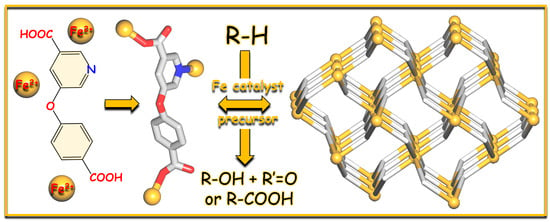
Synthesis, Structural Features, and Catalytic Activity of an Iron(II) 3D Coordination Polymer Driven by an Ether-Bridged Pyridine-Dicarboxylate
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Physical Measurements
2.2. Synthesis of [Fe(µ3-Hcpna)2]n (1)
2.3. X-ray Crystallography
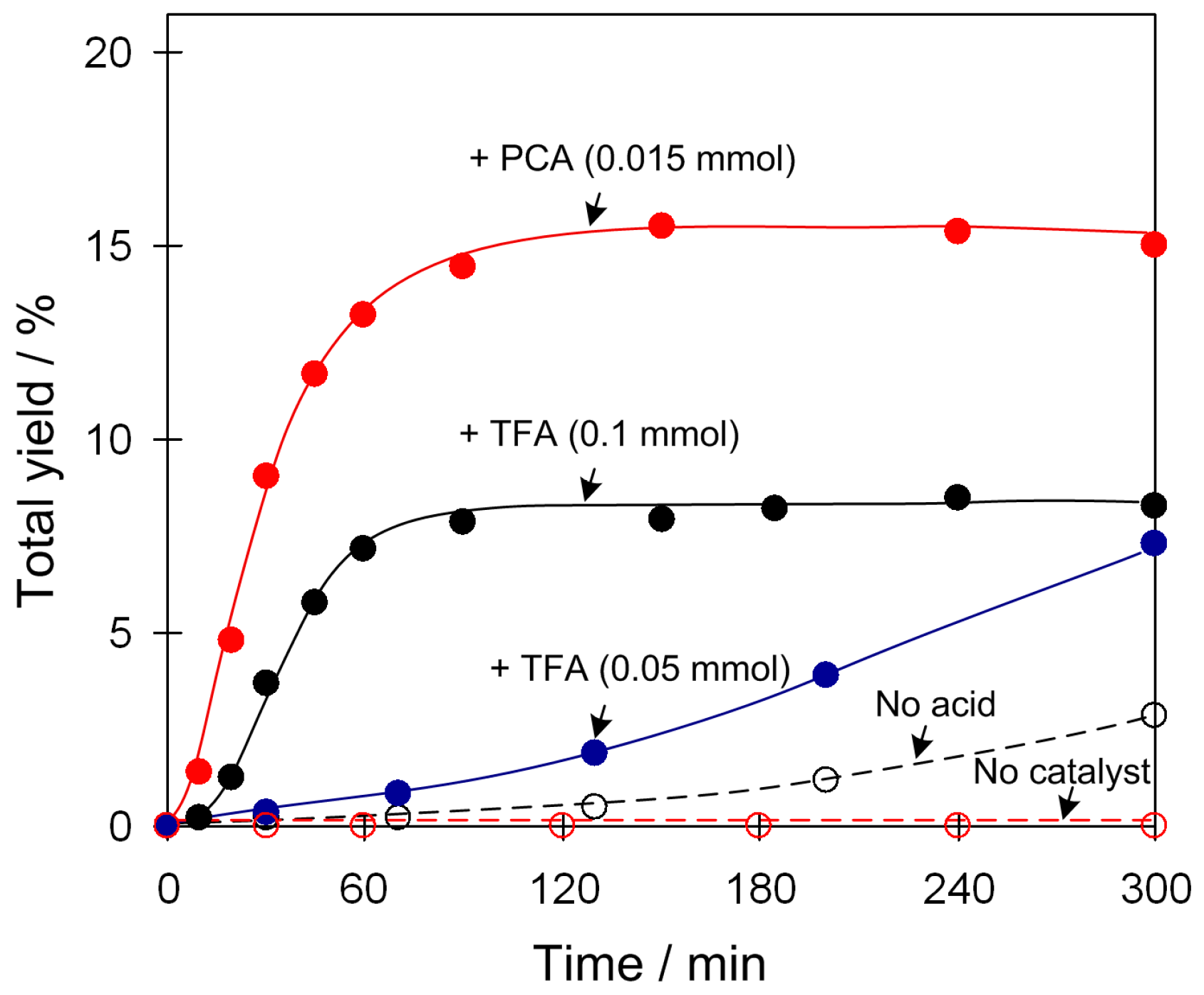
2.4. Catalytic Oxidation of Cyclic Alkanes
2.5. Catalytic Oxidation of Propane
2.6. Catalytic Carboxylation of Alkanes
3. Results and Discussion
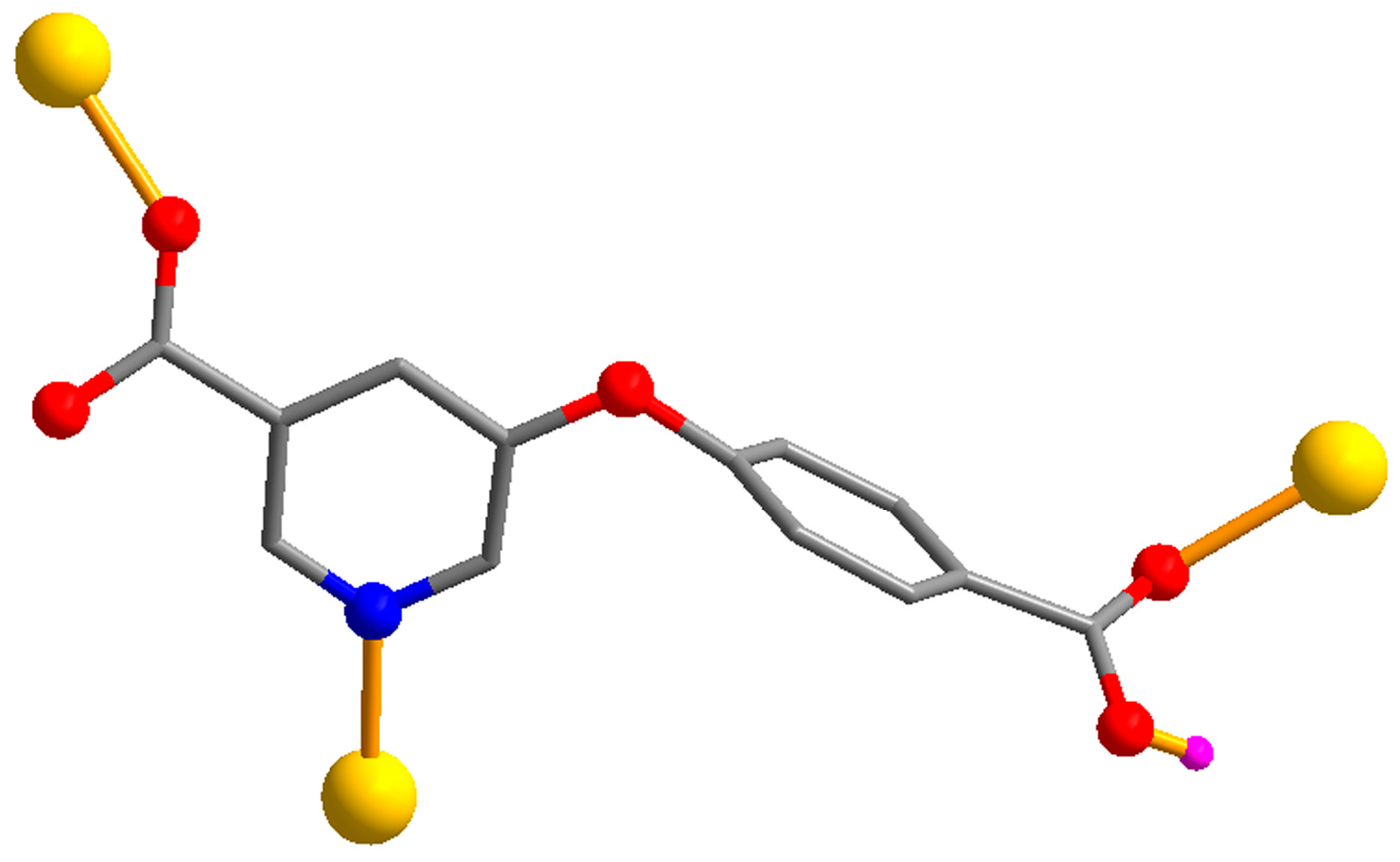
3.1. Synthesis and Characterization
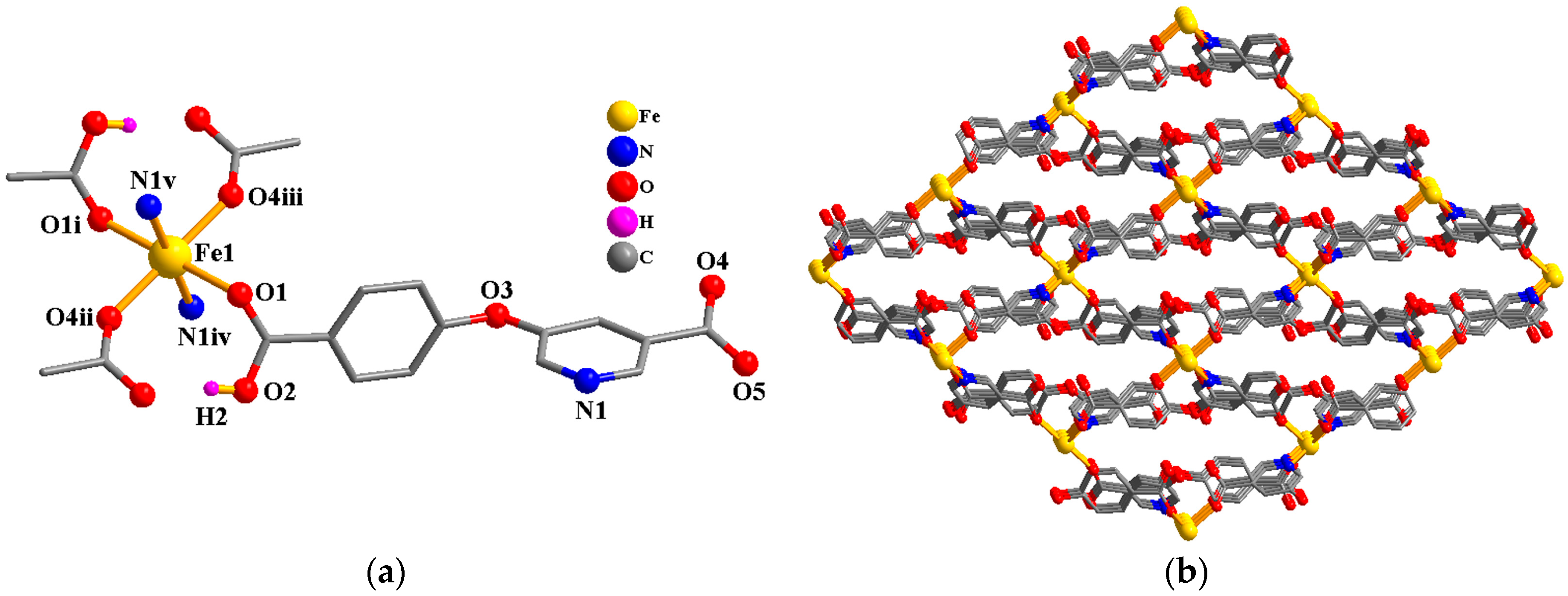
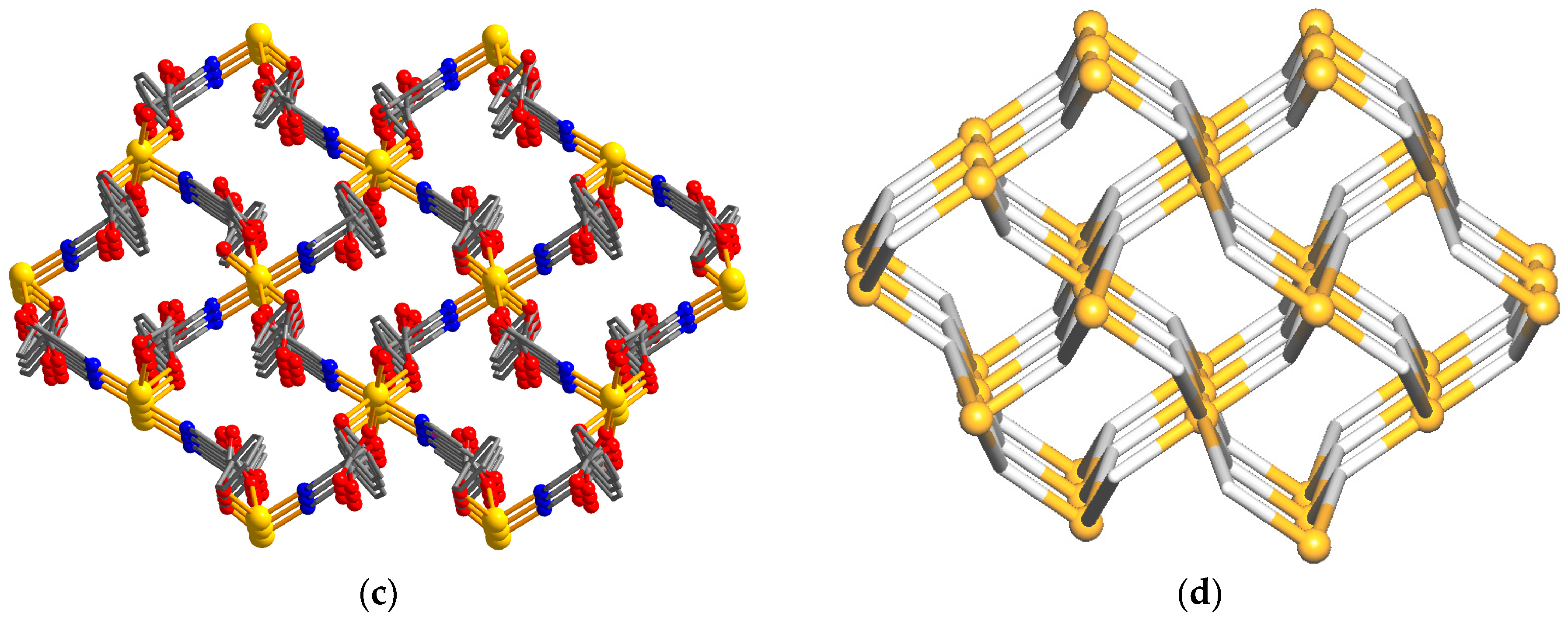
3.2. Description of Structural and Topological Features
3.3. TGA and PXRD
3.4. Catalytic Functionalization of Alkanes
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Glover, T.G.; Mu, B. (Eds.) Gas Adsorption in Metal-Organic Frameworks: Fundamentals and Applications; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Hashemi, L.; Morsali, A. Pillared Metal-Organic Frameworks: Properties and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Adil, K.; Belmabkhout, Y.; Pillai, R.S.; Cadiau, A.; Bhatt, P.M.; Assen, A.H.; Maurin, G.; Eddaoudi, M. Gas/vapour separation using ultra-microporous metal-organic frameworks: Insights into the structure/separation relationship. Chem. Soc. Rev. 2017, 46, 3402–3430. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Qian, G. (Eds.) Metal-Organic Frameworks for Photonics Applications; Springer: Berlin, Germany, 2014. [Google Scholar]
- Cui, Y.J.; Yue, Y.F.; Qian, G.D.; Chen, B.L. Luminescent functional metal-organic frameworks. Chem. Rev. 2012, 112, 1126–1162. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Z.; Zhang, R.; Jiao, L.; Jiang, H.L. Metal-organic framework-derived porous materials for catalysis. Coord. Chem. Rev. 2018, 362, 1–23. [Google Scholar] [CrossRef]
