Synergetic Toughening Effect of Carbon Nanotubes and β-Nucleating Agents on the Polypropylene Random Copolymer/Styrene-Ethylene-Butylene- Styrene Block Copolymer Blends
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Sample Preparation
2.3. Characterization and Testing
2.3.1. Differential Scanning Calorimetry
2.3.2. Scanning Electron Microscopy (SEM)
2.3.3. X-Ray Characterization
2.3.4. Dynamical Mechanical Analysis
2.3.5. Transmission Electron Microscope (TEM)
2.3.6. Impact Tests
2.3.7. Tensile Tests
3. Results and Discussion
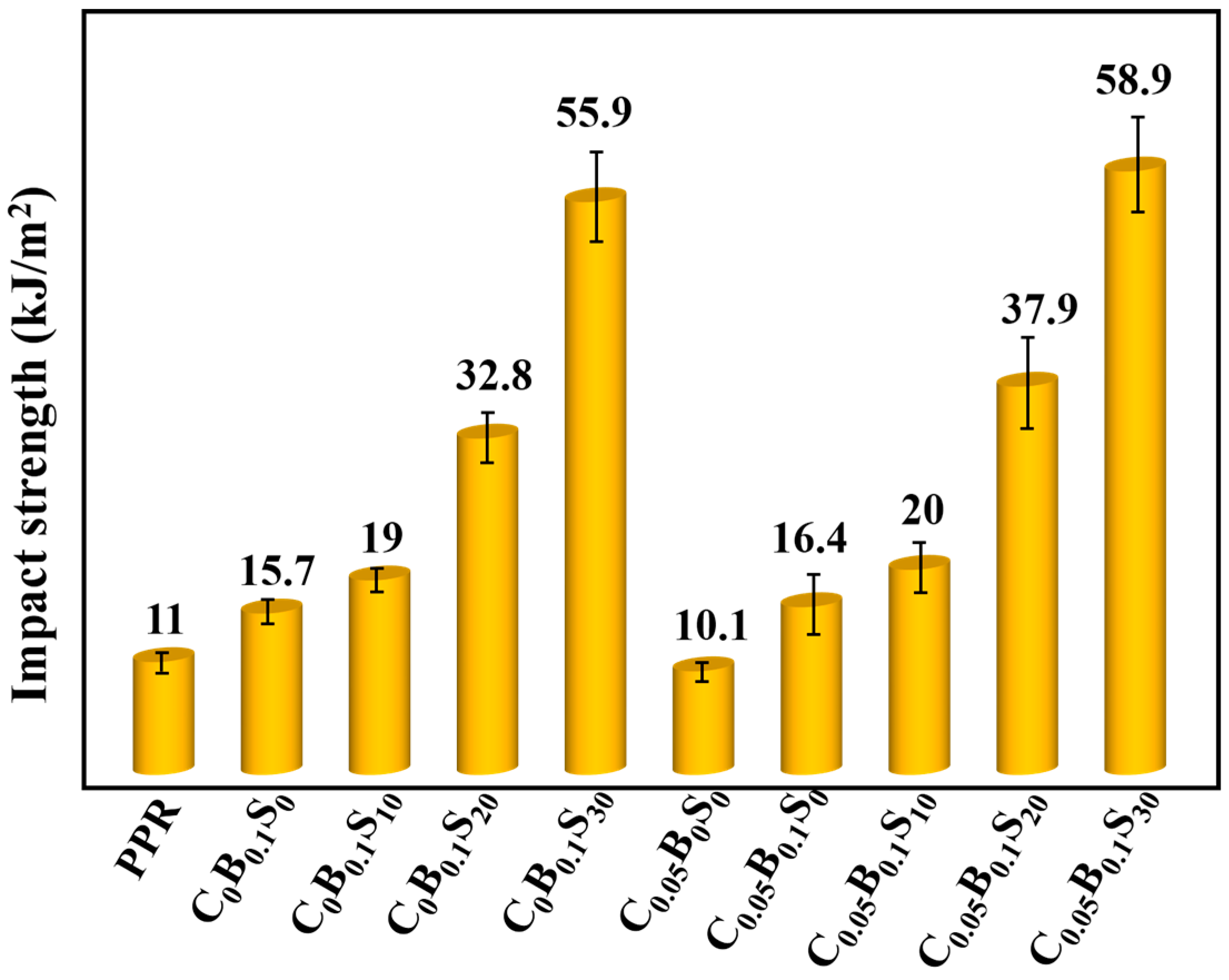
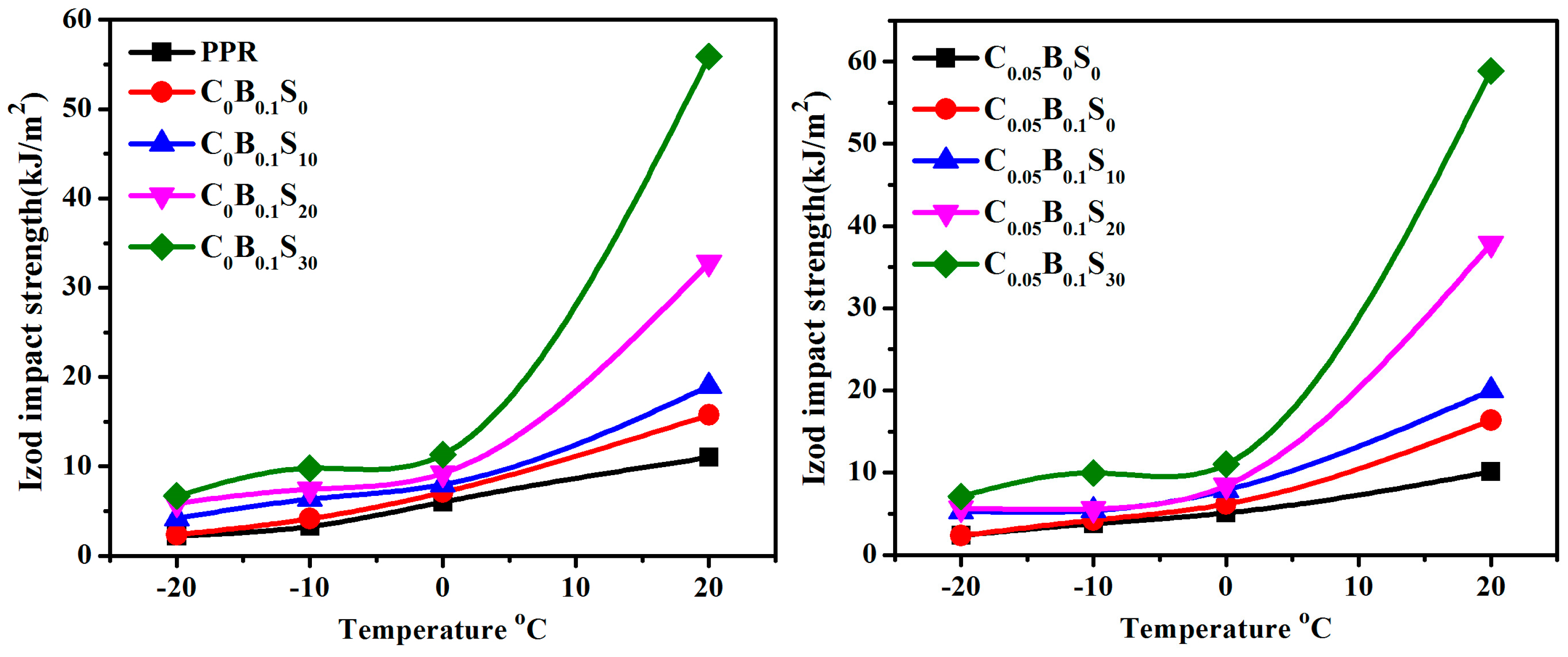
3.1. Notched Izod Impact Performance
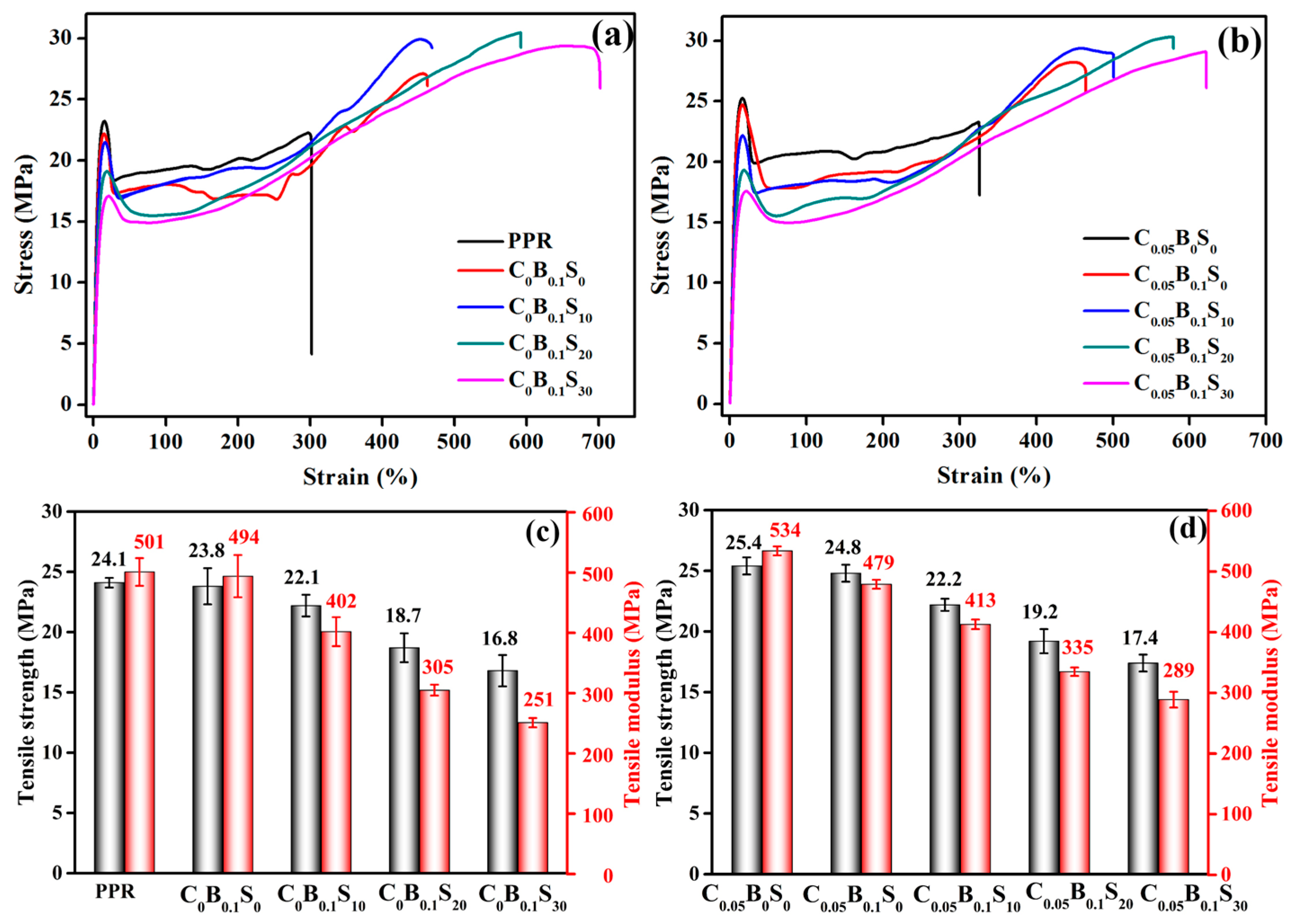
3.2. Tensile Properties
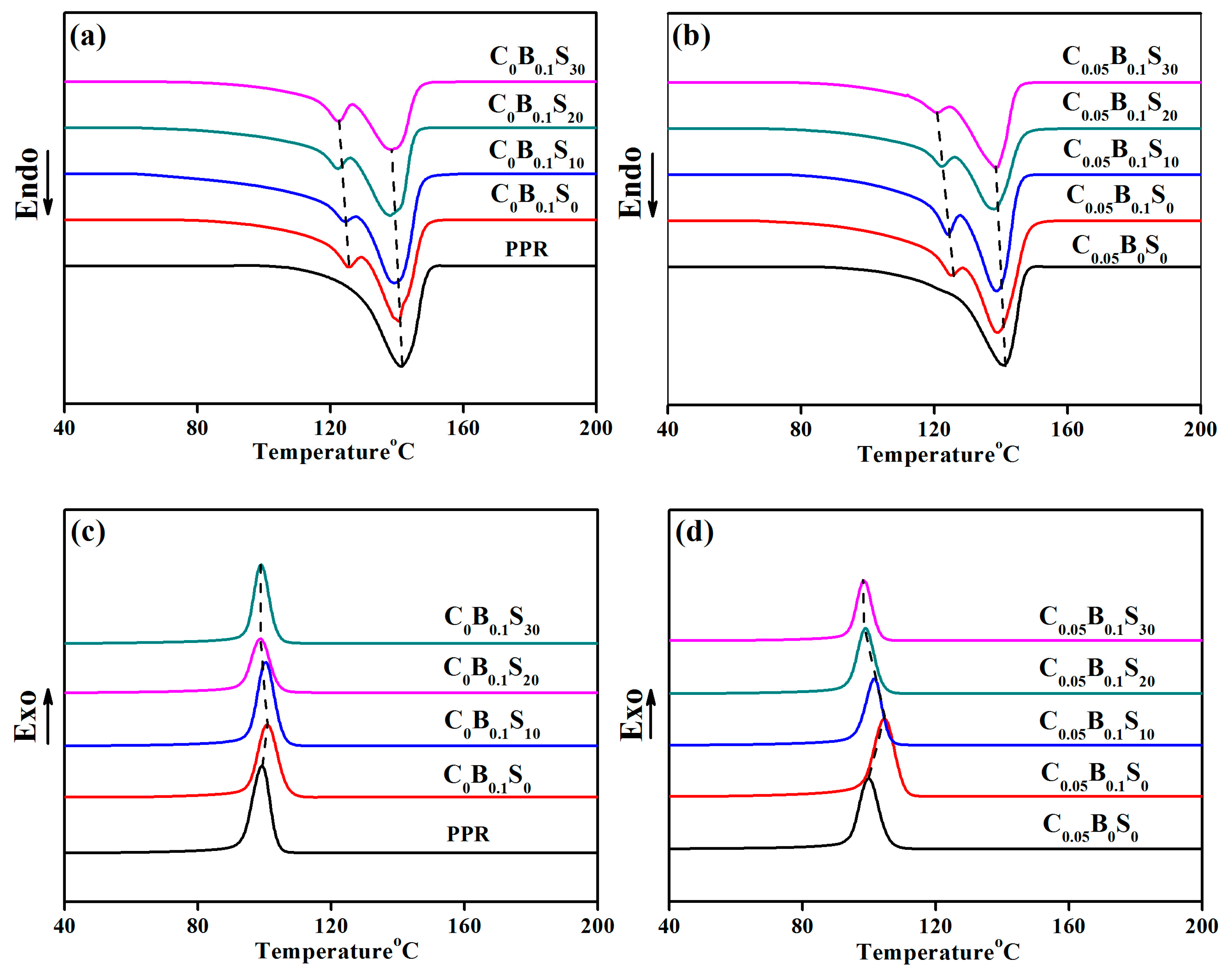
3.3. Melting and Crystallization Behavior
3.4. Phase Morphology
3.5. Glass Transition Temperatures
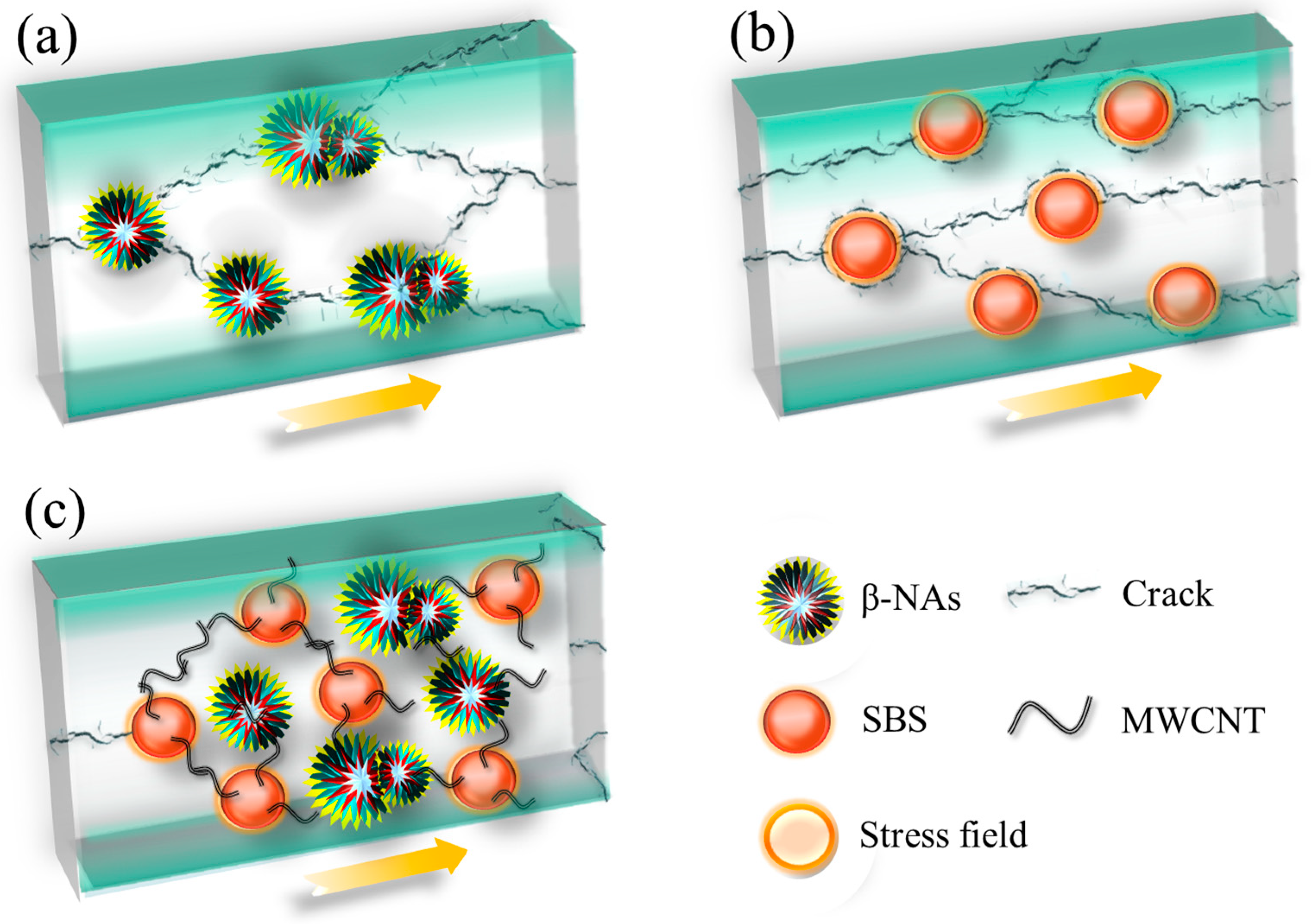
3.6. Toughening Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cao, J.; Zheng, Y.Y.; Lin, T.F. Synergistic toughening effect of β-nucleating agent and long chain branching on polypropylene random copolymer. Polym. Test. 2016, 55, 318–327. [Google Scholar] [CrossRef]
- De Rosa, C.; Auriemma, F.; Ruiz de Ballesteros, O.; Resconi, L.; Camurati, I. Tailoring the Physical Properties of Isotactic Polypropylene through Incorporation of Comonomers and the Precise Control of Stereo- and Regioregularity by Metallocene Catalysts. Chem. Mater. 2007, 19, 5122–5130. [Google Scholar] [CrossRef]
