High-Performance Biobased Unsaturated Polyester Nanocomposites with Very Low Loadings of Graphene †
Abstract
1. Introduction
2. Experimentals
2.1. Materials
2.2. Synthesis of Neat UPR
2.3. Synthesis of UPR/TO
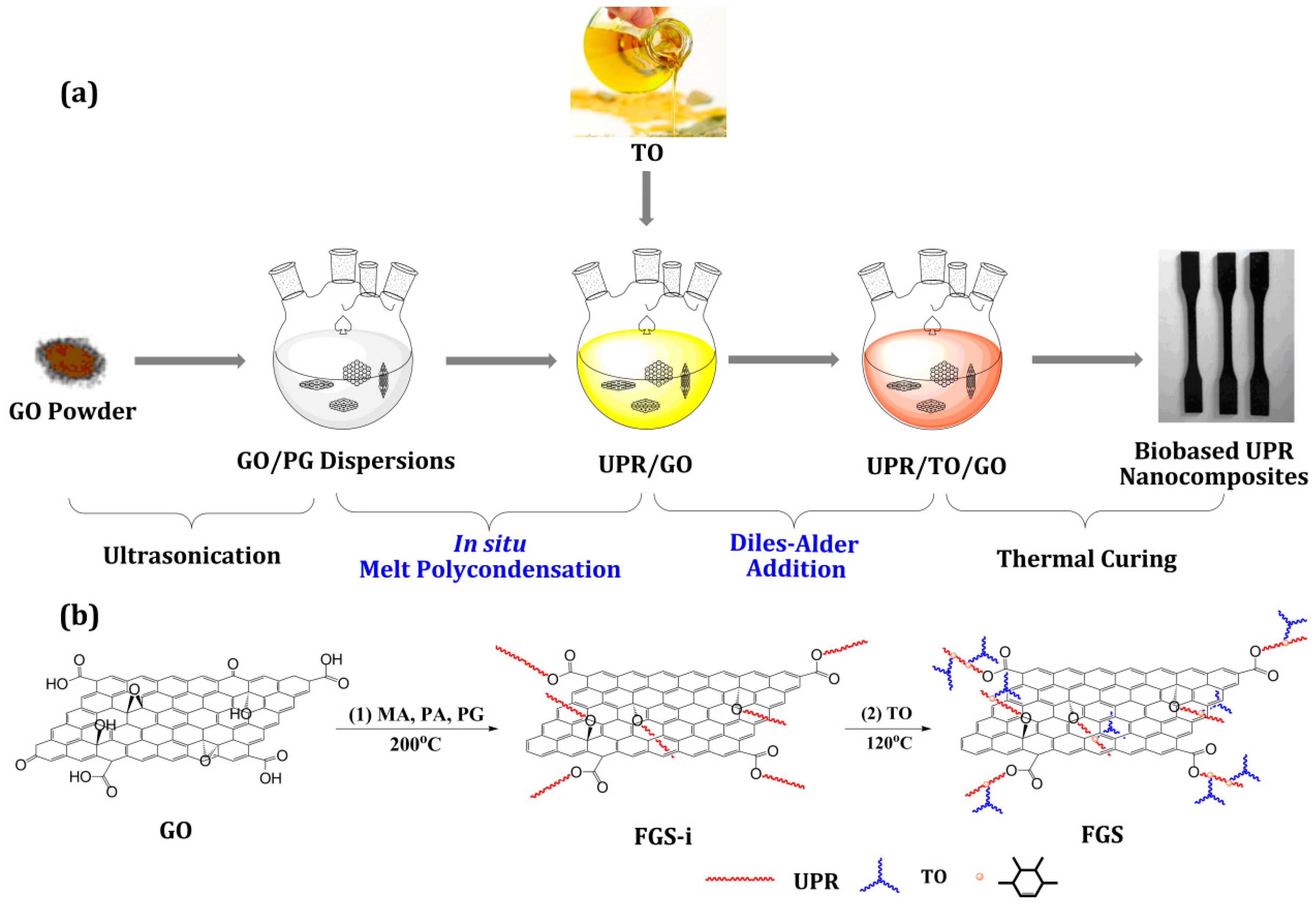
2.4. Preparation of UPR/TO/GO Composites
2.5. Curing of UPR, UPR/TO, and UPR/TO/GO Composites
2.6. Characterization
3. Results and Discussion
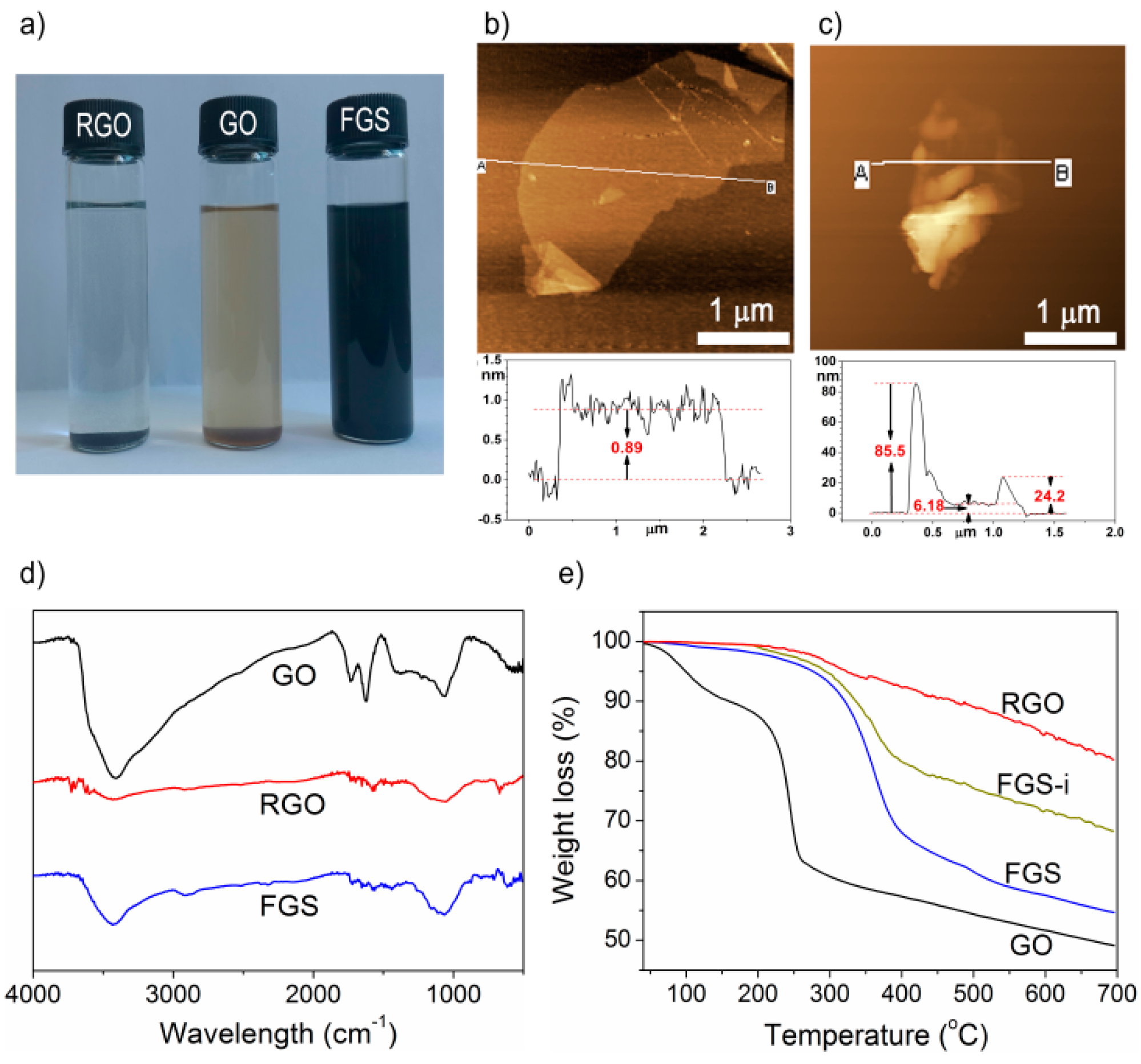
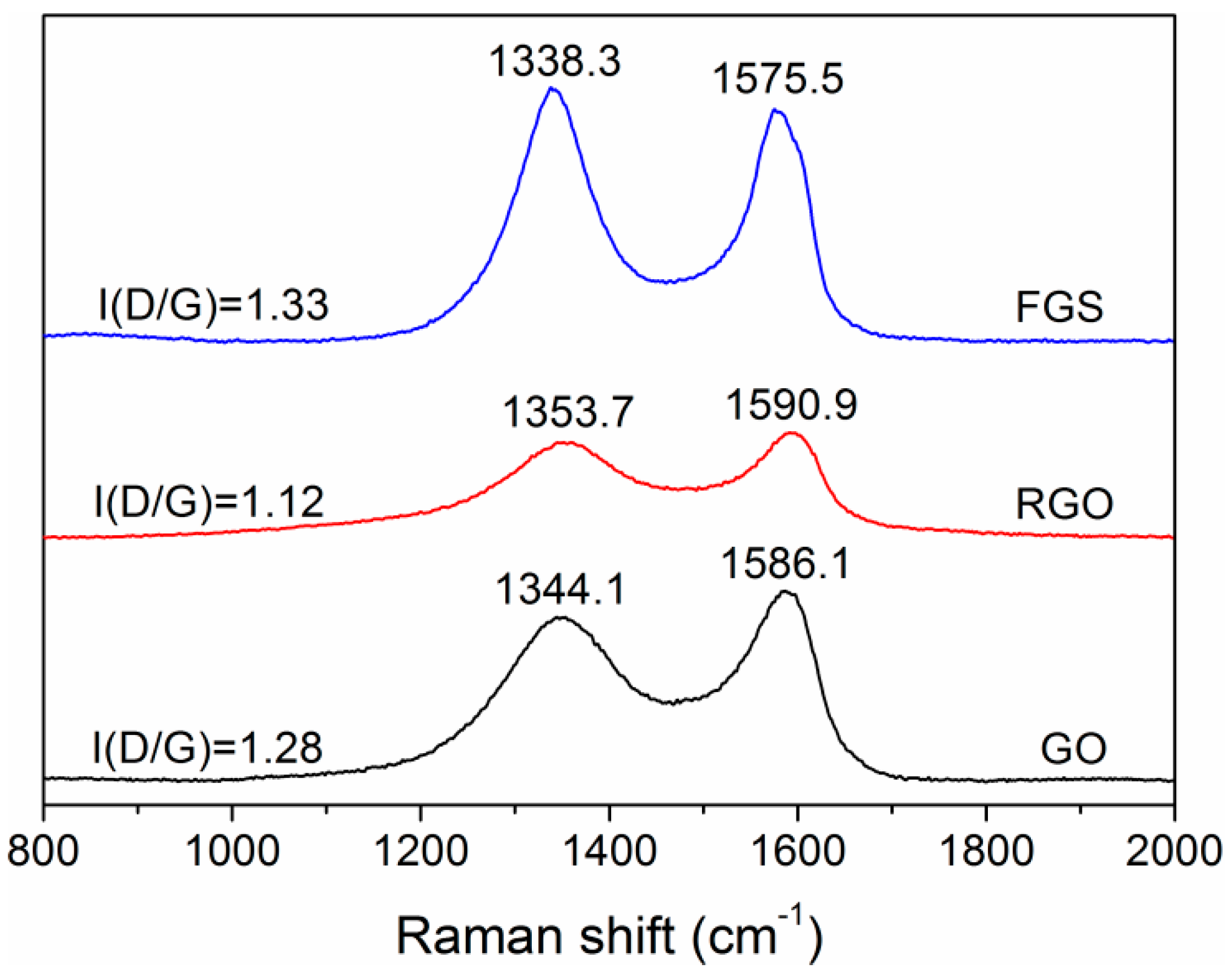
3.1. Structural Characterization of FGS
3.2. Dispersions of Graphene in Polymer Matrix
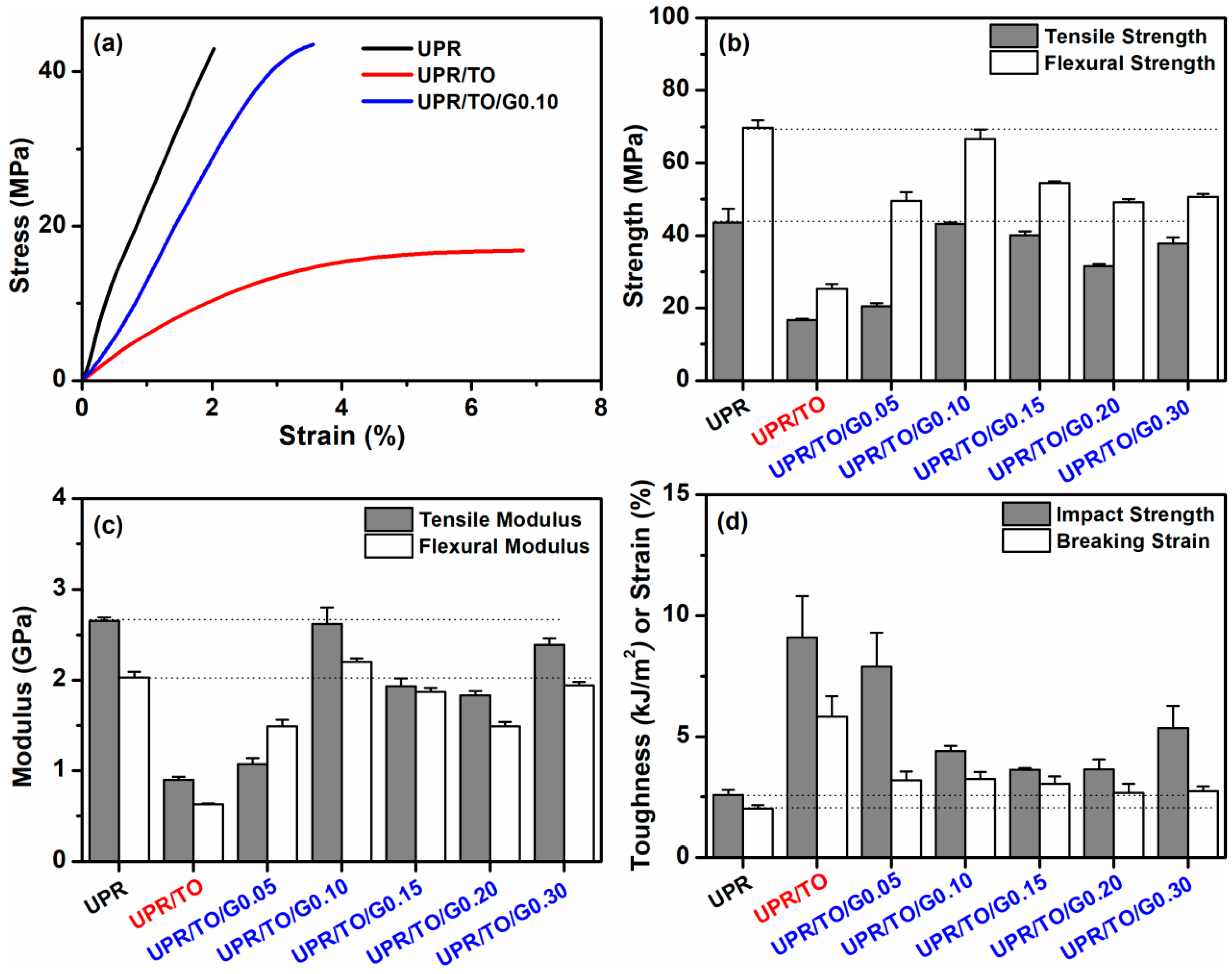
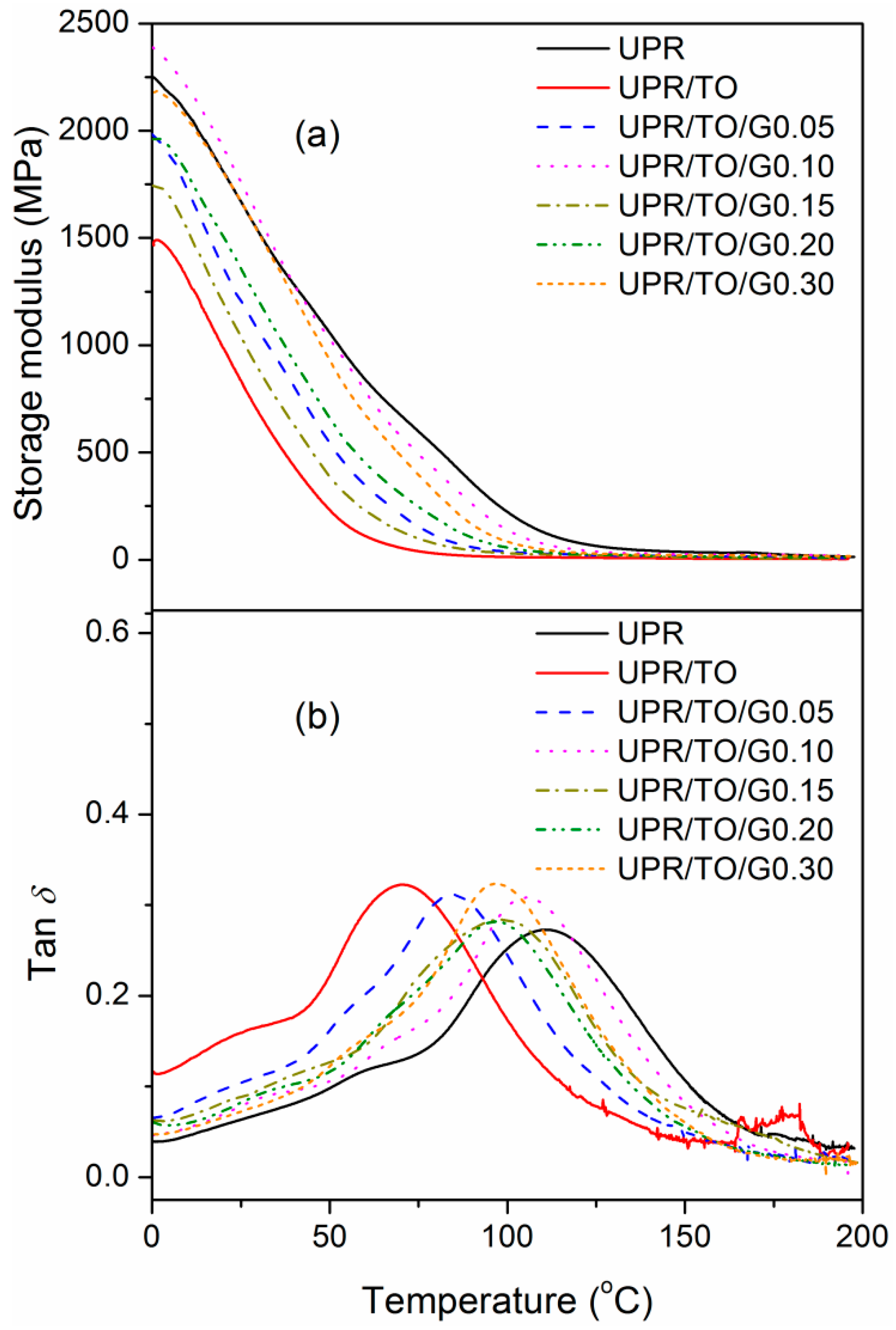
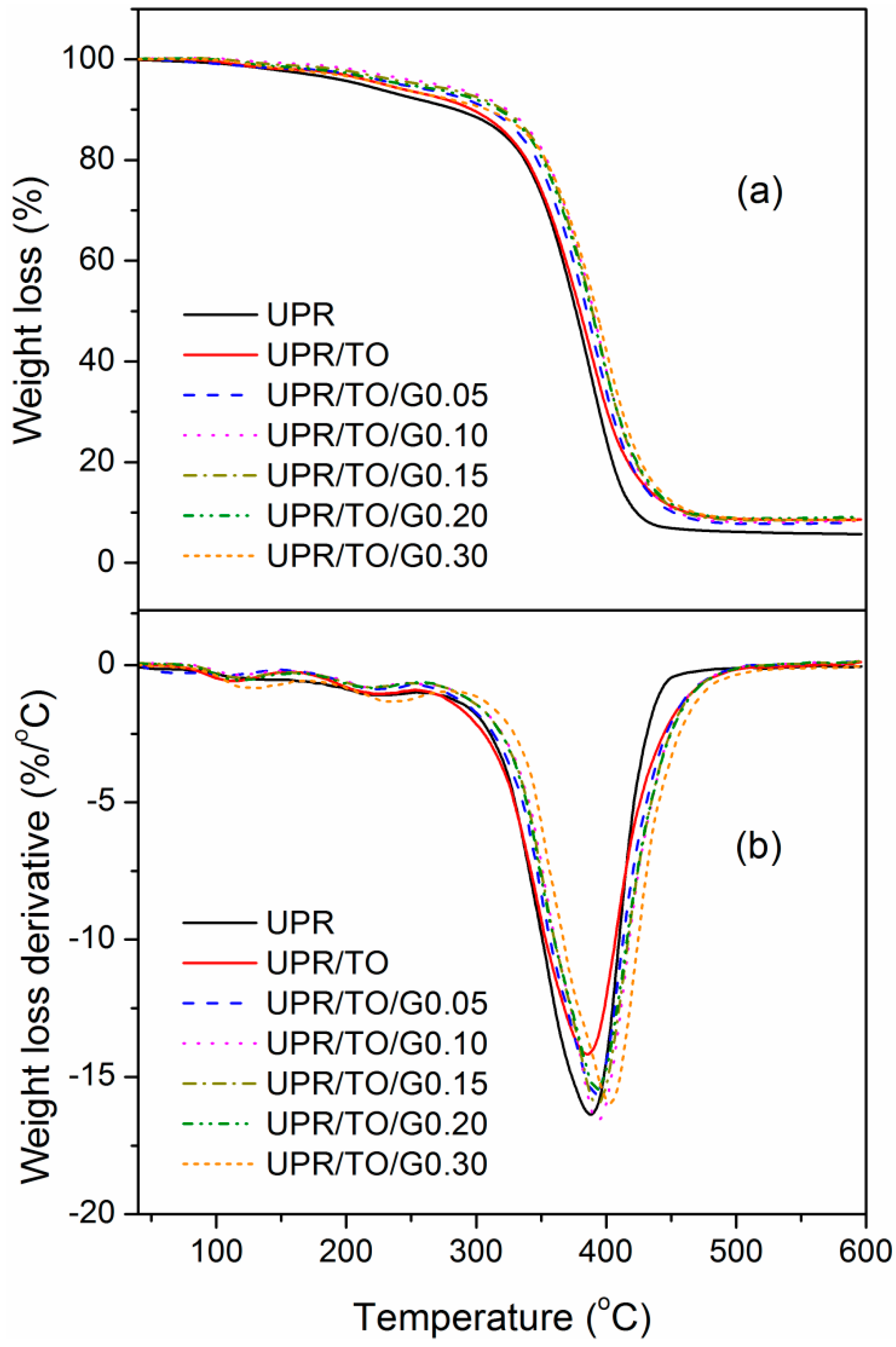
3.3. Properties of the UPR/TO/GO Biobased Nanocomposites
4. Conclusions
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Mighani, H. Synthesis of Thermally Stable Polyesters. In Polyester; Saleh, H.E.-D.M., Ed.; Intech Open: Rijeka, Croatia, 2012; pp. 3–17. [Google Scholar]
- Penczek, P.; Czub, P.; Pielichowski, J. Unsaturated polyester resins: Chemistry and technology. In Crosslinking in Materials Science; Ameduri, B., Ed.; Springer: Berlin, Germany, 2005; pp. 1–95. [Google Scholar]
- Raquez, J.M.; Deléglise, M.; Lacrampe, M.F.; Krawczak, P. Thermosetting (bio)materials derived from renewable resources: A critical review. Prog. Polym. Sci. 2010, 35, 487–509. [Google Scholar] [CrossRef]
