Thermochemically Stable Liquid-Crystalline Gold(I) Complexes Showing Enhanced Room Temperature Phosphorescence
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
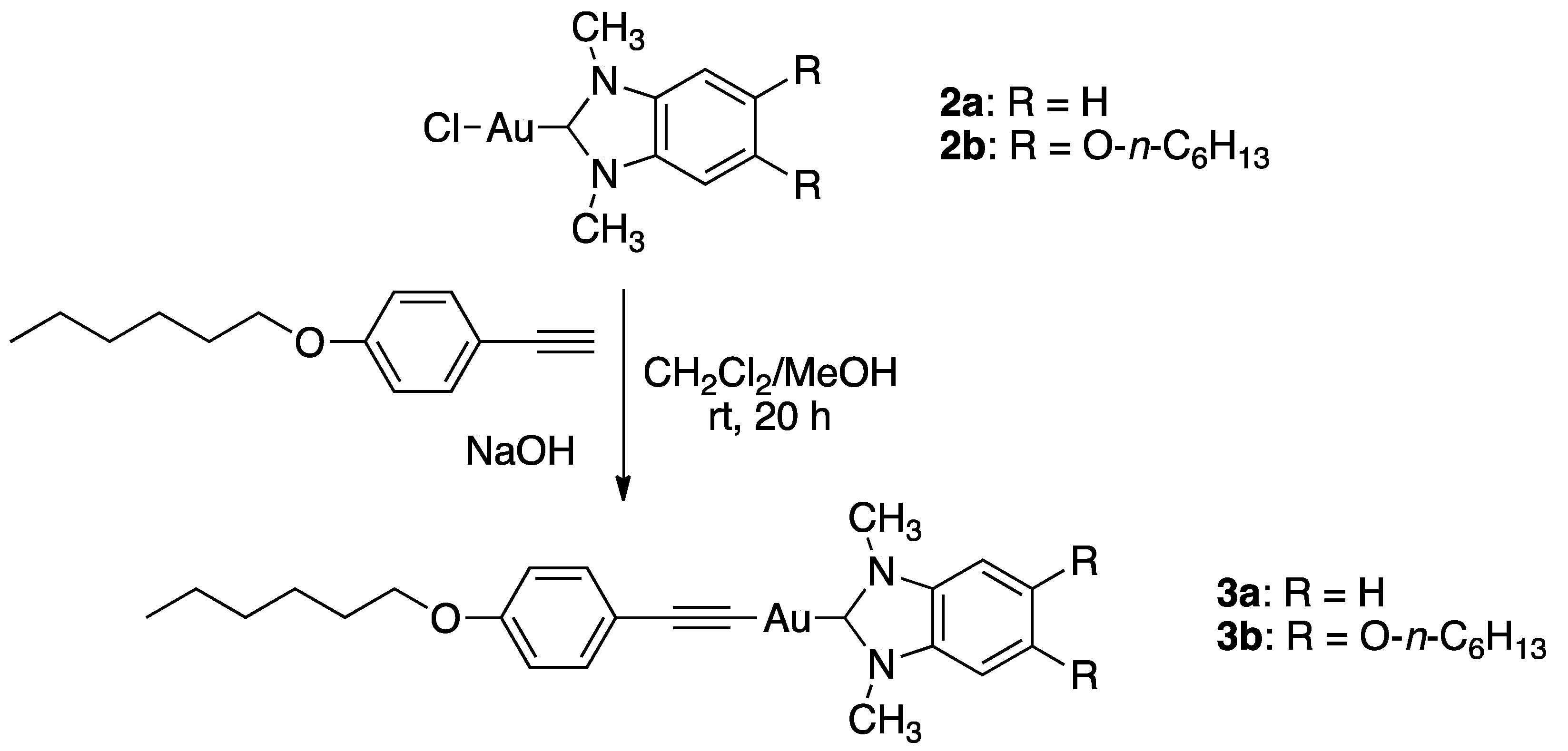
2.2. Synthesis of Au Complex 3a
2.3. Synthesis of Au Complex 3b
2.4. Single Crystal X-ray Structure Analysis
2.5. Thermochemical and Photoluminescence Properties
3. Results and Discussion
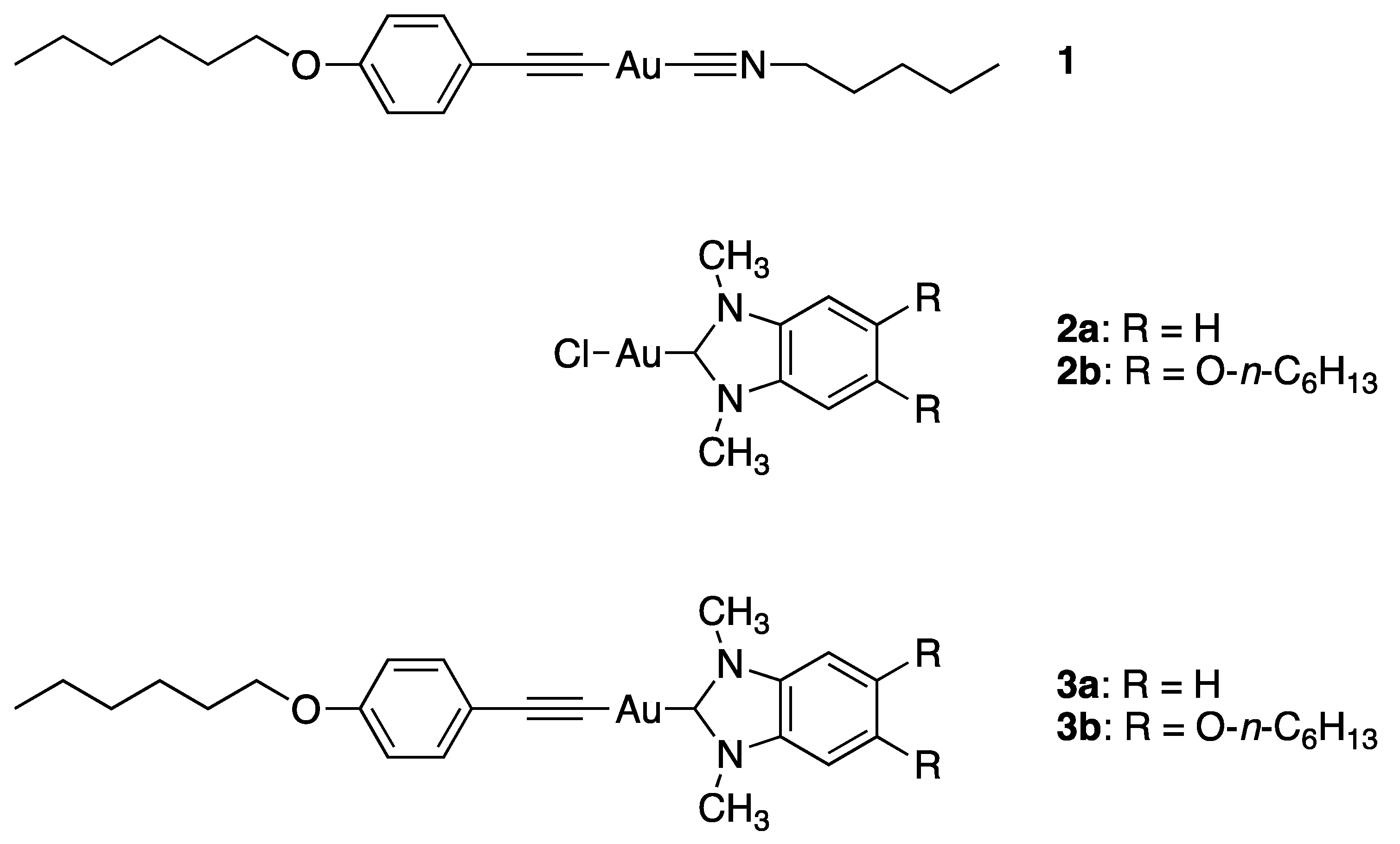
3.1. Synthesis and Structural Characterization of Complexes
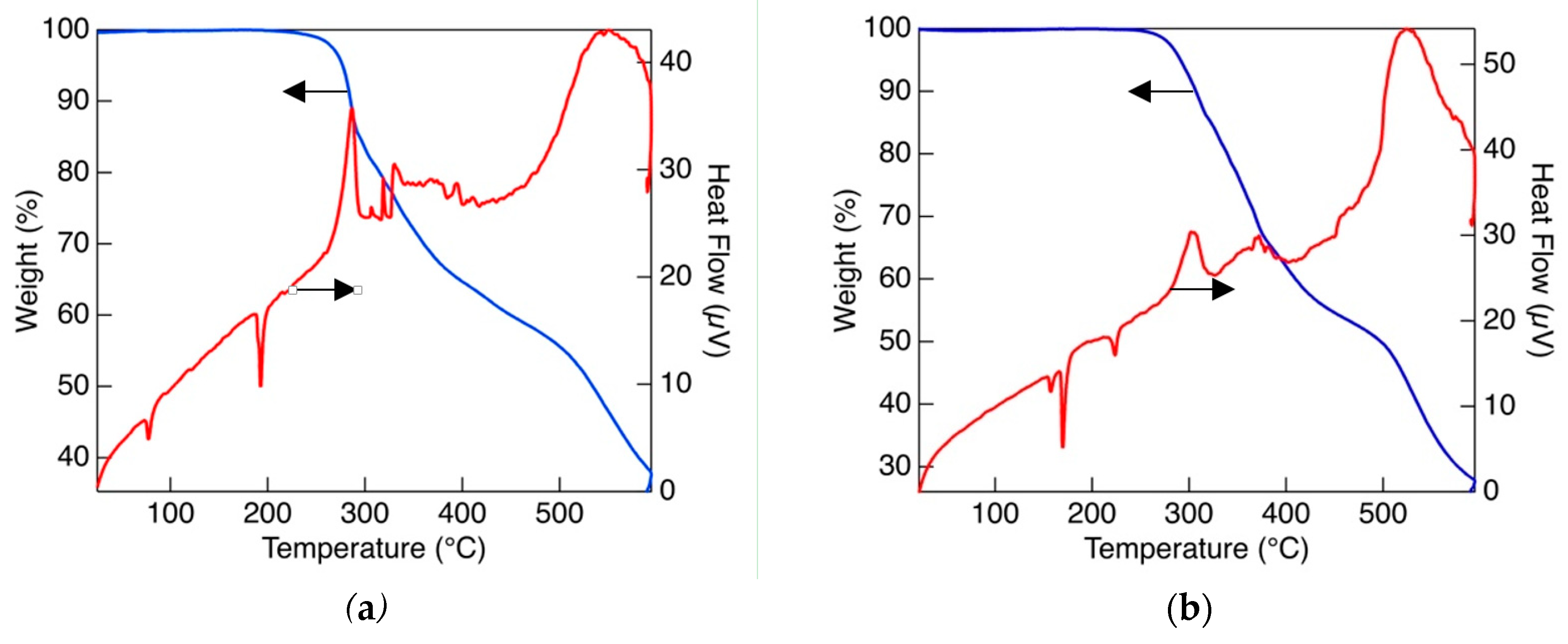
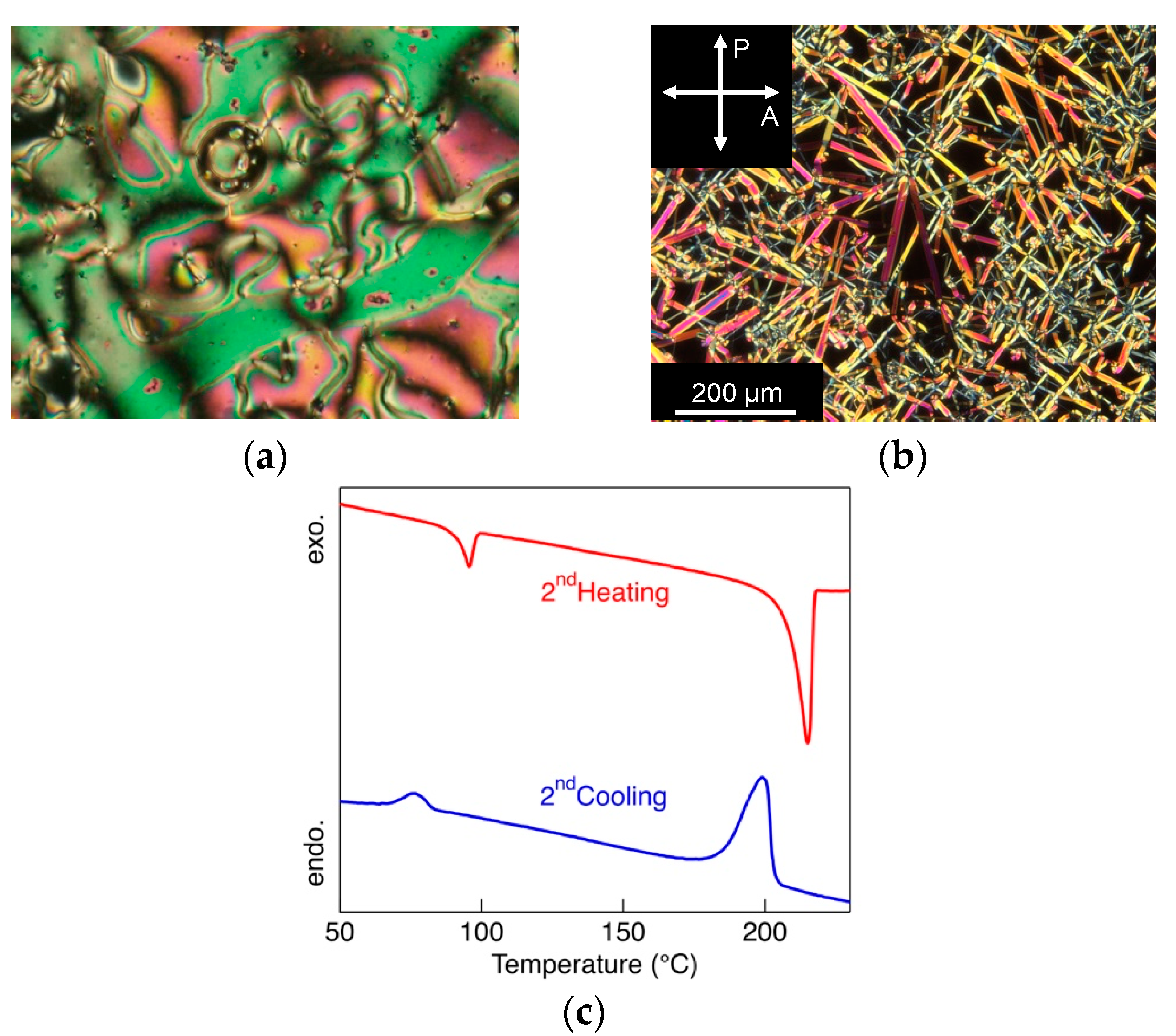
3.2. Thermal Properties
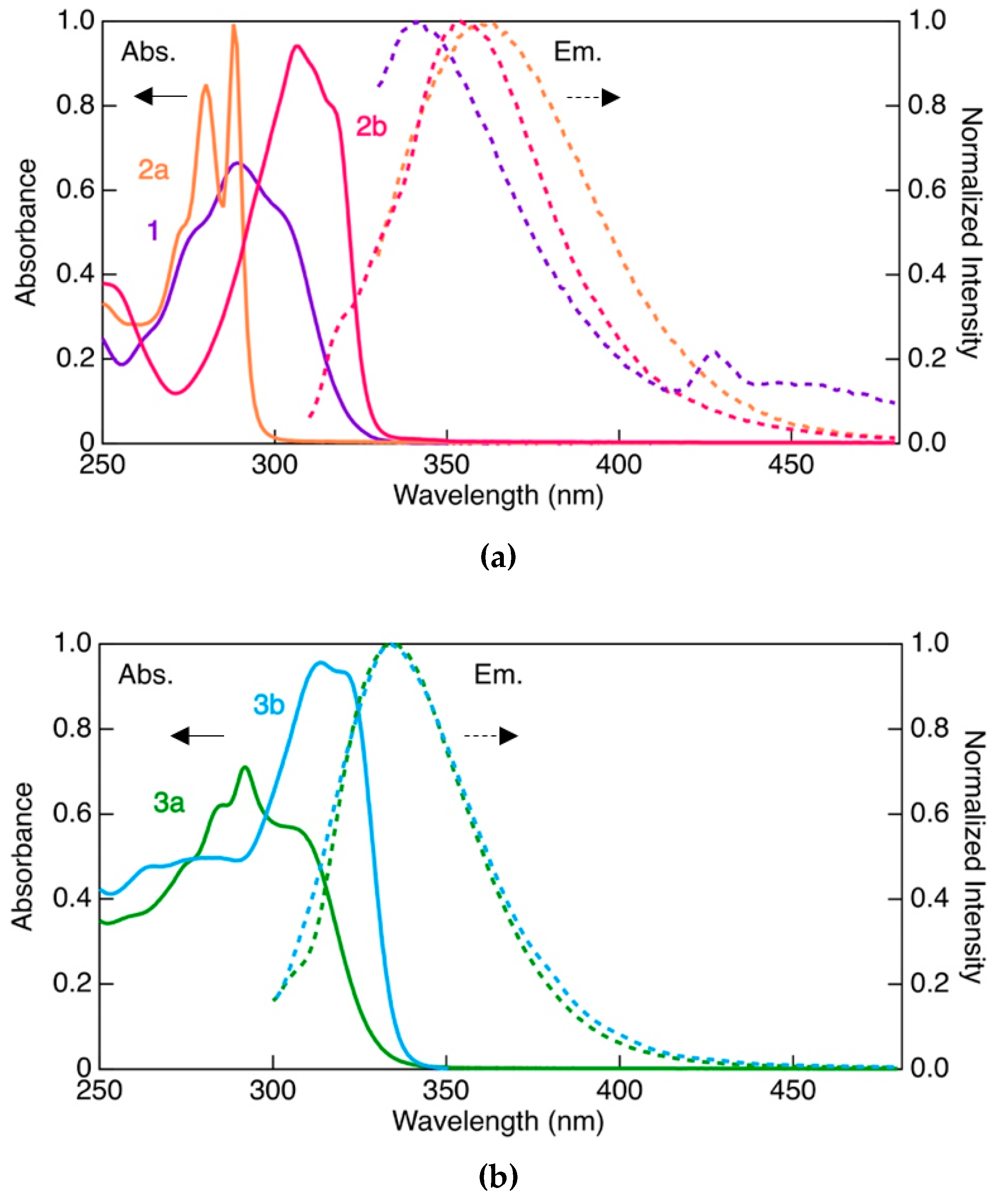
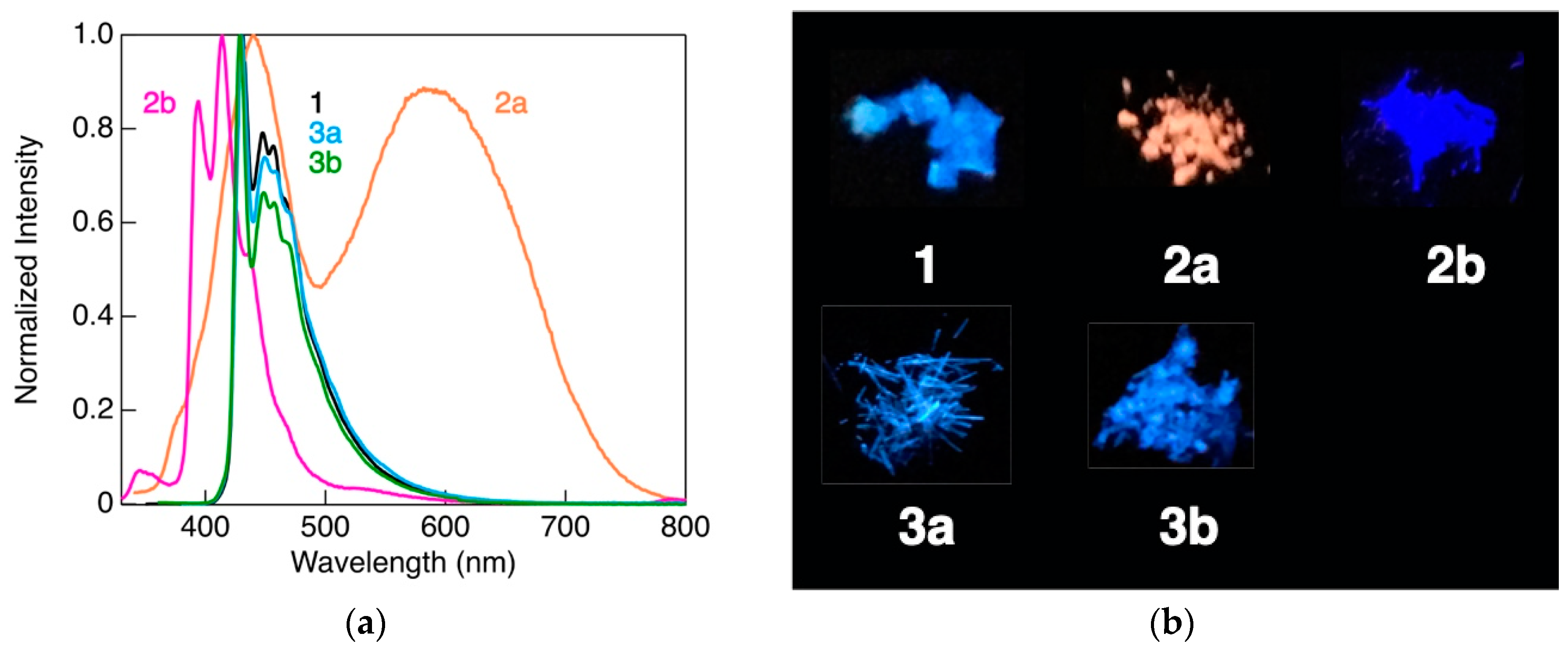
3.3. Photophysical Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lim, X.Z. The nanolight revolution is coming. Nature 2016, 531, 26–28. [Google Scholar] [CrossRef]
- Song, J.; Li, J.; Li, X.; Xu, L.; Dong, Y.; Zeng, H. Quantum dot light-emitting diodes based on inorganic perovskite cesium lead halides (CsPbX3). Adv. Mater. 2015, 27, 7162–7167. [Google Scholar] [CrossRef]
- Feng, G.; Liu, B. Aggregation-induced emission (AIE) dots: Emerging theranostic nanolights. Acc. Chem. Res. 2018, 51, 1404–1414. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Wang, Y.; Cai, C.; Lin, H. Activating room temperature long afterglow of carbon dots via covalent fixation. Chem. Mater. 2017, 29, 4866–4873. [Google Scholar] [CrossRef]
