Chemical and Electrochemical Synthesis of Polypyrrole Using Carrageenan as a Dopant: Polypyrrole/Multi-Walled Carbon Nanotube Nanocomposites
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Carrageenan Solutions
2.3. Chemical Synthesis of Polypyrrole
2.4. Chemical Synthesis of PPy/CNT Composites
2.5. Electrochemical Synthesis of PPy
2.6. Preparation of Carrageenan-CNT Dispersions
2.7. Preparation of Films by Evaporative Casting
3. Characterization Techniques
3.1. UV-Vis Spectroscopy
3.2. Electrical Conductivity Measurements
3.3. Optical Microscopy
3.4. Scanning Electron Microscope
3.5. Tensile Testing
3.6. Thermogravimetric Measurements
3.7. Cyclic Voltammetry
4. Results and Discussion
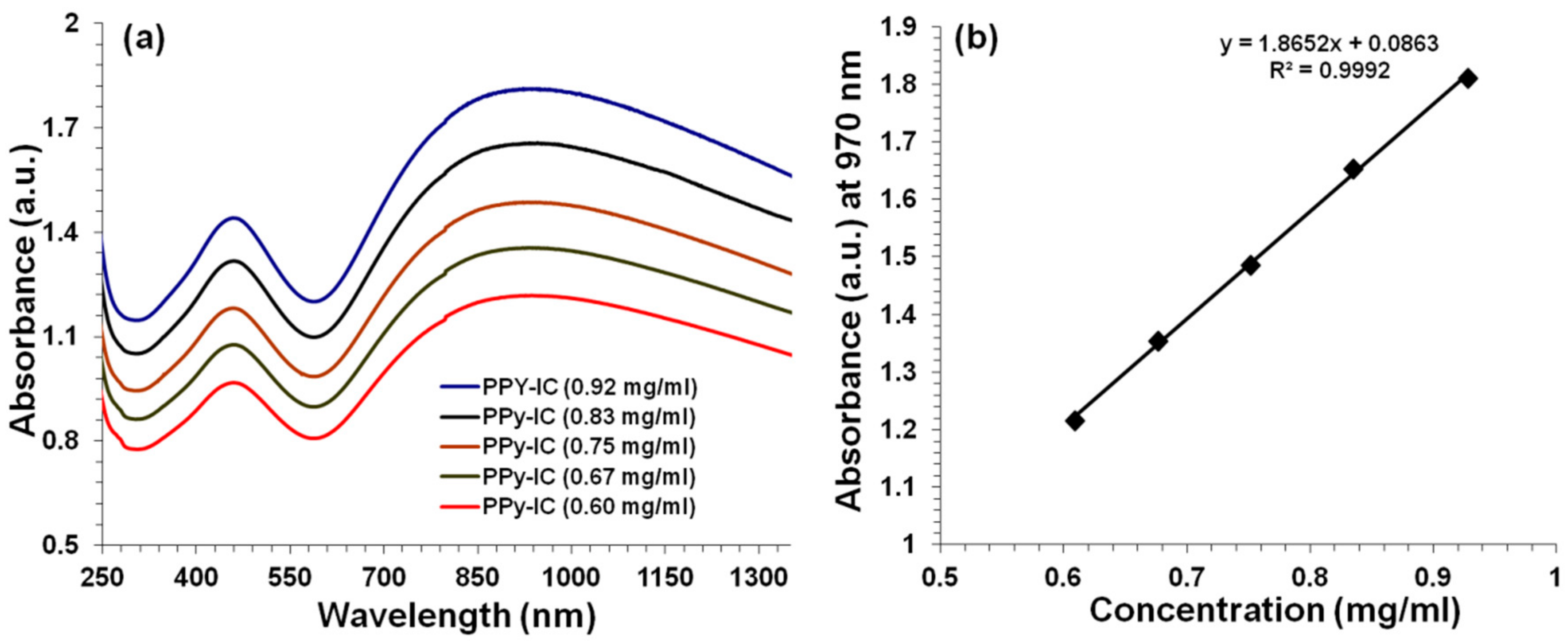
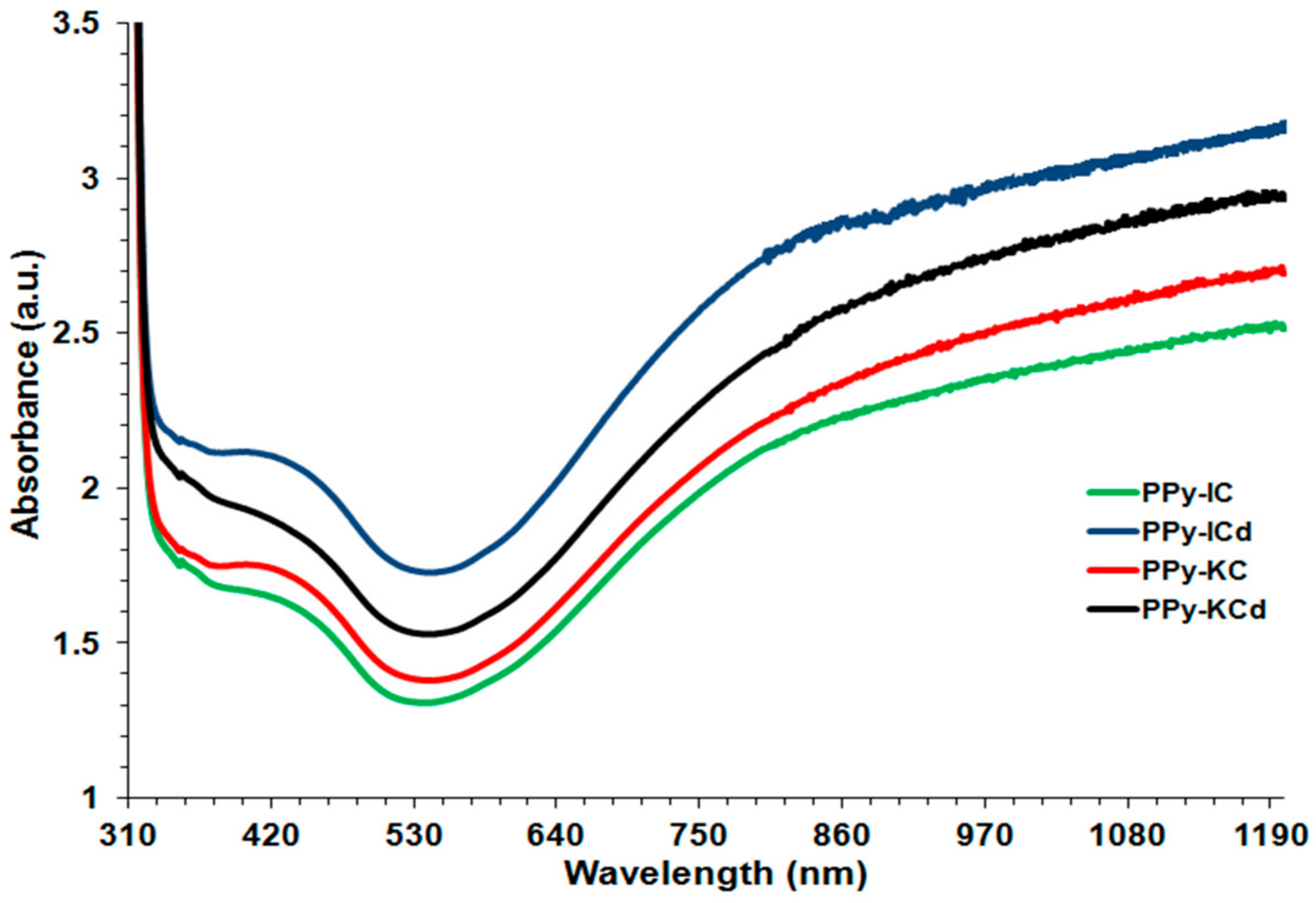
4.1. UV-Visible Spectroscopy
4.2. Microscopy of Composite Films
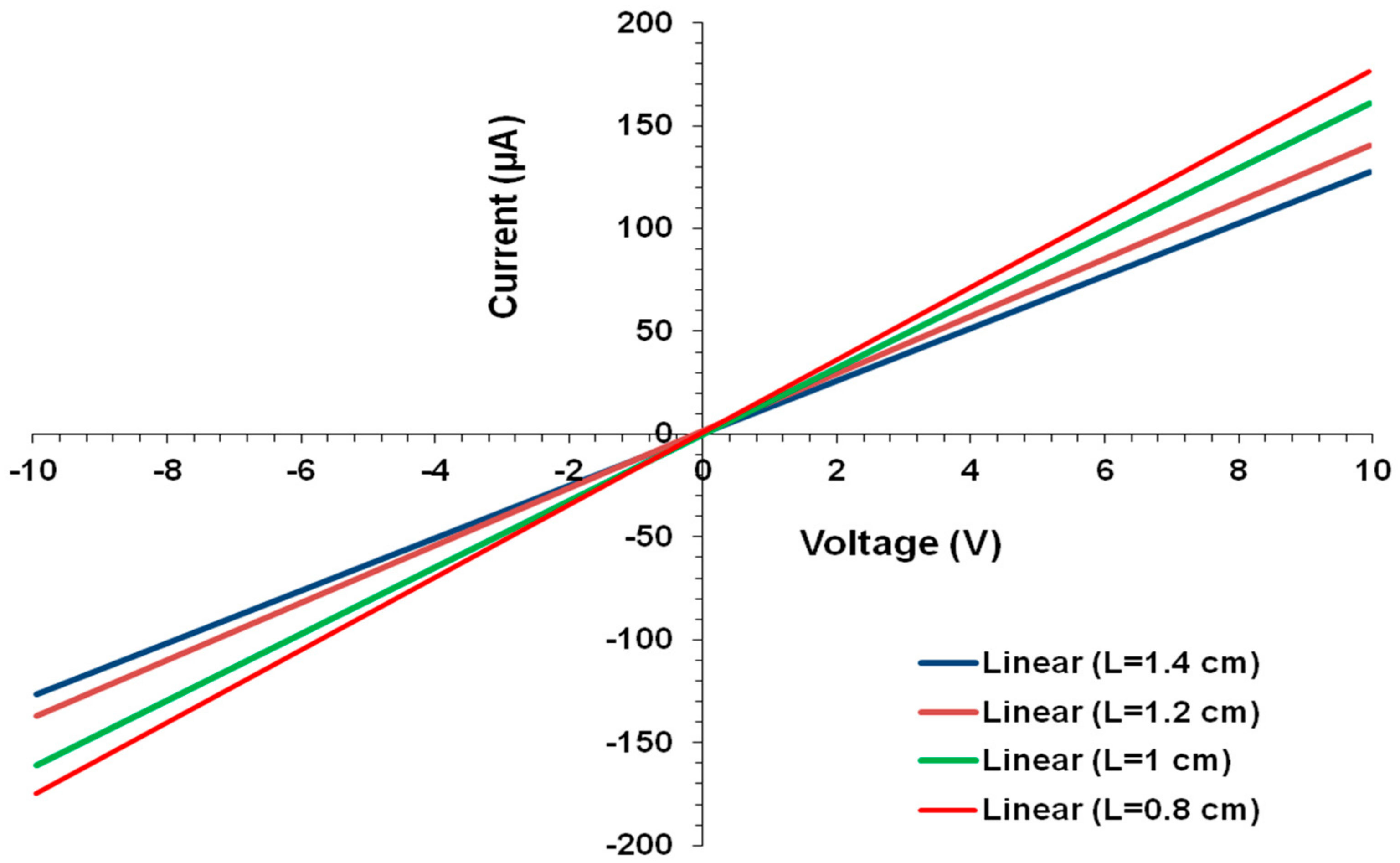
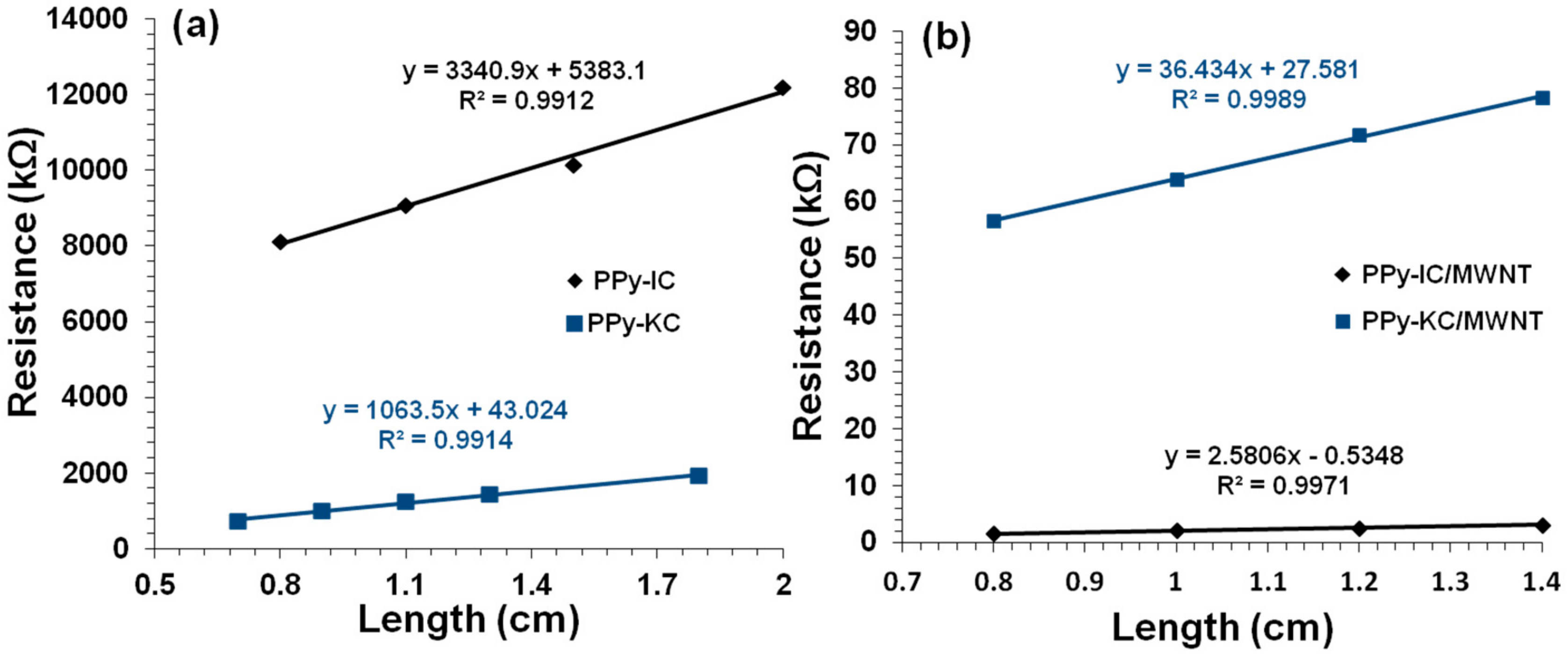
4.3. Electrical Conductivity of Composite Films
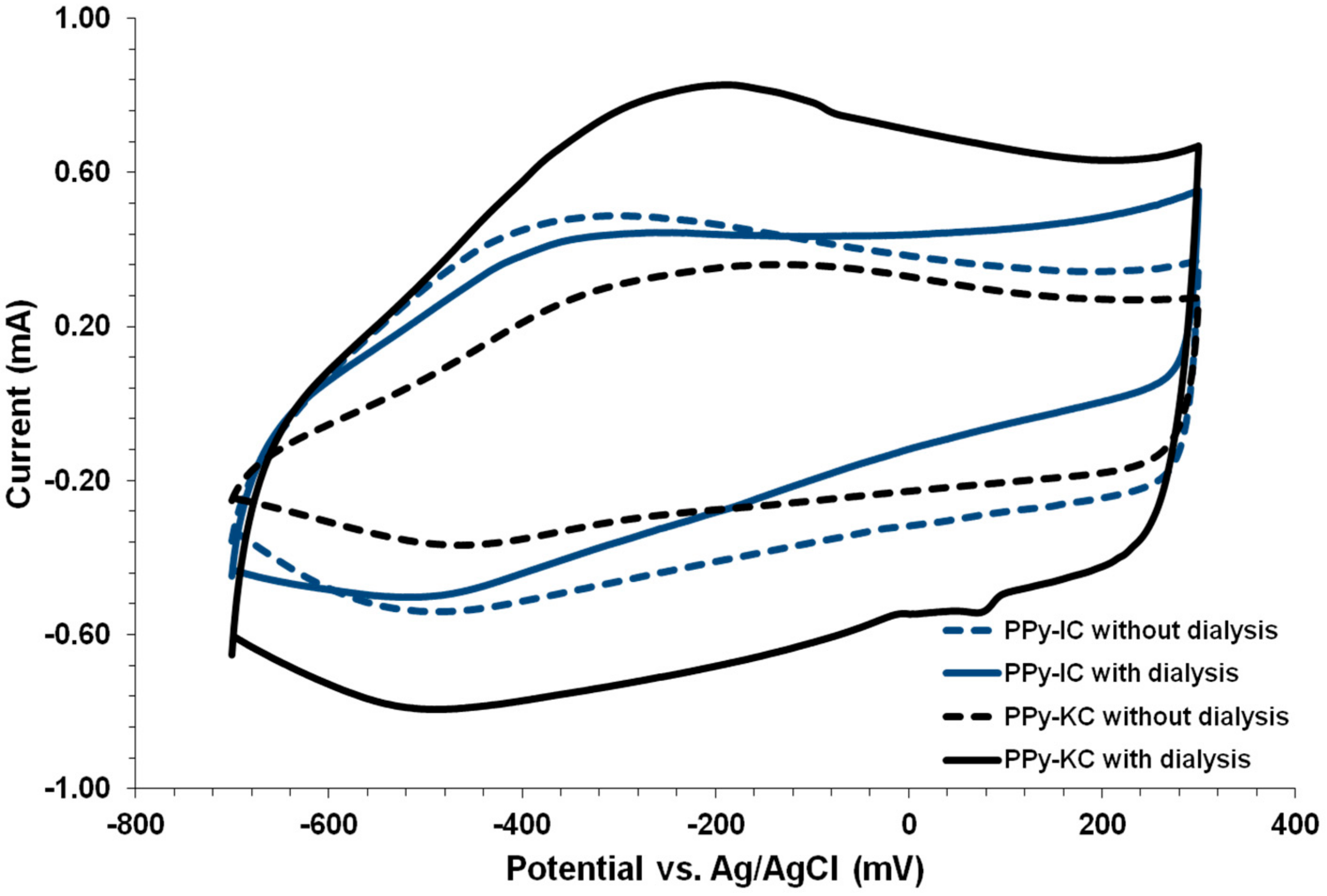
4.4. Cyclic Voltammetry of PPy Films
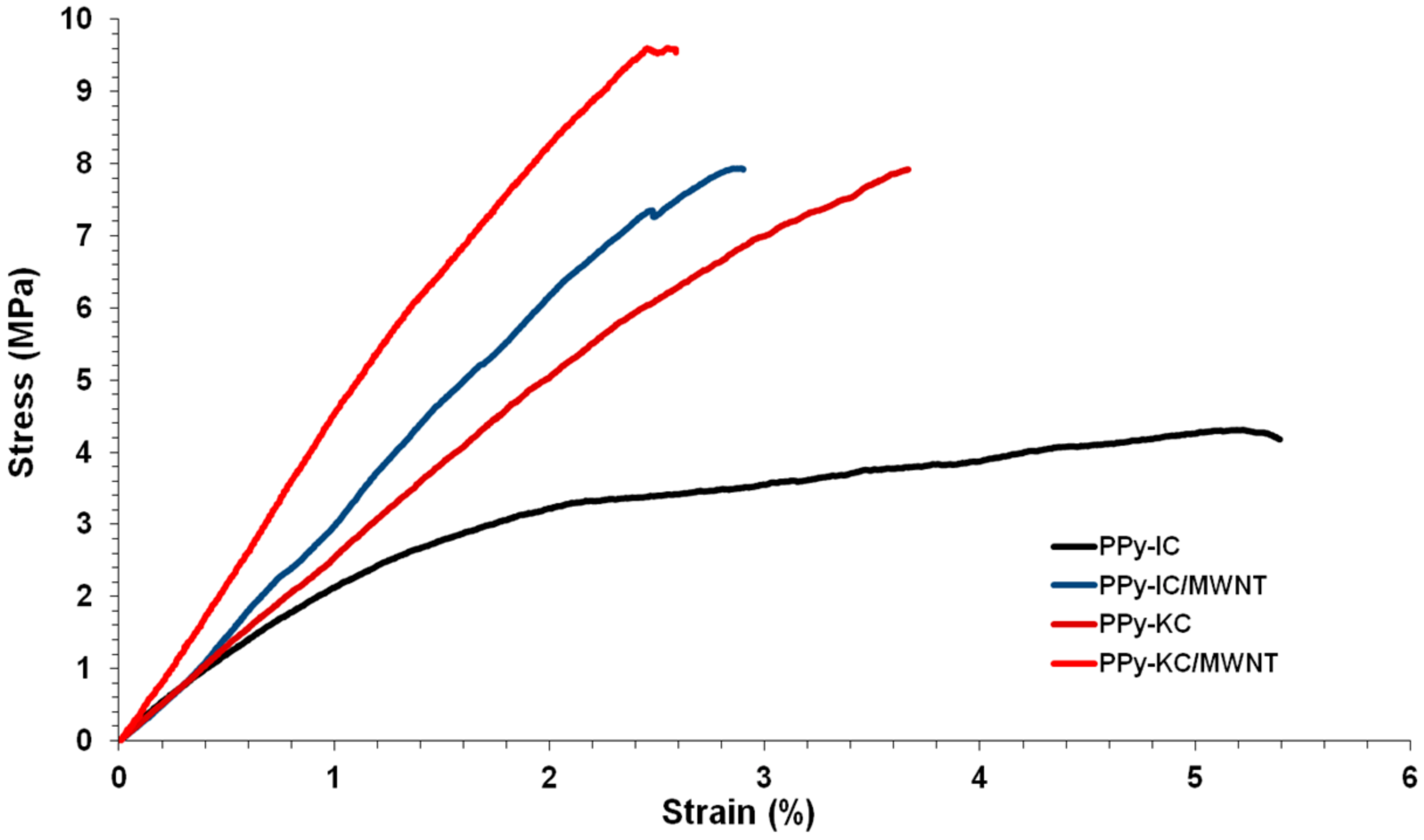
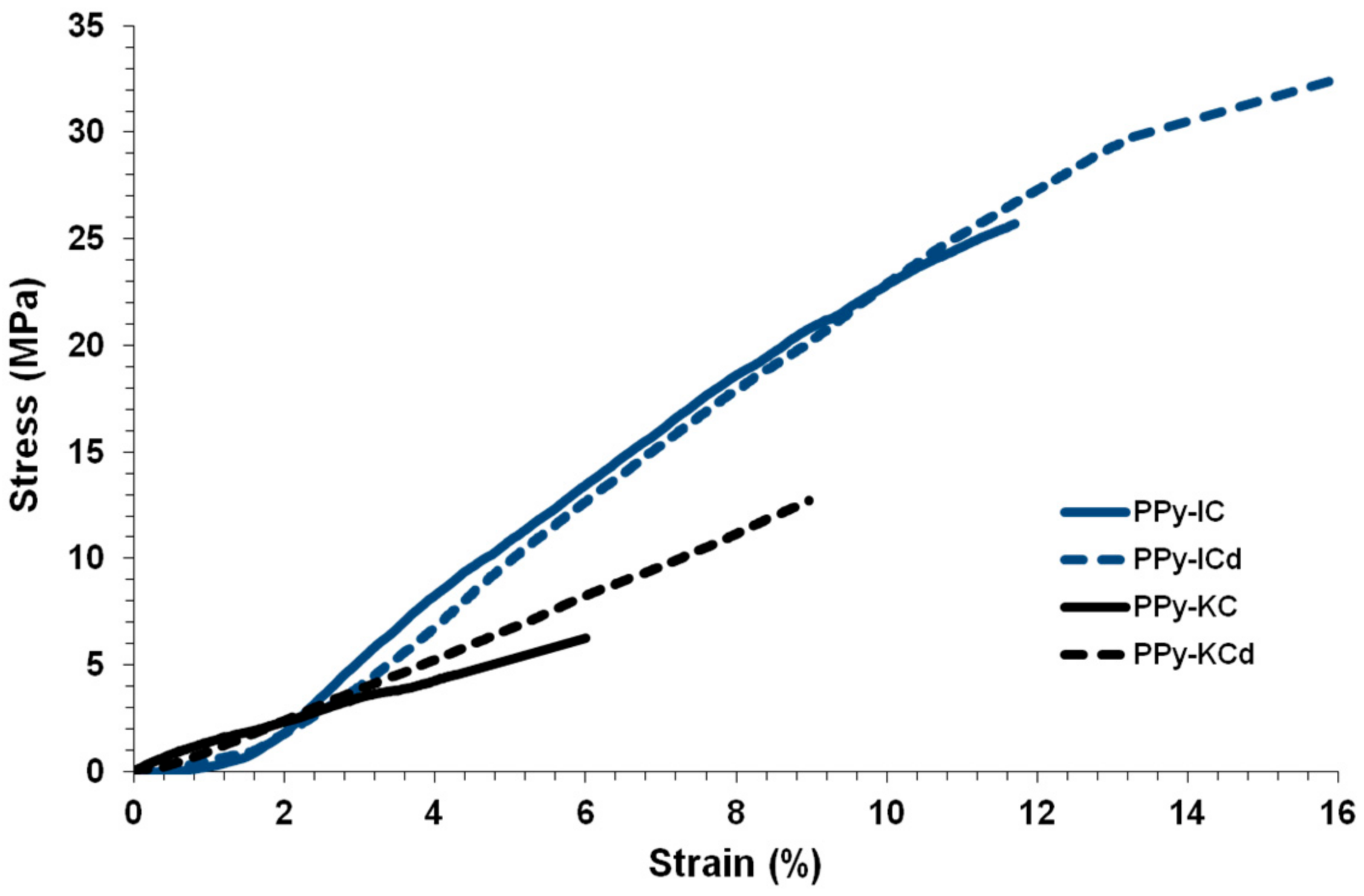
4.5. Mechanical Properties of Composite Films
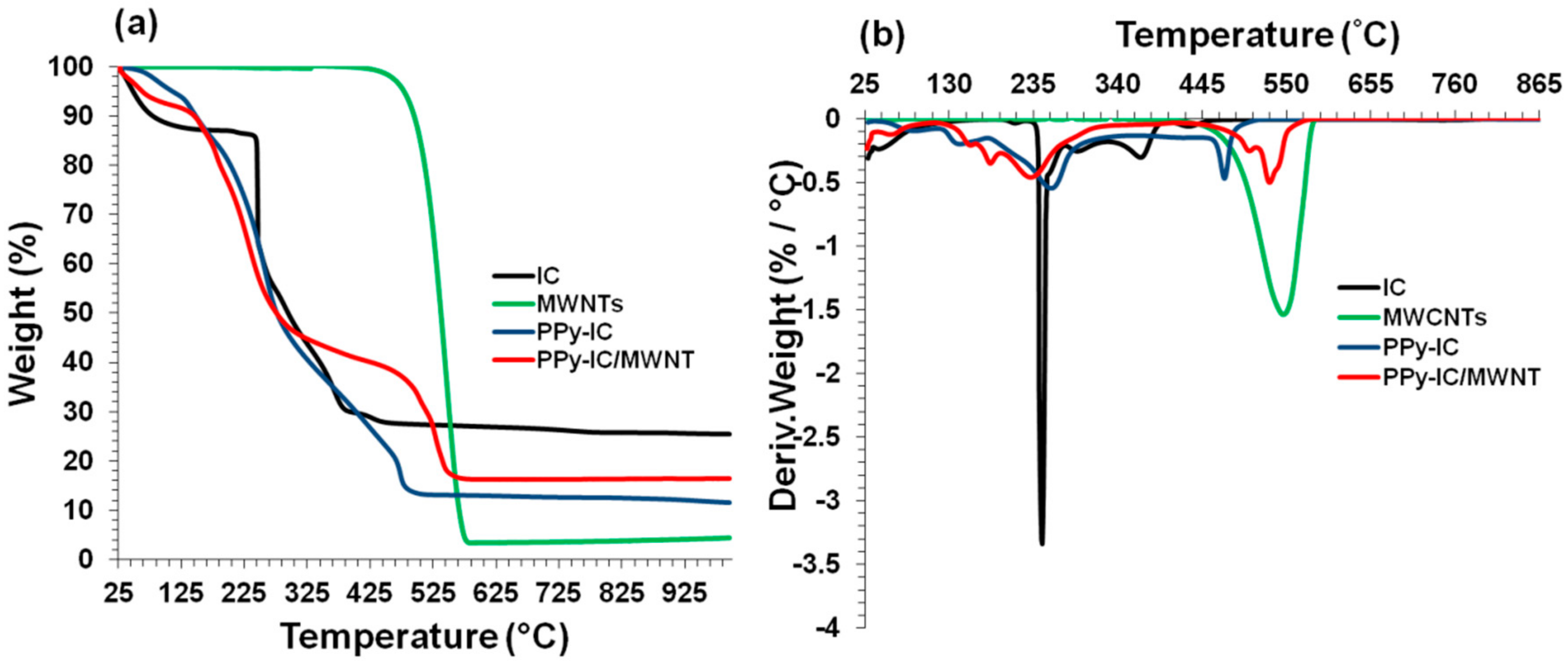
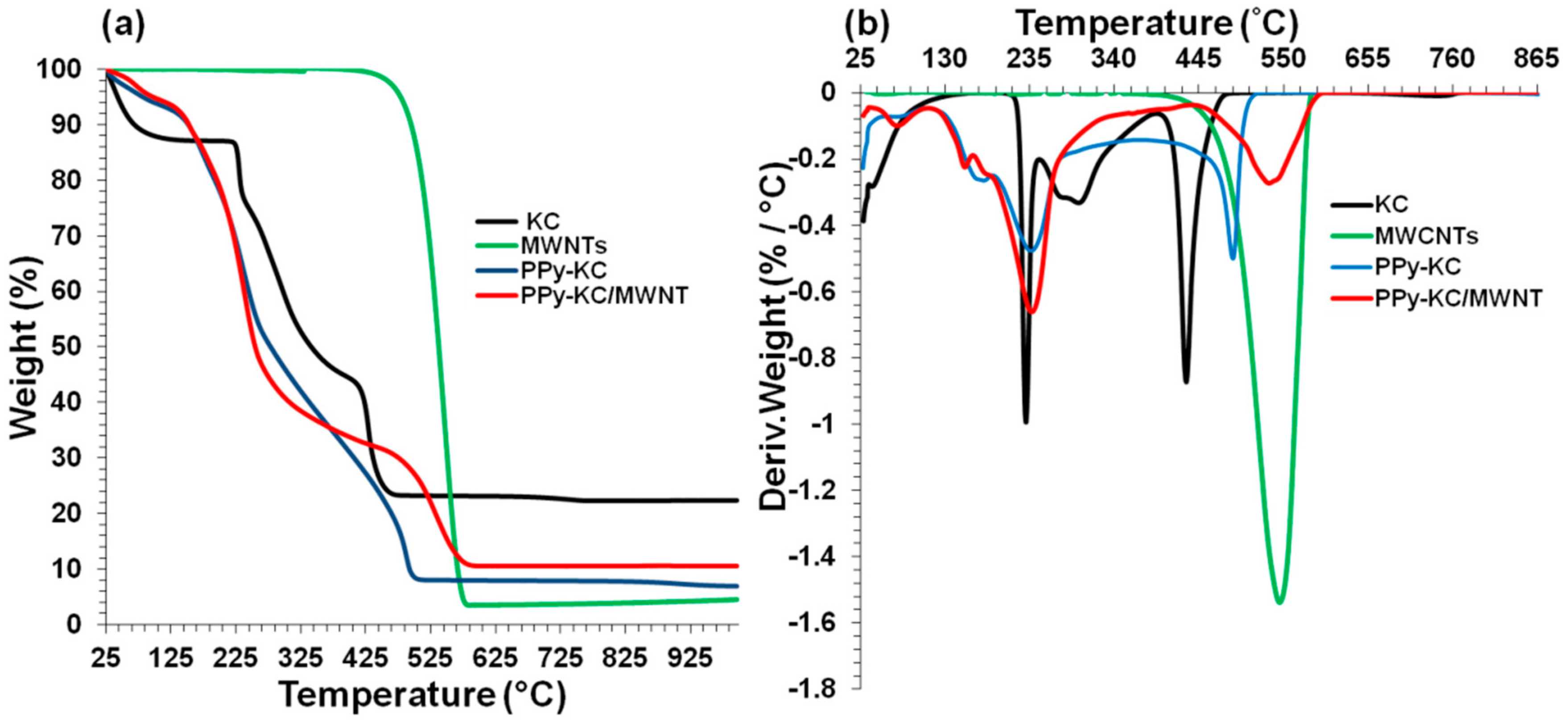
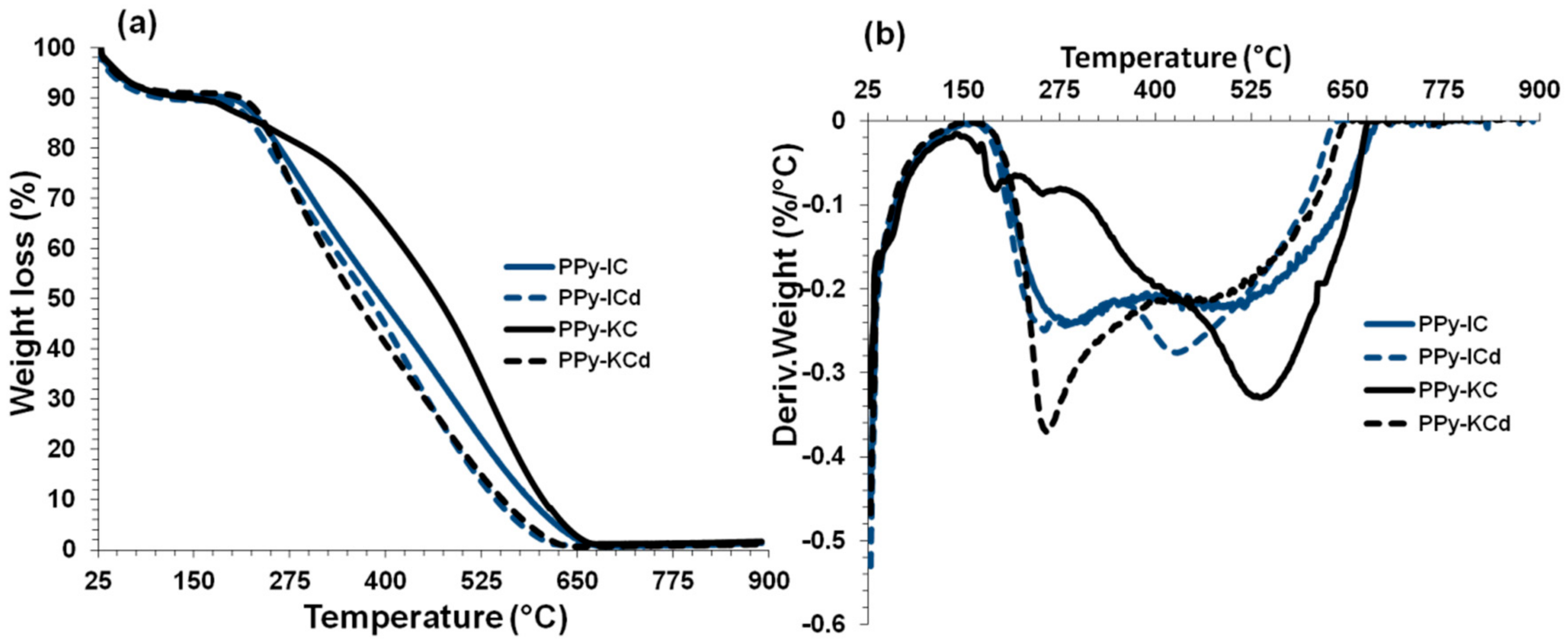
4.6. Thermal Stability of Composite Films
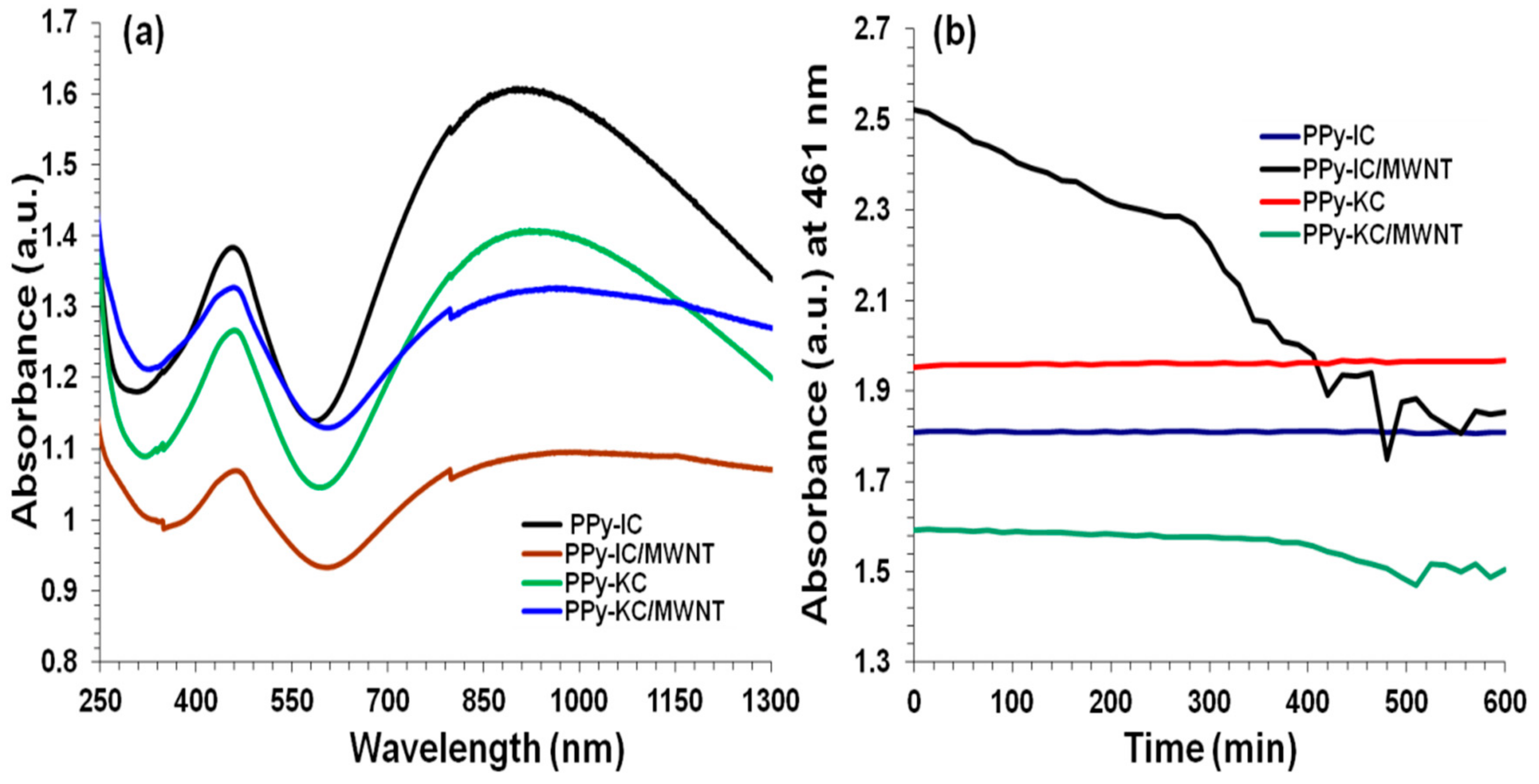
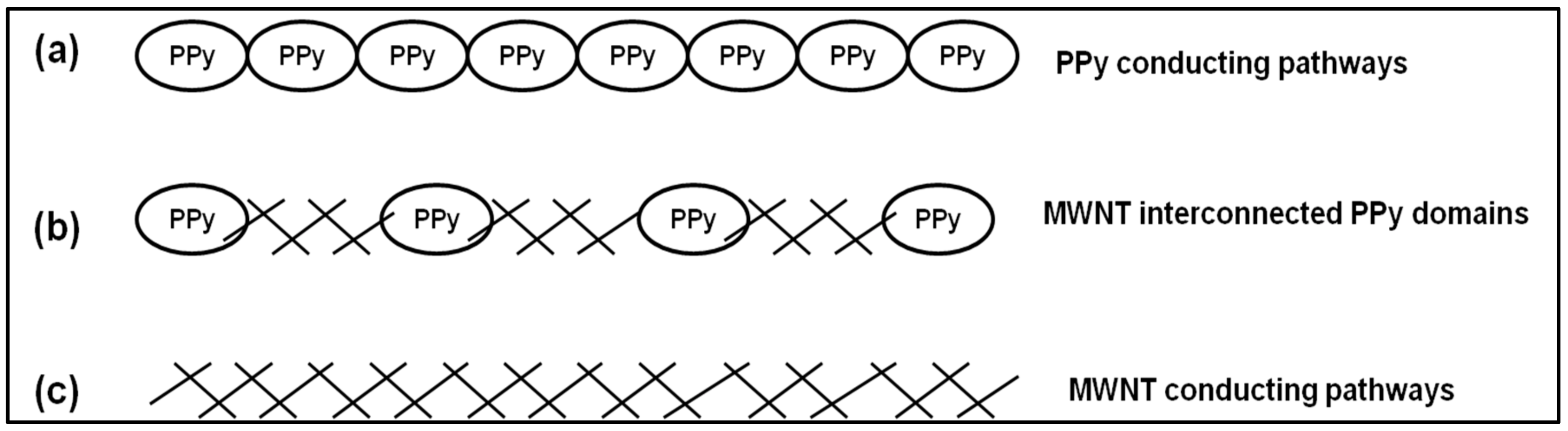
4.7. Incorporation of MWNTs
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Skotheim, T.; Elsenbaumer, R.L.; Reynolds, J.R. Handbook of Conducting Polymers; Marcel Dekker: New York, NY, USA, 1998. [Google Scholar]
- De Barros, R.A.; Martins, C.R.; de Azevedo, W.M. Writing with conducting polymer. Synth. Met. 2005, 155, 35–38. [Google Scholar] [CrossRef]
- Wallace, G.G.; Spinks, G.M.; Teasdale, P.R. Conductive Electroactive Polymers: Intelligent Materials Systems; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Kumar, D.; Sharma, R.C. Advances in conductive polymers. Eur. Polym. J. 1998, 34, 1053–1060. [Google Scholar] [CrossRef]
