Controlled Two-Step Formation of Faceted Perovskite Rare-Earth Scandate Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
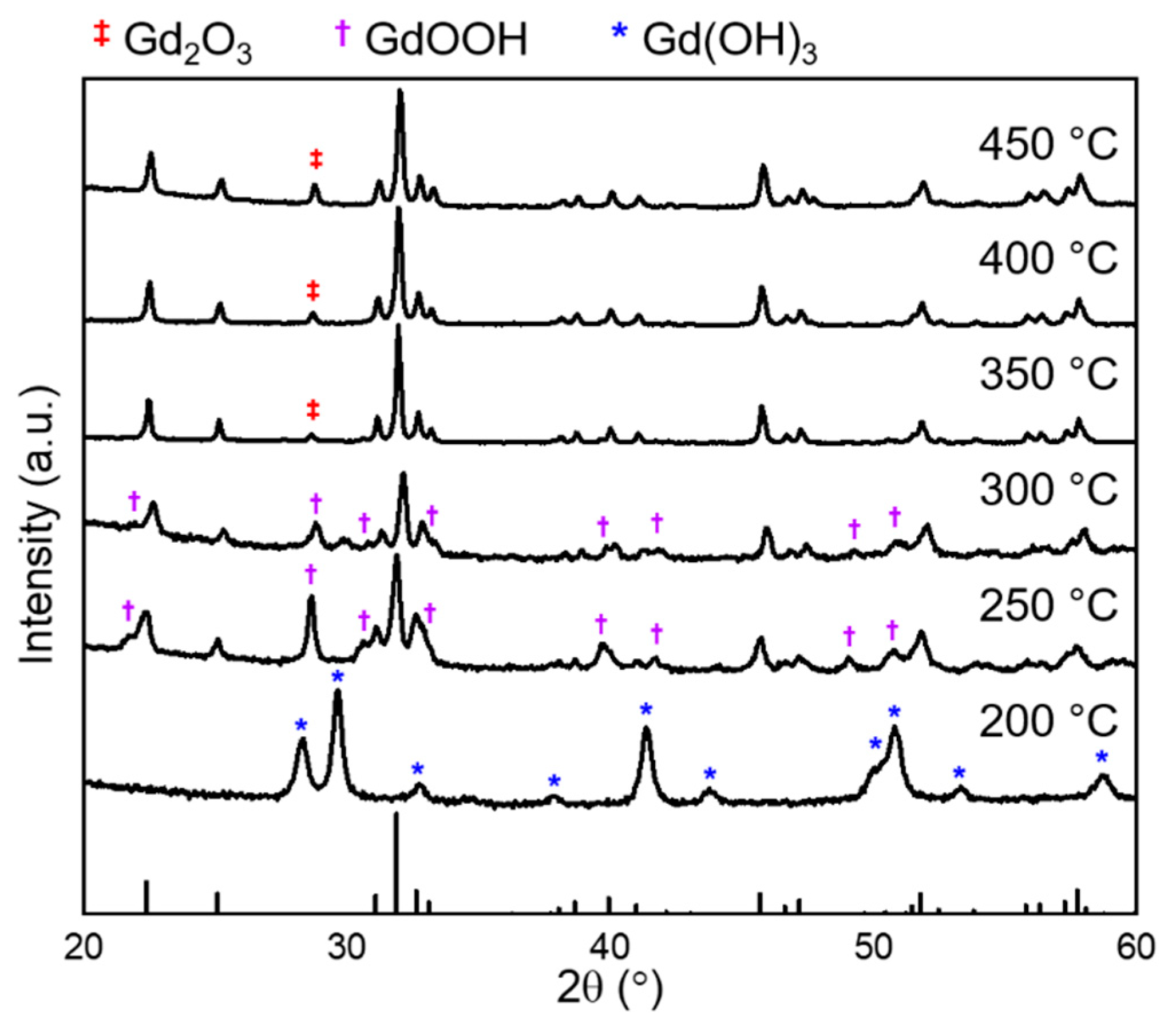
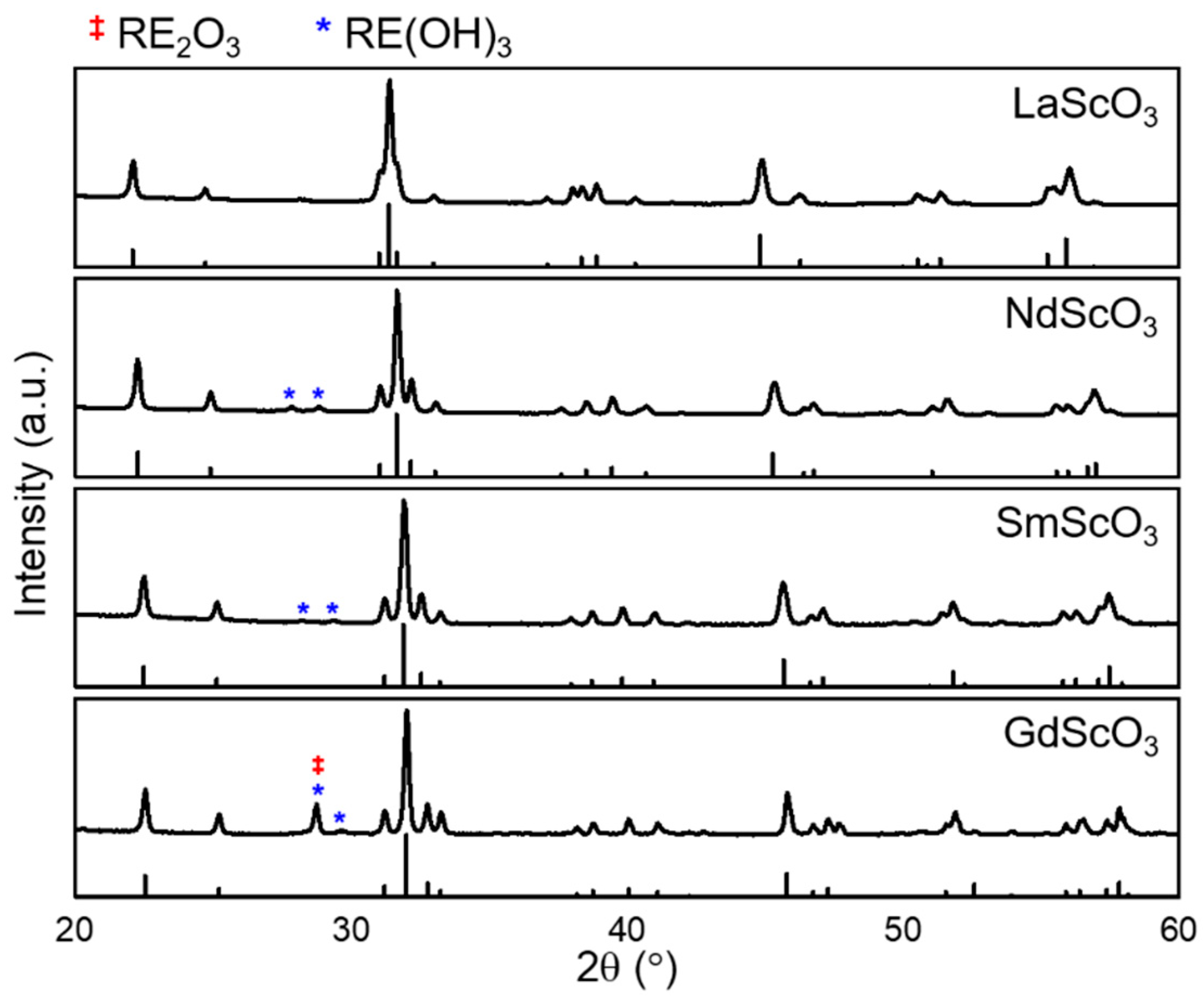
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moshfegh, A.Z. Nanoparticle Catalysts. J. Phys. D Appl. Phys. 2009, 42, 233001. [Google Scholar] [CrossRef]
- Ringe, E.; Sharma, B.; Henry, A.-I.; Marks, L.D.; Van Duyne, R.P. Single Nanoparticle Plasmonics. Phys. Chem. Chem. Phys. 2013, 15, 4110–4129. [Google Scholar] [CrossRef]
- Raj, S.; Jose, S.; Sumod, U.S.; Sabitha, M. Nanotechnology in Cosmetics: Opportunities and Challenges. J. Pharm. Bioallied Sci. 2012, 4, 186–193. [Google Scholar] [CrossRef]
- Basak, A.K.; Pramanik, A.; Islam, M.N.; Anandakrishnan, V. Challenges and Recent Developments on Nanoparticle-Reinforced Metal Matrix Composites. In Fillers and Reinforcements for Advanced Nanocomposites; Dong, Y., Umer, R., Lau, A.K.-T., Eds.; Woodhead Publishing: Sawston, Cambridge, UK, 2015; pp. 349–367. [Google Scholar]
- De Jong, W.H.; Borm, P.J.A. Drug Delivery and Nanoparticles: Applications and Hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef]
- Escudero, A.; Becerro Ana, I.; Carrillo-Carrión, C.; Núñez Nuria, O.; Zyuzin Mikhail, V.; Laguna, M.; González-Mancebo, D.; Ocaña, M.; Parak Wolfgang, J. Rare earth based nanostructured materials: synthesis, functionalization, properties and bioimaging and biosensing applications. Nanophotonics 2017, 6, 881–921. [Google Scholar] [CrossRef]
- Escudero, A.; Carrillo-Carrión, C.; Zyuzin, M.V.; Parak, W.J. Luminescent Rare-earth-based Nanoparticles: A Summarized Overview of their Synthesis, Functionalization, and Applications. Top. Curr. Chem. 2016, 374, 48. [Google Scholar] [CrossRef] [PubMed]
- Roduner, E. Size Matters: Why Nanomaterials Are Different. Chem. Soc. Rev. 2006, 35, 583–592. [Google Scholar] [CrossRef]
