Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate): Enhancement Strategies for Advanced Applications
Abstract
1. Introduction
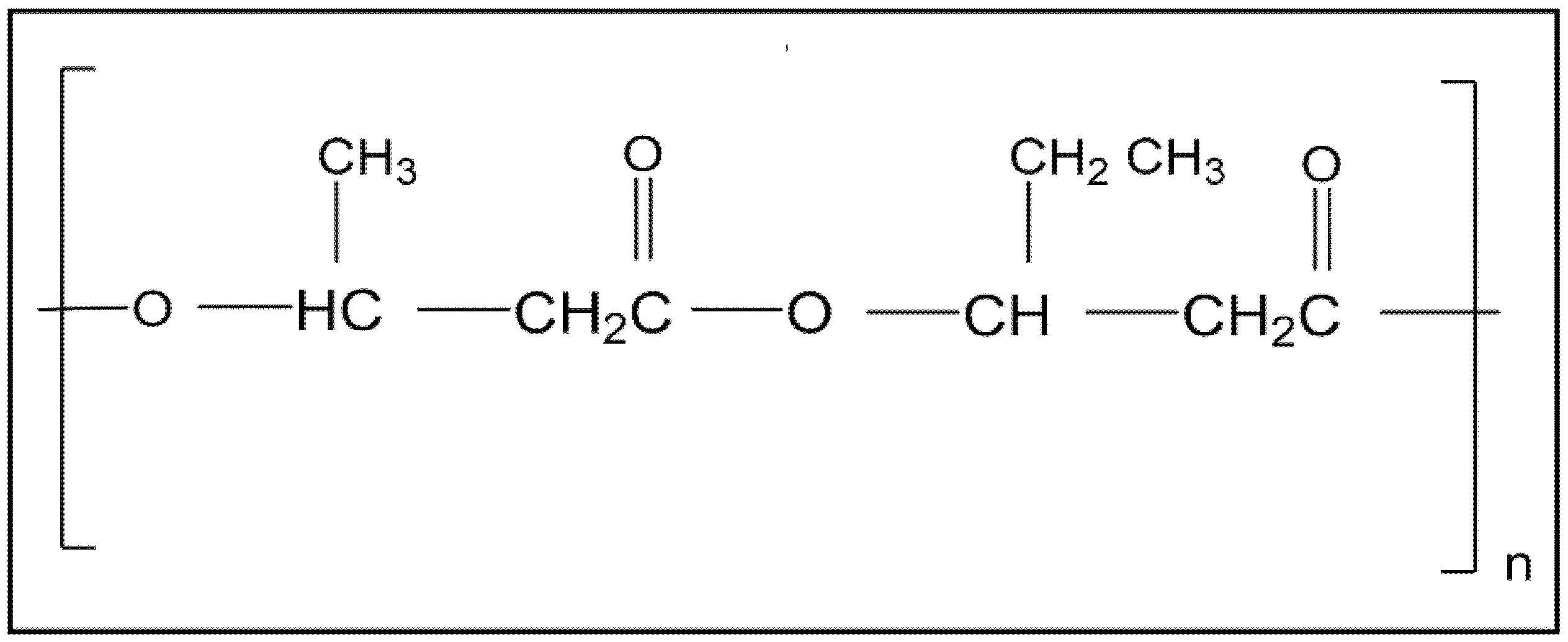
2. PHBV: Chemical Structure and Properties
2.1. Biodegradability
2.2. Molecular Weight
3. Mechanical Reinforcement
3.1. Reinforcement with Other Polymers
3.2. Reinforcement with Natural Fibers
3.3. Reinforcement with Nanomaterials
3.3.1. Carbon Nanomaterials
3.3.2. Nanocellulose
3.3.3. Nanoclays
3.3.4. Nanometals
4. Improvement of Thermal Properties
5. Enhancement of Wettability
5.1. Treatment with Plasma
5.2. Chemical Treatment
5.3. Ultraviolet Radiation
6. Water Absorption Properties
7. Antimicrobial Activity
8. Development of Porous PHBV Scaffolds
8.1. Electrospun PHBV Scaffolds
8.2. PHBV Scaffolds by Leaching
8.3. PHBV Scaffolds by 3D Printing
9. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PHAs | polyhydroxyalkanoates |
| HA | hydroxyalkanoate |
| PHB | polyhydroxybutyrate or poly(3-hydroxybutyrate) |
| PHBHHx | copolymers of 3-hydroxybutyrate and 3-hydroxyhexanoate |
| PHO | poly(3-hydroxyoctanoate) |
| PHBV or PHBHV | poly(3-hydroxybutyrate-co-3-hydroxyvalerate) or poly(hydroxybutyrate-co-hydroxyvalerate) or poly(3-hydroxybutyric acid-co-3-hydroxyvaleric acid) |
| 3HV | 3-hydroxyvalerate |
| MW | molecular weight |
| ZnO | zinc oxide |
| IPNs | interpenetrating polymer networks |
| PLA | polylactic acid |
| PTAcEt | poly(3-thiophene ethyl acetate) |
| PE | polyethylene |
| PHEMA | poly(hydroxyethyl methacrylate) |
| HEMA | hydroxyethyl methacrylate |
| GO | graphene oxide |
| rGO | reduced graphene oxide |
| CNTs | carbon nanotubes |
| SWCNTs | single-wall carbon nanotubes |
| MWCNTs | multi-wall carbon nanotubes |
| PHBV-g-MWCNTs | PHBV-grafted multi-walled carbon nanotubes |
| CNFs | carbon nanofibers |
| GNS | graphene nanosheets |
| CNCs | cellulose nanocrystals |
| CNFs | cellulose nanofibrils |
| NFC | nanofibrillated cellulose |
| POM | polarised optical microscope |
| PBAT | poly(butylene adipate-co-terephthalate) |
| RWF | recycled wood fibre |
| NC | nanoclay |
| WS2 | tungsten disulphide |
| INT-WS2 | tungsten disulphide inorganic nanotubes |
| BN | boron nitride |
| ABOw | aluminium borate whiskers |
| TiO2 | titanium dioxide |
| SEM | scanning electron microscopy |
| TEM | transmission electron microscopy |
| PCL | poly(ε-caprolactone) |
| PBS | poly(butylene succinate) |
| TEC | triethyl citrate |
| DCP | dicumyl peroxide |
| UV | ultraviolet |
| CuO | copper oxide |
| CS | chitosan |
| COS | chitooligosaccharide |
| HAc | hyaluronic acid |
| SPIONs | superparamagnetic iron oxide nanoparticles |
| CEF | antibiotic ceftiofur |
| PEO | polyethylene oxide |
| Hap | hydroxyapatite |
| SCPL | solvent casting particulate leaching |
| SLS | selective laser sintering |
| FDM | fused deposition modelling |
| CAD | computer aided design |
| RP | rapid prototyping |
| µ-CT | microcomputerised tomography |
| CS | calcium silicate |
References
- Ruys, A. Biomimetic Biomaterials: Structure and Applications; Ruys, A.J., Ed.; Woodhead Publishing: Cambridge, UK, 2014; ISBN 978-0-85709-416-2. [Google Scholar]
- Ratner, B.D.; Hoffman, A.S.; Schoen, F.J.; Lemons, J.E. Biomaterials Science: An Introduction to Materials in Medicine; Academic Press: Toronto, ON, Canada, 2012; ISBN 008087780X. [Google Scholar]
- Tadesse, T. Solid and Hazardous Waste Management. Waste Manag. 2004, 120. [Google Scholar] [CrossRef]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Philip, S.; Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Biodegradable polymers with a range of applications. J. Chem. Technol. Biotechnol. 2007, 82, 233–247. [Google Scholar] [CrossRef]
- Khanna, S.; Srivastava, A.K. Recent advances in microbial polyhydroxyalkanoates. Process Biochem. 2005, 40, 607–619. [Google Scholar] [CrossRef]
- Li, Z.; Yang, J.; Loh, X.J. Polyhydroxyalkanoates: Opening doors for a sustainable future. NPG Asia Mater. 2016, 8, e265. [Google Scholar] [CrossRef]
- Braunegg, G.; Lefebvre, G.; Genser, K.F. Polyhydroxyalkanoates, biopolyesters from renewable resources: Physiological and engineering aspects. J. Biotechnol. 1998, 65, 127–161. [Google Scholar] [CrossRef]
- Sudesh, K.; Abe, H.; Yoi, D. Synthesis, structure and properties of polyhydroxyalkanoates: Biological polyesters. Prog. Polym. Sci. 2000, 25, 1503–1555. [Google Scholar] [CrossRef]
- Lee, S. Plastic bacteria Progress and prospects for polyhydroxyalkanoate production in bacteria. Trends Biotechnol. 1996, 14, 431–438. [Google Scholar] [CrossRef]
- Serafim, L.S.; Lemos, P.C.; Oliveira, R.; Reis, M.A.M. Optimization of polyhydroxybutyrate production by mixed cultures submitted to aerobic dynamic feeding conditions. Biotechnol. Bioeng. 2004, 87, 145–1160. [Google Scholar] [CrossRef] [PubMed]
- Avérous, L.; Pollet, E. Biodegradable Polymers; Springer: London, UK, 2012; pp. 13–39. [Google Scholar]
- Anderson, A.J.; Dawes, E. Ocurrence, metabolism, metabolic role and industrial uses of bacterial Polyhydroxyalkanoates. Microbiol. Rev. 1990, 54, 450–472. [Google Scholar] [PubMed]
- Sevastianov, V.I.; Perova, N.V.; Shishatskaya, E.I.; Kalacheva, G.S.; Volova, T.G. Production of purified polyhydroxyalkanoates (PHAs) for applications in contact with blood. J. Biomater. Sci. Polym. Ed. 2003, 14, 1029–1042. [Google Scholar] [CrossRef] [PubMed]
- Juliana Ramsay, B.R. Polyhydroxyalkanoates, Separation, Purification, and Manufacturing Methods. In Encyclopedia of Bioprocess Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2002; pp. 1–23. [Google Scholar]
- Lee, S.Y.; Choi, J., II; Han, K.; Song, J.Y. Removal of endotoxin during purification of poly(3-hydroxybutyrate) from gram-negative bacteria. Appl. Environ. Microbiol. 1999, 65, 2762–2764. [Google Scholar] [PubMed]
- Bugnicourt, E. Polyhydroxyalkanoate (PHA): Review of synthesis, characteristics, processing and potential applications in packaging. Express Polym. 2014, 8, 791–808. [Google Scholar] [CrossRef]
- Kushwah, B.S.; Kushwah, A.V.S.; Singh, V. Towards understanding polyhydroxyalkanoates and their use. J. Polym. Res. 2016, 23. [Google Scholar] [CrossRef]
- Bucci, D.Z.; Tavares, L.B.B.; Sell, I. PHB packaging for the storage of food products. Polym. Test. 2005, 24, 564–571. [Google Scholar] [CrossRef]
- Steinbüchel, A. Biopolymers; Wiley-VCH: Weinheim, Germany, 2001; ISBN 9783527302253. [Google Scholar]