- Liu, J.; Wang, N.; Yu, Y.; Yan, Y.; Zhang, H.; Li, J.; Yu, J. Carbon dots in zeolites: A new class of thermally activated delayed fluorescence materials with ultralong lifetimes. Sci. Adv. 2017, 3, e1603171. [Google Scholar] [CrossRef]
- Hong, Y.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission. Chem. Soc. Rev. 2011, 40, 5361–5388. [Google Scholar] [CrossRef] [PubMed]
- Dang, D.; Qiu, Z.; Han, T.; Liu, Y.; Chen, M.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. 1 + 1 >> 2: Dramatically enhancing the emission efficiency of TPE-based AIEgens but keeping their emission color through tailored alkyl linkages. Adv. Funct. Mater. 2018, 28, 1707210. [Google Scholar] [CrossRef]
- Lintang, H.O.; Kinbara, K.; Tanaka, K.; Yamashita, T.; Aida, T. Self-repair of a one-dimensional molecular assembly in mesoporous silica by a nanoscopic template effect. Angew. Chem. Int. Ed. 2010, 49, 4241–4245. [Google Scholar] [CrossRef]
- Mocanu, A.S.; Amela-Cortes, M.; Molard, Y.; Cîrcu, V.; Cordier, S. Liquid crystal properties resulting from synergetic effects between non-mesogenic organic molecules and a one nanometre sized octahedral transition metal cluster. Chem. Commun. 2011, 47, 2056–2058. [Google Scholar] [CrossRef]
