Epitaxial Order Driven by Surface Corrugation: Quinquephenyl Crystals on a Cu(110)-(2×1)O Surface
Abstract
:1. Introduction
2. Materials and Methods
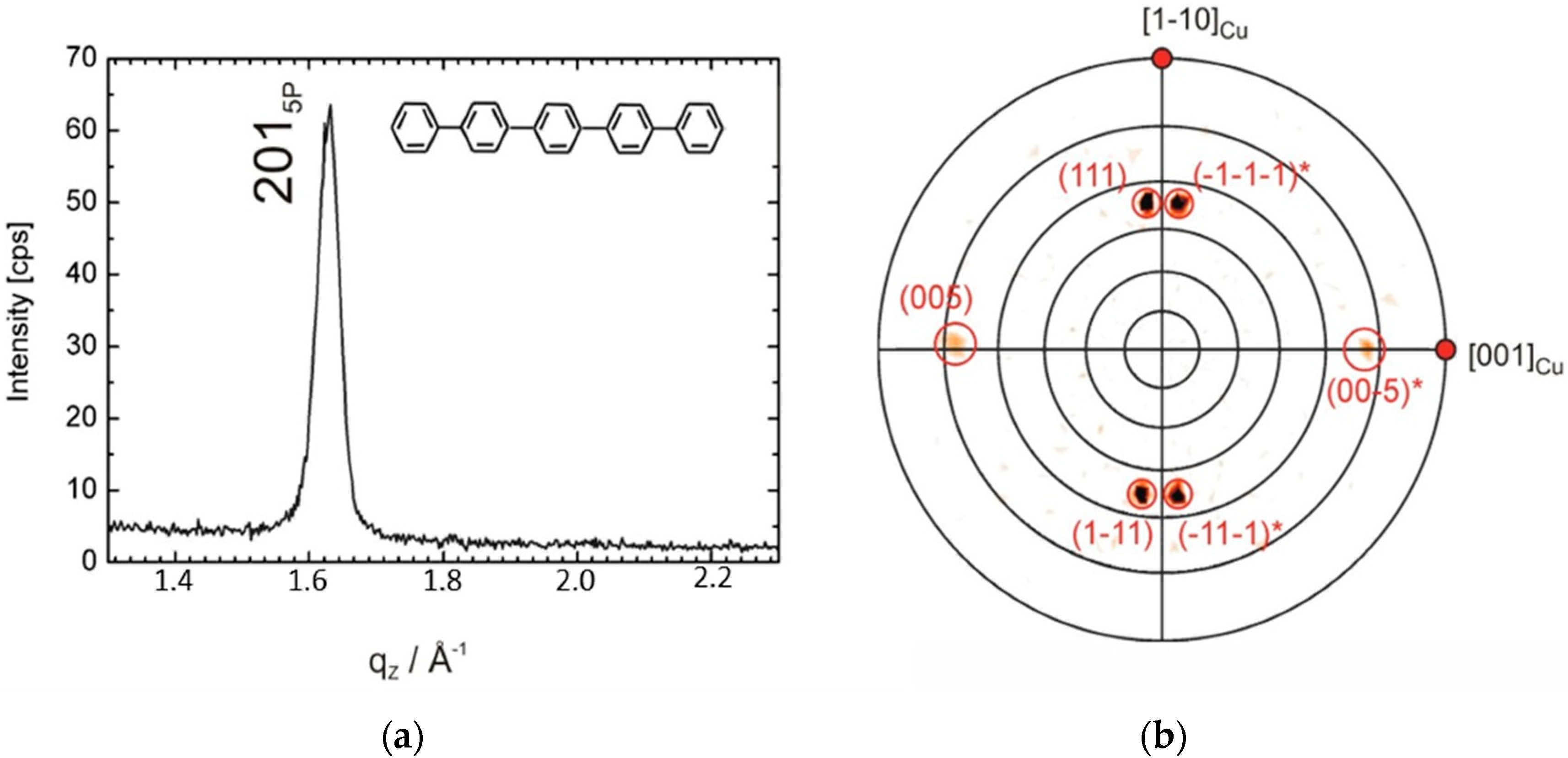
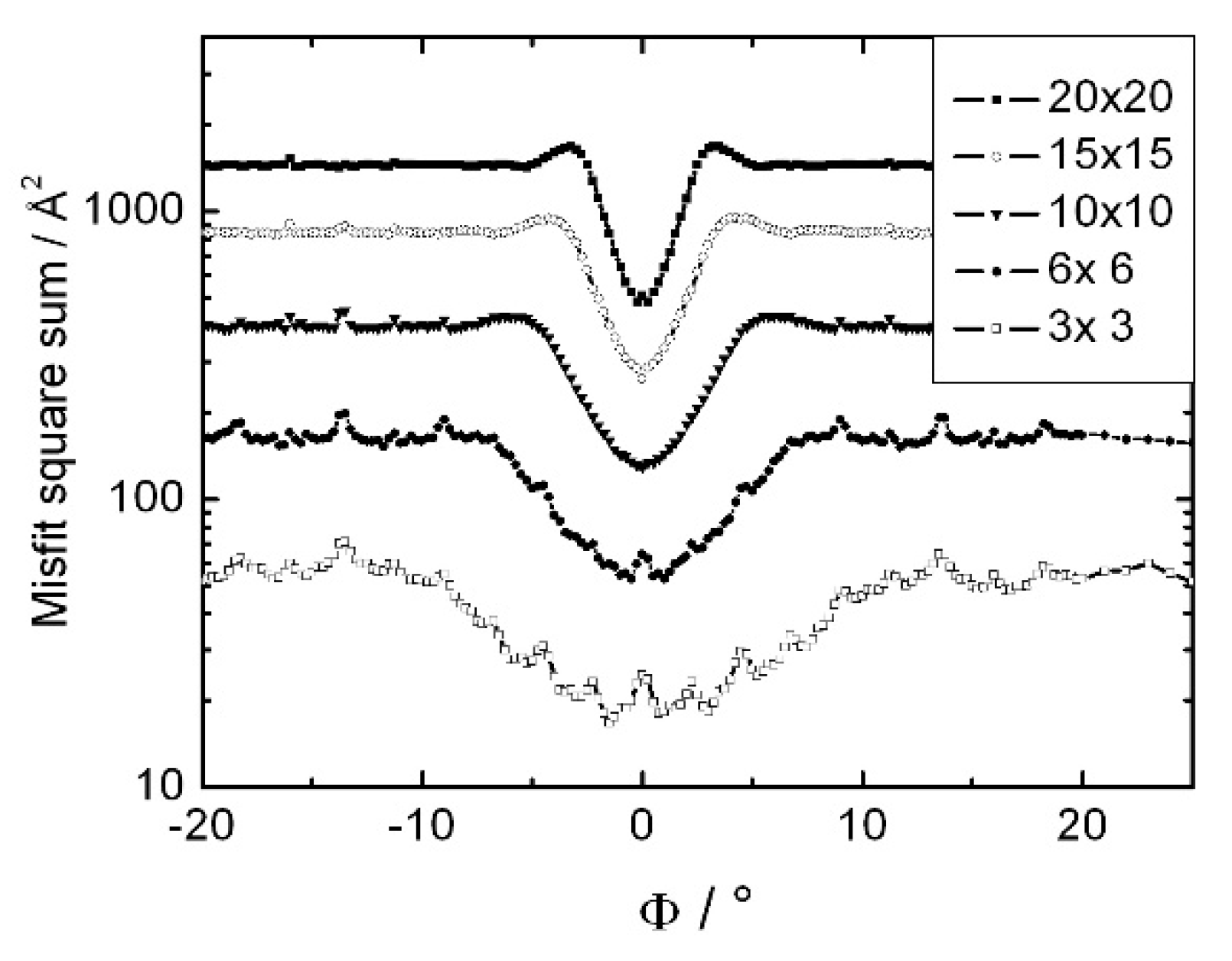
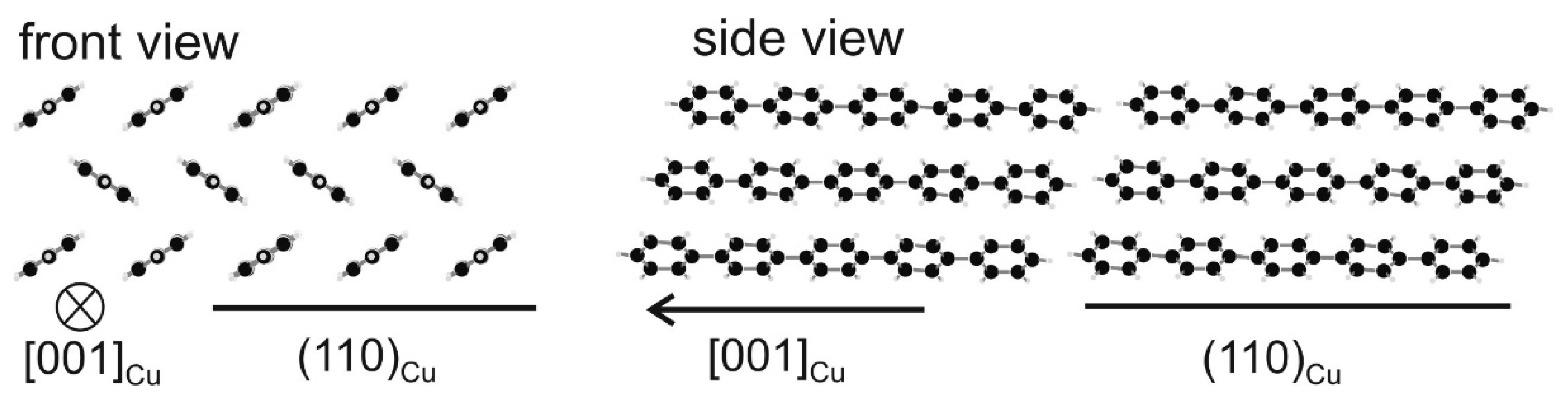
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mitschke, U.; Bäuerle, P. The electroluminescence of organic materials. J. Mater. Chem. 2000, 10, 1471–1507. [Google Scholar] [CrossRef]
- Resel, R. Surface induced crystallographic order in sexiphenyl thin films. J. Phys. Condens. Matter 2008, 20, 184009. [Google Scholar] [CrossRef]
- Forrest, S.R. Ultrathin Organic Films Grown by Organic Molecular Beam Deposition and Related Techniques. Chem. Rev. 1997, 97, 1793–1896. [Google Scholar] [CrossRef] [PubMed]
- Hooks, D.E.; Fritz, T.; Ward, M.D. Epitaxy and Molecular Organization on Solid Substrates. Adv. Mater. 2001, 13, 227–241. [Google Scholar] [CrossRef]
- Mannsfeld, S.C.B.; Leo, K.; Fritz, T. Line-on-Line Coincidence: A New Type of Epitaxy Found in Organic-Organic Heterolayers. Phys. Rev. Lett. 2005, 94, 056104. [Google Scholar] [CrossRef] [PubMed]
- Mannsfeld, S.C.B.; Fritz, T. Understanding organic–inorganic heteroepitaxial growth of molecules on crystalline substrates: Experiment and theory. Phys. Rev. B 2005, 71, 235405. [Google Scholar] [CrossRef]
- Balzer, F.; Rubahn, H.-G. Chain-length dependent para-phenylene film- and needle-growth on dielectrics. Surf. Sci. 2004, 548, 170–182. [Google Scholar] [CrossRef]
- Witte, G.; Wöll, C. Growth of aromatic molecules on solid substrates for applications in organic electronics. J. Mater. Res. 2004, 19, 1889–1916. [Google Scholar] [CrossRef]
- Sassella, A.; Campione, M.; Borghesi, A. Organic epitaxy. Riv. Nuovo Cim. 2008, 31, 457–490. [Google Scholar]
