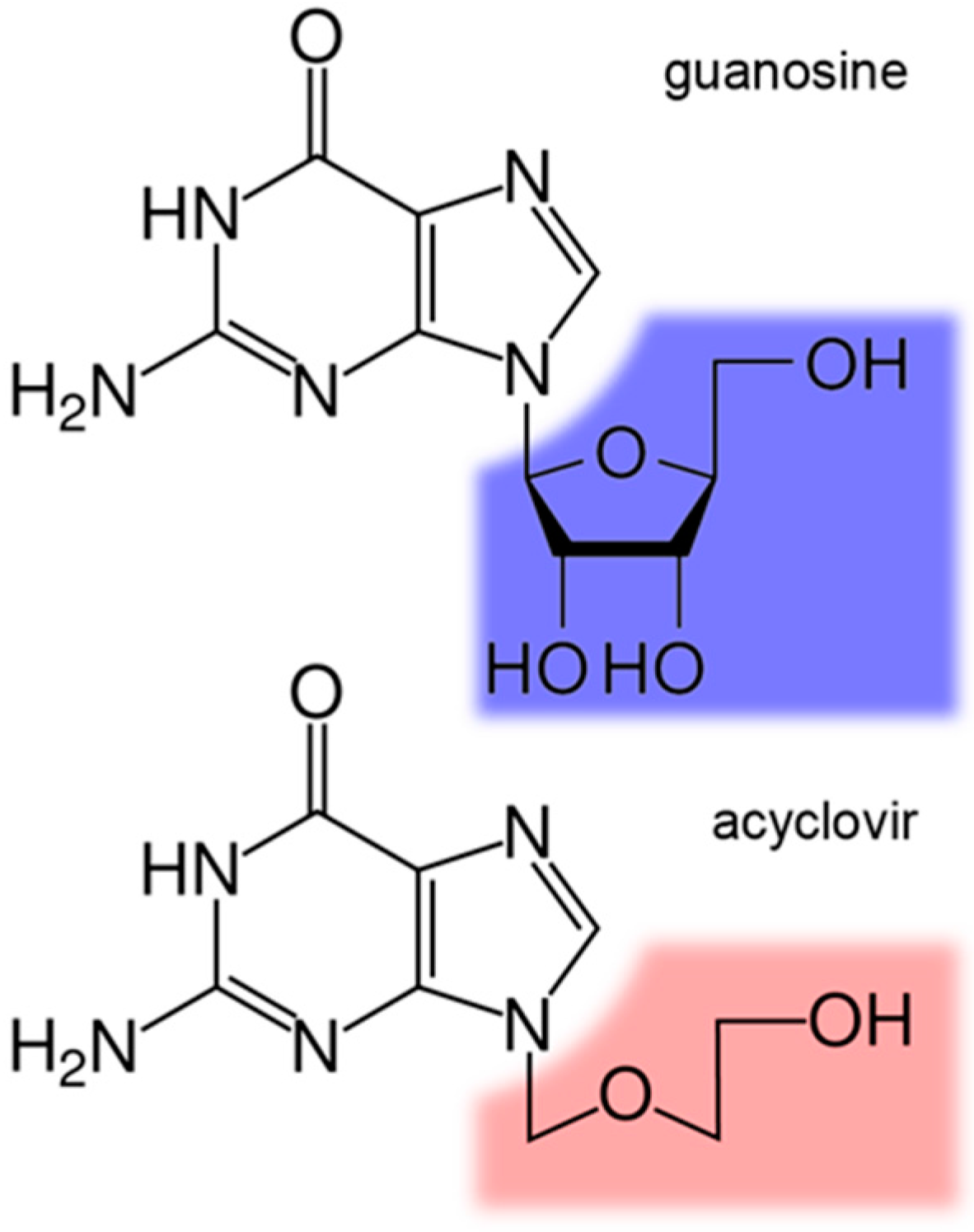
Structural and Theoretical Evidence of the Depleted Proton Affinity of the N3-Atom in Acyclovir
Abstract
