Synthesis and Adsorbing Properties of Tabular {001} Calcite Crystals
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
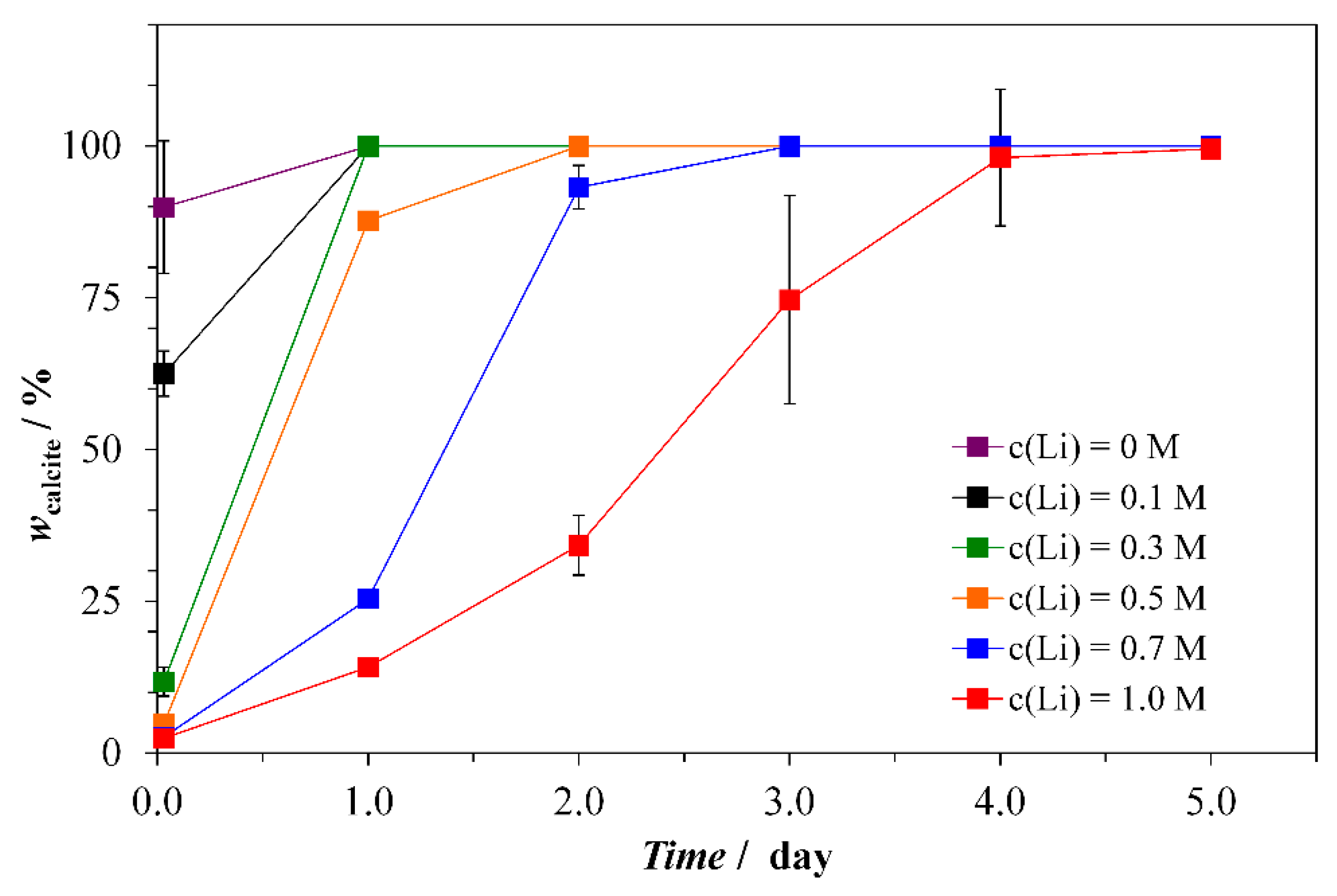
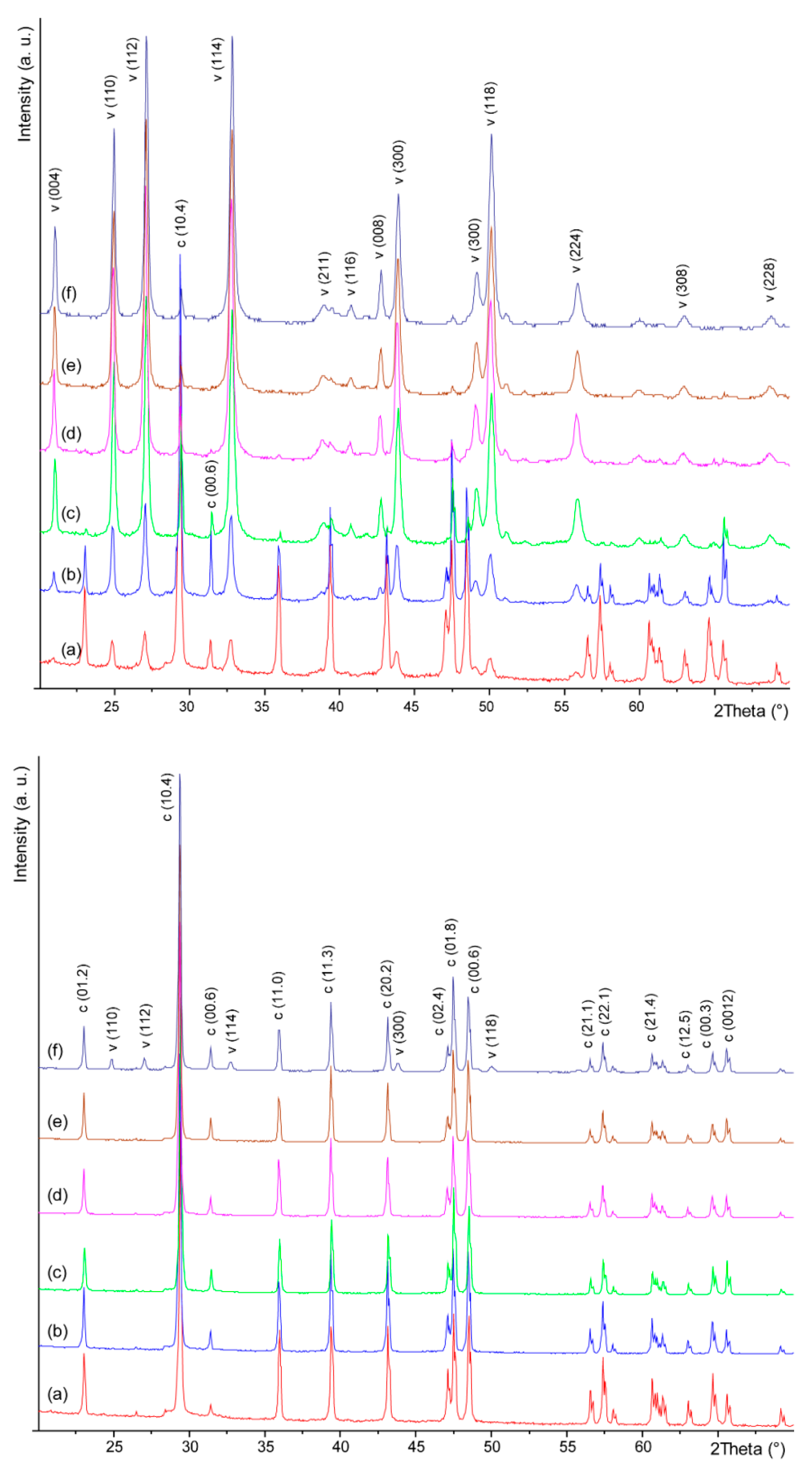
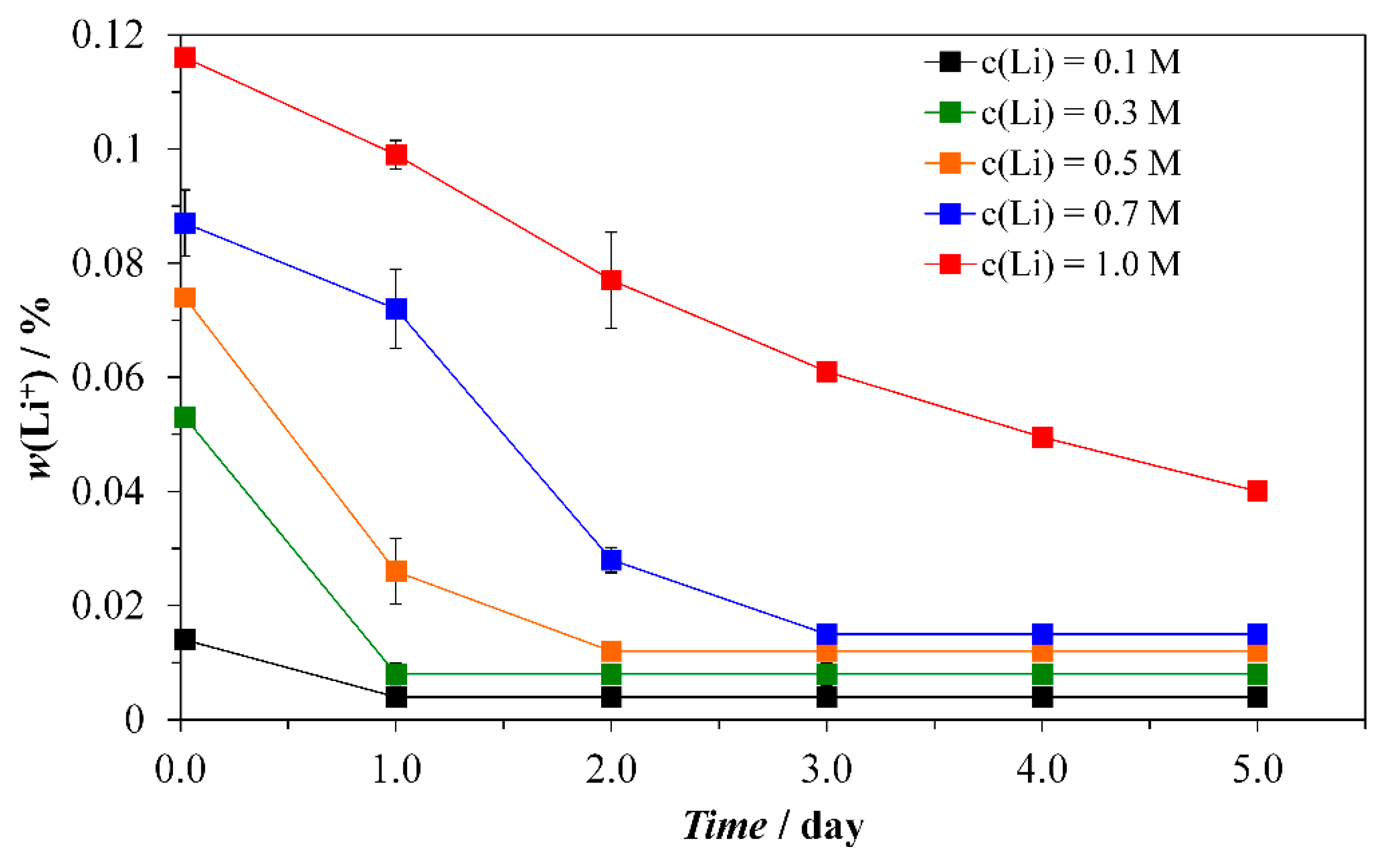
3.1. Synthesis and Characterization of Thin Tabular {00.1} Calcite Crystals
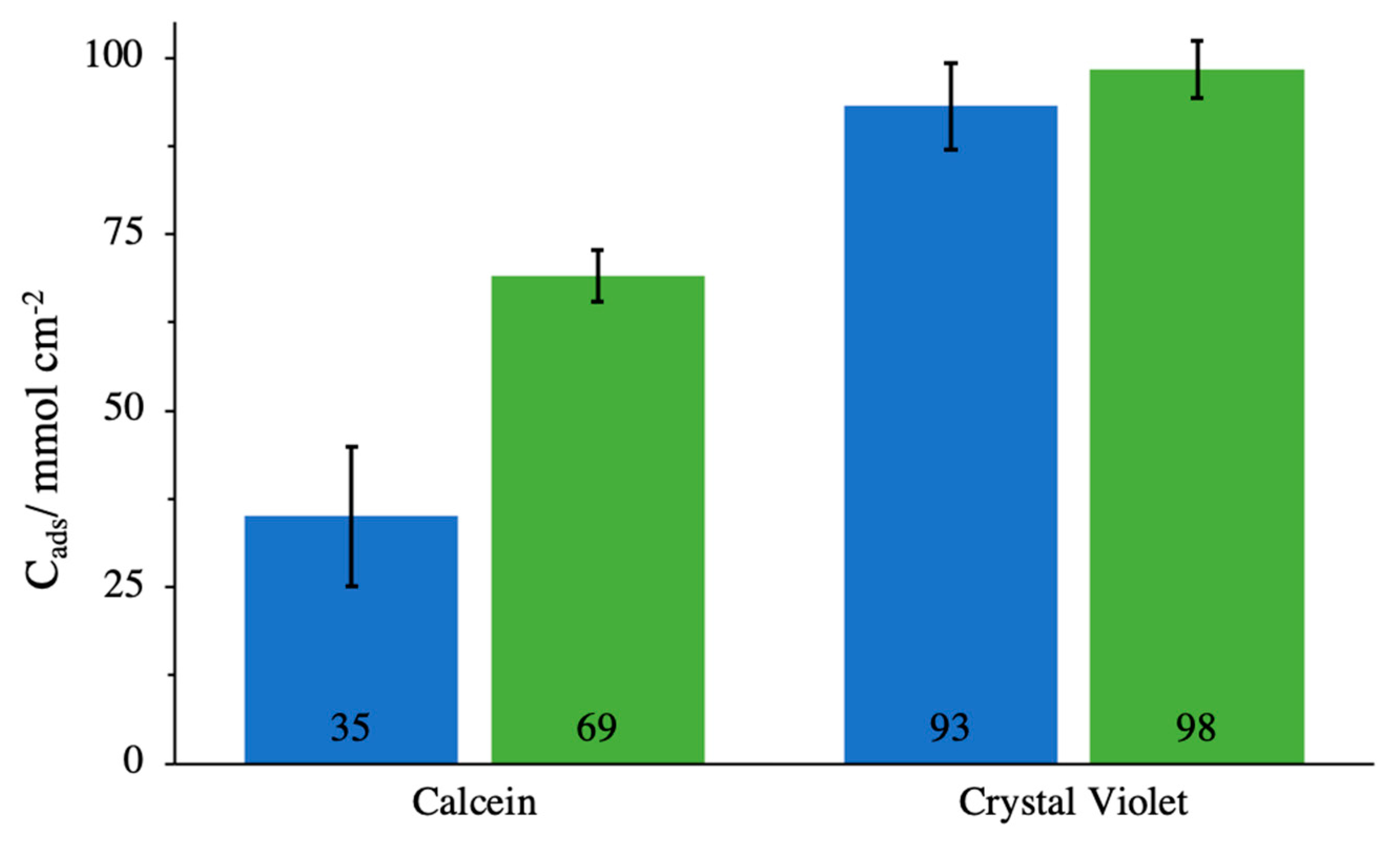
3.2. Adsorption of Model Molecules
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Magnabosco, G.; Giosia, M.D.; Polishchuk, I.; Weber, E.; Fermani, S.; Bottoni, A.; Zerbetto, F.; Pelicci, P.G.; Pokroy, B.; Rapino, S.; et al. Calcite Single Crystals as Hosts for Atomic-Scale Entrapment and Slow Release of Drugs. Adv. Healthc. Mater. 2015, 4, 1510–1516. [Google Scholar] [CrossRef] [PubMed]
- Nudelman, F.; Sommerdijk, N.A. Biomineralization as an inspiration for materials chemistry. Angew. Chem. Int. Ed. 2012, 51, 6582–6596. [Google Scholar] [CrossRef] [PubMed]
- Green, D.C.; Ihli, J.; Thornton, P.D.; Holden, M.A.; Marzec, B.; Kim, Y.Y.; Kulak, A.N.; Levenstein, M.A.; Tang, C.; Lynch, C.; et al. 3D visualization of additive occlusion and tunable full-spectrum fluorescence in calcite. Nat. Commun. 2016, 7, 13524. [Google Scholar] [CrossRef] [PubMed]
- Weiner, S.; Addadi, L. Crystallization pathways in biomineralization. Annu. Rev. Mater. Res. 2011, 41, 21–40. [Google Scholar] [CrossRef]
- Addadi, L.; Weiner, S. Interactions between acidic proteins and crystals: Stereochemical requirements in biomineralization. Proc. Natl. Acad. Sci. USA 1985, 82, 4110–4114. [Google Scholar] [CrossRef]
