Does Thermosalient Effect Have to Concur with a Polymorphic Phase Transition? The Case of Methscopolamine Bromide
Abstract
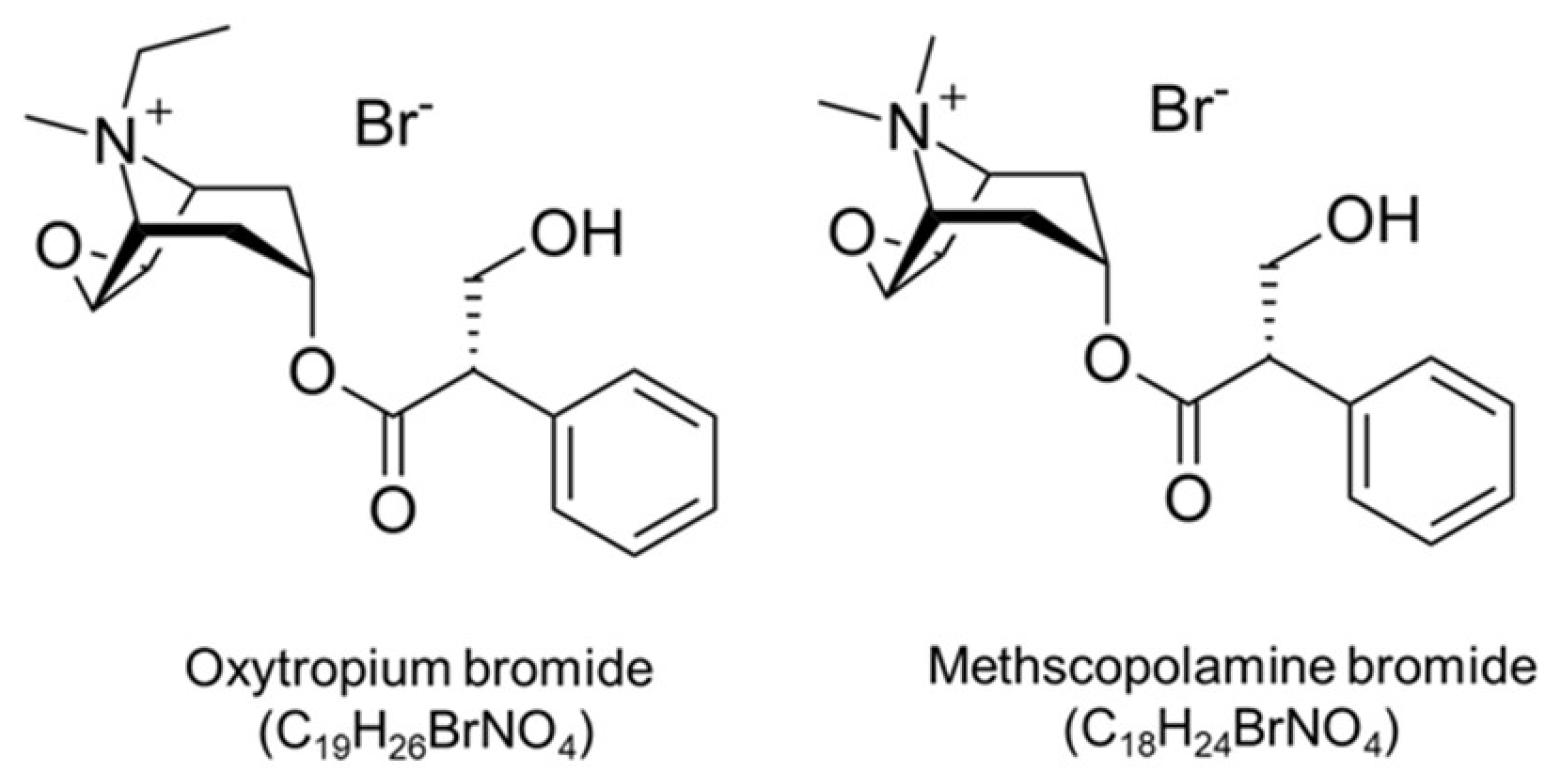
1. Introduction
2. Materials and Methods
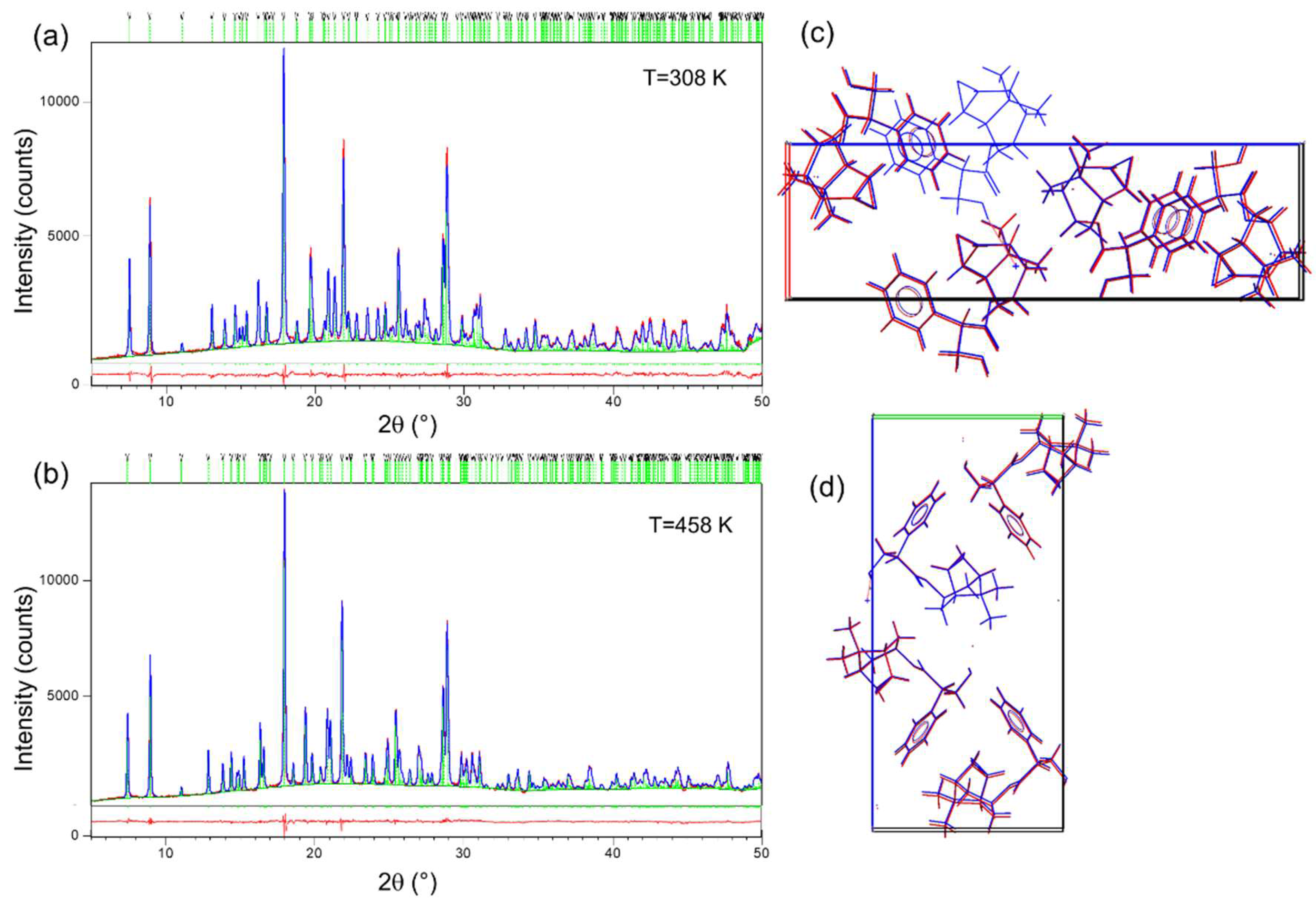
2.1. X-Ray Powder Diffraction (XRPD)
2.2. Thermal Analysis
2.3. Hot-Stage Microscopy
3. Results and Discussion
3.1. Hot-Stage Microscopy and Thermal Analysis
- Jumping (in terms of number of crystal that jumped, their frequency, or strength of the jumps) did not depend on the heating/cooling rate.
