Two Tetranuclear Butterfly-Shaped Co(II) Complexes: Structure, Mass Spectrometric, and Magnetism
Abstract
1. Introduction
2. Results and Discussion
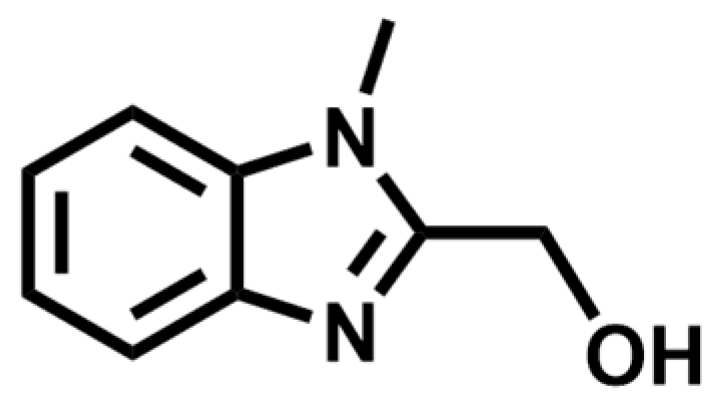
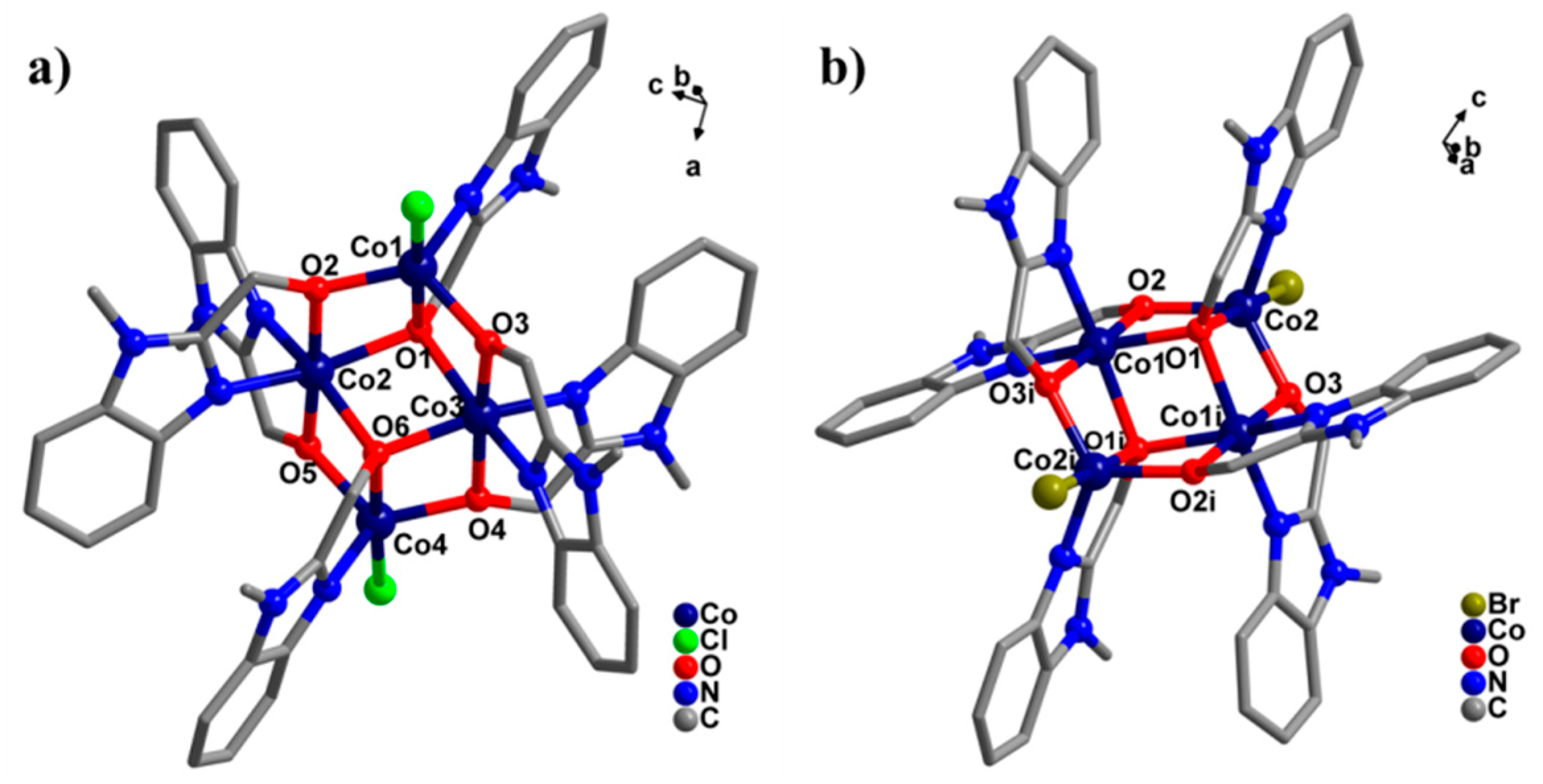
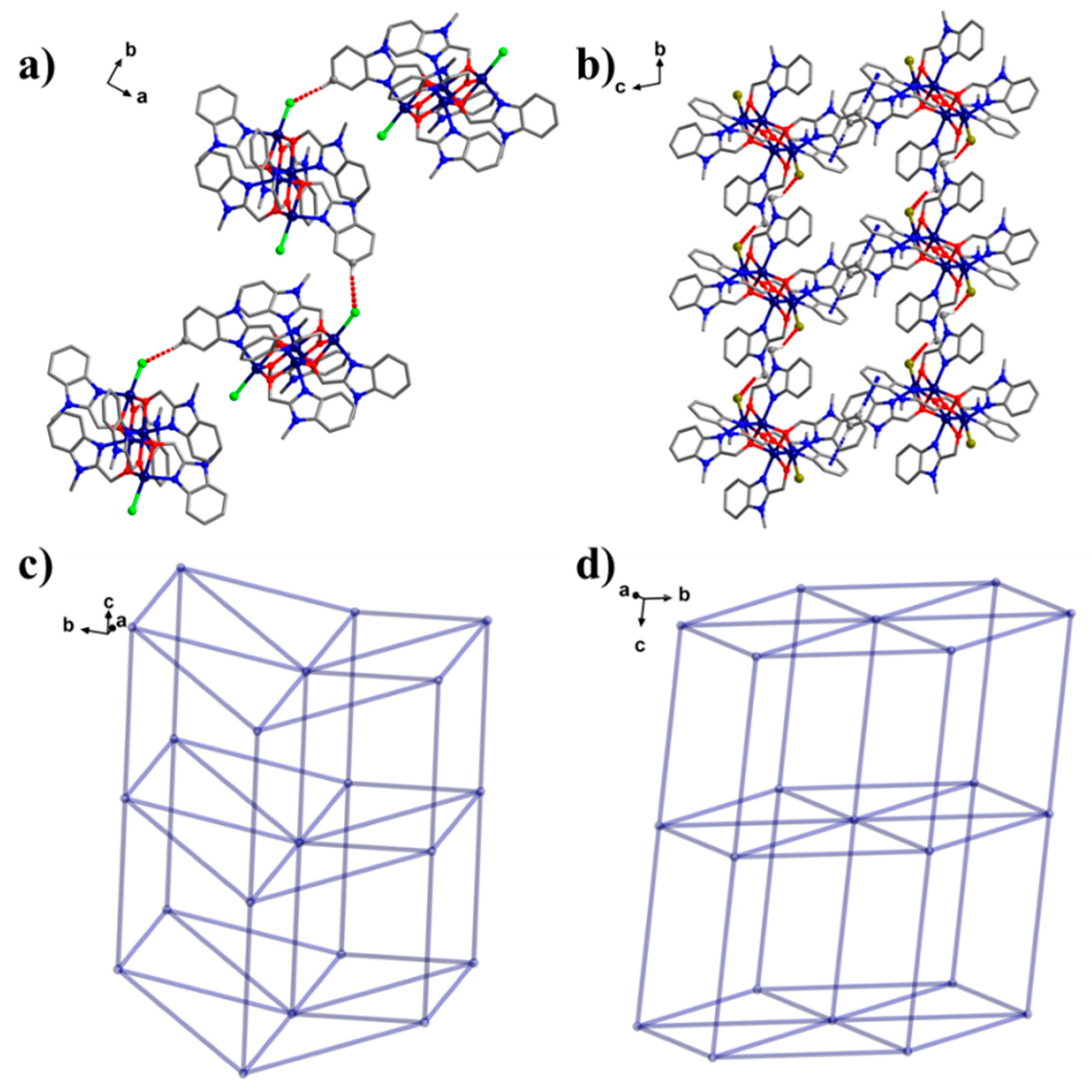
2.1. Crystal Structure
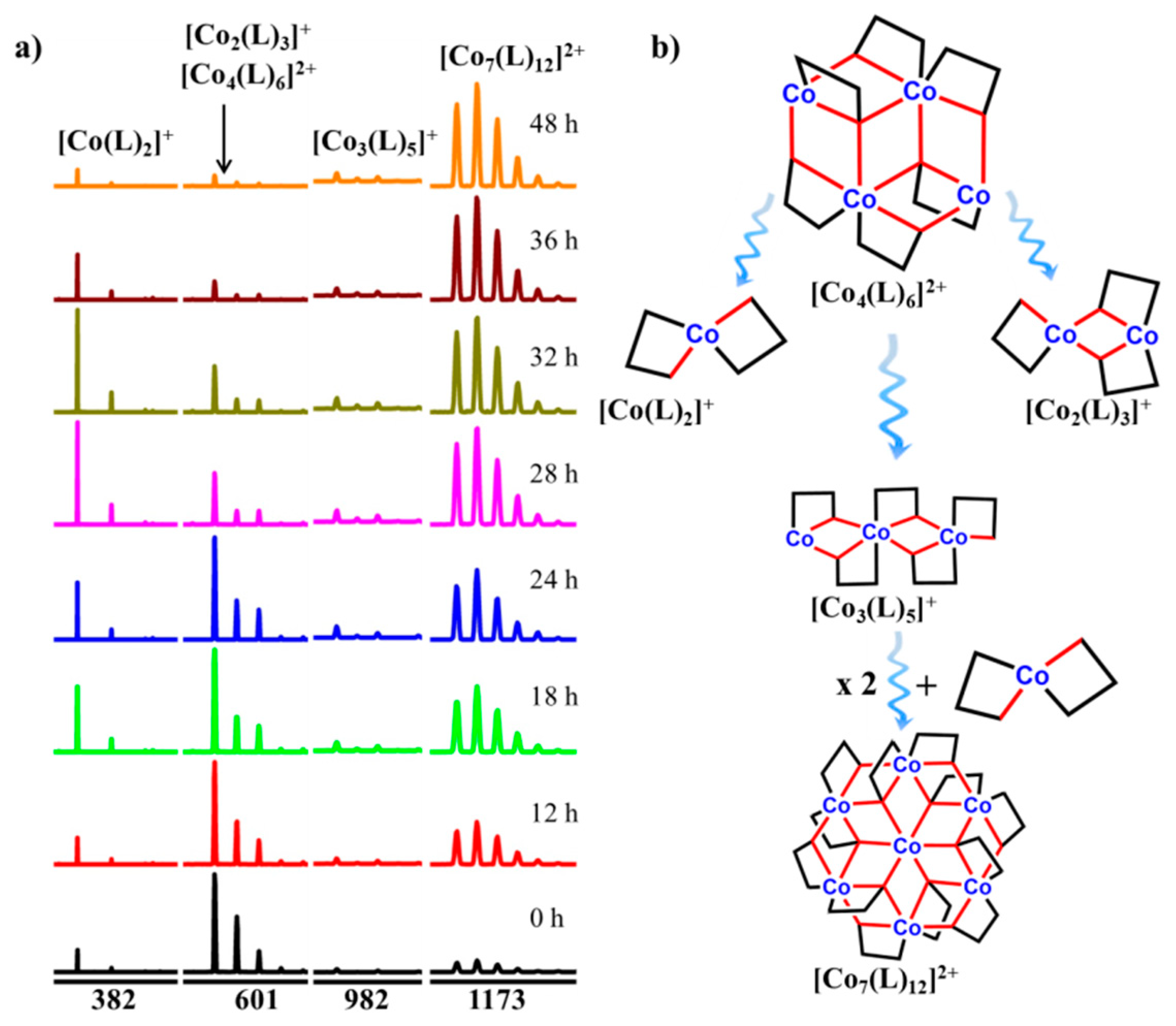
2.2. Electrospray Ionization Mass Spectrometry
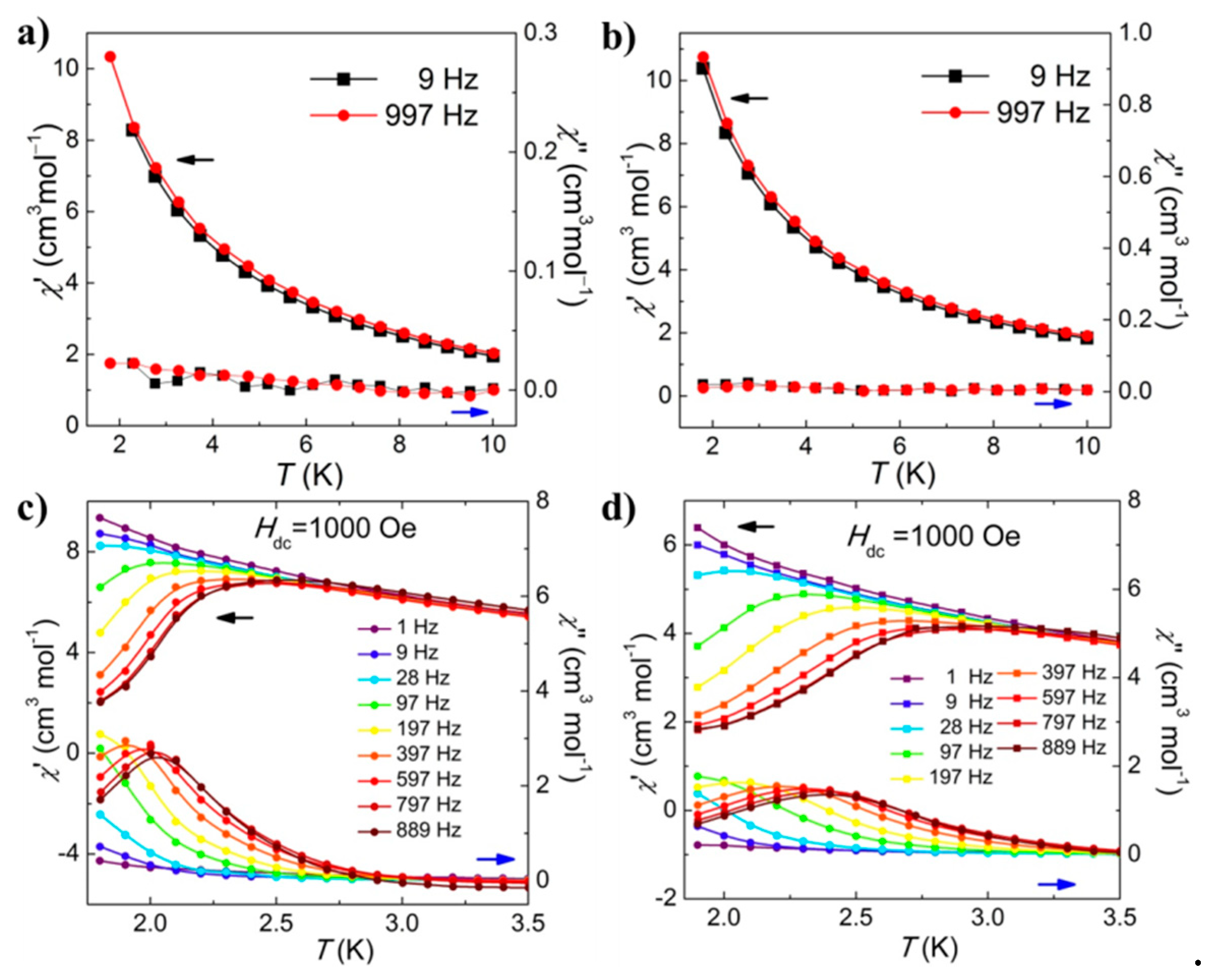
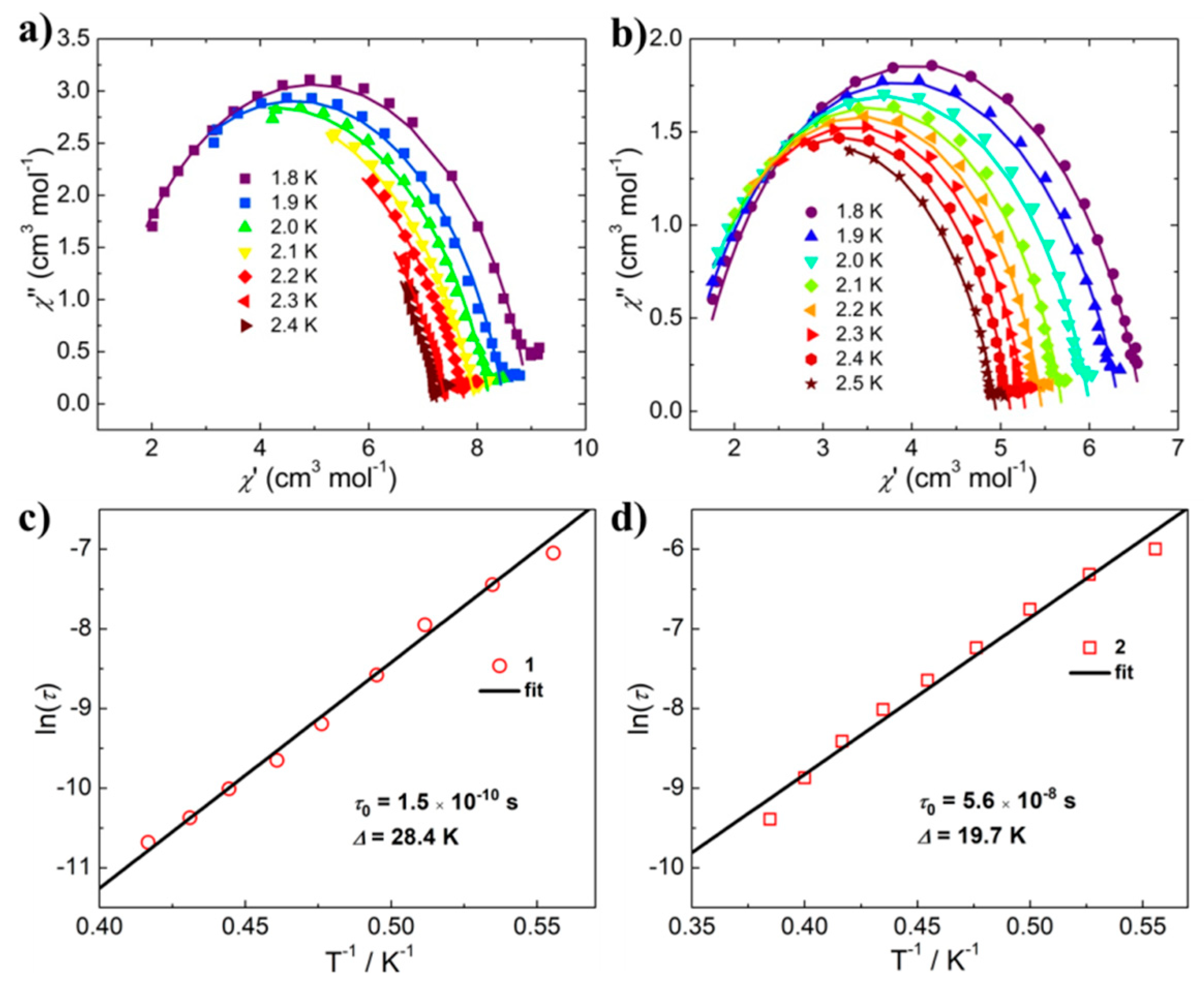
2.3. Magnetic Properties.
3. Conclusions
4. Experimental Section
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sessoli, R.; Gatteschi, D.; Caneschi, A.; Novak, M.A. Magnetic bistability in a metal-ion cluster. Nature. 1993, 365, 141–143. [Google Scholar] [CrossRef]
- Murrie, M. Cobalt(II) single-molecule magnets. Chem. Soc. Rev. 2010, 39, 1986–1995. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Guo, Y.N.; Tang, J. Recent advances in dysprosium-based single molecule magnets: Structural overview and synthetic strategies. Coord. Chem. Rev. 2013, 257, 1728–1763. [Google Scholar] [CrossRef]
