Design and Optimization of a Hyper-Branched Polyimide Proton Exchange Membrane with Ultra-High Methanol-Permeation Resistivity for Direct Methanol Fuel Cells Applications
Abstract
1. Introduction
2. Experimental
2.1. Materials
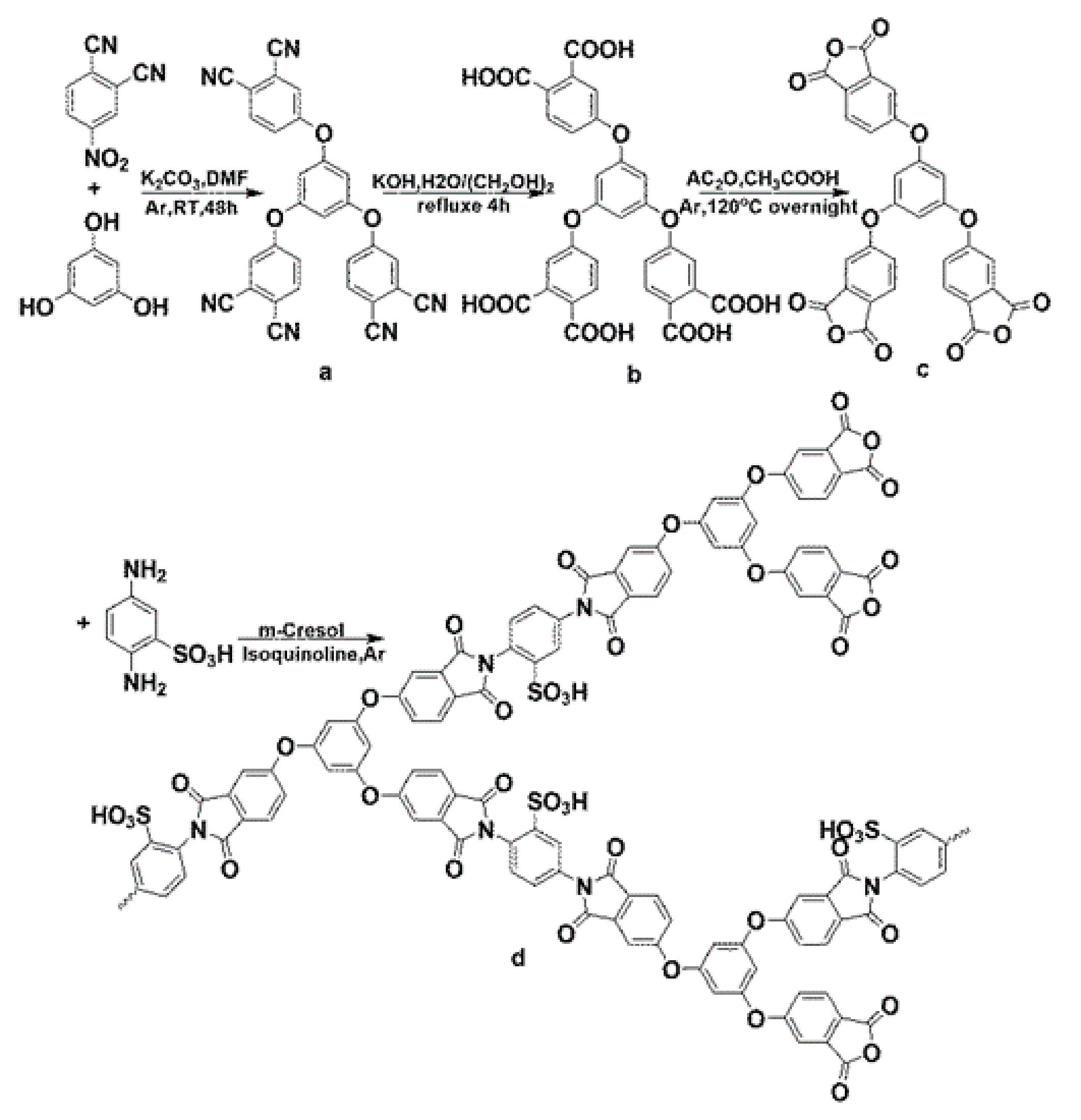
2.2. Synthesis
2.3. Membrane Preparation
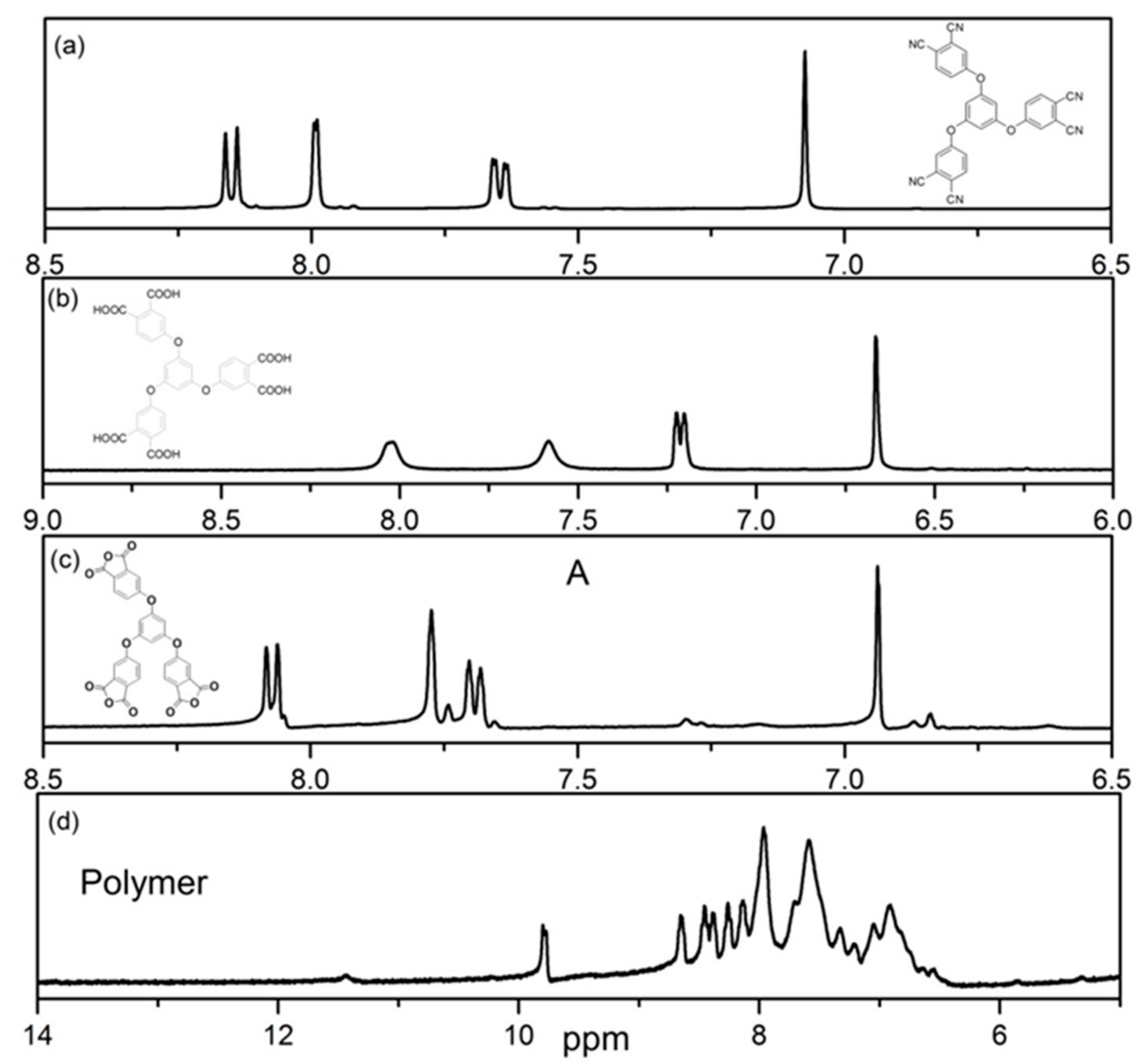
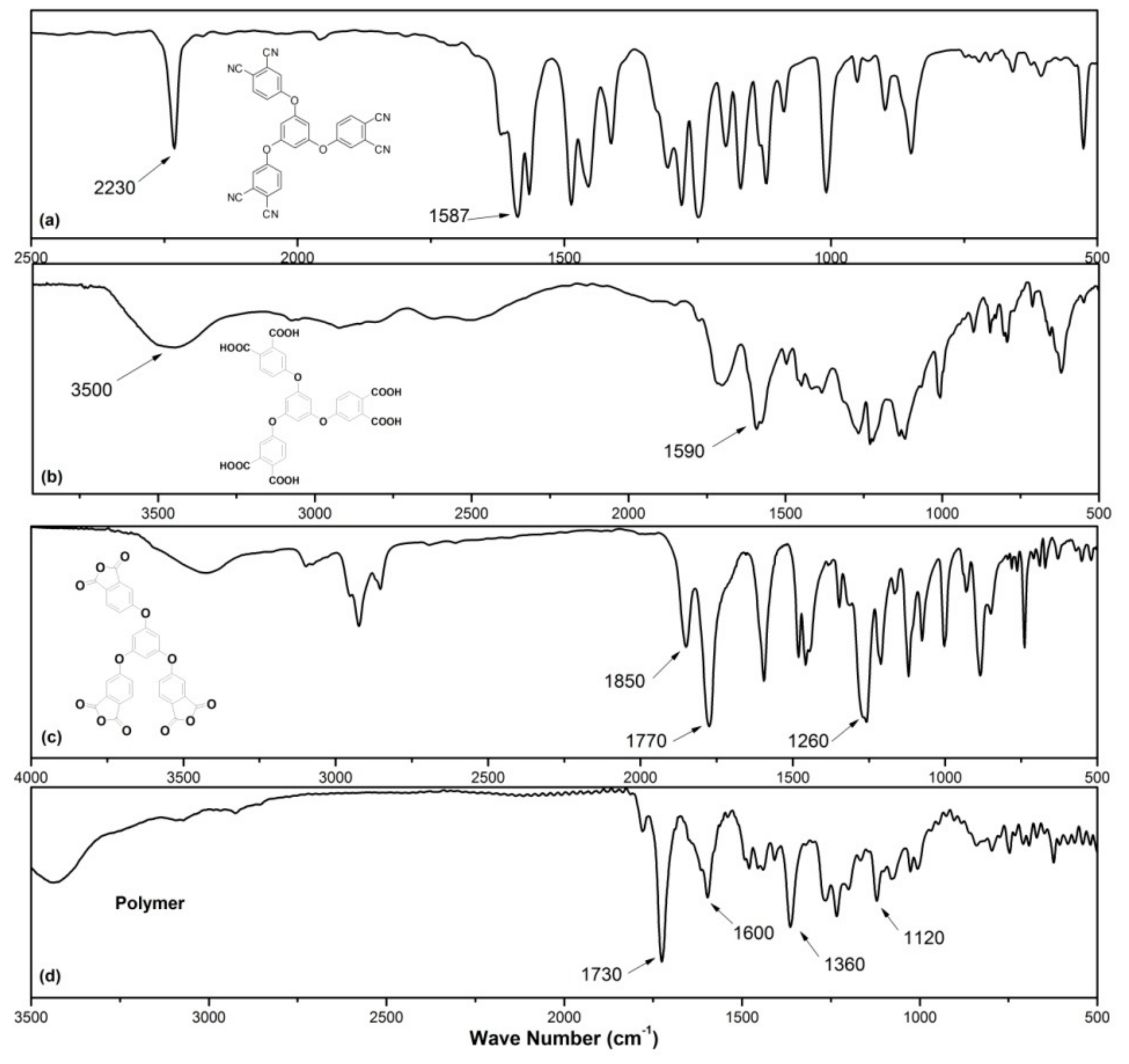
2.4. Measurements and Characterizations
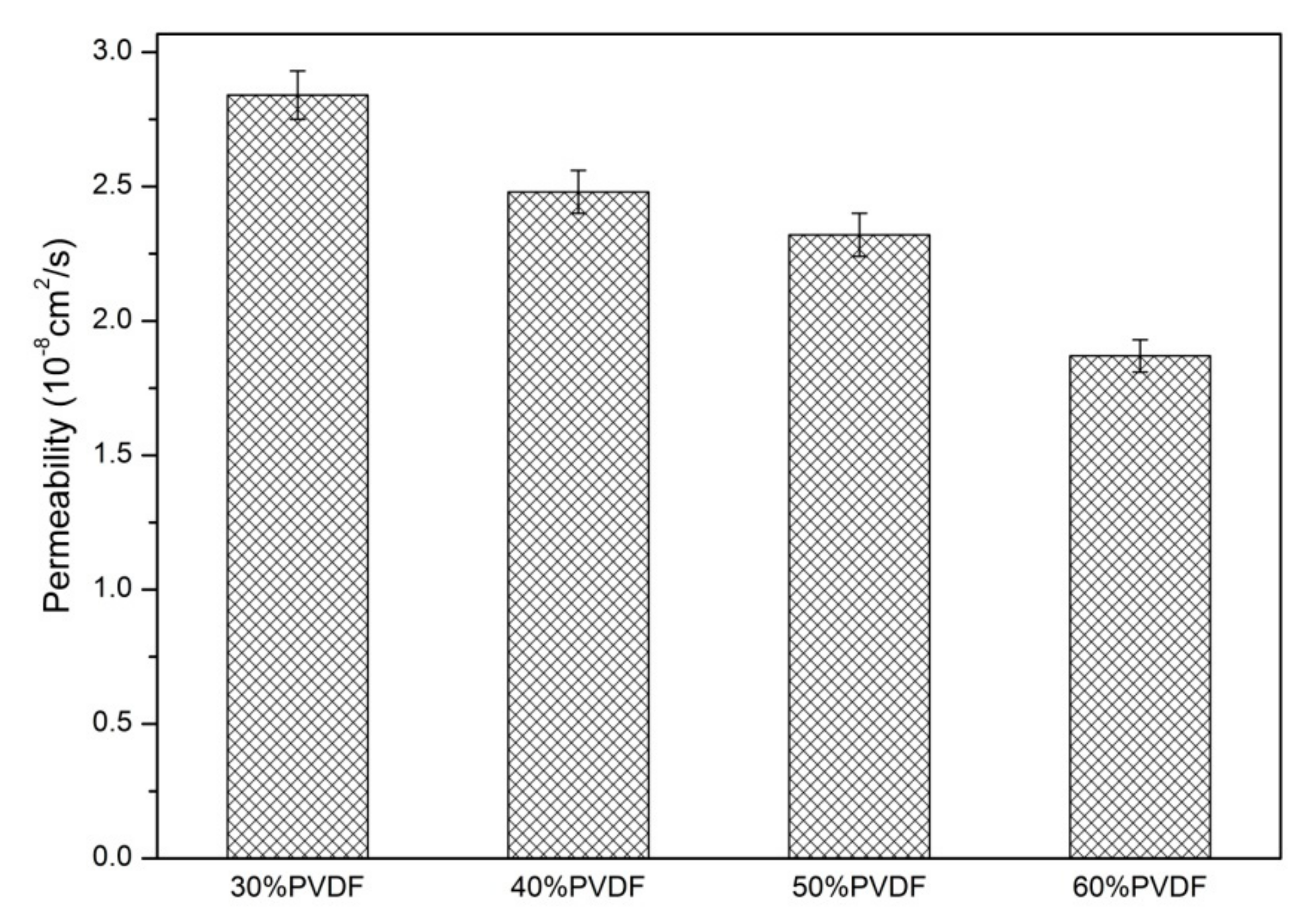
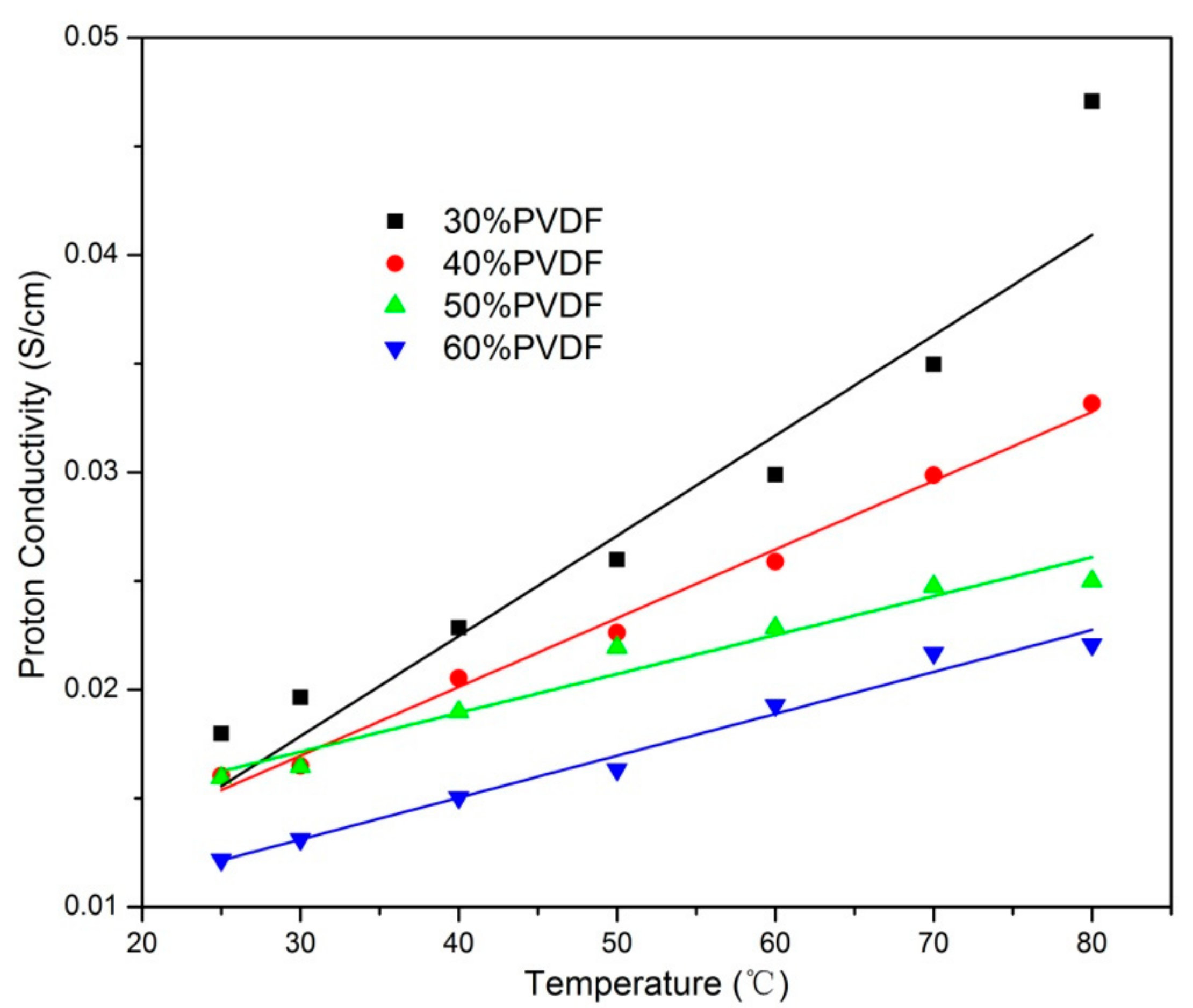
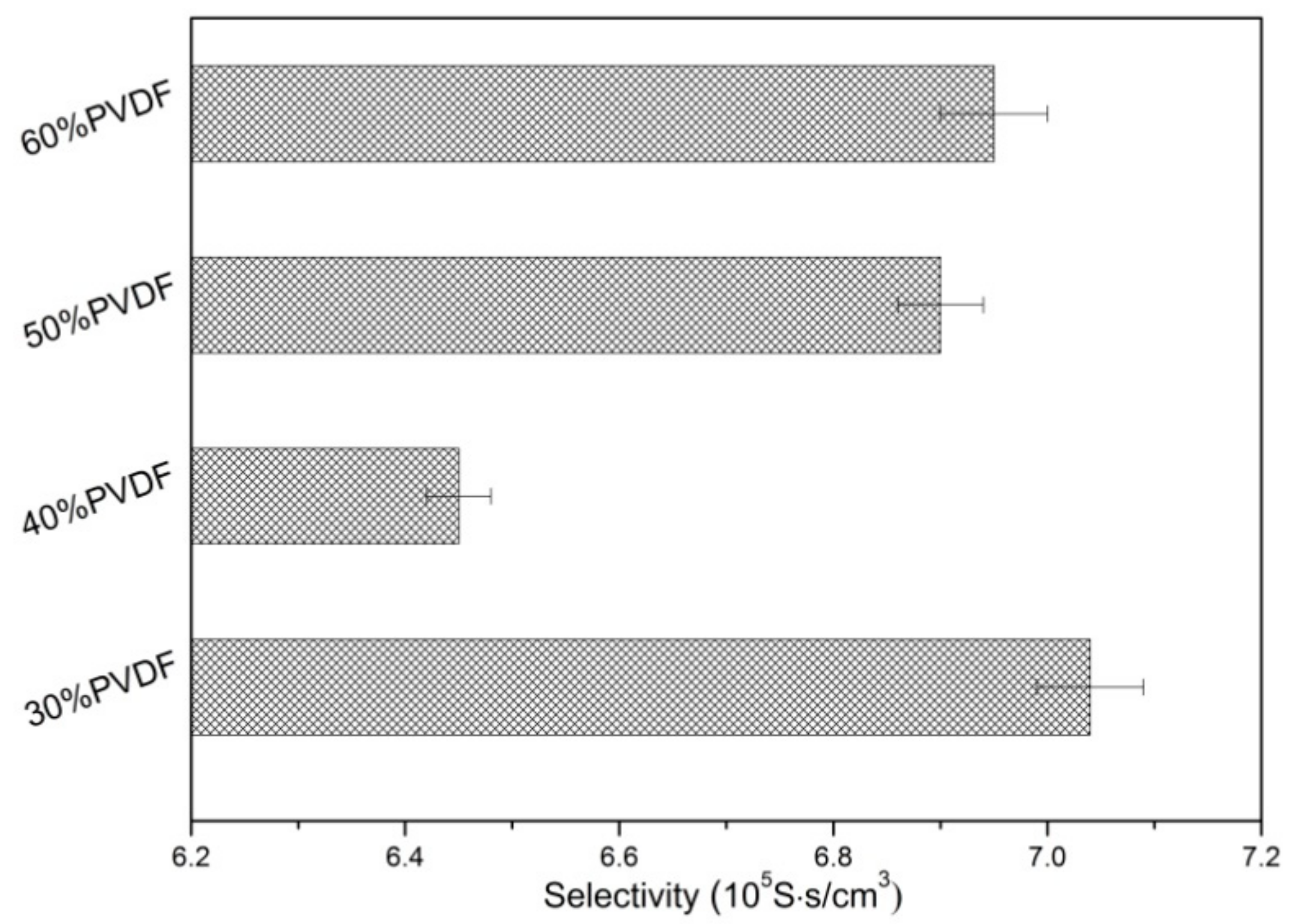
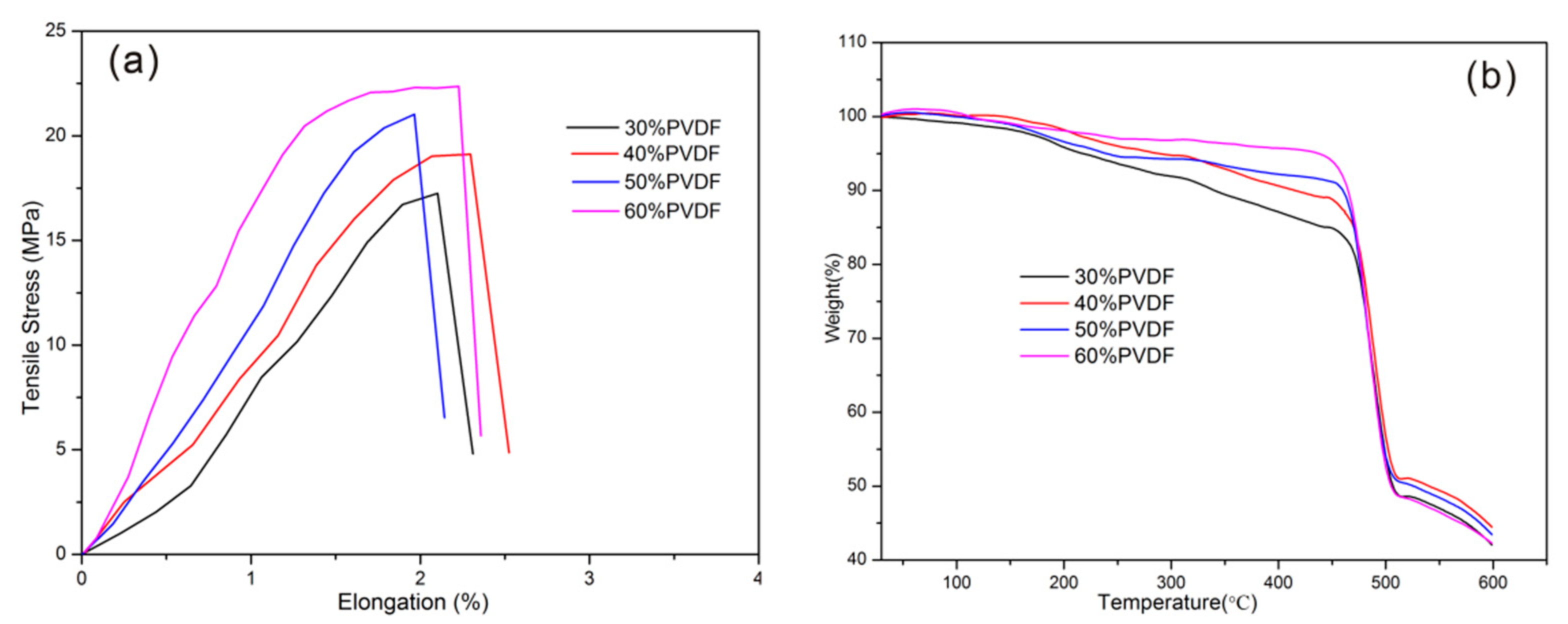
3. Results and Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Winter, M.; Brodd, R.J. What Are Batteries, Fuel Cells, and Supercapacitors? Chem. Rev. 2014, 104, 4245–4270. [Google Scholar] [CrossRef]
- Yandrasits, M.A.; Hamrock, S.J. Membranes for PEM Fuel Cells: 3M Research Activites. In Fuel Cell Chemistry and Opration; Yandrasits, M.A., Zawodzinski, T.A., Jr., Hamrock, S.J., Eds.; ACS Publications: Washington, DC, USA, 2010; pp. 15–29. [Google Scholar]
- Gubler, L. Polymer Design Strategies for Radiation-Grafted Fuel Cell Membranes. Adv. Energy Mater. 2014, 4, 1300827. [Google Scholar] [CrossRef]
- Zhang, H.; Shen, P.K. Recent Development of Polymer Electrolyte Membranes for Fuel Cells. Chem. Rev. 2012, 112, 2780–2832. [Google Scholar] [CrossRef] [PubMed]
- Vasileiadis, S.; Ziaka-Vasileiadou, Z. Efficient catalytic reactors-processors for fuel cells and synthesis applications. Sep. Purif. Technol. 2004, 34, 213–225. [Google Scholar] [CrossRef]
- Vasileiadis, S.; Ziaka, Z. Small Scale Reforming Separation Systems with Nanomembrane Reactors for Direct Fuel Cell Applications. J. Nano Res. 2010, 12, 105–113. [Google Scholar] [CrossRef]
