Relaxation Dynamics in Polyethylene Glycol/Modified Hydrotalcite Nanocomposites
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Nanocomposite Preparation
2.3. Characterization
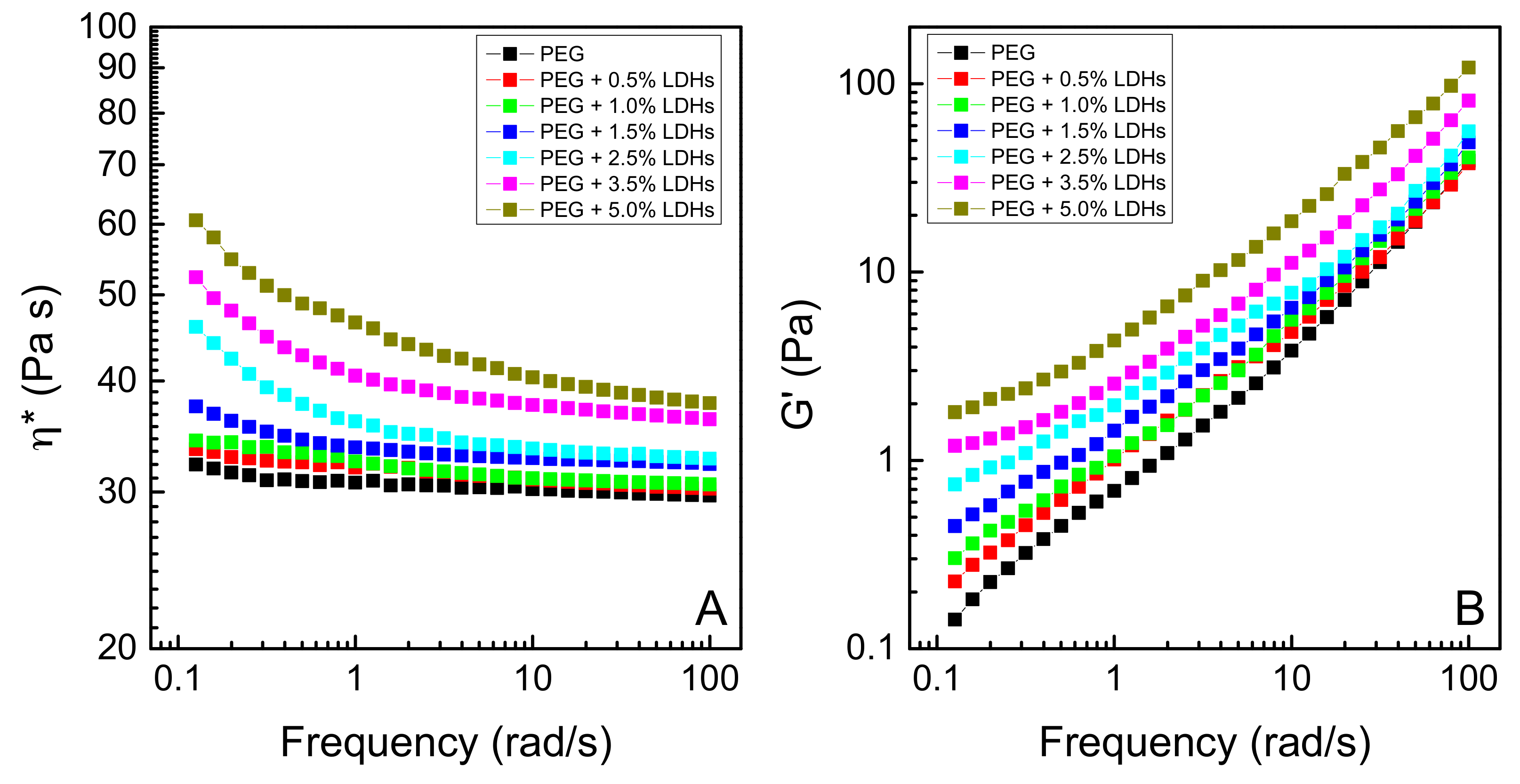
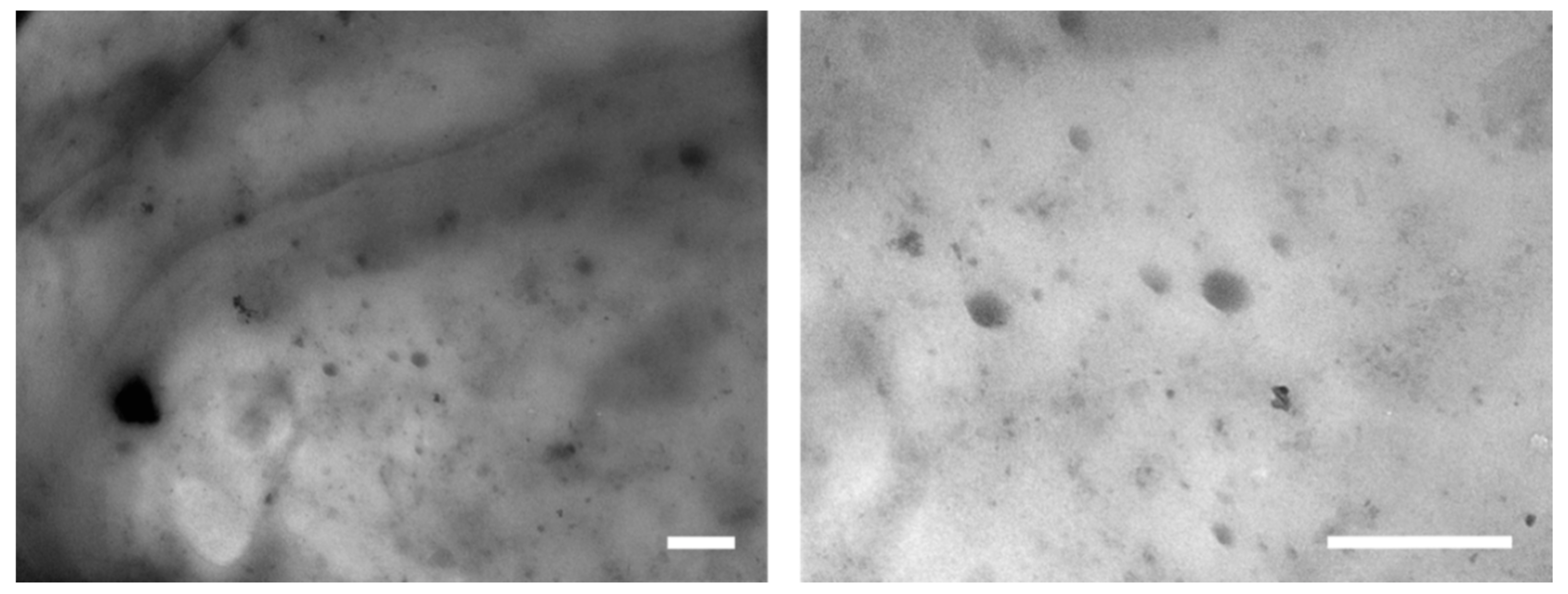
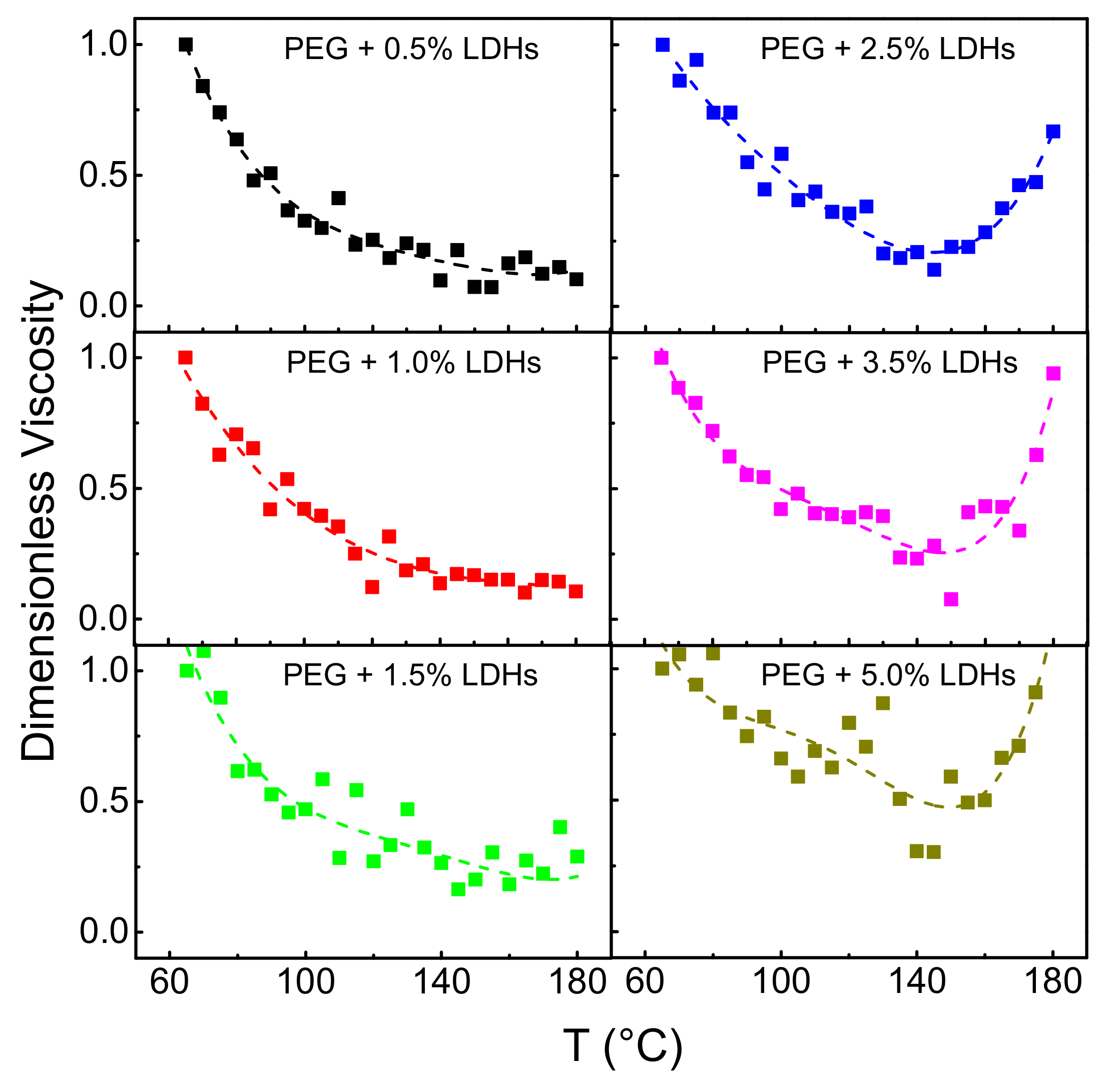
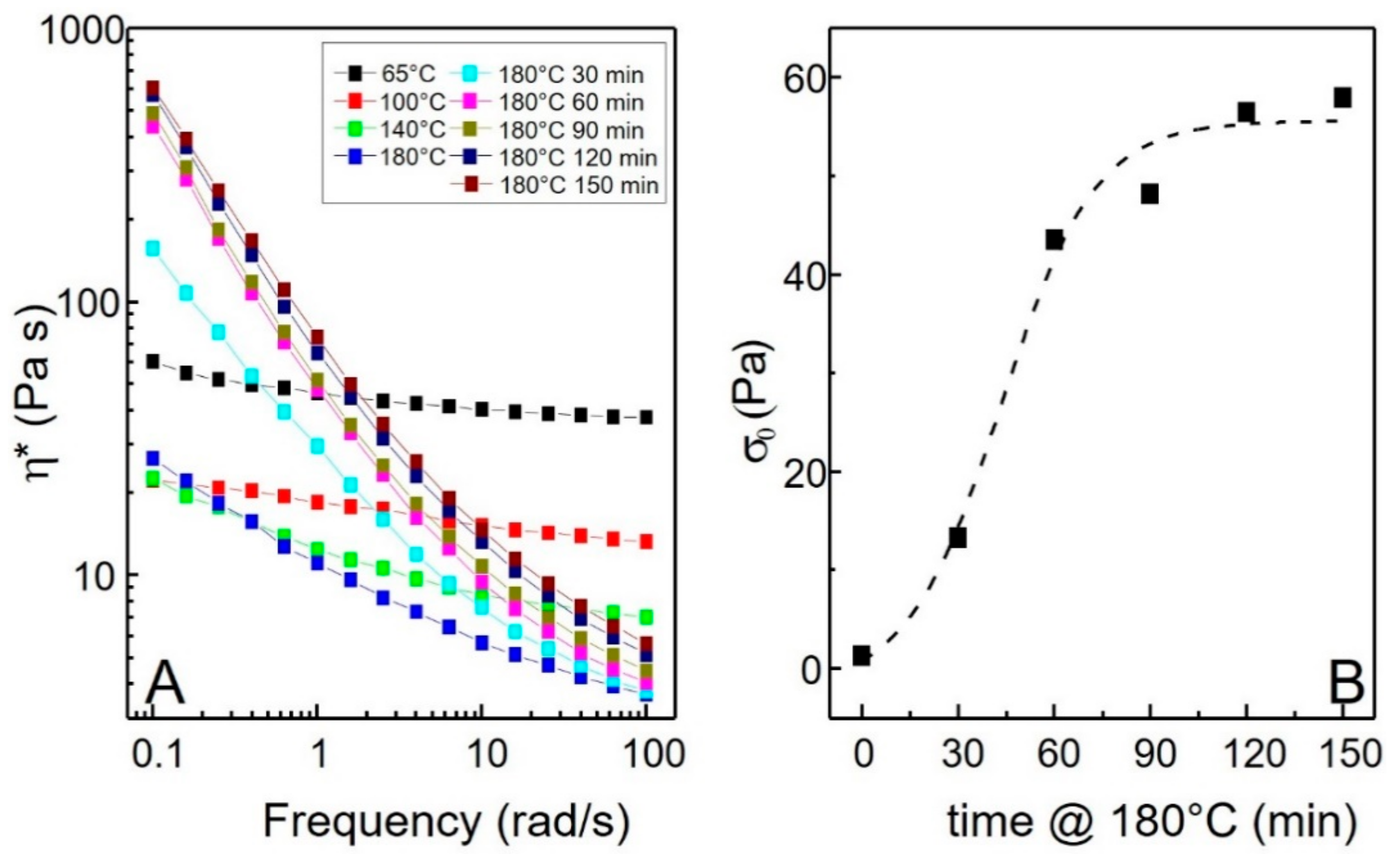
3. Results and Discussion
3.1. Effect of LDHs Loading
3.2. Effect of Temperature
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kotal, M.; Bhowmick, A.K. Polymer nanocomposites from modified clays: Recent advances and challenges. Progr. Polym. Sci. 2015, 51, 127–187. [Google Scholar] [CrossRef]
- Wu, S.; Peng, S.; Wang, C.H. Multifunctional Polymer Nanocomposites Reinforced by Aligned Carbon Nanomaterials. Polymers 2018, 10, 542. [Google Scholar] [CrossRef]
- Quaresimin, M.; Schulte, K.; Zappalorto, M.; Chandrasekaran, S. Toughening mechanisms in polymer nanocomposites: From experiments to modelling. Compos. Sci. Technol. 2016, 123, 187–204. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Luceño Sánchez, J.A.; Peña Capilla, R.; García Díaz, P. Recent Developments in Graphene/Polymer Nanocomposites for Application in Polymer Solar Cells. Polymers 2018, 10, 217. [Google Scholar] [CrossRef]
- Nikolic, M.S.; Petrovic, R.; Veljovic, D.; Cosovic, V.; Stankovic, N.; Djonlagic, J. Effect of sepiolite organomodification on the performance of PCL/sepiolite nanocomposites. Eur. Polym. J. 2017, 97, 198–209. [Google Scholar] [CrossRef]
