Combinations of Antimicrobial Polymers with Nanomaterials and Bioactives to Improve Biocidal Therapies
Abstract
:1. Introduction
2. Co-Administration of Two Different Antimicrobial Polymers
2.1. Improving Antimicrobial Activity of Chitosan by Grafting Biocidal Polymers
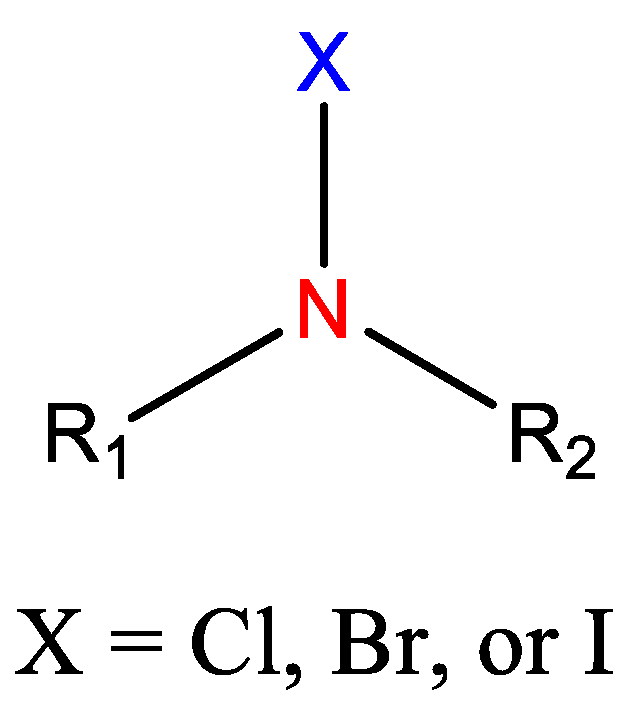
2.2. N-Halamines Based Polymers and Polycations Working in Tandem
| Schematic Representation of the Polymeric Agent | Surface | Micro-Organism | Percentage Reduction of Bacteria 1 (Representative Examples) | Contact Time | Ref. | |
|---|---|---|---|---|---|---|
| HQ1 |  | Cotton (C) Poly(ethylene terephthalate) (PET) | MRSA E. coli | MRSA: 2.0 × 106 CFU/mL Unmodified-C = 0% HQ1-g-C = 27.2% Chlorinated H1-g-C = 100% | 5 min | [54] |
| HQ2 |  | Cotton (C) | S. aureus E. coli | S. aureus: 6.67 × 105 CFU/mL Unmodified-C = N/A HQ2-g-C = 89.55% Chlorinated X-g-C = 100% | 5 min | [55] |
| HQ2 |  | Mesoporous silica SBA-15 | S. aureus E. coli | E. coli: 3.3 × 107 CFU/mL SBA-15: 2.27% SAB-15-g-HQ6 = 24.1% Chlorinated SAB-15-g-HQ6 = 100% | 10 min | [56] |
| H1-Q1 |  | Cellulose fiber (Ce) | S. aureus E. coli | E. coli: 106 to 107 CFU/mL aUnmodified-Ce = 1.0% H1-g-Ce = 99.0% Q1-g-Ce = 93.0% Chlorinated H3Q1-g-Ce = 99.5% | 5 min | [57] |
| HQ3 |  | Macroporous crosslinked chloromethylated polystyrene (CMPS) resin | S. aureus E. coli | E. coli: 1.8 × 107 CFU/mL Unmodified-CMPS = 32.39% HQ3-g-CMPS = 63.69% Chlorinated HQ3-g-CMPS = 99.99% | 5 min | [58] |
| HQ4 |  | Polypropylene (PP) | L. mono-cytogenes | L. monocytogenes: 1.0 × 106 CFU/mL aUnmodified-PP ≈ 0% Chlorinated-PP ≈ 0% HQ4-g-PP ≈ 99.9% Chlorinated HQ4-g-PP > 99.99% | 120 min | [59] |
| H2, Q2 |  | Cotton (C) | S. aureus E. coli | E. coli: 2.51 × 106 CFU/mL C + H2 = 2.27% Chlorinated-C + H2 = 100% C + Q2 = 46.3% Chlorinated C + H2 + Q2 = 100% | 30 min | [60] |
| HQ5 |  | Cotton (C) | S. aureus E. coli | E. coli: 1.93 × 106 CFU/mL Unmodified C = 30.57% C-g-HQ5 = 51.4% Chlorinated C-g-HQ5 = 100% | 30 min | [61] |
2.3. Nitric Oxide co-Administration Systems
3. Enhancing Efficiency of Antimicrobial Cationic Polymers by Incorporating Metal Oxides and Metal Nanoparticles
4. Synergistic Effect Between Antimicrobial Polymers and Carbon Nanostructures
5. Combination of Antimicrobial Cationic Polymers with Antibiotics
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Nomenclature
| AgBr | silver bromide |
| AgNPs | silver nanoparticles |
| AM | acrylamide |
| AMR | antimicrobial resistance |
| AuNps | gold nanoparticles |
| BPAM | benzophenone based quaternary ammonium molecules |
| CMPS | macroporous crosslinked chloromethylated polystyrene |
| CNTs | carbon nanotubes |
| CS | chitosan |
| CuNPs | copper nanoparticles |
| DADMAC | diallyl dimethyl ammonium chloride |
| DMDEPAC | N,N-dimethyl-N-dodecyl-N-(1,2-epoxypropyl) ammonium chloride |
| DMOEPAC | N,N-dimethyl-N-octadecyl-N-(1,2-epoxypropyl) ammonium chloride |
| DNA | deoxyribonucleic acid |
| GMs | graphene-based materials |
| GO | graphene oxide |
| MIC | minimum inhibitory concentration |
| MMT | montmorillonite |
| MNPs | magnetic nanoparticles |
| MRS | more resistant strains |
| MWCNT | multi wall carbon nanotubes |
| NO | nitric oxide |
| NPs | nanoparticles |
| NPVP | poly(4-vinyl-N-hexylpyridinium) bromide) |
| PAMAM | poly(amidoamine) |
| PDMAEMA | poly(dimethyl aminoethyl methacrylate) |
| PEG | poly(ethylene glycol) |
| PET | poly(ethylene terephthalate) |
| PMMA | poly(methyl methacrylate) |
| PP | polypropylene |
| PPI | poly(propylene imine) |
| PTBAM | poly[2-(tert-butylaminoethyl) methacrylate] |
| PTPB | (4-penten-1-yl) triphenylphosphonium bromide |
| PVA | poly(vinyl alcohol) |
| QAS | quaternary ammonium salts |
| QPS | quaternary phosphonium salts |
| RNA | ribonucleic acid |
| ROS | reactive oxygen species |
| SNAP | S-nitroso-N-acetylpenicillamine |
| SSD | silver sulfadiazine |
| ZnO | zinc oxide |
References
- O’Neill, J. Antimicrobial Resistance: Tackling a crisis for the health and wealth of nations. PLoS Med. 2016, 13, e1002130. [Google Scholar]
- Nguyen, D.; Nguyen, T.-K.; Rice, S.A.; Boyer, C. CO-Releasing Polymers Exert Antimicrobial Activity. Biomacromolecules 2015, 16, 2776–2786. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, X.; Saravanan, R.; Li, C.M.; Leong, S.S.J. Antimicrobial macromolecules: Synthesis methods and future applications. RSC Adv. 2012, 2, 4031–4044. [Google Scholar] [CrossRef]
- French, G.L. The continuing crisis in antibiotic resistance. Int. J. Antimicrob. Agents 2010, 36, S3–S7. [Google Scholar] [CrossRef]
- Dever, L.A.; Dermody, T.S. Mechanisms of bacterial resistance to antibiotics. Arch. Intern. Med. 1991, 151, 886–895. [Google Scholar] [CrossRef] [PubMed]
- Ng, V.W.L.; Ke, X.; Lee, A.L.Z.; Hedrick, J.L.; Yang, Y.Y. Synergistic Co-Delivery of Membrane-Disrupting Polymers with Commercial Antibiotics against Highly Opportunistic Bacteria. Adv. Mater. 2013, 25, 6730–6736. [Google Scholar] [CrossRef]
- Fischbach, M.A. Combination therapies for combating antimicrobial resistance. Curr. Opin. Microbiol. 2011, 14, 519–523. [Google Scholar] [CrossRef] [Green Version]
- Cottarel, G.; Wierzbowski, J. Combination drugs, an emerging option for antibacterial therapy. Trends Biotechnol. 2007, 25, 547–555. [Google Scholar] [CrossRef]
- Bollenbach, T. Antimicrobial interactions: Mechanisms and implications for drug discovery and resistance evolution. Curr. Opin. Microbiol. 2015, 27, 1–9. [Google Scholar] [CrossRef]
- Tängdén, T. Combination antibiotic therapy for multidrug-resistant Gram-negative bacteria. Ups. J. Med. Sci. 2014, 119, 149–153. [Google Scholar] [CrossRef]
- Bassetti, M.; Righi, E. New antibiotics and antimicrobial combination therapy for the treatment of gram-negative bacterial infections. Curr. Opin. Crit. Care 2015, 21, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Tyers, M.; Wright, G.D. Drug combinations: A strategy to extend the life of antibiotics in the 21st century. Nat. Rev. Microbiol. 2019, 17, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Allahverdiyev, A.M.; Kon, K.V.; Abamor, E.S.; Bagirova, M.; Rafailovich, M. Coping with antibiotic resistance: Combining nanoparticles with antibiotics and other antimicrobial agents. Expert Rev. Anti. Infect. Ther. 2011, 9, 1035–1052. [Google Scholar] [CrossRef] [PubMed]