- Krikorian, M.; Liu, S.; Swager, T.M. Columnar liquid crystallinity and mechanochromism in cationic platinum (II) complexes. J. Am. Chem. Soc. 2014, 136, 2952–2955. [Google Scholar] [CrossRef]
- Kawano, S.I.; Ishida, Y.; Tanaka, K. Columnar liquid-crystalline metallomacrocycles. J. Am. Chem. Soc. 2015, 137, 2295–2302. [Google Scholar] [CrossRef]
- Concellón, A.; Marcos, M.; Romero, P.; Serrano, J.L.; Termine, R.; Golemme, A. Not only columns: High hole mobility in a discotic nematic mesophase formed by metal-containing porphyrin-core dendrimers. Angew. Chem. Int. Ed. 2017, 129, 1279–1283. [Google Scholar] [CrossRef]
- Fujisawa, K.; Mitsuhashi, F.; Anukul, P.; Taneki, K.; Younis, O.; Tsutsumi, O. Photoluminescence behavior of liquid-crystalline gold(I) complexes with a siloxane group controlled by molecular aggregate structures in condensed phases. Polym. J. 2018, 50, 761–769. [Google Scholar] [CrossRef]
- Sathyanarayana, A.; Nakamura, S.; Hisano, K.; Tsutsumi, O.; Srinivas, K.; Prabusankar, G. Controlling the solid-state luminescence of gold(I) N-heterocyclic carbene complexes through changes in the structure of molecular aggregates. Sci. China Chem. 2018, 61, 957–965. [Google Scholar] [CrossRef]
