Structural Transformations in Crystals Induced by Radiation and Pressure. Part 7. Molecular and Crystal Geometries as Factors Deciding about Photochemical Reactivity under Ambient and High Pressures
Abstract
1. Introduction
- -
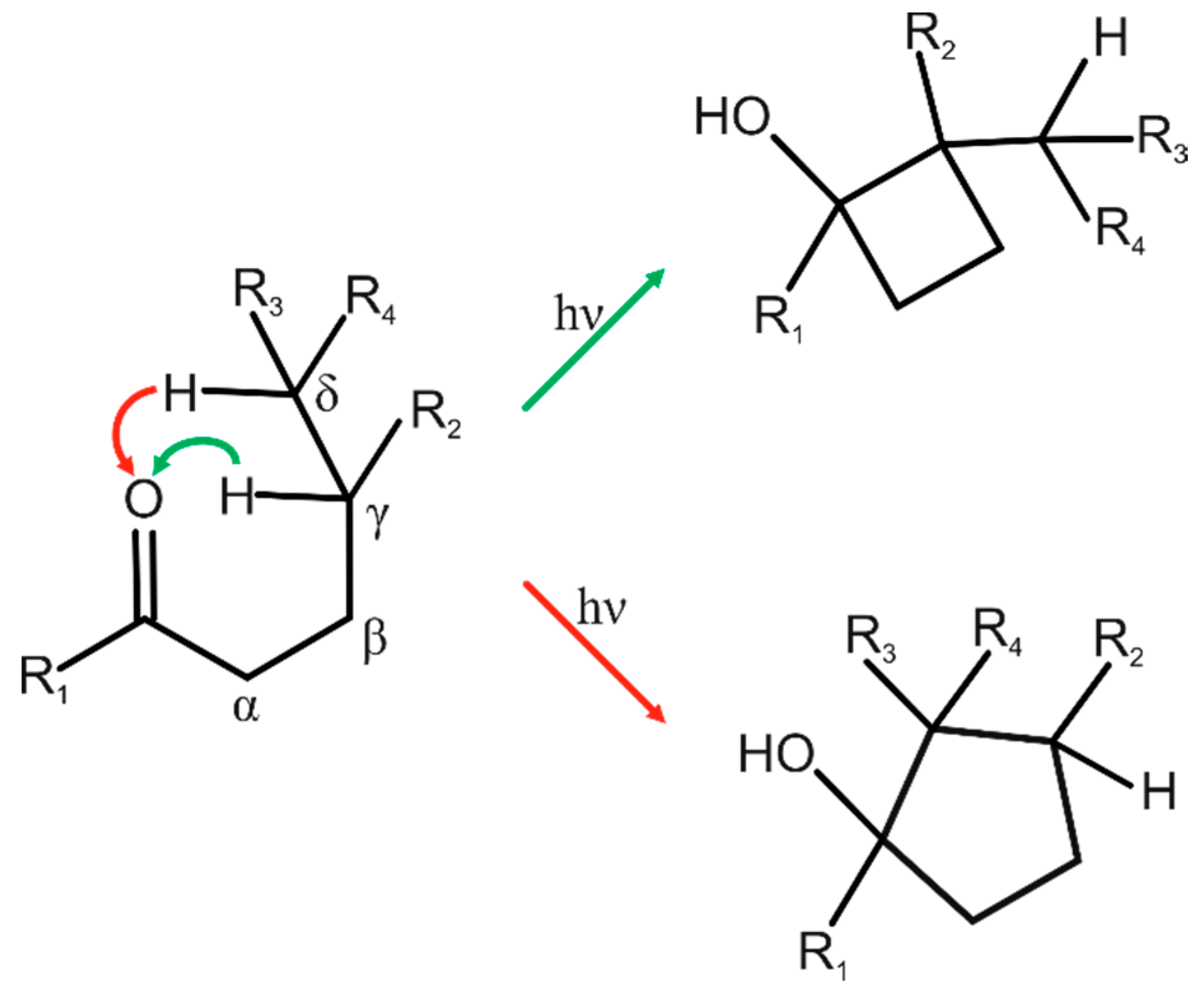
- the distance between the γ-hydrogen atom and the carbonyl oxygen atom (d), with the ideal value less than 2.7 Å;
- -
- the distance between the reacting carbon atoms (D), with the ideal value less than 3.2 Å;
- -
- the C=O∙∙∙H angle between the carbonyl group and the γ-hydrogen atom (Δ), with the ideal values in the range 90°–120°;
- -
- the C–H∙∙∙O angle formed by the γ-carbon, γ-hydrogen, and carbonyl oxygen (θ), with the ideal value 180°;
- -
- the torsion angle describing the deviation of the γ-hydrogen from the plane of the carbonyl group (ω), with the ideal value 0°.