- Yu, D.Y.; Li, L.B.; Wu, M.H.; Crittenden, J.C. Enhanced photocatalytic ozonation of organic pollutants using an iron-based metal-organic framework. Appl. Catal. B Environ. 2019, 251, 66–75. [Google Scholar] [CrossRef]
- Gu, J.Z.; Wen, M.; Cai, Y.; Shi, Z.F.; Nesterov, D.S.; Kirillova, M.V.; Kirillov, A.M. Cobalt(II) coordination polymers assembled from unexplored pyridine-carboxylic acid: Structural diversity and catalytic oxidation of alcohols. Inorg. Chem. 2019, 58, 5875–5885. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.; Ramachandran, V.; Clark, R.J.; Zhou, H.D.; Toby, B.H.; Dalal, N.S.; Kroto, H.W.; Cheetham, A.K. Multiferroic behavior associated with an order-disorder hydrogen bonding transition in metal-organic frameworks (MOFs) with the perovskite ABX3 architecture. J. Am. Chem. Soc. 2009, 131, 13625–13627. [Google Scholar] [CrossRef] [PubMed]
- Minguez, E.G.; Coronado, E. Magnetic functionalities in MOFs: From the framework to the pore. Chem. Soc. Rev. 2018, 47, 533–557. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Fan, R.; Song, Y.; Wang, A.; Xing, K.; Du, X.; Wang, P.; Yang, Y. A highly sensitive turn-on ratiometric luminescent probe based on postsynthetic modification of Tb3+@Cu-MOF for H2S detection. J. Mater. Chem. C 2017, 5, 9943–9951. [Google Scholar] [CrossRef]
- Jaros, S.W.; Sokolnicki, J.; Wołoszyn, A.; Haukka, M.; Kirillov, A.M.; Smoleński, P. A novel 2D coordination network built from hexacopper(I)-iodide clusters and cagelike aminophosphine blocks for reversible “turn-on” sensing of aniline. J. Mater. Chem. C 2018, 6, 1670–1678. [Google Scholar] [CrossRef]
- Mao, N.N.; Hu, P.; Yu, F.; Chen, X.; Zhuang, G.L.; Zhang, T.L.; Li, B. A series of transition metal coordination polymers based on a rigid bi-functional carboxylate–triazolate tecton. CrystEngComm 2017, 19, 4586–4594. [Google Scholar] [CrossRef]