- Preeyanuch, A.; Sugimoto, N.; Sakamoto, K.; Rokusha, Y.; Taneki, K.; Fujisawa, K.; Tsutsumi, O. Effects of aromatic core and flexible terminal chain structures on the properties of luminous liquid-crystalline gold(I) complexes for functional materials. Mol. Cryst. Liq. Cryst. 2018, 662, 176–187. [Google Scholar]
- Yamada, S.; Rokusha, Y.; Kawano, R.; Fujisawa, K.; Tsutsumi, O. Mesogenic gold complexes showing aggregation-induced enhancement of phosphorescence in both crystalline and liquid-crystalline phases. Faraday Discuss. 2017, 196, 269–283. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Yamaguchi, S.; Tsutsumi, O. Electron-density distribution tuning for enhanced thermal stability of luminescent gold complexes. J. Mater. Chem. C 2017, 5, 7977–7984. [Google Scholar] [CrossRef]
- Kawano, R.; Younis, O.; Ando, A.; Rokusha, Y.; Yamada, S.; Tsutsumi, O. Photoluminescence from Au(I) complexes exhibiting color sensitivity to the structure of the molecular aggregates. Chem. Lett. 2016, 45, 66–68. [Google Scholar] [CrossRef]
- Younis, O.; Rokusha, Y.; Sugimoto, N.; Fujisawa, K.; Yamada, S.; Tsutsumi, O. Effects of molecular structure and aggregated structure on photoluminescence properties of liquid-crystalline gold(I) complexes with various aromatic rings. Mol. Cryst. Liq. Cryst. 2015, 617, 21–31. [Google Scholar] [CrossRef]