- Fertier, L.; Koleilat, H.; Stemmelen, M.; Giani, O.; Joly-Duhamel, C.; Lapinte, V.; Robin, J.-J. The use of renewable feedstock in UV-curable materials—A new age for polymers and green chemistry. Prog. Polym. Sci. 2013, 38, 932–962. [Google Scholar] [CrossRef]
- Zhang, C.Q.; Garrison, T.F.; Madbouly, S.A.; Kessler, M.R. Recent advances in vegetable oil-based polymers and their composites. Prog. Polym. Sci. 2017, 71, 91–143. [Google Scholar] [CrossRef]
- Meier, M.A.R.; Metzger, J.O.; Schubert, U.S. Plant oil renewable resources as green alternatives in polymer science. Chem. Soc. Rev. 2007, 36, 1788–1802. [Google Scholar] [CrossRef] [PubMed]
- Biermann, U.; Bornscheuer, U.; Meier, M.A.R.; Metzger, J.O.; Schäfer, H.J. Oils and fats as renewable raw materials in chemistry. Angew. Chem. Inter. Edit. 2011, 50, 3854–3871. [Google Scholar] [CrossRef] [PubMed]
- Mehta, G.; Mohanty, A.K.; Misra, M.; Drzal, L.T. Biobased resin as a toughening agent for biocomposites. Green Chem. 2004, 6, 254–258. [Google Scholar] [CrossRef]
- Miyagawa, H.; Mohanty, A.K.; Burgueno, R.; Drzal, L.T.; Misra, M. Development of biobased unsaturated polyester containing functionalized linseed oil. Ind. Eng. Chem. Res. 2006, 45, 1014–1018. [Google Scholar] [CrossRef]
- Ghorui, S.; Bandyopadhyay, N.R.; Ray, D.; Sengupta, S.; Kar, T. Use of maleated castor oil as biomodifier in unsaturated polyester resin/fly ash composites. Ind. Crop. Prod. 2011, 34, 893–899. [Google Scholar] [CrossRef]