- Rowsell, J.L.C.; Spencer, E.C.; Eckert, J.; Howard, J.A.K.; Yaghi, O.M. Gas adsorption sites in a large-pore metal-organic framework. Science 2005, 309, 1350–1354. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, T.A.; Santos, C.I.M.; André, V.; Kłak, J.; Kirillova, M.V.; Kirillov, A.M. Copper(II) Coordination Polymers Self-assembled from Aminoalcohols and Pyromellitic Acid: Highly Active Pre-catalysts for the Mild Water-promoted Oxidation of Alkanes. Inorg. Chem. 2016, 55, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Sotnik, S.A.; Polunin, R.A.; Kiskin, M.A.; Kirillov, A.M.; Dorofeeva, V.N.; Gavrilenko, K.S.; Eremenko, I.L.; Novotortsev, V.M.; Kolotilov, S.V. Heterometallic Coordination Polymers Assembled from Trigonal Trinuclear Fe2Ni-Pivalate Blocks and Polypyridine Spacers: Topological Diversity, Sorption, and Catalytic Properties. Inorg. Chem. 2015, 54, 5169–5181. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.Z.; Gao, Z.Q.; Tang, Y. pH and auxiliary ligand influence on the structural variations of 5(2′-carboxylphenyl) nicotate coordination polymers. Cryst. Growth Des. 2012, 12, 3312–3323. [Google Scholar] [CrossRef]
- Dong, Y.B.; Jiang, Y.Y.; Li, J.; Ma, J.P.; Liu, F.L.; Tang, B.; Huang, R.Q.; Batten, S.R. Temperature-dependent synthesis of metal-organic frameworks based on a flexible tetradentate ligand with bidirectional coordination donors. J. Am. Chem. Soc. 2007, 129, 4520–4521. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.Z.; Cui, Y.H.; Liang, X.X.; Wu, J.; Lv, D.Y.; Kirillov, A.M. Structurally distinct metal-organicand H-bonded networks derived from 5-(6-carboxypyridin-3-yl)isophthalic acid: Coordination and template effect of 4,4′-bipyridine. Cryst. Growth Des. 2016, 16, 4658–4670. [Google Scholar] [CrossRef]
- Jaros, S.W.; da Silva, M.F.C.G.; Florek, M.; Oliveira, M.C.; Smoleński, P.; Pombeiro, A.J.L.; Kirillov, A.M. Aliphatic dicarboxylate directed assembly of silver(I) 1,3,5-triaza-7-phosphaadamantane coordination networks: Topological versatility and antimicrobial activity. Cryst. Growth Des. 2014, 14, 5408–5417. [Google Scholar] [CrossRef]