- Fujisawa, K.; Yamada, S.; Yanagi, Y.; Yoshioka, Y.; Kiyohara, A.; Tsutsumi, O. Tuning the photoluminescence of condensed-phase cyclic trinuclear Au(I) complexes through control of their aggregated structures by external stimuli. Sci. Rep. 2015, 5, 7934. [Google Scholar] [CrossRef]
- Sugimoto, N.; Tamai, S.; Fujisawa, K.; Tsutsumi, O. Photoluminescent gold(I) complex with biphenyl acetylene ligand showing stable nematic liquid-crystalline phase. Mol. Cryst. Liq. Cryst. 2014, 601, 97–106. [Google Scholar] [CrossRef]
- Fujisawa, K.; Okuda, Y.; Izumi, Y.; Nagamatsu, A.; Rokusha, Y.; Sadaike, Y.; Tsutsumi, O. Reversible thermal-mode control of luminescence from liquid-crystalline gold(I) complexes. J. Mater. Chem. C 2014, 2, 3549–3555. [Google Scholar] [CrossRef]
- Fujisawa, K.; Kawakami, N.; Onishi, Y.; Izumi, Y.; Tamai, S.; Sugimoto, N.; Tsutsumi, O. Photoluminescent properties of liquid crystalline gold(I) isocyanide complexes with a rod-like molecular structure. J. Mater. Chem. C 2013, 1, 5359–5366. [Google Scholar] [CrossRef]
- Schmidbaur, H. The aurophilicity phenomenon: A decade of experimental findings, theoretical concepts and emerging applications. Gold Bull. 2000, 33, 3–10. [Google Scholar] [CrossRef]
