
- Van Krevelen, D.W.; Hoftyzer, P.J. Properties of Polymers: Their Estimation and Correlation with Chemical Structure; Elsevier: New York, NY, USA, 1976. [Google Scholar]
- Realinho, V.; Haurie, L.; Antunes, M.; Velasco, J.I. Thermal stability and fire behaviour of flame retardant high density rigid foams based on hydromagnesite-filled polypropylene composites. Compos. Part B Eng. 2014, 58, 553–558. [Google Scholar] [CrossRef]
- Yoon, O.J.; Jung, C.Y.; Sohn, I.Y.; Kim, H.J.; Hong, B.; Jhon, M.S.; Lee, N.-E. Nanocomposite nanofibers of poly(d,l-lactic-co-glycolic acid) and graphene oxide nanosheets. Compos. Part A Appl. Sci. Manuf. 2011, 42, 1978–1984. [Google Scholar] [CrossRef]
- Ji, X.; Xu, Y.; Zhang, W.; Cui, L.; Liu, J. Review of functionalization, structure and properties of graphene/polymer composite fibers. Compos. Part A Appl. Sci. Manuf. 2016, 87, 29–45. [Google Scholar] [CrossRef]
- Wang, B.; Chen, Z.; Zhang, J.; Cao, J.; Wang, S.; Tian, Q.; Gao, M.; Xu, Q. Fabrication of PVA/graphene oxide/TiO2 composite nanofibers through electrospinning and interface sol–gel reaction: Effect of graphene oxide on PVA nanofibers and growth of TiO2. Colloids Surf. A Physicochem. Eng. Asp. 2014, 457, 318–325. [Google Scholar] [CrossRef]
- Gibson, L.J.; Ashby, M.F. Cellular Solids: Structure and Properties; Cambridge University Press: Cambridge, UK, 1999; ISBN 131602542X. [Google Scholar]
- Kumar, V.; Nadella, K.V. Microcellular Foams. In Handbook of Polymer Foams; Eaves, D., Ed.; Rapa Technology Limited: Shawbury, UK, 2004; Volume 25, pp. 243–268. [Google Scholar]
- Zhou, J.; Yao, Z.; Chen, Y.; Wei, D.; Xu, T. Fabrication and mechanical properties of phenolic foam reinforced with graphene oxide. Polym. Compos. 2014, 35, 581–586. [Google Scholar]