- Uyar, T.; Toppare, L.; Hacaloglu, J. Characterization of electrochemically synthesized p-toluene sulfonic acid doped polypyrrole by direct insertion probe pyrolysis mass spectrometry. J. Anal. Appl. Pyrolysis 2002, 64, 1–13. [Google Scholar] [CrossRef]
- Campomanes, R.; Bittencourt, S.E.; Campos, J.S.C. Study of conductivity of polypyrrol-poly(vinyl alcohol) composites obtained photochemically. Synth. Met. 1999, 102, 1230–1231. [Google Scholar] [CrossRef]
- Mathai, C.J.; Saravanan, S.; Jayalekshmi, S.; Venkitachalam, S.; Anantharaman, M.R. Conduction mechanism in plasma polymerized aniline thin films. Mater. Lett. 2003, 57, 2253–2257. [Google Scholar] [CrossRef]
- Molina, J.; Esteves, M.F.; Fernndez, J.; Bonastre, J.; Cases, F. Polyaniline coated conducting fabrics. Chemical and electrochemical characterization. Eur. Polym. J. 2011, 47, 2003–2015. [Google Scholar] [CrossRef]
- Adhikari, B.; Majumdar, S. Polymers in sensor applications. Prog. Polym. Sci. 2004, 29, 699–766. [Google Scholar] [CrossRef]
- Harun, M.; Saion, E.; Kassim, A.; Yahya, N.; Mahmud, E. Conjugated conducting polymers: A brief overview. UCSI J. Adv. Sci. Arts 2007, 2, 63–68. [Google Scholar]
- Beadle, P.M.; Armes, S.P. Synthesis and characterization of novel polypyrrole colloids. Synth. Met. 1993, 55, 1114–1118. [Google Scholar] [CrossRef]
- Omastova, M.r.; Trchova, M.; Kovarova, J.; Stejskal, J. Synthesis and structural study of polypyrroles prepared in the presence of surfactants. Synth. Met. 2003, 138, 447–455. [Google Scholar] [CrossRef]
- Jang, K.S.; Han, S.S.; Suh, J.S.; Oh, E.J. Synthesis and characterization of alcohol soluble polypyrrole. Synth. Met. 2001, 119, 107–108. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, D.Y.; Kim, C.Y. Synthesis of soluble polypyrrole of the doped state in organic solvents. Synth. Met. 1995, 74, 103–106. [Google Scholar] [CrossRef]
- Shen, Y.; Wan, M. In situ doping polymerization of pyrrole with sulfonic acid as a dopant. Synth. Met. 1998, 96, 127–132. [Google Scholar] [CrossRef]
- Stenger, J.D. Intrinsically electrically conducting polymers. Synthesis, characterization, and their applications. Prog. Polym. Sci. 1998, 23, 57–79. [Google Scholar] [CrossRef]
