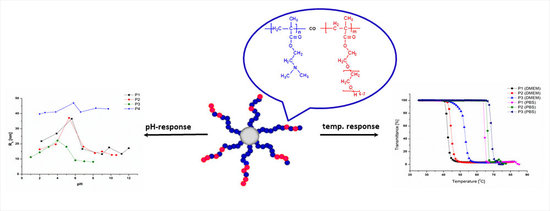
Synthesis, Characterization and Cytotoxicity of Novel Thermoresponsive Star Copolymers of N,N′-Dimethylaminoethyl Methacrylate and Hydroxyl-Bearing Oligo(Ethylene Glycol) Methacrylate
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
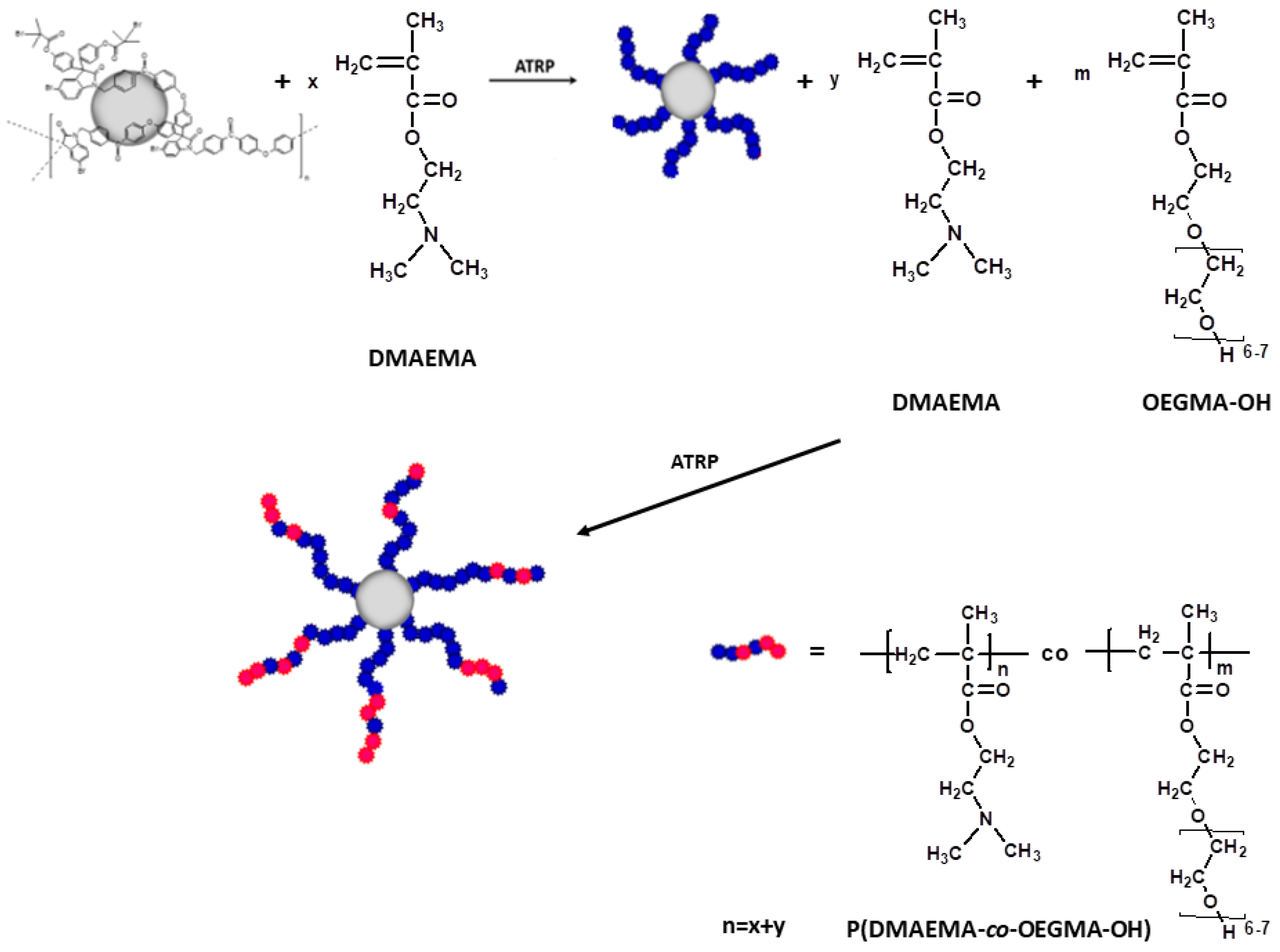
2.2. Synthesis of Star Polymers with Poly[N,N-Dimethylaminoethyl Methacrylate-co-Hydroxyl-Bearing Oligo(Ethylene Glycol) Methacrylate] Arms (P(DMAEMA-co-OEGMA-OH))
2.3. Cell Culture
2.4. Cytotoxicity of Star Polymers
2.5. Methods
3. Results and Discussion
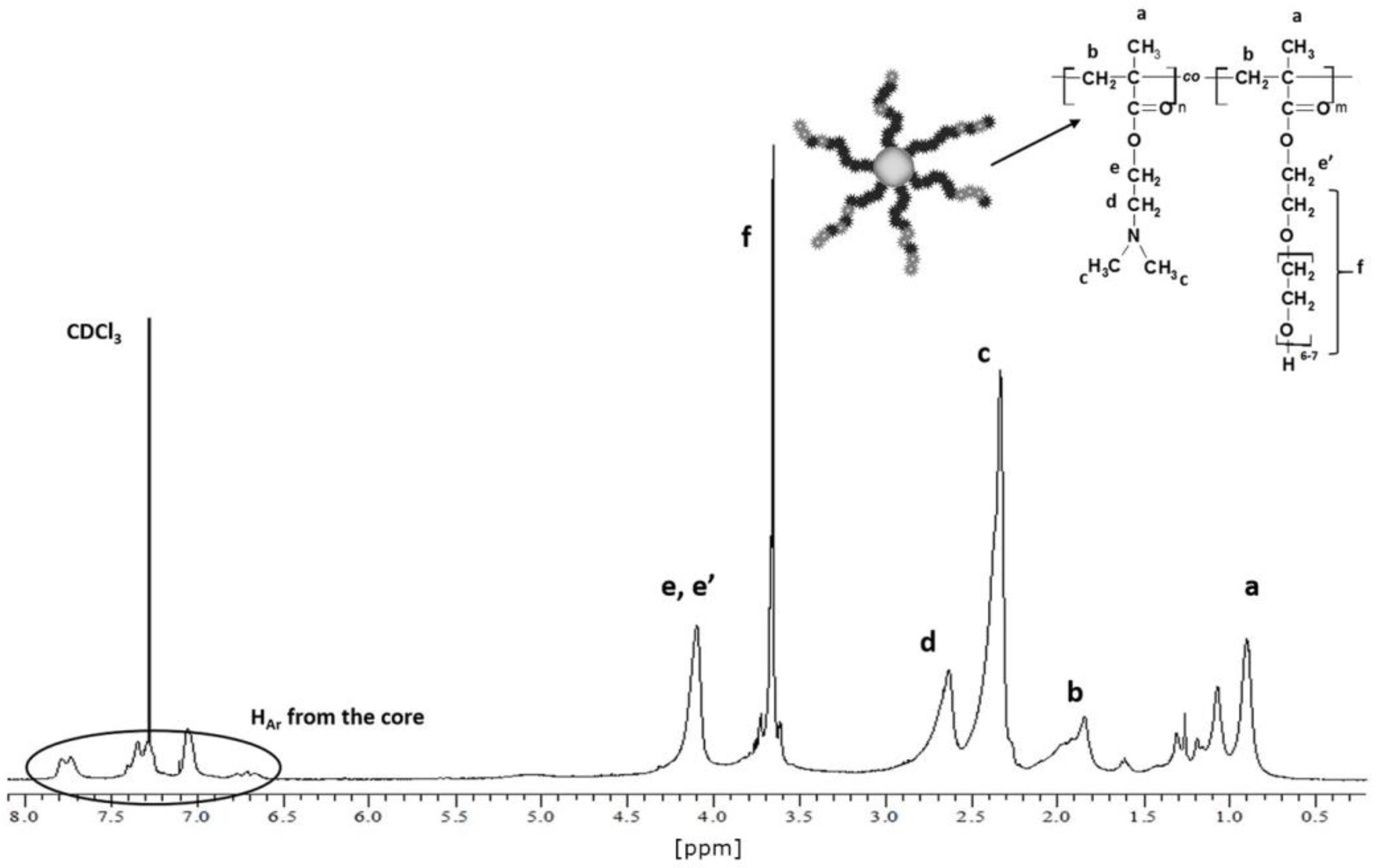
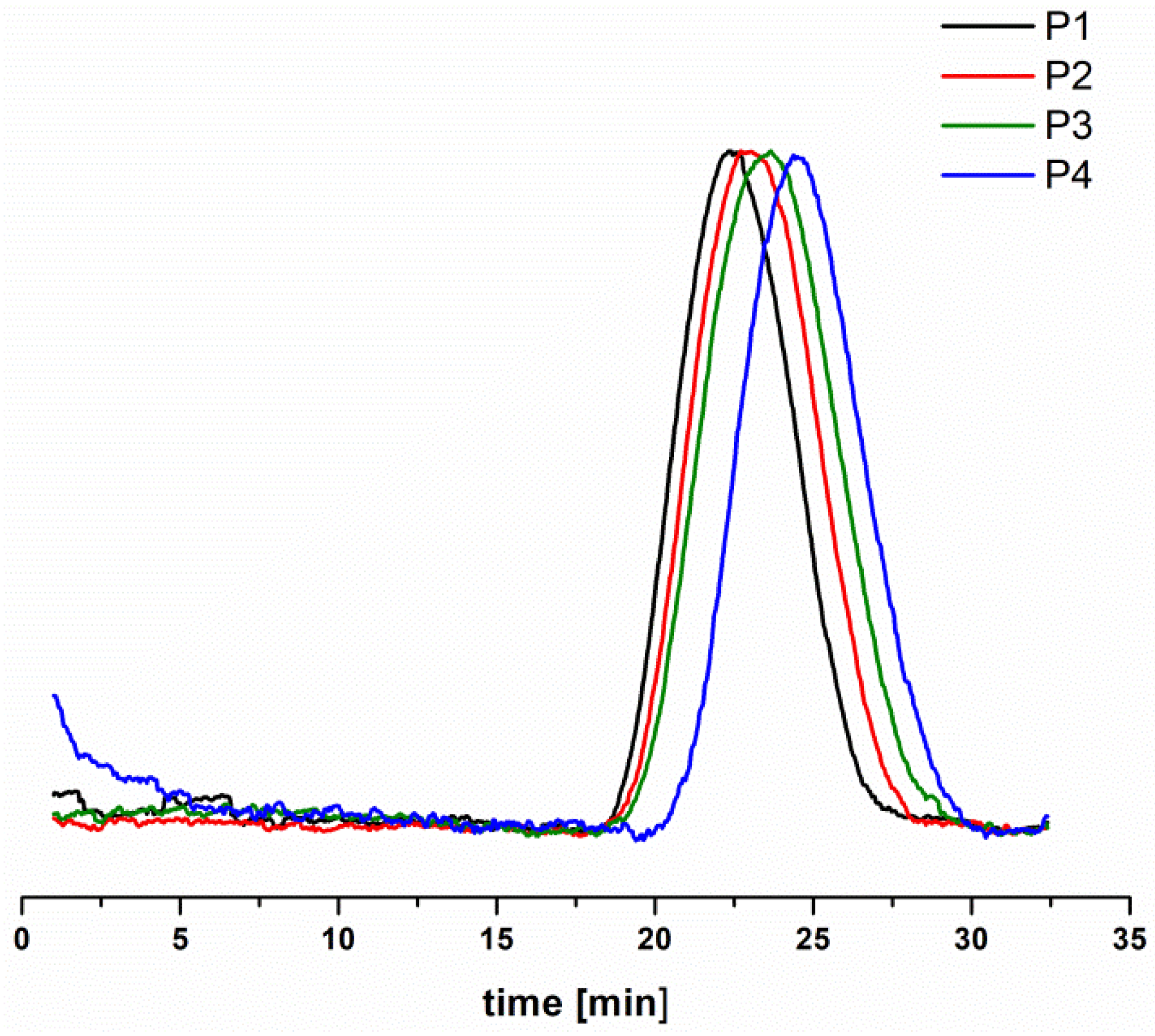
3.1. Synthesis of Star Polymers with Poly[N,N-Dimethylaminoethyl Methacrylate-co-Hydroxyl-Bearing Oligo(Ethylene Glycol) Methacrylate] Arms (P(DMAEMA-co-OEGMA-OH))
3.2. Solution Behavior of the (P(DMAEMA-co-OEGMA-OH)) Star Copolymers
3.3. Temperature-Responsive Properties of the P(DMAEMA-co-OEGMA-OH) Stars
3.4. pH Responsivity and Zeta Potential of the P(DMAEMA-co-OEGMA-OH) Stars
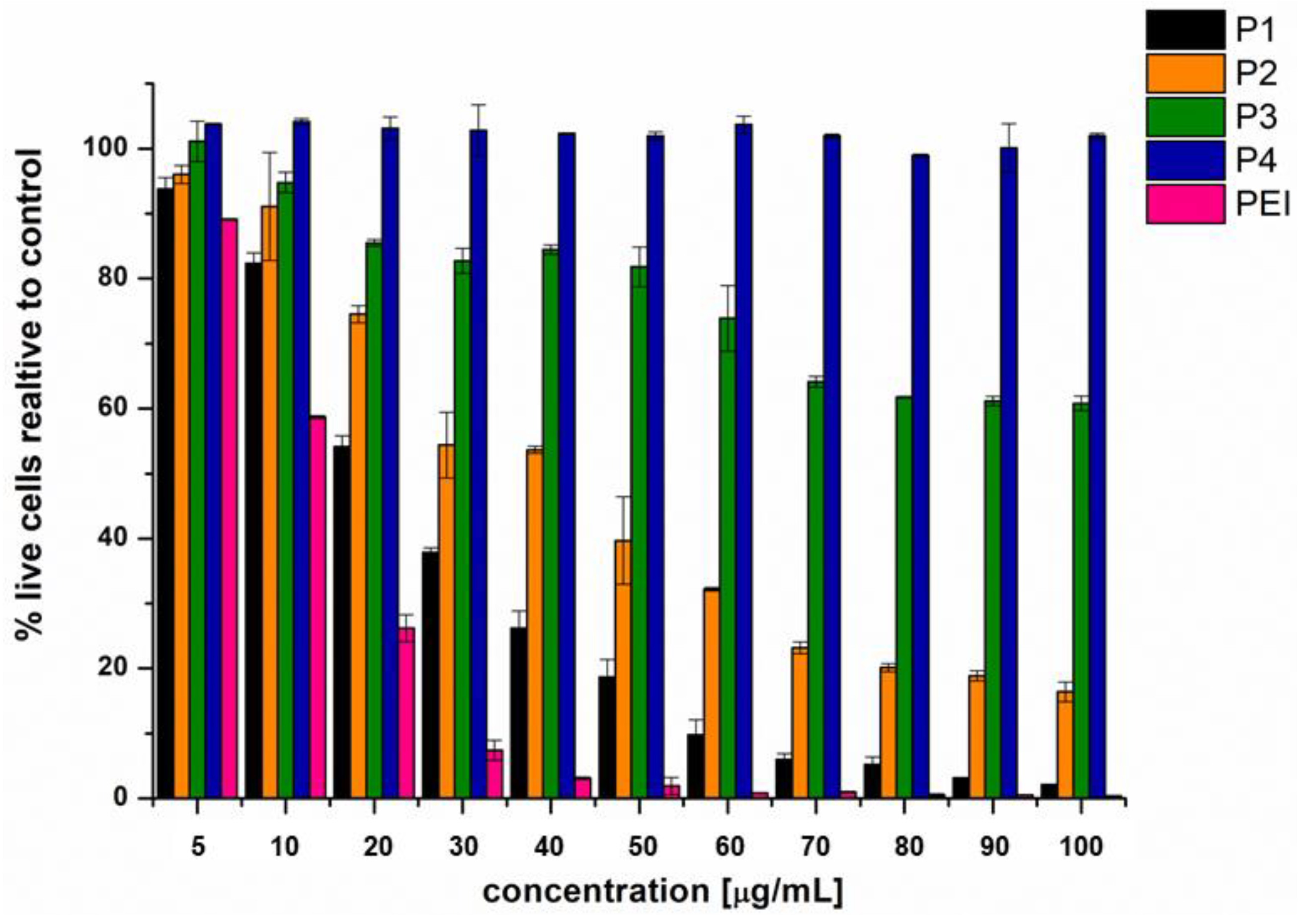
3.5. Cytotoxicity of the Obtained Star Polymers
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ren, J.M.; McKenzie, T.G.; Fu, Q.; Wong, E.H.H.; Xu, J.; An, Z.; Shanmugam, S.; Davis, T.P.; Boyer, C.; Qiao, G.G. Star polymers. Chem. Rev. 2016, 116, 6743–6836. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wang, W.; Li, J. Star polymers: Advances in biomedical applications. Prog. Polym. Sci. 2015, 46, 55–85. [Google Scholar] [CrossRef]
- Kowalczuk, A.; Trzcinska, R.; Trzebicka, B.; Müller, A.H.E.; Dworak, A.; Tsvetanov, C.B. Loading of polymer nanocarriers: Factors, mechanisms and applications. Prog. Polym. Sci. 2014, 39, 43–86. [Google Scholar] [CrossRef]