- Yang, H.; Li, F.; Shan, C.; Han, D.; Zhang, Q.; Niu, L.; Ivaska, A. Covalent functionalization of chemically converted graphene sheets via silane and its reinforcement. J. Mater. Chem. 2009, 19, 4632–4638. [Google Scholar] [CrossRef]
- Chen, L.; Jin, H.; Xu, Z.; Shan, M.; Tian, X.; Yang, C.; Wang, Z.; Cheng, B. A design of gradient interphase reinforced by silanized graphene oxide and its effect on carbon fiber/epoxy interface. Mater. Chem. Phys. 2014, 145, 186–196. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, S.; Ji, J.; Li, Y.; Zhang, G.; Zhang, F.; Fan, X. Primary and tertiary amines bifunctional graphene oxide for cooperative catalysis. Nanoscale 2013, 5, 6030–6033. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Su, S.; Kasner, M.L.; Shah, P.; Patel, K.; Madarang, C.J. Formation of highly stable dispersions of silane-functionalized reduced graphene oxide. Chem. Phys. Lett. 2010, 501, 68–74. [Google Scholar] [CrossRef]
- Lin, Y.; Jin, J.; Song, M. Preparation and characterisation of covalent polymer functionalized graphene oxide. J. Mater. Chem. 2011, 21, 3455–3461. [Google Scholar] [CrossRef]
- Fang, M.; Wang, K.; Lu, H.; Yang, Y.; Nutt, S. Covalent polymer functionalization of graphene nanosheets and mechanical properties of composites. J. Mater. Chem. 2009, 19, 7098–7105. [Google Scholar] [CrossRef]
- Boukhvalov, D.W.; Katsnelson, M.I. Chemical functionalization of graphene with defects. Nano Lett. 2008, 8, 4373–4379. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Jang, J.; Nagase, S. Hydrazine and thermal reduction of graphene oxide: Reaction mechanisms, product structures, and reaction design. J. Phys. Chem. C 2009, 114, 832–842. [Google Scholar] [CrossRef]