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ihmels, H.; Scheffer, J.R. The Norrish type II reaction in the crystalline state: Toward a better understanding of the geometric requirements for γ-hydrogen atom abstraction. Tetrahedron 1999, 55, 885–907. [Google Scholar] [CrossRef]
- Xia, W.; Scheffer, J.R.; Botoshansky, M.; Kaftory, M. Photochemistry of 1-Isopropylcycloalkyl Aryl Ketones: Ring Size Effects, Medium Effects, and Asymmetric Induction. Org. Lett. 2005, 7, 1315–1318. [Google Scholar] [CrossRef] [PubMed]
- Koshima, H.; Ide, Y.; Fukano, M.; Fujii, K.; Uekusa, H. Single-crystal-to-single-crystal photocyclization of 4-(2,4,6-triisopropylbenzoyl)benzoic acid in the salt crystal with (S)-phenylethylamine. Tetrahedron Lett. 2008, 49, 4346–4348. [Google Scholar] [CrossRef]
- Koshima, H.; Kawanishi, H.; Nagano, M.; Yu, H.; Shiro, M.; Hosoya, T.; Uekusa, H.; Ohashi, Y. Absolute Asymmetric Photocyclization of Isopropylbenzophenone Derivatives Using a Cocrystal Approach Involving Single-Crystal-to-Single-Crystal Transformation. J. Org. Chem. 2005, 70, 4490–4497. [Google Scholar] [CrossRef] [PubMed]
- Cheung, E.; Rademacher, K.; Scheffer, J.R.; Trotter, J. An Investigation of the Solid-State Photochemistry of α-Mesitylacetophenone Derivatives: Asymmetric Induction Studies and Crystal Structure–Reactivity Relationships. Tetrahedron 2000, 56, 6739–6751. [Google Scholar] [CrossRef]
- Boldyreva, E.V. The concept of the ‘reaction cavity’: A link between solution and solid-state chemistry. Solid State Ion. 1997, 101–103, 843–849. [Google Scholar] [CrossRef]
- Boldyreva, E.V. Crystal-structure aspects of solid-state innersphere isomerization in nitro(nitrito)pentaamminecobalt(III) complexes. Russ. J. Coord. Chem. 2001, 27, 297–323. [Google Scholar] [CrossRef]
- Tapilin, V.M.; Bulgakov, N.N.; Chupakhin, A.P.; Politov, A.A.; Druganov, A.G. On mechanochemical dimerization of anthracene. Different possible reaction pathways. J. Struct. Chem. 2010, 51, 635–641. [Google Scholar] [CrossRef]
- Moorthy, J.N.; Venkatakrishnan, P.; Savitha, G.; Weiss, R.G. Cis → trans and trans → cis isomerizations of styrylcoumarins in the solid state. Importance of the location of free volume in crystal lattices. Photochem. Photobiol. Sci. 2006, 5, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Sreevidya, T.V.; Cao, D.; Lavy, T.; Botoshansky, M.; Kaftory, M. Unexpected Molecular Flip in Solid-State Photodimerization. Cryst. Growth Des. 2013, 13, 936–941. [Google Scholar] [CrossRef]
- Zheng, S.; Wang, Y.; Yu, Z.; Lin, Q.; Coppens, P. Direct Observation of a Photoinduced Nonstabilized Nitrile Imine Structure in the Solid State. J. Am. Chem. Soc. 2009, 131, 18036–18037. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, S.; Ito, Y.; Hosomi, H.; Ohba, S. Structures and Photoreactivities of 2,4,6-Triisopropylbenzophenones. Acta Crystallogr. B 1998, 54, 895–906. [Google Scholar] [CrossRef]
- Konieczny, K.; Bąkowicz, J.; Galica, T.; Siedlecka, R.; Turowska-Tyrk, I. The reasons for the photochemical behaviour of propylamine 4-(2,4,6-triisopropylbenzoyl)benzoate during the Norrish–Yang reaction. CrystEngComm 2017, 19, 3044–3050. [Google Scholar] [CrossRef]
- Konieczny, K.; Bąkowicz, J.; Siedlecka, R.; Galica, T.; Turowska-Tyrk, I. Photoinduced Structural Changes as the Factor Influencing the Direction of the Photochemical Reaction in the Crystal. Cryst. Growth Des. 2017, 17, 1347–1352. [Google Scholar] [CrossRef]
- Boehler, R. New diamond cell for single-crystal X-ray diffraction. Rev. Sci. Instrum. 2006, 77, 115103. [Google Scholar] [CrossRef]
- Angel, R.J.; Allan, D.R.; Miletich, R.; Finger, L.W. The Use of Quartz as an Internal Pressure Standard in High-Pressure Crystallography. J. Appl. Cryst. 1997, 30, 461–466. [Google Scholar] [CrossRef]
- Enkelmann, V.; Wegner, G.; Novak, K.; Wagener, K.B. Single-Crystal-to-Single-Crystal Photodimerization of Cinnamic Acid. J. Am. Chem. Soc. 1993, 115, 10390–10391. [Google Scholar] [CrossRef]
- Novak, K.; Enkelmann, V.; Wegner, G.; Wagener, K.B. Crystallographic Study of a Single Crystal to Single Crystal Photodimerization and Its Thermal Reverse Reaction. Angew. Chem. Int. Ed. Engl. 1993, 32, 1614–1616. [Google Scholar] [CrossRef]
- Rigaku Oxford Difraction. CrysAlisPro; Rigaku Oxford Diffraction: Wrocław, Poland, 2016. [Google Scholar]
- Bruker Corporation. APEX2, SAINT, Bruker Corporation: Madison, WI, USA, 2012.
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. A 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Farrugia, L.J. WinGX and ORTEP for Windows: An update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Macrae, C.F.; Bruno, J.A.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; van de Streek, J.; Wood, P.A. Mercury CSD 2.0—New features for the visualization and investigation of crystal structures. J. Appl. Cryst. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer17, University of Western Australia: Perth, Australia, 2017.