- Hosier, I.; Alamo, R.; Esteso, P.; Isasi, J.; Mandelkern, L. Formation of the α and γpolymorphs in random metallocene-propylene copolymers. Effect of concentration and type of comonomer. Macromolecules 2003, 36, 5623–5636. [Google Scholar] [CrossRef]
- Luo, F.; Wang, J.; Bai, H.W.; Wang, K.; Deng, H.; Zhang, Q.; Chen, F.; Fu, Q.; Na, B. Synergistic toughening of polypropylene random copolymer at low temperature: β-Modification and annealing. Mater. Sci. Eng. A 2011, 528, 7052–7059. [Google Scholar] [CrossRef]
- Obadal, M.; Cermak, R.; Baran, N.; Stoklasa, K.; Simonik, J. Impact Strength of β-nucleated polypropylene. Int. Polym. Process. 2004, 19, 35–39. [Google Scholar] [CrossRef]
- Xiong, F.; Guan, R.; Xiao, Z.X.; Xiang, B.L.; Lu, D.P. Influence of β-nucleators on iPP crystallization and morphology. Polym.-Plast. Technol. Eng. 2007, 46, 97–103. [Google Scholar] [CrossRef]
- Cao, J.; Lv, Q.F. Crystalline structure, morphology and mechanical properties of β-nucleated controlled-rheology polypropylene random copolymers. Polym. Test. 2011, 30, 899–906. [Google Scholar] [CrossRef]
- Papageorgiou, D.G.; Chrissafis, K.; Bikiaris, D.N. β-Nucleated Polypropylene: Processing, Properties and Nanocomposites. Polym. Rev. 2015, 55, 596–629. [Google Scholar] [CrossRef]
- Tang, W.; Tang, J.; Yuan, H.; Jin, R. Crystallization behavior and mechanical properties of polypropylene random copolymer/poly (ethylene-octene) blends. J. Appl. Polym. Sci. 2011, 122, 461–468. [Google Scholar] [CrossRef]