- Zhu, Z.H.; Ma, X.F.; Wang, H.L.; Zou, H.H.; Mo, K.Q.; Zhang, Y.Q.; Yang, Q.Z.; Li, B.; Liang, F.P. A triangular Dy3 single-molecule toroic with high inversion energy barrier: magnetic properties and multiple-step assembly mechanism. Inorg. Chem. Front. 2018, 5, 3155–3162. [Google Scholar] [CrossRef]
- Wang, H.L.; Ma, X.F.; Peng, J.M.; Zhu, Z.H.; Li, B.; Zou, H.H.; Liang, F.P. Tracking the Stepwise Formation of the Dysprosium Cluster (Dy10) with Multiple Relaxation Behavior. Inorg. Chem. 2019, 58, 9169–9174. [Google Scholar] [CrossRef]
- Ma, X.F.; Wang, H.L.; Zhu, Z.H.; Li, B.; Mo, K.Q.; Zou, H.H.; Liang, F.P. Formation of nanocluster {Dy12} containing Dy-exclusive vertex-sharing [Dy4(μ3-OH)4] cubanes via simultaneous multitemplate guided and step-by-step assembly. Dalt. Trans. 2019, 48, 11338–11344. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.L.; Peng, J.M.; Zhu, Z.H.; Mo, K.Q.; Ma, X.F.; Li, B.; Zou, H.H.; Liang, F.P. Step-by-Step and Competitive Assembly of Two Dy(III) Single-Molecule Magnets with Their Performance Tuned by Schiff Base Ligands. Cryst. Growth Des. 2019, 19, 5369–5375. [Google Scholar] [CrossRef]
