Recent Advances in Engineered Stem Cell-Derived Cell Sheets for Tissue Regeneration
Abstract
1. Introduction
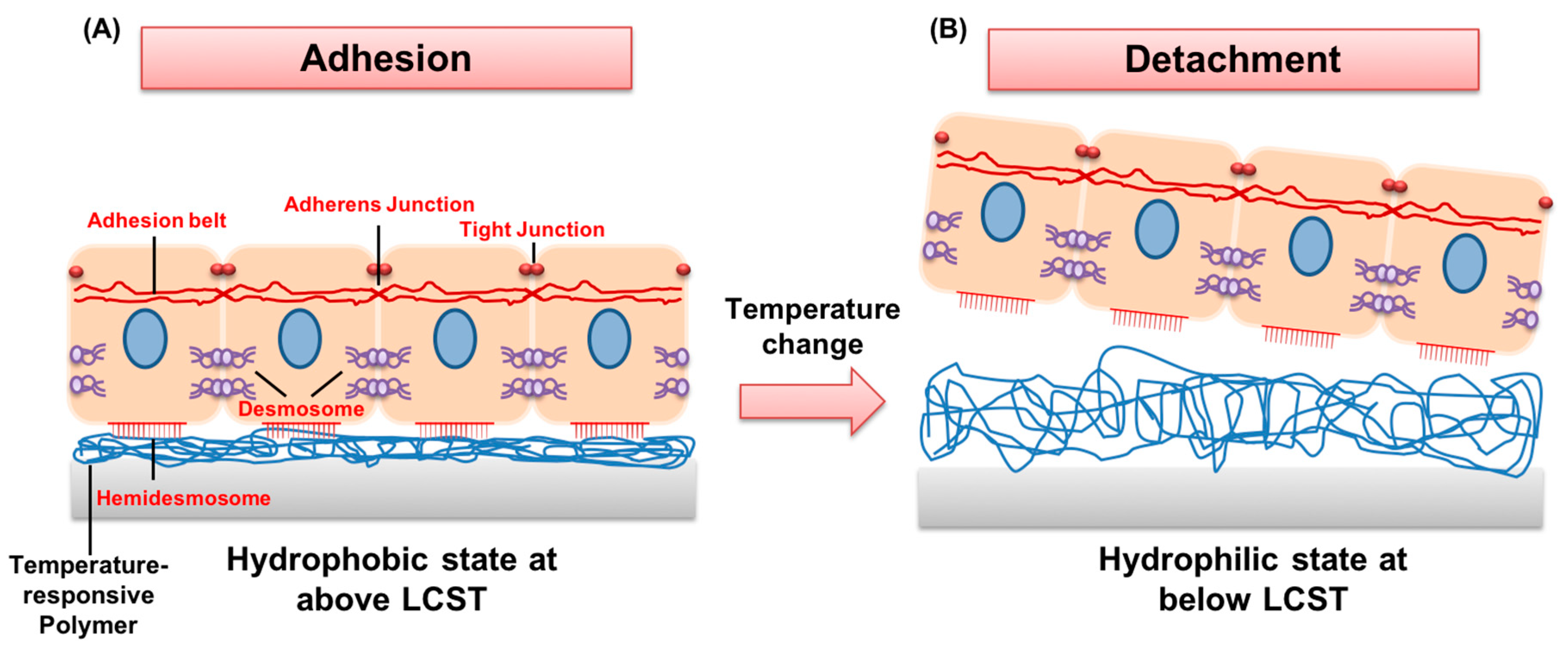
2. The Principles of Temperature-Responsive Behaviors of Polymers in Terms of Their Critical Solution Temperature
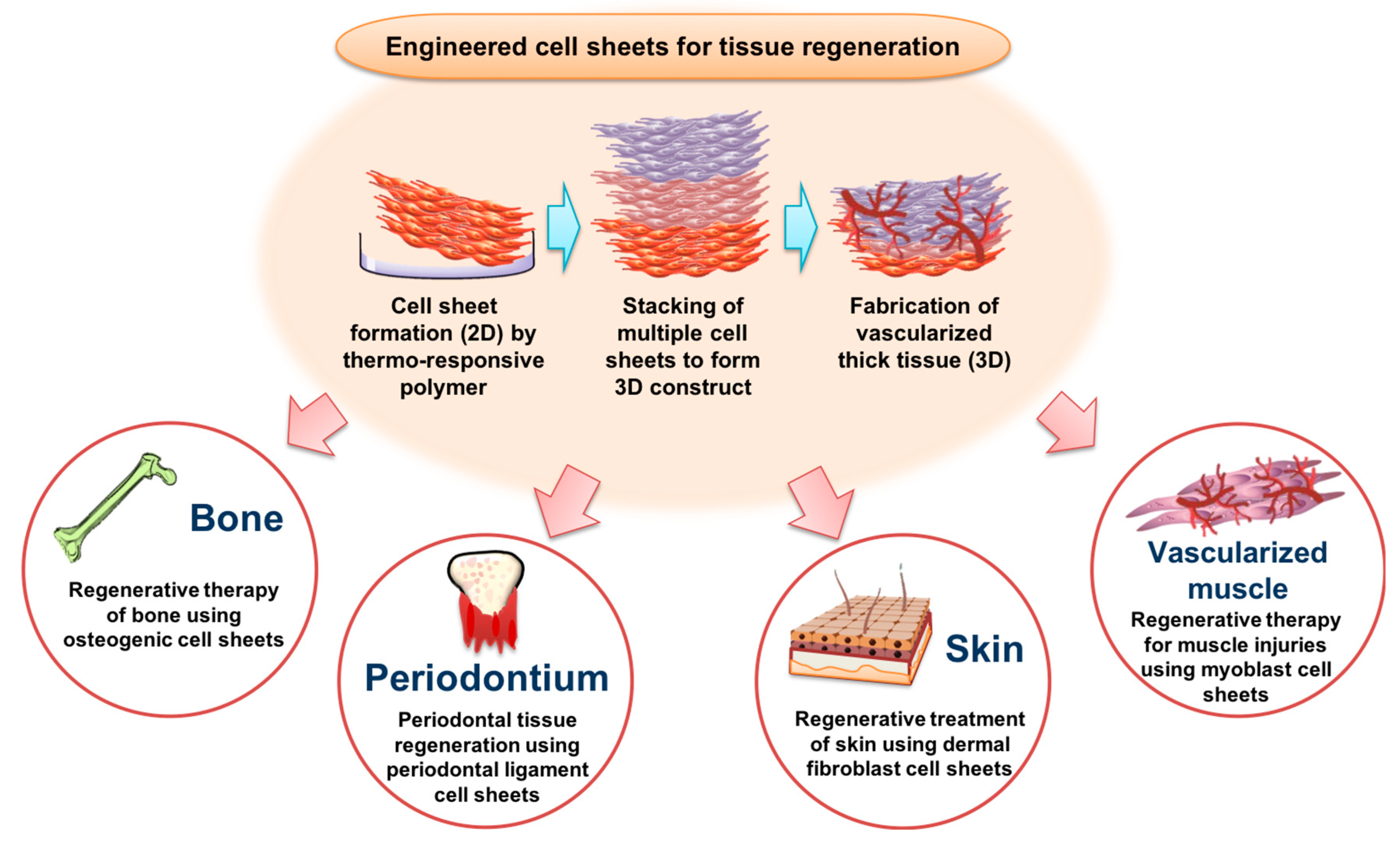
3. Potential Biomedical Applications of Temperature-Responsive Polymers in Regenerative Medicine and Tissues Engineering
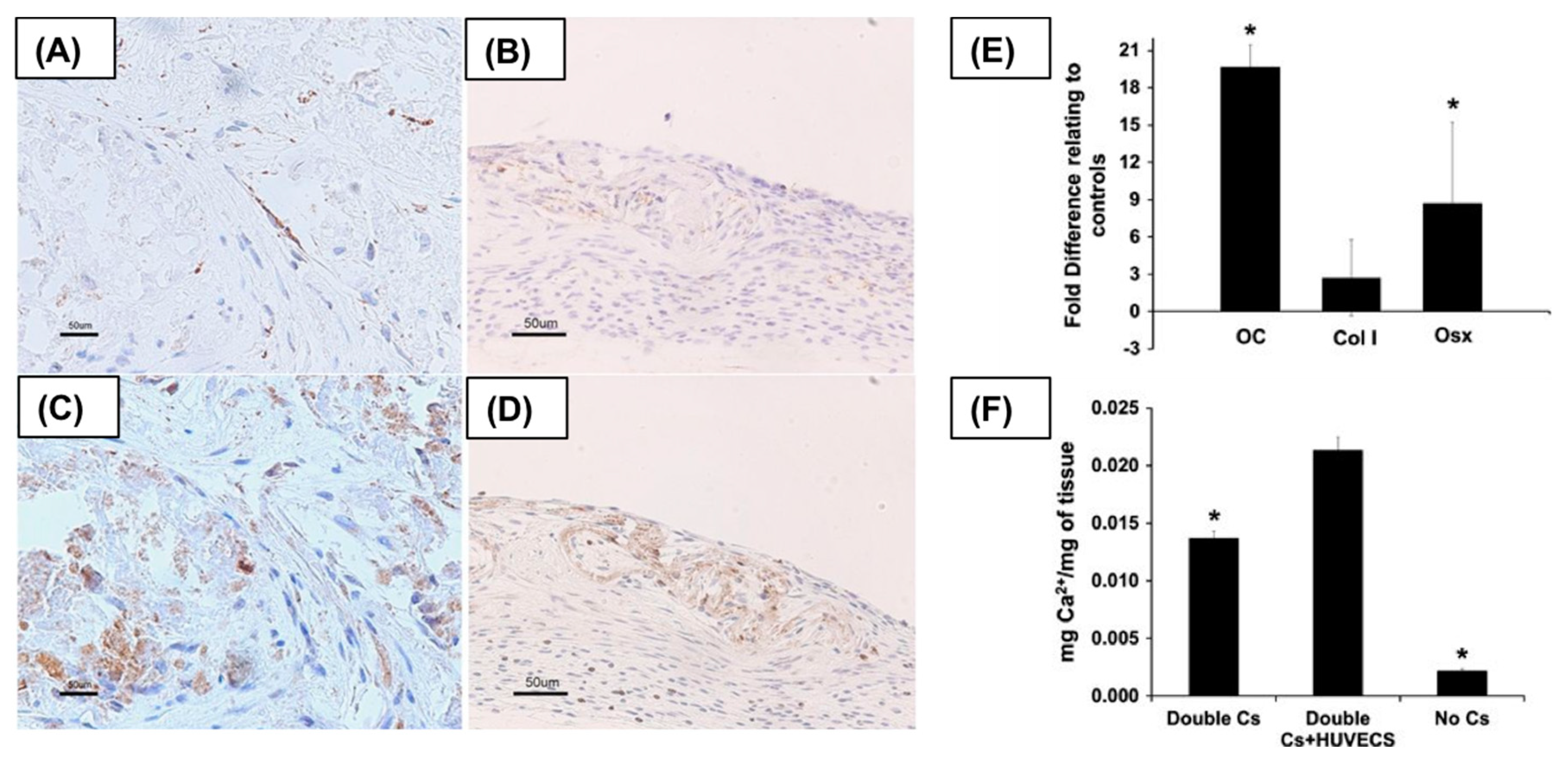
3.1. Engineered Bone Tissue Regeneration by CSE
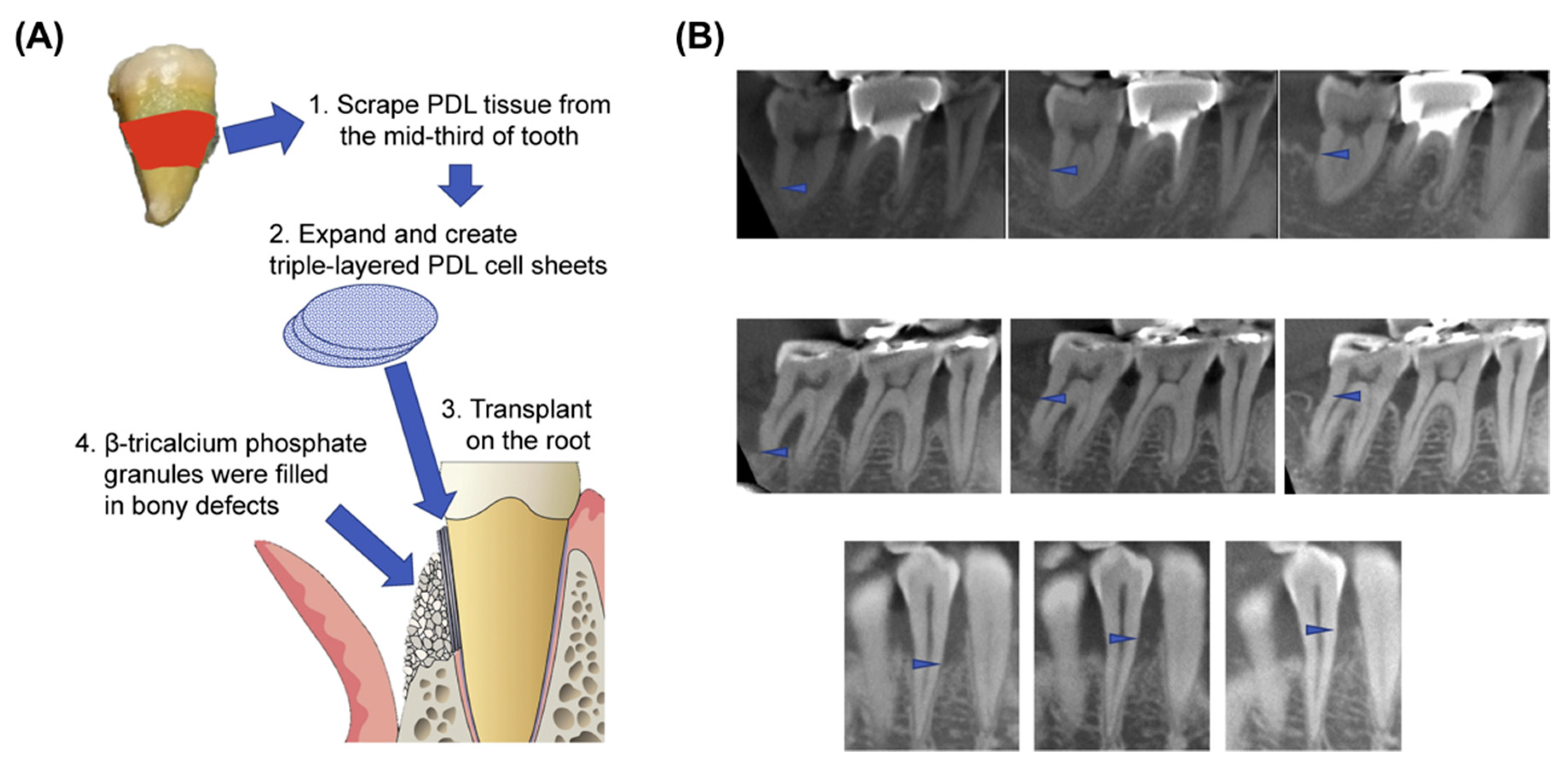
3.2. Engineered Periodontal Tissue Regeneration by CSE
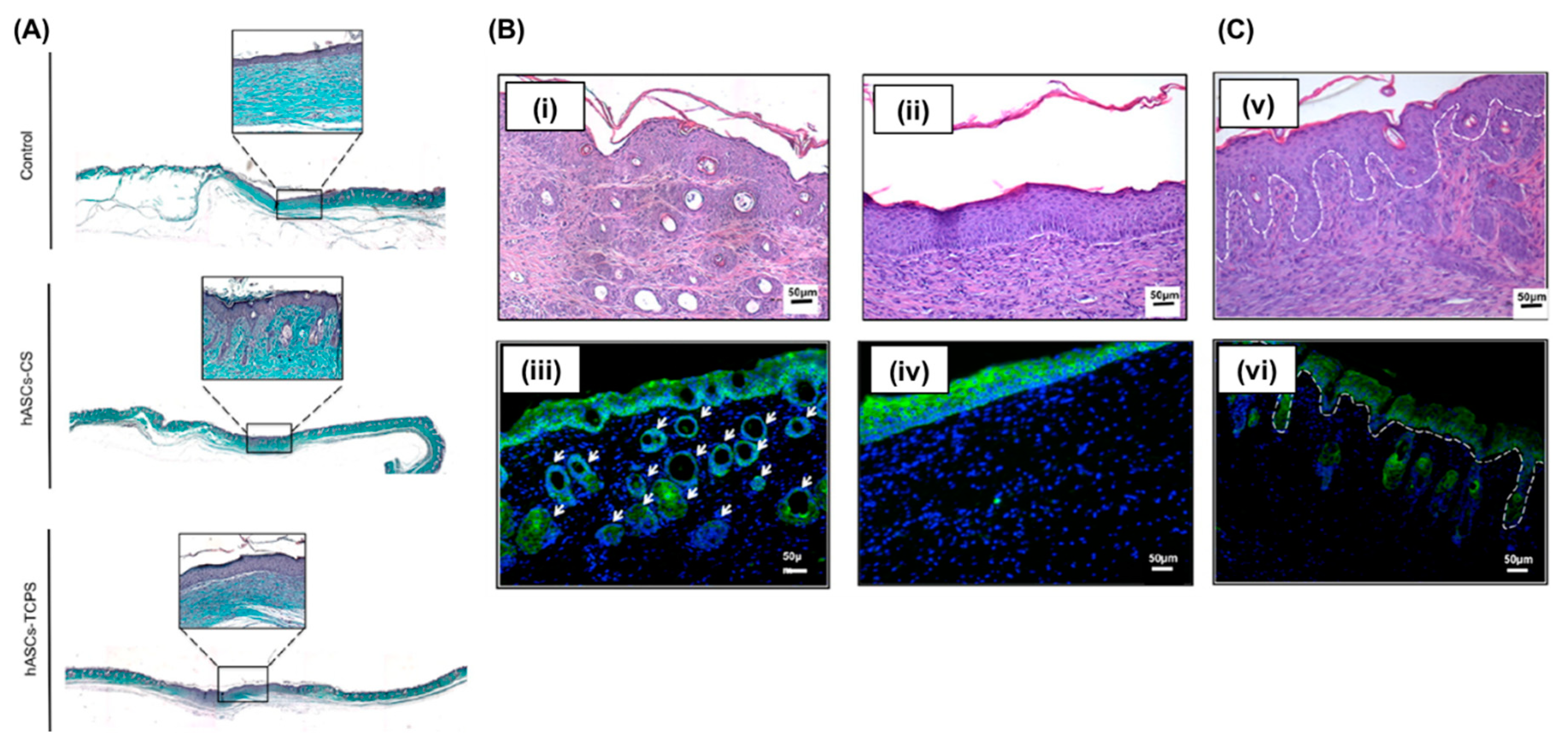
3.3. Engineered Skin Regeneration by CSE
3.4. Engineered Muscle Regeneration by Pre-Vascularized CSE
4. Recent Clinical Studies of CSE-Based Approaches
5. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| hASC | Human adipose-derived stem cells |
| β-TCP | β-tricalciumphosphate |
| CSE | Cell sheet engineering |
| LCST | Lower critical solution temperature |
| LV | Left ventricular |
| MI | Myocardial infarction |
| PDL | Periodontal ligament |
| PSC | Pluripotent stem cells |
| SVF | Stromal vascular fraction |
| TCPS | Tissue culture polystyrene |
| UCST | Upper critical solution temperature |
| VEGF | Vascular endothelial growth factor |
| AVL | Arteriovenous loop |
| VPT | Volume phase transition |
| hESCs | Human embryonic stem cells |
| hMSCs | Human mesenchymal stem cells |
| hBM-MSCs | Human bone marrow-derived mesenchymal stem cells |
| hAFSCs | Human amniotic fluid stem cells |
| PNIPAAm | Poly(N-isopropylacrylamide) |
| MEO2MA | 2-(2-methoxyethoxy) ethyl methacrylate |
| OEGMA | Oligo(ethylene glycol) methacrylate |
| hUCBMSCs | Human umbilical cord blood mesenchymal stem cells |
| CTX | Cardiotoxin |
| ECM | Extracellular matrix |
| PVME | Poly(vinyl methyl ether) |
| PVMEMA | Poly(vinyl methyl ether and maleic acid) |
| PGA | Poly(glycolic acid) |
| TA | Tibialis anterior |
| ASCs | Adipose-derived stem cells |
| PDMS | Polydimethylsiloxane |
| MC | Methylcellulose |
| PDEA | Poly(N,N-diethylacrylamide) |
| PEG | Poly(ethylene glycol) |
| PVA | Poly(vinyl alcohol) |
| PAA | Poly(acrylic acid) |
| PHEMA | Poly(2-hydroxyethyl methacrylate) |
| PLA | Poly(lactic acid) |
| PCL | Poly(caprolactone) |
| PNVCL | Poly(N-vinylcaprolactam) |
| PMAA | Poly(methacrylic acid) |
| PEO | Poly(ethylene oxide) |
| n-BMA | n-butyl methacrylate |
| PPO | Poly(propylene oxide) |
| PEA | Poly(ethyl acrylate) |
| p(NIPAAm-co-MMA) | Poly(N-isopropyl acryl amide-co-methacrylic acid) |
| SE | Secretome of ASCs |
| VC | Vitamin C |
| VEGF | Vascular endothelial growth factor |
| HGF | Hepatocyte growth factor |
| LV | Left ventricular |
| PDLSCs | PDL-derived stem cells |
References
- Ratcliffe, E.; Glen, K.E.; Naing, M.W.; Williams, D.J. Current status and perspectives on stem cell-based therapies undergoing clinical trials for regenerative medicine: Case studies. Br. Med. Bull. 2013, 108, 73–94. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.; Phadke, A.; Varghese, S. Engineered microenvironments for self-renewal and musculoskeletal differentiation of stem cells. Regen. Med. 2011, 6, 505–524. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.W.; Hwang, Y.; Brafman, D.; Hagan, T.; Phung, C.; Varghese, S. Engineering cell-material interfaces for long-term expansion of human pluripotent stem cells. Biomaterials 2013, 34, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Ben-David, U.; Benvenisty, N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat. Rev. Cancer 2011, 11, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Hu, X.; Wang, J. Concise review: Optimized strategies for stem cell-based therapy in myocardial repair: Clinical translatability and potential limitation. Stem Cells 2018, 36, 482–500. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; Baum, E.; Gibbons, D.; Tekamp-Olson, P. Developing a case study model for successful translation of stem cell therapies. Cell Stem Cell 2010, 6, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Hwang, N.S.; Zhang, C.; Hwang, Y.S.; Varghese, S. Mesenchymal stem cell differentiation and roles in regenerative medicine. Wiley Interdiscip. Rev. Syst. Biol. Med. 2009, 1, 97–106. [Google Scholar] [CrossRef]
- Jung, N.; Park, S.; Choi, Y.; Park, J.W.; Hong, Y.B.; Park, H.H.; Yu, Y.; Kwak, G.; Kim, H.S.; Ryu, K.H.; et al. Tonsil-derived mesenchymal stem cells differentiate into a schwann cell phenotype and promote peripheral nerve regeneration. Int. J. Mol. Sci. 2016, 17, 1867. [Google Scholar] [CrossRef]