- Ong, C.W.; Liao, S.-C.; Chang, T.H.; Hsu, H.-F. In situ synthesis of hexakis(alkoxy)diquinoxalino[2,3-a:2′,3′-c]phenazines: Mesogenic phase transition of the electron-deficient discotic compounds. J. Org. Chem. 2004, 69, 3181–3185. [Google Scholar] [CrossRef]
- Wang, H.M.J.; Chen, C.Y.L.; Lin, I.J.B. Synthesis, structure, and spectroscopic properties of gold(I)-carbene complexes. Organometallics 1999, 18, 1216–1223. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXS-2014, Program for crystal structure solution; University of Göttingen: Göttingen, Germany, 2014. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Schmidbaur, H.; Schier, A. Aurophilic interactions as a subject of current research: An update. Chem. Soc. Rev. 2012, 41, 370–412. [Google Scholar] [CrossRef]
- Suezawa, H.; Yoshida, T.; Umezawa, Y.; Tsuboyama, S.; Nishio, M. CH/π interactions implicated in the crystal structure of transition metal compounds – A database study. Eur. J. Inorg. Chem. 2002, 3148–3155. [Google Scholar] [CrossRef]
- Yam, V.W.-W.; Au, V.K.-M.; Leung, S.Y.-L. Light-emitting self-assembled materials based on d8 and d10 transition metal complexes. Chem. Rev. 2015, 115, 7589–7728. [Google Scholar] [CrossRef]