- Spackamn, M.A.; McKinnon, J.J. Fingerprinting intermolecular interactions in molecular crystals. CrystEngComm 2002, 4, 378–392. [Google Scholar] [CrossRef]
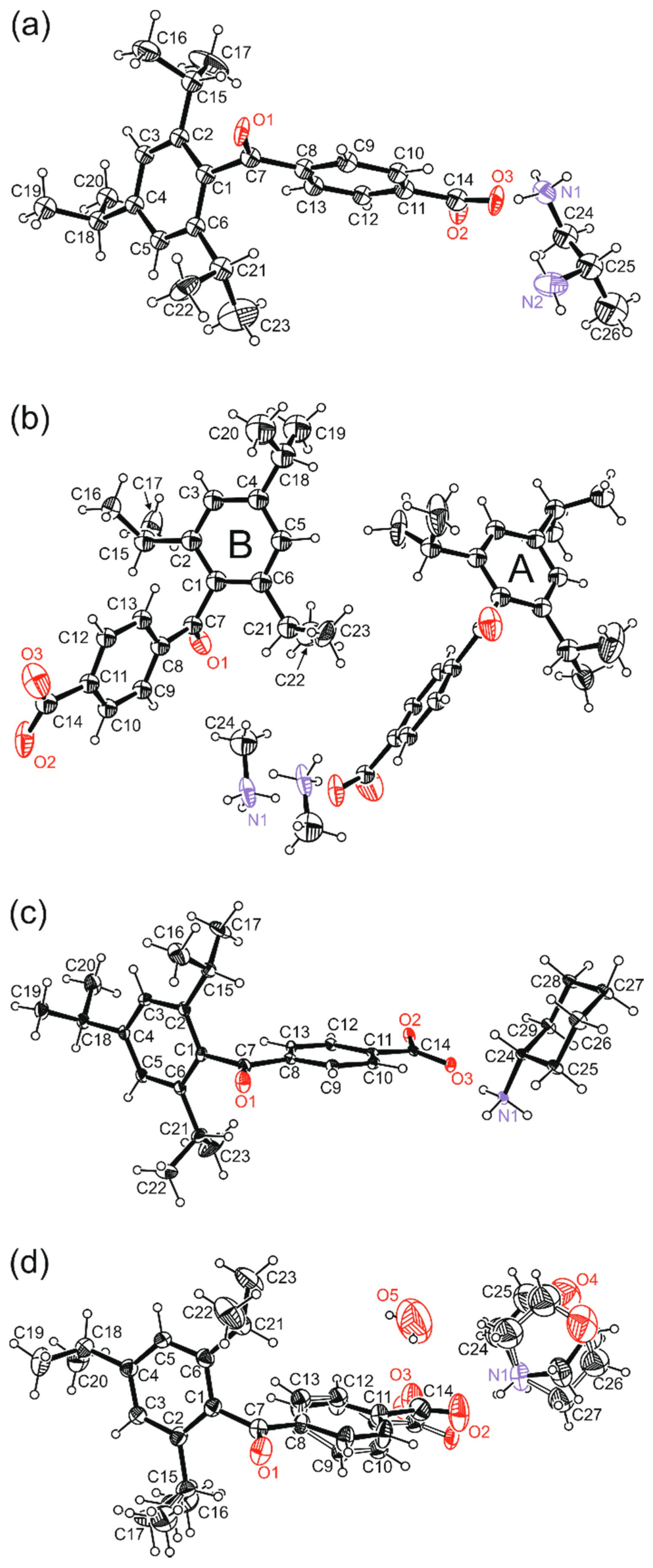
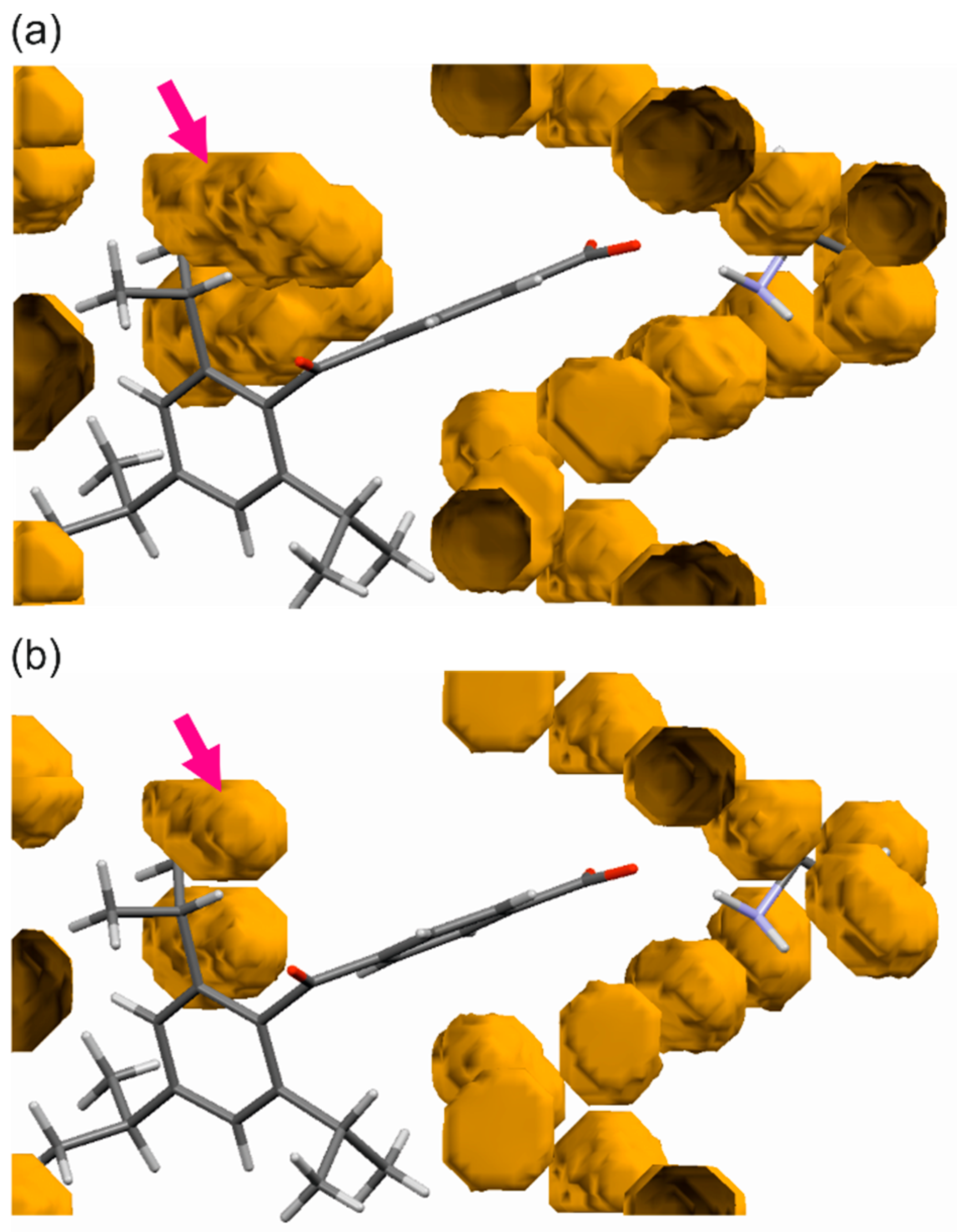
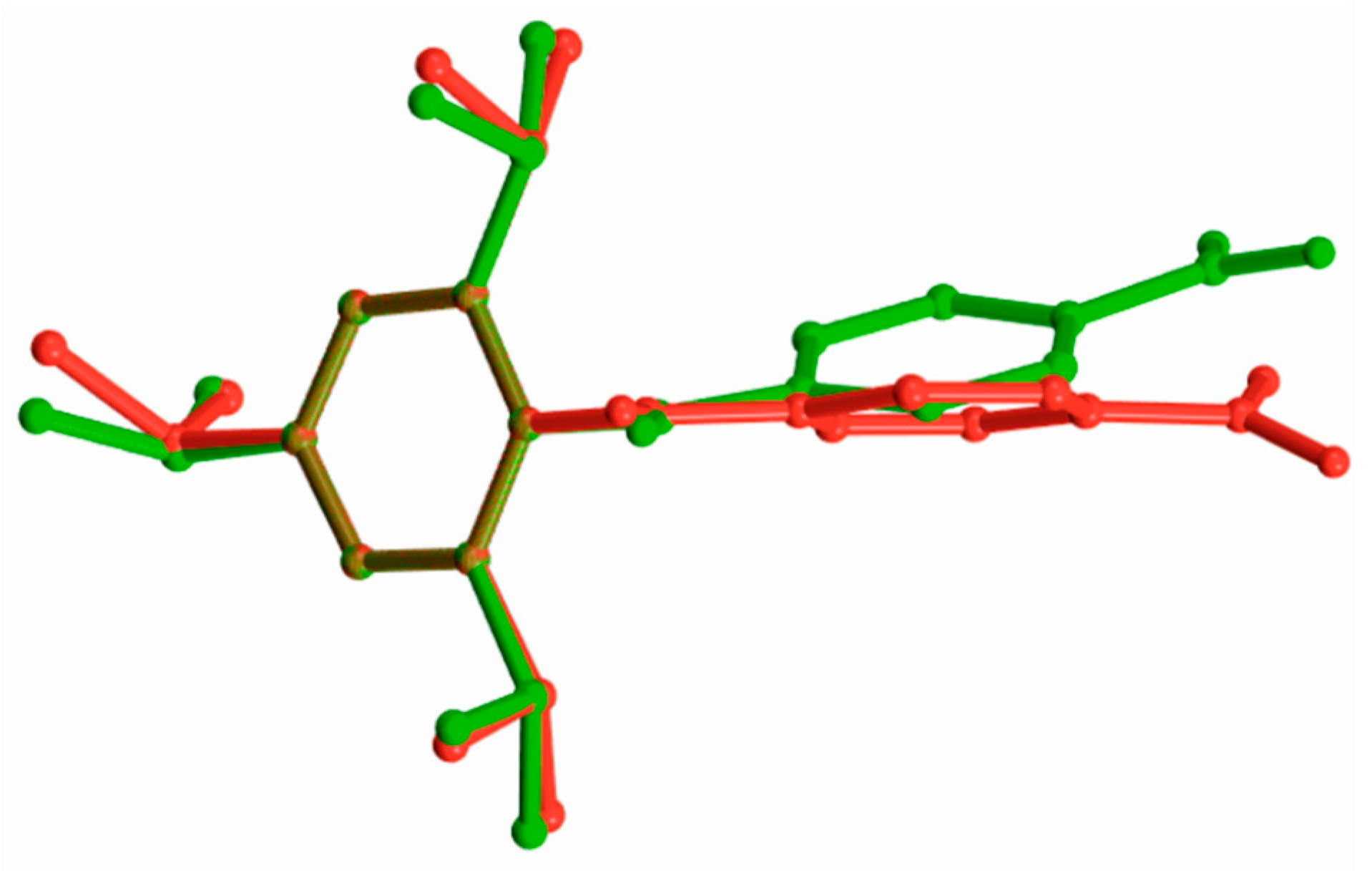
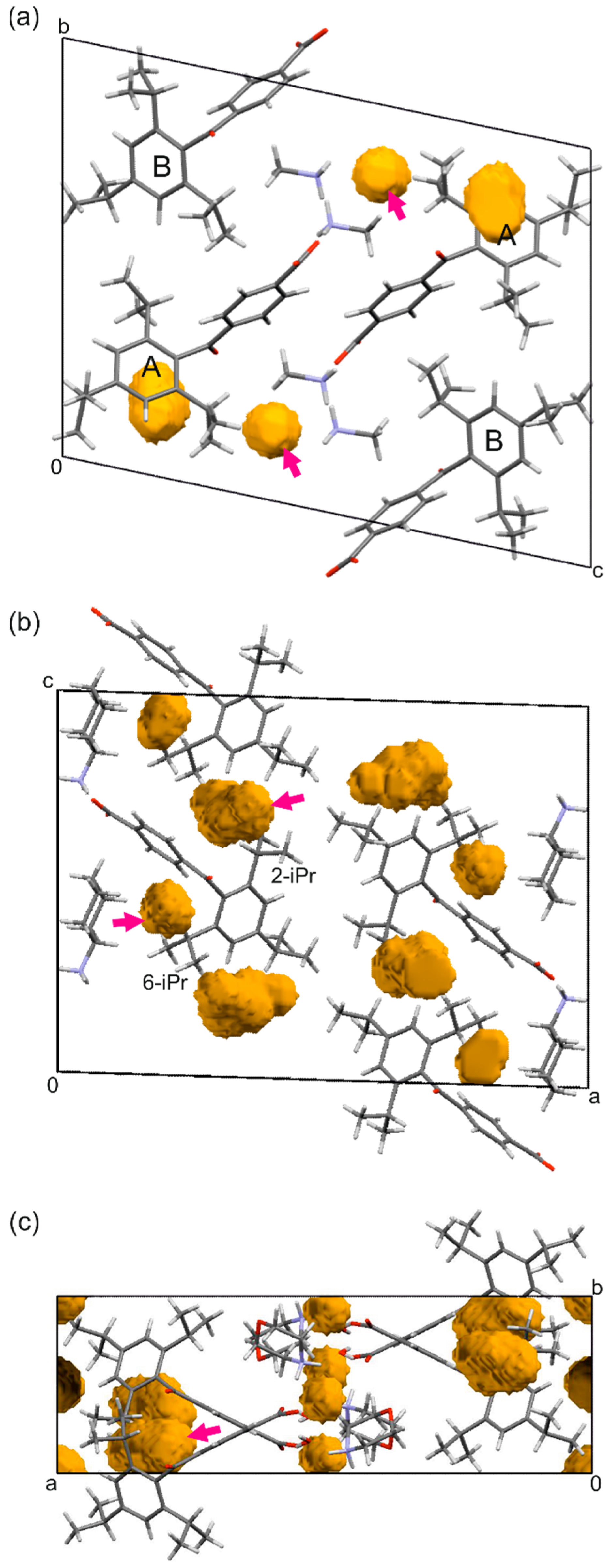
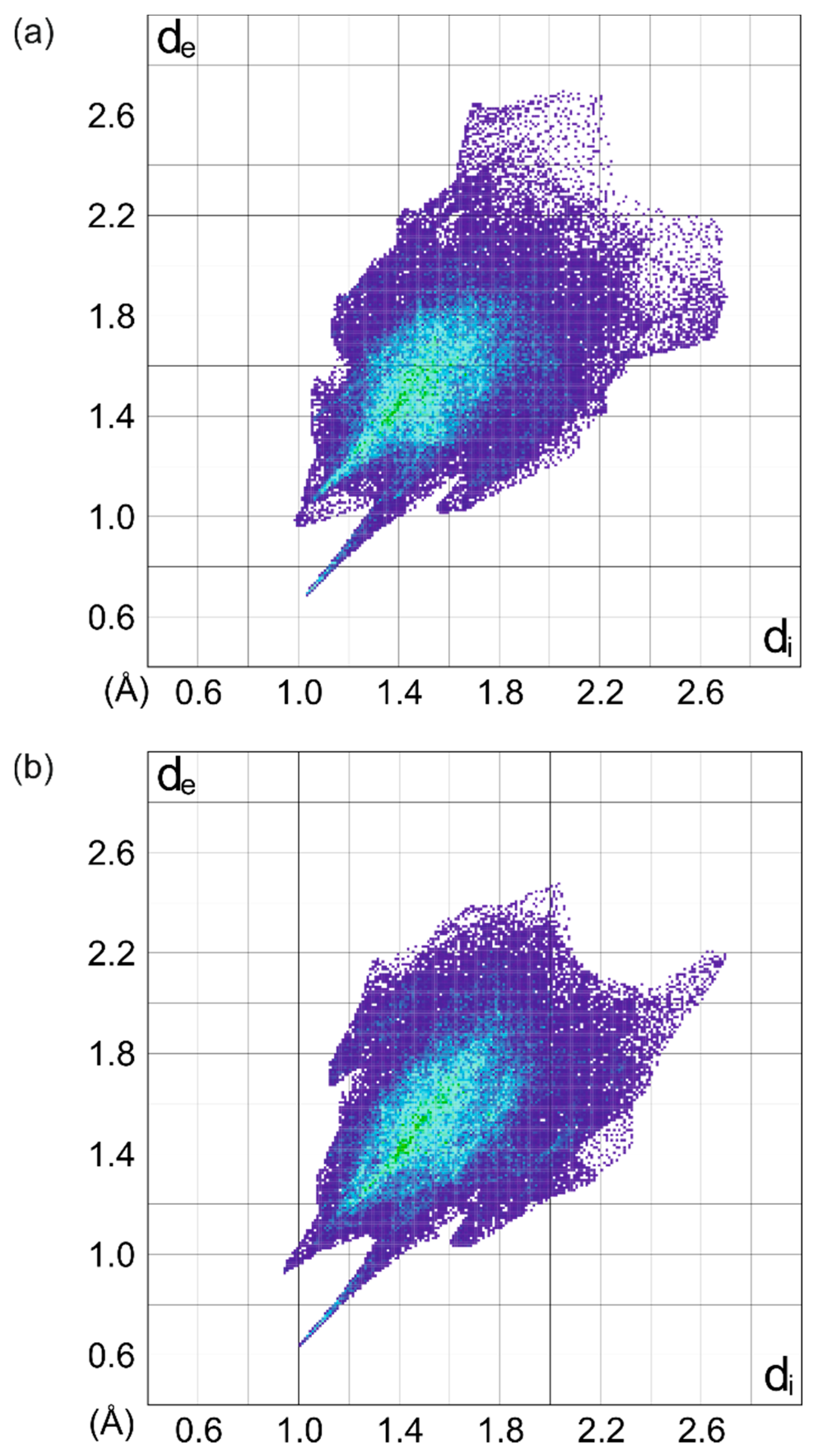
| Compound | (1) | (2) | (3) | (4) | |
|---|---|---|---|---|---|
| Pressure | 0.1 MPa | 1.3 GPa | 1.0 GPa | 0.1 MPa | 0.1 MPa |
| Chemical formula | C26H38N2O3 | C26H38N2O3 | C24H33NO3 | C29H41NO3 | C27H39NO5 |
| Formula weight | 426.58 | 426.58 | 383.51 | 451.63 | 457.59 |
| Crystal dimensions/mm | 0.26 × 0.22 × 0.04 | 0.25 × 0.18 × 0.04 | 0.19 × 0.16 × 0.11 | 0.40 × 0.10 × 0.03 | 0.30 × 0.20 × 0.06 |
| Crystal system | Monoclinic | Monoclinic | Triclinic | Monoclinic | Monoclinic |
| Space group | P21/c | P21/c | P | P21/c | P21/c |
| a/Å | 25.4217(11) | 25.34(3) | 6.1205(7) | 24.434(3) | 25.9788(19) |
| b/Å | 8.5125(4) | 8.2143(11) | 16.6401(17) | 6.3455(8) | 8.4472(9) |
| c/Å | 11.8343(5) | 11.4907(13) | 21.596(4) | 17.538(2) | 12.2065(11) |