- Siedenbiedel, F.; Tiller, J.C. Antimicrobial Polymers in Solution and on Surfaces: Overview and Functional Principles. Polymers (Basel) 2012, 4, 46–71. [Google Scholar] [CrossRef] [Green Version]
- Muñoz, A.; Fernández, M. Polymeric materials with antimicrobial activity. Prog. Polym. Sci. 2012, 37, 281–339. [Google Scholar] [CrossRef]
- Milović, N.M.; Wang, J.; Lewis, K.; Klibanov, A.M. Immobilized N-alkylated polyethylenimine avidly kills bacteria by rupturing cell membranes with no resistance developed. Biotechnol. Bioeng. 2005, 90, 715–722. [Google Scholar] [CrossRef]
- Ng, V.W.L.; Tan, J.P.K.; Leong, J.; Voo, Z.X.; Hedrick, J.L.; Yang, Y.Y. Antimicrobial Polycarbonates: Investigating the Impact of Nitrogen-Containing Heterocycles as Quaternizing Agents. Macromolecules 2014, 47, 1285–1291. [Google Scholar] [CrossRef]
- Kenawy, E.-R.; Worley, S.D.; Broughton, R. The Chemistry and Applications of Antimicrobial Polymers: A State-of-the-Art Review. Biomacromolecules 2007, 8, 1359–1384. [Google Scholar] [CrossRef]
- Jain, A.; Duvvuri, L.S.; Farah, S.; Beyth, N.; Domb, A.J.; Khan, W. Antimicrobial Polymers. Adv. Healthc. Mater. 2014, 3, 1969–1985. [Google Scholar] [CrossRef]
- Álvarez-Paino, M.; Muñoz-Bonilla, A.; Fernández-García, M. Antimicrobial Polymers in the Nano-World. Nanomaterials 2017, 7, 48. [Google Scholar] [CrossRef]
- Arora, A.; Mishra, A. Antibacterial Polymers—A Mini Review. Mater. Today Proc. 2018, 5, 17156–17161. [Google Scholar] [CrossRef]
- Pang, L.Q.; Zhong, L.J.; Zhou, H.F.; Wu, X.E.; Chen, X.D. Grafting of ionic liquids on stainless steel surface for antibacterial application. Coll. Surf. B Biointerfaces 2015, 126, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Rabea, E.I.; Badawy, M.E.T.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as Antimicrobial Agent: Applications and Mode of Action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef]
- Li, Q.; Mahendra, S.; Lyon, D.Y.; Brunet, L.; Liga, M.V.; Li, D.; Alvarez, P.J.J. Antimicrobial nanomaterials for water disinfection and microbial control: Potential applications and implications. Water Res. 2008, 42, 4591–4602. [Google Scholar] [CrossRef]
- Hu, Y.; Du, Y.; Yang, J.; Kennedy, J.F.; Wang, X.; Wang, L. Synthesis, characterization and antibacterial activity of guanidinylated chitosan. Carbohydr. Polym. 2007, 67, 66–72. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, J.; Chen, Y. Synthesis and antimicrobial activity of polymeric guanidine and biguanidine salts. Polymer (Guildf) 1999, 40, 6189–6198. [Google Scholar] [CrossRef]
- Martins, F.A.; Facchi, P.S.; Follmann, D.H.; Pereira, G.A.; Rubira, F.A.; Muniz, C.E. Antimicrobial Activity of Chitosan Derivatives Containing N-Quaternized Moieties in Its Backbone: A Review. Int. J. Mol. Sci. 2014, 15, 20800–20832. [Google Scholar] [CrossRef]
- Liu, J.; Pu, H.; Liu, S.; Kan, J.; Jin, C. Synthesis, characterization, bioactivity and potential application of phenolic acid grafted chitosan: A review. Carbohydr. Polym. 2017, 174, 999–1017. [Google Scholar] [CrossRef]
- Kim, D.-H.; Je, J.-Y. Antimicrobial Activity of Gallic Acid-Grafted-Chitosan Against Fish Pathogens. J. Carbohydr. Chem. 2015, 34, 163–171. [Google Scholar] [CrossRef]
- Kim, J.-H.; Yu, D.; Eom, S.-H.; Kim, S.-H.; Oh, J.; Jung, W.; Kim, Y.-M. Synergistic Antibacterial Effects of Chitosan-Caffeic Acid Conjugate against Antibiotic-Resistant Acne-Related Bacteria. Mar. Drugs 2017, 15, 167. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-S.; Eom, S.-H.; Kim, Y.-M.; Kim, H.S.; Yim, M.-J.; Lee, S.-H.; Kim, D.-H.; Je, J.-Y. Antibacterial and synergic effects of gallic acid-grafted-chitosan with β-lactams against methicillin-resistant Staphylococcus aureus (MRSA). Can. J. Microbiol. 2014, 60, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.M. Binding of a quaternary ammonium polymer-grafted-chitosan onto a chemically modified wool fabric surface: Assessment of mechanical, antibacterial and antifungal properties. RSC Adv. 2015, 5, 35497–35505. [Google Scholar] [CrossRef]
- Chylińska, M.; Kaczmarek, H.; Burkowska-But, A. Preparation and characteristics of antibacterial chitosan films modified with N-halamine for biomedical application. Coll. Surf. B Biointerfaces 2019, 176, 379–386. [Google Scholar] [CrossRef]
- Omidi, S.; Kakanejadifard, A. Modification of chitosan and chitosan nanoparticle by long chain pyridinium compounds: Synthesis, characterization, antibacterial, and antioxidant activities. Carbohydr. Polym. 2019, 208, 477–485. [Google Scholar] [CrossRef]
- Su, Y.; Tian, L.; Yu, M.; Gao, Q.; Wang, D.; Xi, Y.; Yang, P.; Lei, B.; Ma, P.X.; Li, P. Cationic peptidopolysaccharides synthesized by “click” chemistry with enhanced broad-spectrum antimicrobial activities. Polym. Chem. 2017, 8, 3788–3800. [Google Scholar] [CrossRef]
- Hou, Z.; Shankar, Y.V.; Liu, Y.; Ding, F.; Subramanion, J.L.; Ravikumar, V.; Zamudio-Vázquez, R.; Keogh, D.; Lim, H.; Tay, M.Y.F.; et al. Nanoparticles of Short Cationic Peptidopolysaccharide Self-Assembled by Hydrogen Bonding with Antibacterial Effect against Multidrug-Resistant Bacteria. ACS Appl. Mater. Interfaces 2017, 9, 38288–38303. [Google Scholar] [CrossRef]
- Sun, S.; An, Q.; Li, X.; Qian, L.; He, B.; Xiao, H. Synergistic effects of chitosan–guanidine complexes on enhancing antimicrobial activity and wet-strength of paper. Bioresour. Technol. 2010, 101, 5693–5700. [Google Scholar] [CrossRef]
- Liu, Y.; Li, M.; Qiao, M.; Ren, X.; Huang, T.-S.; Buschle-Diller, G. Antibacterial membranes based on chitosan and quaternary ammonium salts modified nanocrystalline cellulose. Polym. Adv. Technol. 2017, 28, 1629–1635. [Google Scholar] [CrossRef]
- Xue, Y.; Pan, Y.; Xiao, H.; Zhao, Y. Novel quaternary phosphonium-type cationic polyacrylamide and elucidation of dual-functional antibacterial/antiviral activity. RSC Adv. 2014, 4, 46887–46895. [Google Scholar] [CrossRef]
- Kenawy, E.-R.; Mahmoud, Y.A.G. Biologically Active Polymers, 6. Macromol. Biosci. 2003, 3, 107–116. [Google Scholar] [CrossRef]
- Hui, F.; Debiemme-Chouvy, C. Antimicrobial N-Halamine Polymers and Coatings: A Review of Their Synthesis, Characterization, and Applications. Biomacromolecules 2013, 14, 585–601. [Google Scholar] [CrossRef] [PubMed]