- Rondin, J.; Bouquey, M.; Muller, R.; Serra, C.A.; Martin, G.; Sonntag, P. Dispersive mixing efficiency of an elongational flow mixer on PP/EPDM blends: Morphological analysis and correlation with viscoelastic properties. Polym. Eng. Sci. 2014, 54, 1444–1457. [Google Scholar] [CrossRef]
- Abreu, F.; Forte, M.; Liberman, S. SBS and SEBS block copolymers as impact modifiers for polypropylene compounds. J. Appl. Polym. Sci. 2005, 95, 254–263. [Google Scholar] [CrossRef]
- Liu, P.W.; Cottrill, A.L.; Kozawa, D.; Koman, V.B.; Parviz, D.; Liu, A.T.X.; Yang, J.F.; Tran, T.Q.; Wong, M.H.; Wang, S.; et al. Emerging trends in 2D nanotechnology that are redefining our understanding of “Nanocomposites”. Nano Today 2018, 21, 18–40. [Google Scholar] [CrossRef]
- Khoshnevis, H.; Myomint, S.; Yedinak, E.; Tran, T.Q.; Zadhoush, A.; Youssefi, M.; Pasquali, M.; Duong, H.M. Super high-rate fabrication of high-purity carbon nanotube aerogels from floating catalyst method for oil spill cleaning. Chem. Phys. Lett. 2018, 693, 146–151. [Google Scholar] [CrossRef]
- Zamani, M.M.; Fereidoon, A.; Sabet, A. Multi-walled carbon nanotube-filled polypropylene nanocomposites: High velocity impact response and mechanical properties. Iran. Polym. J. 2012, 21, 887–894. [Google Scholar] [CrossRef]
- Taraghi, I.; Fereidoon, A.; Zamani, M.M.; Mohyeddin, A. Mechanical, thermal, and viscoelastic properties of polypropylene/glass hybrid composites reinforced with multiwalled carbon nanotubes. J. Compos. Mater. 2015, 49, 3557–3566. [Google Scholar] [CrossRef]