- Guo-Qiang, C.; Qiong, W. The application of polyhydroxyalkanoates as tissue engineering materials. Biomaterials 2005, 26, 6565–6578. [Google Scholar]
- Wang, L.; Du, J.; Cao, D.; Wang, Y. Recent Advances and the Application of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) as Tissue Engineering Materials. J. Macromol. Sci. Part A 2013, 50, 885–893. [Google Scholar] [CrossRef]
- Chang, H.M.; Wang, Z.H.; Luo, H.N.; Xu, M.; Ren, X.Y.; Zheng, G.X.; Wu, B.J.; Zhang, X.H.; Lu, X.Y.; Chen, F.; et al. Poly(3-hydroxybutyrate-co-3-hydroxyhexanoate)-based scaffolds for tissue engineering. Braz. J. Med. Biol. Res. 2014, 47, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.; Liu, X.-B.; Lao, Y.-H.; Wu, L.; Wang, D.; Zuo, Z.-Q. Anti-infective biomaterials with surface-decorated tachyplesin I. Biomaterials 2018. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Wu, L.P.; Liu, X.B.; Hou, J.; Zhao, L.L.; Chen, J.Y.; Zhao, D.H.; Xiang, H. Biodegradation and biocompatibility of haloarchaea-produced poly(3-hydroxybutyrate-co-3-hydroxyvalerate) copolymers. Biomaterials 2017, 139, 172–186. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Wu, L.P.; Hou, J.; Zhao, D.; Xiang, H. Biosynthesis, characterization, and hemostasis potential of tailor-made poly(3-hydroxybutyrate-co-3-hydroxyvalerate) produced by Haloferax mediterranei. Biomacromolecules 2015, 16, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Hou, J.; Zhang, F.; Ai, G.; Li, M.; Cai, S.; Liu, H.; Wang, L.; Wang, Z.; Zhang, S.; et al. Multiple propionyl coenzyme a-supplying pathways for production of the bioplastic poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate) in Haloferax mediterranei. Appl. Environ. Microbiol. 2013, 79, 2922–2931. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.B.; Wu, L.P.; Hou, J.; Chen, J.Y.; Han, J.; Xiang, H. Environmental biodegradation of haloarchaea-produced poly(3-hydroxybutyrate-co-3-hydroxyvalerate) in activated sludge. Appl. Microbiol. Biotechnol. 2016, 100, 6893–6902. [Google Scholar] [CrossRef] [PubMed]
- Rao, U.; Kumar, R.; Balaji, S.; Sehgal, P.K. A novel biocompatible poly (3-hydroxy-co-4-hydroxybutyrate) blend as a potential biomaterial for tissue engineering. J. Bioact. Compat. Polym. 2010, 25, 419–436. [Google Scholar] [CrossRef]
- Smith, J.R.; Lamprou, D.A. Polymer coatings for biomedical applications: A review. Trans. IMF 2014, 92, 9–19. [Google Scholar] [CrossRef]
- Riekes, M.K.; Junior, L.R.; Pereira, R.N.; Borba, P.A.; Fernandes, D.; Stulzer, H.K. Development and evaluation of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) and polycaprolactone microparticles of nimodipine. Curr. Pharm. Des. 2013, 19, 7264–7270. [Google Scholar] [CrossRef] [PubMed]
- Vilos, C.; Morales, F.A.; Solar, P.A.; Herrera, N.S.; Gonzalez-Nilo, F.D.; Aguayo, D.A.; Mendoza, H.L.; Comer, J.; Bravo, M.L.; Gonzalez, P.A.; et al. Paclitaxel-PHBV nanoparticles and their toxicity to endometrial and primary ovarian cancer cells. Biomaterials 2013, 34, 4098–4108. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xue, K.; Li, H.; Sun, J.; Liu, K. Improvement of PHBV scaffolds with bioglass for cartilage tissue engineering. PLoS ONE 2013, 8, e71563. [Google Scholar] [CrossRef] [PubMed]
- Gheibi, A.; Khoshnevisan, K.; Ketabchi, N.; Derakhshan, M.A.; Babadi, A.A. Application of Electrospun Nanofibrous PHBV Scaffold in Neural Graft and Regeneration: A Mini-Review. Nanomed. Res. J. 2016, 1, 107–111. [Google Scholar]
- Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Li, W.; Ding, Y.; Rai, R.; Roether, J.A.; Schubert, D.W.; Boccaccini, A.R. Preparation and characterization of PHBV microsphere/45S5 bioactive glass composite scaffolds with vancomycin releasing function. Mater. Sci. Eng. C 2014, 41, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Sultana, N.; Wang, M. PHBV/PLLA-based composite scaffolds fabricated using an emulsion freezing/freeze-drying technique for bone tissue engineering: Surface modification and in vitro biological evaluation. Biofabrication 2012, 4, 015003. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.; Bastioli, C.; Marini, L.; Würd, D.E. Biopolymers Online—Environmental Assessment of Bio-Based Polymers and Natural Fibres; Utrecht University: Utrecht, The Netherlands, 2005; Volume 59. [Google Scholar]
- Ikada, Y.; Tsuji, H. Biodegradable polyesters for medical and ecological applications. Macromol. Rapid Commun. 2000, 21, 117–132. [Google Scholar] [CrossRef]
- Chen, G.Q. Biofunctionalization of polymers and their applications. In Advances in Biochemical Engineering/Biotechnology; Springer: Berlin/Heidelberg, Germany, 2011; Volume 125, pp. 29–45. [Google Scholar]
- Muthuraj, R.; Misra, M.; Mohanty, A.K. Biodegradable compatibilized polymer blends for packaging applications: A literature review. J. Appl. Polym. Sci. 2018, 135, 45726. [Google Scholar] [CrossRef]
- Xu, Z.; Song, L.; Dai, X.; Chai, X. PHBV polymer supported denitrification system efficiently treated high nitrate concentration wastewater: Denitrification performance, microbial community structure evolution and key denitrifying bacteria. Chemosphere 2018, 197, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Erceg, M.; Kovačić, T.; Perinović, S. Isothermal degradation of poly (3-hydroxybutyrate)/organically modified montmorillonite nanocomposites. Polym. Compos. 2010, 31, 272–278. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Liu, G.M.; Weng, W.Q.; Ding, J.Y.; Liu, S.J. Engineering of Ralstonia eutropha for the production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from glucose. J. Biotechnol. 2015, 195, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhu, Y.; Gu, P.; Li, Y.; Fan, X.; Song, D.; Ji, Y.; Li, Q. Biosynthesis of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) by bacterial community from propylene oxide saponification wastewater residual sludge. Int. J. Biol. Macromol. 2017, 98, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Kunasundari, B.; Sudesh, K. Isolation and recovery of microbial polyhydroxyalkanoates. Express Polym. Lett. 2011, 5, 620–634. [Google Scholar] [CrossRef]
- Weng, Y.X.; Wang, Y.; Wang, X.L.; Wang, Y.Z. Biodegradation behavior of PHBV films in a pilot-scale composting condition. Polym. Test. 2010, 29, 579–587. [Google Scholar] [CrossRef]