- Smith, A.M.; Nie, S. Semiconductor Nanocrystals: Structure, Properties, and Band Gap Engineering. Acc. Chem. Res. 2010, 43, 190–200. [Google Scholar] [CrossRef]
- Kelly, K.L.; Coronado, E.; Zhao, L.L.; Schatz, G.C. The Optical Properties of Metal Nanoparticles: The Influence of Size, Shape, and Dielectric Environment. J. Phys. Chem. B 2003, 107, 668–677. [Google Scholar] [CrossRef]
- Kreibig, U. Electronic Properties of Small Silver Particles: The Optical Constants and Their Temperature Dependence. J. Phys. F Met. Phys. 1974, 4, 999–1014. [Google Scholar] [CrossRef]
- Varghese, J.; Whatmore, R.W.; Holmes, J.D. Ferroelectric Nanoparticles, Wires and Tubes: Synthesis, Characterisation and Applications. J. Mater. Chem. C 2013, 1, 2618–2638. [Google Scholar] [CrossRef]
- Ge, H.; Huang, Y.; Hou, Y.; Xiao, H.; Zhu, M. Size Dependence of the Polarization and Dielectric Properties of KNbO3 Nanoparticles. RSC Adv. 2014, 4, 23344–23350. [Google Scholar] [CrossRef]
- Cheng, H.; Yang, N.; Lu, Q.; Zhang, Z.; Zhang, H. Syntheses and Properties of Metal Nanomaterials with Novel Crystal Phases. Adv. Mater. 2018, 30, 1707189. [Google Scholar] [CrossRef]
- Lee, I.; Morales, R.; Albiter, M.A.; Zaera, F. Synthesis of Heterogeneous Catalysts with Well Shaped Platinum Particles to Control Reaction Selectivity. Proc. Natl. Acad. Sci. USA 2008, 105, 15241–15426. [Google Scholar] [CrossRef]
- Collins, G.; Schmidt, M.; O’Dwyer, C.; Holmes Justin, D.; McGlacken Gerard, P. The Origin of Shape Sensitivity in Palladium-Catalyzed Suzuki–Miyaura Cross Coupling Reactions. Angew. Chem. Int. Ed. 2014, 53, 4142–4145. [Google Scholar] [CrossRef]
- Geng, G.; Chen, P.; Guan, B.; Jiang, L.; Xu, Z.; Di, D.; Tu, Z.; Hao, W.; Yi, Y.; Chen, C.; et al. Shape-Controlled Metal-Free Catalysts: Facet-Sensitive Catalytic Activity Induced by the Arrangement Pattern of Noncovalent Supramolecular Chains. ACS Nano 2017, 11, 4866–4876. [Google Scholar] [CrossRef]
