Over-Expression, Secondary Structure Characterization, and Preliminary X-ray Crystallographic Analysis of Xenopus tropicalis Ependymin
Abstract
1. Introduction
2. Materials and Methods
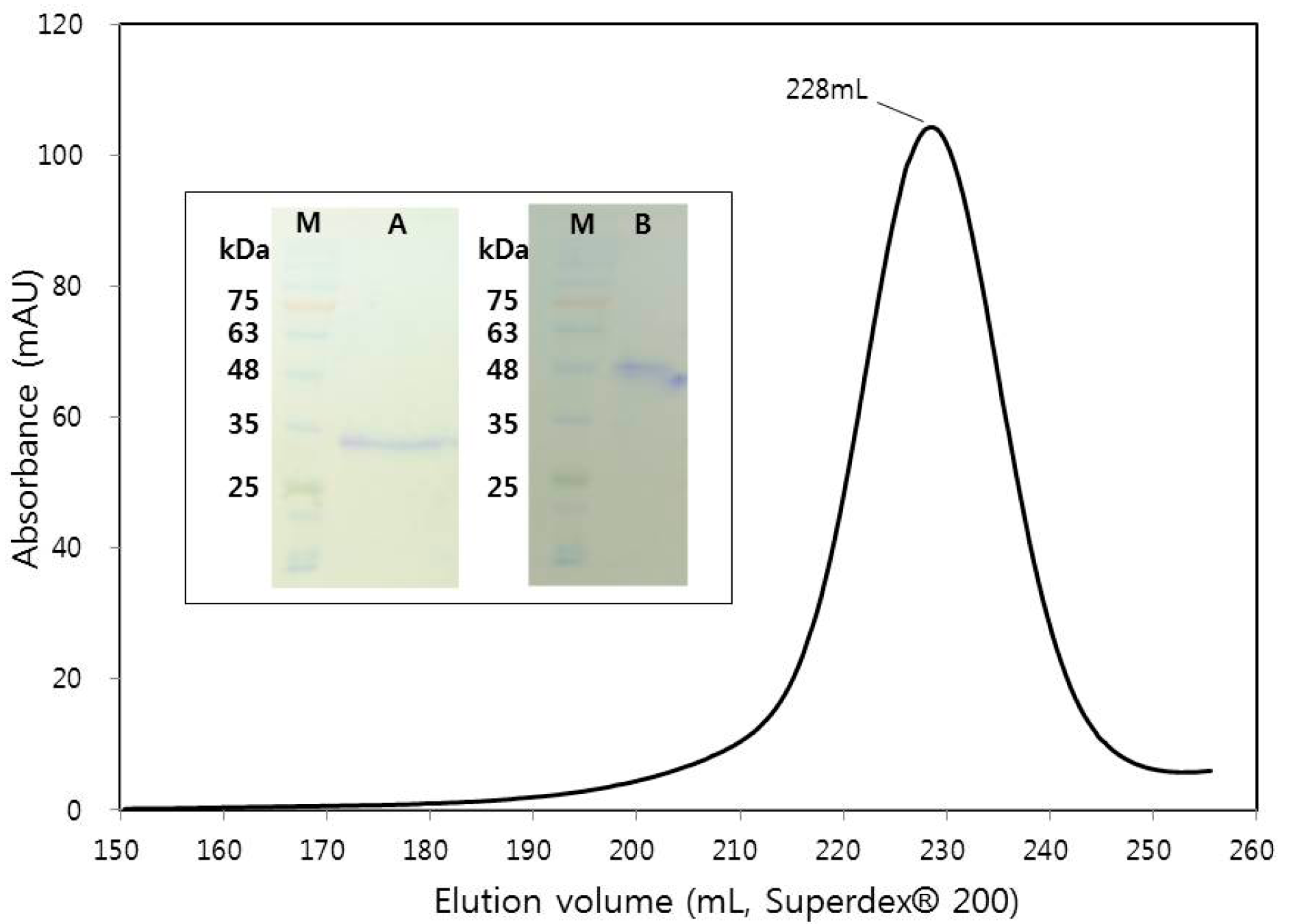
2.1. Macromolecule Production
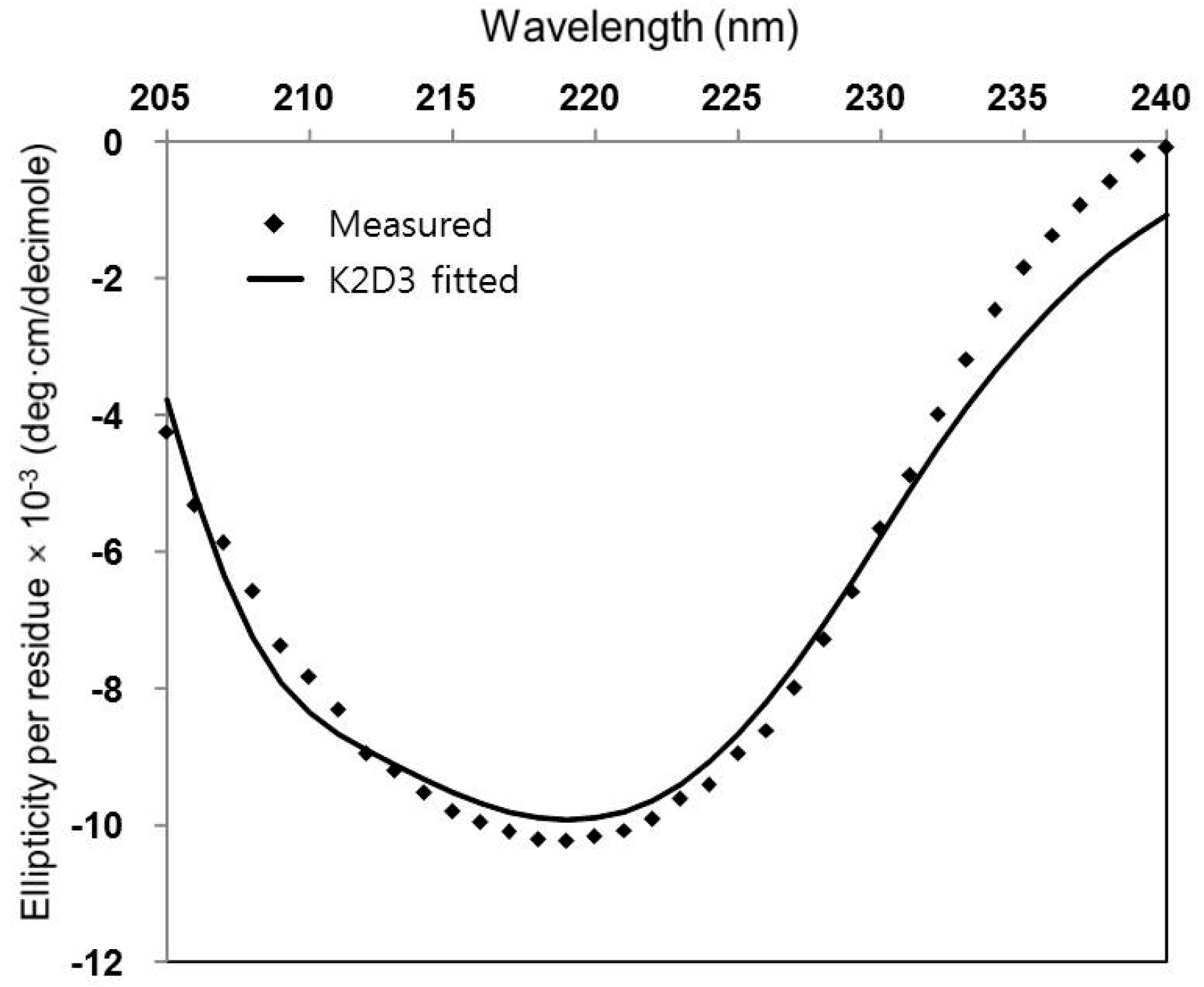
2.2. Circular Dichroism (CD)
2.3. Crystallization
2.4. Data Collection and Processing
3. Results and Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shashoua, V.E. Brain metabolism and the acquisition of new behaviors. I. Evidence for specific changes in the pattern of protein synthesis. Brain Res. 1976, 111, 347–364. [Google Scholar] [CrossRef]
- Benowitz, L.I.; Shashoua, V.E. Localization of a brain protein metabolically linked with behavioral plasticity in the goldfish. Brain Res. 1977, 136, 227–242. [Google Scholar] [CrossRef]
- Shashoua, V.E. Brain protein metabolism and the acquisition of new patterns of behavior. Proc. Natl. Acad. Sci. USA 1977, 74, 1743–1747. [Google Scholar] [CrossRef] [PubMed]
- Shashoua, V.E. Ependymin, a brain extracellular glycoprotein, and CNS plasticity. Ann. N. Y. Acad. Sci. 1991, 627, 94–114. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R. Cell-adhesion molecules in memory formation. Behav. Brain Res. 1995, 66, 65–72. [Google Scholar] [CrossRef]
- Tang, S.J.; Sun, K.H.; Sun, G.H.; Lin, G.; Lin, W.W.; Chuang, M.J. Cold-induced ependymin expression in zebrafish and carp brain: Implications for cold acclimation. FEBS Lett. 1999, 459, 95–99. [Google Scholar] [CrossRef]
- Sneddon, L.U.; Schmidt, R.; Fang, Y.; Cossins, A.R. Molecular correlates of social dominance: A novel role for ependymin in aggression. PLoS ONE 2011, 6, e18181. [Google Scholar] [CrossRef] [PubMed]
- Apostolopoulos, J.; Sparrow, R.L.; McLeod, J.L.; Collier, F.M.; Darcy, P.K.; Slater, H.R.; Ngu, C.; Gregorio-King, C.C.; Kirkland, M.A. Identification and characterization of a novel family of mammalian ependymin-related proteins (MERPs) in hematopoietic, nonhematopoietic, and malignant tissues. DNA Cell Biol. 2001, 20, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Gregorio-King, C.C.; McLeod, J.L.; Collier, F.M.; Collier, G.R.; Bolton, K.A.; Van Der Meer, G.J.; Apostolopoulos, J.; Kirkland, M.A. MERP1: A mammalian ependymin-related protein gene differentially expressed in hematopoietic cells. Gene 2002, 286, 249–257. [Google Scholar] [CrossRef]