- Zhou, X.; Weston, J.; Chalkova, E.; Hofmann, M.A.; Ambler, C.M.; Allcock, H.R.; Lvov, S.N. High temperature transport properties of polyphosphazene membranes for direct methanol fuel cells. Electrochim. Acta 2003, 48, 2173–2180. [Google Scholar] [CrossRef]
- Chen, Z.; Holmberg, B.; Li, W.; Wang, X.; Deng, W.; Munoz, R.; Yan, Y. Nafion/Zeolite Nanocomposite Membrane by in Situ Crystallization for a Direct Methanol Fuel Cell. Chem. Mater. 2006, 18, 5669–5675. [Google Scholar] [CrossRef]
- Kreuer, K.-D. Proton Conductivity: Materials and Applications. Chem. Mater. 1996, 8, 610–641. [Google Scholar] [CrossRef]
- Heinzel, A.; Barragán, V.M. A review of the state-of-the-art of the methanol crossover in direct methanol fuel cells. J. Power Sources 1999, 84, 70–74. [Google Scholar] [CrossRef]
- Savadogo, O. Emerging membranes for electrochemical systems: Part II. High temperature composite membranes for polymer electrolyte fuel cell (PEFC) applications. J. Power Sources 2004, 127, 135–161. [Google Scholar] [CrossRef]
- Li, L.; Deng, B.; Ji, Y.; Yu, Y.; Xie, L.; Li, J.; Lu, X. A novel approach to prepare proton exchange membranes from fluoropolymer powder by pre-irradiation induced graft polymerization. J. Membr. Sci. 2010, 346, 113–120. [Google Scholar] [CrossRef]
- Alkan Gürsel, S.; Gubler, L.; Gupta, B.; Scherer, G.G. Radiation Grafted Membranes. In Fuel Cells I; Scherer, G.G., Ed.; Springer: Berlin, Germany, 2008; pp. 157–217. [Google Scholar]
- Nakabayashi, K.; Matsumoto, K.; Ueda, M. Synthesis and properties of sulfonated multiblock copoly(ether sulfone)s by a chain extender. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 3947–3957. [Google Scholar] [CrossRef]
- Kim, D.S.; Robertson, G.P.; Kim, Y.S.; Guiver, M.D. Copoly(arylene ether)s Containing Pendant Sulfonic Acid Groups as Proton Exchange Membranes † NRCC Publication No. 50899. Macromolecules 2009, 42, 957–963. [Google Scholar] [CrossRef]
