Effects of a Phosphorus Flame Retardant System on the Mechanical and Fire Behavior of Microcellular ABS
Abstract
1. Introduction
2. Materials and Methods
3. Results
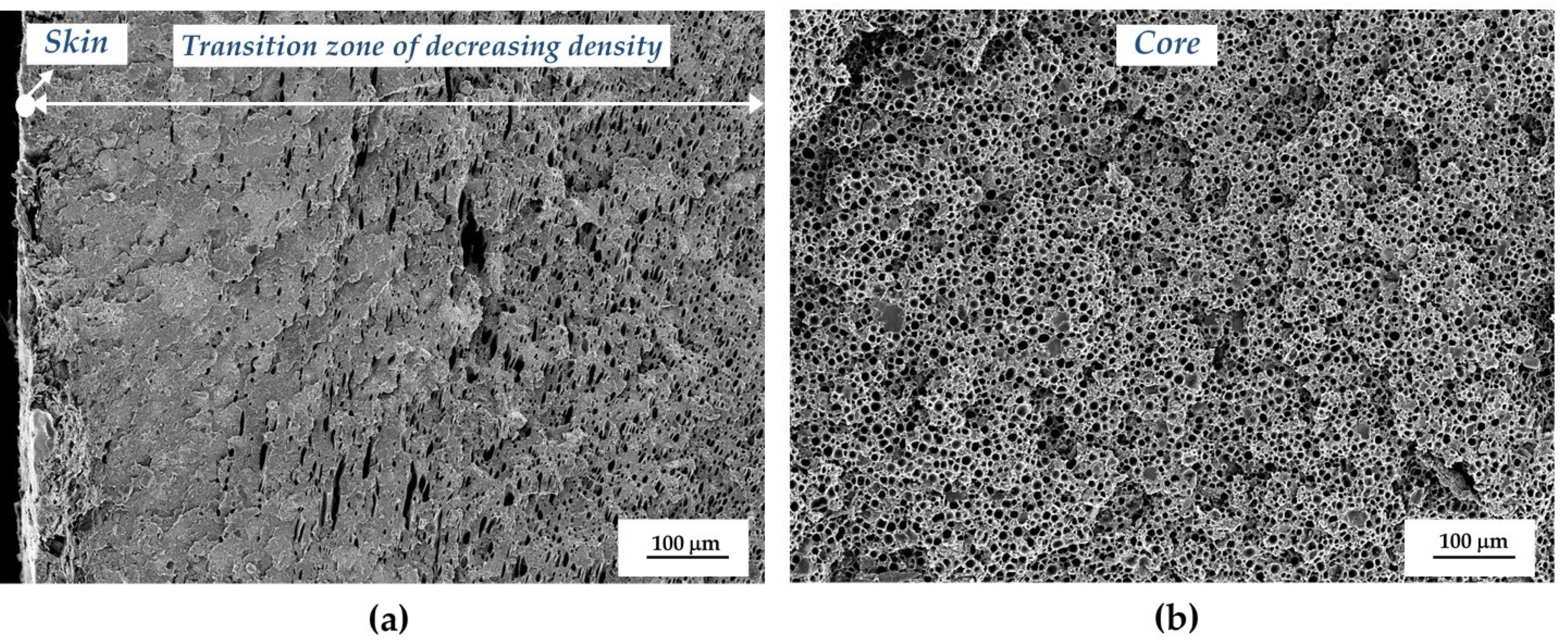
3.1. Structure of the Microcellular Parts
