Growth and Characterization of Tetraphenylphosphonium Bromide Crystal
Abstract
:1. Introduction
2. Results and Discussion
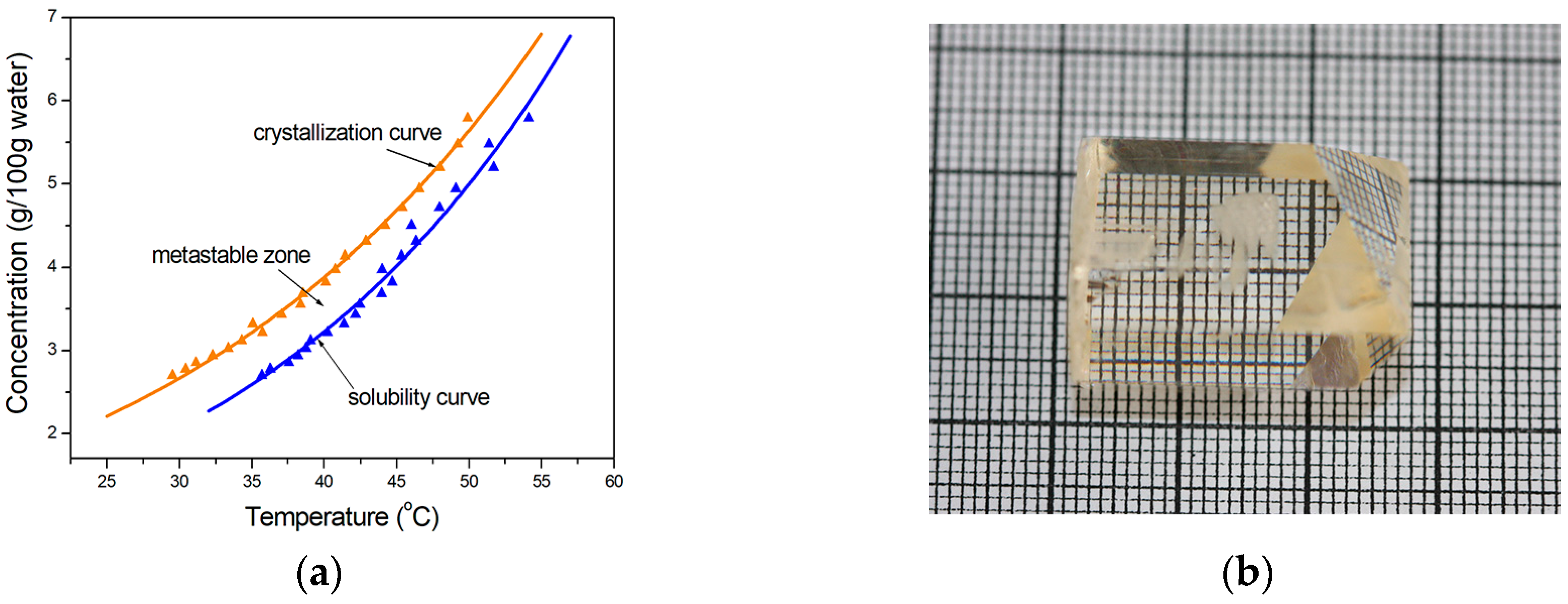
2.1. Crystal Growth
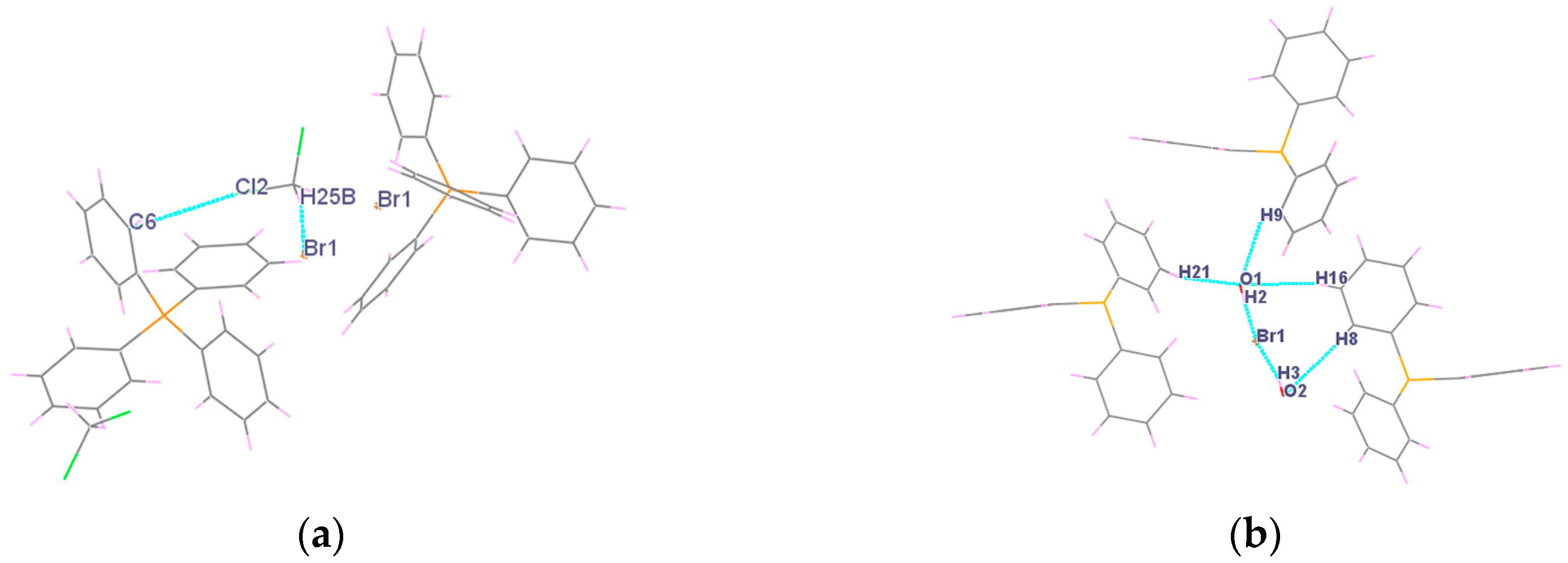
2.2. X-ray Structural Analysis
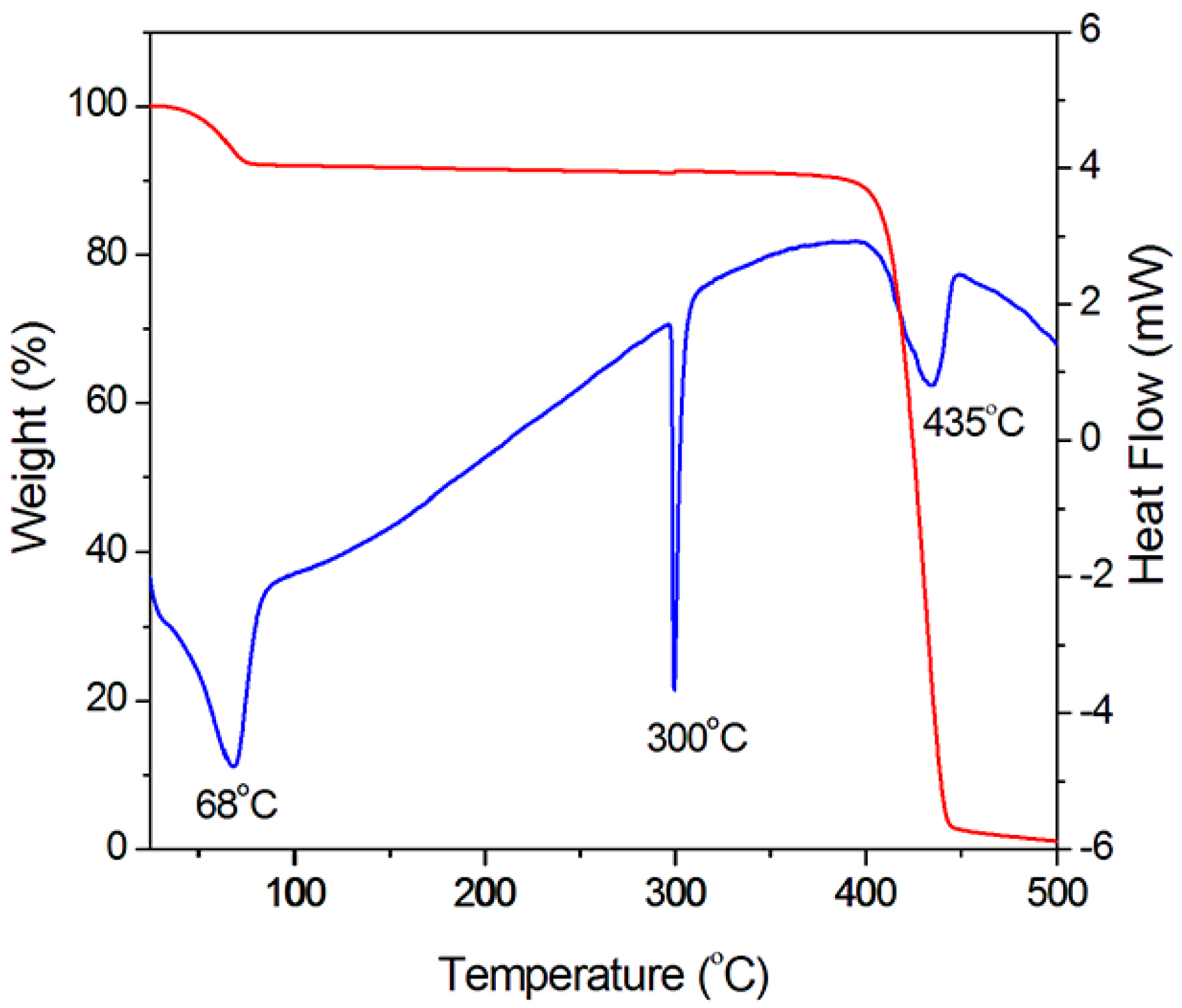
2.3. Thermal Property
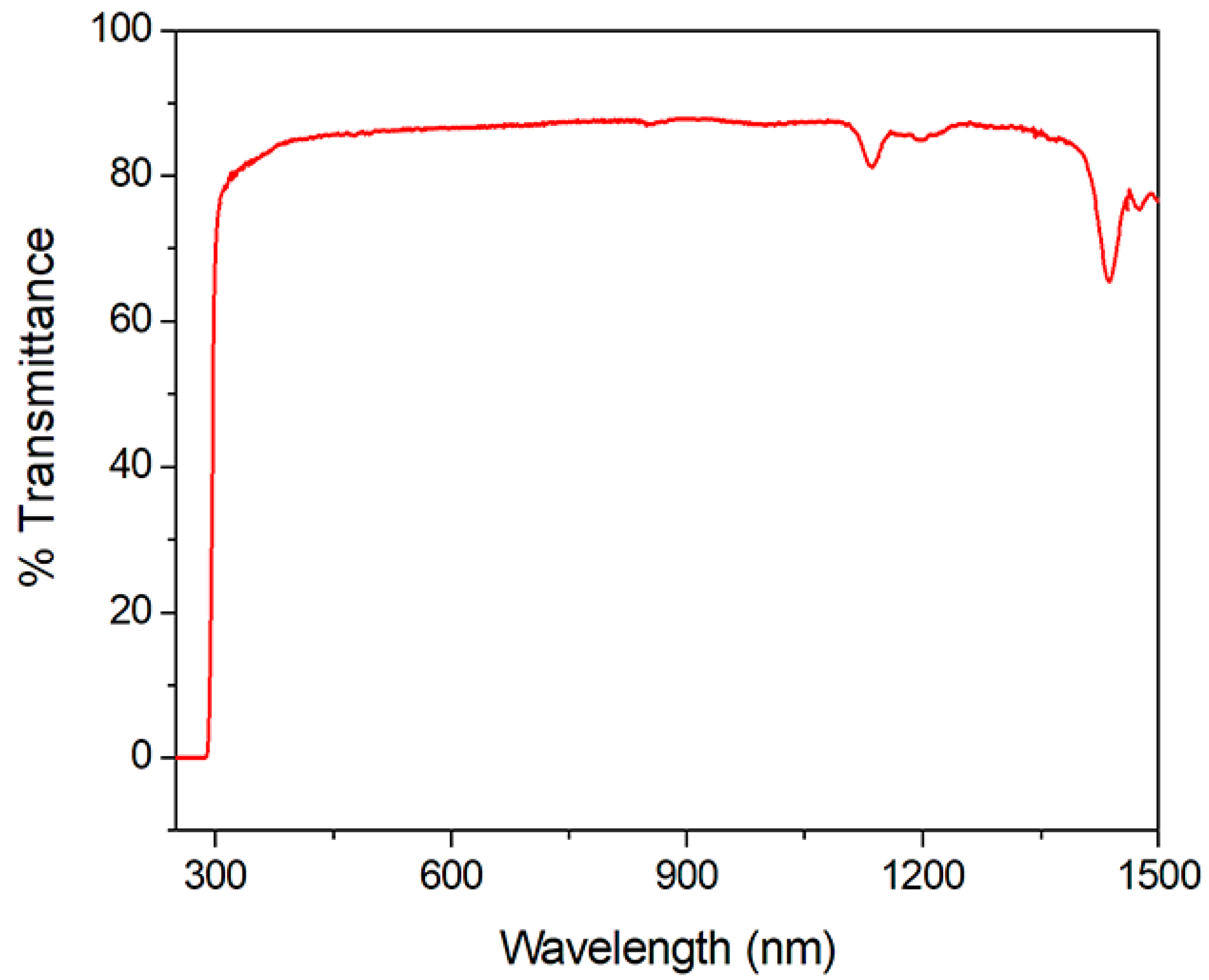
2.4. Optical Transmission Spectrum
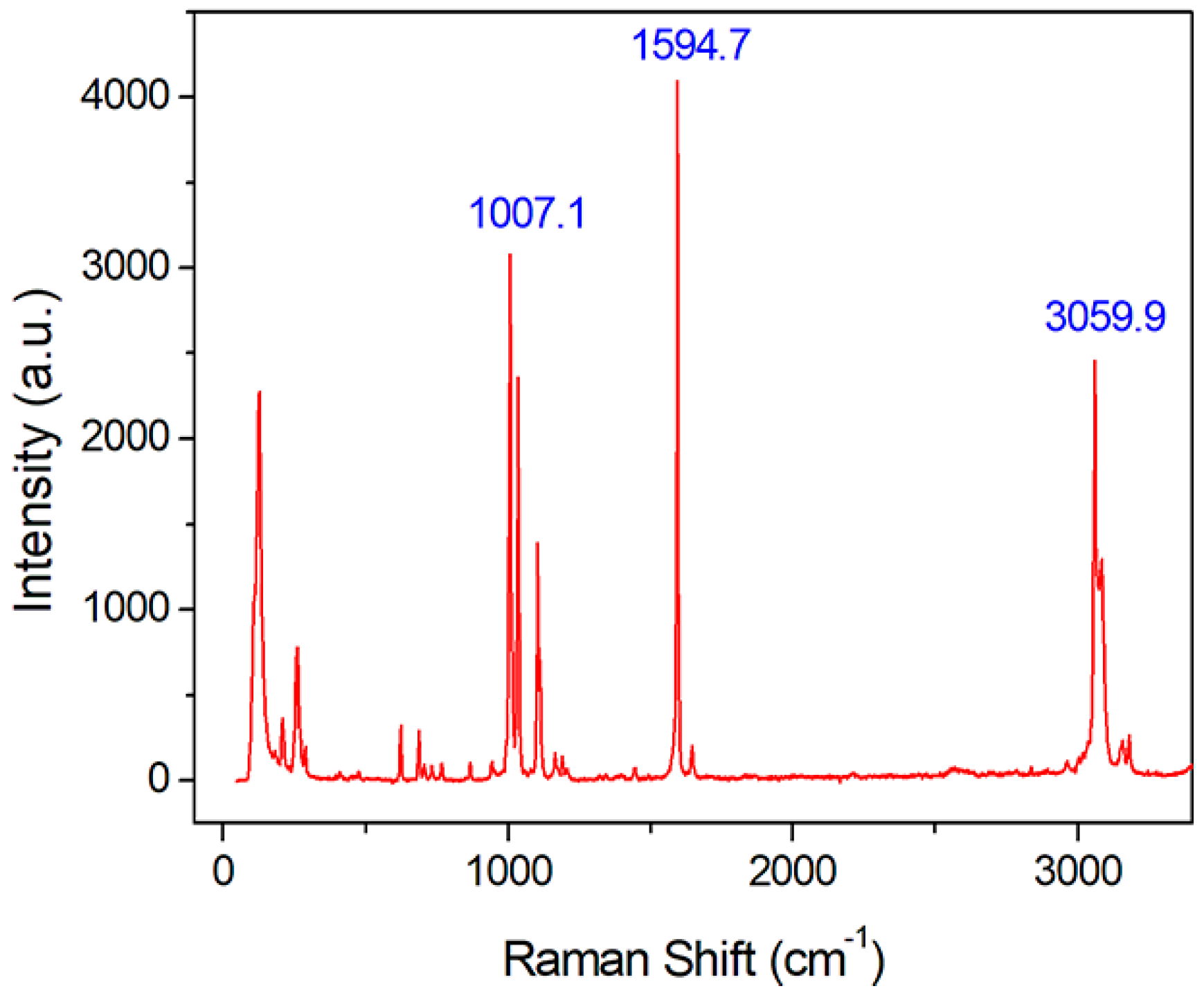
2.5. Raman Scattering Property
3. Materials and Methods
3.1. Reagents
3.2. Solubility and Crystallization Curves
3.3. Single-Crystal X-ray Diffraction
3.4. Thermal and Optical Transmission Measurement
3.5. Raman Scattering Spectrum
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bandoli, G.; Bortolozzo, G.; Clemente, D.A.; Croatto, U.; Panattoni, C. Crystal and molecular structure of triphenylphosphine oxide. J. Chem. Soc. A 1970, 2778–2780. [Google Scholar] [CrossRef]
- Codding, P.W.; Kerr, K.A. Triphenylphosphine sulfide. Acta Crystallogr. B 1978, B34, 3785–3787. [Google Scholar] [CrossRef]
