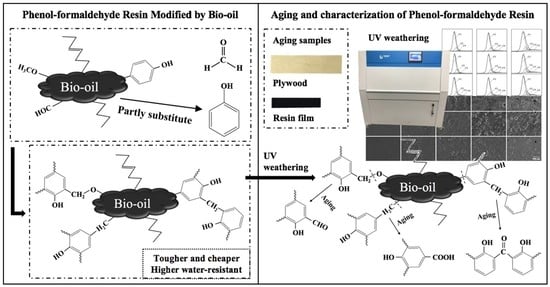
Aging Properties of Phenol-Formaldehyde Resin Modified by Bio-Oil Using UV Weathering
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis and Characterization of Resins
2.3. Preparation of Aging Materials
2.4. UV Weathering
2.5. Analysis
3. Results and Discussion
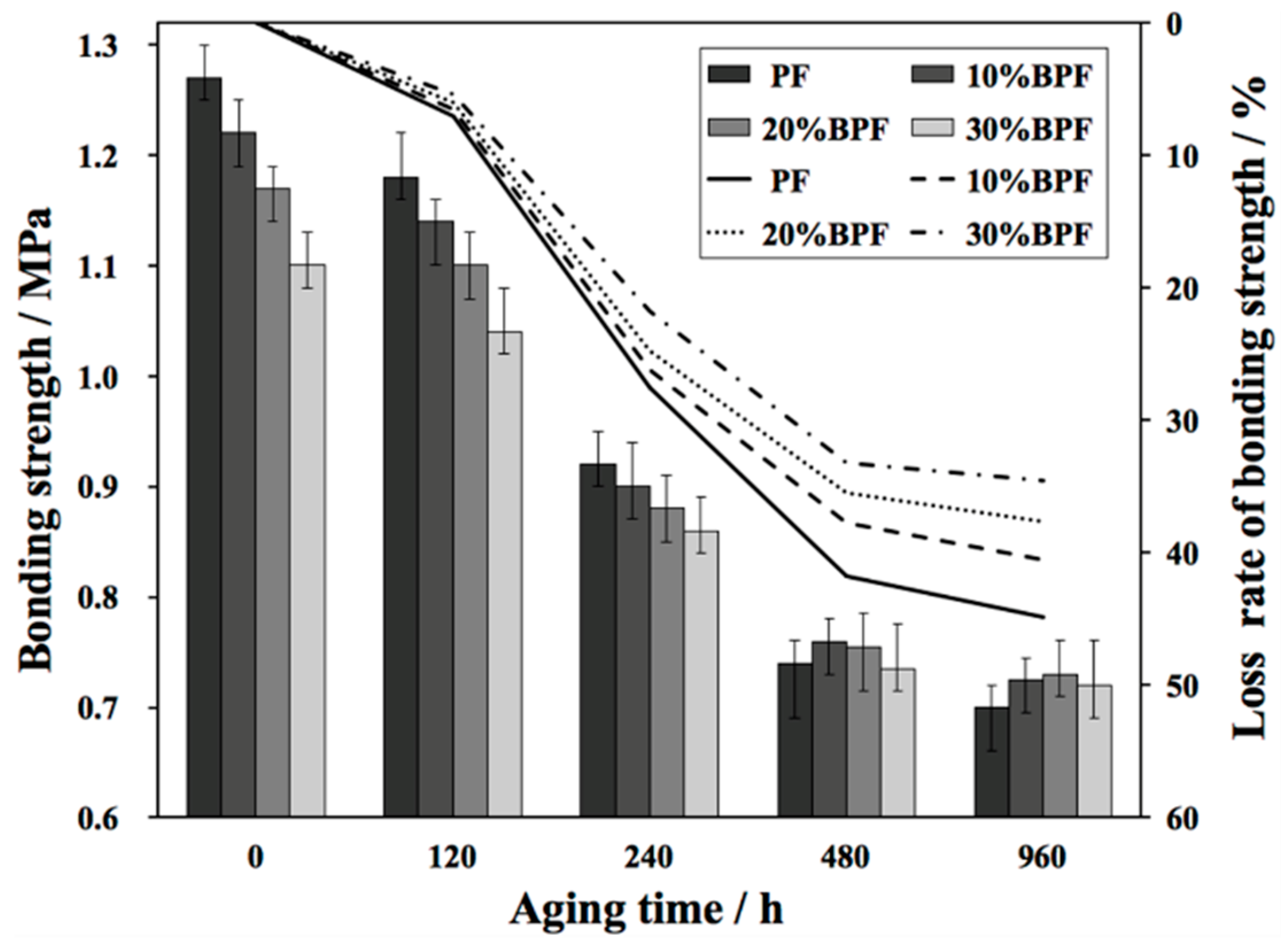
3.1. Bonding Strength
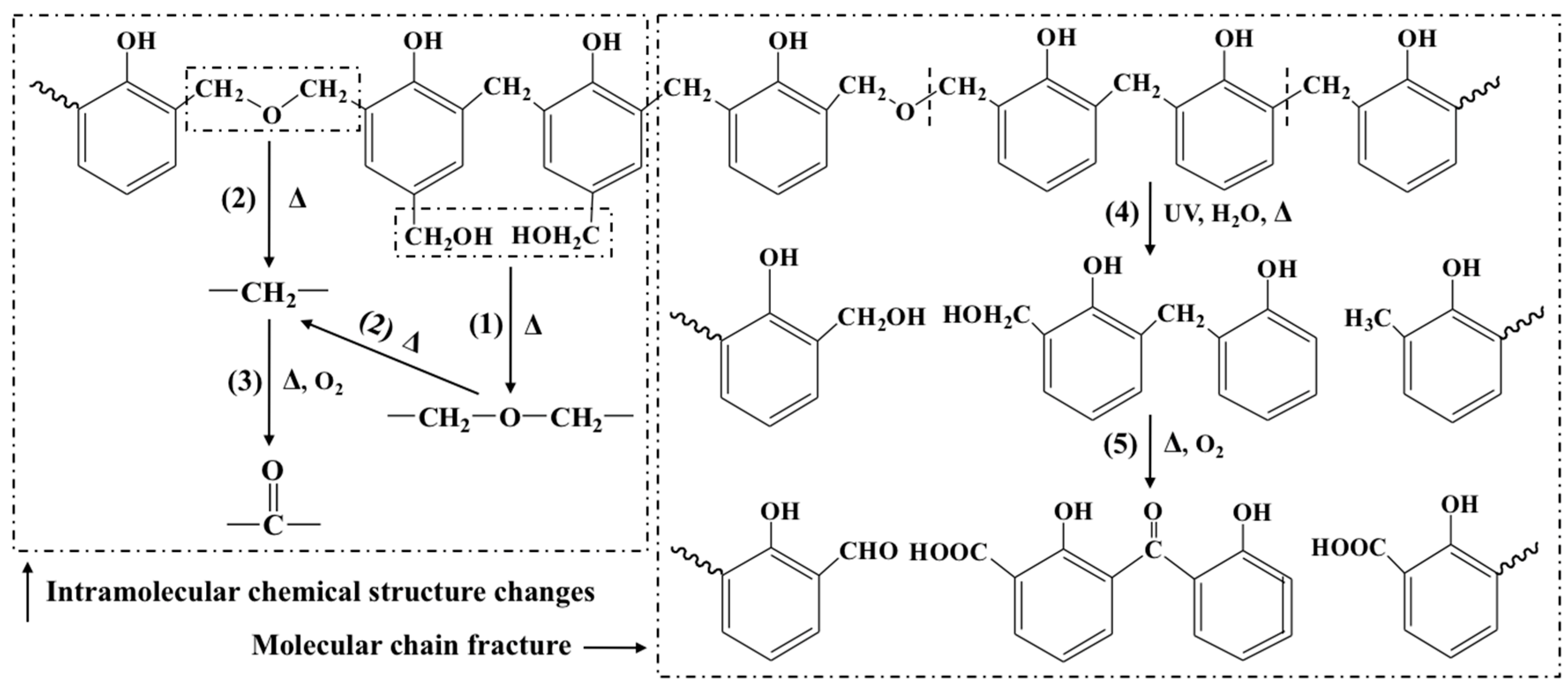
3.2. Microstructure
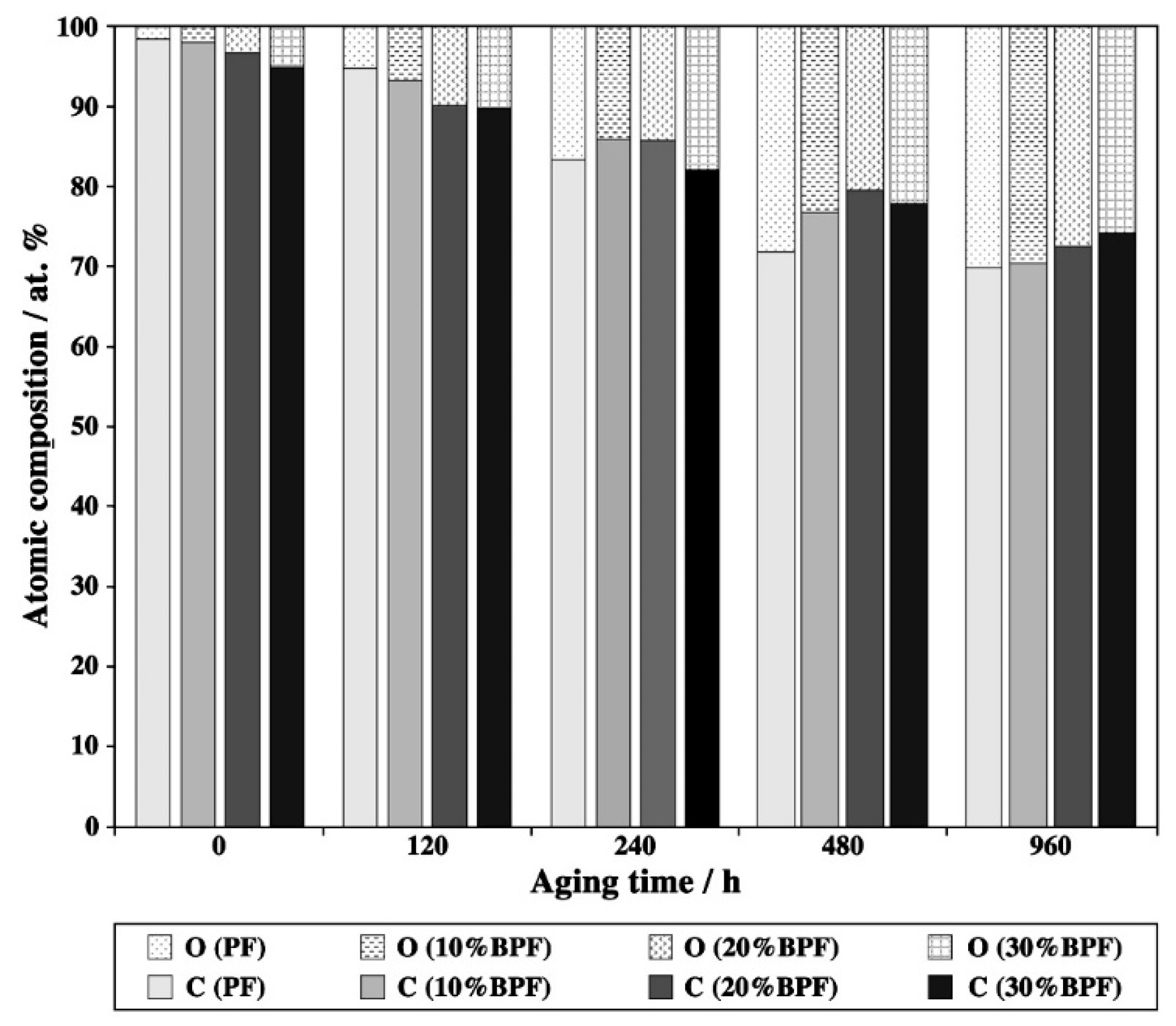
3.3. XPS Analysis
3.4. NMR Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hirano, K.; Asami, M. Phenolic resins—100 years of progress and their future. React. Funct. Polym. 2013, 73, 256–269. [Google Scholar] [CrossRef]
- Pilato, L. Phenolic resins: 100 Years and still going strong. React. Funct. Polym. 2013, 73, 270–277. [Google Scholar] [CrossRef]
- Stoeckel, F.; Konnerth, J.; Gindl-Altmutter, W. Mechanical properties of adhesives for bonding wood—A review. Int. J. Adhes. Adhes. 2013, 45, 32–41. [Google Scholar] [CrossRef]
- Pethrick, R.A. Design and ageing of adhesives for structural adhesive bonding—A review. Proc. Inst. Mech. Eng. Part L 2014, 229, 1–31. [Google Scholar] [CrossRef]
- Cherian, B.M.; Leao, A.L.; Chaves, M.R.; Souza, S.F.; Sain, M.; Narine, S.S. Environmental ageing studies of chemically modified micro and nanofibril phenol formaldehyde composites. Ind. Crops Prod. 2013, 49, 471–483. [Google Scholar] [CrossRef]
- Guo, D.; Zhan, M.; Wang, K. Microstructure evolution of ammonia-catalyzed phenolic resin during thermooxidative aging. J. Appl. Polym. Sci. 2012, 126, 2010–2016. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, Z.; Xiao, S.; Liu, H. A novel thermal degradation mechanism of phenol-formaldehyde type resins. Thermochim. Acta 2008, 476, 39–43. [Google Scholar] [CrossRef]