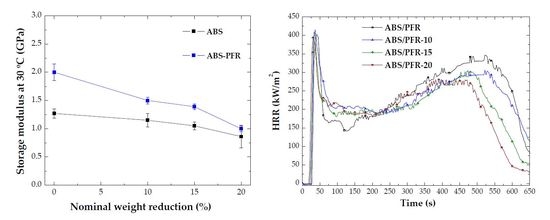
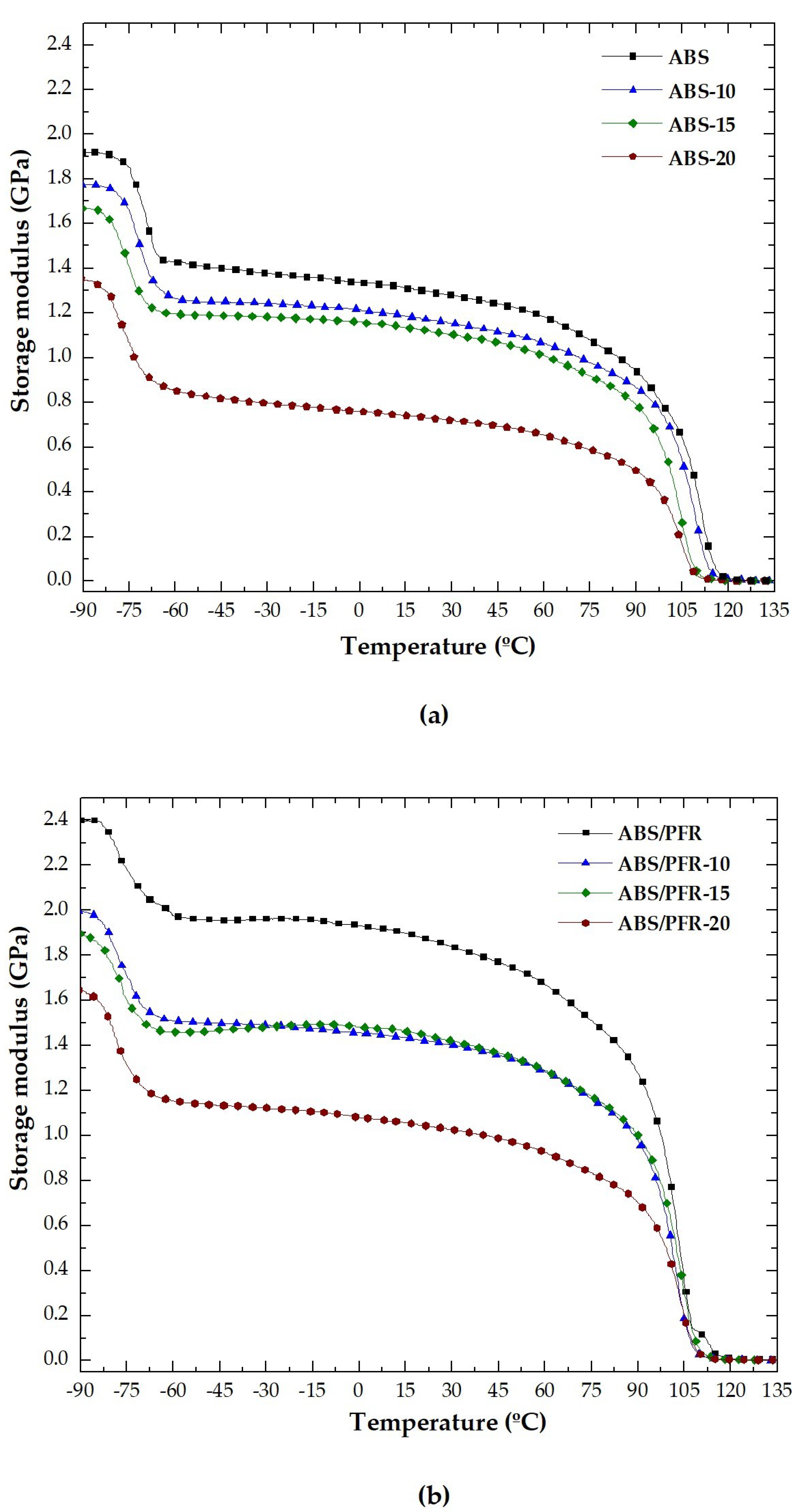
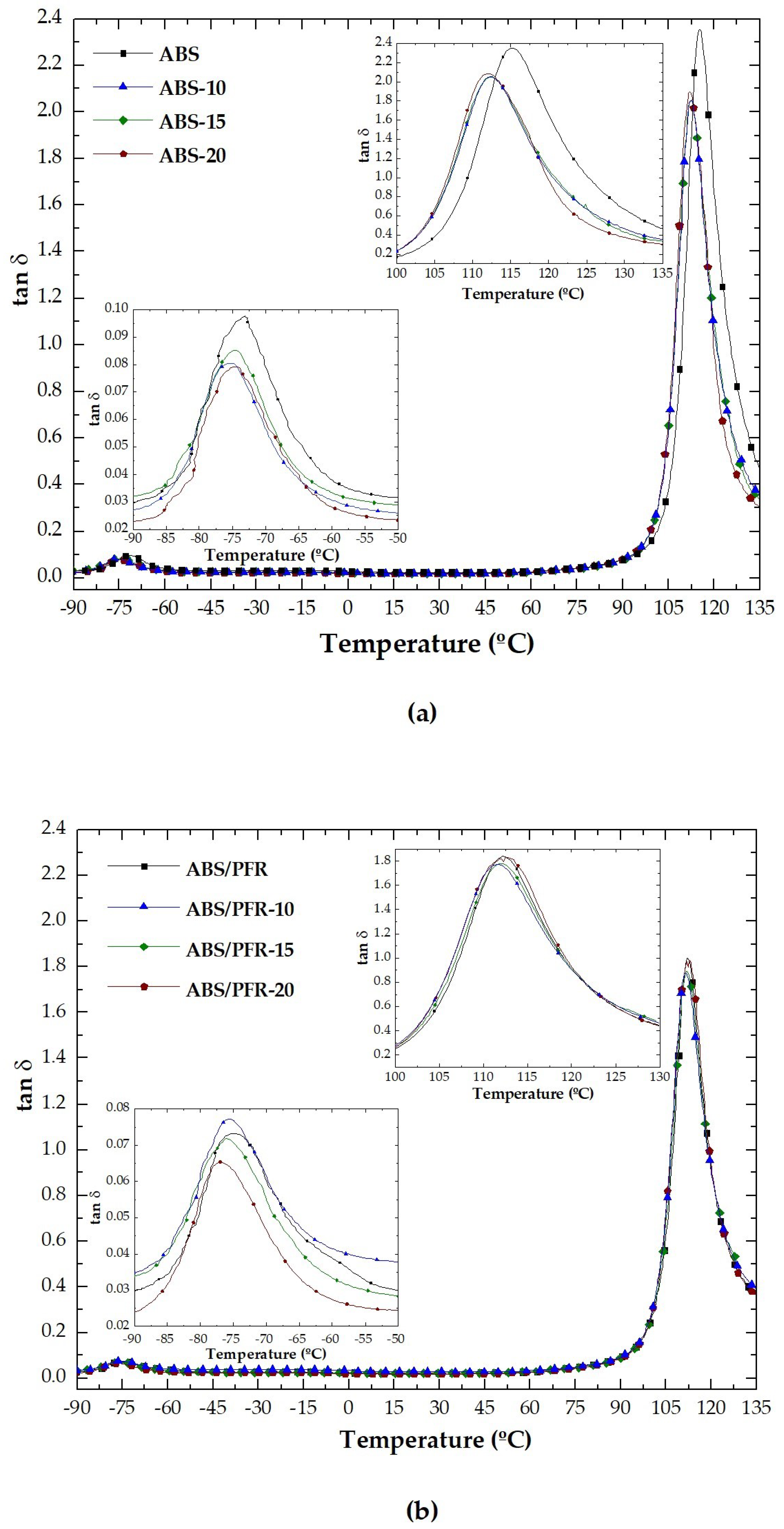
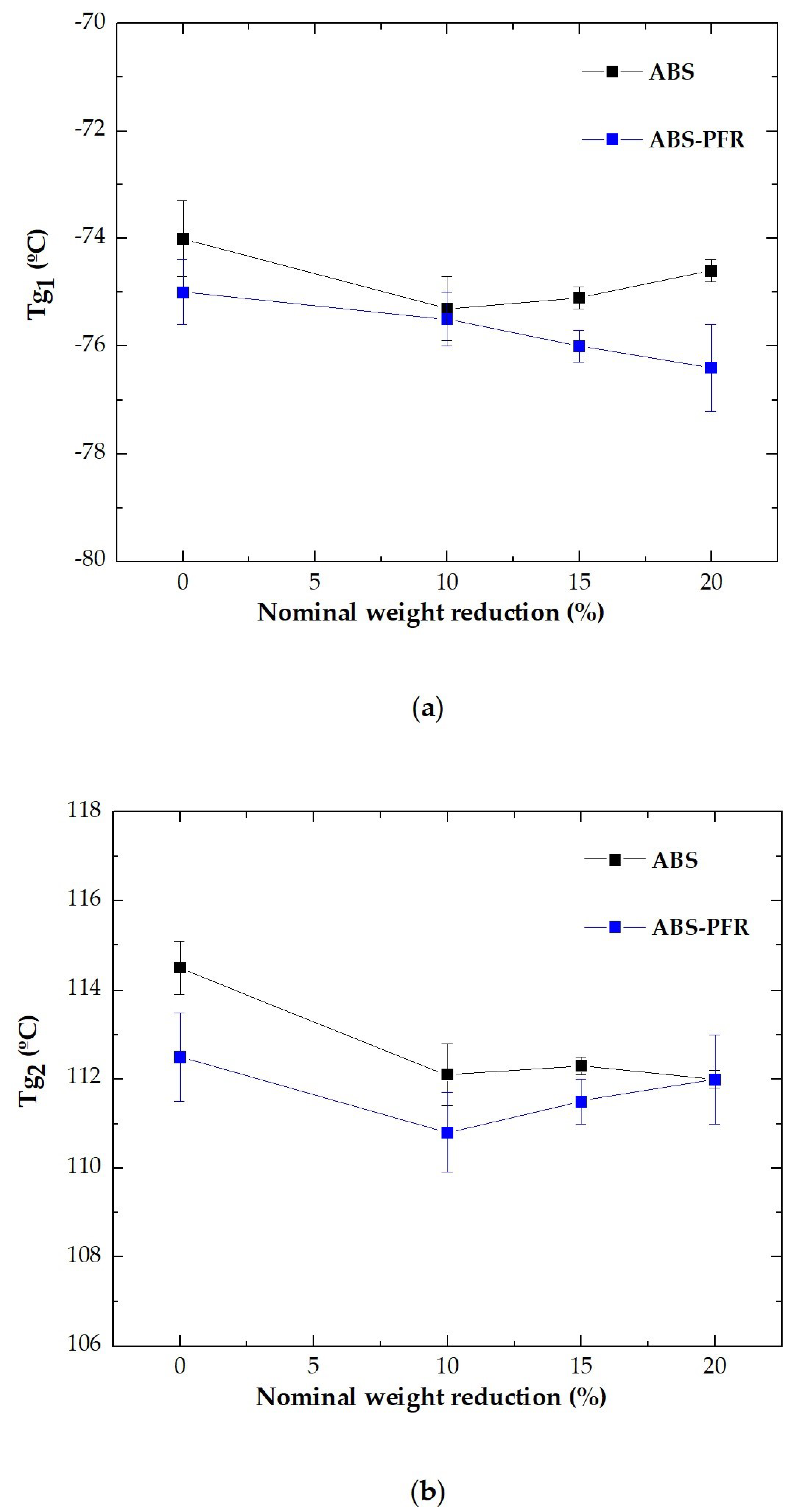
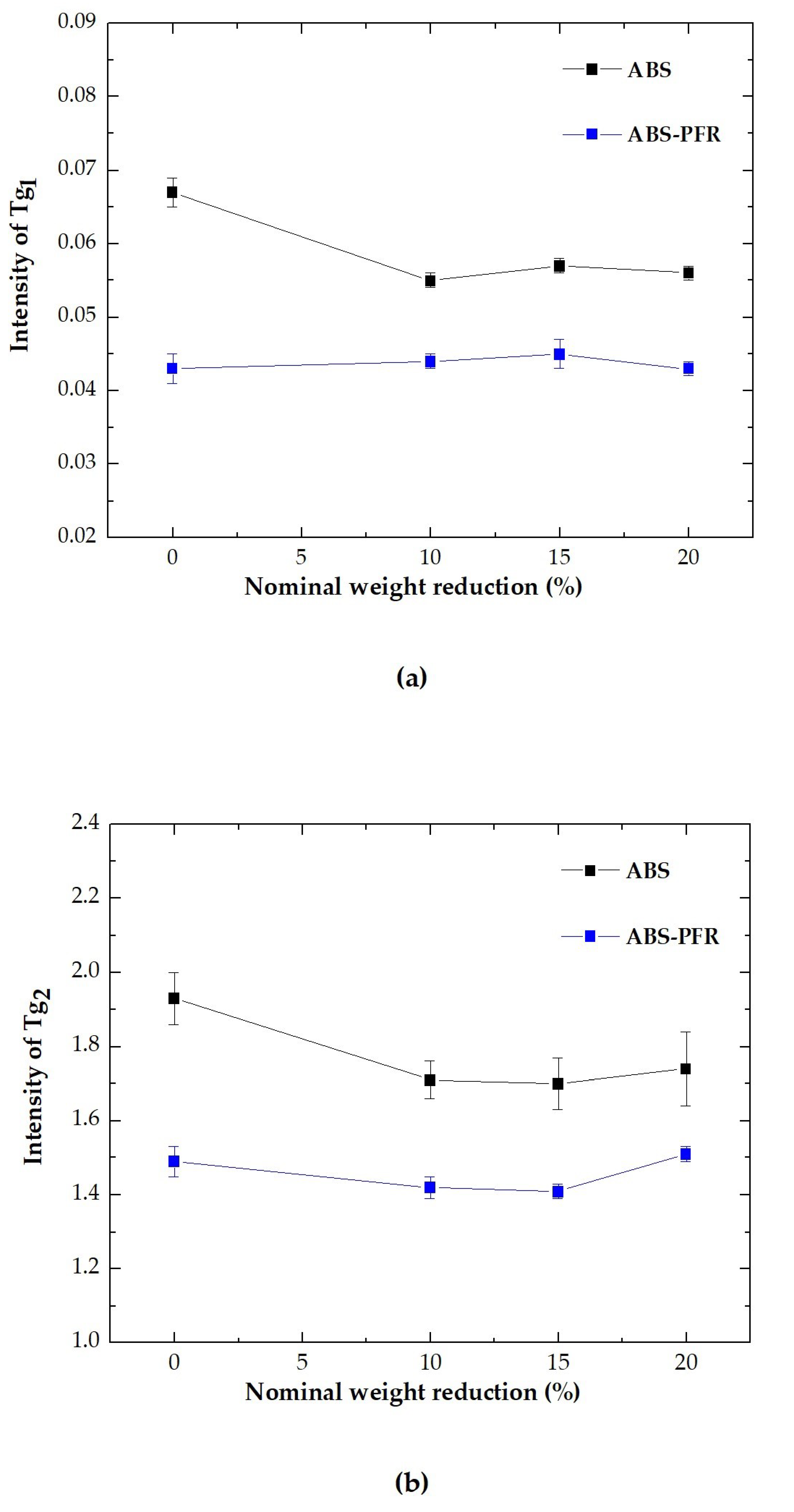
3.2. Dynamic-Mechanical-Thermal Behavior