- Jumping did not depend on the size, shape, or orientation of the crystals. As expected, jumps of the smaller crystals were more forceful, whereas the more massive crystals would only slightly move or turn over to another facet.
- The number of crystals that jumped decreased drastically with consecutive heating/cooling runs. For example, if 10 crystals jumped during the first heating run, only 2–3 would jump in the second heating run.
- Crystals continued to jump sporadically when temperature was maintained for some time within the jumping temperature interval between 323 K and 333 K. Time period of jumping depended on the temperature. At 323 K, crystals continued to jump for 10 minutes, whereas the jumps ceased after 1–2 minutes at 343 K.
- No breaking or cracking of the crystals were observed during the jumping.
- Overall, the jumps of scopolamine bromide crystals were less energetic compared to the crystals of oxitropium bromide.
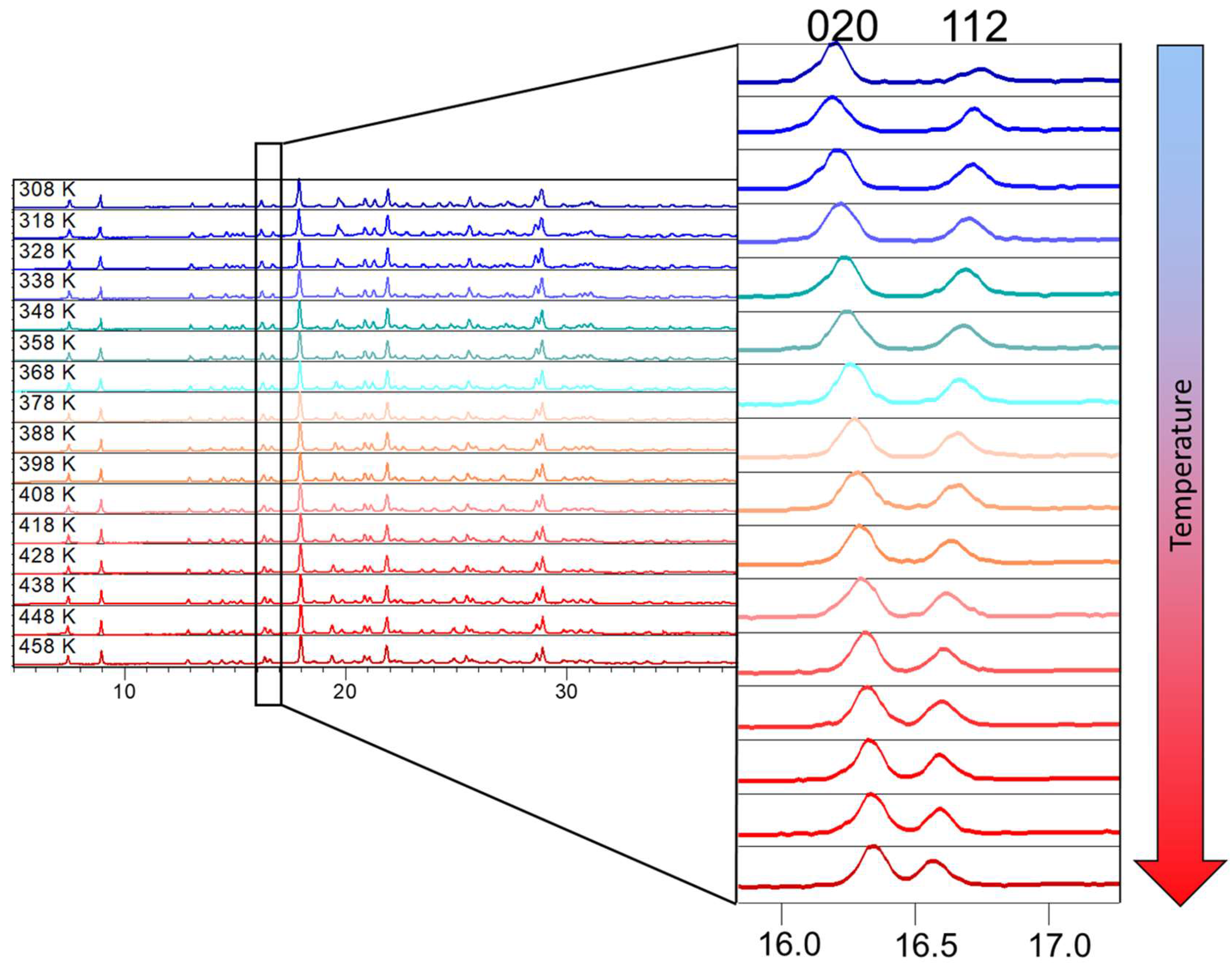
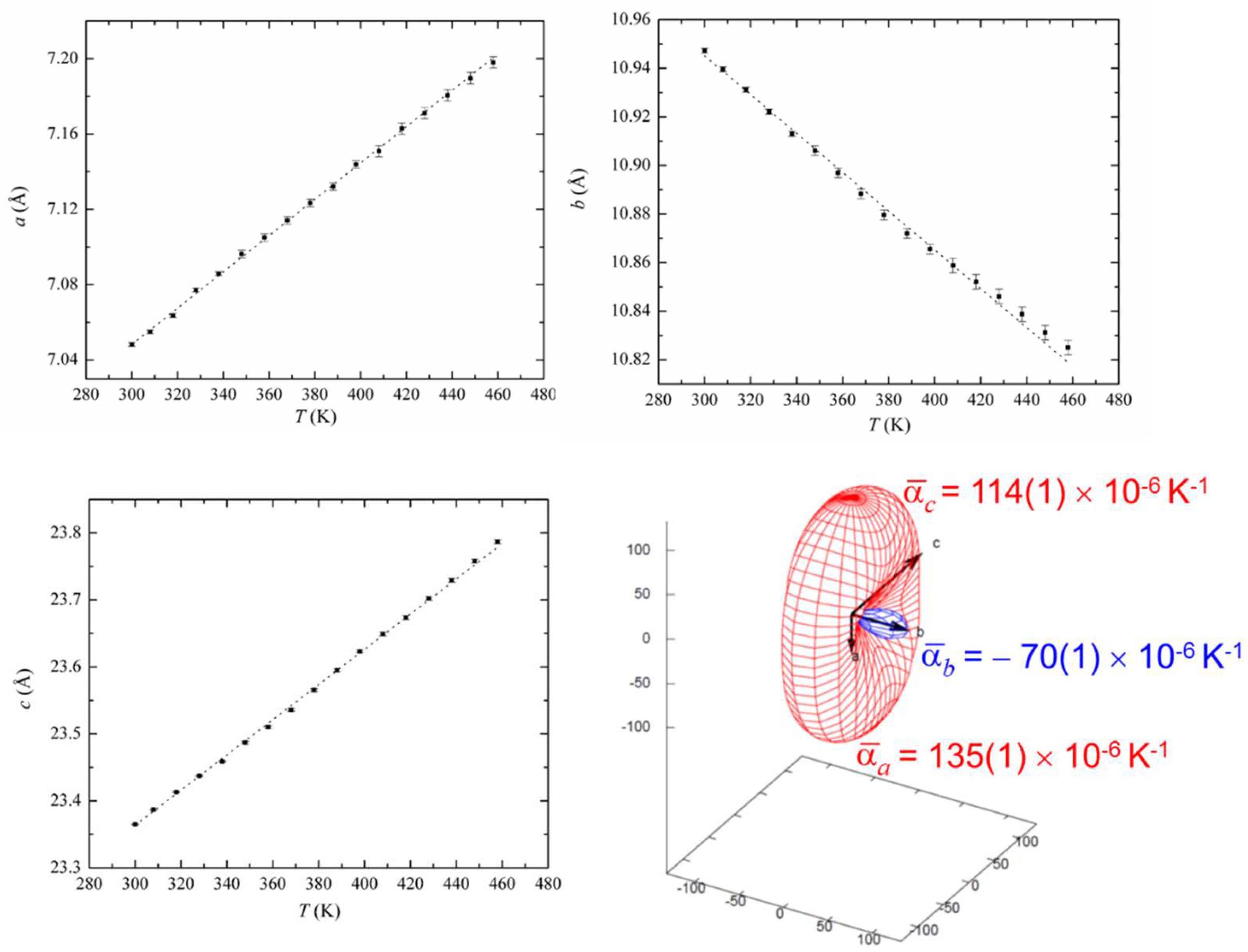
3.2. In Situ Variable Temperature X-ray Powder Diffraction (VT XRPD)
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nath, N.K.; Panda, M.K.; Sahoo, S.C.; Naumov, P. Thermally induced and photoinduced mechanical effects in molecular single crystals—A revival. Cryst. Eng. Comm. 2014, 16, 1850. [Google Scholar] [CrossRef]
- Karothu, D.P.; Weston, J.; Desta, I.T.; Naumov, P. Shape-memory and self-healing effects in mechanosalient molecular crystals. J. Am. Chem. Soc. 2016, 138, 13298–13306. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, S.C.; Sinha, S.B.; Kiran, M.S.R.N.; Ramamurty, U.; Dericioglu, A.F.; Reddy, C.M.; Naumov, P. Kinematic and mechanical profile of the self-actuation of thermosalient crystal twins of 1,2,4,5-tetrabromobenzene: A molecular crystalline analogue of a bimetallic strip. J. Am. Chem. Soc. 2013, 135, 13843–13850. [Google Scholar] [CrossRef] [PubMed]