- Llufriu, S.; Sepulveda, M.; Blanco, Y.; Marin, P.; Moreno, B.; Berenguer, J.; Gabilondo, I.; Martinez-Heras, E.; Sola-Valls, N.; Arnaiz, J.A.; et al. Randomized placebo-controlled phase ii trial of autologous mesenchymal stem cells in multiple sclerosis. PLoS ONE 2014, 9, e113936. [Google Scholar] [CrossRef]
- Vacanti, C.A.; Vacanti, J.P. The science of tissue engineering. Orthop. Clin. N. Am. 2000, 31, 351–356. [Google Scholar] [CrossRef]
- Lodi, D.; Iannitti, T.; Palmieri, B. Stem cells in clinical practice: Applications and warnings. J. Exp. Clin. Cancer Res. 2011, 30, 9. [Google Scholar] [CrossRef] [PubMed]
- Borlongan, C.V.; Glover, L.E.; Tajiri, N.; Kaneko, Y.; Freeman, T.B. The great migration of bone marrow-derived stem cells toward the ischemic brain: Therapeutic implications for stroke and other neurological disorders. Prog. Neurobiol. 2011, 95, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Yamout, B.; Hourani, R.; Salti, H.; Barada, W.; El-Hajj, T.; Al-Kutoubi, A.; Herlopian, A.; Baz, E.K.; Mahfouz, R.; Khalil-Hamdan, R.; et al. Bone marrow mesenchymal stem cell transplantation in patients with multiple sclerosis: A pilot study. J. Neuroimmunol. 2010, 227, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.P.; Sun, Z.; Miyagi, Y.; McDonald Kinkaid, H.; Zhang, L.; Weisel, R.D.; Li, R.K. Differentiation of allogeneic mesenchymal stem cells induces immunogenicity and limits their long-term benefits for myocardial repair. Circulation 2010, 122, 2419–2429. [Google Scholar] [CrossRef] [PubMed]
- Ayala, R.; Zhang, C.; Yang, D.; Hwang, Y.; Aung, A.; Shroff, S.S.; Arce, F.T.; Lal, R.; Arya, G.; Varghese, S. Engineering the cell-material interface for controlling stem cell adhesion, migration, and differentiation. Biomaterials 2011, 32, 3700–3711. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.H.; Vincent, L.G.; Fuhrmann, A.; Choi, Y.S.; Hribar, K.C.; Taylor-Weiner, H.; Chen, S.; Engler, A.J. Interplay of matrix stiffness and protein tethering in stem cell differentiation. Nat. Mater. 2014, 13, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Place, E.S.; George, J.H.; Williams, C.K.; Stevens, M.M. Synthetic polymer scaffolds for tissue engineering. Chem. Soc. Rev. 2009, 38, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.S. Hydrogels for biomedical applications. Ann. N. Y. Acad. Sci. 2001, 944, 62–73. [Google Scholar] [CrossRef]
- Tibbitt, M.W.; Anseth, K.S. Hydrogels as extracellular matrix mimics for 3d cell culture. Biotechnol. Bioeng. 2009, 103, 655–663. [Google Scholar] [CrossRef]
- Hwang, N.S.; Varghese, S.; Zhang, Z.; Elisseeff, J. Chondrogenic differentiation of human embryonic stem cell-derived cells in arginine-glycine-aspartate-modified hydrogels. Tissue Eng. 2006, 12, 2695–2706. [Google Scholar] [CrossRef]
- Kim, H.D.; Heo, J.; Hwang, Y.; Kwak, S.Y.; Park, O.K.; Kim, H.; Varghese, S.; Hwang, N.S. Extracellular-matrix-based and arg-gly-asp-modified photopolymerizing hydrogels for cartilage tissue engineering. Tissue Eng. Part A 2015, 21, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Yamada, N.; Okano, T.; Sakai, H.; Karikusa, F.; Sawasaki, Y.; Sakurai, Y. Thermo-responsive polymeric surfaces; control of attachment and detachment of cultured cells. Die Makromol. Chem. Rapid Commun. 1990, 11, 571–576. [Google Scholar] [CrossRef]
- Yang, J.; Yamato, M.; Kohno, C.; Nishimoto, A.; Sekine, H.; Fukai, F.; Okano, T. Cell sheet engineering: Recreating tissues without biodegradable scaffolds. Biomaterials 2005, 26, 6415–6422. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, Y.; Kikuchi, A.; Yamato, M.; Okano, T. Ultrathin poly(n-isopropylacrylamide) grafted layer on polystyrene surfaces for cell adhesion/detachment control. Langmuir 2004, 20, 5506–5511. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, R.M.P.; Mano, J.F.; Reis, R.L. Smart thermoresponsive coatings and surfaces for tissue engineering: Switching cell-material boundaries. Trends Biotechnol. 2007, 25, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Nash, M.E.; Carroll, W.M.; Nikoloskya, N.; Yang, R.; Connell, C.O.; Gorelov, A.V.; Dockery, P.; Liptrot, C.; Lyng, F.M.; Garcia, A.; et al. Straightforward, one-step fabrication of ultrathin thermoresponsive films from commercially available pnipam for cell culture and recovery. ACS Appl. Mater. Interfaces 2011, 3, 1980–1990. [Google Scholar] [CrossRef]
- Pan, Y.V.; Wesley, R.A.; Luginbuhl, R.; Denton, D.D.; Ratner, B.D. Plasma polymerized n-isopropylacrylamide: Synthesis and characterization of a smart thermally responsive coating. Biomacromolecules 2001, 2, 32–36. [Google Scholar] [CrossRef]
- Tang, Z.; Akiyama, Y.; Okano, T. Temperature-responsive polymer modified surface for cell sheet engineering. Polymers 2012, 4, 1478–1498. [Google Scholar] [CrossRef]