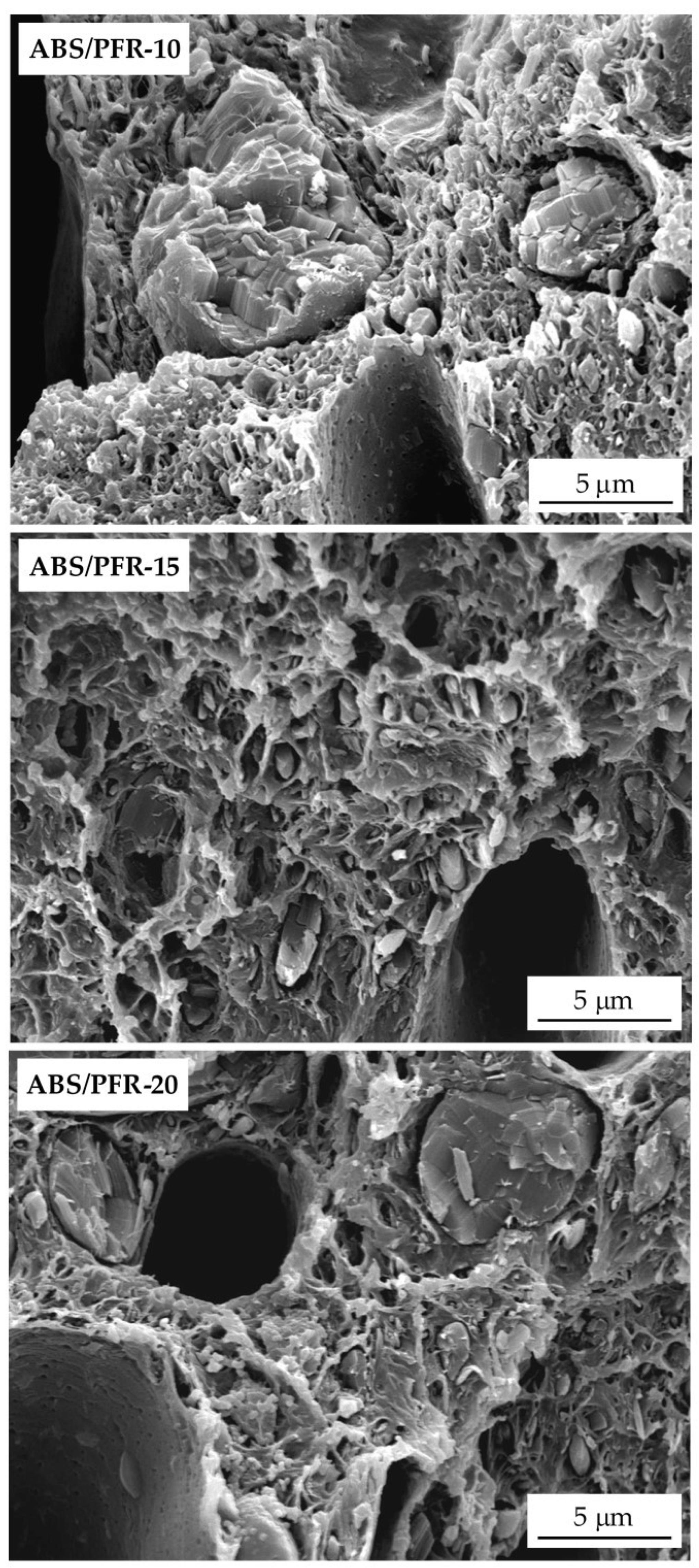
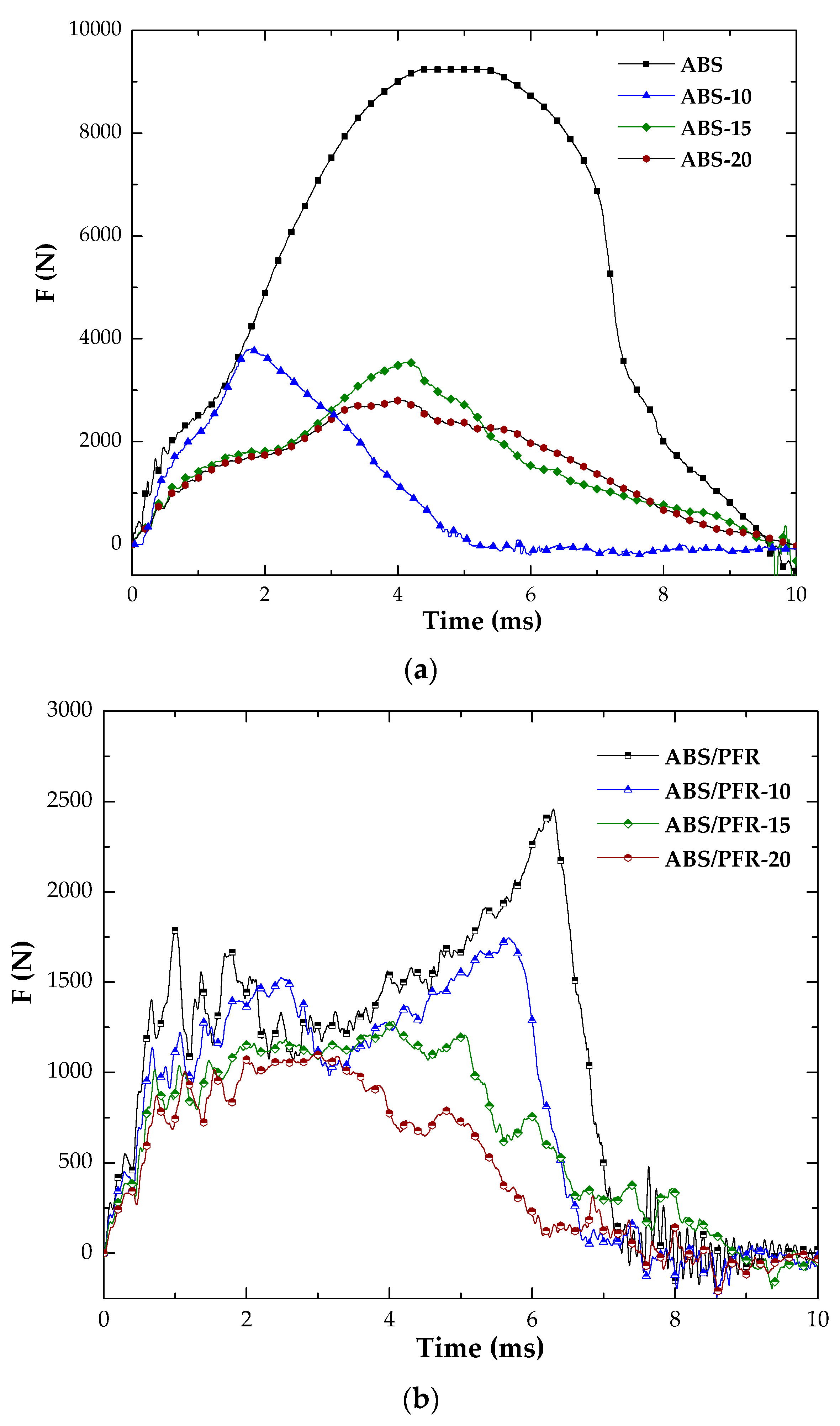
3.3. Fracture Behavior
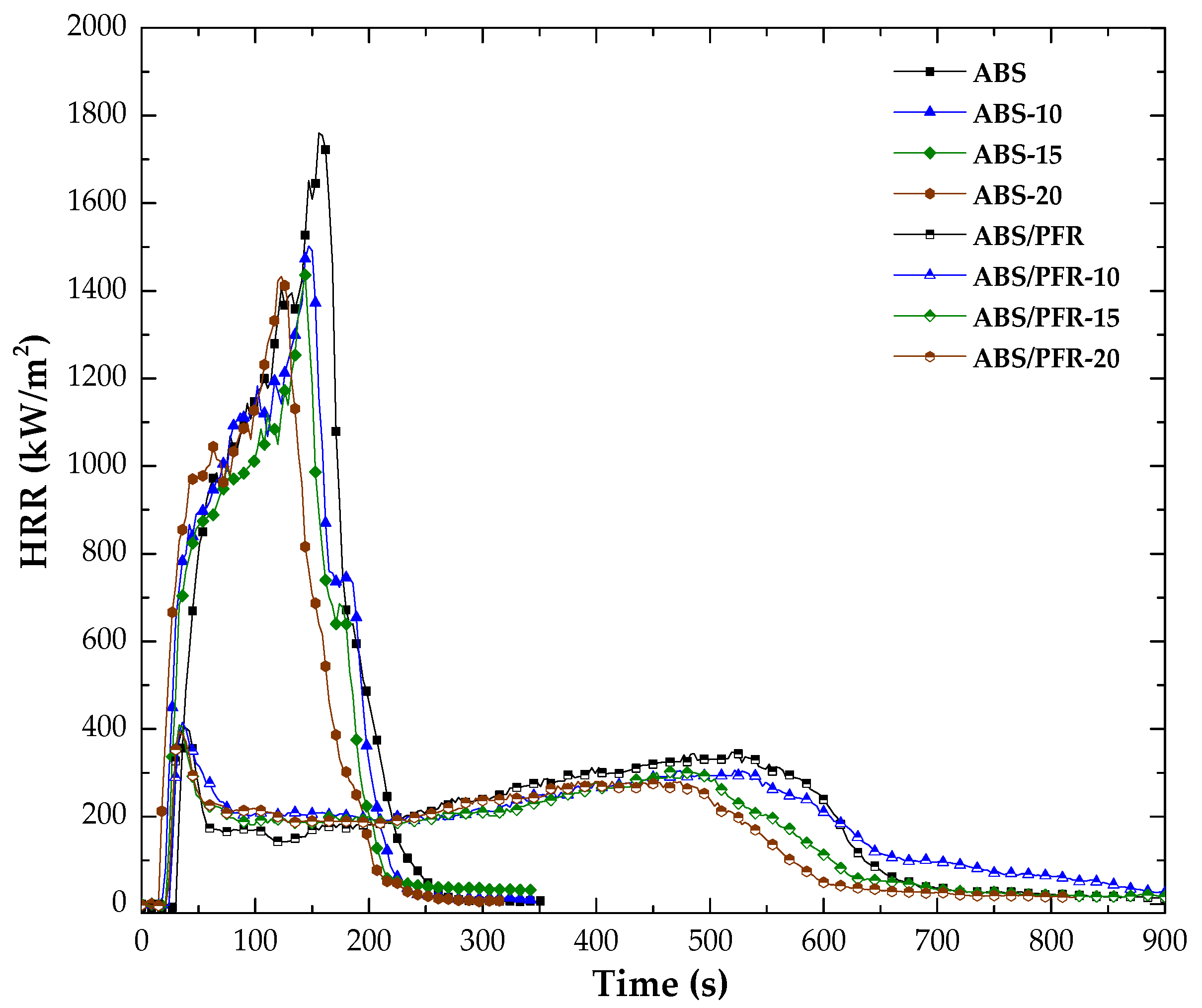
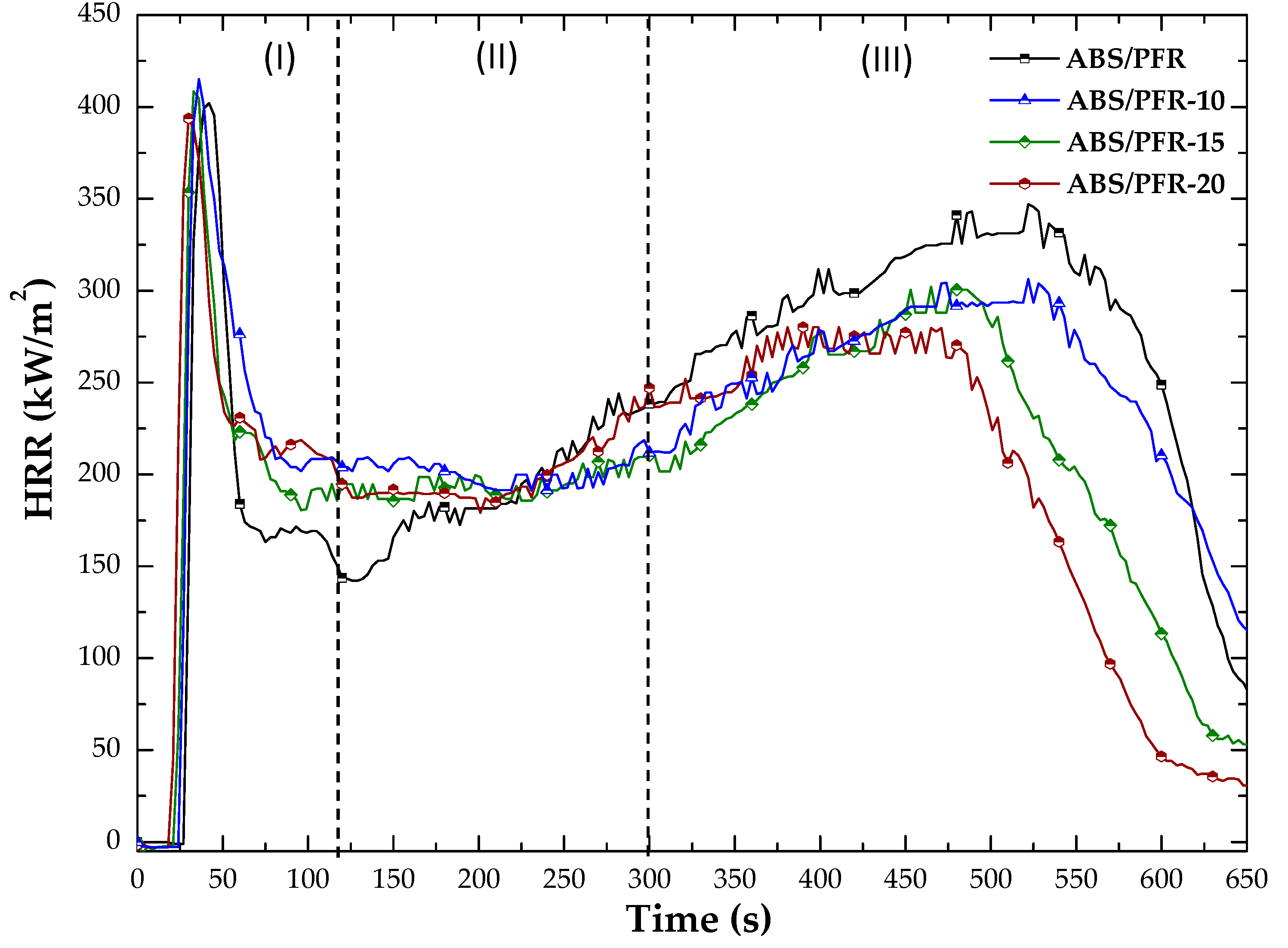
3.4. Fire Behavior
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kattas, L.; Gastrock, F.; Levin, I.; Cacciatore, A. Plastic additives. In Modern Plastics Handbook, 1st ed.; Harper, C.A., Ed.; McGraw-Hill: Lutherville, MD, USA, 2000; p. 284. ISBN 978-0070267145. [Google Scholar]
- Bocchini, S.; Camino, G. Halogen-Containing flame retardants. In Fire Retardancy of Polymeric Materials, 2nd ed.; Wilkie, C.A., Morgan, A.B., Eds.; CRC Press: New York, NY, USA, 2010; p. 88. ISBN 978-1-4200-8399-6. [Google Scholar]
- EU Regulation 143/2011; European Commission: Brussels, Belgium, 2011.