- Otero, T.F.; Sansinena, J.M. Bilayer dimensions and movement in artificial muscles. Bioelectrochem. Bioenerg. 1997, 42, 117–122. [Google Scholar] [CrossRef]
- Kong, Y.; Wang, C.; Yang, Y.; Too, C.O.; Wallace, G.G. A battery composed of a polypyrrole cathode and a magnesium alloy anode—toward a bioelectric battery. Synth. Met. 2012, 162, 584–589. [Google Scholar] [CrossRef]
- Geetha, S.; Rao, C.R.K.; Vijayan, M.; Trivedi, D.C. Biosensing and drug delivery by polypyrrole. Anal. Chim. Acta 2006, 568, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Kim, H.K.; Byun, S.W.; Jeong, S.H.; Hong, Y.K.; Joo, J.S.; Song, K.T.; Kim, J.K.; Lee, C.J.; Lee, J.Y. Pet fabric/polypyrrole composite with high electrical conductivity for emi shielding. Synth. Met. 2002, 126, 233–239. [Google Scholar] [CrossRef]
- Campo, V.L.; Kawano, D.F.; Silva, D.B.; Carvalho, I. Carrageenans: Biological properties, chemical modifications and structural analysis—a review. Carbohydr. Polym. 2009, 77, 167–180. [Google Scholar] [CrossRef]
- Ermak, I.M.; Barabanova, A.O.; Kukarskikh, T.A.; Solovyova, T.F.; Bogdanovich, R.N.; Polyakova, A.M.; Astrina, O.P.; Maleyev, V.V. Natural polysaccharide carrageenan inhibits toxic effect of gram-negative bacterial endotoxins. Bull. Exp. Bio. Med. 2006, 141, 230–232. [Google Scholar] [CrossRef]
- Rokka, S.; Rantamäki, P. Protecting probiotic bacteria by microencapsulation: Challenges for industrial applications. Eur. Food Res. Technol. 2010, 231, 1–12. [Google Scholar] [CrossRef]
- Grenha, A.; Gomes, M.E.; Rodrigues, M.; Santo, V.E.; Mano, J.F.; Neves, N.M.; Reis, R.L. Development of new chitosan/carrageenan nanoparticles for drug delivery applications. J. Biomed. Mater. Res. Part A 2010, 92, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Lundin, V.; Herland, A.; Berggren, M.; Jager, E.W.H.; Teixeira, A.I. Control of neural stem cell survival by electroactive polymer substrates. PLoS ONE 2011, 6, e18624. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jiang, M. Toward genolelectronics: Nucleic acid doped conducting polymers. Langmuir 2000, 16, 2269–2274. [Google Scholar] [CrossRef]