- Chen, Y.S.; Tsou, P.C.; Lo, J.M.; Tsai, H.C.; Wang, Y.Z.; Hsiue, G.H. Poly(n-isopropylacrylamide) hydrogels with interpenetrating multiwalled carbon nanotubes for cell sheet engineering. Biomaterials 2013, 34, 7328–7334. [Google Scholar] [CrossRef]
- Matsuura, K.; Utoh, R.; Nagase, K.; Okano, T. Cell sheet approach for tissue engineering and regenerative medicine. J. Control. Release 2014, 190, 228–239. [Google Scholar] [CrossRef]
- Sarkar, N. Thermal gelation properties of methyl and hydroxypropyl methylcellulose. J. Appl. Polym. Sci. 1979, 24, 1073–1087. [Google Scholar] [CrossRef]
- Chen, C.H.; Tsai, C.C.; Chen, W.; Mi, F.L.; Liang, H.F.; Chen, S.C.; Sung, H.W. Novel living cell sheet harvest system composed of thermoreversible methylcellulose hydrogels. Biomacromolecules 2006, 7, 736–743. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Sekine, H.; Yamato, M.; Okano, T. Cell sheet-based myocardial tissue engineering: New hope for damaged heart rescue. Curr. Pharm. Des. 2009, 15, 2807–2814. [Google Scholar] [CrossRef] [PubMed]
- Thirumala, S.; Gimble, J.M.; Devireddy, R.V. Methylcellulose based thermally reversible hydrogel system for tissue engineering applications. Cells 2013, 2, 460–475. [Google Scholar] [CrossRef] [PubMed]
- Yamato, M.; Utsumi, M.; Kushida, A.; Konno, C.; Kikuchi, A.; Okano, T. Thermo-responsive culture dishes allow the intact harvest of multilayered keratinocyte sheets without dispase by reducing temperature. Tissue Eng. 2001, 7, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.A.; Lucero, A.E.; Cooperstein, M.A.; Canavan, H.E. The effects of cell culture parameters on cell release kinetics from thermoresponsive surfaces. J. Appl. Biomater. Biomech. JABB 2008, 6, 81–88. [Google Scholar] [PubMed]
- Lima, L.H.; Morales, Y.; Cabral, T. Ocular biocompatibility of poly-n-isopropylacrylamide (pnipam). J. Ophthalmol. 2016, 2016, 5356371. [Google Scholar] [CrossRef]
- Fujimoto, K.L.; Ma, Z.; Nelson, D.M.; Hashizume, R.; Guan, J.; Tobita, K.; Wagner, W.R. Synthesis, characterization and therapeutic efficacy of a biodegradable, thermoresponsive hydrogel designed for application in chronic infarcted myocardium. Biomaterials 2009, 30, 4357–4368. [Google Scholar] [CrossRef]
- Wang, T.; Wu, D.-Q.; Jiang, X.-J.; Zhang, X.-Z.; Li, X.-Y.; Zhang, J.-F.; Zheng, Z.-B.; Zhuo, R.; Jiang, H.; Huang, C. Novel thermosensitive hydrogel injection inhibits post-infarct ventricle remodelling. Eur. J. Heart Fail. 2009, 11, 14–19. [Google Scholar] [CrossRef]
- Sheffield, C.; Meyers, K.; Johnson, E.; Rajachar, R. Application of composite hydrogels to control physical properties in tissue engineering and regenerative medicine. Gels 2018, 4, 51. [Google Scholar] [CrossRef]
- Zhu, J.; Marchant, R.E. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev. Med. Devices 2011, 8, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Olejniczak, M.N.; Piechocki, K.; Kozanecki, M.; Koynov, K.; Adamus, A.; Wach, R.A. The influence of selected nsaids on volume phase transition in poly(2-(2-methoxyethoxy)ethyl methacrylate) hydrogels. J. Mater. Chem. B 2016, 4, 1528–1534. [Google Scholar] [CrossRef]
- Wu, T.-Y.; Zrimsek, A.B.; Bykov, S.V.; Jakubek, R.S.; Asher, S.A. Hydrophobic collapse initiates the poly(n-isopropylacrylamide) volume phase transition reaction coordinate. J. Phys. Chem. B 2018, 122, 3008–3014. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.; Tomlins, P.; Sahota, T. Thermoresponsive gels. Gels 2017, 3, 4. [Google Scholar] [CrossRef]
- Seuring, J.; Agarwal, S. Polymers with upper critical solution temperature in aqueous solution. Macromol. Rapid Commun. 2012, 33, 1898–1920. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Hoffman, A.S.; Stayton, P.S. Poly(n-isopropylacrylamide-co-propylacrylic acid) copolymers that respond sharply to temperature and ph. Biomacromolecules 2006, 7, 1381–1385. [Google Scholar] [CrossRef] [PubMed]
- Lanzalaco, S.; Armelin, E. Poly(n-isopropylacrylamide) and copolymers: A review on recent progresses in biomedical applications. Gels 2017, 3, 36. [Google Scholar] [CrossRef]
- Tauer, K.; Gau, D.; Schulze, S.; Völkel, A.; Dimova, R. Thermal property changes of poly(n-isopropylacrylamide) microgel particles and block copolymers. Colloid Polym. Sci. 2009, 287, 299. [Google Scholar] [CrossRef]
- Okano, T.; Yamada, N.; Sakai, H.; Sakurai, Y. A novel recovery system for cultured cells using plasma-treated polystyrene dishes grafted with poly(n-isopropylacrylamide). J. Biomed. Mater. Res. 1993, 27, 1243–1251. [Google Scholar] [CrossRef]
- Alarcon, C.D.H.; Pennadam, S.; Alexander, C. Stimuli responsive polymers for biomedical applications. Chem. Soc. Rev. 2005, 34, 276–285. [Google Scholar] [CrossRef]