- Tiekink, E.R.T.; Kang, J.-G. Luminescence properties of phosphinegold(I) halides and thiolates. Coord. Chem. Rev. 2009, 253, 1627–1648. [Google Scholar] [CrossRef]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission: Together we shine, united we soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef] [PubMed]
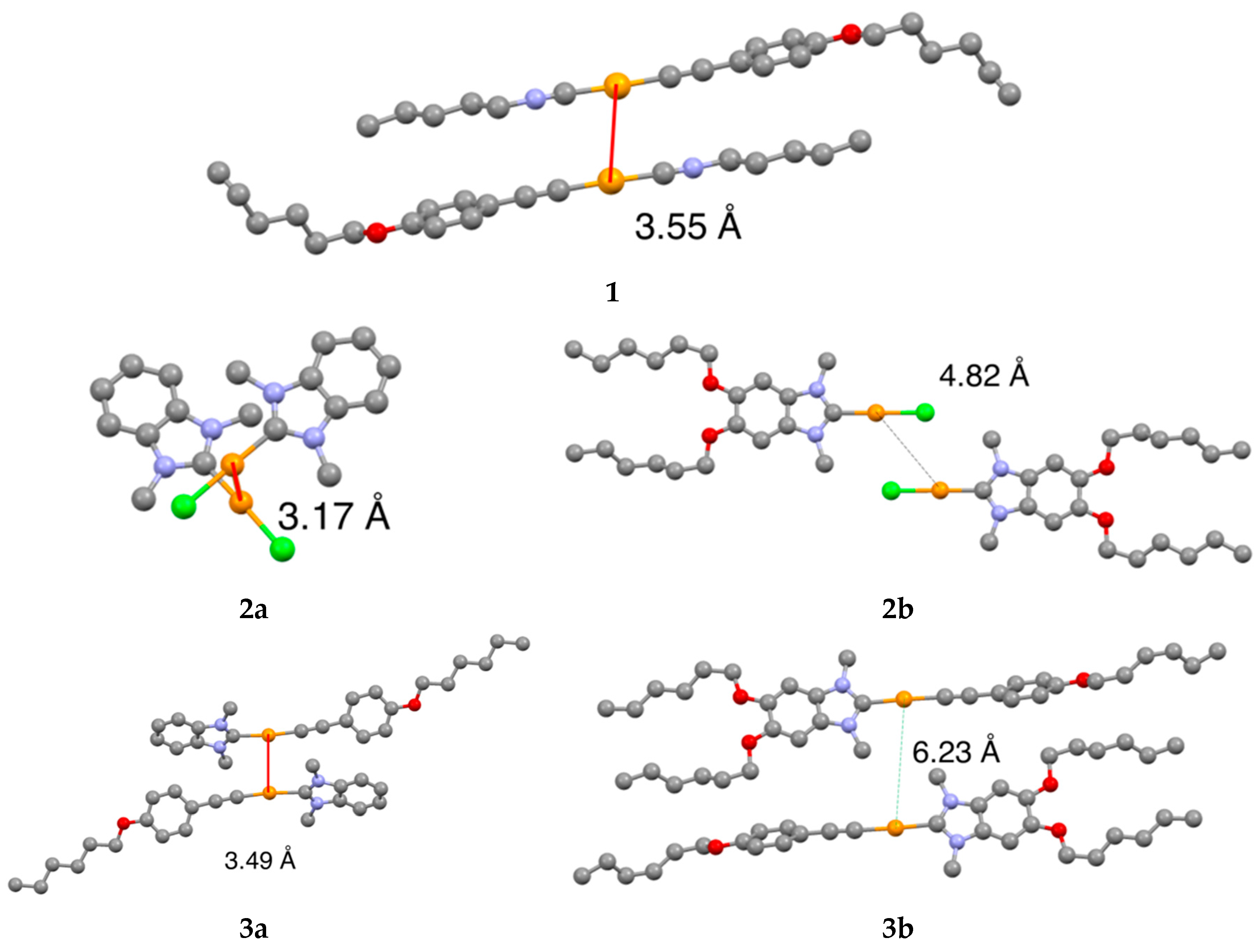
| Complex | Intermolecular Au–Au Distance (Å) | Bond Length of Au−CNHC (Å) | Tdec (°C) |
|---|---|---|---|
| 1 | 3.55 | – | 159 |
| 2a | 3.17 | 1.98 | 298 |
| 2b | 4.82 | 2.00 | 282 |
| 3a | 3.49 | 2.10 | 278 |
| 3b | 6.23 | 2.01 | 291 |
| Complex | Phase Sequences T (°C) [ΔH (kJ mol−1)] | |
|---|---|---|
| 1 | heating | Cr 89 [7] N 93 [0.2] I |
| cooling | Cr 70 [−6] N 94 [−0.3] I | |
| 2a | heating | Cr 293 I |
| cooling | Cr 277 I | |
| 2b | heating | Cr1 78 [1] Cr2 132 [3.4] Cr3 135 [18] I |
| cooling | Cr1 78 [−1.2] Cr2 117 [−23] I | |
| 3a | heating | Cr1 70 [5.6] Cr2 187 [3.8] Cr3 189 [5.6] I |
| cooling | Cr1 40 [−5.2] Cr2 182 [−17] I | |
| 3b | heating | Cr1 91 [4.7] Cr2 207 [29] I |
| cooling | Cr1 82 [−2.9] Cr2 190 SmA 203 [−23] I | |
| Complex | λmaxlum [nm] | τ [μs] | Φ | kr [s−1] | knr [s−1] |
|---|---|---|---|---|---|
| 1 | 431 | 52 | 0.50 | 9.6 × 103 | 9.6 × 103 |
| 2a | 440 | 0.34 | 0.05 | 1.4 × 105 | 2.8 × 106 |
| 590 | 5.4 | 0.11 | 2.0 × 104 | 1.7 × 105 | |
| 2b | 414 | 12 | 0.12 | 1.0 × 104 | 7.6 × 104 |