- Hini, S.; Reznick, G.; Yaakov, Y.B.; Georlette, P. Proceedings of the Confefrence Recent Advances Flame Retardancy Polymer Material; Business Communications Corp.: Stamford, CT, USA, 2002. [Google Scholar]
- Tullo, A. Great lakes to phase out flame retardants. Chem. Eng. News 2003, 81, 13. [Google Scholar] [CrossRef]
- Maley, A.M.; Falk, K.A.; Hoover, L.; Earlywine, E.B.; Seymour, M.D.; DeYoung, P.A.; Blum, A.; Stapleton, H.M.; Peaslee, G.F. Detection of halogenated flame retardants in polyurethane foam by particle induced X-ray emission. Nucl. Instrum. Methods Phys. Res. B 2015, 358, 21–25. [Google Scholar] [CrossRef]
- Dasari, A.; Yu, Z.-Z.; Cai, G.-P.; Mai, Y.-W. Recent developments in the fire retardancy of polymeric materials. Prog. Polym. Sci. 2013, 38, 1357–1387. [Google Scholar] [CrossRef]
- Lu, S.Y.; Hamerton, I. Recent developments in the chemistry of halogen-free flame retardant polymers. Prog. Polym. Sci. 2002, 27, 1661–1712. [Google Scholar] [CrossRef]
- Daniel, Y.G.; Howell, B.A. Flame retardant properties of isosorbide bis-phosphorus esters. Polym. Degrad. Stab. 2017, 140, 25–31. [Google Scholar] [CrossRef]
- Jian, R.K.; Chen, L.; Chen, S.Y.; Long, J.W.; Wang, Y.Z. A novel flame-retardant acrylonitrile-butadiene-styrene system based on aluminum isobutylphosphinate and red phosphorus: Flame retardance, thermal degradation and pyrolysis behavior. Polym. Degrad. Stab. 2014, 109, 184–193. [Google Scholar] [CrossRef]
- Xing, W.Y.; Yang, W.; Yang, W.J.; Hu, Q.H.; Si, J.Y.; Lu, H.D.; Yang, B.H.; Song, L.; Hu, Y.; Yuen, R.K.K. Functionalized carbon nanotubes with phosphorus- and Nitrogen-containing agents: Effective reinforce for thermal, mechanical, and flame-retradant properties of polystyrene nanocomposites. ACS Appl. Mater. Interfaces 2016, 8, 26266–26274. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, X.L.; Fang, Z.P. Synergistic effects of expandable graphite and ammonium polyphosphate with a new carbon source derived from biomass in flame retardant ABS. J. Appl. Polym. Sci. 2013, 128, 2424–2432. [Google Scholar] [CrossRef]
- Wang, S.; Hu, Y.; Zong, R.; Tang, Y.; Chen, Z.; Fan, W. Preparation and characterization of flame retardant ABS/montmorillonite nanocomposite. Appl. Clay Sci. 2004, 25, 49–55. [Google Scholar] [CrossRef]
- Nyambo, C.; Songtipya, P.; Manias, E.; Jimenez-Gasco, M.M.; Wilkie, C.A. Effect of MgAl-layered double hydroxide exchanged with linear alkyl carboxylates on fire-retardancy of PMMA and PS. J. Mater. Chem. 2008, 18, 4827–4838. [Google Scholar] [CrossRef]
- Ma, H.Y.; Tong, L.F.; Xu, Z.B.; Fang, Z.P. Functionalizing carbon nanotubes by grafting on intumescent flame retardant: Nanocomposite synthesis, morphology, rheology, and flammability. Adv. Funct. Mater. 2008, 18, 414–421. [Google Scholar] [CrossRef]
- Beydokhti, K.K.; Behravesh, A.H.; Azdast, T. An experimental study on mechanical and microstructural properties of microcellular foams of ABS composites. Iran. Polym. J. 2006, 15, 555–567. [Google Scholar]
- Murray, R.E.; Weller, J.E.; Kumar, V. Solid-state microcellular acrylonitrile-butadiene-styrene foams. Cell. Polym. 2000, 19, 413–425. [Google Scholar]
- Forest, C.; Chaumont, P.; Cassagnau, P.; Swoboda, B.; Sonntag, P. Generation of nanocellular foams from ABS terpolymers. Eur. Polym. J. 2015, 65, 209–220. [Google Scholar] [CrossRef]
- Yoon, T.J.; Kong, W.; Kwon, D.E.; Park, B.K.; Lee, W.; Lee, Y.-W. Preparation of solid-state micro- and nanocellular acrylonitrile-butadiene-styrene (ABS) foams using sub- and supercritical CO2 as blowing agents. J. Supercrit. Fluids 2017, 124, 30–37. [Google Scholar] [CrossRef]
- Linul, E.; Serban, D.A.; Marsavina, L.; Sadowski, T. Assessment of collapse diagrams of rigid polyurethane foams under dynamic loading conditions. Arch. Civ. Mech. Eng. 2017, 3, 457–466. [Google Scholar] [CrossRef]
- Serban, D.A.; Weissenborn, O.; Geller, S.; Marsavina, L.; Gude, M. Evaluation of the mechanical and morphological properties of long fibre reinforced polyurethane rigid foams. Polym. Test. 2016, 49, 121–127. [Google Scholar] [CrossRef]
- Bledzki, A.K.; Kuhn-Gajdzik, J. Microcellular of glass fibre reinforced PC/ABS: Effect of the processing condition on the morphology and mechanical properties. Cell. Polym. 2010, 29, 27–43. [Google Scholar] [CrossRef]
- Mohyeddin, A.; Fereidoon, A.; Taraghi, I. Study of microstructure and flexural properties of microcellular acrylonitrile-butadiene-styrene nanocomposite foams: Experimental results. Appl. Math. Mech. 2015, 36, 487–498. [Google Scholar] [CrossRef]
- Gómez-Monterde, J.; Schulte, M.; Ilijevic, S.; Hain, J.; Sánchez-Soto, M.; Santana, O.O.; Maspoch, M.L. Effect of microcellular foaming on the fracture behaviour of ABS polymer. J. Appl. Polym. Sci. 2016, 133, 43010. [Google Scholar] [CrossRef]
- Wei, W.M.; Hu, S.F.; Zhang, R.; Xu, C.C.; Zhang, F.; Liu, Q.T. Enhanced electrical properties of graphite/ABS composites prepared via supercritical CO2 processing. Polym. Bull. 2017, 74, 4279–4295. [Google Scholar] [CrossRef]