- Markovsky, E.; Baabur-Cohen, H.; Eldar-Boock, A.; Omer, L.; Tiram, G.; Ferber, S.; Ofek, P.; Polyak, D.; Scomparin, A.; Satchi-Fainaro, R. Administration, distribution, metabolism and elimination of polymer therapeutics. J. Controll. Release 2012, 161, 446–460. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; De Keersmaecker, H.; Corneillie, S.; Yu, F.; Mizuno, H.; Zhang, G.; Hofkens, J.; Mendrek, B.; Kowalczuk, A.; Smet, M. Tunable ratiometric fluorescence sensing of intracellular ph by aggregation-induced emission-active hyperbranched polymer nanoparticles. Chem. Mater. 2015, 27, 3450–3455. [Google Scholar] [CrossRef]
- Yang, K.; Liang, H.; Lu, J. Multifunctional star polymer with reactive and thermosensitive arms and fluorescently labeled core: Synthesis and its protein conjugate. J. Mater. Chem. 2011, 21, 10390–10398. [Google Scholar] [CrossRef]
- Rezaei, S.J.T.; Nabid, M.R.; Niknejad, H.; Entezami, A.A. Folate-decorated thermoresponsive micelles based on star-shaped amphiphilic block copolymers for efficient intracellular release of anticancer drugs. Int. J. Pharm. 2012, 437, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Guo, X.; Qu, Q.; Tang, Z.; Wang, Y.; Zhou, S. Actively targeted delivery of anticancer drug to tumor cells by redox-responsive star-shaped micelles. Biomaterials 2014, 35, 8711–8722. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zaidi, S.S.A.; Hasnain, A.; Guo, J.; Ren, X.; Xia, S.; Zhang, W.; Feng, Y. Multitargeting peptide-functionalized star-shaped copolymers with comblike structure and a poss-core to effectively transfect endothelial cells. ACS Biomater. Sci. Eng. 2018, 4, 2155–2168. [Google Scholar] [CrossRef]
- Cho, H.Y.; Srinivasan, A.; Hong, J.; Hsu, E.; Liu, S.; Shrivats, A.; Kwak, D.; Bohaty, A.K.; Paik, H.-J.; Hollinger, J.O.; et al. Synthesis of biocompatible peg-based star polymers with cationic and degradable core for sirna delivery. Biomacromolecules 2011, 12, 3478–3486. [Google Scholar] [CrossRef] [PubMed]