- Lee, M.; Park, J.K.; Lee, H.-S.; Lane, O.; Moore, R.B.; McGrath, J.E.; Baird, D.G. Effects of block length and solution-casting conditions on the final morphology and properties of disulfonated poly(arylene ether sulfone) multiblock copolymer films for proton exchange membranes. Polymer 2009, 50, 6129–6138. [Google Scholar] [CrossRef]
- Wang, C.; Shin, D.W.; Lee, S.Y.; Kang, N.R.; Lee, Y.M.; Guiver, M.D. Poly(arylene ether sulfone) proton exchange membranes with flexible acid side chains. J. Membr. Sci. 2012, 405–406, 68–78. [Google Scholar] [CrossRef]
- Devanathan, R. Recent developments in proton exchange membranes for fuel cells. Energy Environ. Sci. 2008, 1, 101–119. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, C.; Xiao, G.; Yan, D. Synthesis and properties of sulfonated poly(arylene ether phosphine oxide)s for proton exchange membranes. J. Power Sources 2009, 188, 57–63. [Google Scholar] [CrossRef]
- Zhang, W.; Gogel, V.; Friedrich, K.A.; Kerres, J. Novel covalently cross-linked poly(etheretherketone) ionomer membranes. J. Power Sources 2006, 155, 3–12. [Google Scholar] [CrossRef]
- Ding, J.; Chuy, C.; Holdcroft, S. Enhanced Conductivity in Morphologically Controlled Proton Exchange Membranes: Synthesis of Macromonomers by SFRP and Their Incorporation into Graft Polymers. Macromolecules 2002, 35, 1348–1355. [Google Scholar] [CrossRef]
- Chikashige, Y.; Chikyu, Y.; Miyatake, K.; Watanabe, M. Poly(arylene ether) Ionomers Containing Sulfofluorenyl Groups for Fuel Cell Applications. Macromolecules 2005, 38, 7121–7126. [Google Scholar] [CrossRef]
- Bai, Z.; Dang, T.D. Direct Synthesis of Fully Sulfonated Polyarylenethioether Sulfones as Proton-Conducting Polymers for Fuel Cells. Macromol. Rapid Commun. 2006, 27, 1271–1277. [Google Scholar] [CrossRef]