| α/° | 101.627(15) | ||||
| β/° | 95.110(3) | 94.67(3) | 96.245(15) | 91.774(4) | 99.931(7) |
| γ/° | 91.507(9) | ||||
| V/Å3 | 2550.8(2) | 2384(3) | 2138.9(5) | 2718.0(6) | 2638.6(4) |
| Z | 4 | 4 | 4 | 4 | 4 |
| Dx/Mgm−3 | 1.111 | 1.189 | 1.191 | 1.104 | 1.152 |
| μ/mm−1 | 0.07 | 0.08 | 0.08 | 0.07 | 0.08 |
| T/K | 299 | 299 | 299 | 130 | 299 |
| Reflections collected | 9335 | 11,637 | 10,631 | 28,358 | 9294 |
| Reflections independent | 4483 | 1363 | 2286 | 4790 | 4644 |
| Reflections observed | 2059 | 594 | 1083 | 2962 | 2062 |
| Rint | 0.061 | 0.207 | 0.119 | 0.104 | 0.049 |
| R (F2 > 2σ (F2)), wR, S | 0.077, 0.176, 1.01 | 0.075, 0.220, 0.98 | 0.109, 0.353, 1.05 | 0.075, 0.171, 1.09 | 0.098, 0.289, 1.03 |
| Δρmax, Δρmin /eÅ−3 | 0.17, −0.16 | 0.12, −0.13 | 0.21, −0.21 | 0.25, −0.20 | 0.30, −0.37 |
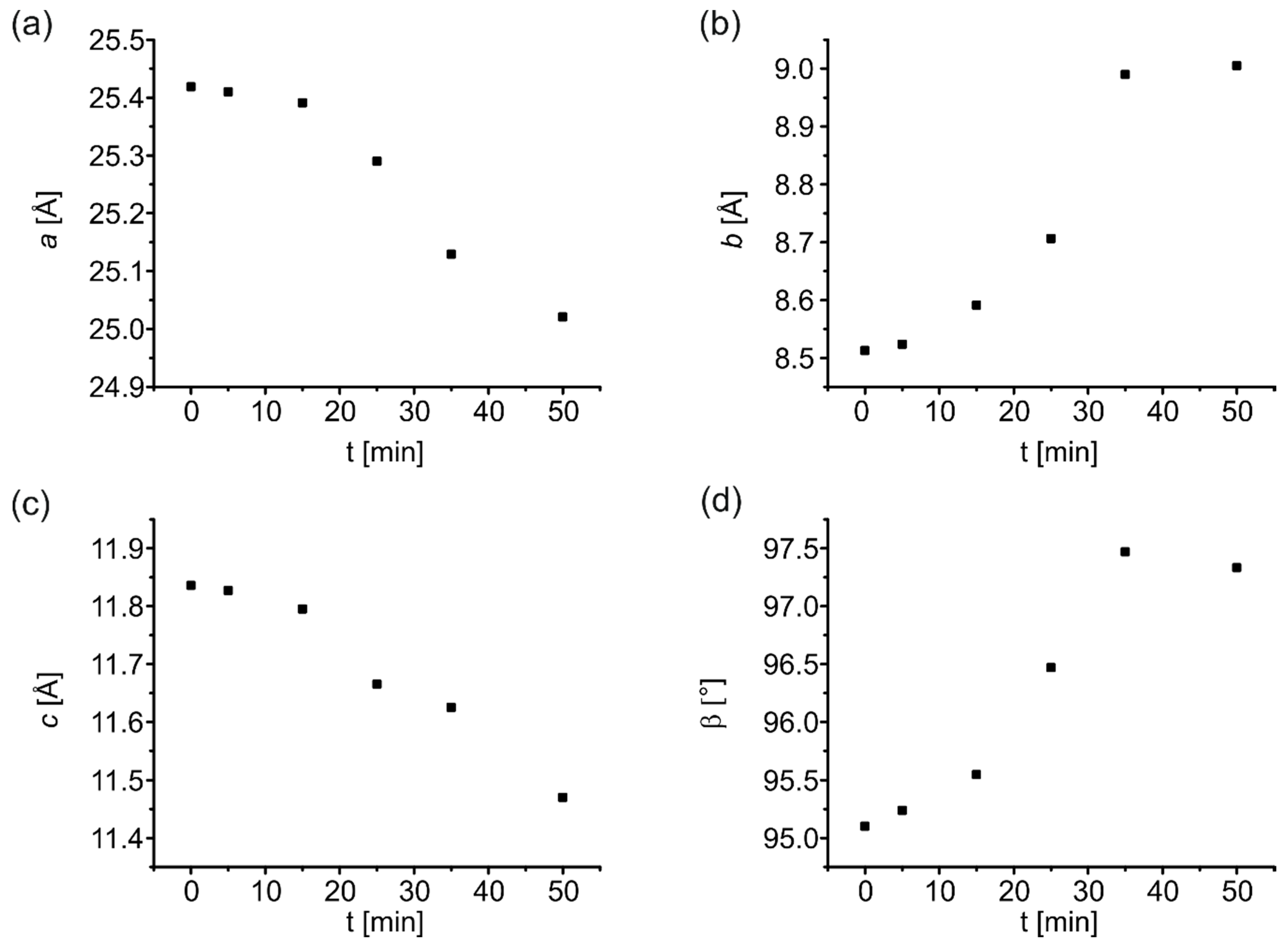
| t/min | a/Å | b/Å | c/Å | α/° | β/° | γ/° | V/Å3 |
|---|---|---|---|---|---|---|---|
| (1) at 0.1 MPa | |||||||
| 0 | 25.4217(11) | 8.5125(4) | 11.8343(5) | 95.110(3) | 2550.8(2) | ||