- Tran, T.Q.; Headrick, R.J.; Bengio, E.A.; Myint, S.M.; Khoshnevis, H.; Jamali, V.; Duong, H.M.; Pasquali, M. Purification and dissolution of carbon nanotube fibers spun from the floating catalyst method. ASC Appl. Mater. Interfaces 2017, 9, 37112–37119. [Google Scholar] [CrossRef] [PubMed]
- Dasari, A.; Zhang, Q.X.; Yu, Z.Z.; Mai, Y.W. Toughening polypropylene and its nanocomposites with submicrometer voids. Macromolecules 2010, 43, 5734–5739. [Google Scholar] [CrossRef]
- Ma, L.F.; Wang, W.K.; Bao, R.Y.; Yang, W.; Xie, B.H.; Yang, M.B. Toughening of polypropylene with beta-nucleated thermoplastic vulcanizates based on polypropylene/ethylene-propylene-diene rubber blends. Mater. Des. 2013, 51, 536–543. [Google Scholar] [CrossRef]
- Varga, J.; Mudra, I.; Ehrenstein, G.W. Highly active thermally stable β-nucleating agents for isotactic polypropylene. J. Appl. Polym. Sci. 1999, 74, 2357–2368. [Google Scholar] [CrossRef]
- Li, M.; Li, G.; Zhang, Z.; Dai, X.; Mai, K. Enhanced β-crystallization in polypropylene random copolymer with a supported β-nucleating agent. Thermochim. Acta 2014, 598, 36–44. [Google Scholar] [CrossRef]
- Ren, Q.L.; Zhang, Q.L.; Wang, L.; Yi, J.J.; Feng, J.C. Synergistic toughening effect of olefin block copolymer and highly effective β-nucleating agent on the low temperature toughness of polypropylene random copolymer. Ind. Eng. Chem. Res. 2017, 56, 5277–5283. [Google Scholar] [CrossRef]
- Chen, Y.H.; Wu, Z.Q.; Fan, Q.; Yang, S.; Song, E.C.; Zhang, Q.Y. Great toughness reinforcement of isotactic polypropylene/elastomer blends with quasi-cocontinuous phase morphology by traces of β-nucleating agents and carbon nanotubes. Compos. Sci. Technol. 2018, 167, 277–284. [Google Scholar] [CrossRef]