- Cerkez, I.; Kocer, H.B.; Worley, S.D.; Broughton, R.M.; Huang, T.S. N-halamine copolymers for biocidal coatings. React. Funct. Polym. 2012, 72, 673–679. [Google Scholar] [CrossRef]
- Sun, G.; Wheatley, W.B.; Worley, S.D. A new cyclic N-halamine biocidal polymer. Ind. Eng. Chem. Res. 1994, 33, 168–170. [Google Scholar] [CrossRef]
- Sun, G.; Allen, L.C.; Luckie, E.P.; Wheatley, W.B.; Worley, S.D. Disinfection of Water by N-Halamine Biocidal Polymers. Ind. Eng. Chem. Res. 1995, 34, 4106–4109. [Google Scholar] [CrossRef]
- Qian, L.; Sun, G. Durable and regenerable antimicrobial textiles: Synthesis and applications of 3-methylol-2,2,5,5-tetramethyl-imidazolidin-4-one (MTMIO). J. Appl. Polym. Sci. 2003, 89, 2418–2425. [Google Scholar] [CrossRef]
- Kocer, H.B.; Cerkez, I.; Worley, S.D.; Broughton, R.M.; Huang, T.S. Polymeric Antimicrobial N-Halamine Epoxides. ACS Appl. Mater. Interfaces 2011, 3, 2845–2850. [Google Scholar] [CrossRef]
- Chen, Z.; Sun, Y. N-Halamine-Based Antimicrobial Additives for Polymers: Preparation, Characterization, and Antimicrobial Activity. Ind. Eng. Chem. Res. 2006, 45, 2634–2640. [Google Scholar] [CrossRef]
- Kocer, H.B.; Cerkez, I.; Worley, S.D.; Broughton, R.M.; Huang, T.S. N-Halamine Copolymers for Use in Antimicrobial Paints. ACS Appl. Mater. Interfaces 2011, 3, 3189–3194. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, G. Novel Refreshable N-Halamine Polymeric Biocides: N-Chlorination of Aromatic Polyamides. Ind. Eng. Chem. Res. 2004, 43, 5015–5020. [Google Scholar] [CrossRef]
- Liang, J.; Chen, Y.; Barnes, K.; Wu, R.; Worley, S.D.; Huang, T.S. N-halamine/quat siloxane copolymers for use in biocidal coatings. Biomaterials 2006, 27, 2495–2501. [Google Scholar] [CrossRef] [PubMed]
- Kou, L.; Liang, J.; Ren, X.; Kocer, H.B.; Worley, S.D.; Tzou, Y.M.; Huang, T.S. Synthesis of a Water-Soluble Siloxane Copolymer and Its Application for Antimicrobial Coatings. Ind. Eng. Chem. Res. 2009, 48, 6521–6526. [Google Scholar] [CrossRef]
- Cerkez, I.; Kocer, H.B.; Worley, S.D.; Broughton, R.M.; Huang, T.S. N-Halamine Biocidal Coatings via a Layer-by-Layer Assembly Technique. Langmuir 2011, 27, 4091–4097. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Pu, T.; Zhanel, G.; Zhao, N.; Ens, W.; Liu, S. New Biocide with Both N-Chloramine and Quaternary Ammonium Moieties Exerts Enhanced Bactericidal Activity. Adv. Healthc. Mater. 2012, 1, 609–620. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Y.; Ren, X.; Huang, T.S. Antimicrobial cotton containing N-halamine and quaternary ammonium groups by grafting copolymerization. Appl. Surf. Sci. 2014, 296, 231–236. [Google Scholar] [CrossRef]
- Wang, Y.; Yin, M.; Lin, X.; Li, L.; Li, Z.; Ren, X.; Sun, Y. Tailored synthesis of polymer-brush-grafted mesoporous silicas with N-halamine and quaternary ammonium groups for antimicrobial applications. J. Colloid Interface Sci. 2019, 533, 604–611. [Google Scholar] [CrossRef]
- Hu, B.; Chen, X.; Zuo, Y.; Liu, Z.; Xing, X. Dual action bactericides: Quaternary ammonium/N-halamine-functionalized cellulose fiber. J. Appl. Polym. Sci. 2014, 131, 1–7. [Google Scholar] [CrossRef]
- Jie, Z.; Yan, X.; Zhao, L.; Worley, S.D.; Liang, J. Eco-friendly synthesis of regenerable antimicrobial polymeric resin with N-halamine and quaternary ammonium salt groups. RSC Adv. 2014, 4, 6048–6054. [Google Scholar] [CrossRef]
- Bastarrachea, L.J.; Goddard, J.M. Antimicrobial Coatings with Dual Cationic and N-Halamine Character: Characterization and Biocidal Efficacy. J. Agric. Food Chem. 2015, 63, 4243–4251. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, K.; Li, R.; Ren, X.; Huang, T.S. Antibacterial cotton treated with N-halamine and quaternary ammonium salt. Cellulose 2013, 20, 3123–3130. [Google Scholar] [CrossRef]
- Zhiming, J.; Mingyu, Q.; Xuehong, R.; Ping, Z.; Tung-Shi, H. Preparation of antibacterial cellulose with s-triazine-based quaternarized N-halamine. J. Appl. Polym. Sci. 2017, 134. [Google Scholar]
- Müller, R.; Eidt, A.; Hiller, K.-A.; Katzur, V.; Subat, M.; Schweikl, H.; Imazato, S.; Ruhl, S.; Schmalz, G. Influences of protein films on antibacterial or bacteria-repellent surface coatings in a model system using silicon wafers. Biomaterials 2009, 30, 4921–4929. [Google Scholar] [CrossRef] [PubMed]
- Kügler, R.; Bouloussa, O.; Rondelez, F. Evidence of a charge-density threshold for optimum efficiency of biocidal cationic surfaces. Microbiology 2005, 151, 1341–1348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nablo, B.J.; Prichard, H.L.; Butler, R.D.; Klitzman, B.; Schoenfisch, M.H. Inhibition of implant-associated infections via nitric oxide release. Biomaterials 2005, 26, 6984–6990. [Google Scholar] [CrossRef] [PubMed]
- Brisbois, E.J.; Bayliss, J.; Wu, J.; Major, T.C.; Xi, C.; Wang, S.C.; Bartlett, R.H.; Handa, H.; Meyerhoff, M.E. Optimized polymeric film-based nitric oxide delivery inhibits bacterial growth in a mouse burn wound model. Acta Biomater. 2014, 10, 4136–4142. [Google Scholar] [CrossRef] [Green Version]
- Carpenter, A.W.; Schoenfisch, M.H. Nitric oxide release: Part II. Therapeutic applications. Chem. Soc. Rev. 2012, 41, 3742–3752. [Google Scholar] [CrossRef] [PubMed]
- Wo, Y.; Brisbois, E.J.; Bartlett, R.H.; Meyerhoff, M.E. Recent advances in thromboresistant and antimicrobial polymers for biomedical applications: Just say yes to nitric oxide (NO). Biomater. Sci. 2016, 4, 1161–1183. [Google Scholar] [CrossRef]
- Sun, B.; Slomberg, D.L.; Chudasama, S.L.; Lu, Y.; Schoenfisch, M.H. Nitric Oxide-Releasing Dendrimers as Antibacterial Agents. Biomacromolecules 2012, 13, 3343–3354. [Google Scholar] [CrossRef] [Green Version]
- Worley, B.V.; Soto, R.J.; Kinsley, P.C.; Schoenfisch, M.H. Active Release of Nitric Oxide-Releasing Dendrimers from Electrospun Polyurethane Fibers. ACS Biomater. Sci. Eng. 2016, 2, 426–437. [Google Scholar] [CrossRef]
- Worley, B.V.; Schilly, K.M.; Schoenfisch, M.H. Anti-Biofilm Efficacy of Dual-Action Nitric Oxide-Releasing Alkyl Chain Modified Poly(amidoamine) Dendrimers. Mol. Pharm. 2015, 12, 1573–1583. [Google Scholar] [CrossRef]