- Zhang, X.Z.; Yang, Y.Y.; Wang, F.J.; Chung, T.S. Thermosensitive poly(n-isopropylacrylamide-co-acrylic acid) hydrogels with expanded network structures and improved oscillating swelling-deswelling properties. Langmuir 2002, 18, 2013–2018. [Google Scholar] [CrossRef]
- Kim, S.; Healy, K.E. Synthesis and characterization of injectable poly(n-isopropylacrylamide-co-acrylic acid) hydrogels with proteolytically degradable cross-links. Biomacromolecules 2003, 4, 1214–1223. [Google Scholar] [CrossRef] [PubMed]
- Varghese, S.; Lele, A.K.; Mashelkar, R.A. Designing new thermoreversible gels by molecular tailoring of hydrophilic-hydrophobic interactions. J. Chem. Phys. 2000, 112, 3063–3070. [Google Scholar] [CrossRef]
- Klouda, L.; Mikos, A.G. Thermoresponsive hydrogels in biomedical applications. Eur. J. Pharm. Biopharm. 2008, 68, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Chevillard, C.; Axelos, M.A.V. Phase separation of aqueous solution of methylcellulose. Colloid Polym. Sci. 1997, 275, 537–545. [Google Scholar] [CrossRef]
- Nasatto, P.; Pignon, F.; Silveira, J.; Duarte, M.; Noseda, M.; Rinaudo, M. Methylcellulose, a cellulose derivative with original physical properties and extended applications. Polymers 2015, 7, 777. [Google Scholar] [CrossRef]
- Arvidson, S.A.; Lott, J.R.; McAllister, J.W.; Zhang, J.; Bates, F.S.; Lodge, T.P.; Sammler, R.L.; Li, Y.; Brackhagen, M. Interplay of phase separation and thermoreversible gelation in aqueous methylcellulose solutions. Macromolecules 2013, 46, 300–309. [Google Scholar] [CrossRef]
- Kobayashi, K.; Huang, C.-I.; Lodge, T.P. Thermoreversible gelation of aqueous methylcellulose solutions. Macromolecules 1999, 32, 7070–7077. [Google Scholar] [CrossRef]
- Lutz, J.F.; Akdemir, O.; Hoth, A. Point by point comparison of two thermosensitive polymers exhibiting a similar lcst: Is the age of poly(nipam) over? J. Am. Chem. Soc. 2006, 128, 13046–13047. [Google Scholar] [CrossRef]
- Lutz, J.-F.; Hoth, A. Preparation of ideal peg analogues with a tunable thermosensitivity by controlled radical copolymerization of 2-(2-methoxyethoxy)ethyl methacrylate and oligo(ethylene glycol) methacrylate. Macromolecules 2006, 39, 893–896. [Google Scholar] [CrossRef]
- Schuh, R.; Panotopoulos, J.; Puchner, S.E.; Willegger, M.; Hobusch, G.M.; Windhager, R.; Funovics, P.T. Vascularised or non-vascularised autologous fibular grafting for the reconstruction of a diaphyseal bone defect after resection of a musculoskeletal tumour. Bone Jt. J. 2014, 96-B, 1258–1263. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Huang, D.; Huang, Y.; Liu, Y.; Lin, B.; Yu, C.; Mou, Y.; Wu, W.; Zhang, H.; Lin, H. Efficacy of combined therapy of periosteum and bone allograft in a critical-sized defect model in new zealand white rabbits. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2014, 20, 2394–2403. [Google Scholar]
- Calori, G.M.; Mazza, E.; Colombo, M.; Ripamonti, C. The use of bone-graft substitutes in large bone defects: Any specific needs? Injury 2011, 42, S56–S63. [Google Scholar] [CrossRef] [PubMed]
- Pirraco, R.P.; Iwata, T.; Yoshida, T.; Marques, A.P.; Yamato, M.; Reis, R.L.; Okano, T. Endothelial cells enhance the in vivo bone-forming ability of osteogenic cell sheets. Lab. Investig. 2014, 94, 663. [Google Scholar] [CrossRef] [PubMed]
- Ishijima, M.; Hirota, M.; Park, W.; Honda, M.J.; Tsukimura, N.; Isokawa, K.; Ishigami, T.; Ogawa, T. Osteogenic cell sheets reinforced with photofunctionalized micro-thin titanium. J. Biomater. Appl. 2015, 29, 1372–1384. [Google Scholar] [CrossRef] [PubMed]
- Akahane, M.; Nakamura, A.; Ohgushi, H.; Shigematsu, H.; Dohi, Y.; Takakura, Y. Osteogenic matrix sheet-cell transplantation using osteoblastic cell sheet resulted in bone formation without scaffold at an ectopic site. J. Tissue Eng. Regen. Med. 2008, 2, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Akahane, M.; Shigematsu, H.; Tadokoro, M.; Ueha, T.; Matsumoto, T.; Tohma, Y.; Kido, A.; Imamura, T.; Tanaka, Y. Scaffold-free cell sheet injection results in bone formation. J. Tissue Eng. Regen. Med. 2010, 4, 404–411. [Google Scholar] [CrossRef]
- Kira, T.; Akahane, M.; Omokawa, S.; Shimizu, T.; Kawate, K.; Onishi, T.; Tanaka, Y. Bone regeneration with osteogenic matrix cell sheet and tricalcium phosphate: An experimental study in sheep. World J. Orthop. 2017, 8, 754–760. [Google Scholar] [CrossRef]
- Pirraco, R.P.; Obokata, H.; Iwata, T.; Marques, A.P.; Tsuneda, S.; Yamato, M.; Reis, R.L.; Okano, T. Development of osteogenic cell sheets for bone tissue engineering applications. Tissue Eng. Part A 2011, 17, 1507–1515. [Google Scholar] [CrossRef]
- Shotorbani, B.B.; Andre, H.; Barzegar, A.; Zarghami, N.; Salehi, R.; Alizadeh, E. Cell sheet biofabrication by co-administration of mesenchymal stem cells secretome and vitamin c on thermoresponsive polymer. J. Mater. Sci. Mater. Med. 2018, 29, 170. [Google Scholar] [CrossRef]