- Abraham, T.N.; Ratna, D.; Siengchin, S.; Karger-Kocsis, J. Rheological and thermal properties of Poly(ethylene oxide)/multiwall carbon nanotube composites. J. Appl. Polym. Sci. 2008, 110, 2094–2101. [Google Scholar] [CrossRef]
- Dintcheva, N.T.; Morici, E.; Arrigo, R.; La Mantia, F.P. Interaction in POSS-poly(ethylene-co-acrylic acid) nanocomposites. Polym. J. 2014, 46, 160–166. [Google Scholar] [CrossRef]
- La Mantia, F.P.; Morreale, M. Improving the properties of polypropylene-wood flour composites by utilization of maleated adhesion promoters. Compos. Interfaces 2007, 14, 685–698. [Google Scholar] [CrossRef]
- Kim, S.Y.; Meyer, H.W.; Saalwachter, K.; Zukoski, C.F. Polymer dynamics in PEG-silica mamocomposites: Effects of polymer molecular weight, temperature and solven dilution. Macromolecules 2012, 45, 4225–4237. [Google Scholar] [CrossRef]
- Cassagnau, P. Melt rheology of organoclay and fumed silica nanocomposites. Polymer 2008, 49, 2183–2196. [Google Scholar] [CrossRef]
- Wang, X.; Gao, Y.; Mao, K.; Xue, G.; Chen, T.; Zhu, J.; Li, B.; Sun, P.; Jin, Q.; Ding, D. Unusual rheological behavior of liquid polybutadiene rubber/clay mamocomposite gels: The role of polymer-clay interactions, clay exfoliation, and clay orientation and disorientation. Macromolecules 2006, 39, 6653–6660. [Google Scholar] [CrossRef]