- Backlund, C.J.; Worley, B.V.; Schoenfisch, M.H. Anti-biofilm action of nitric oxide-releasing alkyl-modified poly(amidoamine) dendrimers against Streptococcus mutans. Acta Biomater. 2016, 29, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, A.W.; Worley, B.V.; Slomberg, D.L.; Schoenfisch, M.H. Dual Action Antimicrobials: Nitric Oxide Release from Quaternary Ammonium-Functionalized Silica Nanoparticles. Biomacromolecules 2012, 13, 3334–3342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Privett, B.J.; Deupree, S.M.; Backlund, C.J.; Rao, K.S.; Johnson, C.B.; Coneski, P.N.; Schoenfisch, M.H. Synergy of Nitric Oxide and Silver Sulfadiazine against Gram-Negative, Gram-Positive, and Antibiotic-Resistant Pathogens. Mol. Pharm. 2010, 7, 2289–2296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Storm, W.L.; Johnson, J.A.; Worley, B.V.; Slomberg, D.L.; Schoenfisch, M.H. Dual action antimicrobial surfaces via combined nitric oxide and silver release. J. Biomed. Mater. Res. Part. A 2014, 103, 1974–1984. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Slomberg, D.L.; Schoenfisch, M.H. Nitric oxide-releasing chitosan oligosaccharides as antibacterial agents. Biomaterials 2014, 35, 1716–1724. [Google Scholar] [CrossRef]
- Pant, J.; Gao, J.; Goudie, M.J.; Hopkins, S.P.; Locklin, J.; Handa, H. A multi-defense strategy: Enhancing bactericidal activity of a medical grade polymer with a nitric oxide donor and surface-immobilized quaternary ammonium compound. Acta Biomater. 2017, 58, 421–431. [Google Scholar] [CrossRef]
- Namivandi-Zangeneh, R.; Sadrearhami, Z.; Bagheri, A.; Sauvage-Nguyen, M.; Ho, K.K.K.; Kumar, N.; Wong, E.H.H.; Boyer, C. Nitric Oxide-Loaded Antimicrobial Polymer for the Synergistic Eradication of Bacterial Biofilm. ACS Macro Lett. 2018, 7, 592–597. [Google Scholar] [CrossRef]
- Caminade, A.-M.; Fruchon, S.; Turrin, C.-O.; Poupot, M.; Ouali, A.; Maraval, A.; Garzoni, M.; Maly, M.; Furer, V.; Kovalenko, V.; et al. The key role of the scaffold on the efficiency of dendrimer nanodrugs. Nat. Commun. 2015, 6, 7722. [Google Scholar] [CrossRef]
- Zou, P.; Laird, D.; Riga, E.K.; Deng, Z.; Dorner, F.; Perez-Hernandez, H.-R.; Guevara-Solarte, D.L.; Steinberg, T.; Al-Ahmad, A.; Lienkamp, K. Antimicrobial and cell-compatible surface-attached polymer networks – how the correlation of chemical structure to physical and biological data leads to a modified mechanism of action. J. Mater. Chem. B 2015, 3, 6224–6238. [Google Scholar] [CrossRef]
- Martinez-Gutierrez, F.; Olive, P.L.; Banuelos, A.; Orrantia, E.; Nino, N.; Sanchez, E.M.; Ruiz, F.; Bach, H.; Av-Gay, Y. Synthesis, characterization, and evaluation of antimicrobial and cytotoxic effect of silver and titanium nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 681–688. [Google Scholar] [CrossRef]
- Dallas, P.; Sharma, V.K.; Zboril, R. Silver polymeric nanocomposites as advanced antimicrobial agents: Classification, synthetic paths, applications, and perspectives. Adv. Colloid Interface Sci. 2011, 166, 119–135. [Google Scholar] [CrossRef] [PubMed]
- Ozay, O.; Akcali, A.; Otkun, M.T.; Silan, C.; Aktas, N.; Sahiner, N. P(4-VP) based nanoparticles and composites with dual action as antimicrobial materials. Colloids Surfaces B Biointerfaces 2010, 79, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Mei, L.; Zhang, X.; Wang, Y.; Zhang, W.; Lu, Z.; Luo, Y.; Zhao, Y.; Li, C. Multivalent polymer-Au nanocomposites with cationic surfaces displaying enhanced antimicrobial activity. Polym. Chem. 2014, 5, 3038–3044. [Google Scholar] [CrossRef]
- Hajipour, M.J.; Fromm, K.M.; Akbar Ashkarran, A.; Jimenez de Aberasturi, D.; de Larramendi, I.R.; Rojo, T.; Serpooshan, V.; Parak, W.J.; Mahmoudi, M. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef] [Green Version]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef]
- Lara, H.H.; Garza-Treviño, E.N.; Ixtepan-Turrent, L.; Singh, D.K. Silver nanoparticles are broad-spectrum bactericidal and virucidal compounds. J. Nanobiotechnology 2011, 9, 1–8. [Google Scholar] [CrossRef]
- Marambio-Jones, C.; Hoek, E. V A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J. Nanoparticle Res. 2010, 12, 1531–1551. [Google Scholar] [CrossRef]
- Mei, L.; Lu, Z.; Zhang, X.; Li, C.; Jia, Y. Polymer-Ag Nanocomposites with Enhanced Antimicrobial Activity against Bacterial Infection. ACS Appl. Mater. Interfaces 2014, 6, 15813–15821. [Google Scholar] [CrossRef]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramirez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar] [CrossRef] [Green Version]
- Reidy, B.; Haase, A.; Luch, A.; Dawson, K.; Lynch, I. Mechanisms of Silver Nanoparticle Release, Transformation and Toxicity: A Critical Review of Current Knowledge and Recommendations for Future Studies and Applications. Materials (Basel) 2013, 6, 2295–2350. [Google Scholar] [CrossRef] [Green Version]
- Murphy, C.J.; Gole, A.M.; Stone, J.W.; Sisco, P.N.; Alkilany, A.M.; Goldsmith, E.C.; Baxter, S.C. Gold Nanoparticles in Biology: Beyond Toxicity to Cellular Imaging. Acc. Chem. Res. 2008, 41, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Robinson, S.M.; Gupta, A.; Saha, K.; Jiang, Z.; Moyano, D.F.; Sahar, A.; Riley, M.A.; Rotello, V.M. Functional Gold Nanoparticles as Potent Antimicrobial Agents against Multi-Drug-Resistant Bacteria. ACS Nano 2014, 8, 10682–10686. [Google Scholar] [CrossRef] [PubMed]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sambhy, V.; MacBride, M.M.; Peterson, B.R.; Sen, A. Silver Bromide Nanoparticle/Polymer Composites: Dual Action Tunable Antimicrobial Materials. J. Am. Chem. Soc. 2006, 128, 9798–9808. [Google Scholar] [CrossRef] [PubMed]
- Tiller, J.C.; Lee, S.B.; Lewis, K.; Klibanov, A.M. Polymer surfaces derivatized with poly(vinyl-N-hexylpyridinium) kill airborne and waterborne bacteria. Biotechnol. Bioeng. 2002, 79, 465–471. [Google Scholar] [CrossRef]
- Liau, S.Y.; Read, D.C.; Pugh, W.J.; Furr, J.R.; Russell, A.D. Interaction of silver nitrate with readily identifiable groups: Relationship to the antibacterialaction of silver ions. Lett. Appl. Microbiol. 1997, 25, 279–283. [Google Scholar] [CrossRef]
- Choi, O.; Deng, K.K.; Kim, N.-J.; Ross Jr, L.; Surampalli, R.Y.; Hu, Z. The inhibitory effects of silver nanoparticles, silver ions, and silver chloride colloids on microbial growth. Water Res. 2008, 42, 3066–3074. [Google Scholar] [CrossRef]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Song, J.; Kang, H.; Lee, C.; Hwang, S.H.; Jang, J. Aqueous Synthesis of Silver Nanoparticle Embedded Cationic Polymer Nanofibers and Their Antibacterial Activity. ACS Appl. Mater. Interfaces 2011, 4, 460–465. [Google Scholar] [CrossRef]