- Gao, S. Molecular Nanomagnets and Related Phenomena; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Christou, G. Single-molecule magnets: a molecular approach to nanoscale magnetic materials. Polyhedron 2005, 24, 2065–2075. [Google Scholar] [CrossRef]
- Wang, H.L.; Ma, X.F.; Zou, H.H.; Wang, K.; Li, B.; Chen, Z.L.; Liang, F.P. Mixed chelating ligands used to regulate the luminescence of Ln(iii) complexes and single-ion magnet behavior in Dy-based analogues. Dalton. Trans. 2018, 47, 15929–15940. [Google Scholar] [CrossRef]
- Fortier, S.; Le Roy, J.J.; Chen, C.H.; Vieru, V.; Murugesu, M.; Chibotaru, L.F.; Mindiola, D.J.; Caulton, K.G. A Dinuclear Cobalt Complex Featuring Unprecedented Anodic and Cathodic Redox Switches for Single-Molecule Magnet Activity. J. Am. Chem. Soc. 2013, 135, 14670–14678. [Google Scholar] [CrossRef]
- Miyasaka, H.; Yamashita, M. A look at molecular nanosized magnets from the aspect of inter-molecular interactions. Dalton Trans. 2007, 399–406. [Google Scholar] [CrossRef]
- Yang, E.C.; Hendrickson, D.N.; Wernsdorfer, W.; Nakano, M.; Zakharov, L.N.; Sommer, R.D.; Rheingold, A.L.; Ledezma-Gairaud, M.; Christou, G. Cobalt single-molecule magnet. J. Appl. Phys. 2002, 91, 7382–7384. [Google Scholar] [CrossRef]
- Kostakis, G.E.; Perlepes, S.P.; Blatov, V.A.; Proserpio, D.M.; Powell, A.K. High-nuclearity cobalt coordination clusters: Synthetic, topological and magnetic aspects. Coord. Chem. Rev. 2012, 256, 1246–1278. [Google Scholar] [CrossRef]
- Klinke, F.J.; Das, A.; Demeshko, S.; Dechert, S.; Meyer, F. Inducing Single Molecule Magnetic Behavior in a [Co4O4] Cubane via a Pronounced Solvatomagnetic Effect. Inorg. Chem. 2014, 53, 2976–2982. [Google Scholar] [CrossRef]
- Rechkemmer, Y.; Breitgoff, F.D.; van der Meer, M.; Atanasov, M.; Hakl, M.; Orlita, M.; Neugebauer, P.; Neese, F.; Sarkar, B.; van Slageren, J. A four-coordinate cobalt (II) single-ion magnet with coercivity and a very high energy barrier. Nat. Commun. 2016, 7, 10467. [Google Scholar] [CrossRef] [PubMed]