- Müllegger, S.; Salzmann, I.; Resel, R.; Winkler, A. Epitaxial growth of quaterphenyl thin films on gold(111). Appl. Phys. Lett. 2003, 83, 4536–4538. [Google Scholar] [CrossRef]
- Hollerer, M.; Pachmajer, S.; Lüftner, D.; Butej, B.; Reinisch, E.-M.; Puschnig, P.; Koller, G.; Ramsey, M.G.; Sterrer, M. Growth study of para-sexiphenyl on Ag(001): From single molecule to crystalline film. Surf. Sci. 2018, 678, 149–156. [Google Scholar] [CrossRef]
- France, C.B.; Parkinson, B.A. Physical and electronic structure of p-sexiphenyl on Au(111). Appl. Phys. Lett. 2003, 82, 1194. [Google Scholar] [CrossRef]
- Müllegger, S.; Salzmann, I.; Resel, R.; Hlawacek, G.; Teichert, C.; Winkler, A. Growth kinetics, structure, and morphology of para-quaterphenyl thin films on gold(111). J. Chem. Phys. 2004, 121, 2272–2277. [Google Scholar] [CrossRef] [PubMed]
- Müllegger, S.; Winkler, A. Hexaphenyl thin films on clean and carbon covered Au(111) studied with TDS and LEED. Surf. Sci. 2006, 600, 1290–1299. [Google Scholar] [CrossRef]
- Haber, T.; Muellegger, S.; Winkler, A.; Resel, R. Temperature-induced epitaxial growth modes of para-sexiphenyl on Au(111). Phys. Rev. B 2006, 74, 045419. [Google Scholar] [CrossRef]
- Koller, G.; Berkebile, S.; Krenn, J.; Tzvetkov, G.; Hlawacek, G.; Lengyel, O.; Netzer, F.P.; Teichert, C.; Resel, R.; Ramsey, M.G. Oriented Sexiphenyl Single Crystal Nanoneedles on TiO2 (110). Adv. Mater. 2004, 16, 2159–2162. [Google Scholar] [CrossRef]
- Hu, Y.; Maschek, K.; Sun, L.D.; Hohage, M.; Zeppenfeld, P. Para-Sexiphenyl thin film growth on Cu(110) and Cu(110)-(2x1)O surfaces. Surf. Sci. 2006, 600, 762–769. [Google Scholar] [CrossRef]
- Oehzelt, M.; Grill, L.; Berkebile, S.; Koller, G.; Netzer, F.P.; Ramsey, M.G. The molecular orientation of para-sexiphenyl on Cu(110) and Cu(110) p(2×1)O. ChemPhysChem 2007, 8, 1707–1712. [Google Scholar] [CrossRef]