- Forghani, A.; Kriegh, L.; Hogan, K.; Chen, C.; Brewer, G.; Tighe, T.B.; Devireddy, R.; Hayes, D. Fabrication and characterization of cell sheets using methylcellulose and pnipaam thermoresponsive polymers: A comparison study. J. Biomed. Mater. Res. Part A 2017, 105, 1346–1354. [Google Scholar] [CrossRef] [PubMed]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Tobita, M.; Cagri Uysal, A.; Ogawa, R.; Hyakusoku, H.; Mizuno, H. Periodontal Tissue Regeneration with Adipose-Derived Stem Cells. Tissue Eng. Part A 2008, 14, 945–953. [Google Scholar] [CrossRef] [PubMed]
- Morikuni, T.; Hiroshi, M. Periodontal disease and periodontal tissue regeneration. Curr. Stem Cell Res. Ther. 2010, 5, 168–174. [Google Scholar]
- Yu, N.; Oortgiesen, D.A.W.; Bronckers, A.L.J.J.; Yang, F.; Walboomers, X.F.; Jansen, J.A. Enhanced periodontal tissue regeneration by periodontal cell implantation. J. Clin. Periodontol. 2013, 40, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Bartold, P.M.; Xiao, Y.; Lyngstaadas, S.P.; Paine, M.L.; Snead, M.L. Principles and applications of cell delivery systems for periodontal regeneration. Periodontol. 2000 2006, 41, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Grusovin, M.G.; Papanikolaou, N.; Coulthard, P.; Worthington, H.V. Enamel matrix derivative (emdogain(r)) for periodontal tissue regeneration in intrabony defects. Cochrane Database Syst. Rev. 2009. [Google Scholar] [CrossRef]
- Hynes, K.; Menicanin, D.; Gronthos, S.; Bartold, P.M. Clinical utility of stem cells for periodontal regeneration. Periodontol. 2000 2012, 59, 203–227. [Google Scholar] [CrossRef]
- Iwata, T.; Yamato, M.; Tsuchioka, H.; Takagi, R.; Mukobata, S.; Washio, K.; Okano, T.; Ishikawa, I. Periodontal regeneration with multi-layered periodontal ligament-derived cell sheets in a canine model. Biomaterials 2009, 30, 2716–2723. [Google Scholar] [CrossRef]
- Iwata, T.; Yamato, M.; Ishikawa, I.; Ando, T.; Okano, T. Tissue engineering in periodontal tissue. Anat. Rec. 2014, 297, 16–25. [Google Scholar] [CrossRef]
- Tsumanuma, Y.; Iwata, T.; Kinoshita, A.; Washio, K.; Yoshida, T.; Yamada, A.; Takagi, R.; Yamato, M.; Okano, T.; Izumi, Y. Allogeneic transplantation of periodontal ligament-derived multipotent mesenchymal stromal cell sheets in canine critical-size supra-alveolar periodontal defect model. BioRes. Open Access 2016, 5, 22–36. [Google Scholar] [CrossRef] [PubMed]
- Iwata, T.; Yamato, M.; Washio, K.; Yoshida, T.; Tsumanuma, Y.; Yamada, A.; Onizuka, S.; Izumi, Y.; Ando, T.; Okano, T.; et al. Periodontal regeneration with autologous periodontal ligament-derived cell sheets—A safety and efficacy study in ten patients. Regen. Ther. 2018, 9, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, M.; Yamato, M.; Kikuchi, A.; Okano, T.; Ishikawa, I. Human periodontal ligament cell sheets can regenerate periodontal ligament tissue in an athymic rat model. Tissue Eng. 2005, 11, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Tsumanuma, Y.; Iwata, T.; Washio, K.; Yoshida, T.; Yamada, A.; Takagi, R.; Ohno, T.; Lin, K.; Yamato, M.; Ishikawa, I.; et al. Comparison of different tissue-derived stem cell sheets for periodontal regeneration in a canine 1-wall defect model. Biomaterials 2011, 32, 5819–5825. [Google Scholar] [CrossRef] [PubMed]
- Ojeh, N.; Pastar, I.; Tomic-Canic, M.; Stojadinovic, O. Stem cells in skin regeneration, wound healing, and their clinical applications. Int. J. Mol. Sci. 2015, 16, 25476–25501. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Paquet-Fifield, S.; Schluter, H.; Li, A.; Aitken, T.; Gangatirkar, P.; Blashki, D.; Koelmeyer, R.; Pouliot, N.; Palatsides, M.; Ellis, S.; et al. A role for pericytes as microenvironmental regulators of human skin tissue regeneration. J. Clin. Investig. 2009, 119, 2795–2806. [Google Scholar] [CrossRef]
- Hanson, S.E.; Bentz, M.L.; Hematti, P. Mesenchymal stem cell therapy for nonhealing cutaneous wounds. Plast. Reconstr. Surg. 2010, 125, 510–516. [Google Scholar] [CrossRef]
- Li, M.; Zhao, Y.; Hao, H.; Han, W.; Fu, X. Mesenchymal stem cell–based therapy for nonhealing wounds: Today and tomorrow. Wound Repair Regen. 2015, 23, 465–482. [Google Scholar] [CrossRef]
- You, H.-J.; Namgoong, S.; Han, S.-K.; Jeong, S.-H.; Dhong, E.-S.; Kim, W.-K. Wound-healing potential of human umbilical cord blood–derived mesenchymal stromal cells in vitro—A pilot study. Cytotherapy 2015, 17, 1506–1513. [Google Scholar] [CrossRef]