- Shankar, K.; Mondal, A.; Li, Y.; Journaux, Y.; Baruah, J.B. Hydroxide-Bridged Mixed-Valence Tetranuclear Cobalt 4-Nitrophenol Inclusion Complex Showing Single Molecule Magnet Property. ChemistrySelect 2017, 2, 7792–7798. [Google Scholar] [CrossRef]
- Plaul, D.; Böhme, M.; Ostrovsky, S.; Tomkowicz, Z.; Görls, H.; Haase, W.; Plass, W. Modeling Spin Interactions in a Triangular Cobalt (II) Complex with Triaminoguanidine Ligand Framework: Synthesis, Structure, and Magnetic Properties. Inorg. Chem. 2018, 57, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Espin, J.; Zarzuela, R.; Statuto, N.; Juanhuix, J.; Maspoch, D.; Imaz, I.; Chudnovsky, E.; Tejada, J. Narrowing the Zero-Field Tunneling Resonance by Decreasing the Crystal Symmetry of Mn12 Acetate. J. Am. Chem. Soc. 2016, 138, 9065–9068. [Google Scholar] [CrossRef]
- Erler, P.; Schmitt, P.; Barth, N.; Irmler, A.; Bouvron, S.; Huhn, T.; Groth, U.; Pauly, F.; Gragnaniello, L.; Fonin, M. Highly Ordered Surface Self-Assembly of Fe4 Single Molecule Magnets. Nano Lett. 2015, 15, 4546–4552. [Google Scholar] [CrossRef]
- Zaleski, C.M.; Depperman, E.C.; Dendrinou-Samara, C.; Alexiou, M.; Kampf, J.W.; Kessissoglou, D.P.; Kirk, M.L.; Pecoraro, V.L. Metallacryptate Metallacryptate single-molecule magnets: Effect of lower molecular symmetry on blocking temperature. J. Am. Chem. Soc. 2005, 127, 12862–12872. [Google Scholar] [CrossRef]
- Kirchon, A.; Feng, L.; Drake, H.F.; Joseph, E.A.; Zhou, H.C. From fundamentals to applications: a toolbox for robust and multifunctional MOF materials. Chem. Soc. Rev. 2018, 47, 8611–8638. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.F.; Guo, L.Y.; Xu, J.H.; Jagodič, M.; Jagličić, Z.; Wang, W.-G.; Zhuang, G.L.; Wang, Z.; Tung, C.H.; Sun, D. Multifaceted Bicubane Co4 Clusters: Magnetism, Photocatalytic Oxygen Evolution, and Electrical Conductivity. Eur. J. Inorg. Chem. 2016, 3253–3261. [Google Scholar] [CrossRef]