- Staiti, P.; Lufrano, F.; Aricò, A.S.; Passalacqua, E.; Antonucci, V. Sulfonated polybenzimidazole membranes—Preparation and physico-chemical characterization. J. Membr. Sci. 2001, 188, 71–78. [Google Scholar] [CrossRef]
- Genova-Dimitrova, P.; Baradie, B.; Foscallo, D.; Poinsignon, C.; Sanchez, J.Y. Ionomeric membranes for proton exchange membrane fuel cell (PEMFC): Pulfonated polysulfone associated with phosphatoantimonic acid. J. Membr. Sci. 2001, 185, 59–71. [Google Scholar] [CrossRef]
- Seo, D.W.; Lim, Y.D.; Lee, S.H.; Hossain, M.A.; Islam, M.M.; Lee, H.C.; Jang, H.H.; Kim, W.G. Preparation and characterization of block copolymers containing multi-sulfonated unit for proton exchange membrane fuel cell. Electrochim. Acta 2012, 86, 352–359. [Google Scholar] [CrossRef]
- Seo, D.-W.; Lim, Y.-D.; Lee, S.-H.; Jeong, Y.-G.; Hong, T.-W.; Kim, W.-G. Preparation and characterization of sulfonated amine-poly(ether sulfone)s for proton exchange membrane fuel cell. Int. J. Hydrogen Energy 2010, 35, 13088–13095. [Google Scholar] [CrossRef]
- Jones, D.J.; Rozière, J. Recent advances in the functionalisation of polybenzimidazole and polyetherketone for fuel cell applications. J. Membr. Sci. 2001, 185, 41–58. [Google Scholar] [CrossRef]
- Tan, N.; Chen, Y.; Xiao, G.; Yan, D. Synthesis and properties of sulfonated polybenzothiazoles with benzimidazole moieties as proton exchange membranes. J. Membr. Sci. 2010, 356, 70–77. [Google Scholar] [CrossRef]
- Carbone, A.; Pedicini, R.; Portale, G.; Longo, A.; D’Ilario, L.; Passalacqua, E. Sulphonated poly(ether ether ketone) membranes for fuel cell application: Thermal and structural characterisation. J. Power Sources 2006, 163, 18–26. [Google Scholar] [CrossRef]