- Hassan, W.U.; Greiser, U.; Wang, W. Role of adipose-derived stem cells in wound healing. Wound Repair Regen. 2014, 22, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Elloumi-Hannachi, I.; Yamato, M.; Okano, T. Cell sheet engineering: A unique nanotechnology for scaffold-free tissue reconstruction with clinical applications in regenerative medicine. J. Intern. Med. 2010, 267, 54–70. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, Y.; Shimizu, T.; Sasagawa, T.; Sekine, H.; Sakaguchi, K.; Kikuchi, T.; Sekine, W.; Sekiya, S.; Yamato, M.; Umezu, M.; et al. Fabrication of functional three-dimensional tissues by stacking cell sheets in vitro. Nat. Protoc. 2012, 7, 850–858. [Google Scholar] [CrossRef]
- Cerqueira, M.T.; Pirraco, R.P.; Santos, T.C.; Rodrigues, D.B.; Frias, A.M.; Martins, A.R.; Reis, R.L.; Marques, A.P. Human adipose stem cells cell sheet constructs impact epidermal morphogenesis in full-thickness excisional wounds. Biomacromolecules 2013, 14, 3997–4008. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Grahovac, T.; Oh, S.J.; Ieraci, M.; Rubin, J.P.; Marra, K.G. Evaluation of a multi-layer adipose-derived stem cell sheet in a full-thickness wound healing model. Acta Biomater. 2013, 9, 5243–5250. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Iwata, T.; Morikawa, S.; Yamato, M.; Okano, T.; Uchigata, Y. Allogeneic transplantation of an adipose-derived stem cell sheet combined with artificial skin accelerates wound healing in a rat wound model of type 2 diabetes and obesity. Diabetes 2015, 64, 2723. [Google Scholar] [CrossRef] [PubMed]
- Moschouris, K.; Firoozi, N.; Kang, Y. The application of cell sheet engineering in the vascularization of tissue regeneration. Regen. Med. 2016, 11, 559–570. [Google Scholar] [CrossRef]
- Akiyama, H.; Ito, A.; Kawabe, Y.; Kamihira, M. Genetically engineered angiogenic cell sheets using magnetic force-based gene delivery and tissue fabrication techniques. Biomaterials 2010, 31, 1251–1259. [Google Scholar] [CrossRef]
- Baldwin, J.; Antille, M.; Bonda, U.; De-Juan-Pardo, E.M.; Khosrotehrani, K.; Ivanovski, S.; Petcu, E.B.; Hutmacher, D.W. In vitro pre-vascularisation of tissue-engineered constructs a co-culture perspective. Vasc. Cell 2014, 6, 13. [Google Scholar] [CrossRef]
- Makarevich, P.I.; Boldyreva, M.A.; Gluhanyuk, E.V.; Efimenko, A.Y.; Dergilev, K.V.; Shevchenko, E.K.; Sharonov, G.V.; Gallinger, J.O.; Rodina, P.A.; Sarkisyan, S.S.; et al. Enhanced angiogenesis in ischemic skeletal muscle after transplantation of cell sheets from baculovirus-transduced adipose-derived stromal cells expressing vegf165. Stem Cell Res. Ther. 2015, 6, 204. [Google Scholar] [CrossRef]
- Shibuya, M. Vascular endothelial growth factor and its receptor system: Physiological functions in angiogenesis and pathological roles in various diseases. J. Biochem. 2013, 153, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Tamaki, T.; Soeda, S.; Hashimoto, H.; Saito, K.; Sakai, A.; Nakajima, N.; Masuda, M.; Fukunishi, N.; Uchiyama, Y.; Terachi, T.; et al. 3d reconstitution of nerve-blood vessel networks using skeletal muscle-derived multipotent stem cell sheet pellets. Regen. Med. 2013, 8, 437–451. [Google Scholar] [CrossRef] [PubMed]
- Masumoto, H.; Ikuno, T.; Takeda, M.; Fukushima, H.; Marui, A.; Katayama, S.; Shimizu, T.; Ikeda, T.; Okano, T.; Sakata, R.; et al. Human ips cell-engineered cardiac tissue sheets with cardiomyocytes and vascular cells for cardiac regeneration. Sci. Rep. 2014, 4, 6716. [Google Scholar] [CrossRef] [PubMed]
- Kamata, S.; Miyagawa, S.; Fukushima, S.; Nakatani, S.; Kawamoto, A.; Saito, A.; Harada, A.; Shimizu, T.; Daimon, T.; Okano, T.; et al. Improvement of cardiac stem cell sheet therapy for chronic ischemic injury by adding endothelial progenitor cell transplantation: Analysis of layer-specific regional cardiac function. Cell Transpl. 2014, 23, 1305–1319. [Google Scholar] [CrossRef] [PubMed]
- Bora, P.; Majumdar, A.S. Adipose tissue-derived stromal vascular fraction in regenerative medicine: A brief review on biology and translation. Stem Cell Res. Ther. 2017, 8, 145. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.; Cerqueira, M.T.; Santos, T.C.; Sampaio-Marques, B.; Ludovico, P.; Marques, A.P.; Pirraco, R.P.; Reis, R.L. Cell sheet engineering using the stromal vascular fraction of adipose tissue as a vascularization strategy. Acta Biomater. 2017, 55, 131–143. [Google Scholar] [CrossRef]
- Rayatpisheh, S.; Heath, D.E.; Shakouri, A.; Rujitanaroj, P.O.; Chew, S.Y.; Chan-Park, M.B. Combining cell sheet technology and electrospun scaffolding for engineered tubular, aligned, and contractile blood vessels. Biomaterials 2014, 35, 2713–2719. [Google Scholar] [CrossRef]
- Takahashi, H.; Shimizu, T.; Nakayama, M.; Yamato, M.; Okano, T. The use of anisotropic cell sheets to control orientation during the self-organization of 3d muscle tissue. Biomaterials 2013, 34, 7372–7380. [Google Scholar] [CrossRef]