- Haq, M.; Burgueno, R.; Mohanty, A.; Misra, M. Hybrid bio-based composites from blends of unsaturated polyester and soybean oil reinforced with nanoclay and natural fibers. Compos. Sci. Technol. 2008, 68, 3344–3351. [Google Scholar] [CrossRef]
- Haq, M.; Burgueno, R.; Mohanty, A.K.; Misra, M. Bio-based unsaturated polyester/layered silicate nanocomposites: Characterization and thermo-physical properties. Compos. Part A 2009, 40, 540–547. [Google Scholar] [CrossRef]
- Haq, M.; Burgueno, R.; Mohanty, A.K.; Misra, M. Bio-based polymer nanocomposites from UPE/EML blends and nanoclay: Development, experimental characterization and limits to synergistic performance. Compos. Part A 2011, 42, 41–49. [Google Scholar] [CrossRef]
- Liu, C.G.; Li, J.; Lei, W.; Zhou, Y.H. Development of biobased unsaturated polyester resin containinghighly functionalized castor oil. Ind. Crop. Prod. 2014, 52, 329–337. [Google Scholar] [CrossRef]
- Lu, J.; Hong, C.K.; Wool, R.P. Bio-based nanocomposites from functionalized plant oils and layered silicate. J. Polym. Sci. Polym. Phys. 2004, 42, 1441–1450. [Google Scholar] [CrossRef]
- Lu, J.; Wool, R.P. Additive toughening effects on new bio-based thermosetting resins from plant oils. Compos. Sci. Technol. 2008, 68, 1025–1033. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.B.