- Wu, W.-P.; Li, Z.-S.; Liu, B.; Liu, P.; Xi, Z.-P.; Wang, Y.-Y. Double-step CO2 sorption and guest-induced single-crystal-to-single-crystal transformation in a flexible porous framework. Dalton Trans. 2015, 44, 10141–10145. [Google Scholar] [CrossRef]
- Liang, Y.T.; Yang, G.P.; Liu, B.; Yan, Y.T.; Xi, Z.P.; Wang, Y.Y. Four super water-stable lanthanide-organic frameworks with active uncoordinated carboxylic and pyridyl groups for selective luminescence sensing of Fe3+. Dalton Trans. 2015, 44, 13325–13330. [Google Scholar] [CrossRef]
- Gu, J.Z.; Kirillov, A.M.; Wu, J.; Lv, D.Y.; Tang, Y.; Wu, J.C. Synthesis, structural versatility, luminescent and magnetic properties of a series of coordination polymers constructed from biphenyl-2,4,4′-tricarboxylate and different N-donor ligands. CrystEngComm 2013, 15, 10287–10303. [Google Scholar] [CrossRef]
- Shao, Y.L.; Cui, Y.H.; Gu, J.Z.; Wu, J.; Wang, Y.W.; Kirillov, A.M. Exploring biphenyl-2,4,4′-tricarboxylic acid as a flexible building block for the hydrothermal self-assembly of diverse metal-organic and supramolecular networks. CrystEngComm 2016, 18, 765–778. [Google Scholar] [CrossRef]
- Gu, J.Z.; Liang, X.X.; Cui, Y.H.; Wu, J.; Kirillov, A.M. Exploring 4-(3-carboxyphenyl)picolinic acid as a semirigid building block for the hydrothermal self-assembly of diverse metal-organic and supramolecular networks. CrystEngComm 2017, 19, 117–128. [Google Scholar] [CrossRef]
- Gu, J.Z.; Wen, M.; Liang, X.X.; Shi, Z.; Kirillova, M.V.; Kirillov, A.M. Multifunctional aromatic carboxylic acids as versatile building blocks for hydrothermal design of coordination polymers. Crystals 2018, 8, 83. [Google Scholar] [CrossRef]
- Gu, J.Z.; Liang, X.X.; Cai, Y.; Wu, J.; Shi, Z.F.; Kirillov, A.M. Hydrothermal assembly, structures, topologies, luminescence, and magnetism of a novel series of coordination polymers driven by a trifunctional nicotinic acid building block. Dalton Trans. 2017, 46, 10908–10925. [Google Scholar] [CrossRef]