- Magnabosco, G.; Polishchuk, I.; Erez, J.; Fermani, S.; Pokroy, B.; Falini, G. Insights on the interaction of calcein with calcium carbonate and its implications in biomineralization studies. CrystEngComm 2018, 20, 4221–4224. [Google Scholar] [CrossRef]
- Ukrainczyk, M.; Stelling, J.; Vučak, M.; Neumann, T. Influence of etidronic acid and tartaric acid on the growth of different calcite morphologies. J. Cryst. Growth 2013, 369, 21–31. [Google Scholar] [CrossRef]
- Wang, L.; Ruiz-Agudo, E.; Putnis, C.V.; Putnis, A. Direct observations of the modification of calcite growth morphology by Li+through selectively stabilizing an energetically unfavourable face. CrystEngComm 2011, 13, 3962–3966. [Google Scholar] [CrossRef]
- Njegić-Džakula, B.; Falini, G.; Brečević, L.; Skoko, Ž.; Kralj, D. Effects of initial supersaturation on spontaneous precipitation of calcium carbonate in the presence of charged poly-l-amino acids. J. Colloid Interface Sci. 2010, 343, 553–563. [Google Scholar] [CrossRef]
- Lowenstam, H.A.; Weiner, S. On Biomineralization; Oxford University Press: Oxford, UK, 1989; ISBN 0195364198. [Google Scholar]
- Falini, G.; Fermani, S.; Goffredo, S. Coral biomineralization: A focus on intra-skeletal organic matrix and calcification. Semin. Cell Dev. Biol. 2015, 46, 17–26. [Google Scholar] [CrossRef]
- Aizenberg, J.; Tkachenko, A.; Weiner, S.; Addadi, L.; Hendler, G. Calcitic microlenses as part of the photoreceptor system in brittlestars. Nature 2001, 412, 819–822. [Google Scholar] [CrossRef] [PubMed]