- Avella, M.; Martuscelli, E.; Raimo, M. Properties of blends and composites based on poly(3-hydroxy)butyrate (PHB) and poly(3-hydroxybutyrate-hydroxyvalerate) (PHBV) copolymers. J. Mater. Sci. 2000, 35, 523–545. [Google Scholar] [CrossRef]
- Malafaya, P.B.; Silva, G.A.; Reis, R.L. Natural-origin polymers as carriers and scaffolds for biomolecules and cell delivery in tissue engineering applications. Adv. Drug Deliv. Rev. 2007, 59, 207–233. [Google Scholar] [CrossRef] [PubMed]
- Modi, S.J.; Cornish, K.; Koelling, K.; Vodovotz, Y. Fabrication and improved performance of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) for packaging by addition of high molecular weight natural rubber. J. Appl. Polym. Sci. 2016, 133, 9. [Google Scholar] [CrossRef]
- Oner, M.; Ilhan, B. Fabrication of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) biocomposites with reinforcement by hydroxyapatite using extrusion processing. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 65, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Ha, C.S.; Cho, W.J. Miscibility, properties, and biodegradability of microbial polyester containing blends. Prog. Polym. Sci. 2002, 27, 759–809. [Google Scholar] [CrossRef]
- Yan, L.; Kasal, B.; Huang, L. A review of recent research on the use of cellulosic fibres, their fibre fabric reinforced cementitious, geo-polymer and polymer composites in civil engineering. Compos. Part B Eng. 2016, 92, 94–132. [Google Scholar] [CrossRef]
- Aldor, I.S.; Kim, S.W.; Jones Prather, K.L.; Keasling, J.D. Metabolic engineering of a novel propionate-independent pathway for the production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) in recombinant Salmonella enterica serovar typhimurium. Appl. Environ. Microbiol. 2002, 68, 3848–3854. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Fei, Q.; Zhang, Y.H.; Wang, X.Z.; Fan, D.; Chang, H.N. Thermal Properties and Biodegradability Studies of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate). J. Polym. Environ. 2012, 20, 23–28. [Google Scholar] [CrossRef]
- Haris, P.I.; Parvez, I.; Chapman, D. New Biomedical Materials: Basic and Applied Studies; IOS Press: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Yu, L.; Dean, K.; Li, L. Polymer blends and composites from renewable resources. Prog. Polym. Sci. 2006, 31, 576–602. [Google Scholar] [CrossRef]
- Azevedo, H.S.; Reis, R.L. Understanding the enzymatic degradation of biodegradable polymers and strategies to control their degradation rate. Biodegrad. Syst. Tissue Eng. Regen. Med. 2005, 177–201. [Google Scholar] [CrossRef]
- Mergaert, J.; Anderson, C.; Wouters, A.; Swings, J.; Kersters, K.F.E.M.S. Biodegradation of polyhydroxyalkanoates. FEMS Microbiol. Rev. 1992, 9, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Netravali, A.N. A study of physical and mechanical properties of poly (hydroxybutyrate-co-hydroxyvalerate) during composting. Polym. Degrad. Stab. 2003, 80, 59–66. [Google Scholar] [CrossRef]
- Shabina, M.; Afzal, M.; Hameed, S. Bacterial polyhydroxyalkanoates-eco-friendly next generation plastic: Production, biocompatibility, biodegradation, physical properties and applications. Green Chem. Lett. Rev. 2015, 8, 56–77. [Google Scholar]
- Braga, N.F.; da Silva, A.P.; Arantes, T.M.; Lemes, A.P.; Cristovan, F.H. Physical-chemical properties of nanocomposites based on poly (3-hydroxybutyrate-co-3-hydroxyvalerate) and titanium dioxide nanoparticles. Mater. Res. Express 2018, 5, 015303. [Google Scholar] [CrossRef]
- Abdalkarim, S.Y.H.; Yu, H.-Y.; Song, M.-L.; Zhou, Y.; Yao, J.; Ni, Q.-Q. In vitro degradation and possible hydrolytic mechanism of PHBV nanocomposites by incorporating cellulose nanocrystal-ZnO nanohybrids. Carbohydr. Polym. 2017, 176, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Bradel, R.; Reichert, K.H. Modeling of molar mass distribution of poly (D-(–)-3-hydroxybutyrate) during bacterial synthesis. Macromol. Chem. Phys. 1993, 194, 1983–1990. [Google Scholar] [CrossRef]
- Park, T.G. Degradation of poly (d,l-lactic acid) microspheres: Effect of molecular weight. J. Control. Release 1994, 30, 161–173. [Google Scholar] [CrossRef]
- Hsiao, L.J.; Lin, J.H.; Sankatumvong, P.; Wu, T.M.; Li, S.Y. The Feasibility of Thermophilic Caldimonas manganoxidans as a Platform for Efficient PHB Production. Appl. Biochem. Biotechnol. 2016, 180, 852–871. [Google Scholar] [CrossRef] [PubMed]
- Kim, O.; Gross, R.A.; Rutherford, D.R. Bioengineen’ng of poly(B-hydroxyalkanoates) for advanced material applications: Incorporation of cyano and nitrophenoxy side chain substituents. Can. J. Microb. 1995, 41, 32–43. [Google Scholar] [CrossRef]
- Koller, M.; Salerno, A.; Dias, M.; Reiterer, A.; Braunegg, G. Modern biotechnological polymer synthesis: A review. Food Technol. Biotechnol. 2010, 48, 255–269. [Google Scholar]
- Tan, G.Y.A.; Chen, C.L.; Li, L.; Ge, L.; Wang, L.; Razaad, I.M.N.; Li, Y.; Zhao, L.; Mo, Y.; Wang, J.Y. Start a research on biopolymer polyhydroxyalkanoate (PHA): A review. Polymers (Basel) 2014, 6, 706–754. [Google Scholar] [CrossRef]
- Lagaron, J.M.; Lopez-Rubio, A. Nanotechnology for bioplastics: Opportunities, challenges and strategies. Trends Food Sci. Technol. 2011, 22, 611–617. [Google Scholar] [CrossRef]
- Fei, B.; Chen, C.; Chen, S.; Peng, S.; Zhuang, Y.; An, Y.; Dong, L. Crosslinking of poly[(3-hydroxybutyrate)-co-(3-hydroxyvalerate)] using dicumyl peroxide as initiator. Polym. Int. 2004, 53, 937–943. [Google Scholar] [CrossRef]
- Krikštanavičienė, K.; Stanys, S.; Jonaitienė, V. Comparative Investigation of Mechanical–Physical Characteristics of Biodegradable and Non-Degradable Yarns. Autex Res. J. 2014, 14, 61–72. [Google Scholar] [CrossRef]
- Modi, S.; Koelling, K.; Vodovotz, Y. Assessing the mechanical, phase inversion, and rheological properties of poly-[(R)-3-hydroxybutyrate-co-(R)-3-hydroxyvalerate](PHBV) blended with poly-(l-lactic acid)(PLA). Eur. Polym. J. 2013, 49, 3681–3690. [Google Scholar] [CrossRef]
- Recco, M.S.; Floriano, A.C.; Tada, D.B.; Lemes, A.P.; Lang, R.; Cristovan, F.H. Poly (3-hydroxybutyrate-co-valerate)/poly(3-thiophene ethyl acetate) blends as a electroactive biomaterial substrate for tissue engineering application. RSC Adv. 2016, 6, 25330–25338. [Google Scholar] [CrossRef]
- Norrrahim, M.N.F.; Ariffin, H.; Hassan, M.A.; Ibrahim, N.A.; Nishida, H. Performance evaluation and chemical recyclability of a polyethylene/poly (3-hydroxybutyrate-co-3-hydroxyvalerate) blend for sustainable packaging. RSC Adv. 2013, 3, 24378–24388. [Google Scholar] [CrossRef]