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Qi, X.; Boey, F.; Zhang, H. Graphene-based composites. Chem. Soc. Rev. 2012, 41, 666–686. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.B.; Kundalwal, S.I.; Kumar, S. Gas barrier performance of graphene/polymer nanocomposites. Carbon 2016, 98, 313–333. [Google Scholar] [CrossRef]
- Li, X.G.; McKenna, G.B. Ultrathin Polymer films: Rubbery stiffening, fragility, and Tg reduction. Macromolecules 2015, 48, 6329–6336. [Google Scholar] [CrossRef]
- Kim, H.; Abdala, A.A.; Macosko, C.W. Graphene/polymer nanocomposites. Macromolecules 2010, 43, 6515–6530. [Google Scholar] [CrossRef]
- Hu, H.T.; Wang, X.B.; Wang, J.C.; Wan, L.; Liu, F.M.; Zheng, H.; Chen, R.; Xu, C. Preparation and properties of graphene nanosheets-polystyrene nanocomposites via in situ emulsion polymerization. Chem. Phys. Lett. 2010, 484, 247–253. [Google Scholar] [CrossRef]
- Patole, A.S.; Patole, S.P.; Kang, H.; Yoo, J.B.; Kim, T.H.; Ahn, J.H. A facile approach to the fabrication of graphene/polystyrene nanocomposite by in situ microemulsion polymerization. J. Colloid. Interface Sci. 2010, 350, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Rafiee, M.A.; Rafiee, J.; Srivastava, I.; Wang, Z.; Song, H.H.; Yu, Z.Z.; Koratkar, N. Fracture and fatigue in graphene nanocomposites. Small 2010, 6, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Bao, C.L.; Guo, Y.Q.; Song, L.; Kan, Y.C.; Qian, X.D.; Hu, Y. In situ preparation of functionalized graphene oxide/epoxy nanocomposites with effective reinforcements. J. Mater. Chem. 2011, 21, 13290–13298. [Google Scholar] [CrossRef]