- Addadi, L.; Raz, S.; Weiner, S. Taking advantage of disorder: Amorphous calcium carbonate and its roles in biomineralization. Adv. Mater. 2003, 15, 959–970. [Google Scholar] [CrossRef]
- Ukrainczyk, M.; Kontrec, J.; Kralj, D. Precipitation of different calcite crystal morphologies in the presence of sodium stearate. J. Colloid Interface Sci. 2009, 329, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Njegić-Džakula, B.; Brečević, L.; Falini, G.; Kralj, D. Calcite Crystal Growth Kinetics in the Presence of Charged Synthetic Polypeptides. Cryst. Growth Des. 2009, 9, 2425–2434. [Google Scholar] [CrossRef]
- Njegić-Džakula, B.; Brečević, L.; Falini, G.; Kralj, D. Kinetic Approach to Biomineralization: Interactions of Synthetic Polypeptides with Calcium Carbonate Polymorphs. Croat. Chem. Acta 2011, 84, 301–314. [Google Scholar] [CrossRef]
- Magnabosco, G.; Polishchuk, I.; Pokroy, B.; Rosenberg, R.; Cölfen, H.; Falini, G. Synthesis of calcium carbonate in trace water environments. Chem. Commun. 2017, 53, 4811–4814. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.J.; Dove, P.M.; Wasylenki, L.E.; De Yoreo, J.J. Morphological consequences of differential Mg2+incorporation at structurally distinct steps on calcite. Am. Mineral. 2004, 89, 714–720. [Google Scholar] [CrossRef]
- Rajam, S.; Mann, S. Selective stabilization of the (001) face of calcite in the presence of lithium. J. Chem. Soc. Chem. Commun. 1990, 0, 1789–1791. [Google Scholar] [CrossRef]
- Song, R.Q.; Cölfen, H. Additive controlled crystallization. CrystEngComm 2011, 13, 1249–1276. [Google Scholar] [CrossRef]