- Rao, W.H.; Xu, H.X.; Xu, Y.J.; Qi, M.; Liao, W.; Xu, S.M.; Wang, Y.Z. Persistently flame-retardant flexible polyurethane foams by a novel phosphorus-containing polyol. Chem. Eng. J. 2018, 343, 198–206. [Google Scholar] [CrossRef]
- Yuan, Y.; Ma, C.; Shi, Y.Q.; Song, L.; Hu, Y.; Hu, W.Z. Highly-efficient reinforcement and flame retardancy of rigid polyurethane foam with phosphorus-containing additive and nitrogen-containing compound. Mater. Chem. Phys. 2018, 211, 42–53. [Google Scholar] [CrossRef]
- Wendels, S.; Chavez, T.; Bonnet, M.; Salmeia, K.A.; Gaan, S. Recent Developments in Organophosphorus Flame Retardants Containing P-C Bond and Their Applications. Materials 2017, 10, 784. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, Y.C.; Li, Y.C.; Shao, Q.; Yan, X.R.; Han, C.; Wang, Z.; Liu, Z.; Guo, Z.H. Flame-retardant rigid polyurethane foam with a phosphorus-nitrogen single intumescent flame retardant. Polym. Adv. Technol. 2018, 29, 668–676. [Google Scholar] [CrossRef]
- Luo, F.B.; Wu, K.; Li, D.F.; Zheng, J.; Guo, H.L.; Zhao, Q.; Lu, M.G. A novel intumescent flame retardant with nanocellulose as charring agent and its flame retardancy in polyurethane foam. Polym. Compos. 2017, 38, 2762–2770. [Google Scholar] [CrossRef]
- Kuranska, M.; Cabulis, U.; Auguscik, M.; Prociak, A.; Ryszkowska, J.; Kirpluks, M. Bio-based polyurethane-polyisocyanurate composites with an intumescent flame retardant. Polym. Degrad. Stab. 2016, 127, 11–19. [Google Scholar] [CrossRef]
- Gao, M.; Wu, W.H.; Liu, S.; Wang, Y.; Shen, T.F. Thermal degradation and flame retardancy of rigid polyurethane foams containing a novel intumescent flame retardant. J. Therm. Anal. Calorim. 2014, 117, 1419–1425. [Google Scholar] [CrossRef]
- Wang, W.; Pan, Y.; Pan, H.F.; Yang, W.; Liew, K.M.; Song, L.; Hu, Y. Synthesis and characterization of MnO2 nanosheets based multilayer coating and applications as a flame retardant for flexible polyurethane foam. Compos. Sci. Technol. 2016, 123, 212–221. [Google Scholar] [CrossRef]
- Xie, H.Y.; Ye, Q.; Si, J.Y.; Yang, W.; Lu, H.D.; Zhang, Q.Z. Synthesis of a carbon nanotubes/ZnAl-layered double hydroxide composite as a novel flame retardant for flexible polyurethane foams. Polym. Adv. Technol. 2016, 27, 651–656. [Google Scholar] [CrossRef]
- Wang, X.C.; Geng, T.; Han, J.; Liu, C.T.; Shen, C.Y.; Turng, L.S.; Yang, H.E. Effects of Nanoclays on the Thermal Stability and Flame Retardancy of Microcellular Thermoplastic Polyurethane Nanocomposites. Polym. Compos. 2018, 39, E1429–E1440. [Google Scholar] [CrossRef]
- Cheng, J.J.; Qu, W.J.; Sun, S.H. Effects of flame-retardant flax-fiber on enhancing performance of the rigid polyurethane foams. J. Appl. Polym. Sci. 2018, 135, 46436. [Google Scholar] [CrossRef]
- Yue, D.Z.; Oribayo, O.; Rempel, G.L.; Pan, Q.M. Liquefaction of waste pine wood and its application in the synthesis of a flame retardant polyurethane foam. RSC Adv. 2017, 7, 30334–30344. [Google Scholar] [CrossRef]
- Wang, X.; Pan, Y.T.; Wan, J.T.; Wang, D.Y. An eco-friendly way to fire retardant flexible polyurethane foam: Layer-by-layer assembly of fully bio-based substances. RSC Adv. 2014, 4, 46164–46169. [Google Scholar] [CrossRef]
- Zhang, S.; Ji, W.F.; Han, Y.; Gu, X.Y.; Li, H.F.; Sun, J. Flame-retardant expandable polystyrene foams coated with ethanediol-modified melamine-formaldehyde resin and microencapsulated ammonium polyphosphate. J. Appl. Polym. Sci. 2018, 135, 46471. [Google Scholar] [CrossRef]
- Zhu, Z.M.; Xu, Y.J.; Liao, W.; Xu, S.M.; Wang, Y.Z. Highly Flame Retardant Expanded Polystyrene Foams from Phosphorus-Nitrogen-Silicon Synergistic Adhesives. Ind. Eng. Chem. 2017, 56, 4649–4658. [Google Scholar] [CrossRef]
- Hamdani-Devarennes, S.; El Hage, R.; Dumazert, L.; Sonnier, R.; Ferry, L.; Lopez-Cuesta, J.M.; Bert, C. Water-based flame retardant coating using nano-boehmite for expanded polystyrene (EPS) foam. Prog. Org. Chem. 2016, 99, 32–46. [Google Scholar] [CrossRef]
- Levchik, S.V.; Wei, E.D. New developments in flame retardancy of styrene thermoplastics and foams. Polym. Int. 2008, 57, 431–448. [Google Scholar] [CrossRef]
- Realinho, V.; Haurie, L.; Formosa, J.; Velasco, J.I. Flame retardancy effect of combined ammonium polyphosphate and aluminium diethyl phosphinate in acrylonitrile-butadiene-styrene. Polym. Degrad. Stab. 2018, 155, 208–219. [Google Scholar] [CrossRef]
- Sims, G.; Khunniteekool, C. Cell size measurement of polymeric foams. Cell. Polym. 1994, 13, 137–146. [Google Scholar]
- Liu, X. Application of dynamic mechanical thermal analysis on polymer material. Eng. Plast. Appl. 2010, 7, 84–86. [Google Scholar]
- Li, Y.C.; Wu, X.L.; Song, J.F.; Li, J.F.; Shao, Q.; Cao, N.; Lu, N.; Guo, Z.H. Reparation of recycled acrylonitrile-butadiene-styrene by pyromellitic dianhydride: Reparation performance evaluation and property analysis. Polymer 2017, 124, 41–47. [Google Scholar] [CrossRef]