- Takahashi, H.; Nakayama, M.; Shimizu, T.; Yamato, M.; Okano, T. Anisotropic cell sheets for constructing three-dimensional tissue with well-organized cell orientation. Biomaterials 2011, 32, 8830–8838. [Google Scholar] [CrossRef]
- Yoon, J.-K.; Misra, M.; Yu, S.J.; Kim, H.Y.; Bhang, S.H.; Song, S.Y.; Lee, J.-R.; Ryu, S.; Choo, Y.W.; Jeong, G.-J.; et al. Thermosensitive, stretchable, and piezoelectric substrate for generation of myogenic cell sheet fragments from human mesenchymal stem cells for skeletal muscle regeneration. Adv. Funct. Mater. 2017, 27, 1703853. [Google Scholar] [CrossRef]
- Shimizu, T.; Sekine, H.; Yang, J.; Isoi, Y.; Yamato, M.; Kikuchi, A.; Kobayashi, E.; Okano, T. Polysurgery of cell sheet grafts overcomes diffusion limits to produce thick, vascularized myocardial tissues. FASEB J. 2006, 20, 708–710. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, M.; Miyagawa, S.; Fukushima, S.; Saito, A.; Miki, K.; Ito, E.; Sougawa, N.; Kawamura, T.; Daimon, T.; Shimizu, T.; et al. Enhanced survival of transplanted human induced pluripotent stem cell-derived cardiomyocytes by the combination of cell sheets with the pedicled omental flap technique in a porcine heart. Circulation 2013, 128, S87–S94. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.; Chen, C.H.; Lin, W.W.; Hwang, S.M.; Hsieh, P.C.; Lai, P.H.; Yeh, Y.C.; Chang, Y.; Sung, H.W. Direct intramyocardial injection of mesenchymal stem cell sheet fragments improves cardiac functions after infarction. Cardiovasc. Res. 2008, 77, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Yeh, Y.C.; Lee, W.Y.; Yu, C.L.; Hwang, S.M.; Chung, M.F.; Hsu, L.W.; Chang, Y.; Lin, W.W.; Tsai, M.S.; Wei, H.J.; et al. Cardiac repair with injectable cell sheet fragments of human amniotic fluid stem cells in an immune-suppressed rat model. Biomaterials 2010, 31, 6444–6453. [Google Scholar] [CrossRef] [PubMed]
- Ohki, T.; Yamato, M.; Ota, M.; Takagi, R.; Murakami, D.; Kondo, M.; Sasaki, R.; Namiki, H.; Okano, T.; Yamamoto, M. Prevention of esophageal stricture after endoscopic submucosal dissection using tissue-engineered cell sheets. Gastroenterology 2012, 143, 582–588. [Google Scholar] [CrossRef]
- Tatsumi, K.; Okano, T. Hepatocyte transplantation: Cell sheet technology for liver cell transplantation. Curr. Transplant. Rep. 2017, 4, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.; Xie, A.W.; Yamato, M.; Okano, T.; Wong, J.Y. Stacking of aligned cell sheets for layer-by-layer control of complex tissue structure. Biomaterials 2011, 32, 5625–5632. [Google Scholar] [CrossRef]
- Sasagawa, T.; Shimizu, T.; Sekiya, S.; Haraguchi, Y.; Yamato, M.; Sawa, Y.; Okano, T. Design of prevascularized three-dimensional cell-dense tissues using a cell sheet stacking manipulation technology. Biomaterials 2010, 31, 1646–1654. [Google Scholar] [CrossRef]
- Sawa, Y.; Miyagawa, S.; Sakaguchi, T.; Fujita, T.; Matsuyama, A.; Saito, A.; Shimizu, T.; Okano, T. Tissue engineered myoblast sheets improved cardiac function sufficiently to discontinue lvas in a patient with dcm: Report of a case. Surg. Today 2012, 42, 181–184. [Google Scholar] [CrossRef]
- Miyagawa, S.; Domae, K.; Yoshikawa, Y.; Fukushima, S.; Nakamura, T.; Saito, A.; Sakata, Y.; Hamada, S.; Toda, K.; Pak, K.; et al. Phase i clinical trial of autologous stem cell-sheet transplantation therapy for treating cardiomyopathy. J. Am. Heart Assoc. 2017, 6. [Google Scholar] [CrossRef]
- Yoshikawa, Y.; Miyagawa, S.; Toda, K.; Saito, A.; Sakata, Y.; Sawa, Y. Myocardial regenerative therapy using a scaffold-free skeletal-muscle-derived cell sheet in patients with dilated cardiomyopathy even under a left ventricular assist device: A safety and feasibility study. Surg. Today 2017, 48, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Feng, F.; Akiyama, K.; Liu, Y.; Yamaza, T.; Wang, T.M.; Chen, J.H.; Wang, B.B.; Huang, G.T.; Wang, S.; Shi, S. Utility of pdl progenitors for in vivo tissue regeneration: A report of 3 cases. Oral Dis. 2010, 16, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.M.; Gao, L.N.; Tian, B.M.; Zhang, X.Y.; Zhang, Y.J.; Dong, G.Y.; Lu, H.; Chu, Q.; Xu, J.; Yu, Y.; et al. Treatment of periodontal intrabony defects using autologous periodontal ligament stem cells: A randomized clinical trial. Stem Cell Res. Ther. 2016, 7, 33. [Google Scholar] [CrossRef] [PubMed]

| Applications | Methods for Cell Sheet Formation | Summarized Results | Refs. |
|---|---|---|---|
| Bone | Physical detachment | In vivo bone formation by transplanting bone marrow cell sheets | [68] |
| Bone | Poly(N-isopropylacrylamide) | New bone formation at day seven post-transplantation upon transplantation of rat bone marrow stromal cell sheet into subcutaneous site. | [69] |