- Walha, F.; Lamnawar, K.; Maazouz, A.; Jaziri, M. Rheological, Morphological and Mechanical Studies of Sustainably Sourced Polymer Blends Based on Poly(Lactic Acid) and Polyamide 11. Polymers 2016, 8, 61. [Google Scholar] [CrossRef]
- Pötschke, P.; Fornes, T.D.; Paul, D.R. Rheological behavior of multiwalled carbon nanotube/polycarbonate composites. Polymer 2002, 43, 3247–3255. [Google Scholar] [CrossRef]
- Chatterjee, T.; Krishnamoorti, R. Rheology of polymer carbon nanotubes composites. Soft Matter 2013, 9, 9515–9529. [Google Scholar] [CrossRef] [PubMed]
- Filippone, G.; Salzano de Luna, M. A unifying approach for the linear viscoelasticity of polymer nanocomposites. Macromolecules 2012, 45, 8853–8860. [Google Scholar] [CrossRef]
- Zakiyan, S.E.; Azizi, H.; Ghasemi, I. Influence of chain mobility on rheological, dielectric and electromagnetic interference shielding properties of poly methyl-methacrylate composites filled with graphene and carbon nanotube. Comp. Sci. Technol. 2017, 142, 10–19. [Google Scholar] [CrossRef]
- Pan, Y.; Li, L. Percolation and gel-like behavior of multiwalled carbon nanotube/polypropylene composites influenced by nanotube aspect ratio. Polymer 2013, 54, 1218–1226. [Google Scholar] [CrossRef]
- Galgali, G.; Ramesh, C.; Lele, A. A Rheological Study on the Kinetics of Hybrid Formation in Polypropylene Nanocomposites. Macromolecules 2001, 34, 852–858. [Google Scholar] [CrossRef]
