Study on the Use of Microbial Cellulose as a Biocarrier for 1,3-Dihydroxy-2-Propanone and Its Potential Application in Industry
Abstract
:1. Introduction
2. Materials and Methods
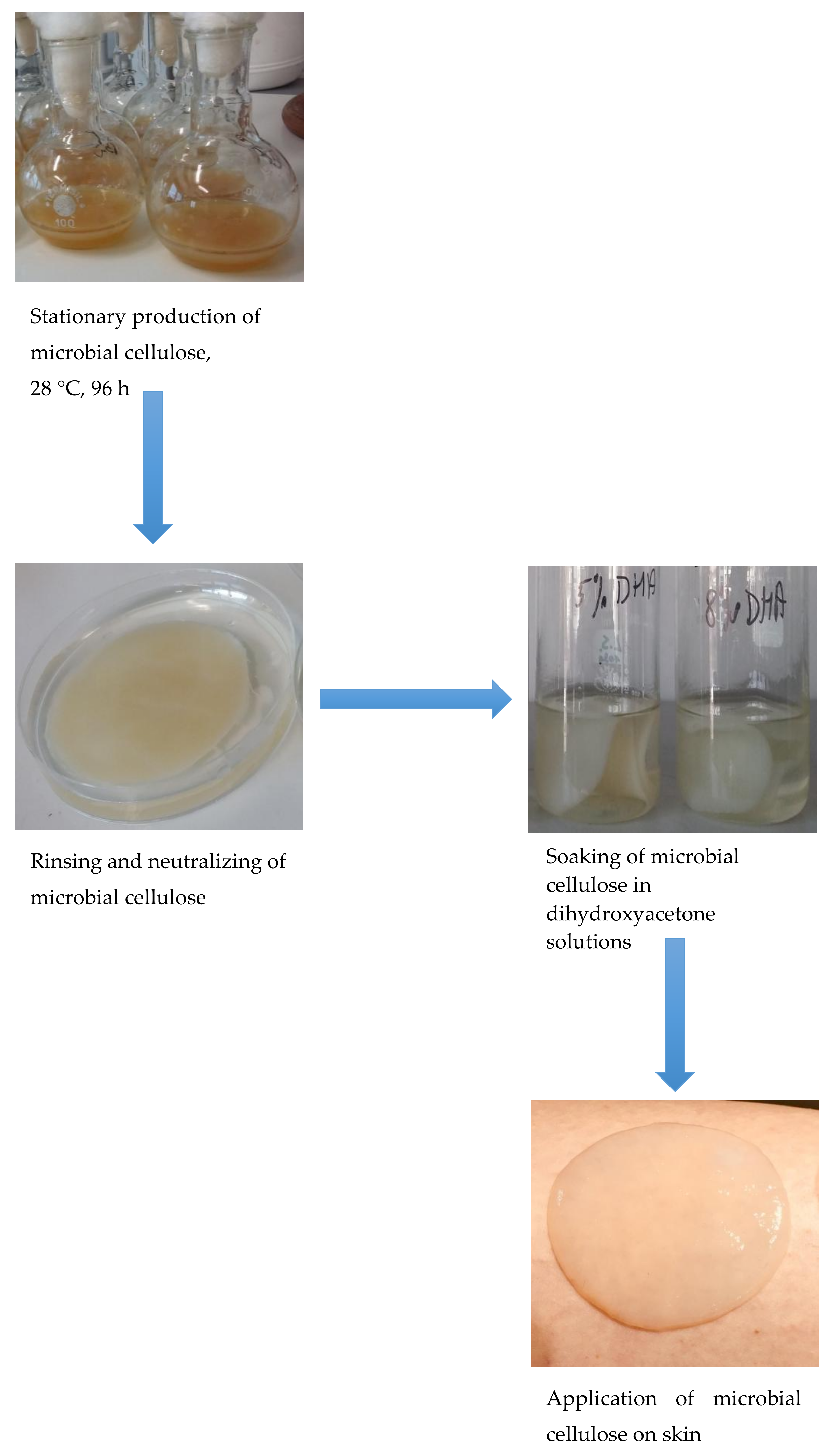
2.1. Growing culture of G. xylinus
2.2. Production of MC by G. xylinus Strain
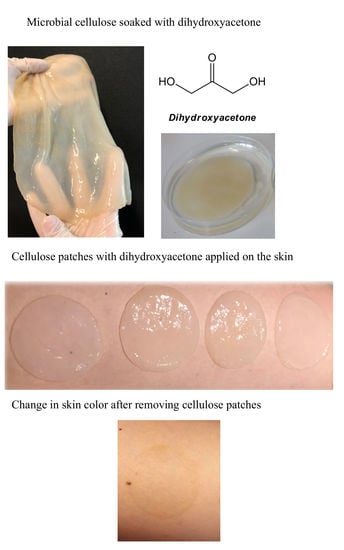
2.3. Soaking the MC with DHA