| 5 | 25.410(2) | 8.5235(8) | 11.8269(9) | 95.237(7) | 2550.8(4) | ||
| 15 | 25.391(4) | 8.591(2) | 11.795(2) | 95.548(17) | 2561(1) | ||
| 25 | 25.29(2) | 8.706(9) | 11.665(12) | 96.47(7) | 2552(4) | ||
| 35 | 25.130(3) | 8.969(11) | 11.625(14) | 97.47(1) | 2598(5) | ||
| 50 | 25.021(5) | 9.0053(18) | 11.470(3) | 97.332(17) | 2563.3(9) | ||
| (2) at 1.0 GPa | |||||||
| 5 | 6.102(3) | 16.123(8) | 22.295(19) | 100.55(6) | 97.68(10) | 90.37(5) | 2136(2) |
| (3) at 0.1 MPa | |||||||
| 35 | 24.202(9) | 6.3227(18) | 18.035(5) | 90.766(12) | 2759.6(15) | ||
| d [Å] | D [Å] | Δ [°] | θ [°] | ω [°] | |||
|---|---|---|---|---|---|---|---|
| Ideal value | <2.7 | 90–120 | 180 | 0 | |||
| Literature range a | 2.39–3.07 | 2.82–3.12 | 52.0–88.0 | 104.3–131.6 | 50.8–85.5 | ||
| (1) | |||||||
| 2-isopropyl, γ-H | 0.1 MPa | 2.63 | 2.948(6) | 67.0 | 122.5 | 72.9 | |
| 6-isopropyl, γ-H | 3.10 | 2.943(6) | 47.0 | 116.0 | 88.4 | ||
| 2-isopropyl, δ-H | 3.39 | 3.935(6) | 95.6 | 110.9 | 39.3 | ||
| 6-isopropyl, δ-H | 3.49 | 3.782(6) | 79.2 | 120.1 | 58.9 | ||
| 2-isopropyl, γ-H | 1.3 GPa | 2.5 | 2.94(2) | 72 | 126 | 69 | |