- Klausler, O.; Clauß, S.; Lubke, L.; Trachsel, J.; Niemz, P. Influence of moisture on stress-strain behaviour of adhesives used for structural bonding of wood. Int. J. Adhes. Adhes. 2013, 44, 57–65. [Google Scholar] [CrossRef]
- Yue, K.; Chen, Z.; Lu, W.; Liu, W.; Li, M.; Shao, Y.; Tang, L.; Wan, L. Evaluating the mechanical and fire-resistance properties of modified fast-growing Chinese fir timber with boric-phenol-formaldehyde resin. Constr. Build. Mater. 2017, 154, 956–962. [Google Scholar] [CrossRef]
- Kobera, L.; Czernek, J.; Streckova, M.; Urbanova, M.; Abbrent, S.; Brus, J. Structure and distribution of cross-links in boron-modified phenol-formaldehyde resins designed for soft magnetic composites: A multiple-quantum 11B-11B MAS NMR correlation spectroscopy study. Macromolecules 2015, 48, 4874–4881. [Google Scholar] [CrossRef]
- Zhang, Y.; Shen, S.; Liu, Y. The effect of titanium incorporation on the thermal stability of phenol-formaldehyde resin and its carbonization microstructure. Polym. Degrad. Stab. 2013, 98, 514–518. [Google Scholar] [CrossRef]
- Huskic, M.; Zagar, E. Catalytic activity of mineral montmorillonite on the reaction of phenol with formaldehyde. Appl. Clay Sci. 2017, 136, 158–163. [Google Scholar] [CrossRef]
- Fang, Q.; Cui, H.; Du, G. Montmorillonite reinforced phenol formaldehyde resin: Preparation, characterization, and application in wood bonding. Int. J. Adhes. Adhes. 2014, 49, 33–37. [Google Scholar] [CrossRef]