- Mendrek, B.; Sieroń, Ł.; Żymełka-Miara, I.; Binkiewicz, P.; Libera, M.; Smet, M.; Trzebicka, B.; Sieroń, A.L.; Kowalczuk, A.; Dworak, A. Nonviral plasmid DNA carriers based on N,N′-dimethylaminoethyl methacrylate and di(ethylene glycol) methyl ether methacrylate star copolymers. Biomacromolecules 2015, 16, 3275–3285. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.J.; Zhang, Z.X.; Ping, Y.; Li, J.; Kang, E.T.; Neoh, K.G. Star-shaped cationic polymers by atom transfer radical polymerization from β-cyclodextrin cores for nonviral gene delivery. Biomacromolecules 2009, 10, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Han, F.; Lin, C.; Wen, X.; Zhao, P. Synthesis of bioreducible core crosslinked star polymers with N,N′-bis(acryloyl)cystamine crosslinker via aqueous ethanol dispersion raft polymerization. Polymer 2018, 146, 378–385. [Google Scholar] [CrossRef]
- Wei, X.; Moad, G.; Muir, B.W.; Rizzardo, E.; Rosselgong, J.; Yang, W.; Thang, S.H. An arm-first approach to cleavable mikto-arm star polymers by raft polymerization. Macromol. Rapid Commun. 2014, 35, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Georgiou, T.K.; Vamvakaki, M.; Phylactou, L.A.; Patrickios, C.S. Synthesis, characterization, and evaluation as transfection reagents of double-hydrophilic star copolymers: Effect of star architecture. Biomacromolecules 2005, 6, 2990–2997. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lu, B.; Fan, Q.; Wu, J.; Wei, L.; Hou, J.; Guo, X.; Liu, Z. Synthesis and self-assembly behavior of ph-responsive star-shaped poss-(pcl-p(dmaema-co-pegma))16 inorganic/organic hybrid block copolymer for the controlled intracellular delivery of doxorubicin. RSC Adv. 2016, 6, 61630–61640. [Google Scholar] [CrossRef]
- Lu, B.-B.; Wei, L.-L.; Meng, G.-H.; Hou, J.; Liu, Z.-Y.; Guo, X.-H. Synthesis of self-assemble ph-responsive cyclodextrin block copolymer for sustained anticancer drug delivery. Chin. J. Polym. Sci. 2017, 35, 924–938. [Google Scholar] [CrossRef]