- Nimmrich, I.; Erdmann, S.; Melchers, U.; Chtarbova, S.; Finke, U.; Hentsch, S.; Hoffmann, I.; Oertel, M.; Hoffmann, W.; Müller, O. The novel ependymin related gene UCC1 is highly expressed in colorectal tumor cells. Cancer Lett. 2001, 165, 71–79. [Google Scholar] [CrossRef]
- Müller-Schmid, A.; Rinder, H.; Lottspeich, F.; Gertzen, E.M.; Hoffmann, W. Ependymins from the cerebrospinal fluid of salmonid fish: Gene structure and molecular characterization. Gene 1992, 118, 189–196. [Google Scholar] [CrossRef]
- Königstorfer, A.; Sterrer, S.; Hoffmann, W. Biosynthesis of ependymins from goldfish brain. J. Biol. Chem. 1989, 264, 13689–13692. [Google Scholar] [PubMed]
- Shashoua, V.E.; Adams, D.; Boyer-Boiteau, A. CMX-8933, a peptide fragment of the glycoprotein ependymin, promotes activation of AP-1 transcription factor in mouse neuroblastoma and rat cortical cell cultures. Neurosci. Lett. 2001, 312, 103–107. [Google Scholar] [CrossRef]
- Adams, D.S.; Hasson, B.; Boyer-Boiteau, A.; El-Khishin, A.; Shashoua, V.E. A peptide fragment of ependymin neurotrophic factor uses protein kinase C and the mitogen-activated protein kinase pathway to activate c-Jun N-terminal kinase and a functional AP-1 containing c-Jun and c-Fos proteins in mouse NB2a cells. J. Neurosci. Res. 2003, 72, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Kaska, J. Ependymin Mechanism of Action: Full Length EPN vs Peptide CMX-8933. Master’s Thesis, Worcester Polytechnic Institute, Worcester, MA, USA, 28 May 2003. [Google Scholar]
- Suárez-Castillo, E.C.; García-Arrarás, J.E. Molecular evolution of the ependymin protein family: A necessary update. BMC Evol. Biol. 2007, 7, 23. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.C.; von Hippel, P.H. Calculation of protein extinction coefficients from amino acid sequence data. Anal. Biochem. 1989, 182, 319–326. [Google Scholar] [CrossRef]
- Louis-Jeune, C.; Andrade-Navarro, M.A.; Perez-Iratxeta, C. Prediction of protein secondary structure from circular dichroism using theoretically derived spectra. Proteins 2012, 80, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Otwinowski, Z.; Minor, W. Processing of X-ray Diffraction Data Collected in Oscillation Mode. Methods Enzymol. 1997, 276, 307–326. [Google Scholar] [CrossRef] [PubMed]