- Sun, Y.-F.; Liu, S.-B.; Meng, F.-L.; Liu, J.-Y.; Jin, Z.; Kong, L.-T.; Liu, J.-H. Metal Oxide Nanostructures and Their Gas Sensing Properties: A Review. Sensors 2012, 12, 2610–2631. [Google Scholar] [CrossRef]
- Rao, X.; Su, X.; Yang, C.; Wang, J.; Zhen, X.; Ling, D. From Spindle-Like β-FeOOH Nanoparticles to α-Fe2O3 Polyhedral Crystals: Shape Evolution, Growth Mechanism and Gas Sensing Property. CrystEngComm 2013, 15, 7250–7256. [Google Scholar] [CrossRef]
- Jońca, J.; Ryzhikov, A.; Kahn, M.L.; Fajerwerg, K.; Chaudret, B.; Chapelle, A.; Menini, P.; Fau, P. Shape-Controlled ZnO Nanostructures for Gas Sensing Applications. Procedia Eng. 2014, 87, 907–910. [Google Scholar] [CrossRef][Green Version]
- Subbiah, R.; Veerapandian, M.; Yun, K.S. Nanoparticles: Functionalization and Multifunctional Applications in Biomedical Sciences. Curr. Med. Chem. 2010, 17, 4559–4577. [Google Scholar] [CrossRef]
- Mout, R.; Moyano, D.F.; Rana, S.; Rotello, V.M. Surface Functionalization of Nanoparticles for Nanomedicine. Chem. Soc. Rev. 2012, 41, 2539–2544. [Google Scholar] [CrossRef]
- Kolhatkar, A.G.; Jamison, A.C.; Litvinov, D.; Willson, R.C.; Lee, T.R. Tuning the Magnetic Properties of Nanoparticles. Int. J. Mol. Sci. 2013, 14, 15977–16009. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Wang, T. The Size and Shape Dependence of Ferromagnetism in Nanomagnets. J. Nanomater. 2012, 2012, 897023. [Google Scholar] [CrossRef]
- Cao, L.-F.; Xie, D.; Guo, M.-X.; Park, H.S.; Fujita, T. Size and Shape Effects on Curie Temperature of Ferromagnetic Nanoparticles. Trans. Nonferrous Met. Soc. China 2007, 17, 1451–1455. [Google Scholar] [CrossRef]
- Gratton, S.E.A.; Ropp, P.A.; Pohlhaus, P.D.; Luft, J.C.; Madden, V.J.; Napier, M.E.; DeSimone, J.M. The effect of particle design on cellular internalization pathways. Proc. Natl. Acad. Sci. USA 2008, 105, 11613–11618. [Google Scholar] [CrossRef]
- Hoffman, D.W.; Cahn, J.W. A Vector Thermodynamics for Anisotropic Surfaces: I. Fundamentals and Application to Plane Surface Junctions. Surf. Sci. 1972, 31, 368–388. [Google Scholar] [CrossRef]