- Yang, Y.Q.; Zhao, B.; Li, Z.D.; Lin, W.J.; Zhang, C.Y.; Guo, X.D.; Wang, J.F.; Zhang, L.J. Ph-sensitive micelles self-assembled from multi-arm star triblock co-polymers poly(ε-caprolactone)-b-poly(2-(diethylamino)ethyl methacrylate)-b-poly(poly(ethylene glycol) methyl ether methacrylate) for controlled anticancer drug delivery. Acta Biomater. 2013, 9, 7679–7690. [Google Scholar] [CrossRef] [PubMed]
- Forbes, D.C.; Frizzell, M.C.H.; Peppas, N.A. Polycationic nanoparticles synthesized using ARGET ATRP for drug delivery. Eur. J. Pharm. Biopharm. 2013, 84, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Kowalczuk, A.; Mendrek, B.; Żymełka-Miara, I.; Libera, M.; Marcinkowski, A.; Trzebicka, B.; Smet, M.; Dworak, A. Solution behavior of star polymers with oligo(ethylene glycol) methyl ether methacrylate arms. Polymer 2012, 53, 5619–5631. [Google Scholar] [CrossRef]
- Mendrek, B. Zachowanie gwieździstych kopolimerów metakrylanów w roztworach. Polimery 2016, 61, 413–420. [Google Scholar] [CrossRef]
- Mendrek, B.; Sieroń, Ł.; Libera, M.; Smet, M.; Trzebicka, B.; Sieroń, A.L.; Dworak, A.; Kowalczuk, A. Polycationic star polymers with hyperbranched cores for gene delivery. Polymer 2014, 55, 4551–4562. [Google Scholar] [CrossRef]
- Ali, M.M.; Stöver, H.D.H. Well-defined amphiphilic thermosensitive copolymers based on poly(ethylene glycol monomethacrylate) and methyl methacrylate prepared by atom transfer radical polymerization. Macromolecules 2004, 37, 5219–5227. [Google Scholar] [CrossRef]
- Kowalczuk, A.; Vandendriessche, A.; Trzebicka, B.; Mendrek, B.; Szeluga, U.; Cholewiński, G.; Smet, M.; Dworak, A.; Dehaen, W. Core-shell nanoparticles with hyperbranched poly(arylene-oxindole) interiors. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 1120–1135. [Google Scholar] [CrossRef]
- Hölter, D.; Burgath, A.; Frey, H. Degree of branching in hyperbranched polymers. Acta Polym. 1997, 48, 30–35. [Google Scholar] [CrossRef]
- Huang, X.; Xiao, Y.; Lang, M. Synthesis of star-shaped pcl-based copolymers via one-pot atrp and their self-assembly behavior in aqueous solution. Macromol. Res. 2012, 20, 597–604. [Google Scholar] [CrossRef]