- Yavari, F.; Rafiee, M.A.; Rafiee, J.; Yu, Z.Z.; Koratkar, N. Dramatic increase in fatigue life in hierarchical graphene composites. ACS Appl. Mater. Inter. 2010, 2, 2738–2743. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, Z.; Huang, Y.; Xie, H.; Liu, Z.; Chen, Y.; Lei, W.; Hu, L.; Zhou, Y.; Cheng, R. One-pot preparation of unsaturated polyester nanocomposites containing functionalized graphene sheets via a novel solvent-exchange method. RSC Adv. 2013, 3, 22380–22388. [Google Scholar] [CrossRef]
- Bora, C.; Bharali, P.; Baglari, S.; Dolui, S.K.; Konwar, B.K. Strong and conductive reduced graphene oxide/polyester resin composite films with improved mechanical strength, thermal stability and its antibacterial activity. Compos. Sci. Technol. 2013, 87, 1–7. [Google Scholar] [CrossRef]
- He, S.Y.; Petkovich, N.D.; Liu, K.W.; Qian, Y.Q.; Macosko, C.W.; Stein, A. Unsaturated polyester resin toughening with very low loadings of GO derivatives. Polymer 2017, 110, 149–157. [Google Scholar] [CrossRef]
- Liu, C.G.; Lei, W.; Cai, Z.C.; Chen, J.Q.; Hu, L.H.; Dai, Y.; Zhou, Y. Use of tung oil as a reactive toughening agent in dicyclopentadiene-terminated unsaturated polyester resins. Ind. Crop. Prod. 2013, 49, 412–418. [Google Scholar] [CrossRef]