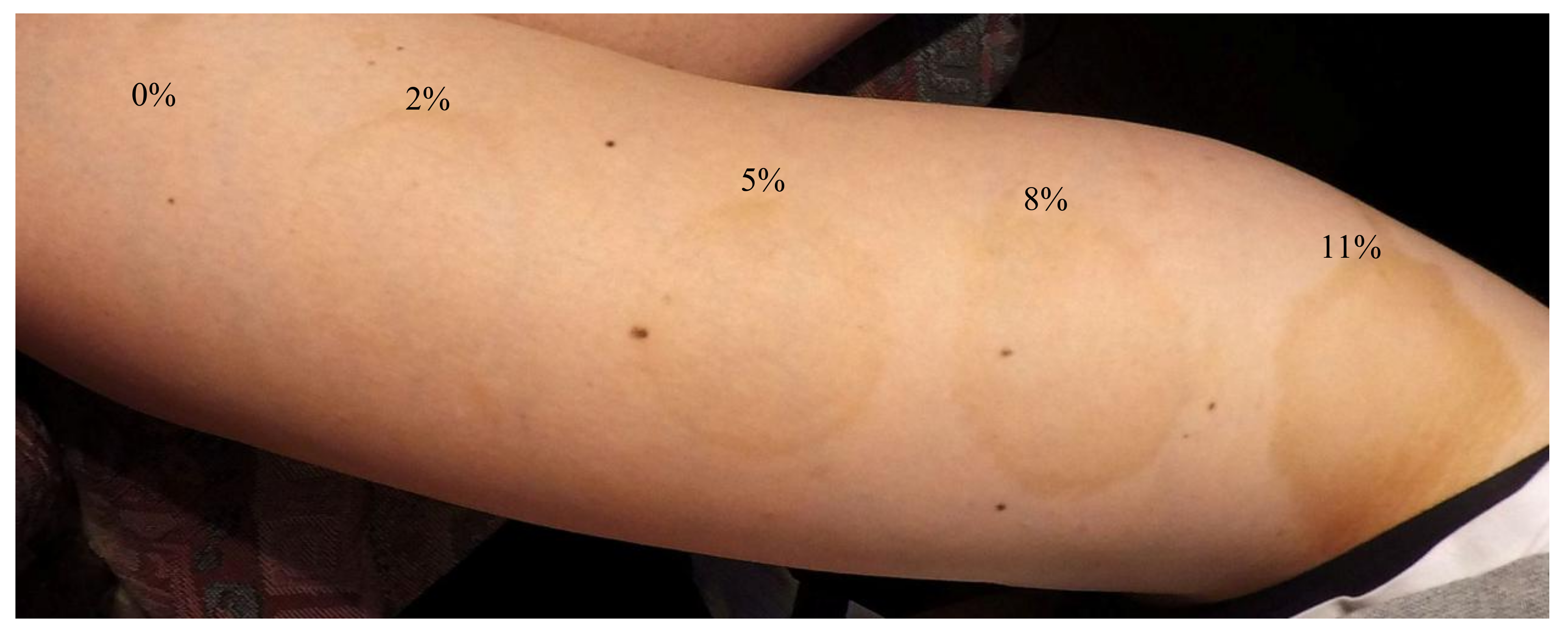
2.4. Application MC with DHA on Skin
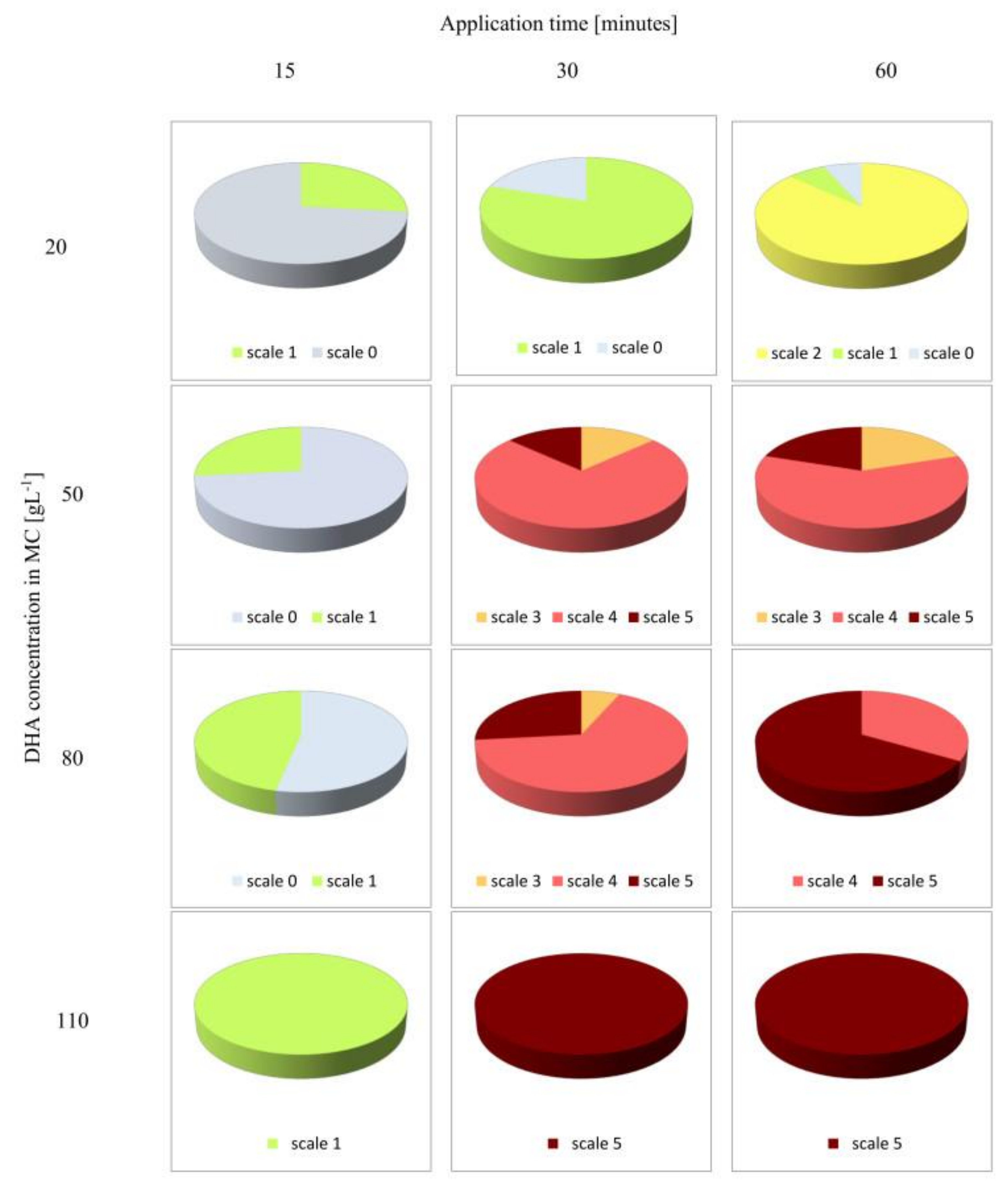
2.5. Visual Assessment of Skin Browning Effects
3. Results and Discussion
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Moosavi-Nasab, M.; Yousefi, M. Biotechnological production of cellulose by Gluconacetobacter xylinus from agricultural waste. Iran J. Biotechnol. 2011, 9, 94–101. [Google Scholar]
- Ramachandran, S.; Fontanille, P.; Pandey, A.; Larroche, C. Gluconic Acid: Properties, Applications and Microbial Production. Food Technol. Biotechnol. 2006, 44, 185–195. Available online: http://www.ftb.com.hr/index.php/archives/80-volume-44-issue-no-2/447 (accessed on 22 March 2018).