- Zhang, L.; Stauffer, W.R.; Jane, E.P.; Sammak, P.J.; Cui, X.T. Enhanced differentiation of embryonic and neural stem cells to neuronal fates on laminin peptides doped polypyrrole. Macromol. Biosci. 2010, 10, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Wang, Q.; Haque, M.A. Doping dependence of electrical and thermal conductivity of nanoscale polyaniline thin films. J. Phys. D Appl. Phys. 2010, 43, 205302. [Google Scholar] [CrossRef]
- Cardoso, M.R.; Lima, M.S.; Lenz, D.M. Polyaniline synthesized with functionalized sulfonic acids for blends manufacture. Mater. Res. 2007, 10, 425–429. [Google Scholar] [CrossRef]
- Zhang, L.; Peng, H.; Kilmartin, P.A.; Soeller, C.; Travas-Sejdic, J. Polymeric acid doped polyaniline nanotubes for oligonucleotide sensors. Electroanalysis 2007, 19, 870–875. [Google Scholar] [CrossRef]
- Cui, X.; Hetke, J.F.; Wiler, J.A.; Anderson, D.J.; Martin, D.C. Electrochemical deposition and characterization of conducting polymer polypyrrole/pss on multichannel neural probes. Sens. Actuators A: Phys. 2001, 93, 8–18. [Google Scholar] [CrossRef]
- Hsu, F.-H.; Wu, T.-M. In situ synthesis and characterization of conductive polypyrrole/graphene composites with improved solubility and conductivity. Synth. Met. 2011, 162, 682–687. [Google Scholar] [CrossRef]
- Collier, J.H.; Camp, J.P.; Hudson, T.W.; Schmidt, C.E. Synthesis and characterization of polypyrrole-hyaluronic acid composite biomaterials for tissue engineering applications. J. Biomed. Mater. Res. 2000, 50, 574–584. [Google Scholar] [CrossRef]
- Higgins, T.M.; Moulton, S.E.; Gilmore, K.J.; Wallace, G.G.; in het Panhuis, M. Gellan gum doped polypyrrole neural prosthetic electrode coatings. Soft Matter 2011, 7, 4690–4695. [Google Scholar] [CrossRef]
- Moreno, J.; Panero, S.; Materazzi, S.; Martinelli, A.; Sabbieti, M.G.; Agas, D.; Materazzi, G. Polypyrrole-polysaccharide thin films characteristics: Electrosynthesis and biological properties. J. Biomed. Mater. Res. Part A 2009, 88A, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shi, W.; Jiang, H.; Wu, G.; Ruan, C.; Ge, D. Heparin-doped affinity electromembranes for thrombin purification. J. Membr. Sci. 2011, 373, 89–97. [Google Scholar] [CrossRef]