- Xu, Z.; Gao, C. In situ polymerization approach to graphene-reinforced nylon-6 composites. Macromolecules 2010, 43, 6716–6723. [Google Scholar] [CrossRef]
- Liu, K.; Chen, L.; Chen, Y.; Wu, J.L.; Zhang, W.Y.; Chen, F.; Fu, Q. Preparation of polyester/reduced graphene oxide composites via in situ melt polycondensation and simultaneous thermo-reduction of graphene oxide. J. Mater. Chem. 2011, 21, 8612–8617. [Google Scholar] [CrossRef]
- Tang, Z.H.; Kang, H.L.; Shen, Z.L.; Guo, B.C.; Zhang, L.Q.; Jia, D.M. Grafting of polyester onto graphene for electrically and thermally conductive composites. Macromolecules 2012, 45, 3444–3451. [Google Scholar] [CrossRef]
- Wei, H.X.; Li, Y.Y.; Chen, J.P.; Zeng, Y.; Yang, G.Q.; Li, Y. Dispersion of reduced graphene oxide in multiple solvents with an imidazolium-modified hexa-peri-hexabenzocoronene. Chem. Asian J. 2012, 7, 2683–2689. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, S.K.; Ray, B.C.; Mallik, A. Role of electrochemically in-house synthesized and functionalized graphene nanofillers in the structural performance of epoxy matrix composites. Phys. Chem. Chem. Phys. 2017, 19, 16219–16230. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, T.; Abdala, A.A.; Stankovich, S.; Dikin, D.A.; Herrera-Alonso, M.; Piner, R.D.; Adamson, D.H.; Schniepp, H.C.; Chen, X.; Ruoff, R.S.; et al. Functionalized graphene sheets for polymer nanocomposites. Nat. Nanotechnol. 