- Lee, K.M.; Han, C.D. Viscoelastic Properties of Functionalized Block Copolymer/Organoclay Nanocomposites. Macromolecules 2003, 36, 804–815. [Google Scholar] [CrossRef]
- Dorigato, A.; Pegoretti, A.; Penati, A. Linear low-density polyethylene/silica micro- and nanocomposites: Dynamic rheological measurements and modelling. Express Polym. Lett. 2010, 4, 115–129. [Google Scholar] [CrossRef]
- Rohlmann, C.O.; Failla, M.D.; Quinzani, L.M. Linear viscoelasticity and structure of polypropylene-montmorillonite nanocomposites. Polymer 2006, 47, 7795–7804. [Google Scholar] [CrossRef]
- Liu, K.; Ronca, S.; Andablo-Reyes, E.; Forte, G.; Rastogi, S. Unique Rheological Response of Ultrahigh Molecular Weight Polyethylenes in the Presence of Reduced Graphene Oxide. Macromolecules 2015, 48, 131–139. [Google Scholar] [CrossRef]
- Gentile, G.; Ambrogi, V.; Cerruti, P.; Di Maio, R.; Nasti, G.; Carfagna, C. Pros and cons of melt annealing on the properties of MWCNT/polypropylene composites. Polym. Degrad. Stabil. 2014, 110, 56–64. [Google Scholar] [CrossRef]
- Shen, Z.; Simon, G.P.; Cheng, Y.B. Comparison of solution intercalation and melt intercalation of polymer–clay nanocomposites. Polymer 2002, 43, 4251–4260. [Google Scholar] [CrossRef]
- Kelarakis, A.; Giannelis, E.P. Crystallization and unusual rheological behavior in poly(ethylene oxide)-clay nanocomposites. Polymer 2011, 52, 2221–2227. [Google Scholar] [CrossRef]