- Titiloye, J.O.; Parker, S.C.; Osguthorpe, D.J.; Mann, S. Predicting the influence of growth additives on the morphology of ionic crystals. J. Chem. Soc. Chem. Commun. 1991, 1494–1496. [Google Scholar] [CrossRef]
- Kitano, Y. The Behavior of Various Inorganic Ions in the Separation of Calcium Carbonate from a Bicarbonate Solution. Bull. Chem. Soc. Jpn. 1962, 35, 1973–1980. [Google Scholar] [CrossRef]
- Bruno, M.; Massaro, F.R.; Prencipe, M.; Aquilano, D. Surface reconstructions and relaxation effects in a centre-symmetrical crystal: The {00.1} form of calcite (CaCO3). CrystEngComm 2010, 12, 3626–3633. [Google Scholar] [CrossRef]
- Pastero, L.; Costa, E.; Bruno, M.; Rubbo, M.; Sgualdino, G.; Aquilano, D. Morphology of calcite (CaCO3) crystals growing from aqueous solutions in the presence of Li+ ions. Surface behavior of the {0001} form. Cryst. Growth Des. 2004, 4, 485–490. [Google Scholar] [CrossRef]
- Pastero, L.; Aquilano, D. CaCO3 (Calcite)/Li2CO3 (zabuyelite) anomalous mixed crystals. Sector zoning and growth mechanisms. Cryst. Growth Des. 2008, 8, 3451–3460. [Google Scholar] [CrossRef]
- Massaro, F.R.; Pastero, L.; Costa, E.; Sgualdino, G.; Aquilano, D. Single and twinned Li2CO3 crystals (zabuyelite) epitaxially grown on {0001} and {101̄4} forms of CaCO3 (calcite) crystals. Cryst. Growth Des. 2008, 8, 2041–2046. [Google Scholar] [CrossRef]
- Aquilano, D.; Pastero, L. Anomalous mixed crystals: A peculiar case of adsorption/absorption. Cryst. Res. Technol. 2013, 48, 819–839. [Google Scholar] [CrossRef]
