Improving the Supercritical CO2 Foaming of Polypropylene by the Addition of Fluoroelastomer as a Nucleation Agent
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Foaming Process
2.4. Sample Characterization
2.5. Morphological Observation of the Foams
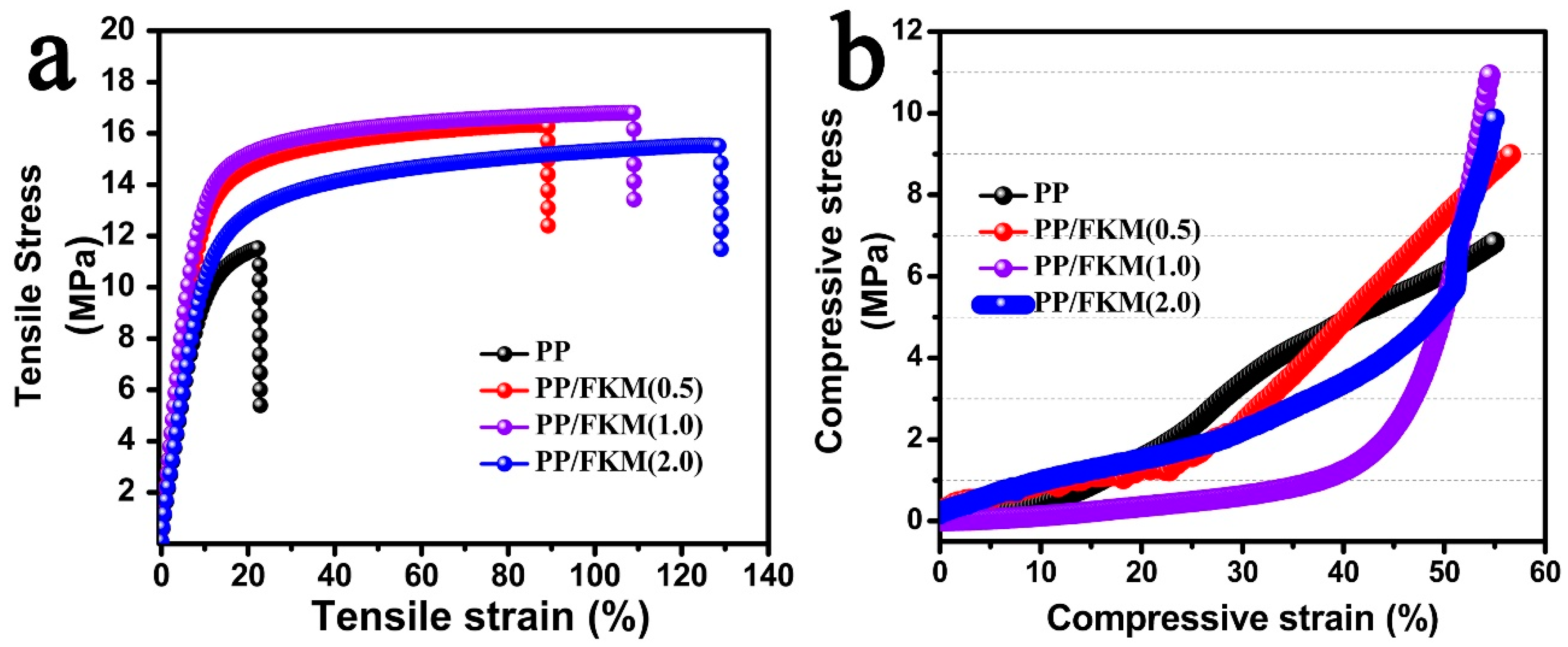
2.6. Mechanical Properties
3. Results and Discussion
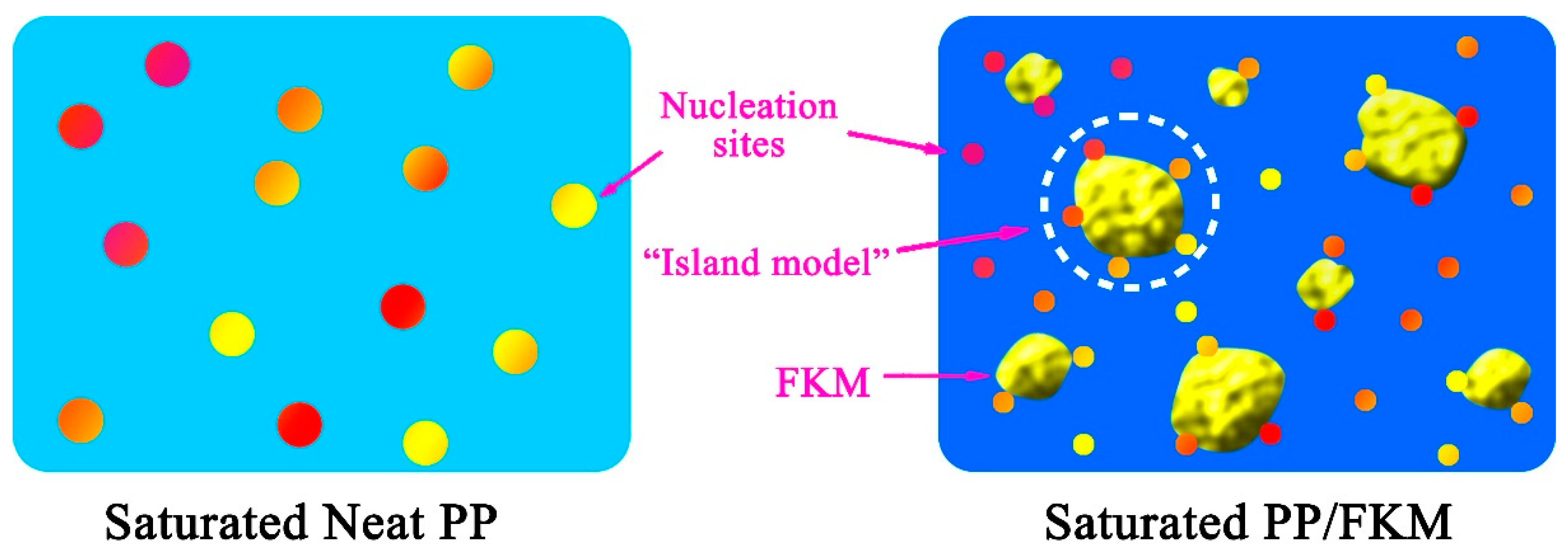
3.1. Microscopic Structure of PP/FKM Blends
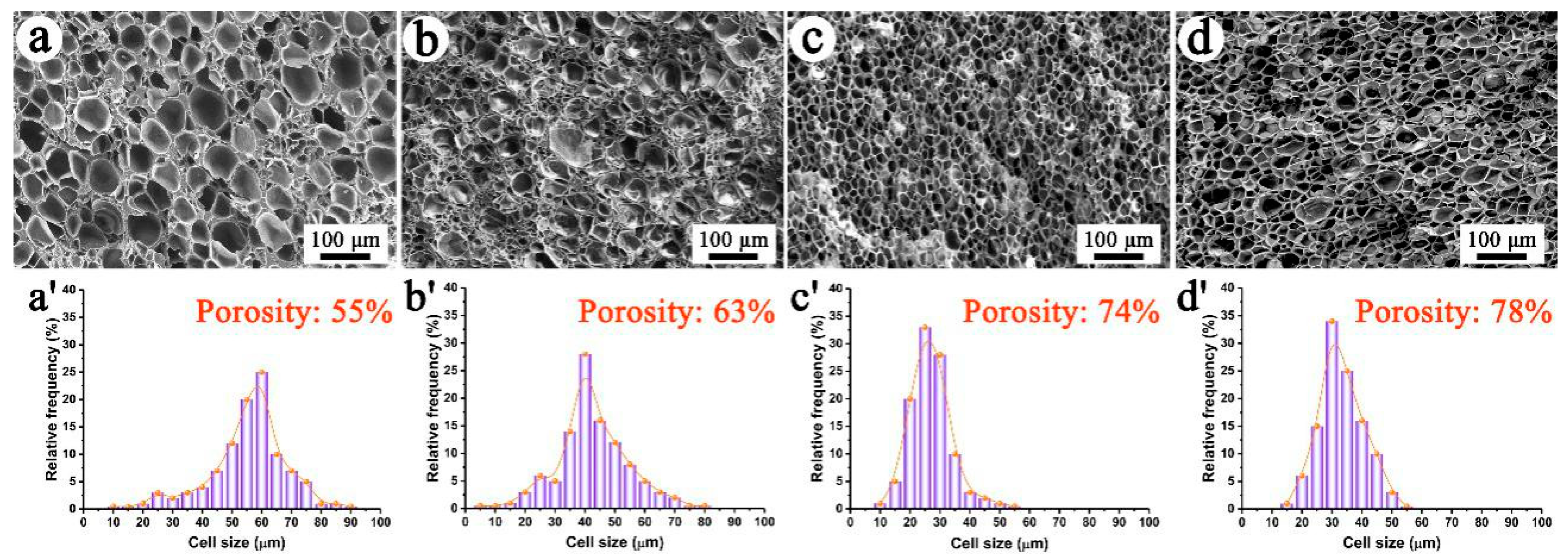
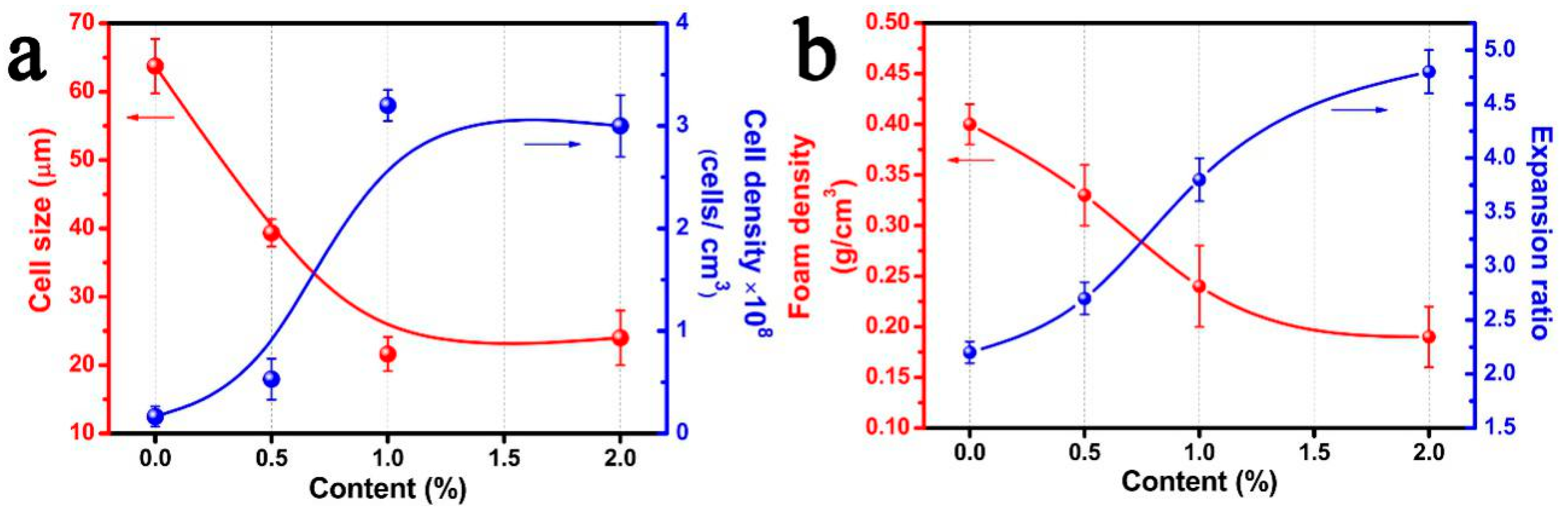
3.2. Morphologies and Properties of PP/FKM Foams
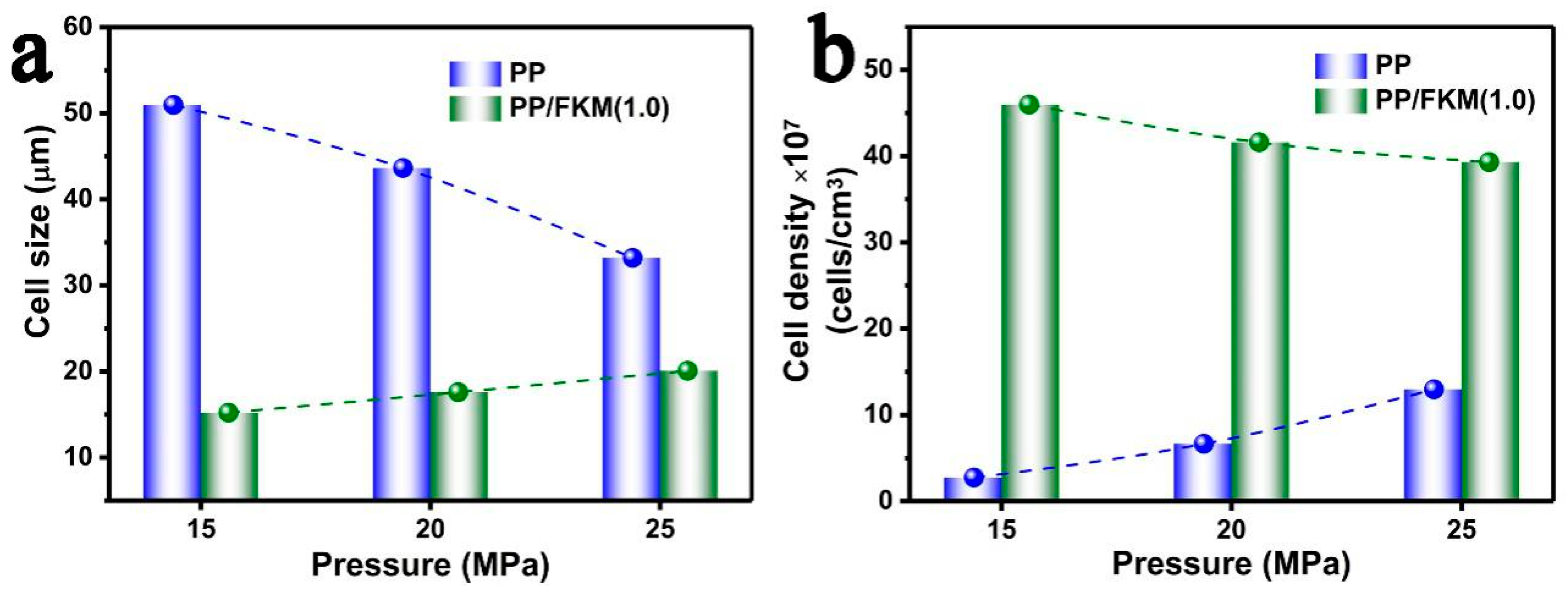
3.3. Effects of Foaming Pressure on the Foaming Behavior of PP/FKM(1.0)
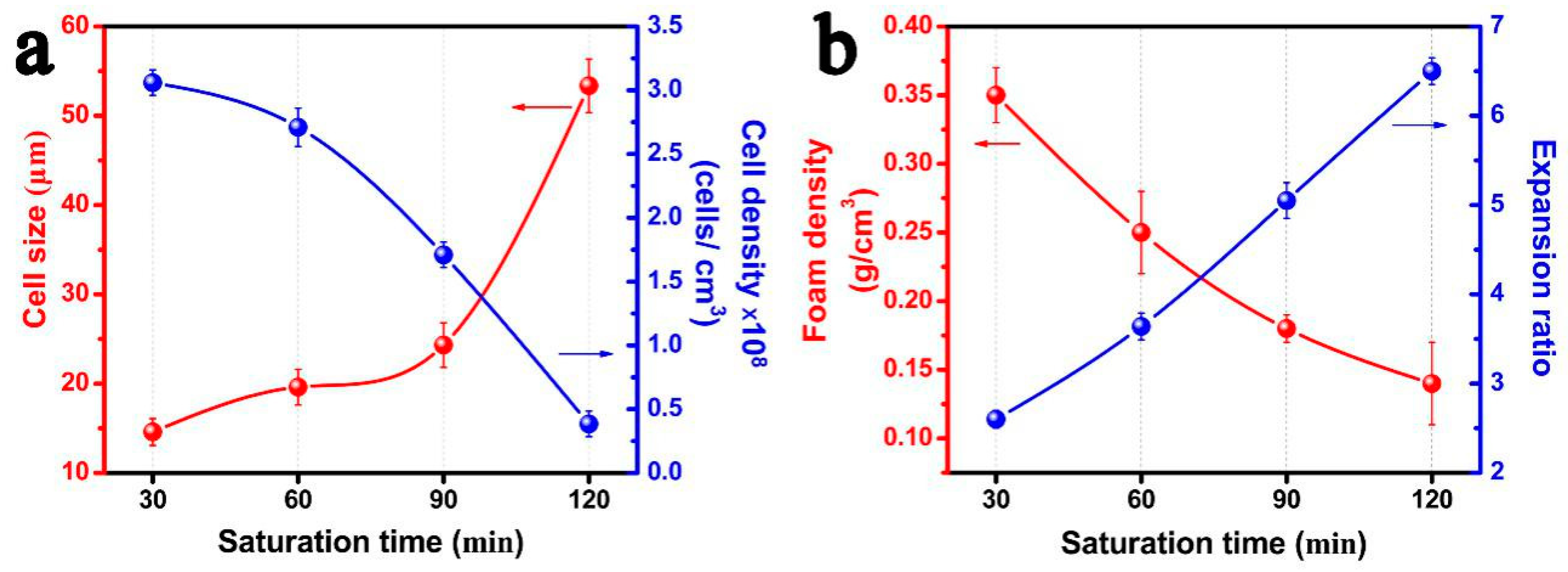
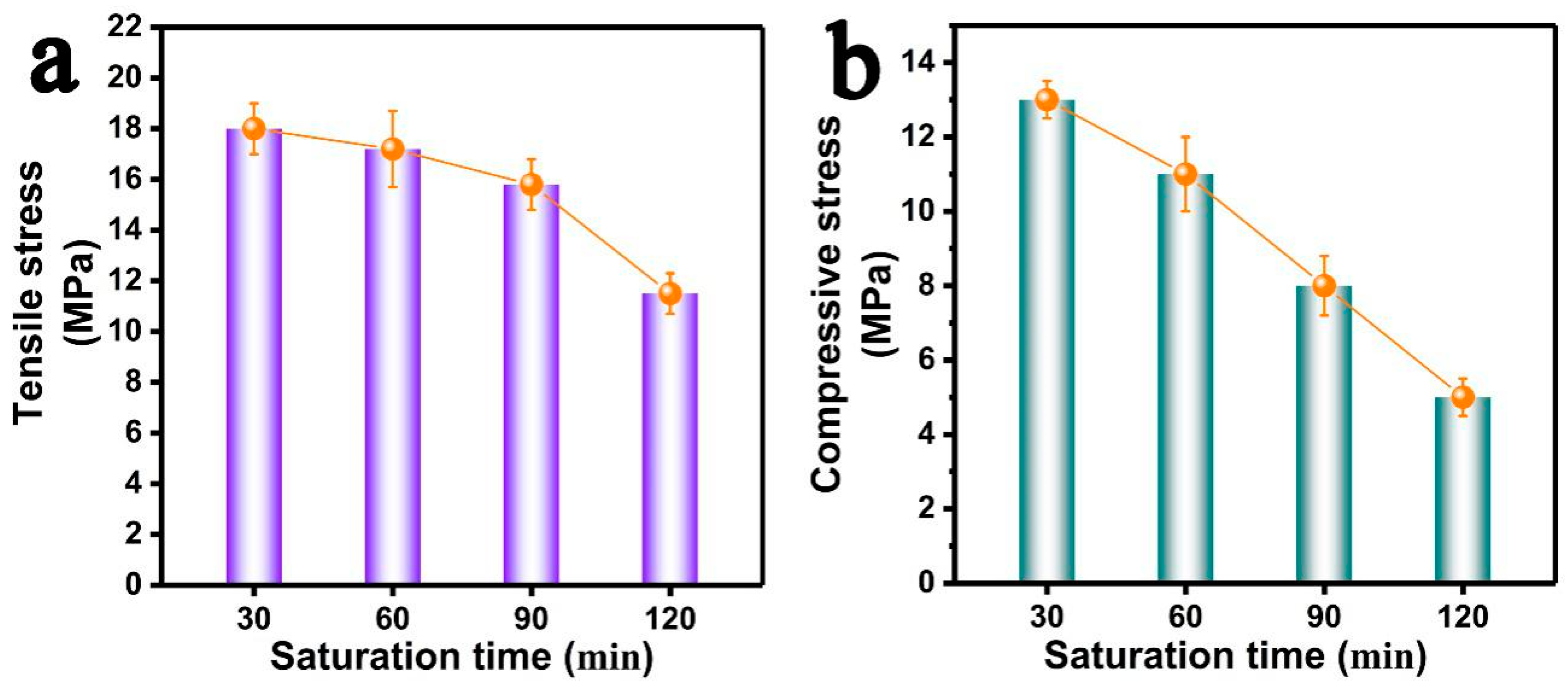
3.4. Effects of Saturation Time on the Foaming Behavior of PP/FKM(1.0)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ameli, A.; Wang, S.; Kazemi, Y.; Park, C.B.; Poetschke, P. A facile method to increase the charge storage capability of polymer nanocomposites. Nano Energy 2015, 15, 54–65. [Google Scholar] [CrossRef]
- Al Tawil, E.; Monnier, A.; Nguyen, Q.T.; Deschrevel, B. Microarchitecture of poly(lactic acid) membranes with an interconnected network of macropores and micropores influences cell behavior. Eur. Polym. J. 2018, 105, 370–388. [Google Scholar] [CrossRef]