2008, 3, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Ray, D.; Banerjee, C.; Bandyopadhyay, N.R.; Mohanty, A.K.; Misra, M. Novel materials from unsaturated polyester resin/styrene/tung oil blends with high impact strengths and enhanced mechanical properties. J. Appl. Polym. Sci. 2011, 119, 2174–2182. [Google Scholar] [CrossRef]
- Chen, D.; Zhu, H.; Liu, T. In situ thermal preparation of polyimide nanocomposite films containing functionalized graphene sheets. ACS Appl. Mater. Inter. 2010, 2, 3702–3708. [Google Scholar] [CrossRef] [PubMed]
- Putz, K.W.; Compton, O.C.; Palmeri, M.J.; Nguyen, S.T.; Brinson, L.C. High-nanofiller-content graphene oxide-polymer nanocomposites via vacuum-assisted self-assembly. Adv. Funct. Mater. 2010, 20, 3322–3329. [Google Scholar] [CrossRef]
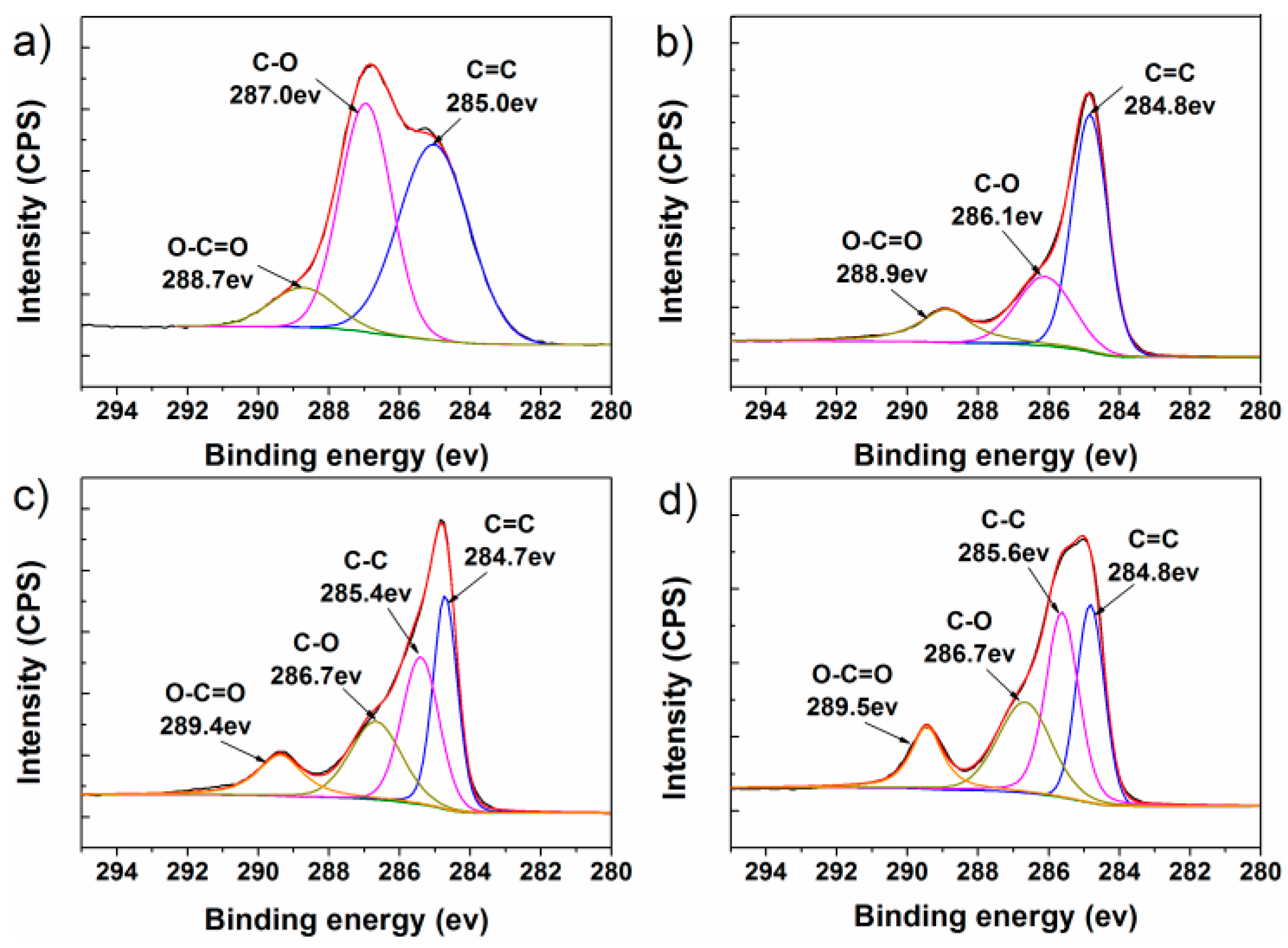

| Sample | Relative Atomic Percentage (%) | C/O Atomic Ratios | |
|---|---|---|---|
| C | O | ||
| GO | 69.0 | 31.0 | 2.23 |
| RGO | 81.9 | 18.1 | 4.53 |
| FGS-i | 78.5 | 21.5 | 3.72 |
| FGS | 80.1 | 19.9 | 4.02 |
| UPR | 62.5 | 37.5 | 1.67 |
| TO | 90.5 | 9.5 | 9.5 |
| Sample | E′25a (GPa) | Tgb (°C) | T5c (°C) | Tpd (°C) | wchare (%) |
|---|---|---|---|---|---|
| UPR | 1.67 | 110.6 | 212.2 | 388.1 | 5.70 |
| UPR/TO | 0.83 | 70.4 | 229.6 | 385.5 | 8.61 |
| UPR/TO/G0.05 | 1.21 | 84.3 | 241.7 | 391.3 | 8.08 |