- Jost, V.; Kopitzky, R. Blending of Polyhydroxybutyrate-co-valerate with Polylactic Acid for Packaging Applications—Reflections on Miscibility and Effects on the Mechanical and Barrier Properties. Chem. Biochem. Eng. Q. 2015, 29, 221–246. [Google Scholar] [CrossRef]
- Gerard, T.; Budtova, T. Morphology and molten-state rheology of polylactide and polyhydroxyalkanoate blends. Eur. Polym. J. 2012, 48, 1110–1117. [Google Scholar] [CrossRef]
- Zembouai, I.; Kaci, M.; Bruzaud, S.; Dumazert, L.; Bourmaud, A.; Mahlous, M.; Lopez-Cuesta, J.M.; Grohens, Y. Gamma irradiation effects on morphology and properties of PHBV/PLA blends in presence of compatibilizer and Cloisite 30B. Polym. Test. 2016, 49, 29–37. [Google Scholar] [CrossRef]
- Martucci, J.F.; Ruseckaite, R.A. Three-Layer Sheets Based on Gelatin and Poly(lactic Acid), Part 1: Preparation and Properties. J. Appl. Polym. Sci. 2010, 118, 3102–3110. [Google Scholar] [CrossRef]
- Fabra, M.J.; López-Rubio, A.; Lagaron, J.M. On the use of different hydrocolloids as electrospun adhesive interlayers to enhance the barrier properties of polyhydroxyalkanoates of interest in fully renewable food packaging concepts. Food Hydrocoll. 2014, 39, 77–84. [Google Scholar] [CrossRef]
- Dole, P.; Joly, C.; Espuche, E.; Alric, I.; Gontard, N. Gas transport properties of starch based films. Carbohydr. Polym. 2004, 58, 335–343. [Google Scholar] [CrossRef]
- Janssen, L.; Moscicki, L. Thermoplastic Starch: A Green Material for Various Industries; Wiley-VCH: Weinheim, Germany, 2009; Volume 78, pp. 3–16. [Google Scholar]
- Requena, R.; Jiménez, A.; Vargas, M.; Chiralt, A. Effect of plasticizers on thermal and physical properties of compression-moulded poly[(3-hydroxybutyrate)-co-(3-hydroxyvalerate)] films. Polym. Test. 2016, 56, 45–53. [Google Scholar] [CrossRef]
- Gursel, I.; Balcik, C.; Arica, Y.; Akkus, O.; Akkas, N.; Hasirci, V. Synthesis and mechanical properties of interpenetrating networks of polyhydroxybutyrate-co-hydroxyvalerate and polyhydroxyethyl methacrylate. Biomaterials 1998, 19, 1137–1143. [Google Scholar] [CrossRef]
- Raquez, J.M.; Deléglise, M.; Lacrampe, M.F.; Krawczak, P. Thermosetting (bio) materials derived from renewable resources: A critical review. Prog. Polym. Sci. 2010, 35, 487–509. [Google Scholar] [CrossRef]
- John, M.J.; Thomas, S. Biofibres and biocomposites. Carbohydr. Polym. 2008, 71, 343–364. [Google Scholar] [CrossRef]
- Sykacek, E.; Hrabalova, M.; Frech, H.; Mundigler, N. Extrusion of five biopolymers reinforced with increasing wood flour concentration on a production machine, injection moulding and mechanical performance. Compos. Part A Appl. Sci. Manuf. 2009, 40, 1272–1282. [Google Scholar] [CrossRef]
- Singh, S.; Mohanty, A.K. Wood fiber reinforced bacterial bioplastic composites: Fabrication and performance evaluation. Compos. Sci. Technol. 2007, 67, 1753–1763. [Google Scholar] [CrossRef]
- Singh, S.; Mohanty, A.K.; Sugie, T.; Takai, Y.; Hamada, H. Renewable resource based biocomposites from natural fiber and polyhydroxybutyrate-co-valerate (PHBV) bioplastic. Compos. Part A Appl. Sci. Manuf. 2008, 39, 875–886. [Google Scholar] [CrossRef]
- Sahari, J.; Sapuan, S.M. Natural fibre reinforced biodegradable polymer composites. Rev. Adv. Mater. Sci 2011, 30, 166–174. [Google Scholar]
- Berthet, M.A.; Angellier-Coussy, H.; Machado, D.; Hilliou, L.; Staebler, A.; Vicente, A.; Gontard, N. Exploring the potentialities of using lignocellulosic fibres derived from three food by-products as constituents of biocomposites for food packaging. Ind. Crops Prod. 2015, 69, 110–122. [Google Scholar] [CrossRef]
- Berthet, M.A.; Angellier-Coussy, H.; Chea, V.; Guillard, V.; Gastaldi, E.; Gontard, N. Sustainable food packaging: Valorising wheat straw fibres for tuning PHBV-based composites properties. Compos. Part A Appl. Sci. Manuf. 2015, 72, 139–147. [Google Scholar] [CrossRef]
- Dahman, Y.; Kamil, A.; Baena, D. Nanotechnology and Functional Materials for Engineers. Nanotechnol. Funct. Mater. Eng. 2017, 47–66. [Google Scholar] [CrossRef]
- Majeed, K.; Jawaid, M.; Hassan, A.; Bakar, A.A.; Khalil, H.A.; Salema, A.A.; Inuwa, I. Potential materials for food packaging from nanoclay/natural fibres filled hybrid composites. Mater. Des. 2013, 46, 391–410. [Google Scholar] [CrossRef]
- Fuchs, J.N.; Goerbig, M.O. Introduction to the physical properties of graphene. Lecture notes. Carbon N. Y. 2008, 472, 8539. [Google Scholar]
- Kalbacova, M.; Broz, A.; Kong, J.; Kalbac, M. Graphene substrates promote adherence of human osteoblasts and mesenchymal stromal cells. Carbon N. Y. 2010, 48, 4323–4329. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and graphene oxide: Synthesis, properties, and applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Nayak, T.R.; Hong, H.; Cai, W. Graphene: A versatile nanoplatform for biomedical applications. Nanoscale 2012, 4, 3833–3842. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.; Chen, Y. Antibacterial activity of graphite, graphite oxide, graphene oxide, and reduced graphene oxide: Membrane and oxidative stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, V.; Lee, I.; Chun, H.H.; Park, H. Graphene reinforced biodegradable poly (3-hydroxybutyrate-co-4-hydroxybutyrate) nano-composites. Express Polym. Lett. 2013, 7, 320–328. [Google Scholar] [CrossRef]
- Pramanik, N.; De, J.; Basu, R.K.; Rath, T.; Kundu, P.P. Fabrication of magnetite nanoparticle doped reduced graphene oxide grafted polyhydroxyalkanoate nanocomposites for tissue engineering application. RSC Adv. 2016, 6, 46116–46133. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56. [Google Scholar] [CrossRef]
- Avouris, P.; Dresselhaus, G.; Dresselhaus, M.S. Carbon Nanotubes: Synthesis, Structure, Properties and Applications; Springer: Berlin/Heidelberg, Germany, 2000. [Google Scholar]
- Liang, W.; Bockrath, M.; Bozovic, D.; Hafner, J.H.; Tinkham, M.; Park, H. Fabry-Perot interference in a nanotube electron waveguide. Nature 2001, 411, 665–669. [Google Scholar] [CrossRef] [PubMed]
- Cristóvan, F.H.; Machado, J.P.B.; Tada, D.B.; Durán, N.; Lemes, A.P. Effect of MWCNT functionalization on thermal and electrical properties of PHBV/MWCNT nanocomposites. J. Mater. Res. 2015, 30, 55–65. [Google Scholar]