- Angot, S.; Murthy, K.S.; Taton, D.; Gnanou, Y. Scope of the copper halide/bipyridyl system associated with calixarene-based multihalides for the synthesis of well-defined polystyrene and poly(meth)acrylate stars. Macromolecules 2000, 33, 7261–7274. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Miller, P.J.; Pyun, J.; Kickelbick, G.; Diamanti, S. Synthesis and characterization of star polymers with varying arm number, length, and composition from organic and hybrid inorganic/organic multifunctional initiators. Macromolecules 1999, 32, 6526–6535. [Google Scholar] [CrossRef]
- Newkome, G.R.; Moorefield, C.N.; Baker, G.R.; Saunders, M.J.; Grossman, S.H. Unimolekulare micellen. Angew. Chem. 1991, 103, 1207–1209. [Google Scholar] [CrossRef]
- Paul, C.; Hiemenz, T.P.L. Polymer Chemistry; CRC Press Taylor and Francis Group: Boca Raton, FL, USA, 2007; ISBN 9781574447798. [Google Scholar]
- Plamper, F.A.; Ruppel, M.; Schmalz, A.; Borisov, O.; Ballauff, M.; Müller, A.H.E. Tuning the thermoresponsive properties of weak polyelectrolytes: Aqueous solutions of star-shaped and linear poly(n,n-dimethylaminoethyl methacrylate). Macromolecules 2007, 40, 8361–8366. [Google Scholar] [CrossRef]
- Ward, M.A.; Georgiou, T.K. Thermoresponsive polymers for biomedical applications. Polymers 2011, 3, 1215–1242. [Google Scholar] [CrossRef]
- Dong, Z.; Wei, H.; Mao, J.; Wang, D.; Yang, M.; Bo, S.; Ji, X. Synthesis and responsive behavior of poly(N,N-dimethylaminoethyl methacrylate) brushes grafted on silica nanoparticles and their quaternized derivatives. Polymer 2012, 53, 2074–2084. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, L.; Ma, J.; Wang, J.; Yu, H.; Xiao, A. Temperature- and ph-responsive star amphiphilic block copolymer prepared by a combining strategy of ring-opening polymerization and reversible addition–fragmentation transfer polymerization. Eur. Polym. J. 2010, 46, 1288–1298. [Google Scholar] [CrossRef]