- Burgess, K.M.N.; Korobkov, I.; Bryce, D.L. A combined solid-state NMR and X-ray crystallography study of the bromide ion environments in triphenylphosphonium bromides. Chem. Eur. J. 2012, 18, 5748–5758. [Google Scholar] [CrossRef] [PubMed]
- Corbridge, D.E.C. Phosphorous: An Outline of Its Chemistry, Biochemistry and Technology, 5th ed.; Elsevier: Amsterdam, The Netherlands, 1980; ISBN 0444893075. [Google Scholar]
- Chu, T.; Vyboishchikov, S.F.; Gabidullin, B.; Nikonov, G.I. Oxidative cleavage of C=S and P=S bonds at an AlI center: Preparation of terminally bound alunimum sulfides. Angew. Chem. Int. Edit. 2016, 55, 13306–13311. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.J.; Lavigne, F.; Coyle, E.E.; Holohan, A.J.; Doonan, B.J. Breaking the ring through a room temperature catalytic Wittig reaction. Chem. Eur. J. 2013, 19, 5854–5858. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, B.; Schuster, O.; Schmidbaur, H. A cyclic hexamer of silver trifluoroacetate supported by for triphenylphosphine sulfide template molecules. Inorg. Chem. 2005, 44, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Kozminykh, V.O.; Igidov, N.M.; Kozminykh, E.N.; Aliev, Z.G. Reactions of 5-aryl-furan-2,3-diones with acylmethylenetriphenylphosphoranes-synthesis and biological-activity of 3(2H)-furanone derivatives. Pharmazie 1993, 48, 99–106. [Google Scholar] [PubMed]
- El-Shahawi, M.S.; Hassan, S.S.M.; Othman, A.M.; Zyada, M.A.; El-Sonbati, M.A. Chemical speciation of chromium(III,VI) employing extractive spectrophotometry and tetraphenylarsonium chloride or tetraphenylphosphonium bromide as ion-pair reagent. Anal. Chim. Acta 2005, 534, 319–326. [Google Scholar] [CrossRef]
- Varma, R.S.; Naicker, K.P.; Liesen, P.J. Palladium chloride and tetraphenylphosphonium bromide intercalated clay as a new catalyst for the Heck reaction. Tetrahedron Lett. 1999, 40, 2075–2078. [Google Scholar] [CrossRef]