- Pastero, L.; Aquilano, D.; Costa, E.; Rubbo, M. 2D epitaxy of lithium carbonate inducing growth mechanism transitions on {0 0 0 1}-K and {0 1 1̄ 8}-S forms of calcite crystals. J. Cryst. Growth 2005, 275, 1625–1630. [Google Scholar] [CrossRef]
- Titiloye, J.O.; Parker, S.C.; Mann, S. Atomistic simulation of calcite surfaces and the influence of growth additives on their morphology. J. Cryst. Growth 1993, 131, 533–545. [Google Scholar] [CrossRef]
- Bohr, J.; Wogelius, R.A.; Morris, P.M.; Stipp, S.L.S. Thickness and structure of the water film deposited from vapour on calcite surfaces. Geochim. Cosmochim. Acta 2010, 74, 5985–5999. [Google Scholar] [CrossRef]
- Sand, K.K.; Yang, M.; Makovicky, E.; Cooke, D.J.; Hassenkam, T.; Bechgaard, K.; Stipp, S.L.S. Binding of Ethanol on Calcite: The Role of the OH Bond and Its Relevance to Biomineralization. Langmuir 2010, 26, 15239–15247. [Google Scholar] [CrossRef] [PubMed]
- Bovet, N.; Yang, M.; Javadi, M.S.; Stipp, S.L.S. Interaction of alcohols with the calcite surface. Phys. Chem. Chem. Phys. 2015, 17, 3490–3496. [Google Scholar] [CrossRef] [PubMed]
- Aschauer, U.; Spagnoli, D.; Bowen, P.; Parker, S.C. Growth modification of seeded calcite using carboxylic acids: Atomistic simulations. J. Colloid Interface Sci. 2010, 346, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Mureşan, L.; Sinha, P.; Maroni, P.; Borkovec, M. Adsorption and surface-induced precipitation of poly(acrylic acid) on calcite revealed with atomic force microscopy. Colloids Surf. A Physicochem. Eng. Asp. 2011, 390, 225–230. [Google Scholar] [CrossRef]