- Shan, G.F.; Gong, X.; Chen, W.P.; Chen, L.; Zhu, M.F. Effect of multi-walled carbon nanotubes on crystallization behavior of poly (3-hydroxybutyrate-co-3-hydroxyvalerate. Colloid Polym. Sci. 2011, 289, 1005–1014. [Google Scholar] [CrossRef]
- Vidhate, S.; Innocentini-Mei, L.; D’Souza, N.A. Mechanical and electrical multifunctional poly (3-hydroxybutyrate-co-3-hydroxyvalerate)—Multiwall carbon nanotube nanocomposites. Polym. Eng. Sci. 2012, 52, 1367–1374. [Google Scholar] [CrossRef]
- Yu, H.Y.; Yao, J.M.; Qin, Z.Y.; Liu, L.; Yang, X.G. Comparison of covalent and noncovalent interactions of carbon nanotubes on the crystallization behavior and thermal properties of poly(3-hydroxybutyrate-co-3-hydroxyvalerate). J. Appl. Polym. Sci. 2013, 130, 4299–4307. [Google Scholar] [CrossRef]
- Yu, H.Y.; Qin, Z.Y.; Sun, B.; Yang, X.G.; Yao, J.M. Reinforcement of transparent poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by incorporation of functionalized carbon nanotubes as a novel bionanocomposite for food packaging. Compos. Sci. Technol. 2014, 94, 96–104. [Google Scholar] [CrossRef]
- Sanchez-Garcia, M.D.; Lagaron, J.M.; Hoa, S.V. Effect of addition of carbon nanofibers and carbon nanotubes on properties of thermoplastic biopolymers. Compos. Sci. Technol. 2010, 70, 1095–1105. [Google Scholar] [CrossRef]
- Tran, P.A.; Zhang, L.; Webster, T.J. Carbon nanofibers and carbon nanotubes in regenerative medicine. Adv. Drug Deliv. Rev. 2009, 61, 1097–1114. [Google Scholar] [CrossRef] [PubMed]
- Llorens-Gámez, M.; Serrano-Aroca, Á. Low-Cost Advanced Hydrogels of Calcium Alginate/Carbon Nanofibers with Enhanced Water Diffusion and Compression Properties. Polymers (Basel) 2018, 10, 405. [Google Scholar] [CrossRef]
- Kim, J.H.; Shim, B.S.; Kim, H.S.; Lee, Y.J.; Min, S.K.; Jang, D.; Kim, J. Review of nanocellulose for sustainable future materials. Int. J. Precis. Eng. Manuf. Technol. 2015, 2, 197–213. [Google Scholar] [CrossRef]
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose nanomaterials review: Structure, properties and nanocomposites. Chem. Soc. Rev. 2011, 40, 3941–3994. [Google Scholar] [CrossRef] [PubMed]
- Jun, D.; Guomin, Z.; Mingzhu, P.; Leilei, Z.; Dagang, L.; Rui, Z. Crystallization and mechanical properties of reinforced PHBV composites using melt compounding: Effect of CNCs and CNFs. Carbohydr. Polym. 2017, 168, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Ten, E.; Bahr, D.F.; Li, B.; Jiang, L.; Wolcott, M.P. Effects of cellulose nanowhiskers on mechanical, dielectric, and rheological properties of poly (3-hydroxybutyrate-co-3-hydroxyvalerate)/cellulose nanowhisker composites. Ind. Eng. Chem. Res. 2012, 5, 2941–2951. [Google Scholar] [CrossRef]
- Yu, H.Y.; Qin, Z.Y.; Zhe, Z.H.O.U. Cellulose nanocrystals as green fillers to improve crystallization and hydrophilic property of poly (3-hydroxybutyrate-co-3-hydroxyvalerate). Prog. Nat. Sci. Mater. Int. 2011, 21, 478–484. [Google Scholar] [CrossRef]
- Yu, H.Y.; Qin, Z.Y.; Liu, Y.N.; Chen, L.; Liu, N.; Zhou, Z. Simultaneous improvement of mechanical properties and thermal stability of bacterial polyester by cellulose nanocrystals. Carbohydr. Polym. 2012, 89, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Srithep, Y.; Ellingham, T.; Peng, J.; Sabo, R.; Clemons, C.; Turng, L.S.; Pilla, S. Melt compounding of poly (3-hydroxybutyrate-co-3-hydroxyvalerate)/nanofibrillated cellulose nanocomposites. Polym. Degrad. Stab. 2013, 98, 1439–1449. [Google Scholar] [CrossRef]
- Carli, L.N.; Crespo, J.S.; Mauler, R.S. PHBV nanocomposites based on organomodified montmorillonite and halloysite: The effect of clay type on the morphology and thermal and mechanical properties. Compos. Part A Appl. Sci. Manuf. 2011, 42, 1601–1608. [Google Scholar] [CrossRef]
- Bittmann, B.; Bouza, R.; Barral, L.; Diez, J.; Ramirez, C. Poly (3-hydroxybutyrate-co-3-hydroxyvalerate)/clay nanocomposites for replacement of mineral oil based materials. Polym. Compos. 2013, 34, 1033–1040. [Google Scholar] [CrossRef]
- Zhao, H.; Cui, Z.; Wang, X.; Turng, L.S.; Peng, X. Processing and characterization of solid and microcellular poly(lactic acid)/polyhydroxybutyrate-valerate (PLA/PHBV) blends and PLA/PHBV/Clay nanocomposites. Compos. Part B Eng. 2013, 51, 79–81. [Google Scholar] [CrossRef]
- Irtegov, Y.; An, V.V.; Vinatier, P.; Sochugov, N.; Zakharov, A. Properties of WS2 Films Prepared by Magnetron Sputtering from a Nanostructured. Adv. Mater. Res. 2014, 872, 197–200. [Google Scholar] [CrossRef]
- Wu, J.S.; Li, J.F.; Zhang, L.; Qian, Z.Y. Effects of environment on dry sliding wear behavior of silver–copper based composites containing tungsten disulfide. Trans. Nonferr. Met. Soc. China 2017, 27, 2202–2213. [Google Scholar] [CrossRef]
- Silverman, T.; Naffakh, M.; Marco, C.; Ellis, G. Morphology and thermal properties of biodegradable poly (hydroxybutyrate-co-hydroxyvalerate)/tungsten disulphide inorganic nanotube nanocomposites. Mater. Chem. Phys. 2016, 170, 145–153. [Google Scholar] [CrossRef]
- Silverman, T.; Naffakh, M.; Marco, C.; Ellis, G. Effect of WS2 Inorganic Nanotubes on Isothermal Crystallization Behavior and Kinetics of Poly(3-Hydroxybutyrate-co-3-hydroxyvalerate). Polymers (Basel) 2018, 10, 166. [Google Scholar] [CrossRef]
- Weng, Q.; Wang, X.; Wang, X.; Bando, Y.; Golberg, D. Functionalized hexagonal boron nitride nanomaterials: Emerging properties and applications. Chem. Soc. Rev. 2016, 45, 3989–4012. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Jin, X.; Xue, Y.; Zhang, X.; Zhang, Y.; Lin, J.; Xu, X.; Meng, F.; Lu, C.Y.; Fan, Y.; et al. Processing and characterizations of nanofiller-modulated poly(3-hydroxybutyrate-co-3-hydroxyvalerate) composites. Polym. Plast. Technol. Eng. 2016, 55, 663–671. [Google Scholar] [CrossRef]
- Öner, M.; Keskin, G.; Kızıl, G.; Pochat-Bohatier, C.; Bechelany, M. Development of poly (3-hydroxybutyrate-co-3-hydroxyvalerate)/boron nitride bionanocomposites with enhanced barrier properties. Polym. Compos. 2017, 10, 24603. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. ZnO-reinforced poly (3-hydroxybutyrate-co-3-hydroxyvalerate) bionanocomposites with antimicrobial function for food packaging. ACS Appl. Mater. Interfaces 2014, 6, 9822–9834. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.S.; Zhu, M.F.; Wu, W.H.; Qin, Z.Y. Reducing the formation of six-membered ring ester during thermal degradation of biodegradable PHBV to enhance its thermal stability. Polym. Degrad. Stab. 2009, 94, 18–24. [Google Scholar] [CrossRef]