- Koini, M.; Haber, T.; Werzer, O.; Berkebile, S.; Koller, G.; Oehzelt, M.; Ramsey, M.G.; Resel, R. Epitaxial order of pentacene on Cu(110)-(2×1)O: One dimensional alignment induced by surface corrugation. Thin Solid Films 2008, 517, 483–487. [Google Scholar] [CrossRef]
- Koini, M.; Haber, T.; Berkebile, S.; Koller, G.; Ramsey, M.G.; Resel, R.; Oehzelt, M. Growth of sexithiophene crystals on Cu(110) and Cu(110)-(2×1)O stripe phase-The influence of surface corrugation. J. Cryst. Growth 2009, 311, 1364–1369. [Google Scholar] [CrossRef]
- Haber, T.; Resel, R.; Andreev, A.; Oehzelt, M.; Smilgies, D.-M.; Sitter, H. Evolution of epitaxial order in para-sexiphenyl on KCl(100). J. Cryst. Growth 2010, 312, 333–339. [Google Scholar] [CrossRef]
- Smilgies, D.-M.; Boudet, N.; Yanagi, H. In-plane alignment of para-sexiphenyl films grown on KCl(001). Appl. Surf. Sci. 2002, 189, 24–29. [Google Scholar] [CrossRef]
- Koini, M. Surface Corrugations and Its Influence on the Growth of Thin Organic Films. Ph.D. Thesis, Graz University of Technology, Graz, Austria, October 2009. [Google Scholar]
- Zeppenfeld, P.; Diercks, V.; David, R.; Picaud, F.; Ramseyer, C.; Girardet, C. Selective adsorption and structure formation of N2 on the nanostructured Cu-CuO stripe phase. Phys. Rev. B 2002, 66, 085414. [Google Scholar] [CrossRef]
- Birkholz, M. Thin Film Analysis by X-ray Scattering; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Schrode, B.; Pachmajer, S.; Dohr, M.; Röthel, C.; Domke, J.; Fritz, T.; Resel, R.; Werzer, O. GIDVis: A comprehensive software tool for geometry-independent grazing-incidence X-ray diffraction data analysis and pole-figure calculations. J. Appl. Crystallogr. 2019, 52, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Toussaint, C.J. Unit-cell dimensions and space group of some polyphenyls. Acta Crystallogr. 1966, 21, 1002–1003. [Google Scholar] [CrossRef]