- Velasco, J.I.; Antunes, M.; Ayyad, O.; López-Cuesta, J.M.; Gaudon, P.; Saiz-Arroyo, C.; Rodríguez-Pérez, M.A.; Saja, J.A.D. Foaming behaviour and cellular structure of LDPE/hectorite nanocomposites. Polymer 2007, 48, 2098–2108. [Google Scholar] [CrossRef]
- Antunes, M.; Velasco, J.I. Multifunctional polymer foams with carbon nanoparticles. Prog. Polym. Sci. 2014, 39, 486–509. [Google Scholar] [CrossRef]
- Nofar, M.; Ameli, A.; Park, C.B. Development of polylactide bead foams with double crystal melting peaks. Polymer 2015, 69, 83–94. [Google Scholar] [CrossRef]
- Hochleitner, G.; Huemmer, J.F.; Luxenhofer, R.; Groll, J. High definition fibrous poly(2-ethyl-2-oxazoline) scaffolds through melt electrospinning writing. Polymer 2014, 55, 5017–5023. [Google Scholar] [CrossRef]
- Sun, J.; Xu, J.; He, Z.; Ren, H.; Wang, Y.; Zhang, L.; Bao, J.-B. Role of nano silica in supercritical CO2 foaming of thermoplastic poly(vinyl alcohol) and its effect on cell structure and mechanical properties. Eur. Polym. J. 2018, 105, 491–499. [Google Scholar] [CrossRef]
- Yang, C.; Xing, Z.; Wang, M.; Zhao, Q.; Wang, M.; Zhang, M.; Wu, G.-Z. Better scCO2 foaming of polypropylene via earlier crystallization with the addition of composite nucleating agent. Ind. Eng. Chem. Res. 2018, 57, 15916–15923. [Google Scholar] [CrossRef]
- Realinho, V.; Haurie, L.; Antunes, M.; Velasco, J.I. Thermal stability and fire behaviour of flame retardant high density rigid foams based on hydromagnesite-filled polypropylene composites. Compos. Part B Eng. 2014, 58, 553–558. [Google Scholar] [CrossRef]