- Suzuki, H.; Kageyama, H.; Yoshida, Y.; Kimura, Y. Tetraphenylphosphonium bromide-catalyzed ‘Halex’ fluorination of chloroaryl sulfonyl chlorides. J. Fluor. Chem. 1991, 55, 335. [Google Scholar] [CrossRef]
- Han, J.; Wu, H.; Teng, M.Y.; Li, Z.Y.; Wang, Y.N.; Wang, L.Y.; Pan, Y. Novel tripod L-prolinamide catalysts based on tribenzyl- and triphenylphosphine oxide for the direct aldo reaction. Syn. Lett. 2009, 6, 933–936. [Google Scholar] [CrossRef]
- Kim, S.S.; Lee, S.H. Asymmetric cyanohydrin synthesis catalyzed by Mn(salen) complex/triphenylphosphine oxide. Syn. Commun. 2005, 35, 751–759. [Google Scholar] [CrossRef]
- Etter, M.C.; Baures, P.W. Triphenylphosphine oxide as a crystallization aid. J. Am. Chem. Soc. 1988, 110, 639–640. [Google Scholar] [CrossRef]
- Liu, X.L.; Wang, G.Q.; Dang, Y.Y.; Zhang, S.J.; Tian, H.L.; Ren, Y.; Tao, X.T. A new potential nonlinear optical hybrid semiorganic crystal of ZnMnCl4(TPPO)4 with attractive physical properties. CrystEngComm 2016, 18, 1818–1824. [Google Scholar] [CrossRef]