- Chang, V.T.; Crispin, M.; Aricescu, A.R.; Harvey, D.J.; Nettleship, J.E.; Fennelly, J.A.; Yu, C.; Boles, K.S.; Evans, E.J.; Stuart, D.I.; et al. Glycoprotein Structural Genomics: Solving the Glycosylation Problem. Structure 2007, 15, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Matthews, B.W. Solvent content of protein crystals. J. Mol. Biol. 1968, 33, 491–497. [Google Scholar] [CrossRef]
- Pike, A.C.; Garman, E.F.; Krojer, T.; von Delft, F.; Carpenter, E.P. An overview of heavy-atom derivatization of protein crystals. Acta Crystallogr. D Struct. Biol. 2016, 72, 303–318. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, O.; Bujacz, A.; Biesiadka, J.; Bujacz, G.; Sikorski, M.; Jaskolski, M. MAD phasing using the (Ta6Br12)2+ cluster: A retrospective study. Acta Crystallogr. D Struct. Biol. 2008, 64, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Sippel, K.H.; Robbins, A.H.; Reutzel, R.; Domsic, J.; Boehlein, S.K.; Govindasamy, L.; Agbandje-McKenna, M.; Rosser, C.J.; McKenna, R. Structure determination of the cancer-associated Mycoplasma hyorhinis protein Mh-p37. Acta Crystallogr. D Struct. Biol. 2008, 64, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- El Omari, K.; Iourin, O.; Kadlec, J.; Fearn, R.; Hall, D.R.; Harlos, K.; Grimes, J.M.; Stuart, D.I. Pushing the limits of sulfur SAD phasing: De novo structure solution of the N-terminal domain of the ectodomain of HCV E1. Acta Crystallogr. D Struct. Biol. 2014, 70, 2197–2203. [Google Scholar] [CrossRef] [PubMed]
- Gorgel, M.; Bøggild, A.; Ulstrup, J.J.; Weiss, M.S.; Müller, U.; Nissen, P.; Boesen, T. Against the odds? De novo structure determination of a pilin with two cysteine residues by sulfur SAD. Acta Crystallogr. D Struct. Biol. 2015, 71, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Foos, N.; Seuring, C.; Schubert, R.; Burkhardt, A.; Svensson, O.; Meents, A.; Chapman, H.N.; Nanao, M.H. X-ray and UV radiation-damage-induced phasing using synchrotron serial crystallography. Acta Crystallogr. D Struct. Biol. 2018, 74, 366–378. [Google Scholar] [CrossRef] [PubMed]



| Source Organism | Xenopus tropicalis |