- Cotanda, P.; Wright, D.B.; Tyler, M.; O’Reilly, R.K. A comparative study of the stimuli-responsive properties of dmaea and dmaema containing polymers. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 3333–3338. [Google Scholar] [CrossRef]
- Badi, N. Non-linear peg-based thermoresponsive polymer systems. Prog. Polym. Sci. 2017, 66, 54–79. [Google Scholar] [CrossRef]
- Park, T.G.; Hoffman, A.S. Sodium chloride-induced phase transition in nonionic poly(n-isopropylacrylamide) gel. Macromolecules 1993, 26, 5045–5048. [Google Scholar] [CrossRef]
- Trzebicka, B.; Szweda, D.; Rangelov, S.; Kowalczuk, A.; Mendrek, B.; Utrata-Wesołek, A.; Dworak, A. (co)polymers of oligo(ethylene glycol) methacrylates—Temperature-induced aggregation in aqueous solution. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 614–623. [Google Scholar] [CrossRef]
- Bütün, V.; Armes, S.P.; Billingham, N.C. Synthesis and aqueous solution properties of near-monodisperse tertiary amine methacrylate homopolymers and diblock copolymers. Polymer 2001, 42, 5993–6008. [Google Scholar] [CrossRef]
- Hu, L.; Chu, L.-Y.; Yang, M.; Wang, H.-D.; Hui Niu, C. Preparation and characterization of novel cationic ph-responsive poly(N,N′-dimethylamino ethyl methacrylate) microgels. J. Colloid Interface Sci. 2007, 311, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Uchida, E.; Uyama, Y.; Ikada, Y. Zeta potential of polycation layers grafted onto a film surface. Langmuir 1994, 10, 1193–1198. [Google Scholar] [CrossRef]
- Rinkenauer, A.C.; Schubert, S.; Traeger, A.; Schubert, U.S. The influence of polymer architecture on in vitro pdna transfection. J. Mater. Chem. B 2015, 3, 7477–7493. [Google Scholar] [CrossRef]
- Lam, S.J.; Wong, E.H.H.; Boyer, C.; Qiao, G.G. Antimicrobial polymeric nanoparticles. Prog. Polym. Sci. 2018, 76, 40–64. [Google Scholar] [CrossRef]
- Cai, J.; Yue, Y.; Rui, D.; Zhang, Y.; Liu, S.; Wu, C. Effect of chain length on cytotoxicity and endocytosis of cationic polymers. Macromolecules 2011, 44, 2050–2057. [Google Scholar] [CrossRef]