- Gu, J.Z.; Cai, Y.; Liang, X.X.; Wu, J.; Shi, Z.F.; Kirillov, A.M. Bringing 5-(3,4-dicarboxylphenyl)picolinic acid to crystal engineering research: Hydrothermal assembly, structural features, and photocatalytic activity of Mn, Ni, Cu, and Zn coordination polymer. CrystEngComm 2018, 20, 906–916. [Google Scholar] [CrossRef]
- Gu, J.Z.; Cai, Y.; Qian, Z.Y.; Wen, M.; Shi, Z.F.; Lv, D.Y.; Kirillov, A.M. A new series of Co, Ni, Zn, and Cd metal-organic architectures driven by an unsymmetrical biphenyl-tricarboxylic acid: Hydrothermal assembly, structural features and properties. Dalton Trans. 2018, 47, 7431–7444. [Google Scholar] [CrossRef]
- Gu, J.Z.; Cai, Y.; Wen, M.; Shi, Z.F.; Kirillov, A.M. A new series of Cd(II) metal-organic architectures driven by soft ether-bridged tricarboxylate spacers: Synthesis, structural and topological versatility, and photocatalytic properties. Dalton Trans. 2017, 46, 14327–14339. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXS-97, Program for X-ray Crystal Structure Determination; University of Gottingen: Göttingen, Germany, 1997. [Google Scholar]
- Sheldrick, G.M. SHELXL-97, Program for X-ray Crystal Structure Refinement; University of Gottingen: Göttingen, Germany, 1997. [Google Scholar]
- Blatov, V.A. Multipurpose crystallochemical analysis with the program package TOPOS. IUCr CompComm Newsl. 2006, 7, 4–38. [Google Scholar]
- Blatov, V.A.; Shevchenko, A.P.; Proserpio, D.M. Applied topological analysis of crystal structures with the program package ToposPro. Cryst. Growth Des. 2014, 14, 3576–3586. [Google Scholar] [CrossRef]
- Lu, W.G.; Gu, J.Z.; Jiang, L.; Tan, M.Y.; Lu, T.B. Achiral and chiral coordination polymers containing helical chains: The chirality transfer between helical chains. Cryst. Growth Des. 2008, 8, 192–199. [Google Scholar] [CrossRef]
- Gu, J.Z.; Gao, Z.Q. Synthesis, crystal structure, magnetic properties of a compound based on eight-coordinated iron(II) ion. Chin. J. Ionrg. Chem. 2012, 28, 1038–1042. [Google Scholar]