- Choi, J.S.; Park, W.H. Effect of biodegradable plasticizers on thermal and mechanical properties of poly(3-hydroxybutyrate). Polym. Test. 2004, 23, 455–460. [Google Scholar] [CrossRef]
- Luo, S.; Netravali, A.N. Effect of 60Co γ-radiation on the properties of poly(hydroxybutyrate-co-hydroxyvalerate). J. Appl. Polym. Sci. 1999, 73, 1059–1067. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, R.; Cai, J.Y.; Liu, Z.; Zheng, Y.; Wang, H.; Li, Q.; He, N. Biosynthesis and Thermal Properties of PHBV Produced from Levulinic Acid by Ralstonia eutropha. PLoS ONE 2013, 8, e60318. [Google Scholar] [CrossRef] [PubMed]
- Mofokeng, J.P.; Luyt, A.S. Morphology and thermal degradation studies of melt-mixed poly (hydroxybutyrate-co-valerate)(PHBV)/poly (ε-caprolactone)(PCL) biodegradable polymer blend nanocomposites with TiO2 as filler. J. Mater. Sci. 2015, 50, 3812–3824. [Google Scholar] [CrossRef]
- Kennouche, S.; Le Moigne, N.; Kaci, M.; Quantin, J.C.; Caro-Bretelle, A.S.; Delaite, C.; Lopez-Cuesta, J.M. Morphological characterization and thermal properties of compatibilized poly (3-hydroxybutyrate-co-3-hydroxyvalerate)(PHBV)/poly (butylene succinate)(PBS)/halloysite ternary nanocomposites. Eur. Polym. J. 2016, 75, 142–162. [Google Scholar] [CrossRef]
- Javadi, A.; Srithep, Y.; Lee, J.; Pilla, S.; Clemons, C.; Gong, S.; Turng, L.S. Processing and characterization of solid and microcellular PHBV/PBAT blend and its RWF/nanoclay composites. Compos. Part A Appl. Sci. Manuf. 2010, 41, 982–990. [Google Scholar] [CrossRef]
- Castro-Mayorga, J.L.; Fabra, M.J.; Pourrahimi, A.M.; Olsson, R.T.; Lagaron, J.M. The impact of zinc oxide particle morphology as an antimicrobial and when incorporated in poly(3-hydroxybutyrate-co-3-hydroxyvalerate) films for food packaging and food contact surfaces applications. Food Bioprod. Process. 2017, 101, 32–44. [Google Scholar] [CrossRef]
- Wang, B.J.; Zhang, Y.J.; Zhang, J.Q.; Gou, Q.T.; Wang, Z.B.; Chen, P.; Gu, Q. Crystallization behavior, thermal and mechanical properties of PHBV/graphene nanosheet composites. Chin. J. Polym. Sci. 2013, 31, 670–678. [Google Scholar] [CrossRef]
- Nakagawa, M.; Teraoka, F.; Fujimoto, S.; Hamada, Y.; Kibayashi, H.; Takahashi, J. Improvement of cell adhesion on poly (L-lactide) by atmospheric plasma treatment. J. Biomed. Mater. Res. Part A 2006, 44, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.; Liu, C.; Zhang, X.; Xiao, M.; Wu, G. Surface Modification of Polyhydroxyalkanoates toward Enhancing Cell Compatibility and Antibacterial Activity. Macromol. Mater. Eng. 2017, 302, 1700258. [Google Scholar] [CrossRef]
- Gogolewski, S.; Mainil-Varlet, P.; Dillon, J.G. Sterility, mechanical properties, and molecular stability of polylactide internal-fixation devices treated with low-temperature plasmas. J. Biomed. Mater. Res. Part A 1996, 32, 227–235. [Google Scholar] [CrossRef]
- Denes, F.S.; Manolache, S. Macromolecular plasma-chemistry: An emerging field of polymer science. Prog. Polym. Sci. 2004, 29, 815–885. [Google Scholar] [CrossRef]
- Chan, C.M.; Ko, T.M.; Hiraoka, H. Polymer surface modification by plasmas and photons. Surf. Sci. Rep. 1996, 24, 1–54. [Google Scholar] [CrossRef]
- Ferreira, B.M.P.; Pinheiro, L.M.P.; Nascente, P.A.P.; Ferreira, M.J.; Duek, E.A.R. Plasma surface treatments of poly(l-lactic acid) (PLLA) and poly(hydroxybutyrate-co-hydroxyvalerate) (PHBV). Mater. Sci. Eng. C 2009, 29, 806–813. [Google Scholar] [CrossRef]
- Lucchesi, C.; Ferreira, B.M.; Duek, E.A.; Santos, A.R.; Joazeiro, P.P. Increased response of Vero cells to PHBV matrices treated by plasma. J. Mater. Sci. Mater. Med. 2008, 19, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Unalan, I.; Colpankan, O.; Albayrak, A.Z.; Gorgun, C.; Urkmez, A.S. Biocompatibility of plasma-treated poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanofiber mats modified by silk fibroin for bone tissue regeneration. Mater. Sci. Eng. C 2016, 68, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.K.; Wang, H.M.D.; Lan, J.C.W. Investigation and characterization of plasma-treated poly(3-hydroxybutyrate) and s(3-hydroxybutyrateco-3-hydroxyvalerate) biopolymers for an in vitro cellular study of mouse adipose-derived stem cells. Polymers (Basel) 2018, 10, 355. [Google Scholar] [CrossRef]
- García-García, J.M.; Quijada-Garrido, I.; López, L.; París, R.; Núñez-López, M.T.; De La Peña Zarzuelo, E.; Garrido, L. The surface modification of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) copolymers to improve the attachment of urothelial cells. Mater. Sci. Eng. C 2013, 33, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zaloga, J.; Ding, Y.; Liu, Y.; Janko, C.; Pischetsrieder, M.; Alexiou, C.; Boccaccini, A.R. Facile preparation of multifunctional superparamagnetic PHBV microspheres containing SPIONs for biomedical applications. Sci. Rep. 2016, 6, 23140. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Gao, Z.; Hu, X.; Wang, Z.; Su, T.; Yang, L.; Yan, S. Blending Modification of PHBV/PCL and its Biodegradation by Pseudomonas mendocina. J. Polym. Environ. 2017, 25, 156–164. [Google Scholar] [CrossRef]
- Wang, Y.; Ke, Y.; Ren, L.; Wu, G.; Chen, X.; Zhao, Q. Surface engineering of PHBV by covalent collagen immobilization to improve cell compatibility. J. Biomed. Mater. Res. Part A 2009, 88, 616–627. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, R.; Lazare, S. Modification of polymer surfaces by far-ultraviolet radiation of low and high (laser) intensities. Polymer (Guildf.) 1985, 26, 1297–1300. [Google Scholar] [CrossRef]
- Ke, Y.; Wang, Y.; Ren, L. Surface modification of PHBV scaffolds via UV polymerization to improve hydrophilicity. J. Biomater. Sci. Polym. Ed. 2010, 21, 1589–1602. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.; Wang, Y.J.; Ren, L.; Zhao, Q.C.; Huang, W. Modified PHBV scaffolds by in situ UV polymerization: Structural characteristic, mechanical properties and bone mesenchymal stem cell compatibility. Acta Biomater. 2010, 6, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Hassaini, L.; Kaci, M.; Benhamida, A.; Bruzaud, S.; Pillin, I.; Grohens, Y. The effects of PHBV-g-MA compatibilizer on morphology and properties of poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/olive husk flour composites. J. Adhes. Sci. Technol. 2016, 30, 2061–2080. [Google Scholar] [CrossRef]