- Commins, P.; Desta, I.T.; Karothu, D.P.; Panda, M.K.; Naumov, P. Crystals on the move: Mechanical effects in dynamic solids. Chem. Commun. 2016, 52, 13941–13954. [Google Scholar] [CrossRef] [PubMed]
- Panda, M.K.; Runčevski, T.; Husain, A.; Dinnebier, R.E.; Naumov, P. Perpetually self-propelling chiral single crystals. J. Am. Chem. Soc. 2015, 137, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Panda, M.K.; Runčevski, T.; Chandra Sahoo, S.; Belik, A.A.; Nath, N.K.; Dinnebier, R.E.; Naumov, P. Colossal positive and negative thermal expansion and thermosalient effect in a pentamorphic organometallic martensite. Nat. Commun. 2014, 5, 4811. [Google Scholar] [CrossRef] [PubMed]
- Panda, M.K.; Centore, R.; Causà, M.; Tuzi, A.; Borbone, F.; Naumov, P. Strong and anomalous thermal expansion precedes the thermosalient effect in dynamic molecular crystals. Sci. Rep. 2016, 6, 29610. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, S.C.; Giannini, M.; Carpenter, M.A.; Zema, M. Cooperative Jahn-Teller effect and the role of strain in the tetragonal-to-cubic phase transition in MgxCu1−xCr2O4. IUCrJ 2016, 3, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, Y.; Itoh, Y.; Aida, T. Jumping crystals of pyrene tweezers: Crystal-to-Crystal transition involving π/π-to-CH/π assembly mode switching. Chem. Asian J. 2017, 12, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Takeda, T.; Akutagawa, T. Anisotropic dissociation of π-π stacking and flipping-motion-induced crystal jumping in alkylacridones and their dicyanomethylene derivatives. Chem. Eur. J. 2016, 22, 7763–7770. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, S.; Gon, M.; Tanaka, K.; Chujo, Y. A flexible, fused, azomethine–boron complex: Thermochromic luminescence and thermosalient behavior in structural transitions between crystalline polymorphs. Chem. Eur. J. 2017, 23, 11827–11833. [Google Scholar] [CrossRef] [PubMed]
- Lončarić, I.; Popović, J.; Despoja, V.; Burazer, S.; Grgičević, I.; Popović, D.; Skoko, Ž. Reversible thermosalient effect of N′-2-propylidene-4- hydroxybenzohydrazide accompanied by an immense negative compressibility: Structural and theoretical arguments aiming toward the elucidation of jumping phenomenon. Cryst. Growth Des. 2017, 17, 4445–4453. [Google Scholar] [CrossRef]
- Khalil, A.; Ahmed, E.; Naumov, P. Metal-coated thermosalient crystals as electrical fuses. Chem. Commun. 2017, 60, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Skoko, Ž.; Zamir, S.; Naumov, P.; Bernstein, J. The thermosalient phenomenon. “Jumping crystals” crystal chemistry of the anticholinergic agent oxitropium bromide. J. Am. Chem. Soc. 2010, 132, 14191–14202. [Google Scholar] [CrossRef] [PubMed]
- Glaser, R.; Shiftan, D.; Drouin, M. The solid-state structures of (–) -scopolamine free base, (–) -scopolamine methobromide, the pseudopolymorphic forms of (–) -scopolamine hydrochloride anhydrate and 1.66hydrate. Can. J. Chem. 2000, 78, 212–223. [Google Scholar] [CrossRef]
- Cliffe, M.J.; Goodwin, A.L. PASCal: A principal axis strain calculator for thermal expansion and compressibility determination. J. Appl. Crystallogr. 2012, 45, 1321–1329. [Google Scholar] [CrossRef]