- Li, M.J.; Yang, Q.; Kong, M.Q.; Huang, Y.J.; Liao, X.; Niu, Y.H.; Zhao, Z.G. Role of dicumyl peroxide on the morphology and mechanical performance of polypropylene random copolymer in microinjection molding. Polym. Adv. Technol. 2018, 29, 171–181. [Google Scholar] [CrossRef]
- Chen, Y.H.; Fang, D.F.; Lei, J.; Li, L.B.; Hsiao, B.S.; Li, Z.M. Shear-Induced Precursor Relaxation-Dependent Growth Dynamics and Lamellar Orientation of beta-Crystals in beta-Nucleated Isotactic Polypropylene. J. Phys. Chem. B 2014, 119, 34–143. [Google Scholar]
- Chen, Y.H.; Zhong, G.J.; Lei, J.; Li, Z.M.; Benjamin, S.H. In-situ synchrotron x-ray scattering study on isotactic polypropylene crystallization under the coexistence of shear flow and carbon nanotubes. Macromolecules 2011, 44, 8080–8092. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, J.Z.; Chen, Y.H.; Qiao, K.; Xu, L.; Ji, X.; Li, Z.M.; Hsiao, B.S. Crystalline Structure Changes in Preoriented Metallocene-Based Isotactic Polypropylene upon Annealing. J. Phys. Chem. B 2013, 117, 7113–7122. [Google Scholar] [CrossRef]
- Saroop, M.; Mathur, G.N. Studies on the dynamically vulcanized polypropylene (PP) butadiene styrene block copolymer (SBS) blends: Mechanical properties. J. Appl. Polym. Sci. 1997, 65, 2691–2701. [Google Scholar] [CrossRef]
- Turnerjones, A.; Cobbold, A.J. The β crystalline form of isotactic polypropylene. J. Polym. Sci. Part B Polym. Lett. 1968, 6, 539–546. [Google Scholar] [CrossRef]