- Voccia, S.; Ignatova, M.; Jérôme, R.; Jérôme, C. Design of Antibacterial Surfaces by a Combination of Electrochemistry and Controlled Radical Polymerization. Langmuir 2006, 22, 8607–8613. [Google Scholar] [CrossRef]
- Stevens, K.N.; Knetsch, M.L.; Sen, A.; Sambhy, V.; Koole, L.H. Disruption and Activation of Blood Platelets in Contact with an Antimicrobial Composite Coating Consisting of a Pyridinium Polymer and AgBr Nanoparticles. ACS Appl. Mater. Interfaces 2009, 1, 2049–2054. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Lee, D.; Sheng, X.; Cohen, R.E.; Rubner, M.F. Two-Level Antibacterial Coating with Both Release-Killing and Contact-Killing Capabilities. Langmuir 2006, 22, 9820–9823. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Wang, L.; Zhou, L.; Su, Y.; Qiu, F.; Wang, D.; Wu, J.; Zhu, X.; Yan, D. The effect of a branched architecture on the antimicrobial activity of poly(sulfone amines) and poly(sulfone amine)/silver nanocomposites. J. Mater. Chem. 2012, 22, 15227–15234. [Google Scholar] [CrossRef]
- Suleman, N.; Kalhapure, R.S.; Mocktar, C.; Rambharose, S.; Singh, M.; Govender, T. Silver salts of carboxylic acid terminated generation 1 poly (propyl ether imine) (PETIM) dendron and dendrimers as antimicrobial agents against S. aureus and MRSA. RSC Adv. 2015, 5, 34967–34978. [Google Scholar] [CrossRef]
- Peña-González, C.E.; Pedziwiatr-Werbicka, E.; Martín-Pérez, T.; Szewczyk, E.M.; Copa-Patiño, J.L.; Soliveri, J.; Pérez-Serrano, J.; Gómez, R.; Bryszewska, M.; Sánchez-Nieves, J.; et al. Antibacterial and antifungal properties of dendronized silver and gold nanoparticles with cationic carbosilane dendrons. Int. J. Pharm. 2017, 528, 55–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Q.; Zheng, Z.; Wang, B.; Mao, H.; Yan, F. Zinc Ion Coordinated Poly(Ionic Liquid) Antimicrobial Membranes for Wound Healing. ACS Appl. Mater. Interfaces 2017, 9, 14656–14664. [Google Scholar] [CrossRef] [PubMed]
- Sierra-Fernandez, A.; De la Rosa-García, S.C.; Yañez-Macías, R.; Guerrero-Sanchez, C.; Gomez-Villalba, L.S.; Gómez-Cornelio, S.; Rabanal, M.E.; Schubert, U.S.; Fort, R.; Quintana, P. Sol–gel synthesis of Mg(OH)2 and Ca(OH)2 nanoparticles: A comparative study of their antifungal activity in partially quaternized p(DMAEMA) nanocomposite films. J. Sol.-Gel Sci. Technol. 2019, 89, 310–321. [Google Scholar] [CrossRef]
- Banerjee, M.; Mallick, S.; Paul, A.; Chattopadhyay, A.; Ghosh, S.S. Heightened Reactive Oxygen Species Generation in the Antimicrobial Activity of a Three Component Iodinated Chitosan-Silver Nanoparticle Composite. Langmuir 2010, 26, 5901–5908. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A. Potential of chitin/chitosan-bearing materials for uranium recovery: An interdisciplinary review. Carbohydr. Polym. 2011, 84, 54–63. [Google Scholar] [CrossRef]
- Cao, X.L.; Cheng, C.; Ma, Y.L.; Zhao, C.S. Preparation of silver nanoparticles with antimicrobial activities and the researches of their biocompatibilities. J. Mater. Sci. Mater. Med. 2010, 21, 2861–2868. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Hong, S.-I.; Park, H.-M.; Ng, P.K.W. Preparation and Characterization of Chitosan-Based Nanocomposite Films with Antimicrobial Activity. J. Agric. Food Chem. 2006, 54, 5814–5822. [Google Scholar] [CrossRef] [PubMed]
- López-Carballo, G.; Higueras, L.; Gavara, R.; Hernández-Muñoz, P. Silver Ions Release from Antibacterial Chitosan Films Containing in Situ Generated Silver Nanoparticles. J. Agric. Food Chem. 2013, 61, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Krishna Rao, K.S.V.; Ramasubba Reddy, P.; Lee, Y.-I.; Kim, C. Synthesis and characterization of chitosan–PEG–Ag nanocomposites for antimicrobial application. Carbohydr. Polym. 2012, 87, 920–925. [Google Scholar] [CrossRef]
- Chen, K.; Ye, W.; Cai, S.; Huang, L.; Zhong, T.; Chen, L.; Wang, X. Green antimicrobial coating based on quaternised chitosan/organic montmorillonite/Ag NPs nanocomposites. J. Exp. Nanosci. 2016, 11, 1360–1371. [Google Scholar] [CrossRef] [Green Version]
- Regiel-Futyra, A.; Kus-Liśkiewicz, M.; Sebastian, V.; Irusta, S.; Arruebo, M.; Stochel, G.; Kyzioł, A. Development of Noncytotoxic Chitosan–Gold Nanocomposites as Efficient Antibacterial Materials. ACS Appl. Mater. Interfaces 2015, 7, 1087–1099. [Google Scholar] [CrossRef]
- Sibbald, G.; Leaper, D.; Queen, D. Iodine made easy. Wounds Int. 2011, 2, s1–s6. [Google Scholar]
- Mallick, S.; Sharma, S.; Banerjee, M.; Ghosh, S.S.; Chattopadhyay, A.; Paul, A. Iodine-Stabilized Cu Nanoparticle Chitosan Composite for Antibacterial Applications. ACS Appl. Mater. Interfaces 2012, 4, 1313–1323. [Google Scholar] [CrossRef]
- Brasil, M.L.S.; Filgueiras, A.L.; Campos, M.B.; Neves, M.S.L.; Eugenio, M.; Sena, L.A.; Sant’Anna, C.B.; da Silva, V.L.; Diniz, C.G.; Sant’Anna, A.C. Synergism in the Antibacterial Action of Ternary Mixtures Involving Silver Nanoparticles, Chitosan and Antibiotics. J. Braz. Chem. Soc. 2018, 29, 2016–2033. [Google Scholar] [CrossRef]
- Li, L.-H.; Deng, J.-C.; Deng, H.-R.; Liu, Z.-L.; Xin, L. Synthesis and characterization of chitosan/ZnO nanoparticle composite membranes. Carbohydr. Res. 2010, 345, 994–998. [Google Scholar] [CrossRef]
- Li, L.-H.; Deng, J.-C.; Deng, H.-R.; Liu, Z.-L.; Li, X.-L. Preparation, characterization and antimicrobial activities of chitosan/Ag/ZnO blend films. Chem. Eng. J. (Amsterdam, The Netherlands) 2010, 160, 378–382. [Google Scholar] [CrossRef]
- Vimala, K.; Mohan, Y.M.; Sivudu, K.S.; Varaprasad, K.; Ravindra, S.; Reddy, N.N.; Padma, Y.; Sreedhar, B.; MohanaRaju, K. Fabrication of porous chitosan films impregnated with silver nanoparticles: A facile approach for superior antibacterial application. Coll. Surf. B Biointerfaces 2010, 76, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Fouda, M.M.G.; El-Aassar, M.R.; Al-Deyab, S.S. Antimicrobial activity of carboxymethyl chitosan/polyethylene oxide nanofibers embedded silver nanoparticles. Carbohydr. Polym. 2013, 92, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Kim, H.-I. Characterization and antibacterial properties of genipin-crosslinked chitosan/poly(ethylene glycol)/ZnO/Ag nanocomposites. Carbohydr. Polym. 2012, 89, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Ray Chowdhuri, A.; Tripathy, S.; Chandra, S.; Roy, S.; Sahu, S.K. A ZnO decorated chitosan-graphene oxide nanocomposite shows significantly enhanced antimicrobial activity with ROS generation. RSC Adv. 2015, 5, 49420–49428. [Google Scholar] [CrossRef]