- Roth, T.; Morningstar, M.L.; Boyer, P.L.; Hughes, S.H.; Buckheit, R.W., Jr.; Michejda, C.J. Synthesis and biological activity of novel nonnucleoside inhibitors of HIV-1 reverse transcriptase. 2-Aryl-substituted benzimidazoles. J. Med. Chem. 1997, 40, 4199–4207. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELXT: Integrated space-group and crystalstructure Determination. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]

| Complex | 1 | 2 |
|---|---|---|
| Formula | C54H49Cl2Co4N12O6 | C57H66Br2Co4N12O9 |
| Formula weight | 1268.67 | 1456.08 |
| T (K) | 293(2) | 293(2) |
| Crystal system | Monoclinic | Triclinic |
| Space group | P21 | P-1 |
| a (Å) | 12.5470(6) | 11.7411(10) |
| b (Å) | 19.9240(9) | 12.3673(10) |
| c (Å) | 12.6993(5) | 12.7465(8) |
| α (°) | 90.00 | 98.212(8) |
| β (°) | 103.403(4) | 93.842(6) |
| γ (°) | 90.00 | 117.605(9) |
| V (Å3) | 3088.2(2) | 1604.5(3) |
| Z | 2 | 4 |
| Dc (g cm−3) | 1.364 | 1.488 |
| μ (mm−1) | 1.197 | 9.915 |
| F(000) | 1294.0 | 728.0 |
| Reflns coll. | 17333 | 8924 |
| Unique reflns | 10017 | 5199 |
| Rint | 0.0419 | 0.0866 |