- Gullo, M.; Giudici, P. Acetic acid bacteria in traditional balsamic vinegar: Phenotypic traits relevant for starter cultures selection. Int. J. Food Microbiol. 2008, 125, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Franken, J.; Brandt, B.A.; Tai, S.L.; Bauer, F.F. Biosynthesis of levan, a bacterial extracellular polysaccharide, in the yeast Saccharomyces cerevisiae. PLoS ONE 2013, 8, e77499. [Google Scholar] [CrossRef] [PubMed]
- Dikshit, P.K.; Padhi, S.K.; Moholkar, V.S. Process optimization and analysis of product inhibition kinetics of crude glycerol fermentation for 1,3-Dihydroxyacetone production. Bioresour. Technol. 2017, 244, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Jahan, F.; Kumar, V.; Rawat, G.; Saxena, R.K. Production of microbial cellulose by a bacterium isolated from fruit. Appl. Biochem. Biotechnol. 2012, 167, 1157–1171. [Google Scholar] [CrossRef] [PubMed]
- Chawla, P.R.; Bajaj, I.B.; Survase, S.A.; Singhal, R.S. Microbial Cellulose: Fermentative Production and Applications. Food Technol. Biotechnol. 2009, 47, 107–124. Available online: http://www.ftb.com.hr/images/pdfarticles/2009/April-June/47-107.pdf (accessed on 22 March 2018).
- An, S.J.; Lee, S.H.; Huh, J.B.; Jeong, S.I.; Park, J.S.; Gwon, H.J.; Kang, E.S.; Jeong, C.M.; Lim, Y.M. Preparation and characterization of resorbable bacterial cellulose membranes treated by electron beam irradiation for guided bone regeneration. Int. J. Mol. Sci. 2017, 18, 2236. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Ullah, N.; Wang, X.; Sun, X.; Li, C.; Bai, Y.; Chen, L.; Li, Z. Characterization of bacterial cellulose by Gluconacetobacter hansenii CGMCC 3917. J. Food Sci. 2015, 80, 2217–2227. [Google Scholar] [CrossRef] [PubMed]
- Halib, N.; Amin, M.C.I.M.; Ahmad, I.; Hashim, Z.M.; Jamal, N. Swelling of Bacterial Cellulose-Acrylic Acid Hydrogels: Sensitivity towards External Stimuli. Sains Malays. 2009, 38, 785–791. Available online: https://ukm.pure.elsevier.com/en/publications/swelling-of-bacterial-cellulose-acrylic-acid-hydrogels-sensitivit (accessed on 5 April 2018).
- Stanisławska, A. Bacterial nanocellulose as a microbiological derived nanomaterial. Adv. Mater. 2016, 16, 45–57. [Google Scholar] [CrossRef]
- Czaja, W.; Krystynowicz, A.; Bielecki, S.; Brown, M., Jr. Microbial cellulose—The natural power to heal wounds. Biomaterials 2006, 27, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Abeer, M.M.; Mohd Admin, M.C.; Martin, C. A review of bacterial cellulose-based drug delivery systems: Their biochemistry, current approaches and future prospects. J. Pharm. Pharmacol. 2014, 66, 1047–1061. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z. Treatment of tympanic membrane perforation using bacterial cellulose: A randomized controlled trial. Braz. J. Otorhinolaryngol. 2016, 82, 618–619. [Google Scholar] [CrossRef] [PubMed]
- Loloei, M.; Rezaee, A.; Roohaghdam, A.S.; Aliofkhazraei, M. Conductive microbial cellulose as a novel biocathode for Cr (VI) bioreduction. Carbohydr. Polym. 2017, 162, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Jang, W.D.; Hwang, J.H.; Kim, H.U.; Ryu, J.Y.; Lee, S.Y. Bacterial cellulose as an example product for sustainable production and consumption. Microb. Biotechnol. 2017, 10, 1181–1185. [Google Scholar] [CrossRef] [PubMed]
- Draelos, M.D.; Zoe, D. Self-Tanning Lotions: Are they a healthy way to achieve a tan? Am. J. Clin. Dermatol. 2002, 3, 317–318. Available online: https://www.ncbi.nlm.nih.gov/pubmed/12069637 (accessed on 22 March 2018). [CrossRef] [PubMed]
- Erni, B.; Siebold, C.; Christen, S.; Srinivas, A.; Oberholzer, A.; Baumann, U. Small substrate, big surprise: Fold, function and phylogeny of dihydroxyacetone kinases. Cell. Mol. Life Sci. 2006, 63, 890–900. [Google Scholar] [CrossRef] [PubMed]
- Gätgens, C.; Degner, U.; Bringer-Meyer, S.; Herrmann, U. Biotransformation of glycerol to dihydroxyacetone by recombinant Gluconobacter oxydans DSM 2343. Appl. Microbiol. Biotechnol. 2007, 76, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Stasiak-Różańska, L.; Błażejak, S. Dihydroksyaceton—Characteristic, application, obtaining. Acta Sci. Pol. Biotechnol. 2012, 11, 17–28. Available online: http://yadda.icm.edu.pl/yadda/element/bwmeta1.element.agro-ae299301-7397-4412-874f-754382ffce9b (accessed on 22 March 2018).