- Costa, I.F.M.; Kirillova, M.V.; André, V.; Fernandes, T.A.; Kirillov, A.M. Tetracopper(II) Cores Driven by an Unexplored Trifunctional Aminoalcohol Sulfonic Acid for Mild Catalytic C–H Functionalization of Alkanes. Catalysts 2019, 9, 321. [Google Scholar] [CrossRef]
- Dias, S.S.P.; Kirillova, M.V.; André, V.; Kłak, J.; Kirillov, A.M. New Tetracopper (II) Cubane Cores Driven by a Diamino Alcohol: Self-assembly Synthesis, Structural and Topological Features, and Magnetic and Catalytic Oxidation Properties. Inorg. Chem. 2015, 54, 5204–5212. [Google Scholar] [CrossRef]
- Dias, S.S.P.; Kirillova, M.V.; André, V.; Kłak, J.; Kirillov, A.M. New tricopper(II) cores self-assembled from aminoalcohol biobuffers and homophthalic acid: Synthesis, structural and topological features, magnetic properties and mild catalytic oxidation of cyclic and linear C5–C8 alkanes. Inorg. Chem. Front. 2015, 2, 525–537. [Google Scholar] [CrossRef]
- Fernandes, T.A.; Santos, C.I.M.; André, V.; Dias, S.S.P.; Kirillova, M.V.; Kirillov, A.M. New aqua-soluble dicopper(II) aminoalcoholate cores for mild and water-assisted catalytic oxidation of alkanes. Catal. Sci. Technol. 2016, 6, 4584–4593. [Google Scholar] [CrossRef]
- Kirillov, A.M.; Shul’pin, G.B. Pyrazinecarboxylic acid and analogs: Highly efficient co-catalysts in the metal-complex-catalyzed oxidation of organic compounds. Coord. Chem. Rev. 2013, 257, 732–754. [Google Scholar] [CrossRef]
- Armakola, E.; Colodrero, R.M.P.; Bazaga-García, M.; Salcedo, I.R.; Choquesillo-Lazarte, D.; Cabeza, A.; Kirillova, M.V.; Kirillov, A.M.; Demadis, K.D. Three-Component Copper-Phosphonate-Auxiliary Ligand Systems: Proton Conductors and Efficient Catalysts in Mild Oxidative Functionalization of Cycloalkanes. Inorg. Chem. 2018, 57, 10656–10666. [Google Scholar] [CrossRef]
- Kirillova, M.V.; Kirillov, A.M.; Pombeiro, A.J.L. Metal-free and copper-promoted single-pot hydrocarboxylation of cycloalkanes to carboxylic acids in aqueous medium. Adv. Synth. Catal. 2009, 351, 2936–2948. [Google Scholar] [CrossRef]