- Yang, X.; Too, C.O.; Sparrow, L.; Ramshaw, J.; Wallace, G.G. Polypyrrole-heparin system for the separation of thrombin. React. Funct. Polym. 2002, 53, 53–62. [Google Scholar] [CrossRef]
- Zhou, D.; Too, C.O.; Wallace, G.G. Synthesis and characterisation of polypyrrole/heparin composites. React. Funct. Polym. 1999, 39, 19–26. [Google Scholar] [CrossRef]
- Hughes, M.; Chen, G.Z.; Shaffer, M.S.P.; Fray, D.J.; Windle, A.H. Electrochemical capacitance of a nanoporous composite of carbon nanotubes and polypyrrole. Chem. Mater. 2002, 14, 1610–1613. [Google Scholar] [CrossRef]
- Coleman, J.N.; Khan, U.; Gun'ko, Y.K. Mechanical reinforcement of polymers using carbon nanotubes. Adv. Mater. 2006, 18, 689–706. [Google Scholar] [CrossRef]
- Woo, H.S.; Czerw, R.; Webster, S.; Carroll, D.L.; Park, J.W.; Lee, J.H. Organic light emitting diodes fabricated with single wall carbon nanotubes dispersed in a hole conducting buffer: The role of carbon nanotubes in a hole conducting polymer. Synth. Met. 2001, 116, 369–372. [Google Scholar] [CrossRef]
- Frackowiak, E.; Jurewicz, K.; Delpeux, S.; Beguin, F. Nanotubular materials for supercapacitors. J. Power Sources 2001, 97–98, 822–825. [Google Scholar] [CrossRef]
- Kong, J.; Franklin, N.R.; Zhou, C.; Chapline, M.G.; Peng, S.; Cho, K.; Dai, H. Nanotube molecular wires as chemical sensors. Science 2000, 287, 622–625. [Google Scholar] [CrossRef] [PubMed]
- Michio, M.; Atsushi, I.; Yuichiro, M. Accumulation of positive charges in organic light-emitting diodes with a double-layer structure. Appl. Phys. Lett. 1999, 75, 1042–1044. [Google Scholar]
- Foroughi, J.; Spinks, G.M.; Wallace, G.G. Effect of synthesis conditions on the properties of wet spun polypyrrole fibres. Synth. Met. 2009, 159, 1837–1843. [Google Scholar] [CrossRef]
- In het Panhuis, M.; Maiti, A.; Dalton, A.B.; van den Noort, A.; Coleman, J.N.; McCarthy, B.; Blau, W.J. Selective interaction in a polymer -single-wall carbon nanotube composite. J. Phys. Chem. B 2002, 107, 478–482. [Google Scholar] [CrossRef]