- Kelarakis, A.; Krysmann, M.J.; Giannelis, E.P. Thermoreversible gelation in poly(ethylene oxide)/carbon black hybrid melts. Polymer 2014, 55, 6278–6281. [Google Scholar] [CrossRef]
- Arrigo, R.; Ronchetti, S.; Montanaro, L.; Malucelli, G. Effects of the nanofiller size and aspect ratio on the thermal and rheological behavior of PEG nanocomposites containing boehmites or hydrotalcites. J. Therm. Anal. Calorim. 2018. [Google Scholar] [CrossRef]
- Gianotti, V.; Antonioli, D.; Sparnacci, K.; Laus, M.; Giammaria, T.J.; Ceresoli, M.; Ferrarese Lupi, F.; Seguini, G.; Perego, M. Characterization of ultra-thin polymeric films by Gas chromatography-Mass spectrometry hyphenated to thermogravimetry. J. Chromatogr. A 2014, 1368, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, C.; Wu, D.; Xie, W.; Wang, K.; Xia, Q.; Yang, H. Rheology of the cellulose nanocrystals filled poly(ε-caprolactone) biocomposites. Polymer 2018, 140, 167–178. [Google Scholar] [CrossRef]
- Rizvi, A.; Park, C.B.; Favis, B.D. Tuning viscoelastic and crystallization properties of polypropylene containing in-situ generated high aspect ratio polyethylene terephthalate fibrils. Polymer 2015, 68, 83–91. [Google Scholar] [CrossRef]
- Vivek, R.; Joseph, K.; Simon, G.P.; Bhattacharyya, A.R. Melt-mixed composites of multi-walled carbon nanotubes and thermotropic liquid crystalline polymer: Morphology, rheology and mechanical properties. Comp. Sci. Technol. 2017, 151, 184–192. [Google Scholar] [CrossRef]