- Cahn, J.L.; Hoffman, D.L. A Vector Thermodynamics for Anisotropic Surfaces—II. Curved and Faceted Surfaces. Acta Metall. 1974, 22, 1205–1214. [Google Scholar] [CrossRef]
- Dinghas, A. Über einen geometrischen Satz von Wulff für die Gleichgewichtsform von Kristallen. Z. Kristallogr. Cryst. Mater. 1943, 105, 304–314. [Google Scholar] [CrossRef]
- Angenent, S.; Gurtin, M.E. Multiphase Thermomechanics with Interfacial Structure 2. Evolution of an Isothermal Interface. Arch. Ration. Mech. Anal. 1989, 108, 323–391. [Google Scholar] [CrossRef]
- Yokoyama, E.; Sekerka, R.F. A Numerical Study of the Combined Effect of Anisotropic Surface Tension and Interface Kinetics on Pattern Formation during the Growth of Two-Dimensional Crystals. J. Cryst. Growth 1992, 125, 389–403. [Google Scholar] [CrossRef]
- Berg, W.F. Crystal Growth from Solutions. Proc. R. Soc. Lond. Ser. A 1938, 164, 79–95. [Google Scholar]
- Ham, F.S. Theory of Diffusion-Limited Precipitation. J. Phys. Chem. Solids 1958, 6, 335–351. [Google Scholar] [CrossRef]
- Burton, W.K.; Cabrera, N.; Frank, F.C. The Growth of Crystals and the Equilibrium Structure of Their Surfaces. Proc. R. Soc. Lond. Ser. A 1951, 243, 299–358. [Google Scholar] [CrossRef]
- Ly, T.; Wen, J.; Marks, L.D. Kinetic Growth Regimes of Hydrothermally Synthesized Potassium Tantalate Nanoparticles. Nano Lett. 2018, 18, 5186–5191. [Google Scholar] [CrossRef] [PubMed]
- Uecker, R.; Velickov, B.; Klimm, D.; Bertram, R.; Bernhagen, M.; Rabe, M.; Albrecht, M.; Fornari, R.; Schlom, D.G. Properties of Rare-Earth Scandate Single Crystals (Re=Nd−Dy). J. Cryst. Growth 2008, 310, 2649–2658. [Google Scholar] [CrossRef]
- Schneider, S.J.; Roth, R.S.; Waring, J.L. Solid state reactions involving oxides of trivalent cations. J. Res. Natl. Bur. Stand. A Phys. Chem. 1961, 65A, 345–374. [Google Scholar] [CrossRef]
- Badie, J.M.; Foex, M. Determination experimentale, calcul et prevision de certains diagrammes Sc2O3-Ln2O3. J. Solid State Chem. 1978, 26, 311–319. [Google Scholar] [CrossRef]