- In het Panhuis, M.; Sainz, R.; Innis, P.C.; Kane-Maguire, L.A.P.; Benito, A.M.; Martinez, M.T.; Moulton, S.E.; Wallace, G.G.; Maser, W.K. Optically active polymer carbon nanotube composite. J. Phys. Chem. B 2005, 109, 22725–22729. [Google Scholar] [CrossRef] [PubMed]
- Song, K.T.; Lee, J.Y.; Kim, H.D.; Kim, D.Y.; Kim, S.Y.; Kim, C.Y. Solvent effects on the characteristics of soluble polypyrrole. Synth. Met. 2000, 110, 57–63. [Google Scholar] [CrossRef]
- Omastova, M.; Trchova, M.; Pionteck, J.r.; Prokes, J.; Stejskal, J. Effect of polymerization conditions on the properties of polypyrrole prepared in the presence of sodium bis(2-ethylhexyl) sulfosuccinate. Synth. Met. 2004, 143, 153–161. [Google Scholar] [CrossRef]
- Stejskal, J. Colloidal dispersions of conducting polymers. J. Polym. Mater. 2001, 18, 225–258. [Google Scholar]
- Brédas, J.; Scott, J.; Yakushi, K.; Street, G. Polarons and bipolarons in polypyrrole: Evolution of the band structure and optical spectrum upon doing. Phys. Rev. B 1984, 30, 1023–1025. [Google Scholar] [CrossRef]
- Huang, J.; Li, X.-H.; Xu, J.-C.; Li, H.-L. Well-dispersed single-walled carbon nanotube/polyaniline composite films. Carbon 2003, 41, 2731–2736. [Google Scholar] [CrossRef]
- Rezaul Karim, M.; Lee, C.J.; Sarwaruddin Chowdhury, A.M.; Nahar, N.; Lee, M.S. Radiolytic synthesis of conducting polypyrrole/carbon nanotube composites. Mater. Lett. 2007, 61, 1688–1692. [Google Scholar]
- Sahoo, N.G.; Jung, Y.C.; So, H.H.; Cho, J.W. Polypyrrole coated carbon nanotubes: Synthesis, characterization, and enhanced electrical properties. Synth. Met. 2007, 157, 374–379. [Google Scholar] [CrossRef]
- Wu, T.-M.; Lin, S.-H. Characterization and electrical properties of polypyrrole/multiwalled carbon nanotube composites synthesized by in situ chemical oxidative polymerization. J. Polym. Sci. Part B Polym. Phys. 2006, 44, 1413–1418. [Google Scholar] [CrossRef]
- Long, Y.; Chen, Z.; Zhang, X.; Zhang, J.; Liu, Z. Electrical properties of multi-walled carbon nanotube/polypyrrole nanocables: Percolation-dominated conductivity. J. Phys. D Appl. Phys. 2004, 37, 1965. [Google Scholar] [CrossRef]