- Baker, K.N.; Fratini, A.V.; Resch, T.; Knachel, H.C.; Adams, W.; Socci, E.; Farmer, B. Crystal structures, phase transitions and energy calculations of poly(p-phenylene) oligomers. Polymer 1993, 34, 1571–1587. [Google Scholar] [CrossRef]
- Kraus, W.; Nolzeb, G. POWDER CELL—A program for the representation and manipulation of crystal structures and calculation of the resulting X-ray powder patterns. J. Appl. Crystallogr. 1996, 29, 301–303. [Google Scholar] [CrossRef]
- Salzmann, I.; Resel, R. STEREOPOLE: Software for the analysis of X-ray diffraction pole figures with IDL. J. Appl. Crystallogr. 2004, 37, 1029–1033. [Google Scholar] [CrossRef]
- Hillier, A.C.; Ward, M.D. Epitaxial interactions between molecular overlayers and ordered substrates. Phys. Rev. B 1996, 54, 14037–14051. [Google Scholar] [CrossRef]
- Last, J.A.; Hooks, D.E.; Hillier, A.C.; Ward, M.D. The Physicochemical Origins of Coincident Epitaxy in Molecular Overlayers: Lattice Modeling vs Potential Energy Calculations. J. Phys. Chem. B 1999, 103, 6723–6733. [Google Scholar] [CrossRef]
- Hoshino, A.; Isoda, S.; Kurata, H.; Kobayashi, T. Scanning tunelling microscope contrast of perylene-3,4,9,10-tetracarbboxylic-dianhydride on graphite and its application to the study of epitaxy. J. Appl. Phys. 1994, 76, 4113–4120. [Google Scholar] [CrossRef]
- Novák, J.; Oehzelt, M.; Berkebile, S.; Koini, M.; Ules, T.; Koller, G.; Haber, T.; Resel, R.; Ramsey, M.G. Crystal growth of para-sexiphenyl on clean and oxygen reconstructed Cu(110) surfaces. Phys. Chem. Chem. Phys. 2011, 13, 14675–14684. [Google Scholar] [CrossRef] [PubMed]