- Kashi, S.; Gupta, R.K.; Baum, T.; Kao, N.; Bhattacharya, S.N. Phase transition and anomalous rheological behavior of polylactide/graphene nanocomposites. Compos. Part B Eng. 2018, 135, 25–34. [Google Scholar] [CrossRef]
- Kashi, S.; Gupta, R.K.; Kao, N.; Bhattacharya, S.N. Viscoelastic properties and physical gelation of poly (butylene adipate-co-terephtalate)/graphene nanoplatelet nanocomposites at elevated temperatures. Polymer 2016, 101, 347–357. [Google Scholar] [CrossRef]
- Kelarakis, A.; Yoon, K.; Somani, R.H.; Chen, X.; Hsiao, B.S.; Chu, B. Rheological study of carbon nanofiber induced physical gelation in polyolefin nanocomposite melt. Polymer 2005, 46, 11591–11599. [Google Scholar] [CrossRef]
- Mansky, P.; Liu, Y.; Huang, E.; Russell, T.P.; Hawker, C. Controlling Polymer-Surface Interactions with Random Copolymer Brushes. Science 1997, 275, 1458–1460. [Google Scholar] [CrossRef]
- Ferrarese Lupi, F.; Giammaria, T.J.; Seguini, G.; Ceresoli, M.; Perego, M.; Antonioli, D.; Gianotti, V.; Sparnacci, K.; Laus, M. Flash Grafting of Functional Random Copolymers for Surface Neutralization. J. Mater. Chem. C 2014, 2, 4909–4917. [Google Scholar] [CrossRef]
- Sparnacci, K.; Antonioli, D.; Gianotti, V.; Laus, M.; Ferrarese Lupi, F.; Giammaria, T.J.; Seguini, G.; Perego, M. Ultra-thin Random Copolymer Grafted Layers for Block Copolymer Self- Assembly. ACS Appl. Mater. Interfaces 2015, 7, 10944–10951. [Google Scholar] [CrossRef] [PubMed]