- Dangtungee, R.; Tengsuthiwat, J.; Boonyasopon, P.; Siengchin, S. Sisal natural fiber/clay-reinforced poly(hydroxybutyrate-co-hydroxyvalerate) hybrid composites. J. Thermoplast. Compos. Mater. 2015, 28, 879–895. [Google Scholar] [CrossRef]
- Wolf, C.; Guillard, V.; Angellier-Coussy, H.; Silva, G.G.D.; Gontard, N. Water vapor sorption and diffusion in wheat straw particles and their impact on the mass transfer properties of biocomposites. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Tang, C.Y.; Chen, D.Z.; Yue, T.M.; Chan, K.C.; Tsui, C.P.; Yu, P.H.F. Water absorption and solubility of PHBHV/HA nanocomposites. Compos. Sci. Technol. 2008, 68, 1927–1934. [Google Scholar] [CrossRef]
- WHO. WHO|Antibiotic Resistance; World Health Organization: Roman, Italy, 2017. [Google Scholar]
- Castro-Mayorga, J.L.; Fabra, M.J.; Lagaron, J.M. Stabilized nanosilver based antimicrobial poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanocomposites of interest in active food packaging. Innov. Food Sci. Emerg. Technol. 2016, 33, 524–533. [Google Scholar] [CrossRef]
- Muñoz-Bonilla, A.; Fernández-García, M. Polymeric Materials with Antimicrobial Activity. Prog. Polym. Sci. 2013, 37, 281–339. [Google Scholar] [CrossRef]
- Benchaar, C.; Calsamiglia, S.; Chaves, A.V.; Fraser, G.R.; Colombatto, D.; McAllister, T.A.; Beauchemin, K. A review of plant-derived essential oils in ruminant nutrition and production. Anim. Feed Sci. Technol. 2008, 145, 209–228. [Google Scholar] [CrossRef]
- Debiagi, F.; Kobayashi, R.K.; Nakazato, G.; Panagio, L.A.; Mali, S. Biodegradable active packaging based on cassava bagasse, polyvinyl alcohol and essential oils. Ind. Crops Prod. 2014, 52, 664–670. [Google Scholar] [CrossRef]
- Emiroğlu, Z.K.; Yemiş, G.P.; Coşkun, B.K.; Candoğan, K. Antimicrobial Activity of soy edible films incorporated with thyme and oregano essential oils on fresh ground beef patties. Meat Sci. 2010, 86, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Kocić-Tanackov, S.; Dimić, G.; Tanackov, I.; Pejin, D.; Mojović, L.; Pejin, J. The inhibitory effect of oregano extract on the growth of Aspergillus spp. and on sterigmatocystin biosynthesis. LWT Food Sci. Technol. 2012, 49, 14–20. [Google Scholar] [CrossRef]
- Kocić-Tanackov, S.; Dimic, G.; Tanackov, I.; Pejin, D.; Mojovic, L.; Pejin, J. Antifungal activity of oregano (Origanum vulgare L.) extract on the growth of Fusarium and Penicillium species isolated from food. Hem. Ind. 2012, 66, 33–41. [Google Scholar] [CrossRef]
- Requena, R.; Jiménez, A.; Vargas, M.; Chiralt, A. Poly[(3-hydroxybutyrate)-co-(3-hydroxyvalerate)] active bilayer films obtained by compression moulding and applying essential oils at the interface. Polym. Int. 2016, 65, 883–891. [Google Scholar] [CrossRef]
- Xing, Z.C.; Chae, W.P.; Baek, J.Y.; Choi, M.J.; Jung, Y.; Kang, I.K. In vitro assessment of antibacterial activity and cytocompatibility of silver-containing PHBV nanofibrous scaffolds for tissue engineering. Biomacromolecules 2010, 11, 1248–1253. [Google Scholar] [CrossRef] [PubMed]
- Castro-Mayorga, J.L.; Martínez-Abad, A.; Fabra, M.J.; Olivera, C.; Reis, M.; Lagarón, J.M. Stabilization of antimicrobial silver nanoparticles by a polyhydroxyalkanoate obtained from mixed bacterial culture. Int. J. Biol. Macromol. 2014, 71, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Castro-Mayorga, J.L.; Fabra-Rovira, M.J.; Cabedo-Mas, L.; Sánchez-Moragas, G.; Lagarón-Cabello, J.M. Antimicrobial nanocomposites and electrospun coatings based on poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and copper oxide nanoparticles for active packaging and coating applications. J. Appl. Polym. Sci. 2018, 135, 45673. [Google Scholar] [CrossRef]
- Abdalkarim, S.Y.H.; Yu, H.Y.; Wang, D.; Yao, J. Electrospun poly (3-hydroxybutyrate-co-3-hydroxy-valerate)/cellulose reinforced nanofibrous membranes with ZnO nanocrystals for antibacterial wound dressings. Cellulose 2017, 24, 2925–2938. [Google Scholar] [CrossRef]
- Hu, S.G.; Jou, C.H.; Yang, M.C. Protein adsorption, fibroblast activity and antibacterial properties of poly(3-hydroxybutyric acid-co-3-hydroxyvaleric acid) grafted with chitosan and chitooligosaccharide after immobilized with hyaluronic acid. Biomaterials 2003, 24, 2685–2693. [Google Scholar] [CrossRef]
- Solar, P.; González, G.; Vilos, C.; Herrera, N.; Juica, N.; Moreno, M.; Simon, F.; Velásquez, L. Multifunctional polymeric nanoparticles doubly loaded with SPION and ceftiofur retain their physical and biological properties. J. Nanobiotechnol. 2015, 13, 14. [Google Scholar] [CrossRef] [PubMed]
- Zine, R.; Sinha, M. Nanofibrous poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/collagen/graphene oxide scaffolds for wound coverage. Mater. Sci. Eng. C 2017, 80, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Raeisdasteh Hokmabad, V.; Davaran, S.; Ramazani, A.; Salehi, R. Design and fabrication of porous biodegradable scaffolds: A strategy for tissue engineering. J. Biomater. Sci. Polym. Ed. 2017, 28, 1797–1825. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-E.; Kim, Y.-J. Inhibition Effect of Low Molecular Weight α-/β-Glucans Loaded Nanofibers on the Growth of Skin Cancer Cells. Polym. Korea 2017, 41, 662–669. [Google Scholar] [CrossRef]
- Monleón-Pradas, M.; Gómez-Ribelles, J.L.; Serrano-Aroca, Á.; Gallego-Ferrer, G.; Suay-Antón, J.; Pissis, P. Porous poly(2-hydroxyethyl acrylate) hydrogels. Polymer (Guildf.) 2001, 42, 4667–4674. [Google Scholar] [CrossRef]
- Serrano-Aroca, Á.; Monleón-Pradas, M.; Gómez-Ribelles, J.L. Macroporous poly(methyl methacrylate) produced by phase separation during polymerisation in solution. Colloid Polym. Sci. 2007, 285, 753–760. [Google Scholar] [CrossRef]
- Serrano-Aroca, Á.; Llorens-Gámez, M. Dynamic mechanical analysis and water vapour sorption of highly porous poly(methyl methacrylate). Polymer (Guildf.) 2017, 125, 58–65. [Google Scholar] [CrossRef]
- Serrano-Aroca, Á.; Campillo-Fernández, A.J.; Gómez-Ribelles, J.L.; Monleón-Pradas, M.; Gallego-Ferrer, G.; Pissis, P. Porous poly(2-hydroxyethyl acrylate) hydrogels prepared by radical polymerisation with methanol as diluent. Polymer (Guildf.) 2004, 45, 8949–8955. [Google Scholar] [CrossRef]