- Yang, J.N.; Nie, S.B.; Zhu, J.B. A comparative study on different rubbery modifiers: Effect on morphologies, mechanical, and thermal properties of PLA blends. J. Appl. Polym. Sci. 2016, 133, 43340. [Google Scholar] [CrossRef]
- Jang, B.Z.; Uhlmann, D.R.; Sande, J.B. Crazing in polypropylene. Polym. Eng. Sci. 1985, 25, 98–104. [Google Scholar] [CrossRef]
- Jang, B.Z.; Uhlmann, D.R.; Sande, J.B.V. The rubber particle size dependence of crazing in polypropylene. Polym. Eng. Sci. 2010, 25, 643–651. [Google Scholar] [CrossRef]
- Borggreve, R.J.M.; Gaymans, R.J.; Schuijer, J.; Housz, J.F.I. Brittle-tough transition in nylon-rubber blends: Effect of rubber concentration and particles size. Polymer 1987, 28, 1489–1496. [Google Scholar] [CrossRef]
- Jang, B.Z.; Uhlmann, D.R.; Sande, J.B.V. Rubber-toughening in polypropylene. J. Appl. Polym. Sci. 1985, 30, 2485–2504. [Google Scholar] [CrossRef]
- Ren, F.; Li, Z.; Xu, L.; Sun, Z.F.; Ren, P.G.; Yan, D.Y.; Li, Z.M. Large-scale preparation of segregated PLA/carbon nanotube composite with high efficient electromagnetic interference shielding and favourable mechanical properties. Compos. Part B-Eng. 2018, 155, 405–413. [Google Scholar] [CrossRef]
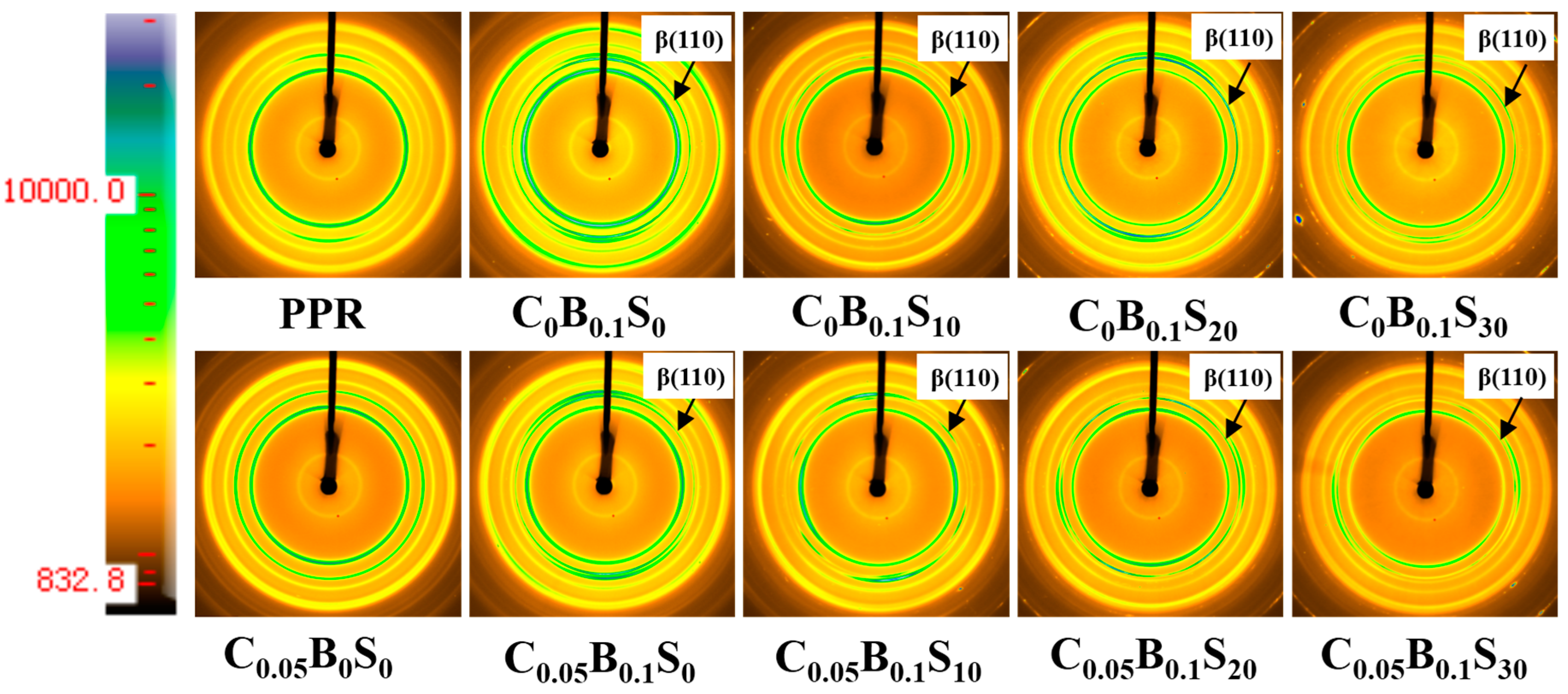
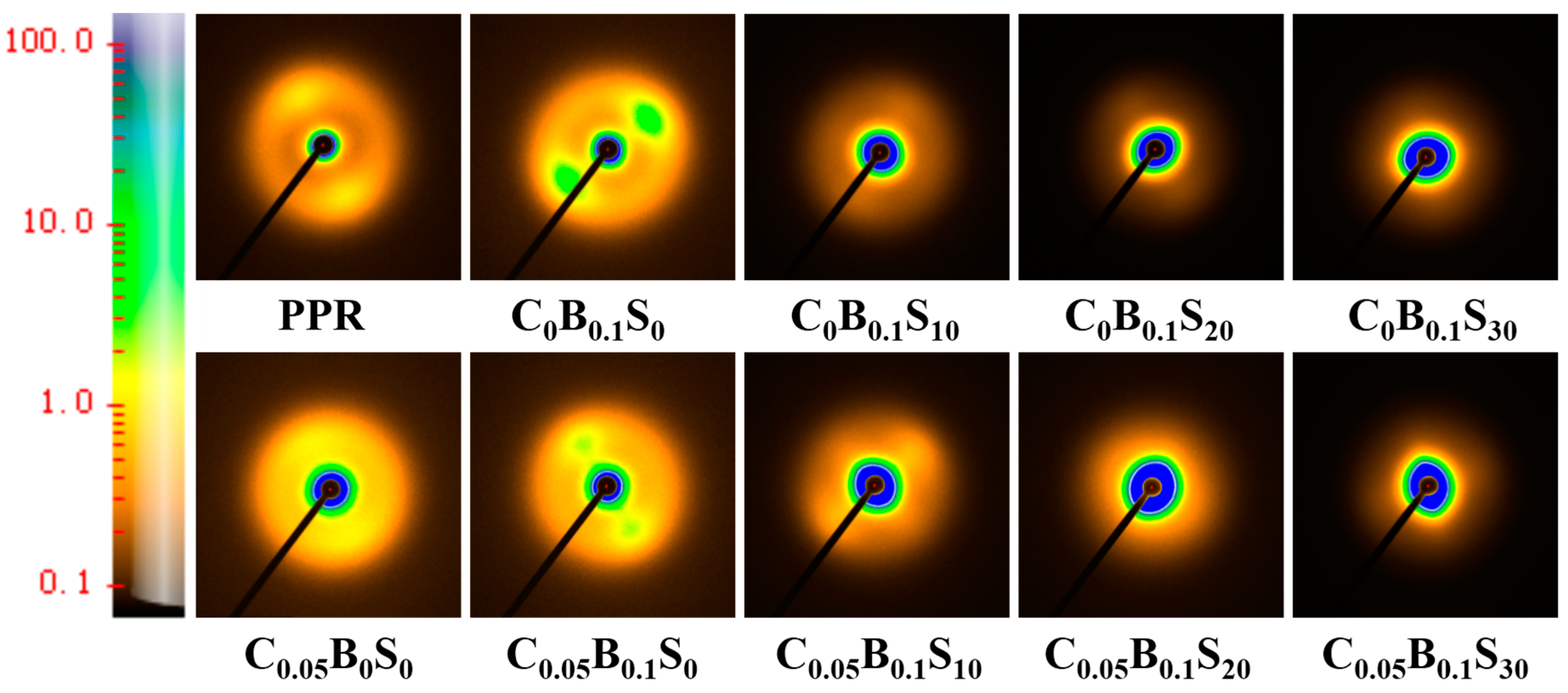

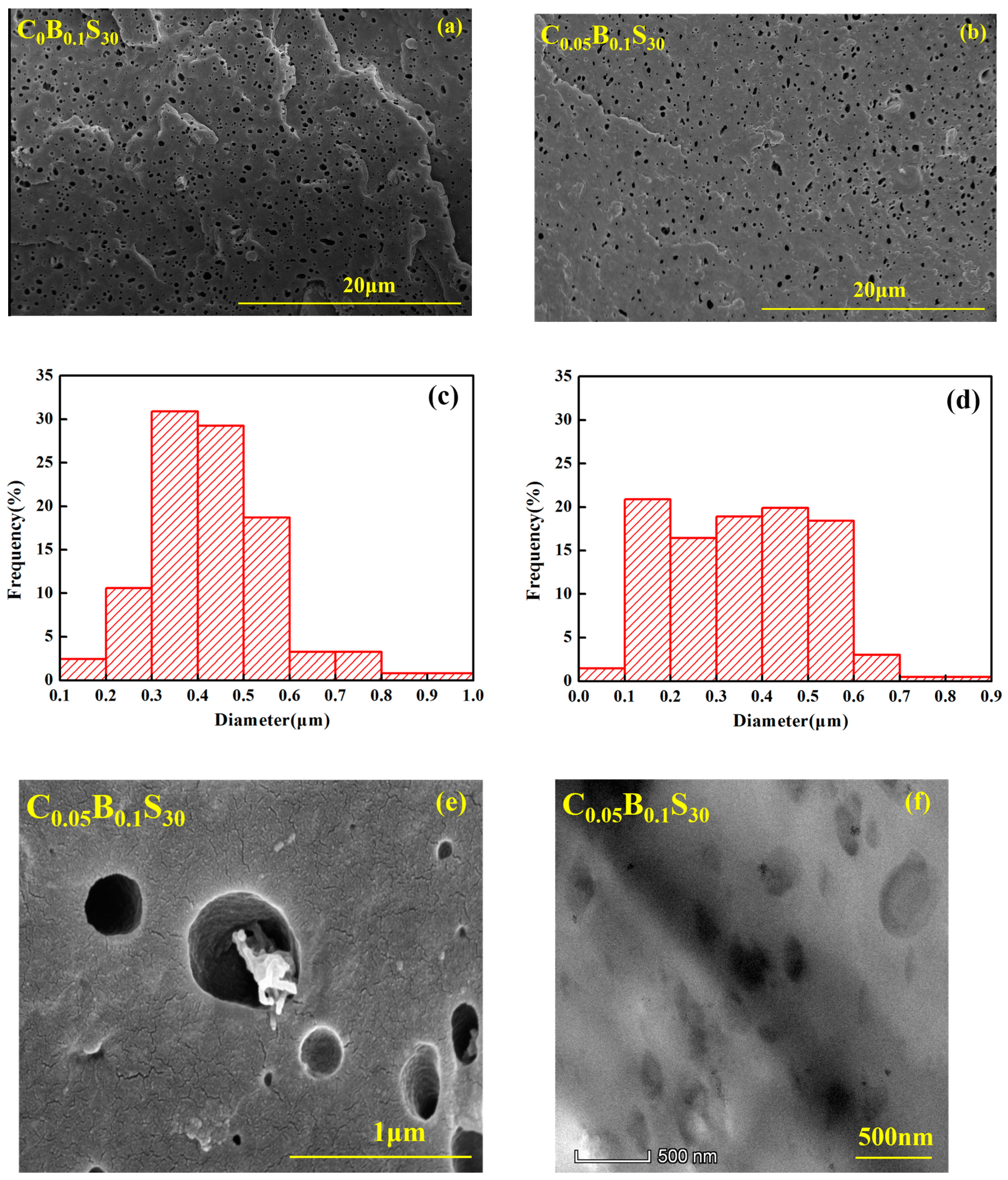
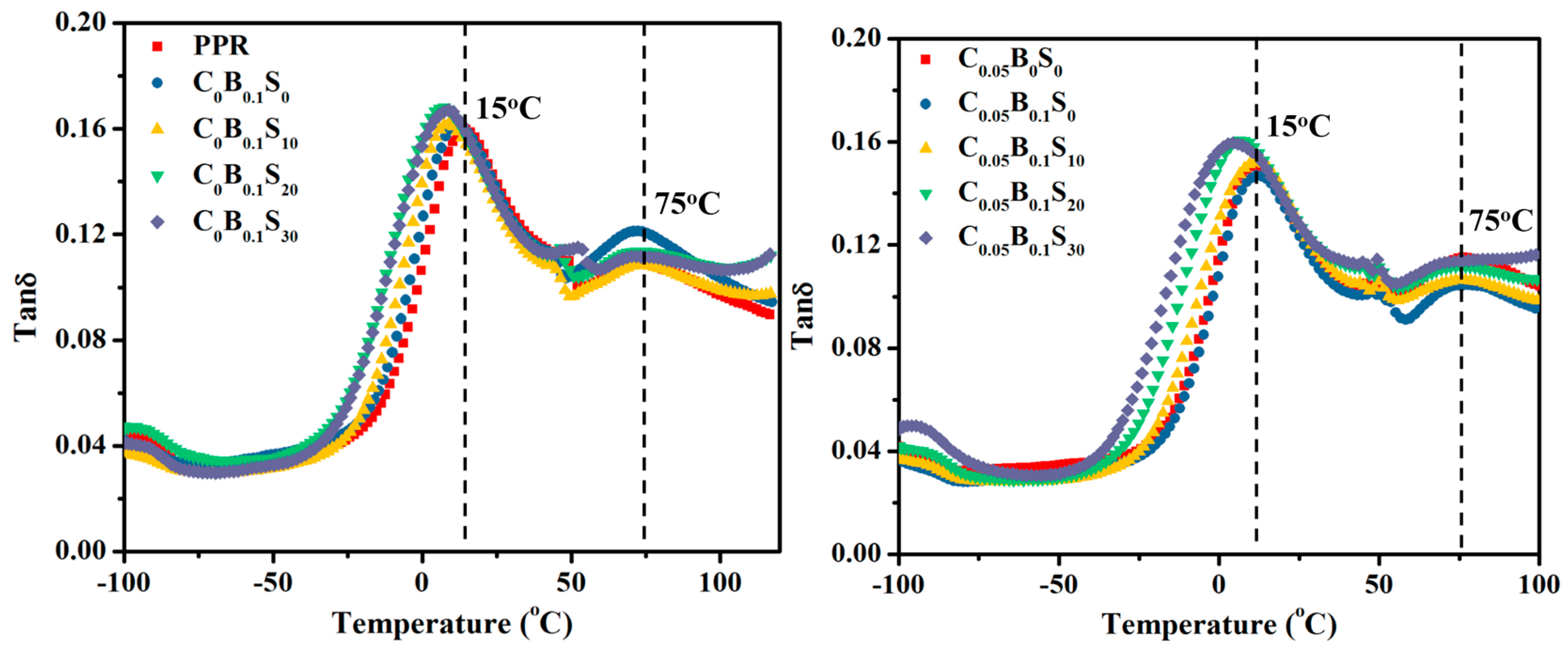
| Samples | PPR (wt %) | SBS (wt %) | TMB-5 (wt %) | MWCNTs (wt %) |
|---|---|---|---|---|
| PPR | 100 | 0 | 0 | 0 |
| C0B0.1S0 | 100 | 0 | 0.1 | 0 |
| C0B0.1S10 | 90 | 10 | 0.1 | 0 |
| C0B0.1S20 | 80 | 20 | 0.1 | 0 |
| C0B0.1S30 | 70 | 30 | 0.1 | 0 |