- Kirillova, M.V.; Kirillov, A.M.; Pombeiro, A.J.L. Mild, single-pot hydrocarboxylation of gaseous alkanes to carboxylic acids in metal-free and copper-promoted aqueous systems. Chem. Eur. J. 2010, 16, 9485–9493. [Google Scholar] [CrossRef]
- Shul’pin, G.B. Metal-catalyzed hydrocarbon oxygenations in solutions: The dramatic role of additives: A review. J. Mol. Catal. A Chem. 2002, 189, 39–66. [Google Scholar] [CrossRef]
- Shul’pin, G.B. Hydrocarbon Oxygenations with Peroxides Catalyzed by Metal Compounds. Mini-Rev. Org. Chem. 2009, 6, 95–104. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Cryst. 2016, B72, 171–179. [Google Scholar] [CrossRef]
- Shilov, A.E.; Shul’pin, G.B. Activation and Catalytic Reactions of Saturated Hydrocarbons in the Presence of Metal Complexes; Kluwer Acad. Publ.: Dordrecht, The Netherlands, 2000. [Google Scholar]
- Bäckvall, J.-E. (Ed.) Modern Oxidation Methods, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Shul’pin, G.B. New Trends in Oxidative Functionalization of Carbon–Hydrogen Bonds: A Review. Catalysts 2016, 6, 50. [Google Scholar] [CrossRef]
- Hagen, J. Industrial Catalysis: A Practical Approach; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Schuchardt, U.; Cardoso, D.; Sercheli, R.; Pereira, R.; da Cruz, R.S.; Guerreiro, M.C.; Mandelli, D.; Spinace, E.V.; Pires, E.L. Cyclohexane Oxidation Continues to be a Challenge. Appl. Catal. A Gen. 2001, 211, 1–17. [Google Scholar] [CrossRef]



| Compound | 1 |
|---|---|
| Chemical formula | C26H16FeN2O10 |
| Molecular weight | 572.26 |
| Crystal system | Monoclinic |
| Space group | P21/n |
| a/Å | 9.4673(7) |
| b/Å | 9.2170(6) |
| c/Å | 14.0358(9) |
| α/ (°) | 90 |
| β/(°) | 108.079(8) |
| γ/ (°) | 90 |
| V/Å3 | 1164.30(15) |
| Z | 2 |
| F(000) | 584 |
| Crystal size/mm | 0.27 × 0.25 × 0.23 |
| θ Range for data collection | 3.770–25.049 |
| Limiting indices | −9 ≤ h ≤ 11, −10 ≤ k ≤ 10, −16 ≤ l ≤ 10 |