| 6-isopropyl, γ-H | 3.2 | 2.93(1) | 43 | 115 | 91 | ||
| 2-isopropyl, δ-H | 3.4 | 3.96(2) | 97 | 108 | 34 | ||
| 6-isopropyl, δ-H | 3.5 | 3.76(1) | 76 | 123 | 63 | ||
| (2) | |||||||
| 2-isopropyl, γ-H | 0.1 MPa d | 3.07 | 2.920(4) | 53.4 | 104.3 | 83.4 | |
| 6-isopropyl, γ-H | 2.89 | 2.931(4) | 54.6 | 115.0 | 83.4 | ||
| 2-isopropyl, δ-H | 3.09 | 3.694(4) | 90.7 | 125.2 | 56.8 | ||
| 6-isopropyl, δ-H | 3.33 | 3.815(5) | 88.8 | 119.6 | 54.3 | ||
| 2-isopropyl, γ-H | A | 1.0 GPa | 2.9 b | 2.917(15) | 53 | 115 b | 84 |
| B | 3.0 b | 2.88(3) | 49 | 113 b | 86 | ||
| 6-isopropyl, γ-H | A | 3.0 c | 2.90(3) | 53 | 108 c | 83 | |
| B | 2.8 b | 2.906(14) | 61 b | 108 c | 78 | ||
| 2-isopropyl, δ-H | A | 3.3 | 3.786(16) | 89 | 121 | 57 | |
| B | 3.2 | 3.62(3) | 83 | 122 | 53 | ||
| 6-isopropyl, δ-H | A | 2.8 b | 3.57(4) b | 92 | 130 b | 56 | |
| B | 2.7 b | 3.505(18) b | 103 | 118 | 58 | ||
| (3) | |||||||
| 2-isopropyl, γ-H | 0.1 MPa | 2.88 | 2.907(5) | 53.9 | 118.7 | 84.6 | |
| 6-isopropyl, γ-H | 2.81 | 2.936(5) | 57.6 | 122.2 | 79.9 | ||
| 2-isopropyl, δ-H | 3.40 | 3.817(6) | 86.1 | 117.6 | 54.7 | ||