- Rodríguez-Hernández, J.C.; Serrano-Aroca, Á.; Gómez-Ribelles, J.L.; Monleón-Pradas, M. Three-dimensional nanocomposite scaffolds with ordered cylindrical orthogonal pores. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 84, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Brígido-Diego, R.; Pérez-Olmedilla, M.; Serrano-Aroca, Á.; Gómez-Ribelles, J.L.; Monleón-Pradas, M.; Gallego-Ferrer, G.; Salmerón-Sánchez, M. Acrylic scaffolds with interconnected spherical pores and controlled hydrophilicity for tissue engineering. J. Mater. Sci. Mater. Med. 2005, 40, 4881–4887. [Google Scholar] [CrossRef]
- Wang, N.; Zhou, Z.; Xia, L.; Dai, Y.; Liu, H. Fabrication and characterization of bioactive β-Ca2SiO4/PHBV composite scaffolds. Mater. Sci. Eng. C 2013, 33, 2294–2301. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Calderone, M.; Cacciotti, I. Electrospun PHBV/PEO co-solution blends: Microstructure, thermal and mechanical properties. Mater. Sci. Eng. C 2013, 33, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhu, M.; Zhang, J.; Zhang, Y.; Liu, Z.; Zhu, Y.; Zhang, C. Three dimensionally printed mesoporous bioactive glass and poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) composite scaffolds for bone regeneration. J. Mater. Chem. B 2014, 2, 6106. [Google Scholar] [CrossRef]
- Kosorn, W.; Sakulsumbat, M.; Uppanan, P.; Kaewkong, P.; Chantaweroad, S.; Jitsaard, J.; Sitthiseripratip, K.; Janvikul, W. PCL/PHBV blended three dimensional scaffolds fabricated by fused deposition modeling and responses of chondrocytes to the scaffolds. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 1141–1150. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Vahabzadeh, S.; Bandyopadhyay, A. Bone tissue engineering using 3D printing. Mater. Today 2013, 16, 496–504. [Google Scholar] [CrossRef]
- Duan, B.; Wang, M.; Zhou, W.Y.; Cheung, W.L.; Li, Z.Y.; Lu, W.W. Three-dimensional nanocomposite scaffolds fabricated via selective laser sintering for bone tissue engineering. Acta Biomater. 2010, 6, 4495–4505. [Google Scholar] [CrossRef] [PubMed]
- Van Blitterswijk, C.; De Boer, J. Tissue Engineering; Academic Press: Oxford, UK, 2014. [Google Scholar]
- Tong, H.W.; Zhang, X.; Wang, M. A new nanofiber fabrication technique based on coaxial electrospinning. Mater. Lett. 2012, 66, 257–260. [Google Scholar] [CrossRef]
- Haider, A.; Haider, S.; Kang, I.K. A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arab. J. Chem. 2015. [Google Scholar] [CrossRef]
- Cho, Y.S.; Hong, M.W.; Kim, S.Y.; Lee, S.J.; Lee, J.H.; Kim, Y.Y.; Cho, Y.S. Fabrication of dual-pore scaffolds using SLUP (salt leaching using powder) and WNM (wire-network molding) techniques. Mater. Sci. Eng. C 2014, 45, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Li, Y.; Zhou, Z.; Dai, Y.; Liu, H.; Liu, H. Icariin delivery porous PHBV scaffolds for promoting osteoblast expansion in vitro. Mater. Sci. Eng. C 2013, 33, 3545–3552. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.-Y.; Xing, Z.-C.; Kwak, G.; Yoon, K.-B.; Park, S.-Y.; Park, L.S.; Kang, I.-K. Fabrication and Characterization of Collagen-Immobilized Porous PHBV/HA Nanocomposite Scaffolds for Bone Tissue Engineering. J. Nanomater. 2012, 2012, 171804. [Google Scholar] [CrossRef]
- Che Johari, N.S.; Aizad, S.; Zubairi, S.I. Efficacy Study of Carrageenan as an Alternative Infused Material (Filler) in Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) Porous 3D Scaffold. Int. J. Polym. Sci. 2017, 2017. [Google Scholar] [CrossRef]
- Bikas, H.; Stavropoulos, P.; Chryssolouris, G. Additive manufacturing methods and modelling approaches: A critical review. Int. J. Adv. Manuf. Technol. 2016, 83, 389–405. [Google Scholar] [CrossRef]
- Bose, S.; Darsell, J.; Hosick, H.; Yang, L.; Sarkar, D.K.; Bandyopadhyay, A. Processing and characterization of porous alumina scaffolds. J. Mater. Sci. Mater. Med. 2002, 13, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Darsell, J.; Bose, S.; Hosick, H.L.; Bandyopadhyay, A. From CT scan to ceramic bone graft. J. Am. Ceram. Soc. 2003, 86, 1076–1080. [Google Scholar] [CrossRef]
- Chia, H.N.; Wu, B.M. Recent advances in 3D printing of biomaterials. J. Biol. Eng. 2015, 9, 4. [Google Scholar] [CrossRef] [PubMed]
- Zein, I.; Hutmacher, D.W.; Tan, K.C.; Teoh, S.H. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials 2002, 23, 1169–1185. [Google Scholar] [CrossRef]
- Duan, B.; Cheung, W.L.; Wang, M. Optimized fabrication of Ca-P/PHBV nanocomposite scaffolds via selective laser sintering for bone tissue engineering. Biofabrication 2011, 3, 015001. [Google Scholar] [CrossRef] [PubMed]
- Shuai, C.; Guo, W.; Gao, C.; Yang, Y.; Xu, Y.; Liu, L.; Qin, T.; Sun, H.; Yang, S.; Feng, P.; et al. Calcium silicate improved bioactivity and mechanical properties of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) scaffolds. Polymers (Basel) 2017, 9, 175. [Google Scholar] [CrossRef]
- Ortega, I.; Dew, L.; Kelly, A.G.; Chong, C.K.; MacNeil, S.; Claeyssens, F. Fabrication of biodegradable synthetic perfusable vascular networks via a combination of electrospinning and robocasting. Biomater. Sci. 2015, 3, 592–596. [Google Scholar] [CrossRef] [PubMed]





| Properties | PHB | PHBV |
|---|---|---|
| Density (g/cm3) | 1.25 | 1.25 |
| Elasticity modulus (GPa) | 0.93 | 2.38 |
| Traction Resistance (MPa) | 21 | 25.9 |
| Elongation (%) | 5.2–8.4 | 1.4 |
| Fusion temperature (°C) | 161 | 153 |
| Glass transition temperature (°C) | −10 | −1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivera-Briso, A.L.; Serrano-Aroca, Á. Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate): Enhancement Strategies for Advanced Applications. Polymers 2018, 10, 732. https://doi.org/10.3390/polym10070732
Rivera-Briso AL, Serrano-Aroca Á. Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate): Enhancement Strategies for Advanced Applications. Polymers. 2018; 10(7):732. https://doi.org/10.3390/polym10070732
Chicago/Turabian StyleRivera-Briso, Ariagna L., and Ángel Serrano-Aroca. 2018. "Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate): Enhancement Strategies for Advanced Applications" Polymers 10, no. 7: 732. https://doi.org/10.3390/polym10070732
APA StyleRivera-Briso, A. L., & Serrano-Aroca, Á. (2018). Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate): Enhancement Strategies for Advanced Applications. Polymers, 10(7), 732. https://doi.org/10.3390/polym10070732