- Lubczak, R. Thermally resistant carbazole-modified phenol-formaldehyde resins. Polym. Int. 2015, 64, 1163–1171. [Google Scholar] [CrossRef]
- Cardona, F.; Kin-Tak, A.L.; Fedrigo, J. Novel phenolic resins with improved mechanical and toughness properties. Appl. Polym. Sci. 2012, 123, 2131–2139. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Chrissafis, K. Thermal study of phenol–formaldehyde resin modified with cashew nut shell liquid. Thermochim. Acta 2011, 512, 105–109. [Google Scholar] [CrossRef]
- Wang, W.; Li, X.; Ye, D.; Cai, L.; Shi, S.Q. Catalytic pyrolysis of larch sawdust for phenol-rich bio-oil using different catalysts. Renew. Energ. 2018, 121, 146–152. [Google Scholar] [CrossRef]
- Kim, J. Production, separation and applications of phenolic-rich bio-oil—A review. Bioresour. Technol. 2015, 178, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenerg. 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Dong, F.; Wang, M.; Wang, Z. Bio-oil as substitute of phenol for synthesis of resol-type phenolic resin as wood adhesive. Int. J. Chem. React. Eng. 2017, 16, 1–7. [Google Scholar] [CrossRef]
- Vithanage, A.E.; Chowdhury, E.; Alejo, L.D.; Pomeroy, P.C.; DeSisto, W.J.; Frederick, B.G.; Gramlich, W.M. Renewably sourced phenolic resins from lignin bio-oil. J. Appl. Polym. Sci. 2017, 134, 44827. [Google Scholar] [CrossRef]
- Aslan, M.; Özbay, G.; Ayrilmis, N. Adhesive characteristics and bonding performance of phenol formaldehyde modified with phenol-rich fraction of crude bio-oil. J. Adhes. Sci. Technol. 2015, 29, 2679–2691. [Google Scholar] [CrossRef]