| 3a | 430 | 32 | 0.43 | 1.3 × 104 | 1.8 × 104 |
| 3b | 429 | 36 | 0.33 | 9.3 × 103 | 1.9 × 104 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuroda, Y.; Nakamura, S.-y.; Srinivas, K.; Sathyanarayana, A.; Prabusankar, G.; Hisano, K.; Tsutsumi, O. Thermochemically Stable Liquid-Crystalline Gold(I) Complexes Showing Enhanced Room Temperature Phosphorescence. Crystals 2019, 9, 227. https://doi.org/10.3390/cryst9050227
Kuroda Y, Nakamura S-y, Srinivas K, Sathyanarayana A, Prabusankar G, Hisano K, Tsutsumi O. Thermochemically Stable Liquid-Crystalline Gold(I) Complexes Showing Enhanced Room Temperature Phosphorescence. Crystals. 2019; 9(5):227. https://doi.org/10.3390/cryst9050227
Chicago/Turabian StyleKuroda, Yuki, Shin-ya Nakamura, Katam Srinivas, Arruri Sathyanarayana, Ganesan Prabusankar, Kyohei Hisano, and Osamu Tsutsumi. 2019. "Thermochemically Stable Liquid-Crystalline Gold(I) Complexes Showing Enhanced Room Temperature Phosphorescence" Crystals 9, no. 5: 227. https://doi.org/10.3390/cryst9050227
APA StyleKuroda, Y., Nakamura, S.-y., Srinivas, K., Sathyanarayana, A., Prabusankar, G., Hisano, K., & Tsutsumi, O. (2019). Thermochemically Stable Liquid-Crystalline Gold(I) Complexes Showing Enhanced Room Temperature Phosphorescence. Crystals, 9(5), 227. https://doi.org/10.3390/cryst9050227