- Busila, M.; Musat, V.; Textor, T.; Mahltig, B. Synthesis and characterization of antimicrobial textile finishing based on Ag:ZnO nanoparticles/chitosan biocomposites. RSC Adv. 2015, 5, 21562–21571. [Google Scholar] [CrossRef]
- Chen, S.; Guo, Y.; Chen, S.; Yu, H.; Ge, Z.; Zhang, X.; Zhang, P.; Tang, J. Facile preparation and synergistic antibacterial effect of three-component Cu/TiO2/CS nanoparticles. J. Mater. Chem. 2012, 22, 9092–9099. [Google Scholar] [CrossRef]
- Liu, B.; Shen, S.; Luo, J.; Wang, X.; Sun, R. One-pot green synthesis and antimicrobial activity of exfoliated Ag NP-loaded quaternized chitosan/clay nanocomposites. RSC Adv. 2013, 3, 9714–9722. [Google Scholar] [CrossRef]
- Žalnėravičius, R.; Paškevičius, A.; Kurtinaitiene, M.; Jagminas, A. Size-dependent antimicrobial properties of the cobalt ferrite nanoparticles. J. Nanoparticle Res. 2016, 18, 300. [Google Scholar] [CrossRef]
- Chen, C.; Chen, L.; Yi, Y.; Chen, C.; Wu, L.-F.; Song, T. Killing of Staphylococcus aureus via Magnetic Hyperthermia Mediated by Magnetotactic Bacteria. Appl. Environ. Microbiol. 2016, 82, 2219–2226. [Google Scholar] [CrossRef] [Green Version]
- Wust, P.; Hildebrandt, B.; Sreenivasa, G.; Rau, B.; Gellermann, J.; Riess, H.; Felix, R.; Schlag, P.M. Hyperthermia in combined treatment of cancer. Lancet Oncol. 2002, 3, 487–497. [Google Scholar] [CrossRef]
- Kim, M.-H.; Yamayoshi, I.; Mathew, S.; Lin, H.; Nayfach, J.; Simon, S.I. Magnetic Nanoparticle Targeted Hyperthermia of Cutaneous Staphylococcus aureus Infection. Ann. Biomed. Eng. 2013, 41, 598–609. [Google Scholar] [CrossRef] [PubMed]
- Pu, L.; Xu, J.; Sun, Y.; Fang, Z.; Chan-Park, M.B.; Duan, H. Cationic polycarbonate-grafted superparamagnetic nanoparticles with synergistic dual-modality antimicrobial activity. Biomater. Sci. 2016, 4, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Cha, C.; Shin, S.R.; Annabi, N.; Dokmeci, M.R.; Khademhosseini, A. Carbon-Based Nanomaterials: Multifunctional Materials for Biomedical Engineering. ACS Nano 2013, 7, 2891–2897. [Google Scholar] [CrossRef] [PubMed]
- Maleki Dizaj, S.; Mennati, A.; Jafari, S.; Khezri, K.; Adibkia, K. Antimicrobial Activity of Carbon-Based Nanoparticles. Adv. Pharm Bull. 2015, 5, 19–23. [Google Scholar]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Saito, R.; Dresselhaus, G.; Dresselhaus, M.S. Physical Properties of Carbon Nanotubes; World Scientific Publishing Co.: Singapore, 1998; p. 272. [Google Scholar]
- Kang, S.; Herzberg, M.; Rodrigues, D.F.; Elimelech, M. Antibacterial Effects of Carbon Nanotubes: Size Does Matter! Langmuir 2008, 24, 6409–6413. [Google Scholar] [CrossRef]
- Pangule, R.C.; Brooks, S.J.; Dinu, C.Z.; Bale, S.S.; Salmon, S.L.; Zhu, G.; Metzger, D.W.; Kane, R.S.; Dordick, J.S. Antistaphylococcal Nanocomposite Films Based on Enzyme−Nanotube Conjugates. ACS Nano 2010, 4, 3993–4000. [Google Scholar] [CrossRef]
- Kang, S.; Pinault, M.; Pfefferle, L.D.; Elimelech, M. Single-Walled Carbon Nanotubes Exhibit Strong Antimicrobial Activity. Langmuir 2007, 23, 8670–8673. [Google Scholar] [CrossRef]
- Liu, S.; Wei, L.; Hao, L.; Fang, N.; Chang, M.W.; Xu, R.; Yang, Y.; Chen, Y. Sharper and Faster “Nano Darts” Kill More Bacteria: A Study of Antibacterial Activity of Individually Dispersed Pristine Single-Walled Carbon Nanotube. ACS Nano 2009, 3, 3891–3902. [Google Scholar] [CrossRef]
- Kang, S.; Mauter, M.S.; Elimelech, M. Physicochemical Determinants of Multiwalled Carbon Nanotube Bacterial Cytotoxicity. Environ. Sci. Technol. 2008, 42, 7528–7534. [Google Scholar] [CrossRef]
- Joo, Y.T.; Jung, K.H.; Kim, M.J.; Kim, Y. Preparation of antibacterial PDMAEMA-functionalized multiwalled carbon nanotube via atom transfer radical polymerization. J. Appl. Polym. Sci. 2013, 127, 1508–1518. [Google Scholar] [CrossRef]
- Murugan, E.; Vimala, G. Effective functionalization of multiwalled carbon nanotube with amphiphilic poly(propyleneimine) dendrimer carrying silver nanoparticles for better dispersability and antimicrobial activity. J. Coll. Interface Sci. 2011, 357, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Vadukumpully, S.; Paul, J.; Valiyaveettil, S. Cationic surfactant mediated exfoliation of graphite into graphene flakes. Carbon N. Y. 2009, 47, 3288–3294. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Won Suk, J.; Potts, J.R.; Ruoff, R.S. Graphene and Graphene Oxide: Synthesis, Properties, and Applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef]
- Liu, S.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.; Kong, J.; Chen, Y. Antibacterial Activity of Graphite, Graphite Oxide, Graphene Oxide, and Reduced Graphene Oxide: Membrane and Oxidative Stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Veerapandian, M.; Zhang, L.-H.; Yun, K.; Kim, S.J. Antibacterial Efficiency of Graphene Nanosheets against Pathogenic Bacteria via Lipid Peroxidation. J. Phys. Chem. C 2012, 116, 17280–17287. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E. Toxicity of Graphene and Graphene Oxide Nanowalls Against Bacteria. ACS Nano 2010, 4, 5731–5736. [Google Scholar] [CrossRef]
- Chen, J.; Peng, H.; Wang, X.; Shao, F.; Yuan, Z.; Han, H. Graphene oxide exhibits broad-spectrum antimicrobial activity against bacterial phytopathogens and fungal conidia by intertwining and membrane perturbation. Nanoscale 2014, 6, 1879–1889. [Google Scholar] [CrossRef]
- Zou, X.; Zhang, L.; Wang, Z.; Luo, Y. Mechanisms of the Antimicrobial Activities of Graphene Materials. J. Am. Chem. Soc. 2016, 138, 2064–2077. [Google Scholar] [CrossRef]
- Liu, T.; Liu, Y.; Liu, M.; Wang, Y.; He, W.; Shi, G.; Hu, X.; Zhan, R.; Luo, G.; Xing, M.; et al. Synthesis of graphene oxide-quaternary ammonium nanocomposite with synergistic antibacterial activity to promote infected wound healing. Burn. Trauma 2018, 6, 16. [Google Scholar] [CrossRef]