- Choi, G.; Oh, S.; Lee, S.; Kim, J. Production of bio-based phenolic resin and activated carbon from bio-oil and biochar derived from fast pyrolysis of palm kernel shells. Bioresour. Technol. 2015, 178, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Chaouch, M.; Diouf, P.N.; Laghdir, A.; Yin, S. Bio-oil from whole-tree feedstock in resol-type phenolic resins. J. Appl. Polym. Sci. 2014, 131, 40014. [Google Scholar] [CrossRef]
- Strzemieckaa, B.; Zieba-Palusb, J.; Voelkela, A.; Lachowiczc, T.; Sochad, E. Examination of the chemical changes in cured phenol-formaldehyde resins during storage. J. Chromatogr. A 2016, 1441, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Özbaya, G.; Ayrilmis, N. Bonding performance of wood bonded with adhesive mixtures composed of phenol-formaldehyde and bio-oil. Ind. Crops Prod. 2015, 66, 68–72. [Google Scholar] [CrossRef]
- Monni, J.; Alvila, L.; Rainio, J.; Pakkanen, T.T. Effects of silane and the lowering of pH on the properties of phenol–formaldehyde resol resins and resin coatings. J. Appl. Polym. Sci. 2007, 104, 1933–1941. [Google Scholar] [CrossRef]
- Okhrimenko, D.V.; Thomsen, A.B.; Ceccato, M.; Johansson, D.B.; Lybye, D.; Bechgaard, K.; Tougaard, S.; Stipp, S.L.S. Impact of curing time on ageing and degradation of phenol-urea-formaldehyde binder. Polym. Degrad. Stab. 2018, 152, 86–94. [Google Scholar] [CrossRef]
- Yu, Y.; Xu, P.; Chen, C.; Chang, J.; Li, L. Formaldehyde emission behavior of plywood with phenol-formaldehyde resin modified by bio-oil under radiant floor heating condition. Build. Environ. 2018. [Google Scholar] [CrossRef]