- Schmidt, D.; Belser, E.; Zulli, F. Self-tanning based on stimulation of melanin biosynthesis. Cosmet. Toilet. 2007, 6, 55–60. Available online: http://www.cosmeticsandtoiletries.com/formulating/function/active/8281582.html (accessed on 22 March 2018).
- Chanprateep, S. Current trends in biodegradable polyhydroxyalkanoates. J. Biosci. Bioeng. 2010, 110, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, B.C.; Kochevar, I.E. Factors influencing sunless tanning with dihydroxyacetone. Br. J. Dermatol. 2003, 149, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Fesq, H.; Brockow, K.; Strom, K.; Mempel, M.; Ring, J.; Abeck, D. Dihydroxyacetone in a newformulation—A powerful therapeutic option in vitiligo. Dermatology 2001, 203, 241–243. [Google Scholar] [CrossRef] [PubMed]
- Mohite, B.V.; Patil, S.V. A novel biomaterial: Bacterial cellulose and its new era applications. Biotechnol. Appl. Biochem. 2014, 61, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Okamura, K.; Araki, Y.; Suzuki, M.; Tomaka, T.; Abe, Y.; Nakano, S.; Yoshizawa, J.; Hozumi, Y.; Inoie, M.; et al. A novel three dimensional imaging method for the measurment of area in vitiligo and chemical leukoderma. J. Dermatol. Sci. 2016, 84, 219–221. [Google Scholar] [CrossRef] [PubMed]
- Amer, A.A.; Gao, X.H. Quality of life in patients with vitiligo: An analysis of the dermatology life quality index outcome over the past two decades. Int. J. Dermatol. 2016, 55, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, A.; Kar, S. Smashed skin grafting—A novel method of vitiligo surgery. Int. J. Dermatol. 2012, 51, 1242–1247. [Google Scholar] [CrossRef] [PubMed]
- Hossain, C.; Porto, D.A.; Hamzavi, I.; Lim, H.W. Camouflaging agents for vitiligo patients. J. Drugs Dermatol. 2016, 15, 384–387. Available online: https://www.ncbi.nlm.nih.gov/pubmed/27050692 (accessed on 22 March 2018). [PubMed]
- Iwanowski, T.; Szlązak, P.; Rustowska, A.; Sokołowska-Wojdyło, M. Efficary of suction blister epidermal grafting with concominant phototherapy in vitiligo treatment. Adv. Dermatol. Allergol. 2017. [Google Scholar] [CrossRef]
- Ko, W.C.; Chen, Y.F. Suction blister epidermal grafts combined with CO2 laser superficial ablation as a good method for treating small-sized vitiligo. Dermatol. Surg. 2009, 35, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S. Camouflaging vitiligo with dihydroxyacetone. Dermatol. Online J. 2008, 14, 23. Available online: https://www.ncbi.nlm.nih.gov/pubmed/19061581 (accessed on 22 March 2018). [PubMed]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stasiak-Różańska, L.; Płoska, J. Study on the Use of Microbial Cellulose as a Biocarrier for 1,3-Dihydroxy-2-Propanone and Its Potential Application in Industry. Polymers 2018, 10, 438. https://doi.org/10.3390/polym10040438
Stasiak-Różańska L, Płoska J. Study on the Use of Microbial Cellulose as a Biocarrier for 1,3-Dihydroxy-2-Propanone and Its Potential Application in Industry. Polymers. 2018; 10(4):438. https://doi.org/10.3390/polym10040438
Chicago/Turabian StyleStasiak-Różańska, Lidia, and Justyna Płoska. 2018. "Study on the Use of Microbial Cellulose as a Biocarrier for 1,3-Dihydroxy-2-Propanone and Its Potential Application in Industry" Polymers 10, no. 4: 438. https://doi.org/10.3390/polym10040438
APA StyleStasiak-Różańska, L., & Płoska, J. (2018). Study on the Use of Microbial Cellulose as a Biocarrier for 1,3-Dihydroxy-2-Propanone and Its Potential Application in Industry. Polymers, 10(4), 438. https://doi.org/10.3390/polym10040438