- Li, W.; Zhang, F.; Yi, S.; Huang, C.; Zhang, H.; Pan, M. Effects of casting solvent on microstructrue and ionic conductivity of anhydrous sulfonated poly(ether ether ketone)-inoic liquid composite membranes. Int. J. Hydrogen Energy 2012, 37, 748–754. [Google Scholar] [CrossRef]
- Kreuer, K.D. On the development of proton conducting polymer membranes for hydrogen and methanol fuel cells. J. Membr. Sci. 2001, 185, 29–39. [Google Scholar] [CrossRef]
- Ma, L.; Cai, W.; Li, J.; Fan, K.; Jiang, Y.; Ma, L.; Cheng, H. A high performance polyamide-based proton exchange membrane fabricated via construction of hierarchical proton conductive channels. J. Power Sources 2016, 302, 189–194. [Google Scholar] [CrossRef]
- Li, J.; Cai, W.; Ma, L.; Zhang, Y.; Chen, Z.; Cheng, H. Towards neat methanol operation of direct methanol fuel cells: A novel self-assembled proton exchange membrane. Chem. Commun. 2015, 51, 6556–6559. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ting, J.W.Y.; Rohan, R.; Cai, W.; Li, J.; Xu, G.; Chen, Z.; Lin, A.; Cheng, H. Fabrication of a proton exchange membrane via blended sulfonimide functionalized polyamide. J. Mater. Sci. 2014, 49, 3442–3450. [Google Scholar] [CrossRef]
- Yu, M.; Wang, W.; Li, C.; Zhai, T.; Lu, X.; Tong, Y. Scalable self-growth of Ni@NiO core-shell electrode with ultrahigh capacitance and super-long cyclic stability for supercapacitors. NPG Asia Mater. 2014, 6, e129. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, C.; Liu, X.; Yang, Z.; Dong, J.; Liu, Y.; Cai, W.; Cheng, H. Fabrication of a polymer electrolyte membrane with uneven side chains for enhancing proton conductivity. RSC Adv. 2016, 6, 79593–79601. [Google Scholar] [CrossRef]