- Iqbal, M.Z.; Katsiotis, M.S.; Alhassan, S.M.; Liberatore, M.W.; Abdala, A.A. Effect of solvent on the uncatalyzed synthesis of aminosilane-functionalized graphene. RSC Adv. 2014, 4, 6830–6839. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.B.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef]
- Chiu, F.-C. A review on conduction mechanisms in dielectric films. Adv. Mater. Sci. Eng. 2014, 2014. [Google Scholar] [CrossRef]
- Antunes, M.; Mudarra, M.; Velasco, J.I. Broad-band electrical conductivity of carbon nanofibre-reinforced polypropylene foams. Carbon N. Y. 2011, 49, 708–717. [Google Scholar] [CrossRef]
- Sichel, E.K. Carbon Black-Polymer Composites: The Physics of Electrically Conducting Composites; Marcel Dekker Inc.: New York, NY, USA, 1982; Volume 3, ISBN 0824716736. [Google Scholar]
- Ryvkina, N.; Tchmutin, I.; Vilčáková, J.; Pelíšková, M.; Sáha, P. The deformation behavior of conductivity in composites where charge carrier transport is by tunneling: Theoretical modeling and experimental results. Synth. Met. 2005, 148, 141–146. [Google Scholar] [CrossRef]
- Hull, D.; Clyne, T.W. An Introduction to Composite Materials; Cambridge University Press: Cambridge, UK, 1996; ISBN 0521388554. [Google Scholar]
- Allaoui, A.; Hoa, S.V.; Pugh, M.D. The electronic transport properties and microstructure of carbon nanofiber/epoxy composites. Compos. Sci. Technol. 2008, 68, 410–416. [Google Scholar] [CrossRef]
- Krenchel, H. Fibre Reinforcement; Alademisk forlag: Copenhagen, Denmark, 1964. [Google Scholar]
- Fisher, F.T.; Bradshaw, R.D.; Brinson, L.C. Fiber waviness in nanotube-reinforced polymer composites—I: Modulus predictions using effective nanotube properties. Compos. Sci. Technol. 2003, 63, 1689–1703. [Google Scholar] [CrossRef]

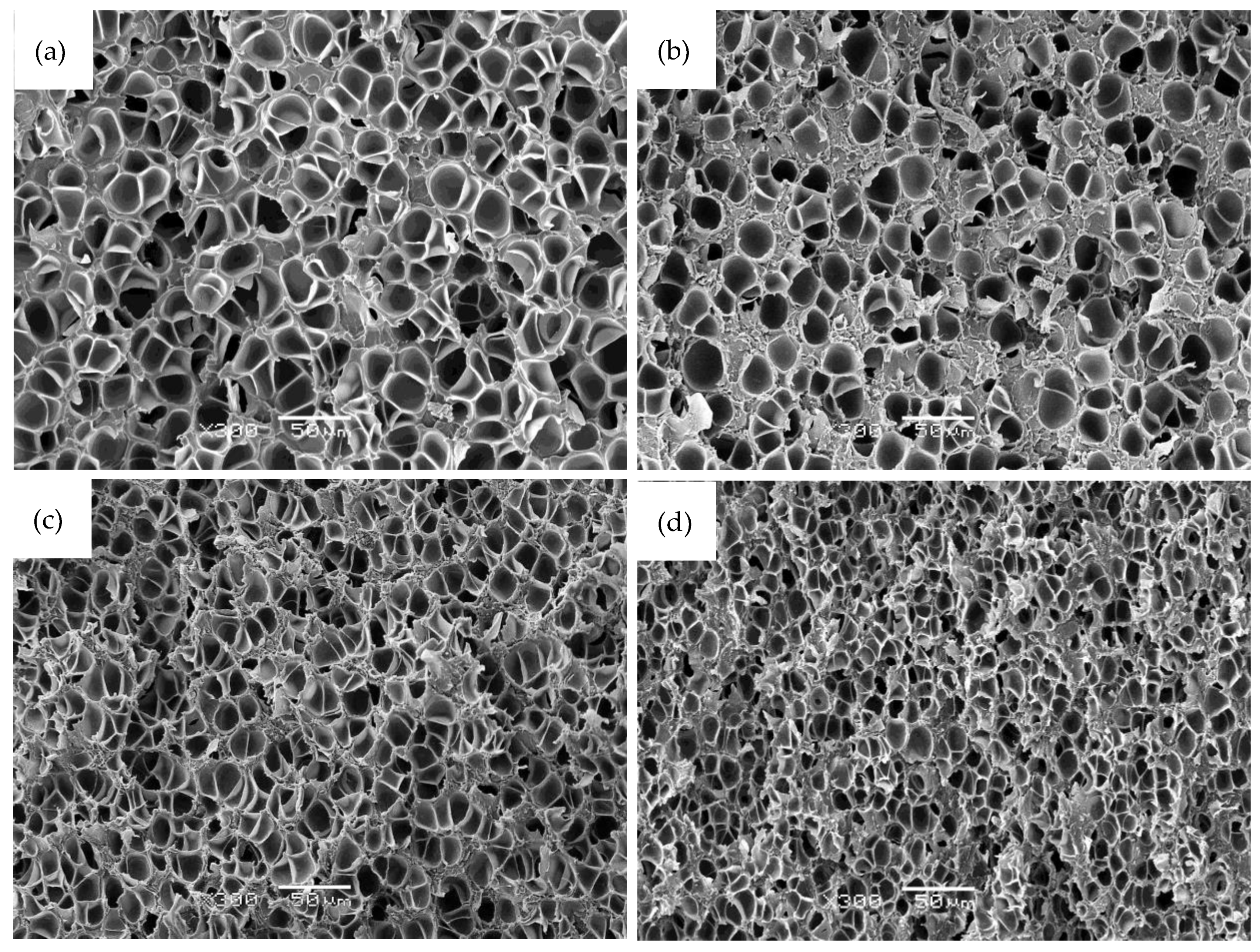
| Sample | GnP (wt %) | GnP (vol%) | Relative Density | Φ (μm) 1 | N0 (cells/cm3) |