| C0.05B0S0 | 100 | 0 | 0 | 0.05 |
| C0.05B0.1S0 | 100 | 0 | 0.1 | 0.05 |
| C0.05B0.1S10 | 90 | 10 | 0.1 | 0.05 |
| C0.05B0.1S20 | 80 | 20 | 0.1 | 0.05 |
| C0.05B0.1S30 | 70 | 30 | 0.1 | 0.05 |
| C0.05B0S30 | 70 | 30 | 0 | 0.05 |
| Samples | Tc/°C | α-form | β-form | Total Xc | ||
|---|---|---|---|---|---|---|
| Tm/°C | Xα | Tm/°C | Xβ | |||
| PPR | 99.2 | 141.1 | 0.19 | 0.19 | ||
| C0B0.1S0 | 100.9 | 140.6 | 0.15 | 125.5 | 0.09 | 0.24 |
| C0B0.1S10 | 100.3 | 140.2 | 0.21 | 124.2 | 0.10 | 0.31 |
| C0B0.1S20 | 98.9 | 139.2 | 0.18 | 123.0 | 0.11 | 0.29 |
| C0B0.1S30 | 98.6 | 138.5 | 0.16 | 122.0 | 0.10 | 0.26 |
| C0.05B0S0 | 99.7 | 141.1 | 0.21 | 0.21 | ||
| C0.05B0.1S0 | 104.7 | 140.0 | 0.18 | 125.5 | 0.12 | 0.30 |
| C0.05B0.1S10 | 101.7 | 139.2 | 0.17 | 124.1 | 0.08 | 0.25 |
| C0.05B0.1S20 | 99.1 | 138.5 | 0.17 | 122.5 | 0.07 | 0.24 |
| C0.05B0.1S30 | 98.1 | 138.2 | 0.16 | 121.1 | 0.06 | 0.22 |
| Samples | Xc | Kβ | Xβ |
|---|---|---|---|
| PPR | 0.35 | -- | -- |
| C0B0.1S0 | 0.36 | 0.12 | 0.05 |
| C0B0.1S10 | 0.31 | 0.24 | 0.08 |
| C0B0.1S20 | 0.29 | 0.28 | 0.08 |
| C0B0.1S30 | 0.28 | 0.31 | 0.09 |
| C0.05B0S0 | 0.33 | -- | --- |
| C0.05B0.1S0 | 0.35 | 0.26 | 0.09 |
| C0.05B0.1S10 | 0.31 | 0.26 | 0.08 |
| C0.05B0.1S20 | 0.30 | 0.26 | 0.08 |
| C0.05B0.1S30 | 0.26 | 0.17 | 0.05 |
| Samples | S (1/nm) | LB (nm) | Lc (nm) | La (nm) |
|---|---|---|---|---|
| PPR | 0.0802 | 12.5 | 4.4 | 8.1 |
| C0B0.1S0 | 0.0788 | 12.7 | 4.6 | 8.1 |
| C0B0.1S10 | 0.0774 | 12.9 | 4.0 | 8.9 |
| C0B0.1S20 | 0.0732 | 13.7 | 4.0 | 9.7 |
| C0B0.1S30 | 0.0710 | 14.1 | 3.9 | 10.2 |