- Bao, J.-B.; Junior, A.N.; Weng, G.-S.; Wang, J.; Fang, Y.-W.; Hu, G.-H. Tensile and impact properties of microcellular isotactic polypropylene (PP) foams obtained by supercritical carbon dioxide. J. Supercrit. Fluids 2016, 111, 63–73. [Google Scholar] [CrossRef]
- Yang, C.-G.; Wang, M.-H.; Zhang, M.-X.; Li, X.-H.; Wang, H.-L.; Xing, Z.; Ye, L.-F.; Wu, G.-Z. Supercritical CO2 Foaming of Radiation Cross-Linked Isotactic Polypropylene in the Presence of TAIC. Molecules 2016, 21, 1660. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Xing, Z.; Wang, M.; Zhao, Q.; Wu, G. Merits of the Addition of PTFE Micropowder in Supercritical Carbon Dioxide Foaming of Polypropylene: Ultrahigh Cell Density, High Tensile Strength, and Good Sound Insulation. Ind. Eng. Chem. Res. 2018, 57, 1498–1505. [Google Scholar] [CrossRef]
- Cui, L.; Wang, P.; Zhang, Y.; Zhou, X.; Xu, L.; Zhang, L.; Zhang, L.; Liu, L.; Guo, X. Glass fiber reinforced and beta-nucleating agents regulated polypropylene: A complementary approach and a case study. J. Appl. Polym. Sci. 2018, 135, 45768. [Google Scholar] [CrossRef]
- Dlouha, J.; Suryanegara, L.; Yano, H. The role of cellulose nanofibres in supercritical foaming of polylactic acid and their effect on the foam morphology. Soft Matter 2012, 8, 8704–8713. [Google Scholar] [CrossRef]