|---|---|---|---|---|---|
| PEI | 0.0 | 0.00 | 0.44 | 14.0 (5.0) | 5.1 × 108 |
| 0.1 GnP | 0.1 | 0.03 | 0.48 | 11.7 (4.2) | 5.6 × 108 |
| 0.4 GnP | 0.4 | 0.11 | 0.49 | 13.6 (4.4) | 3.9 × 108 |
| 0.7 GnP | 0.7 | 0.17 | 0.42 | 5.4 (2.3) | 6.5 × 109 |
| 1.0 GnP | 1.0 | 0.27 | 0.46 | 9.5 (3.3) | 1.1 × 109 |
| 1.5 GnP | 1.5 | 0.35 | 0.40 | 10.0 (4.0) | 1.2 × 109 |
| 2.0 GnP | 2.0 | 0.57 | 0.49 | 7.5 (2.9) | 1.6 × 109 |
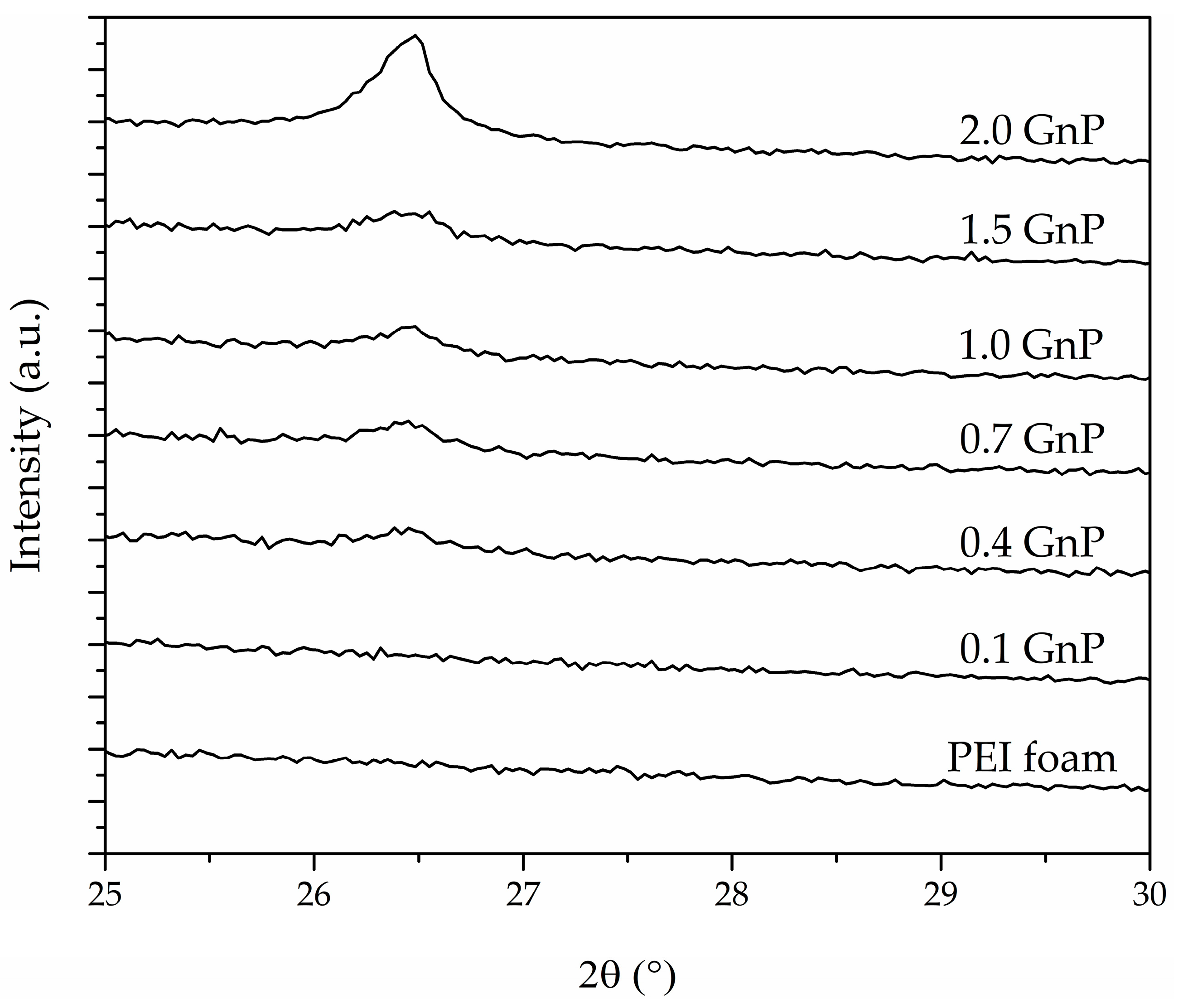
| Sample | Intensity (a.u.) | FWHM (°) |
|---|---|---|
| PEI | - | - |
| 0.1 GnP | - | - |
| 0.4 GnP | 350.5 | 0.23 |
| 0.7 GnP | 481.2 | 0.35 |
| 1.0 GnP | 505.4 | 0.30 |
| 1.5 GnP | 483.5 | 0.40 |
| 2.0 GnP | 1716.6 | 0.29 |
| Sample | Decomposition Temperature (°C) | CR (wt %) | LOI (%) | ||
|---|---|---|---|---|---|
| T5% weight loss | Tmax | T35% weight loss | |||
| PEI | 523.4 | 538.7 | 580.9 | 50.5 | 37.7 |
| 0.1 GnP | 516.8 | 537.3 | 578.3 | 50.8 | 37.8 |
| 0.4 GnP | 518.7 | 536.9 | 580.4 | 51.7 | 38.2 |
| 0.7 GnP | 516.2 | 536.5 | 579.3 | 51.5 | 38.1 |
| 1.0 GnP | 519.0 | 537.0 | 583.0 | 51.9 | 38.3 |
| 1.5 GnP | 517.4 | 536.6 | 582.9 | 52.4 | 38.5 |
| 2.0 GnP | 513.9 | 535.3 | 581.7 | 52.3 | 38.4 |
| Sample | Relative Density | E′ at 30 °C (MPa) | E′spec (MPa·cm3/g) | Glass Transition (°C) | |
|---|---|---|---|---|---|
| Max E″ | Max tanδ | ||||
| scCO2 foams | |||||
| PEI | 0.44 | 738.6 | 1295.8 | 212.0 | 220.9 |
| 0.1 GnP | 0.48 | 702.8 | 1171.3 | 212.1 | 224.4 |
| 0.4 GnP | 0.49 | 884.3 | 1426.3 | 212.5 | 221.4 |
| 0.7 GnP | 0.42 | 630.7 | 1168.0 | 212.6 | 224.0 |
| 1.0 GnP | 0.46 | 751.6 | 1273.9 | 212.8 | 218.2 |
| 1.5 GnP | 0.40 | 642.3 | 1235.2 | 213.0 | 224.2 |
| 2.0 GnP | 0.49 | 922.1 | 1463.7 | 210.9 | 226.5 |
| WVIPS foams 1 | |||||
| 1.0 GnP NS | 0.44 | 742.6 | 1335.6 | 218.0 | 225.0 |