- Cai, X.; Tan, S.; Lin, M.; Xie, A.; Mai, W.; Zhang, X.; Lin, Z.; Wu, T.; Liu, Y. Synergistic Antibacterial Brilliant Blue/Reduced Graphene Oxide/Quaternary Phosphonium Salt Composite with Excellent Water Solubility and Specific Targeting Capability. Langmuir 2011, 27, 7828–7835. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Liu, B.; Wang, J.; Zhang, S.; Lin, Q.; Gong, P.; Ma, L.; Yang, S. A Novel Wound Dressing Based on Ag/Graphene Polymer Hydrogel: Effectively Kill Bacteria and Accelerate Wound Healing. Adv. Funct. Mater. 2014, 24, 3933–3943. [Google Scholar] [CrossRef]
- Kanchanapally, R.; Viraka Nellore, B.P.; Sinha, S.S.; Pedraza, F.; Jones, S.J.; Pramanik, A.; Chavva, S.R.; Tchounwou, C.; Shi, Y.; Vangara, A.; et al. Antimicrobial peptide-conjugated graphene oxide membrane for efficient removal and effective killing of multiple drug resistant bacteria. RSC Adv. 2015, 5, 18881–18887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sundar, K.; Harikarthick, V.; Swarna Karthika, V.; Ravindran, A. Preparation of Chitosan-Graphene Oxide Nanocomposite and Evaluation of Its Antimicrobial Activity. J. Bionanosci. 2014, 8, 2017–2212. [Google Scholar] [CrossRef]
- Piddock, L.J. V The crisis of no new antibiotics—what is the way forward. Lancet Infect. Dis. 2012, 12, 249–253. [Google Scholar] [CrossRef]
- Appelbaum, P.C.; Hunter, P.A. The fluoroquinolone antibacterials: Past, present and future perspectives. Int. J. Antimicrob. Agents 2000, 16, 5–15. [Google Scholar] [CrossRef]
- Decker, E.-M.; Von Ohle, C.; Weiger, R.; Wiech, I.; Brecx, M. A synergistic chlorhexidine/chitosan combination for improved antiplaque strategies. J. Periodontal Res. 2005, 40, 373–377. [Google Scholar] [CrossRef]
- Tin, S.; Sakharkar, K.R.; Lim, C.S.; Sakharkar, M.K. Activity of Chitosans in combination with antibiotics in Pseudomonas aeruginosa. Int. J. Biol. Sci. 2009, 5, 153–160. [Google Scholar] [CrossRef]
- Tré-Hardy, M.; Vanderbist, F.; Traore, H.; Devleeschouwer, M.J. In vitro activity of antibiotic combinations against Pseudomonas aeruginosa biofilm and planktonic cultures. Int. J. Antimicrob. Agents 2008, 31, 329–336. [Google Scholar] [CrossRef]
- He, M.; Zhou, Y.; Xiao, H.; Lu, P. Amphiphilic cationic copolymers with ciprofloxacin: Preparation and antimicrobial activities. N. J. Chem. 2016, 40, 1354–1364. [Google Scholar] [CrossRef]
- He, M.; Xiao, H.; Zhou, Y.; Lu, P. Synthesis, characterization and antimicrobial activities of water-soluble amphiphilic copolymers containing ciprofloxacin and quaternary ammonium salts. J. Mater. Chem. B 2015, 3, 3704–3713. [Google Scholar] [CrossRef]
- Siala, W.; Van Bambeke, F.; Taresco, V.; Piozzi, A.; Francolini, I. Synergistic activity between an antimicrobial polyacrylamide and daptomycin versus Staphylococcus aureus biofilm. Pathog. Dis. 2016, 74, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Felczak, A.; Zawadzka, K.; Wronska, N.; Janaszewska, A.; Klajnert, B.; Bryszewska, M.; Appelhans, D.; Voit, B.; Lisowska, K. Enhancement of antimicrobial activity by co-administration of poly(propylene imine) dendrimers and nadifloxacin. N. J. Chem. 2013, 37, 4156–4162. [Google Scholar] [CrossRef]
- Wrońska, N.; Felczak, A.; Zawadzka, K.; Poszepczyńska, M.; Różalska, S.; Bryszewska, M.; Appelhans, D.; Lisowska, K. Poly(Propylene Imine) Dendrimers and Amoxicillin as Dual-Action Antibacterial Agents. Molecules 2015, 20, 19330–19342. [Google Scholar] [CrossRef] [Green Version]
- Karthikeyan, R.; Karempudi, B.; Rasheed, S.; Vijayaraj, P. Dendritic architechture for the delivery of antibacterial agent against resistant producing strains. Cent. Euro. J. Exp. Bio 2012, 1, 45–48. [Google Scholar]
- Mishra, M.K.; Kotta, K.; Hali, M.; Wykes, S.; Gerard, H.C.; Hudson, A.P.; Whittum-Hudson, J.A.; Kannan, R.M. PAMAM dendrimer-azithromycin conjugate nanodevices for the treatment of Chlamydia trachomatis infections. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 935–944. [Google Scholar] [CrossRef]
- Khalil, H.; Chen, T.; Riffon, R.; Wang, R.; Wang, Z. Synergy between Polyethylenimine and Different Families of Antibiotics against a Resistant Clinical Isolate of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2008, 52, 1635–1641. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Chen, Y.P.; Miller, K.P.; Ganewatta, M.S.; Bam, M.; Yan, Y.; Nagarkatti, M.; Decho, A.W.; Tang, C. Antimicrobial Metallopolymers and Their Bioconjugates with Conventional Antibiotics against Multidrug-Resistant Bacteria. J. Am. Chem. Soc. 2014, 136, 4873–4876. [Google Scholar] [CrossRef]
- Yang, P.; Bam, M.; Pageni, P.; Zhu, T.; Chen, Y.P.; Nagarkatti, M.; Decho, A.W.; Tang, C. Trio Act of Boronolectin with Antibiotic-Metal Complexed Macromolecules toward Broad-Spectrum Antimicrobial Efficacy. ACS Infect. Dis. 2017, 3, 845–853. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, T.-K.; Selvanayagam, R.; Ho, K.K.K.; Chen, R.; Kutty, S.K.; Rice, S.A.; Kumar, N.; Barraud, N.; Duong, H.T.T.; Boyer, C. Co-delivery of nitric oxide and antibiotic using polymeric nanoparticles. Chem. Sci. 2016, 7, 1016–1027. [Google Scholar] [CrossRef]
- Ren, H.; Wu, J.; Colletta, A.; Meyerhoff, M.E.; Xi, C. Efficient Eradication of Mature Pseudomonas aeruginosa Biofilm via Controlled Delivery of Nitric Oxide Combined with Antimicrobial Peptide and Antibiotics. Front. Microbiol. 2016, 7, 1260. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Zhang, J.; Yang, J.; Du, L.; Geng, H.; Cheng, Y. Conjugated Polymers Act Synergistically with Antibiotics to Combat Bacterial Drug Resistance. ACS Appl. Mater. Interfaces 2017, 9, 18512–18520. [Google Scholar] [CrossRef] [PubMed]




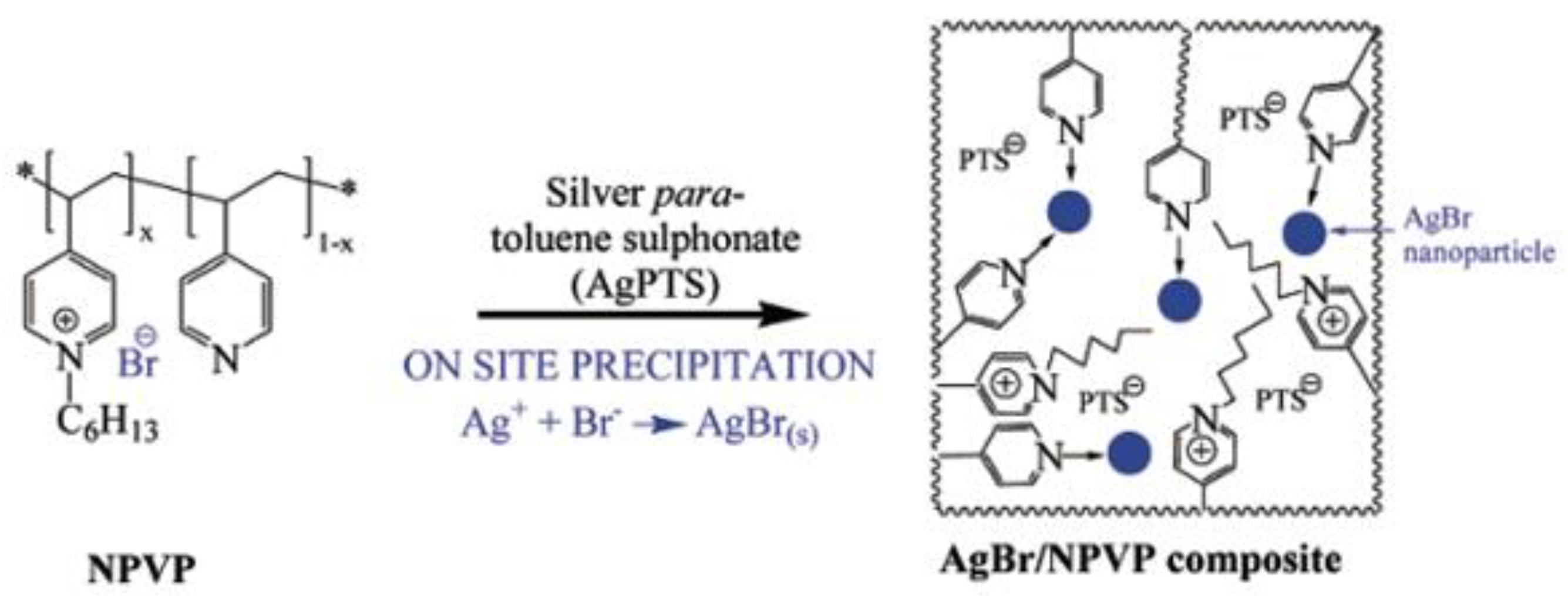
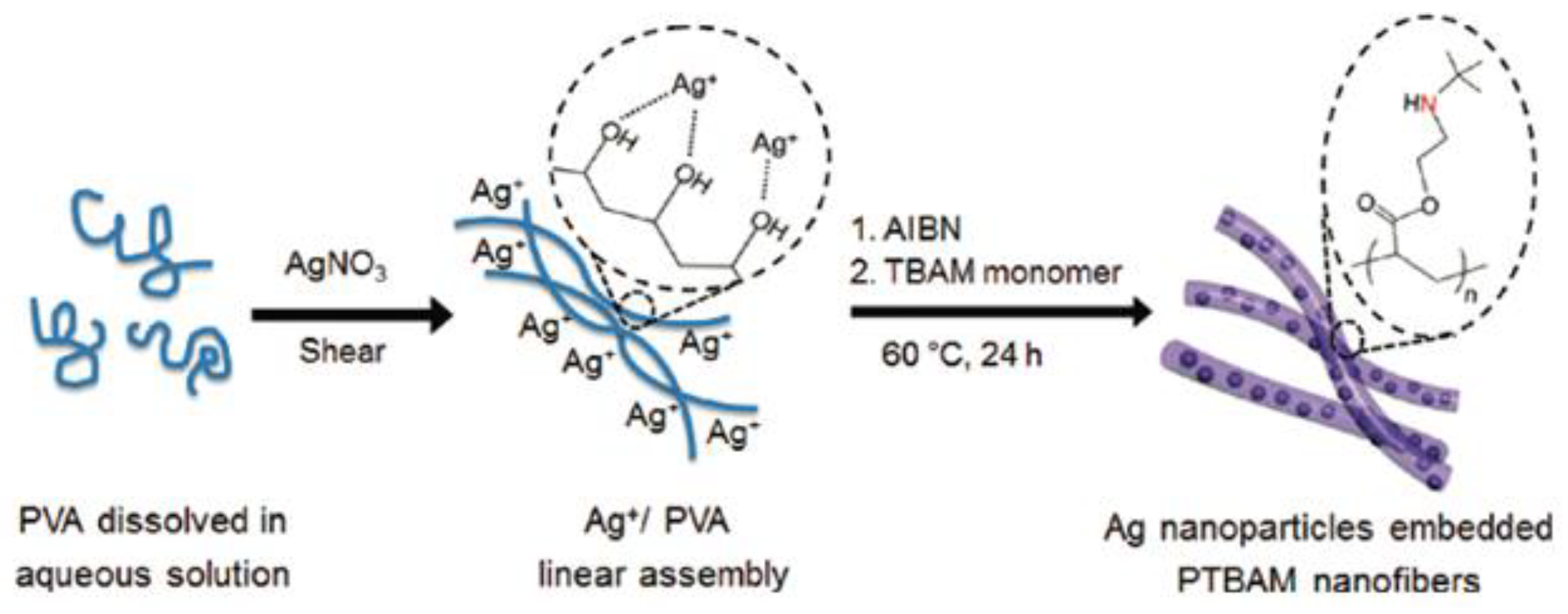


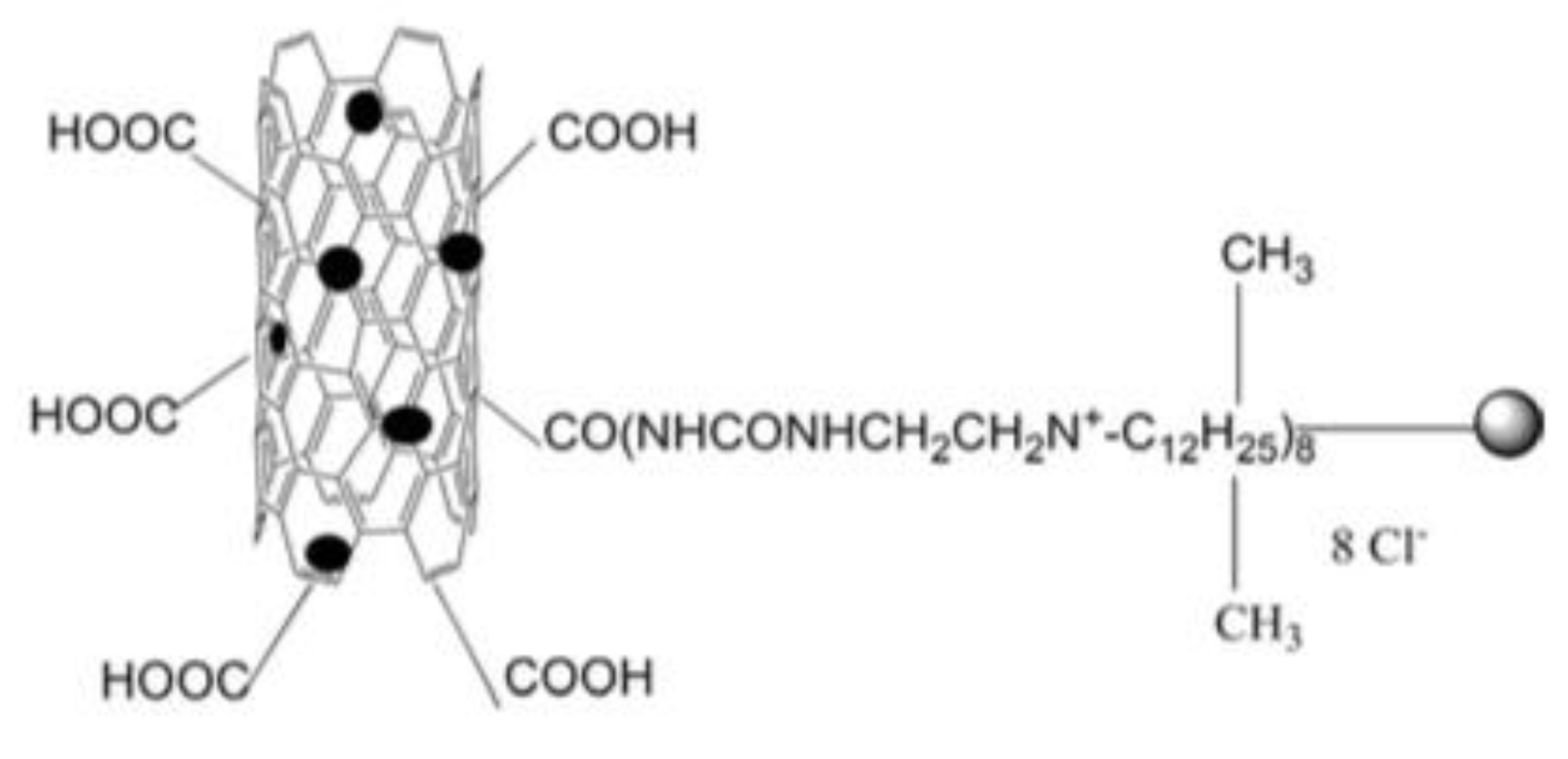

| Schematic Representation of the Antimicrobial Polymer | NP/Salt | Microorganism Tested | Synergistic Effect | Ref |
|---|---|---|---|---|
 | AgBr (10–70 nm) | E. coli B. cereus | Membrane disrupting of the cationic polymer. Long lasting action without depletion of Ag+ ion. The dual system | [94,101] |
 | Ag, Cu | S. aureus P. aeruginosa B. subtilis E. coli | increased the killing rate of bacteria and kept activity for a longer time in comparison with AgBr alone. | [82] |