- Chen, L.; Rende, D.; Schadler, L.S.; Ozisik, R. Polymer nanocomposite foams. J. Mater. Chem. A 2013, 1, 3837–3850. [Google Scholar] [CrossRef]
- Yang, C.; Xing, Z.; Zhang, M.; Zhao, Q.; Wang, M.; Wu, G. Supercritical CO2 foaming of radiation crosslinked polypropylene/high-density polyethylene blend: Cell structure and tensile property. Radiat. Phys. Chem. 2017, 141, 276–283. [Google Scholar] [CrossRef]
- Zhao, J.; Zhao, Q.; Wang, C.; Guo, B.; Park, C.B.; Wang, G. High thermal insulation and compressive strength polypropylene foams fabricated by high-pressure foam injection molding and mold opening of nano-fibrillar composites. Mater. Des. 2017, 131, 1–11. [Google Scholar] [CrossRef]
- Yang, C.G.; Wang, M.H.; Xing, Z.; Zhao, Q.; Wang, M.L.; Wu, G.Z. A new promising nucleating agent for polymer foaming: Effects of hollow molecular-sieve particles on polypropylene supercritical CO2 microcellular foaming. RSC Adv. 2018, 8, 20061–20067. [Google Scholar] [CrossRef]
- Yang, C.G.; Xing, Z.; Zhao, Q.; Wang, M.H.; Wu, G.Z. A strategy for the preparation of closed-cell and crosslinked polypropylene foam by supercritical CO2 foaming. J. Appl. Polym. Sci. 2018, 135, 45809. [Google Scholar] [CrossRef]
- Zhai, W.; Wang, H.; Yu, J.; Dong, J.; He, J. Cell coalescence suppressed by crosslinking structure in polypropylene microcellular foaming. Polym. Eng. Sci. 2010, 48, 1312–1321. [Google Scholar] [CrossRef]