|---|---|
| DNA source | Synthesized DNA |
| Cloning sites | BamHI and NotI |
| Cloning vector | pAcGP67A |
| Expression vector | pAcGP67A |
| Expression host | SF9 (S. frugiperda) |
| Complete amino acid sequence of the construct produced 1 | ADPHHHHHHHHPVTPCEAPLQWEGRIVLYDHNTGKNTRATVTYDAILQRIRILEEKKSFVPCKRFFEYIFLYQEAVMFQIEQVTKICSKNTLTEPWDPYDIPENSTYEDQYYIGGPGDQIPVQEWSDRKPARKYETWVGVYTVKDCYPVQETYTKNDSMTTSTRFFDIKLGISDPSVFNPPSTCEAAQPLLMSGDC |
| Diffraction Source | Pohang Light Source (PLS 7A) (Pohang, Korea) |
|---|---|
| Wavelength (Å) | 0.97934 |
| Temperature (K) | 100 |
| Detector | ADSC Quantum Q270 |
| Crystal-detector distance (mm) | 350 |
| Rotation range per image (°) | 1 |
| Total rotation range (°) | 180 |
| Exposure time per image (s) | 1 |
| Space group | P6122 (or P6522) |
| a, b, c (Å) | 61.05, 61.05, 234.33 |
| α, β, γ (°) | 90.0, 90.0, 120.0 |
| Mosaicity (°) | 0.496 |
| Resolution range (Å) | 50.0–2.70 (2.75–2.70) 1 |
| Total No. of reflections | 129,713 |
| No. of unique reflections | 7871 |
| Completeness (%) | 99.6 (100.0) |
| Redundancy | 16.6 (17.0) |
| 〈I/σ(I)〉 | 43.8 (3.7) |
| Rmerge | 0.118 (1.136) |
| Rp.i.m. | 0.029 (0.271) |
| CC1/2 | (0.886) |
| Overall B factor from Wilson plot (Å2) | 58.9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.K.; Sim, Y.W.; Park, S. Over-Expression, Secondary Structure Characterization, and Preliminary X-ray Crystallographic Analysis of Xenopus tropicalis Ependymin. Crystals 2018, 8, 284. https://doi.org/10.3390/cryst8070284
Park JK, Sim YW, Park S. Over-Expression, Secondary Structure Characterization, and Preliminary X-ray Crystallographic Analysis of Xenopus tropicalis Ependymin. Crystals. 2018; 8(7):284. https://doi.org/10.3390/cryst8070284
Chicago/Turabian StylePark, Jeong Kuk, Yeo Won Sim, and SangYoun Park. 2018. "Over-Expression, Secondary Structure Characterization, and Preliminary X-ray Crystallographic Analysis of Xenopus tropicalis Ependymin" Crystals 8, no. 7: 284. https://doi.org/10.3390/cryst8070284
APA StylePark, J. K., Sim, Y. W., & Park, S. (2018). Over-Expression, Secondary Structure Characterization, and Preliminary X-ray Crystallographic Analysis of Xenopus tropicalis Ependymin. Crystals, 8(7), 284. https://doi.org/10.3390/cryst8070284