- Rawlinson, L.-A.B.; O’Brien, P.J.; Brayden, D.J. High content analysis of cytotoxic effects of pdmaema on human intestinal epithelial and monocyte cultures. J. Controll. Release 2010, 146, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Schallon, A.; Jérôme, V.; Walther, A.; Synatschke, C.V.; Müller, A.H.E.; Freitag, R. Performance of three pdmaema-based polycation architectures as gene delivery agents in comparison to linear and branched pei. React. Funct. Polym. 2010, 70, 1–10. [Google Scholar] [CrossRef]
- Synatschke, C.V.; Schallon, A.; Jérôme, V.; Freitag, R.; Müller, A.H.E. Influence of polymer architecture and molecular weight of poly(2-(dimethylamino)ethyl methacrylate) polycations on transfection efficiency and cell viability in gene delivery. Biomacromolecules 2011, 12, 4247–4255. [Google Scholar] [CrossRef] [PubMed]
- Majewski, A.P.; Stahlschmidt, U.; Jérôme, V.; Freitag, R.; Müller, A.H.E.; Schmalz, H. Pdmaema-grafted core–shell–corona particles for nonviral gene delivery and magnetic cell separation. Biomacromolecules 2013, 14, 3081–3090. [Google Scholar] [CrossRef] [PubMed]
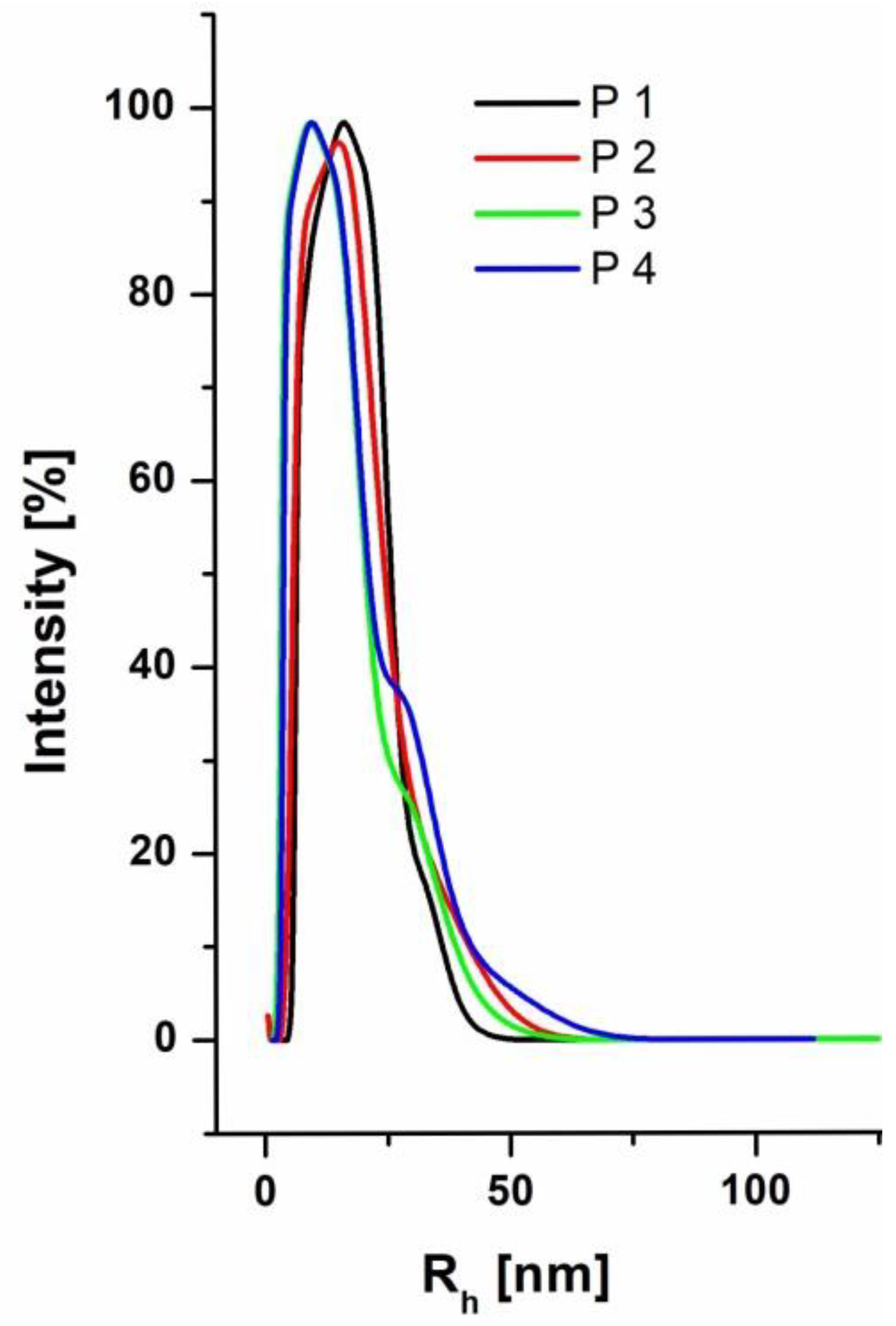
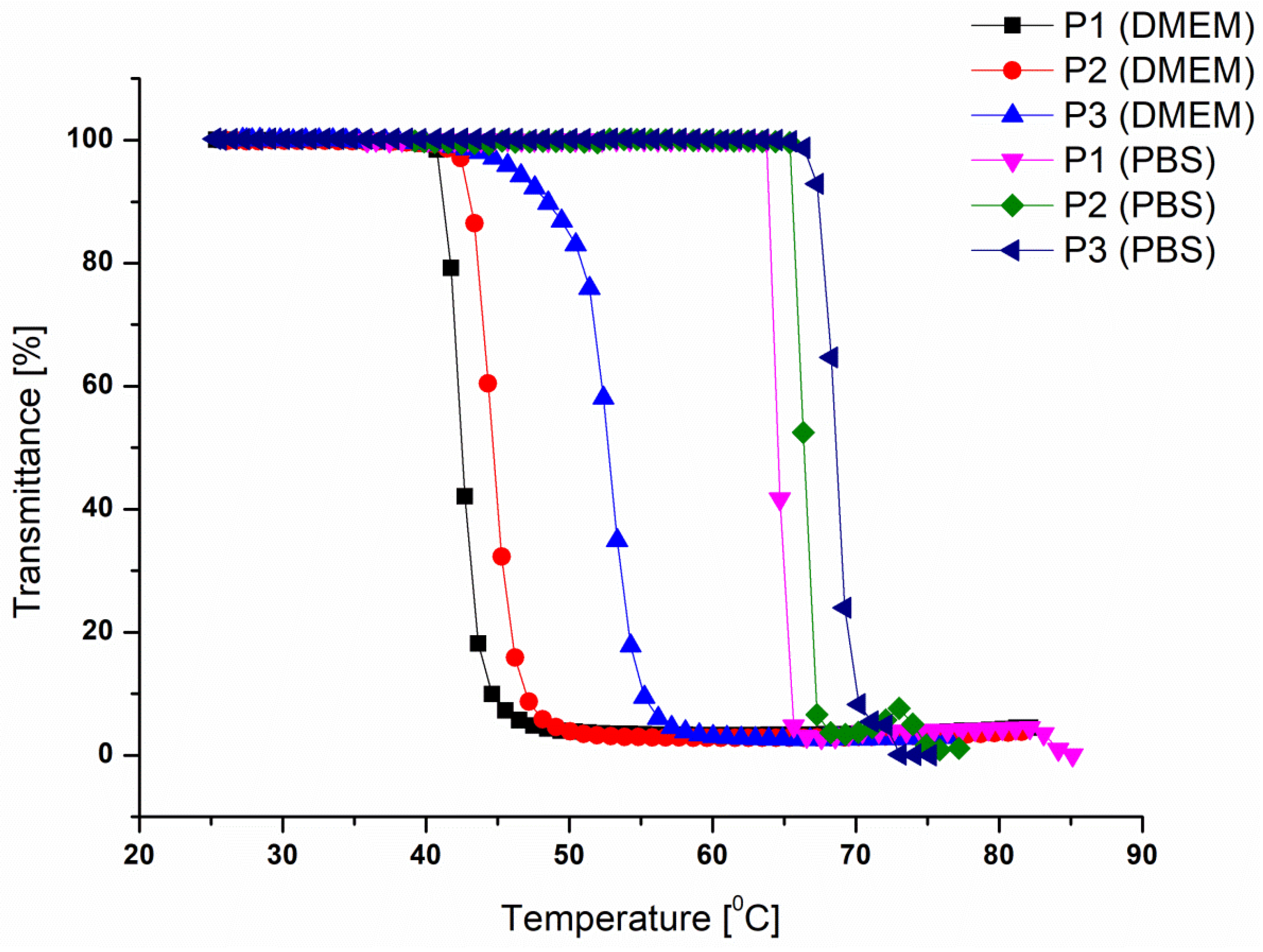
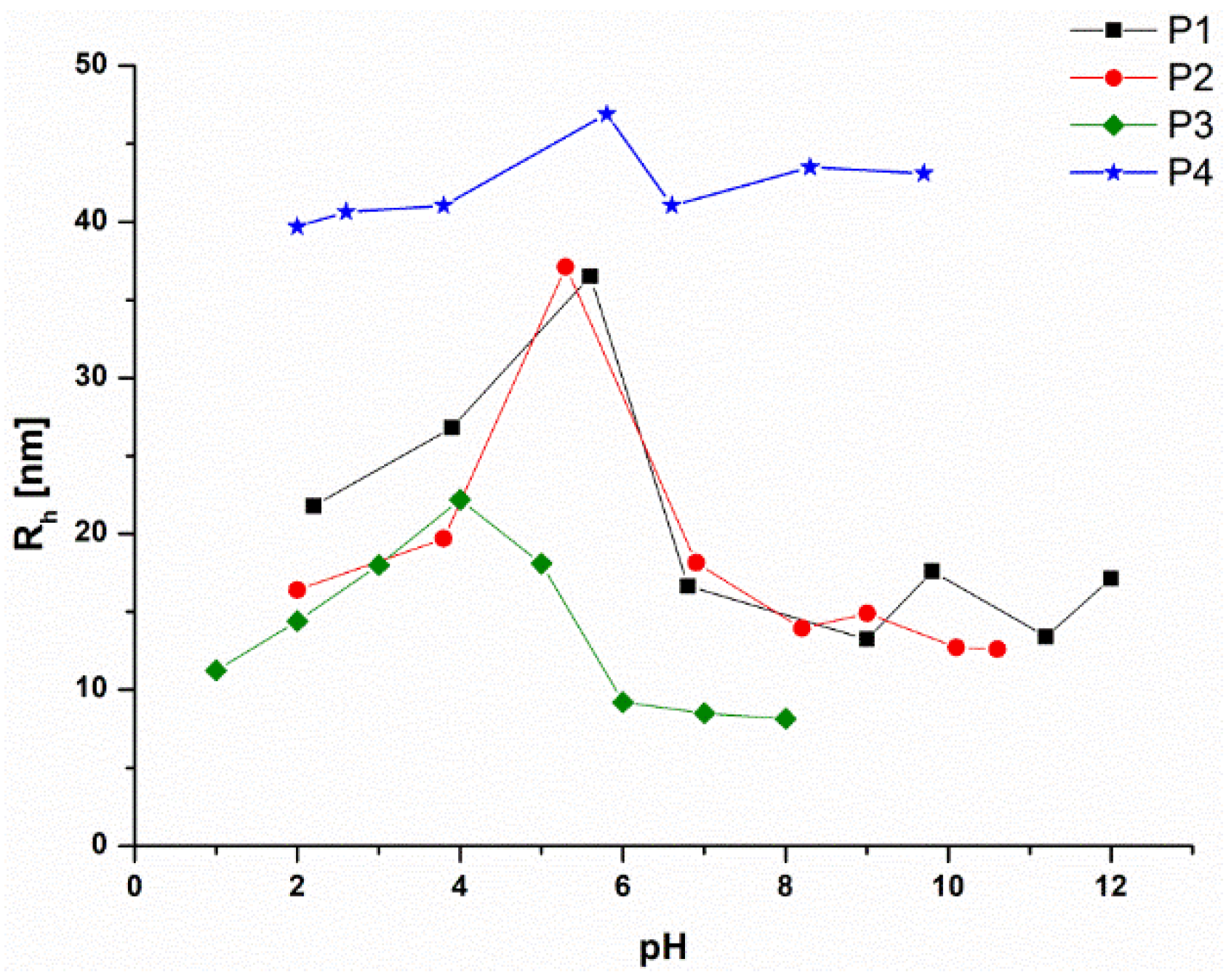
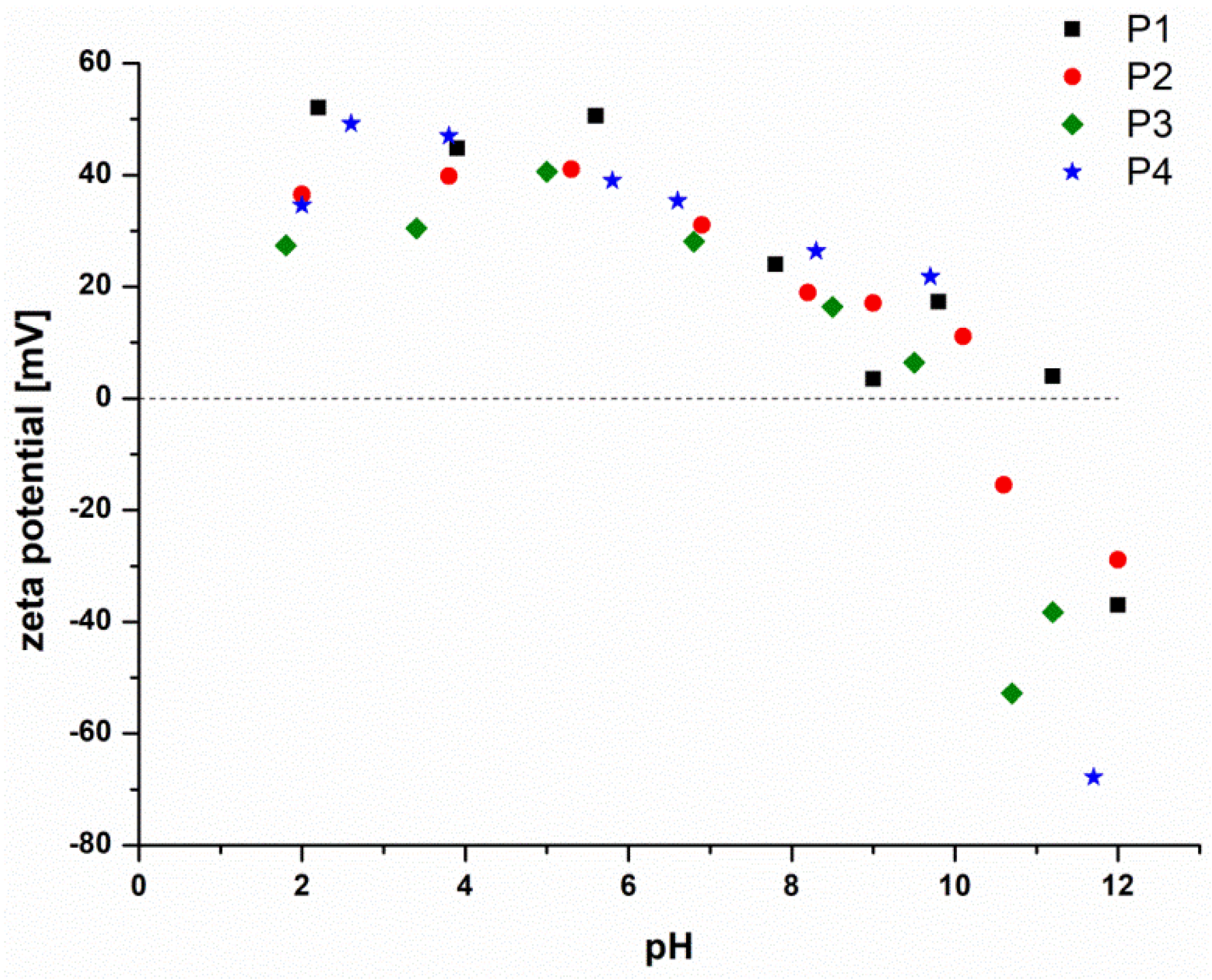
| Star | [DMAEMA]: [OEGMA]:[PArOx] a | Conversion DMAEMA:OEGMA b [%] | OEGMA-OH Content c [mol%] |
|---|---|---|---|
| P1 | 100:10:1 | 44:3 | 2.6 |
| P2 | 50:5:1 | 62:29 | 2.8 |
| P3 | 30:3:1 | 60:56 | 4.2 |
| P4 | 40:10:1 | 36:30 | 10.0 |
| Star | Mna [g/mol] | dn/dca [mL/g] | Mw/Mna | Mtheor b [g/mol] |
|---|---|---|---|---|
| P1 | 257 000 | 0.067 | 2.4 | 240 000 |
| P2 | 159 000 | 0.074 | 2.5 | 172 000 |
| P3 | 132 000 | 0.081 | 2.3 | 117 000 |
| P4 | 100 000 | 0.126 | 2.2 | 114 000 |
| Star | Mn [g/mol] | Contour Length of Arm [nm] | Rh90° [nm] | ||||
|---|---|---|---|---|---|---|---|
| Acetone | EtOH | Water (pH Dissolution) | DMEM | PBS | |||
| P1 | 257,000 | 11.3 | 10.0 ± 2 | 22.4 ± 2 | 17.0 ± 2 (6.8) | 20.0 ± 2 | 14.4 ± 1 |