- Hazen, R.M.; Filley, T.R.; Goodfriend, G.A. Selective adsorption of l- and d-amino acids on calcite: Implications for biochemical homochirality. Proc. Natl. Acad. Sci. USA 2001, 98, 5487–5490. [Google Scholar] [CrossRef] [PubMed]
- Orme, C.A.; Noy, A.; Wierzbicki, A.; Mcbride, M.T.; Grantham, M.; Teng, H.H.; Dove, P.M.; Deyoreo, J.J. Formation of chiral morphologies through selective binding of amino acids to calcite surface steps. Nature 2001, 411, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Wierzbicki, A.; Sikes, C.S.; Madura, J.D.; Drake, B. Atomic force microscopy and molecular modeling of protein and peptide binding to calcite. Calcif. Tissue Int. 1994, 54, 133–141. [Google Scholar] [CrossRef]
- Sonnenberg, L.; Luo, Y.; Schlaad, H.; Seitz, M.; Cölfen, H.; Gaub, H.E. Quantitative single molecule measurements on the interaction forces of poly(l-glutamic acid) with calcite crystals. J. Am. Chem. Soc. 2007, 129, 15364–15371. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Mark Rodger, P.; Harding, J.H.; Stipp, S.L.S. Molecular dynamics simulations of peptides on calcite surface. Mol. Simul. 2009, 35, 547–553. [Google Scholar] [CrossRef]
- Freeman, C.L.; Harding, J.H.; Quigley, D.; Rodger, P.M. Simulations of ovocleidin-17 binding to calcite surfaces and its implications for eggshell formation. J. Phys. Chem. C 2011, 115, 8175–8183. [Google Scholar] [CrossRef]
- Yang, M.; Stipp, S.L.S.; Harding, J. Biological control on calcite crystallization by polysaccharides. Cryst. Growth Des. 2008, 8, 4066–4074. [Google Scholar] [CrossRef]
- Dalas, E. The effect of ultrasonic field on calcium carbonate scale formation. J. Cryst. Growth 2001, 222, 287–292. [Google Scholar] [CrossRef]