- Chua, F.M.; Kuk, Y.; Silverman, P.J. Oxygen chemisorption on Cu(110): An atomic view by scanning tunneling microscopy. Phys. Rev. Lett. 1989, 63, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Herman, M.A.; Richter, W.; Sitter, H. Epitaxy—Physical Principles and Technical Implementation; Springer: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Forker, R.; Meissner, M.; Fritz, T. Classification of epitaxy in reciprocal and real space: Rigid versus flexible lattices. Soft Matter 2017, 13, 1748–1758. [Google Scholar] [CrossRef] [PubMed]
- Resel, R.; Haber, T.; Lengyel, O.; Sitter, H.; Balzer, F.; Rubahn, H.-G. Origins for epitaxial order of sexiphenyl crystals on muscovite(001). Surf. Interface Anal. 2009, 41, 764–770. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Resel, R.; Koini, M.; Novak, J.; Berkebile, S.; Koller, G.; Ramsey, M. Epitaxial Order Driven by Surface Corrugation: Quinquephenyl Crystals on a Cu(110)-(2×1)O Surface. Crystals 2019, 9, 373. https://doi.org/10.3390/cryst9070373
Resel R, Koini M, Novak J, Berkebile S, Koller G, Ramsey M. Epitaxial Order Driven by Surface Corrugation: Quinquephenyl Crystals on a Cu(110)-(2×1)O Surface. Crystals. 2019; 9(7):373. https://doi.org/10.3390/cryst9070373
Chicago/Turabian StyleResel, Roland, Markus Koini, Jiri Novak, Steven Berkebile, Georg Koller, and Michael Ramsey. 2019. "Epitaxial Order Driven by Surface Corrugation: Quinquephenyl Crystals on a Cu(110)-(2×1)O Surface" Crystals 9, no. 7: 373. https://doi.org/10.3390/cryst9070373
APA StyleResel, R., Koini, M., Novak, J., Berkebile, S., Koller, G., & Ramsey, M. (2019). Epitaxial Order Driven by Surface Corrugation: Quinquephenyl Crystals on a Cu(110)-(2×1)O Surface. Crystals, 9(7), 373. https://doi.org/10.3390/cryst9070373