- Pang, J.; Zhang, H.; Li, X.; Liu, B.; Jiang, Z. Poly(arylene ether)s with pendant sulfoalkoxy groups prepared by direct copolymerization method for proton exchange membranes. J. Power Sources 2008, 184, 1–8. [Google Scholar] [CrossRef]
- Xie, H.; Tao, D.; Xiang, X.; Ou, Y.; Bai, X.; Wang, L. Synthesis and properties of highly branched star-shaped sulfonated block poly(arylene ether)s as proton exchange membranes. J. Membr. Sci. 2015, 473, 226–236. [Google Scholar] [CrossRef]
- Miyatake, K.; Zhou, H.; Watanabe, M. Proton Conductive Polyimide Electrolytes Containing Fluorenyl Groups: Synthesis, Properties, and Branching Effect. Macromolecules 2004, 37, 4956–4960. [Google Scholar] [CrossRef]
- Yan, J.; Liu, C.; Wang, Z.; Xing, W.; Ding, M. Water resistant sulfonated polyimides based on 4,4′-binaphthyl-1,1′,8,8′-tetracarboxylic dianhydride (BNTDA) for proton exchange membranes. Polymer 2007, 48, 6210–6214. [Google Scholar] [CrossRef]
- Wang, Y.; Xing, C.; Guan, J.; Li, Y. Towards Flexible Dielectric Materials with High Dielectric Constant and Low Loss: PVDF Nanocomposites with both Homogenously Dispersed CNTs and Ionic Liquids Nanodomains. Polymers 2017, 9, 562. [Google Scholar] [CrossRef]
- Rana, D.; Mandal, B.M.; Bhattacharyya, S.N. Analogue Calorimetric Studies of Blends of Poly(vinyl ester)s and Polyacrylates. Macromolecules 1996, 29, 1579–1583. [Google Scholar] [CrossRef]
- Rana, D.; Mandal, B.M.; Bhattacharyya, S.N. Analogue calorimetry of polymer blends: Poly(styrene-co-acrylonitrile) and poly(phenyl acrylate) or poly(vinyl benzoate). Polymer 1996, 37, 2439–2443. [Google Scholar] [CrossRef]
- Rana, D.; Mandal, B.M.; Bhattacharyya, S.N. Miscibility and phase diagrams of poly(phenyl acrylate) and poly(styrene-co-acrylonitrile) blends. Polymer 1993, 34, 1454–1459. [Google Scholar] [CrossRef]