- Yang, C.; Zhe, X.; Zhang, M.; Wang, M.; Wu, G. Radiation effects on the foaming of atactic polypropylene with supercritical carbon dioxide. Radiat. Phys. Chem. 2017, 131, 35–40. [Google Scholar] [CrossRef]
- Wang, G.; Zhao, G.; Zhang, L.; Mu, Y.; Park, C.B. Lightweight and tough nanocellular PP/PTFE nanocomposite foams with defect-free surfaces obtained using in situ nanofibrillation and nanocellular injection molding. Chem. Eng. J. 2018, 350, 1–11. [Google Scholar] [CrossRef]
- Zheng, W.G.; Lee, Y.H.; Park, C.B. Use of nanoparticles for improving the foaming behaviors of linear PP. J. Appl. Polym. Sci. 2010, 117, 2972–2979. [Google Scholar] [CrossRef]
- Werner, P.; Verdejo, R.; Wöllecke, F.; Altstädt, V.; Sandler, J.K.W.; Shaffer, M.S.P. Carbon Nanofibers Allow Foaming of Semicrystalline Poly(ether ether ketone). Adv. Mater. 2010, 17, 2864–2869. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, H.; Wang, X.; Mi, J. Evaluation of Nanoparticle Effect on Bubble Nucleation in Polymer Foaming. J. Phys. Chem. C 2016, 120, 26841–26851. [Google Scholar] [CrossRef]
- Kakroodi, A.R.; Kazemi, Y.; Nofar, M.; Park, C.B. Tailoring poly(lactic acid) for packaging applications via the production of fully bio-based in situ microfibrillar composite films. Chem. Eng. J. 2017, 308, 772–782. [Google Scholar] [CrossRef]
- Ventura, H.; Sorrentino, L.; Laguna-Gutierrez, E.; Rodriguez-Perez, M.; Ardanuy, M. Gas Dissolution Foaming as a Novel Approach for the Production of Lightweight Biocomposites of PHB/Natural Fibre Fabrics. Polymers 2018, 10, 249. [Google Scholar] [CrossRef]