| Resins | B/P Substitute Rate (%) | Characteristics | |||
|---|---|---|---|---|---|
| pH (25 °C) | Viscosity (25 °C, mPa·s) | Solids Content (%) | Water Absorption (25 °C, 24 h, %) | ||
| PF | 0 | 10.98 ± 0.12 | 117 ± 22 | 47.64 ± 0.28 | 30.26 ± 1.12 |
| 10%BPF | 10 | 10.57 ± 0.15 | 185 ± 40 | 46.42 ± 0.19 | 27.34 ± 0.98 |
| 20%BPF | 20 | 10.27 ± 0.09 | 278 ± 31 | 45.23 ± 0.23 | 24.33 ± 0.73 |
| 30%BPF | 30 | 9.95 ± 0.16 | 636 ± 58 | 42.69 ± 0.21 | 22.32 ± 1.36 |
| Aging Time | Peak Area (%) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C1 (284.8, C–H) a | C2 (285.6, C–O) | C3 (286.5, C=O) | C4 (288.4, O–C=O) | |||||||||||||
| 0 | 1 | 2 | 3 b | 0 | 1 | 2 | 3 | 0 | 1 | 2 | 3 | 0 | 1 | 2 | 3 | |
| 0 | 84.3 | 81.9 | 80.0 | 78.6 | 13.6 | 14.6 | 14.2 | 14.8 | 2.1 | 3.0 | 4.8 | 5.2 | — | 0.5 | 1.0 | 1.4 |
| 120 | 80.3 | 79.8 | 77.9 | 74.0 | 14.2 | 12.8 | 15.3 | 15.1 | 3.1 | 4.7 | 3.7 | 7.3 | 2.4 | 2.7 | 3.1 | 3.6 |
| 240 | 75.1 | 73.2 | 74.3 | 72.3 | 14.8 | 16.5 | 14.4 | 18.0 | 5.6 | 6.6 | 6.7 | 5.5 | 4.5 | 3.7 | 4.6 | 4.2 |
| 480 | 70.9 | 70.3 | 70.9 | 71.2 | 13.9 | 16.9 | 16.0 | 17.4 | 10.0 | 8.2 | 6.6 | 6.3 | 5.2 | 4.6 | 6.5 | 5.1 |
| 960 | 66.2 | 67.0 | 68.1 | 68.4 | 13.4 | 14.7 | 14.2 | 14.9 | 10.8 | 8.7 | 8.6 | 8.2 | 9.6 | 9.6 | 9.1 | 8.5 |
| Chemical Group Structure | Chemical Shirt (ppm) | Calculation | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 20%BPF | PF | ||||||||||
| 0 | 120 | 240 | 480 | 960 | 0 | 120 | 240 | 480 | 960 | ||
| C=O form aldehydes/ketones | 220–200 | 0.98 | 1.08 | 1.11 | 1.19 | 1.24 | 0.85 | 1.10 | 1.26 | 1.37 | 1.32 |
| C=O from carboxylic acids | 186–176 | 0.09 | 0.14 | 0.23 | 0.26 | 0.30 | 0.02 | 0.02 | 0.03 | 0.12 | 0.18 |