- Zakharov, B.A.; Michalchuk, A.A.L.; Morrison, C.A.; Boldyreva, E.V. Anisotropic lattice softening near the structural phase transition in the thermosalient crystal 1,2,4,5-tetrabromobenzene. Phys. Chem. Chem. Phys. 2018, 20, 8523–8532. [Google Scholar] [CrossRef] [PubMed]
- Birkedal, H.; Schwarzenbach, D. Observation of uniaxial negative thermal expansion in an organic crystal. Angew. Chem. Int. Ed. 2002, 41, 754–756. [Google Scholar] [CrossRef]
- Krishnan, R.S.; Srinivasan, R.; Devanarayanan, S. Thermal Expansion of Crystals; Pamplin, B.R., Ed.; Pergamon Press: Oxford, UK, 1979. [Google Scholar]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klaser, T.; Popović, J.; Fernandes, J.A.; Tarantino, S.C.; Zema, M.; Skoko, Ž. Does Thermosalient Effect Have to Concur with a Polymorphic Phase Transition? The Case of Methscopolamine Bromide. Crystals 2018, 8, 301. https://doi.org/10.3390/cryst8070301
Klaser T, Popović J, Fernandes JA, Tarantino SC, Zema M, Skoko Ž. Does Thermosalient Effect Have to Concur with a Polymorphic Phase Transition? The Case of Methscopolamine Bromide. Crystals. 2018; 8(7):301. https://doi.org/10.3390/cryst8070301
Chicago/Turabian StyleKlaser, Teodoro, Jasminka Popović, José A. Fernandes, Serena C. Tarantino, Michele Zema, and Željko Skoko. 2018. "Does Thermosalient Effect Have to Concur with a Polymorphic Phase Transition? The Case of Methscopolamine Bromide" Crystals 8, no. 7: 301. https://doi.org/10.3390/cryst8070301
APA StyleKlaser, T., Popović, J., Fernandes, J. A., Tarantino, S. C., Zema, M., & Skoko, Ž. (2018). Does Thermosalient Effect Have to Concur with a Polymorphic Phase Transition? The Case of Methscopolamine Bromide. Crystals, 8(7), 301. https://doi.org/10.3390/cryst8070301