 | Ag (≈5 nm) Au (≈8 nm) | S. aureus P. aeruginosa | Positive charges and alkyl chains act together to damage the bacterial structure. This fact increases cell permeability allowing AgNps to penetrate and inhibit the function of enzymes and proteins. | [83,88] |
 | Ag | E. coli S. epidermis | Release (Ag+) and contact killing mechanisms (QAS). Long sustainability. | [102] |
 | Ag (8 to 15 nm) | A. niger | Higher branched degree of polymers produces smaller AgNps with better diffusion and interaction, increasing the antimicrobial performance. | [103] |
 | Ag salts | S. aureus MRSA | Dendrimer acted as a template to load silver salts allowing the high local concentration of exposed silver ions in the periphery. | [104] |
 | Ag (1.5 nm) | S. aureus E. coli P. aeruginosa S. hemolyticus C. albicans | Peripheral +NMe3 groups in combination with biocidal silver cations. | [105] |
 | Zn ions | S. aureus E coli C. albicans | Interactions between cations of poly(ionic liquid) and cell wall, which boost the cell membrane permeability causing lysis of the cells. Zn2+ can produce reactive ROS in cells leading to the growth inhibition and death of bacteria. | [106] |
 | Mg(OH)2, Ca(OH)2 (30 nm) | A.niger P. oxalicum | Cationic copolymer provides additional charges on the NPs surfaces promoting affinity to bind to fungal cells, thus improving their interaction with the negatively charged microbial cell surface. | [107] |
| System | Nanoparticle | Microorganism Tested | Synergistic Effect | Ref |
|---|---|---|---|---|
| Porous CS films | Ag (≈12 nm) | E. coli, S. aureus P. aeruginosa, MRSA | The presence of hundreds of porous enables formation of smaller AgNPs, which are more effective than longer. Besides CS absorbs a large amount of water and releases Ag more efficient than chitosan without porous. | [121] |
| Carboxymethyl CS/polyethylene oxide nanofibers (CMCTS/PEO) | Ag (12 to 18 nm) | E. coli, S. aureus P. aeruginosa, C. albicans | The fibrous structure of nanofibers allowed to increase the silver load. | [122] |
| Crosslinked CS/polyethylene glycol nanocomposite films | ZnO Ag < 100 nm | E. coli, S. aureus P. aeruginosa, B. subtilis | Membrane disrupting of the cationic polymer. Ag and ZnO enhanced antibacterial property due to the photocatalysis and metal release process. Generation of active free radicals. | [123] |
| Nancomposite GO-CS/ZnO | GO ZnO | E. coli, S. aureus | GO-ZnO induce ROS production that causes oxidative damage. The interaction bacteria with composite and ZnO-NPs increase its permeability and generate active superoxide ions (O2−), which can react with the peptide linkages in the cell wall of bacteria and thus disrupt. | [124] |
| CS coatings applied on cotton and cotton/polyester CS/Ag, CS/ZnO, CS/Ag-ZnO | Ag (3 to 5 nm) ZnO Ag-doped ZnO (10 to 35 nm) | E. coli, S. aureus | AgNPs disturbs the permeability, respiration and cell division. ZnO NPs produce ROS. Under light conditions, Ag improved the charge transfer, reducing the chance of electron–hole pairs to recombine and promoting the generation of perhydroxyl radicals and other potent oxidizing radicals. | [125] |
| CS NPs | Cu, TiO2 ≈10 nm | E. coli, S. aureus | Negatively charged TiO2 NPs acts as a copper ion carrier, and its surface can absorb positively charged copper ions. Cu in combination with TiO2 can increase the amount of copper in bacteria and subsequently enhances antimicrobial activity. | [126] |
| Quaternized CS-clay (MMT) based nanocomposites | Ag (≈26 nm) | E. coli, S. aureus, P. aeruginosa, B. subtilis | Exfoliated MMT with a large specific surface area adsorbs and fixes microorganisms. QAS disrupt cell membrane allowing AgNPs infiltrate and react with compounds in the cell wall. | [127] |
| Schematic Representation of the Antimicrobial Polymer | Antibiotic | Microorganism Tested | Synergistic Effect | Ref |
|---|---|---|---|---|
 | Ciprofloxacin (CPF) | E. coli | Integrity of the cell membrane was disrupted by hydrophobic moieties (in an optimal concentration). CPF inhibits the activity of the bacterial DNA gyrase, which leads to bacterial cell death. | [161] |
 | Ciprofloxacin (CPF) | E. coli | [162] | |
 | Polypeptide antibiotics: Polymyxin B Polymyxin R | E. coli | Combination of cationic conjugated polymers (CCPs) with polypeptide antibiotics facilitates and accelerates the rupture and collapse of bacterial membranes. | [173] |
 | Penicillin-G Amoxicillin Ampicillin Cefazolin | MRSA | Adsorption of metallopolymer to the negatively charged MRSA surface which promotes damage in the cell walls and at the same time allows the release of complexed antibiotic. | [169] |
 | Penicillin-G | E. coli P. aeruginosa P. vulgaris | Phenylboronic acid binds to peptide-glycan via boron-polyol based boronolectin chemistry, cationic cobalto-cenium moiety interact with negatively charged cell membranes and antibiotic is reinstated with enhanced vitality to attack bacteria | [170] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yañez-Macías, R.; Muñoz-Bonilla, A.; De Jesús-Tellez, M.A.; Maldonado-Textle, H.; Guerrero-Sánchez, C.; Schubert, U.S.; Guerrero-Santos, R. Combinations of Antimicrobial Polymers with Nanomaterials and Bioactives to Improve Biocidal Therapies. Polymers 2019, 11, 1789. https://doi.org/10.3390/polym11111789
Yañez-Macías R, Muñoz-Bonilla A, De Jesús-Tellez MA, Maldonado-Textle H, Guerrero-Sánchez C, Schubert US, Guerrero-Santos R. Combinations of Antimicrobial Polymers with Nanomaterials and Bioactives to Improve Biocidal Therapies. Polymers. 2019; 11(11):1789. https://doi.org/10.3390/polym11111789
Chicago/Turabian StyleYañez-Macías, Roberto, Alexandra Muñoz-Bonilla, Marco A. De Jesús-Tellez, Hortensia Maldonado-Textle, Carlos Guerrero-Sánchez, Ulrich S. Schubert, and Ramiro Guerrero-Santos. 2019. "Combinations of Antimicrobial Polymers with Nanomaterials and Bioactives to Improve Biocidal Therapies" Polymers 11, no. 11: 1789. https://doi.org/10.3390/polym11111789
APA StyleYañez-Macías, R., Muñoz-Bonilla, A., De Jesús-Tellez, M. A., Maldonado-Textle, H., Guerrero-Sánchez, C., Schubert, U. S., & Guerrero-Santos, R. (2019). Combinations of Antimicrobial Polymers with Nanomaterials and Bioactives to Improve Biocidal Therapies. Polymers, 11(11), 1789. https://doi.org/10.3390/polym11111789