- Li, L.; Yan, F.; Xue, G. Preparation of a porous conducting polymer film by electrochemical synthesis-solvent extraction method. J. Appl. Polym. Sci. 2004, 91, 303–307. [Google Scholar] [CrossRef]
- Potschke, P.; Brünig, H.; Janke, A.; Fischer, D.; Jehnichen, D. Orientation of multiwalled carbon nanotubes in composites with polycarbonate by melt spinning. Polymer 2005, 46, 10355–10363. [Google Scholar] [CrossRef]
- Prasad, K.; Meena, R.; Siddhanta, A.K. Microwave-induced rapid one-pot synthesis of κ-carrageenan-g-pmma copolymer by potassium persulphate initiating system. J. Appl. Polym. Sci. 2006, 101, 161–166. [Google Scholar] [CrossRef]
- Wu, T.-M.; Chang, H.-L.; Lin, Y.-W. Synthesis and characterization of conductive polypyrrole/multi-walled carbon nanotubes composites with improved solubility and conductivity. Compos. Sci. Technol. 2009, 69, 639–644. [Google Scholar] [CrossRef]
- Vincekovic, M.; Pustak, A.; Tusek-Bozic, L.; Liu, F.; Ungar, G.; Bujan, M.; Smit, I.; Filipovic-Vincekovic, N. Structural and thermal study of mesomorphic dodecylammonium carrageenates. J. Colloid Interface Sci. 2010, 341, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Snook, G.A.; Gupta, V.; Shaffer, M.; Fray, D.J.; Chen, G.Z. Electrochemical fabrication and capacitance of composite films of carbon nanotubes and polyaniline. J. Mater. Chem. 2005, 15, 2297–2303. [Google Scholar] [CrossRef]



| Sample | σ (S/m) |
|---|---|
| PPy-IC | 0.022 ± 0.004 |
| PPy-IC/MWNT | 21 ± 3 |
| PPy-KC | 0.067 ± 0.012 |
| PPy-KC/MWNT | 2.2 ± 0.4 |
| Film | Thickness (cm) | Rs (Ω·m) | σ (S/m) |
|---|---|---|---|
| PPy-IC | 0.0045 ± 0.002 | 35.2 ± 3.0 | 631 ± 66 |
| PPy-ICd | 0.0011 ± 0.001 | 183 ± 3.2 | 463 ± 50 |
| PPy-KC | 0.0034 ± 0.002 | 43.8 ± 2.7 | 674 ± 49 |
| PPy-KCd | 0.0021 ± 0.001 | 93.2 ± 2.9 | 501 ± 31 |
| Film | TS (MPa) | γ (%) | T (J/m3) | E (MPa) |
|---|---|---|---|---|
| PPy-IC | 4.18 ± 0.97 | 5.39 ± 0.43 | 0.17 ± 0.02 | 232 ± 6 |
| PPy-IC/MWCNT | 7.95 ± 1.16 | 2.93 ± 0.23 | 0.13 ± 0.01 | 361 ± 9 |