- Nishida, I. Precipitation of calcium carbonate by ultrasonic irradiation. Ultrason. Sonochem. 2004, 11, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Berdonosov, S.S.; Znamenskaya, I.V.; Melikhov, I.V. Mechanism of the vaterite-to-calcite phase transition under sonication. Inorg. Mater. 2005, 41, 1308–1312. [Google Scholar] [CrossRef]
- Njegić Džakula, B.; Kontrec, J.; Ukrainczyk, M.; Sviben, S.; Kralj, D. Polymorphic composition and morphology of calcium carbonate as a function of ultrasonic irradiation. Cryst. Res. Technol. 2014, 49, 244–256. [Google Scholar] [CrossRef]
- Luo, J.; Kong, F.; Ma, X. Role of Aspartic Acid in the Synthesis of Spherical Vaterite by the Ca(OH)2–CO2 Reaction. Cryst. Growth Des. 2016, 16, 728–736. [Google Scholar] [CrossRef]
- Arvidson, R.S.; Ertan, I.E.; Amonette, J.E.; Luttge, A. Variation in calcite dissolution rates: A fundamental problem? Geochim. Cosmochim. Acta 2003, 67, 1623–1634. [Google Scholar] [CrossRef]
- De Giudici, G. Surface control vs. diffusion control during calcite dissolution: Dependence of step-edge velocity upon solution pH. Am. Mineral. 2002, 87, 1279–1285. [Google Scholar] [CrossRef]
- Beck, R.; Andreassen, J.P. Spherulitic growth of calcium carbonate. Cryst. Growth Des. 2010, 10, 2934–2947. [Google Scholar] [CrossRef]
- Andreassen, J.P. Formation mechanism and morphology in precipitation of vaterite—Nano-aggregation or crystal growth? J. Cryst. Growth 2005, 274, 256–264. [Google Scholar] [CrossRef]