- Antunes, M.; Mudarra, M.; Velasco, J.I. Broad-band electrical conductivity of carbon nanofibre-reinforced polypropylene foams. Carbon 2011, 49, 708–717. [Google Scholar] [CrossRef]
- Ji, G.; Zhai, W.; Lin, D.; Ren, Q.; Zheng, W.; Jung, D.W. Microcellular Foaming of Poly(lactic acid)/Silica Nanocomposites in Compressed CO2: Critical Influence of Crystallite Size on Cell Morphology and Foam Expansion. Ind. Eng. Chem. Res. 2013, 52, 6390–6398. [Google Scholar] [CrossRef]
- Ren, Q.; Wang, J.; Zhai, W.; Su, S. Solid State Foaming of Poly(lactic acid) Blown with Compressed CO2: Influences of Long Chain Branching and Induced Crystallization on Foam Expansion and Cell Morphology. Ind. Eng. Chem. Res. 2013, 52, 13411–13421. [Google Scholar] [CrossRef]
- Wong, A.; Guo, Y.; Park, C.B. Fundamental mechanisms of cell nucleation in polypropylene foaming with supercritical carbon dioxide—Effects of extensional stresses and crystals. J. Supercrit. Fluids 2013, 79, 142–151. [Google Scholar] [CrossRef]
- Martín-de León, J.; Bernardo, V.; Rodríguez-Pérez, M. Low Density Nanocellular Polymers Based on PMMA Produced by Gas Dissolution Foaming: Fabrication and Cellular Structure Characterization. Polymers 2016, 8, 265. [Google Scholar] [CrossRef]
- Bonavoglia, B.; Giuseppe Storti, A.; Morbidelli, M. Modeling of the Sorption and Swelling Behavior of Semicrystalline Polymers in Supercritical CO2. Ind. Eng. Chem. Res. 2006, 45, 4739–4750. [Google Scholar] [CrossRef]