- Lin, S.-C.; Burks, S.J. Compatible Polyvinylidene Fluoride Blends with Polymers Containing Imide Moieties. U.S. Patent 5959022A, 28 September 1999. [Google Scholar]
- Zhang, Y.; Cai, W.; Si, F.; Ge, J.; Liang, L.; Liu, C.; Xing, W. A modified Nafion membrane with extremely low methanol permeability via surface coating of sulfonated organic silica. Chem. Commun. 2012, 48, 2870–2872. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, L.E.; Wesslén, B.; Jannasch, P. Water absorption and proton conductivity of sulfonated acrylamide copolymers. Electrochim. Acta 2002, 47, 3269–3275. [Google Scholar] [CrossRef]
- Li, Q.; He, R.; Jensen, J.O.; Bjerrum, N.J. Approaches and recent development of polymer electrolyte membranes for fuel cells operating above 100 C. Chem. Mater. 2003, 15, 4896–4915. [Google Scholar] [CrossRef]
- Ansari, Y.; Tucker, T.G.; Huang, W.; Klein, I.S.; Lee, S.-Y.; Yarger, J.L.; Angell, C.A. A flexible all-inorganic fuel cell membrane with conductivity above Nafion, and durable operation at 150 °C. J. Power Sources 2016, 303, 142–149. [Google Scholar] [CrossRef]
- Rana, D.; Sauvant, V.; Halary, J.L. Molecular analysis of yielding in pure and antiplasticized epoxy-amine thermosets. J. Mater. Sci. 2002, 37, 5267–5274. [Google Scholar] [CrossRef]
- Rana, D.; Lee, C.H.; Cho, K.; Lee, B.H.; Choe, S. Thermal and mechanical properties for binary blends of metallocene polyethylene with conventional polyolefins. J. Appl. Polym. Sci. 1998, 69, 2441–2450. [Google Scholar] [CrossRef]
- Rana, D.; Cho, K.; Woo, T.; Lee, B.H.; Choe, S. Blends of ethylene 1-octene copolymer synthesized by Ziegler–Natta and metallocene catalysts. I. Thermal and mechanical properties. J. Appl. Polym. Sci. 1999, 74, 1169–1177. [Google Scholar] [CrossRef]