| 6-isopropyl, δ-H | 3.54 | 3.928(7) | 87.6 | 113.4 | 50.3 | ||
| (4) | |||||||
| 2-isopropyl, γ-H e | 0.1 MPa | 2.65 2.82 | 2.931(6) | 65.2 260.2 | 123.4 2110.8 | 74.5 277.1 | |
| 6-isopropyl, γ-H | 3.12 | 2.938(7) | 47.1 | 111.7 | 88.0 | ||
| 2-isopropyl, δ-H e | 3.47 22.80 | 3.967(15) 23.62(3) | 93.9 298.8 | 110.7 2127.7 | 42.4 247.1 | ||
| 6-isopropyl, δ-H | 3.26 | 3.688(8) | 82.2 | 125.0 | 59.6 | ||
| a/Å | b/Å | c/Å | α/° | β/° | γ/° | V/Å3 |
|---|---|---|---|---|---|---|
| (2) at 0.1 MPa, I2/a | ||||||
| 17.1387(10) | 6.3047(4) | 44.649(3) | 98.744(6) | 4768.4(5) | ||
| (2) at 1.0 GPa, P | ||||||
| 6.1205(7) | 16.6401(17) | 21.596(4) | 101.627(15) | 96.245(15) | 91.507(9) | 2138.9(5) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konieczny, K.; Ciesielski, A.; Bąkowicz, J.; Galica, T.; Turowska-Tyrk, I. Structural Transformations in Crystals Induced by Radiation and Pressure. Part 7. Molecular and Crystal Geometries as Factors Deciding about Photochemical Reactivity under Ambient and High Pressures. Crystals 2018, 8, 299. https://doi.org/10.3390/cryst8070299
Konieczny K, Ciesielski A, Bąkowicz J, Galica T, Turowska-Tyrk I. Structural Transformations in Crystals Induced by Radiation and Pressure. Part 7. Molecular and Crystal Geometries as Factors Deciding about Photochemical Reactivity under Ambient and High Pressures. Crystals. 2018; 8(7):299. https://doi.org/10.3390/cryst8070299
Chicago/Turabian StyleKonieczny, Krzysztof, Arkadiusz Ciesielski, Julia Bąkowicz, Tomasz Galica, and Ilona Turowska-Tyrk. 2018. "Structural Transformations in Crystals Induced by Radiation and Pressure. Part 7. Molecular and Crystal Geometries as Factors Deciding about Photochemical Reactivity under Ambient and High Pressures" Crystals 8, no. 7: 299. https://doi.org/10.3390/cryst8070299
APA StyleKonieczny, K., Ciesielski, A., Bąkowicz, J., Galica, T., & Turowska-Tyrk, I. (2018). Structural Transformations in Crystals Induced by Radiation and Pressure. Part 7. Molecular and Crystal Geometries as Factors Deciding about Photochemical Reactivity under Ambient and High Pressures. Crystals, 8(7), 299. https://doi.org/10.3390/cryst8070299