| 2.0 GnP NS | 0.39 | 568.1 | 1147.7 | 218.4 | 226.7 |
| 1.0 GnP S | 0.26 | 370.4 | 1110.9 | 223.1 | 229.8 |
| 2.0 GnP S | 0.26 | 385.3 | 1170.5 | 223.3 | 228.6 |
| 2.0 CNT S | 0.44 | 442.9 | 776.5 | 221.5 | 227.1 |
| Sample | GnP (wt %) | GnP (vol%) | Porosity (%) | σ (S/m) | σcorr (S/m) 1 |
|---|---|---|---|---|---|
| PEI | 0.0 | 0.00 | 55.3 | 7.18 × 10−16 | 4.60 × 10−16 (9.92 × 10−17) |
| 0.1 GnP | 0.1 | 0.03 | 52.4 | 2.70 × 10−13 | 1.88 × 10−13 (4.03 × 10−14) |
| 0.4 GnP | 0.4 | 0.11 | 51.0 | 3.17 × 10−11 | 2.27 × 10−11 (4.10 × 10−12) |
| 0.7 GnP | 0.7 | 0.17 | 57.7 | 7.16 × 10−11 | 4.99 × 10−11 (1.25 × 10−11) |
| 1.0 GnP | 1.0 | 0.27 | 53.5 | 3.76 × 10−10 | 1.86 × 10−10 (4.00 × 10−11) |
| 1.5 GnP | 1.5 | 0.35 | 59.6 | 5.12 × 10−10 | 3.45 × 10−10 (8.67 × 10−11) |
| 2.0 GnP | 2.0 | 0.57 | 51.0 | 1.12 × 10−10 | 7.70 × 10−11 (1.53 × 10−11) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbasi, H.; Antunes, M.; Velasco, J.I. Polyetherimide Foams Filled with Low Content of Graphene Nanoplatelets Prepared by scCO2 Dissolution. Polymers 2019, 11, 328. https://doi.org/10.3390/polym11020328
Abbasi H, Antunes M, Velasco JI. Polyetherimide Foams Filled with Low Content of Graphene Nanoplatelets Prepared by scCO2 Dissolution. Polymers. 2019; 11(2):328. https://doi.org/10.3390/polym11020328
Chicago/Turabian StyleAbbasi, Hooman, Marcelo Antunes, and José Ignacio Velasco. 2019. "Polyetherimide Foams Filled with Low Content of Graphene Nanoplatelets Prepared by scCO2 Dissolution" Polymers 11, no. 2: 328. https://doi.org/10.3390/polym11020328
APA StyleAbbasi, H., Antunes, M., & Velasco, J. I. (2019). Polyetherimide Foams Filled with Low Content of Graphene Nanoplatelets Prepared by scCO2 Dissolution. Polymers, 11(2), 328. https://doi.org/10.3390/polym11020328