- Solms, N.v.; Zecchin, N.; Rubin, A.; Andersen, S.I.; Stenby, E.H. Direct measurement of gas solubility and diffusivity in poly(vinylidene fluoride) with a high-pressure microbalance. Eur. Polym. J. 2005, 41, 341–348. [Google Scholar] [CrossRef]
- Zirkel, L.; Jakob, M.; Munstedt, H. Foaming of thin films of a fluorinated ethylene propylene copolymer using supercritical carbon dioxide. J. Supercrit. Fluids 2009, 49, 103–110. [Google Scholar] [CrossRef]
- Deverman, G.S.; Yonker, C.R.; Grate, J.W. Thin fluoropolymer films and nanoparticle coatings from the rapid expansion of supercritical carbon dioxide solutions with electrostatic collection. Polymer 2003, 44, 3627–3632. [Google Scholar] [CrossRef]
- Leung, S.N.; Park, C.B.; Xu, D.; Hongbo Li, A.; Fenton, R.G. Computer Simulation of Bubble-Growth Phenomena in Foaming. Ind. Eng. Chem. Res. 2006, 45, 7823–7831. [Google Scholar] [CrossRef]
- Wang, L.; Ishihara, S.; Hikima, Y.; Ohshima, M.; Sekiguchi, T.; Sato, A.; Yano, H. Unprecedented Development of Ultrahigh Expansion Injection-Molded Polypropylene Foams by Introducing Hydrophobic-Modified Cellulose Nanofibers. ACS Appl. Mater. Interfaces 2017, 9, 9250–9254. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Hikima, Y.; Ohshima, M.; Yusa, A.; Yamamoto, S.; Goto, H. Unusual Fabrication of Lightweight Injection-Molded Polypropylene Foams by Using Air as the Novel Foaming Agent. Ind. Eng. Chem. Res. 2018, 57, 3800–3804. [Google Scholar] [CrossRef]