- Sasikala, S.; Meenakshi, S.; Bhat, S.D.; Sahu, A.K. Functionalized Bentonite clay-sPEEK based composite membranes for direct methanol fuel cells. Electrochim. Acta 2014, 135, 232–241. [Google Scholar] [CrossRef]

| Membranes | 30% PVDF | 40% PVDF | 50% PVDF | 60% PVDF | Nafion 117 |
|---|---|---|---|---|---|
| Water uptake (%) | 25.79 ± 0.03 | 17.87 ± 0.04 | 14.11 ± 0.04 | 9.05 ± 0.03 | 20.97 ± 0.05 |
| Volume swelling (%) | 26.95 ± 0.05 | 25.84 ± 0.06 | 24.73 ± 0.04 | 14.87 ± 0.04 | 36.29 ± 0.05 |
| IEC (mmol/g) | 0.6212 ± 0.03 | 0.5931 ± 0.02 | 0.5719 ± 0.03 | 0.3540 ± 0.04 | 0.91 ± 0.05 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, L.; Xu, G.; Li, S.; Ma, J.; Li, J.; Cai, W. Design and Optimization of a Hyper-Branched Polyimide Proton Exchange Membrane with Ultra-High Methanol-Permeation Resistivity for Direct Methanol Fuel Cells Applications. Polymers 2018, 10, 1175. https://doi.org/10.3390/polym10101175
Ma L, Xu G, Li S, Ma J, Li J, Cai W. Design and Optimization of a Hyper-Branched Polyimide Proton Exchange Membrane with Ultra-High Methanol-Permeation Resistivity for Direct Methanol Fuel Cells Applications. Polymers. 2018; 10(10):1175. https://doi.org/10.3390/polym10101175
Chicago/Turabian StyleMa, Liying, Guoxiao Xu, Shuai Li, Jiao Ma, Jing Li, and Weiwei Cai. 2018. "Design and Optimization of a Hyper-Branched Polyimide Proton Exchange Membrane with Ultra-High Methanol-Permeation Resistivity for Direct Methanol Fuel Cells Applications" Polymers 10, no. 10: 1175. https://doi.org/10.3390/polym10101175
APA StyleMa, L., Xu, G., Li, S., Ma, J., Li, J., & Cai, W. (2018). Design and Optimization of a Hyper-Branched Polyimide Proton Exchange Membrane with Ultra-High Methanol-Permeation Resistivity for Direct Methanol Fuel Cells Applications. Polymers, 10(10), 1175. https://doi.org/10.3390/polym10101175