| UPR/TO/G0.10 | 1.76 | 105.2 | 270.6 | 392.9 | 8.37 |
| UPR/TO/G0.15 | 1.04 | 98.4 | 259.4 | 392.9 | 8.76 |
| UPR/TO/G0.20 | 1.36 | 96.4 | 246.1 | 392.9 | 9.16 |
| UPR/TO/G0.30 | 1.67 | 97.1 | 229.1 | 397.8 | 8.55 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, C.; Wang, C.; Tang, J.; Zhang, J.; Shang, Q.; Hu, Y.; Wang, H.; Wu, Q.; Zhou, Y.; Lei, W.; et al. High-Performance Biobased Unsaturated Polyester Nanocomposites with Very Low Loadings of Graphene. Polymers 2018, 10, 1288. https://doi.org/10.3390/polym10111288
Liu C, Wang C, Tang J, Zhang J, Shang Q, Hu Y, Wang H, Wu Q, Zhou Y, Lei W, et al. High-Performance Biobased Unsaturated Polyester Nanocomposites with Very Low Loadings of Graphene. Polymers. 2018; 10(11):1288. https://doi.org/10.3390/polym10111288
Chicago/Turabian StyleLiu, Chengguo, Cuina Wang, Jijun Tang, Jing Zhang, Qianqian Shang, Yun Hu, Hongxiao Wang, Qiong Wu, Yonghong Zhou, Wen Lei, and et al. 2018. "High-Performance Biobased Unsaturated Polyester Nanocomposites with Very Low Loadings of Graphene" Polymers 10, no. 11: 1288. https://doi.org/10.3390/polym10111288
APA StyleLiu, C., Wang, C., Tang, J., Zhang, J., Shang, Q., Hu, Y., Wang, H., Wu, Q., Zhou, Y., Lei, W., & Liu, Z. (2018). High-Performance Biobased Unsaturated Polyester Nanocomposites with Very Low Loadings of Graphene. Polymers, 10(11), 1288. https://doi.org/10.3390/polym10111288