| P2 | 159,000 | 8.2 | 11.0 ± 2 | 20.0 ± 3 | 18.0 ± 1 (6.9) | 19.3 ± 2 | 13.3 ± 2 |
| P3 | 132,000 | 5.0 | 13.6 ± 1 | 20.5 ± 2 | 18.0 ± 2 (6.8) | 11.7 ± 1 | 10.7 ± 3 |
| P4 | 100,000 | 4.3 | 9.0 ± 2 | 26.6 ± 1 | 41.0 ± 2 (6.6) | 27.3 ± 2 | 14.0 ± 1 |
| Star | Mna [g/mol] | OEGMA-OH Content [mol%] | TCP [°C] | |
|---|---|---|---|---|
| PBS | DMEM | |||
| P1 | 257 000 | 2.6 | 64.5 | 42.4 |
| P2 | 159 000 | 2.8 | 66.3 | 44.5 |
| P3 | 132 000 | 4.2 | 68.5 | 52.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendrek, B.; Fus, A.; Klarzyńska, K.; Sieroń, A.L.; Smet, M.; Kowalczuk, A.; Dworak, A. Synthesis, Characterization and Cytotoxicity of Novel Thermoresponsive Star Copolymers of N,N′-Dimethylaminoethyl Methacrylate and Hydroxyl-Bearing Oligo(Ethylene Glycol) Methacrylate. Polymers 2018, 10, 1255. https://doi.org/10.3390/polym10111255
Mendrek B, Fus A, Klarzyńska K, Sieroń AL, Smet M, Kowalczuk A, Dworak A. Synthesis, Characterization and Cytotoxicity of Novel Thermoresponsive Star Copolymers of N,N′-Dimethylaminoethyl Methacrylate and Hydroxyl-Bearing Oligo(Ethylene Glycol) Methacrylate. Polymers. 2018; 10(11):1255. https://doi.org/10.3390/polym10111255
Chicago/Turabian StyleMendrek, Barbara, Agnieszka Fus, Katarzyna Klarzyńska, Aleksander L. Sieroń, Mario Smet, Agnieszka Kowalczuk, and Andrzej Dworak. 2018. "Synthesis, Characterization and Cytotoxicity of Novel Thermoresponsive Star Copolymers of N,N′-Dimethylaminoethyl Methacrylate and Hydroxyl-Bearing Oligo(Ethylene Glycol) Methacrylate" Polymers 10, no. 11: 1255. https://doi.org/10.3390/polym10111255
APA StyleMendrek, B., Fus, A., Klarzyńska, K., Sieroń, A. L., Smet, M., Kowalczuk, A., & Dworak, A. (2018). Synthesis, Characterization and Cytotoxicity of Novel Thermoresponsive Star Copolymers of N,N′-Dimethylaminoethyl Methacrylate and Hydroxyl-Bearing Oligo(Ethylene Glycol) Methacrylate. Polymers, 10(11), 1255. https://doi.org/10.3390/polym10111255