| aR1 [I ≥ 2σ(I)] | 0.0875 | 0.1101 |
| bwR2(all data) | 0.2678 | 0.3229 |
| GOF | 1.037 | 1.072 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, Q.-J.; Chen, M.; Chen, D.-C.; Chen, C.-A. Two Tetranuclear Butterfly-Shaped Co(II) Complexes: Structure, Mass Spectrometric, and Magnetism. Crystals 2019, 9, 477. https://doi.org/10.3390/cryst9090477
Deng Q-J, Chen M, Chen D-C, Chen C-A. Two Tetranuclear Butterfly-Shaped Co(II) Complexes: Structure, Mass Spectrometric, and Magnetism. Crystals. 2019; 9(9):477. https://doi.org/10.3390/cryst9090477
Chicago/Turabian StyleDeng, Qian-Jun, Min Chen, Dong-Chu Chen, and Chang-Ai Chen. 2019. "Two Tetranuclear Butterfly-Shaped Co(II) Complexes: Structure, Mass Spectrometric, and Magnetism" Crystals 9, no. 9: 477. https://doi.org/10.3390/cryst9090477
APA StyleDeng, Q.-J., Chen, M., Chen, D.-C., & Chen, C.-A. (2019). Two Tetranuclear Butterfly-Shaped Co(II) Complexes: Structure, Mass Spectrometric, and Magnetism. Crystals, 9(9), 477. https://doi.org/10.3390/cryst9090477