- Giaquinta, D.M.; zur Loye, H.-C. Structural Predictions in the ABO3 Phase Diagram. Chem. Mater. 1994, 6, 365–372. [Google Scholar] [CrossRef]
- Heeg, T.; Roeckerath, M.; Schubert, J.; Zander, W.; Buchal, C.; Chen, H.Y.; Jia, C.L.; Jia, Y.; Adamo, C.; Schlom, D.G. Epitaxially stabilized growth of orthorhombic LuScO3 thin films. Appl. Phys. Lett. 2007, 90, 192901. [Google Scholar] [CrossRef]
- Zhao, C.; Witters, T.; Brijs, B.; Bender, H.; Richard, O.; Caymax, M.; Heeg, T.; Schubert, J.; Afanas’ev, V.V.; Stesmans, A.; et al. Ternary Rare-Earth Metal Oxide High-k Layers on Silicon Oxide. Appl. Phys. Lett. 2005, 86, 132903. [Google Scholar] [CrossRef]
- Koirala, P.; Mizzi, C.A.; Marks, L.D. Direct Observation of Large Flexoelectric Bending at the Nanoscale in Lanthanide Scandates. Nano Lett. 2018, 18, 3850–3856. [Google Scholar] [CrossRef]
- Myllymaki, P.; Roeckerath, M.; Lopes, J.M.; Schubert, J.; Mizohata, K.; Putkonen, M.; Niinisto, L. Rare Earth Scandate Thin Films by Atomic Layer Deposition: Effect of the Rare Earth Cation Size. J. Mater. Chem. 2010, 20, 4207–4212. [Google Scholar] [CrossRef]
- Christen, H.M.; Jellison, G.E.; Ohkubo, I.; Huang, S.; Reeves, M.E.; Cicerrella, E.; Freeouf, J.L.; Jia, Y.; Schlom, D.G. Dielectric and Optical Properties of Epitaxial Rare-Earth Scandate Films and Their Crystallization Behavior. Appl. Phys. Lett. 2006, 88, 262906. [Google Scholar] [CrossRef]
- Iacopetti, S.; Shekhter, P.; Winter, R.; Tromm, T.C.U.; Schubert, J.; Eizenberg, M. The Asymmetric Band Structure and Electrical Behavior of the GdScO3/GaN System. J. Appl. Phys. 2017, 121, 205303. [Google Scholar] [CrossRef]
- Somorjai, G.A.; Li, Y. Introduction to Surface Chemistry and Catalysis; John Wiley & Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- Sanz-Pérez, E.S.; Murdock, C.R.; Didas, S.A.; Jones, C.W. Direct Capture of CO2 from Ambient Air. Chem. Rev. 2016, 116, 11840–11876. [Google Scholar] [CrossRef]