| PPy-KC | 7.83 ± 1.12 | 3.58 ± 0.35 | 0.16 ± 0.02 | 257 ± 7 |
| PPy-KC/MWCNT | 9.54 ± 1.31 | 2.51 ± 0.21 | 0.14 ± 0.01 | 453 ± 12 |
| Film | TS (MPa) | γ (%) | T (J/m3) | E (MPa) |
|---|---|---|---|---|
| PPy-IC | 25.2 ± 3.6 | 11.6 ± 2.3 | 1.3 ± 0.1 | 256 ± 13 |
| PPy-ICd | 34.6 ± 6.8 | 18.9 ± 6.1 | 1.8 ± 0.3 | 282 ± 16 |
| PPy-KC | 6.5 ± 3.1 | 6.7 ± 1.1 | 0.2 ± 0.1 | 186 ± 14 |
| PPy-KCd | 12.6 ± 5.6 | 8.9 ± 3.7 | 0.5 ± 0.2 | 199 ± 13 |
| Films | Temperature at 5% weight loss (°C) | Temperature at 20% weight loss (°C) | Temperature at 50% weight loss (°C) |
|---|---|---|---|
| MWNTs | 479.0 | 512.0 | 573.0 |
| IC | 048.2 | 246.0 | 295.0 |
| * PPy-IC | 062.9 | 186.0 | 376.0 |
| # PPy-IC | 047.6 | 262.0 | 395.0 |
| # PPy-ICd | 040.8 | 248.0 | 378.0 |
| * PPy-IC/MWNT | 111.2 | 203.0 | 277.0 |
| KC | 043.5 | 232.0 | 341.0 |
| * PPy-KC | 098.0 | 196.0 | 255.0 |
| # PPy-KC | 051.7 | 295.0 | 469.0 |
| # PPy-KCd | 046.7 | 256.0 | 360.0 |
| * PPy-KC/MWNT | 081.7 | 192.0 | 278.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahaman, M.; Aldalbahi, A.; Almoiqli, M.; Alzahly, S. Chemical and Electrochemical Synthesis of Polypyrrole Using Carrageenan as a Dopant: Polypyrrole/Multi-Walled Carbon Nanotube Nanocomposites. Polymers 2018, 10, 632. https://doi.org/10.3390/polym10060632
Rahaman M, Aldalbahi A, Almoiqli M, Alzahly S. Chemical and Electrochemical Synthesis of Polypyrrole Using Carrageenan as a Dopant: Polypyrrole/Multi-Walled Carbon Nanotube Nanocomposites. Polymers. 2018; 10(6):632. https://doi.org/10.3390/polym10060632
Chicago/Turabian StyleRahaman, Mostafizur, Ali Aldalbahi, Mohammed Almoiqli, and Shaykha Alzahly. 2018. "Chemical and Electrochemical Synthesis of Polypyrrole Using Carrageenan as a Dopant: Polypyrrole/Multi-Walled Carbon Nanotube Nanocomposites" Polymers 10, no. 6: 632. https://doi.org/10.3390/polym10060632
APA StyleRahaman, M., Aldalbahi, A., Almoiqli, M., & Alzahly, S. (2018). Chemical and Electrochemical Synthesis of Polypyrrole Using Carrageenan as a Dopant: Polypyrrole/Multi-Walled Carbon Nanotube Nanocomposites. Polymers, 10(6), 632. https://doi.org/10.3390/polym10060632