- Baboo, M.; Dixit, M.; Sharma, K.; Saxena, N.S. Mechanical and thermal characterization of cis-polyisoprene and trans-polyisoprene blends. Polym. Bull. 2011, 66, 661–672. [Google Scholar] [CrossRef]
- Modesti, M.; Besco, S.; Lorenzetti, A.; Causin, V.; Marega, C.; Gilman, J.W.; Fox, D.M.; Trulove, P.C.; De Long, H.C.; Zammarano, M. ABS/clay nanocomposites obtained by a solution technique: Influence of clay organic modifiers. Polym. Degrad. Stab. 2007, 92, 2206–2213. [Google Scholar] [CrossRef]
- Shimbo, M.; Kawashima, H.; Yoshitami, S. Foam injection technology and influence factors of microcellular plastics. In Proceedings of the 2nd International Conference on Thermoplastic Foam, Parsippany, NJ, USA, 24–25 October 2000; pp. 162–168. [Google Scholar]
- Michaeli, W.; Florez, L.; Oberloer, D.; Brinkmann, M. Analysis of the impact properties of structural foams. Cell. Plast. 2009, 45, 321–351. [Google Scholar] [CrossRef]
- Xu, J. Microcellular Injection Molding; John Wiley and Sons: Cambridge, UK, 2010; pp. 62–85. ISBN 978-0-470-46612-4. [Google Scholar]
- Xia, J.; Jian, X.; Li, J.; Wang, X.; Xu, Y. Synergistic effect of montmorillonite and intumescent flame retardant on flame retardance enhancement of ABS. Polym. Plast. Technol. Eng. 2007, 46, 227–232. [Google Scholar] [CrossRef]
- Schartel, B. Uses of fire tests in materials flammability development. In Fire Retardancy of Polymeric Materials, 2nd ed.; Wilkie, C.A., Morgan, A.B., Eds.; CRC Press: New York, NY, USA, 2010; p. 388. ISBN 978-1-4200-8399-6. [Google Scholar]
- Realinho, V.; Antunes, M.; Velasco, J.I. Enhanced fire behavior of Casico-based foams. Polym. Degrad. Stab. 2016, 128, 260–268. [Google Scholar] [CrossRef]






| Material | Complete Part | Core | ||||||
|---|---|---|---|---|---|---|---|---|
| Density (g/cm3) | Relative Density, ρr | Density (g/cm3) | Relative Density, ρrc | ϕVD (µm) | ϕWD (µm) | Nf (cells/cm3) | N0 (cells/cm3) | |
| ABS-10 | 0.939 | 0.900 | 0.782 | 0.774 | 13.6 | 12.6 | 1.72 × 108 | 1.35 × 108 |
| (0.003) | (0.047) | (2.2) | (6.3) | |||||
| ABS-15 | 0.887 | 0.850 | 0.750 | 0.743 | 9.2 | 8.5 | 6.32 × 108 | 6.46 × 108 |
| (0.011) | (0.033) | (1.9) | (1.4) | |||||
| ABS-20 | 0.834 | 0.800 | 0.696 | 0.668 | 19.5 | 18.9 | 9.70 × 107 | 8.07 × 107 |
| (0.001) | (0.015) | (4.9) | (5.6) | |||||
| ABS/PFR-10 | 1.047 | 0.909 | 0.904 | 0.805 | 7.8 | 7.9 | 7.87 × 108 | 6.88 × 108 |
| (0.020) | (0.016) | (0.6) | (0.3) | |||||
| ABS/PFR-15 | 0.988 | 0.862 | 0.834 | 0.743 | 7.6 | 7.6 | 1.15 × 109 | 1.60 × 109 |
| (0.002) | (0.018) | (0.8) | (0.4) | |||||
| ABS/PFR-20 | 0.925 | 0.807 | 0.740 | 0.659 | 9.5 | 8.9 | 7.74 × 108 | 9.68 × 108 |
| (0.005) | (0.010) | (0.9) | (0.7) | |||||
| Material | Fmax * (N) | Fmax Reduction ** (%) | Emax * (J) | Emax Reduction ** (%) | ET * (J) | ET Reduction ** (%) |
|---|---|---|---|---|---|---|
| ABS | 9385 | - | 79.9 | - | 119.6 | - |
| ABS-10 | 3626 | 61.4 | 18.3 | 77.1 | 42.0 | 64.9 |
| ABS-15 | 3314 | 64.7 | 16.6 | 79.2 | 28.9 | 75.8 |
| ABS-20 | 2629 | 72.0 | 13.7 | 82.9 | 25.9 | 78.3 |
| ABS/PFR | 2439 | - | 9.5 | - | 10.7 | - |
| ABS/PFR-10 | 1672 | 31.4 | 7.0 | 26.3 | 7.7 | 28.0 |
| ABS/PFR-15 | 1297 | 46.8 | 5.3 | 44.2 | 6.6 | 38.3 |
| ABS/PFR-20 | 1157 | 52.6 | 3.8 | 60.0 | 5.4 | 49.5 |
| Material | TTI (s) | PHRR1 * (kW/m2) | Time of PHRR1 (s) | PHRR2 * (kW/m2) | Time of PHRR2 (s) | THE (MJ/m2) | Residue (wt %) |
|---|---|---|---|---|---|---|---|
| ABS | 32 | 1760 | 156 | - | - | 191 | 0.52 |
| ABS-10 | 22 | 1502 | 147 | - | - | 178 | 0.50 |
| ABS-15 | 22 | 1436 | 144 | - | - | 168 | 0.49 |
| ABS-20 | 17 | 1432 | 123 | - | - | 157 | 0.47 |
| ABS/PFR | 28 | 402 | 39 | 345 | 522 | 173 | 11.3 |
| ABS/PFR-10 | 28 | 415 | 36 | 306 | 522 | 162 | 11.3 |
| ABS/PFR-15 | 25 | 409 | 33 | 301 | 483 | 140 | 13.0 |
| ABS/PFR-20 | 23 | 409 | 33 | 280 | 468 | 132 | 12.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Realinho, V.; Arencón, D.; Antunes, M.; Velasco, J.I. Effects of a Phosphorus Flame Retardant System on the Mechanical and Fire Behavior of Microcellular ABS. Polymers 2019, 11, 30. https://doi.org/10.3390/polym11010030
Realinho V, Arencón D, Antunes M, Velasco JI. Effects of a Phosphorus Flame Retardant System on the Mechanical and Fire Behavior of Microcellular ABS. Polymers. 2019; 11(1):30. https://doi.org/10.3390/polym11010030
Chicago/Turabian StyleRealinho, Vera, David Arencón, Marcelo Antunes, and José Ignacio Velasco. 2019. "Effects of a Phosphorus Flame Retardant System on the Mechanical and Fire Behavior of Microcellular ABS" Polymers 11, no. 1: 30. https://doi.org/10.3390/polym11010030
APA StyleRealinho, V., Arencón, D., Antunes, M., & Velasco, J. I. (2019). Effects of a Phosphorus Flame Retardant System on the Mechanical and Fire Behavior of Microcellular ABS. Polymers, 11(1), 30. https://doi.org/10.3390/polym11010030