- See the Crystal Structural Data Deposited in Cambridge Crystallographic Data Center with the CCDC Number 270973. Available online: http://webcsd.ccdc.cam.ac.uk (accessed on 15 March 2017).
- Vincent, B.R.; Knop, O.; Linden, A.; Stanley Cameron, T.; Robertson, K.N. Crystal chemistry of tetraradial species. Part 2. Crystal structures of Et4NI, Ph4PBr·H2O, and Ph4PBr·2H2O. Can. J. Chem. 1988, 66, 3060–3069. [Google Scholar] [CrossRef]
- Curcio, J.A.; Petty, C.C. The near infrared absorption spectrum of liquid water. J. Opt. Soc. Am. 1951, 41, 302–304. [Google Scholar] [CrossRef]
- Dollish, F.R.; Fateley, W.G.; Bentley, F.F. Characteristic Raman Frequencies of Organic Compounds; Wiley-Interscience: New York, NY, USA, 1974; ISBN 0471217697. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]

| TPPB (CH2Cl2) | TPPB (2H2O) | ||
|---|---|---|---|
| Symmetry codes: I x, y, z; II − 1/2 + x, 1.5 − y, 1/2 + z | Symmetry codes: I x, y, x; II 1/2 + x, 1.5 − y, 1/2 − z; III 1/2 + x, 1/2 − y, 1/2 − z; IV 1/2 − x, 1 − y, − 1/2 + z; V 1 − x, 1/2 + y, 1 − z | ||
| I Br1...II H25B | 2.772 | I Br1...II H3 | 2.454 |