| Bone | Poly(N-isopropyl acryl amide-co-methacrylic acid) | In vitro osteogenesis of human adipose-derived stem cell sheets validated by Alizarin red staining and qPCR. | [70] |
| Bone | Methylcellulose and poly(N-isopropylacrylamide) | In vitro osteogenesis of human adipose-derived stem cell sheets validated by ALP, Alizarin Red staining and qPCR | [71] |
| PDL | Poly(N-isopropylacrylamide) | Validation of safety and efficacy of autologous PDL-derived cell sheets | [82] |
| PDL | Poly(N-isopropylacrylamide) | Regeneration of PDL tissues in a rat mesial dehiscence model | [83] |
| PDL | Poly(N-isopropylacrylamide) | Newly formed cementum and well-oriented PDL fibers by PDL cell sheet transplantation | [84] |
| PDL | Poly(N-isopropylacrylamide) | New bone and cementum-like tissue formation in a canine periodontal defect model | [79] |
| Skin | Poly(N-isopropylacrylamide) | In vivo engraftment and skin regeneration by multiple stacks of hASC-derived 3D cell sheets in a full-thickness wound model | [93] |
| Skin | Poly(N-isopropylacrylamide) | Newly formed collagen matrix and blood vessel structures in a full-thickness wound model using hASC-derived cell sheets | [94] |
| Skin | Poly(N-isopropylacrylamide) | Accelerated wound closure and dermis tissue formation by rat ASC cell sheets | [96] |
| Cardiac Muscle | Poly(N-isopropylacrylamide) | Enhanced cardiac function by transplanted pre-vascularized cardiac cell sheets in a rat myocardial infarction model | [102] |
| Cardiac Muscle | Poly(N-isopropylacrylamide) | Recovery of the endocardium and capillary density by cardiac stem cell sheet and endothelial progenitors | [104] |
| Cardiac Muscle | Physical detachment | Significant functional recovery of the ischemic epicardium by SVF-derived cell sheets | [106] |
| Smooth Muscle | Poly(N-isopropylacrylamide) and polycaprolactone | Engineered uniaxially aligned and tubular blood vessel structure by smooth muscle cell sheets | [107] |
| Skeletal Muscle | Poly(N-isopropylacrylamide) | Uniaxially aligned skeletal muscle cell tissue formation by skeletal muscle cell sheets | [107,108] |
| Skeletal Muscle | Poly(N-isopropylacrylamide) | Uniaxially aligned smooth muscle cell sheets induced to form blood vessel-like cellular orientation | [108] |
| Skeletal Muscle | Poly(N-isopropylacrylamide) | Well-controlled 3D aligned skeletal cell sheets having physical and biological successful anisotropy | [110] |
| Cardiac Muscle | Poly(N-isopropylacrylamide) | Well-organized microvascular formation and enhanced heart function by cell-dense multiple layered myocardial cell sheet grafts | [111] |
| Cardiac Muscle | Poly(N-isopropylacrylamide) | Enhanced blood supply and cell viability by hiPSC-derived cell sheets | [112] |
| Cardiac Muscle | Methylcellulose | Improved LV functions by rat BM-MSC sheets in a rat myocardial infarction model | [113] |
| Cardiac Muscle | Methylcellulose | Improved LV functions by human AFSC-derived cell sheets in a rat myocardial infarction model | [114] |
| Clinical trial: cardiac muscle | Poly(N-isopropylacrylamide) | Improved cardiac functions by autologous myoblast cell sheets transplantation | [119] |
| Clinical trial: cardiac muscle | Poly(N-isopropylacrylamide) | Improved clinical condition without any arrhythmia and a left ventricular assist system | [120] |
| Clinical trial: cardiac muscle | Poly(N-isopropylacrylamide) | Improved LV functions by autologous cell sheets transplantation | [121] |
| Clinical trial: periodontal ligament | Poly(N-isopropylacrylamide) | Improved periodontitis symptoms by autologous PDL-derived stem cell sheets | [122] |
| Clinical trial: periodontal ligament | Poly(N-isopropylacrylamide) | Improved periodontitis symptoms by autologous PDL-derived stem cell sheets | [123] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Kim, Y.; Park, J.; Hwang, N.S.; Lee, Y.K.; Hwang, Y. Recent Advances in Engineered Stem Cell-Derived Cell Sheets for Tissue Regeneration. Polymers 2019, 11, 209. https://doi.org/10.3390/polym11020209
Kim H, Kim Y, Park J, Hwang NS, Lee YK, Hwang Y. Recent Advances in Engineered Stem Cell-Derived Cell Sheets for Tissue Regeneration. Polymers. 2019; 11(2):209. https://doi.org/10.3390/polym11020209
Chicago/Turabian StyleKim, Hyunbum, Yunhye Kim, Jihyun Park, Nathaniel S. Hwang, Yun Kyung Lee, and Yongsung Hwang. 2019. "Recent Advances in Engineered Stem Cell-Derived Cell Sheets for Tissue Regeneration" Polymers 11, no. 2: 209. https://doi.org/10.3390/polym11020209
APA StyleKim, H., Kim, Y., Park, J., Hwang, N. S., Lee, Y. K., & Hwang, Y. (2019). Recent Advances in Engineered Stem Cell-Derived Cell Sheets for Tissue Regeneration. Polymers, 11(2), 209. https://doi.org/10.3390/polym11020209