- Pozun, Z.D.; Tran, K.; Shi, A.; Smith, R.H.; Henkelman, G. Why Silver Nanoparticles Are Effective for Olefin/Paraffin Separations. J. Phys. Chem. C 2011, 115, 1811–1818. [Google Scholar] [CrossRef]
- Moeller, T.; Kremers, H.E. The Basicity Characteristics of Scandium, Yttrium, and the Rare Earth Elements. Chem. Rev. 1945, 37, 97–159. [Google Scholar] [CrossRef]
- Paull, R.J.; Mansley, Z.R.; Ly, T.; Marks, L.D.; Poeppelmeier, K.R. Synthesis of Gadolinium Scandate from a Hydroxide Hydrogel. Inorg. Chem. 2018, 57, 4104–4108. [Google Scholar] [CrossRef]
- Kozawa, T.; Yanagisawa, K.; Suzuki, Y. Water Vapor-Assisted Solid-State Reaction for the Synthesis of Nanocrystalline BaZrO3 powder. J. Ceram. Soc. Jpn. 2013, 121, 308–312. [Google Scholar] [CrossRef]
- Stampler, E.S.; Sheets, W.C.; Prellier, W.; Marks, T.J.; Poeppelmeier, K.R. Hydrothermal Synthesis of LnMnO3 (Ln = Ho-Lu and Y): Exploiting Amphoterism in Late Rare-Earth Oxides. J. Mater. Chem. 2009, 19, 4375–4381. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paull, R.J.; Ly, T.; Mansley, Z.R.; Poeppelmeier, K.R.; Marks, L.D. Controlled Two-Step Formation of Faceted Perovskite Rare-Earth Scandate Nanoparticles. Crystals 2019, 9, 218. https://doi.org/10.3390/cryst9040218
Paull RJ, Ly T, Mansley ZR, Poeppelmeier KR, Marks LD. Controlled Two-Step Formation of Faceted Perovskite Rare-Earth Scandate Nanoparticles. Crystals. 2019; 9(4):218. https://doi.org/10.3390/cryst9040218
Chicago/Turabian StylePaull, Ryan J., Tiffany Ly, Zachary R. Mansley, Kenneth R. Poeppelmeier, and Laurence D. Marks. 2019. "Controlled Two-Step Formation of Faceted Perovskite Rare-Earth Scandate Nanoparticles" Crystals 9, no. 4: 218. https://doi.org/10.3390/cryst9040218
APA StylePaull, R. J., Ly, T., Mansley, Z. R., Poeppelmeier, K. R., & Marks, L. D. (2019). Controlled Two-Step Formation of Faceted Perovskite Rare-Earth Scandate Nanoparticles. Crystals, 9(4), 218. https://doi.org/10.3390/cryst9040218