| I C6...II Cl2 | 3.354 | I Br1...III H2 | 2.447 |
| I H9...I O1 | 2.692 | ||
| IV H8...I O2 | 2.641 | ||
| IV H21...I O1 | 2.677 | ||
| IV H16...V O1 | 2.699 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, G.; Liu, X.; Ren, Y.; Zhang, C.; Tao, X. Growth and Characterization of Tetraphenylphosphonium Bromide Crystal. Crystals 2017, 7, 154. https://doi.org/10.3390/cryst7060154
Wang G, Liu X, Ren Y, Zhang C, Tao X. Growth and Characterization of Tetraphenylphosphonium Bromide Crystal. Crystals. 2017; 7(6):154. https://doi.org/10.3390/cryst7060154
Chicago/Turabian StyleWang, Guangqiang, Xiaolong Liu, Yan Ren, Chengqian Zhang, and Xutang Tao. 2017. "Growth and Characterization of Tetraphenylphosphonium Bromide Crystal" Crystals 7, no. 6: 154. https://doi.org/10.3390/cryst7060154
APA StyleWang, G., Liu, X., Ren, Y., Zhang, C., & Tao, X. (2017). Growth and Characterization of Tetraphenylphosphonium Bromide Crystal. Crystals, 7(6), 154. https://doi.org/10.3390/cryst7060154