| C0.05B0S0 | 0.0798 | 12.5 | 4.1 | 8.4 |
| C0.05B0.1S0 | 0.0785 | 12.7 | 4.4 | 8.3 |
| C0.05B0.1S10 | 0.0769 | 13.0 | 4.0 | 9.0 |
| C0.05B0.1S20 | 0.0703 | 14.2 | 4.3 | 9.9 |
| C0.05B0.1S30 | 0.0700 | 14.3 | 3.7 | 10.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, P.-G.; Wang, J.; Fan, Q.; Yang, S.; Wu, Z.-Q.; Yan, D.-X.; Chen, Y.-H. Synergetic Toughening Effect of Carbon Nanotubes and β-Nucleating Agents on the Polypropylene Random Copolymer/Styrene-Ethylene-Butylene- Styrene Block Copolymer Blends. Polymers 2019, 11, 29. https://doi.org/10.3390/polym11010029
Ren P-G, Wang J, Fan Q, Yang S, Wu Z-Q, Yan D-X, Chen Y-H. Synergetic Toughening Effect of Carbon Nanotubes and β-Nucleating Agents on the Polypropylene Random Copolymer/Styrene-Ethylene-Butylene- Styrene Block Copolymer Blends. Polymers. 2019; 11(1):29. https://doi.org/10.3390/polym11010029
Chicago/Turabian StyleRen, Peng-Gang, Jin Wang, Qian Fan, Song Yang, Zhi-Qiang Wu, Ding-Xiang Yan, and Yan-Hui Chen. 2019. "Synergetic Toughening Effect of Carbon Nanotubes and β-Nucleating Agents on the Polypropylene Random Copolymer/Styrene-Ethylene-Butylene- Styrene Block Copolymer Blends" Polymers 11, no. 1: 29. https://doi.org/10.3390/polym11010029
APA StyleRen, P.-G., Wang, J., Fan, Q., Yang, S., Wu, Z.-Q., Yan, D.-X., & Chen, Y.-H. (2019). Synergetic Toughening Effect of Carbon Nanotubes and β-Nucleating Agents on the Polypropylene Random Copolymer/Styrene-Ethylene-Butylene- Styrene Block Copolymer Blends. Polymers, 11(1), 29. https://doi.org/10.3390/polym11010029