| Reflections collected/unique (Rint) | 4273/2053 (0.0558) |
| Dc/ (Mg·cm−3) | 1.632 |
| μ/mm−1 | 0.715 |
| Data/restraints/parameters | 2053/0/179 |
| Goodness-of-fit on F2 | 1.029 |
| Final R indices [(I ≥ 2σ(I))] R1, wR2 | 0.0502, 0.0890 |
| R indices (all data) R1, wR2 | 0.0895, 0.1084 |
| Largest diff. peak and hole/(e·Å−3) | 0.329 and −0.376 |
| Catalyst | Product Yield (%)b | ||||
|---|---|---|---|---|---|
| i-Propanol | Acetone | n-Propanol | Propanal | Total | |
| 1/PCA | 6.1 | 11.3 | 3.0 | 1.6 | 22.0 |
| 1/TFA | 2.1 | 2.3 | 1.5 | 1.4 | 7.3 |
| Substrate | Yield (%)b | |||
|---|---|---|---|---|
| Cycloalkane-Carboxylic Acid | Cyclic Ketone | Cyclic alcohol | Total | |
| Cyclopentane | 13.9 | 3.4 | 1.7 | 19.0 |
| Cyclohexane | 20.9 | 1.1 | 0.6 | 22.6 |
| Cycloheptane | 9.2 | 6.5 | 2.5 | 18.2 |
| Cyclooctane | 4.8 | 7.8 | 5.9 | 18.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, N.; Li, Y.; Gu, J.; Kirillova, M.V.; Kirillov, A.M. Synthesis, Structural Features, and Catalytic Activity of an Iron(II) 3D Coordination Polymer Driven by an Ether-Bridged Pyridine-Dicarboxylate. Crystals 2019, 9, 369. https://doi.org/10.3390/cryst9070369
Zhao N, Li Y, Gu J, Kirillova MV, Kirillov AM. Synthesis, Structural Features, and Catalytic Activity of an Iron(II) 3D Coordination Polymer Driven by an Ether-Bridged Pyridine-Dicarboxylate. Crystals. 2019; 9(7):369. https://doi.org/10.3390/cryst9070369
Chicago/Turabian StyleZhao, Na, Yu Li, Jinzhong Gu, Marina V. Kirillova, and Alexander M. Kirillov. 2019. "Synthesis, Structural Features, and Catalytic Activity of an Iron(II) 3D Coordination Polymer Driven by an Ether-Bridged Pyridine-Dicarboxylate" Crystals 9, no. 7: 369. https://doi.org/10.3390/cryst9070369
APA StyleZhao, N., Li, Y., Gu, J., Kirillova, M. V., & Kirillov, A. M. (2019). Synthesis, Structural Features, and Catalytic Activity of an Iron(II) 3D Coordination Polymer Driven by an Ether-Bridged Pyridine-Dicarboxylate. Crystals, 9(7), 369. https://doi.org/10.3390/cryst9070369