- Sparnacci, K.; Antonioli, D.; Perego, M.; Giammaria, T.J.; Ferrarese Lupi, F.; Seguini, G.; Zuccheri, G.; Gianotti, V.; Laus, M. High temperature surface neutralization process with random copolymers for block copolymer self-assembly. Polym. Int. 2017, 66, 459–467. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arrigo, R.; Antonioli, D.; Lazzari, M.; Gianotti, V.; Laus, M.; Montanaro, L.; Malucelli, G. Relaxation Dynamics in Polyethylene Glycol/Modified Hydrotalcite Nanocomposites. Polymers 2018, 10, 1182. https://doi.org/10.3390/polym10111182
Arrigo R, Antonioli D, Lazzari M, Gianotti V, Laus M, Montanaro L, Malucelli G. Relaxation Dynamics in Polyethylene Glycol/Modified Hydrotalcite Nanocomposites. Polymers. 2018; 10(11):1182. https://doi.org/10.3390/polym10111182
Chicago/Turabian StyleArrigo, Rossella, Diego Antonioli, Massimo Lazzari, Valentina Gianotti, Michele Laus, Laura Montanaro, and Giulio Malucelli. 2018. "Relaxation Dynamics in Polyethylene Glycol/Modified Hydrotalcite Nanocomposites" Polymers 10, no. 11: 1182. https://doi.org/10.3390/polym10111182
APA StyleArrigo, R., Antonioli, D., Lazzari, M., Gianotti, V., Laus, M., Montanaro, L., & Malucelli, G. (2018). Relaxation Dynamics in Polyethylene Glycol/Modified Hydrotalcite Nanocomposites. Polymers, 10(11), 1182. https://doi.org/10.3390/polym10111182