| Samples | PP | PP/FKM(0.5) | PP/FKM(1.0) | PP/FKM(2.0) |
|---|---|---|---|---|
| Tm/°C | 165.4 | 165.9 | 166.4 | 167.3 |
| XC/% | 38.2 | 38.8 | 39.7 | 40.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.; Zhao, Q.; Xing, Z.; Zhang, W.; Zhang, M.; Tan, H.; Wang, J.; Wu, G. Improving the Supercritical CO2 Foaming of Polypropylene by the Addition of Fluoroelastomer as a Nucleation Agent. Polymers 2019, 11, 226. https://doi.org/10.3390/polym11020226
Yang C, Zhao Q, Xing Z, Zhang W, Zhang M, Tan H, Wang J, Wu G. Improving the Supercritical CO2 Foaming of Polypropylene by the Addition of Fluoroelastomer as a Nucleation Agent. Polymers. 2019; 11(2):226. https://doi.org/10.3390/polym11020226
Chicago/Turabian StyleYang, Chenguang, Quan Zhao, Zhe Xing, Wenli Zhang, Maojiang Zhang, Hairong Tan, Jixiang Wang, and Guozhong Wu. 2019. "Improving the Supercritical CO2 Foaming of Polypropylene by the Addition of Fluoroelastomer as a Nucleation Agent" Polymers 11, no. 2: 226. https://doi.org/10.3390/polym11020226
APA StyleYang, C., Zhao, Q., Xing, Z., Zhang, W., Zhang, M., Tan, H., Wang, J., & Wu, G. (2019). Improving the Supercritical CO2 Foaming of Polypropylene by the Addition of Fluoroelastomer as a Nucleation Agent. Polymers, 11(2), 226. https://doi.org/10.3390/polym11020226