| C=O from esters | 176–168 | 0.05 | 0.05 | 0.12 | 0.17 | 0.16 | 0.01 | 0.01 | 0.02 | 0.09 | 0.14 |
| Phenolic carbon | 160–148 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Substituted ortho and para aromatic carbons | 137–123 | 4.19 | 4.52 | 4.29 | 4.26 | 4.20 | 4.21 | 4.43 | 4.31 | 4.22 | 4.07 |
| Dimethylene ether bridges | 75–65 | 0.56 | 0.68 | 0.71 | 0.50 | 0.48 | 0.20 | 0.46 | 0.26 | 0.18 | 0.24 |
| Methylol groups | 63–58 | 0.32 | 0.54 | 0.43 | 0.33 | 0.27 | 0.19 | 0.36 | 0.25 | 0.20 | 0.23 |
| Benzylamines | 58–45 | 0.53 | 0.88 | 0.91 | 1.11 | 1.20 | 0.56 | 1.06 | 0.89 | 0.96 | 0.74 |
| Methylene bridges | 45–29 | 2.27 | 2.40 | 2.36 | 2.27 | 2.10 | 2.17 | 2.38 | 2.29 | 2.16 | 2.03 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Xu, P.; Chang, M.; Chang, J. Aging Properties of Phenol-Formaldehyde Resin Modified by Bio-Oil Using UV Weathering. Polymers 2018, 10, 1183. https://doi.org/10.3390/polym10111183
Yu Y, Xu P, Chang M, Chang J. Aging Properties of Phenol-Formaldehyde Resin Modified by Bio-Oil Using UV Weathering. Polymers. 2018; 10(11):1183. https://doi.org/10.3390/polym10111183
Chicago/Turabian StyleYu, Yuxiang, Pingping Xu, Miaomiao Chang, and Jianmin Chang. 2018. "Aging Properties of Phenol-Formaldehyde Resin Modified by Bio-Oil Using UV Weathering" Polymers 10, no. 11: 1183. https://doi.org/10.3390/polym10111183
APA StyleYu, Y., Xu, P., Chang, M., & Chang, J. (2018). Aging Properties of Phenol-Formaldehyde Resin Modified by Bio-Oil Using UV Weathering. Polymers, 10(11), 1183. https://doi.org/10.3390/polym10111183