- Atun, G.; Acar, E.T. Competitive adsorption of basic dyes onto calcite in single and binary component systems. Sep. Sci. Technol. 2010, 45, 1471–1481. [Google Scholar] [CrossRef]


| c(Li+)/mol·dm−3 | w(calcite)/wt % | Particle Size */μm | Aspect Ratio # | I(10.4)/I(00.1) | w(Li)/wt % |
|---|---|---|---|---|---|
| 0 | 100 | 9 | - | 68 | 0 |
| 0.1 | 100 | 11/27 | 11:1 27:1 | 39 | 0.004 ± 0.00017 |
| 0.3 | 100 | 12/32 | 12:1 32:1 | 25 | 0.008 ± 0.00011 |
| 0.5 | 100 | 23/43 | 23:1 43:1 | 30 | 0.012 ± 0.00015 |
| 0.7 | 100 | 23/40 | 23:1 40:1 | 23 | 0.015 ± 0.00023 |
| 1.0 | 98 ± 0.055 | 30/48 | 30:1 48:1 | 21 | 0.049 ± 0.00037 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matijaković, N.; Magnabosco, G.; Scarpino, F.; Fermani, S.; Falini, G.; Kralj, D. Synthesis and Adsorbing Properties of Tabular {001} Calcite Crystals. Crystals 2019, 9, 16. https://doi.org/10.3390/cryst9010016
Matijaković N, Magnabosco G, Scarpino F, Fermani S, Falini G, Kralj D. Synthesis and Adsorbing Properties of Tabular {001} Calcite Crystals. Crystals. 2019; 9(1):16. https://doi.org/10.3390/cryst9010016
Chicago/Turabian StyleMatijaković, Nives, Giulia Magnabosco, Francesco Scarpino, Simona Fermani, Giuseppe Falini, and Damir Kralj. 2019. "Synthesis and Adsorbing Properties of Tabular {001} Calcite Crystals" Crystals 9, no. 1: 16. https://doi.org/10.3390/cryst9010016
APA StyleMatijaković, N., Magnabosco, G., Scarpino, F., Fermani, S., Falini, G., & Kralj, D. (2019). Synthesis and Adsorbing Properties of Tabular {001} Calcite Crystals. Crystals, 9(1), 16. https://doi.org/10.3390/cryst9010016