:1. Introduction
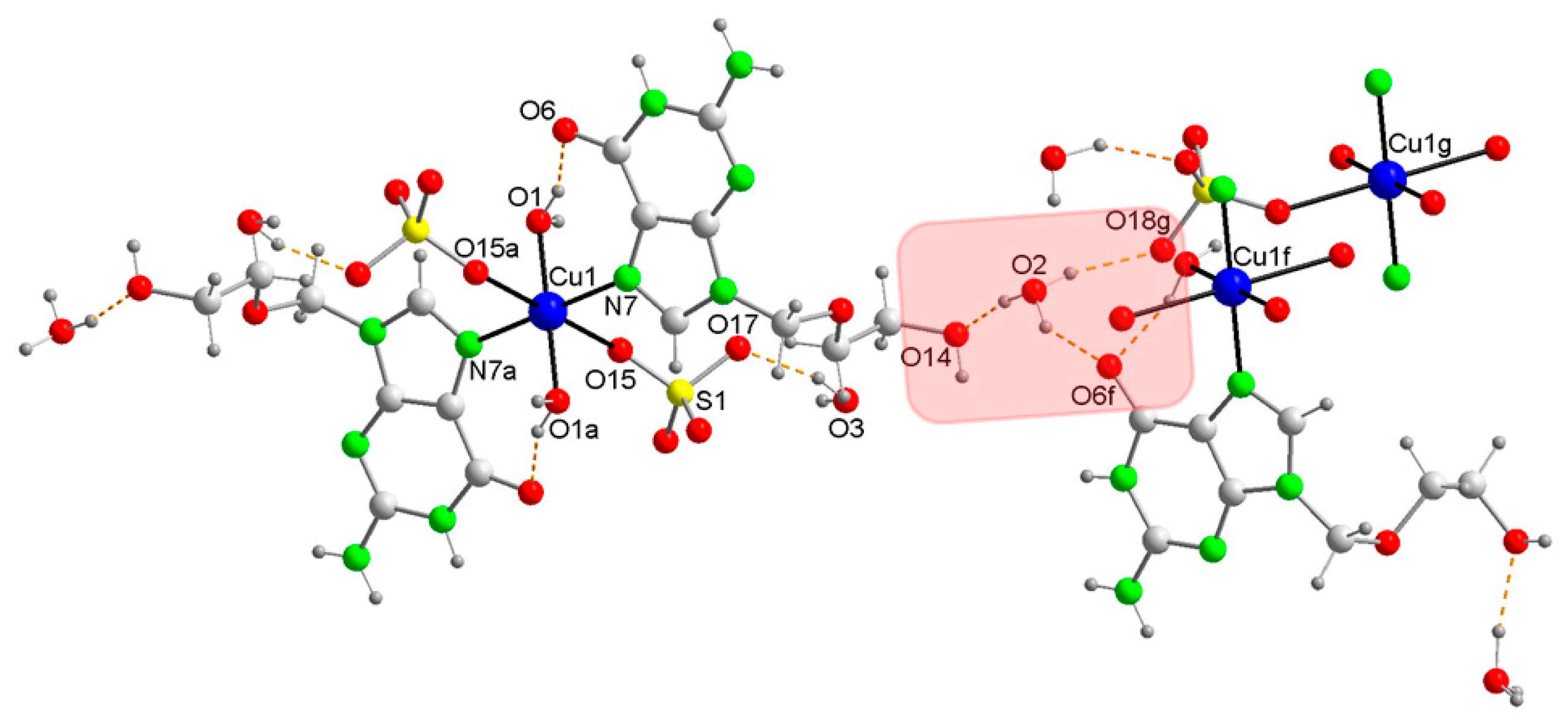
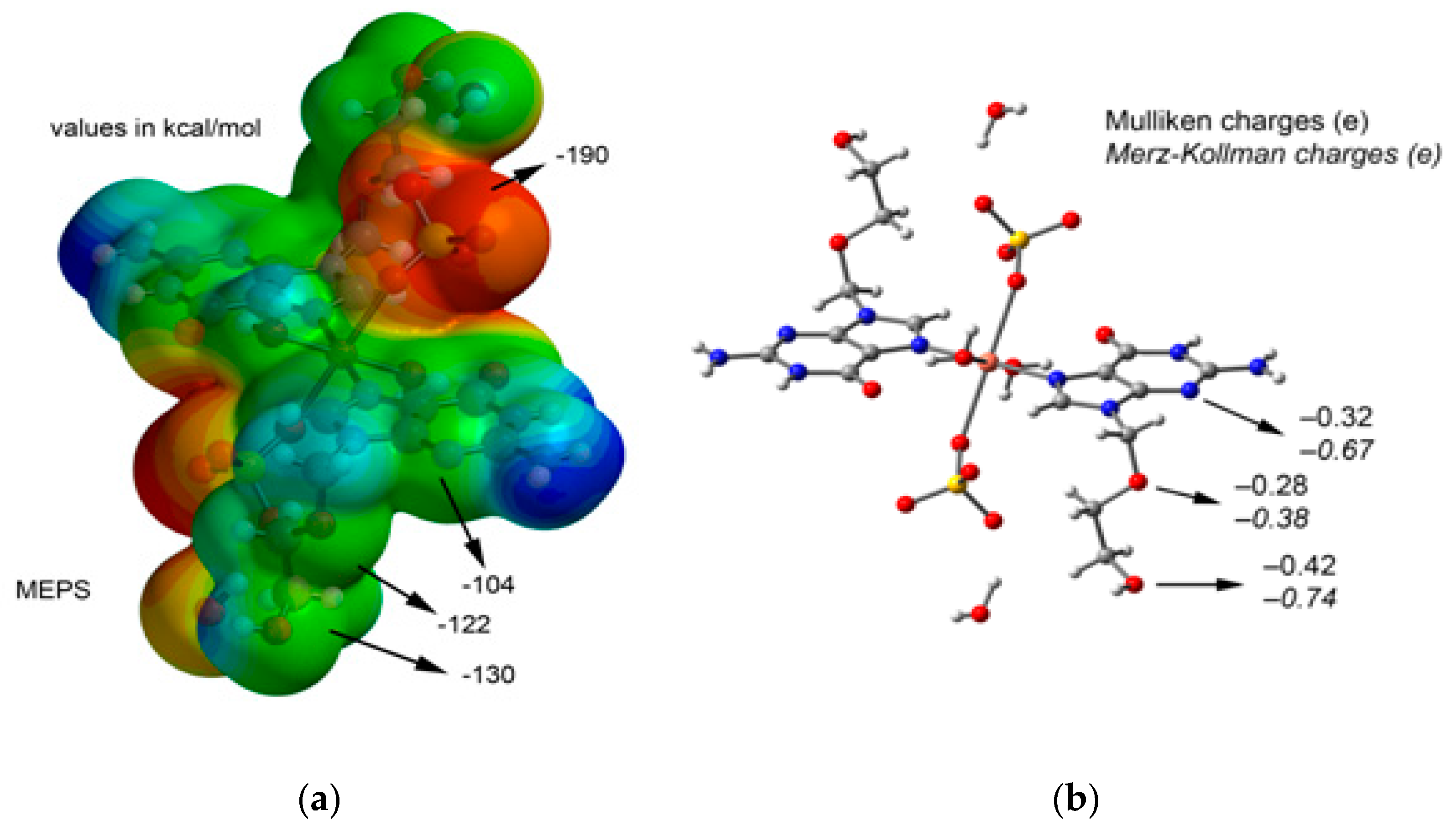
2. Results and Discussion
3. Materials and Methods
3.1. Synthesis of Compound 1
3.2. Crystal Structure Determination
3.3. Theoretical Calculations
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
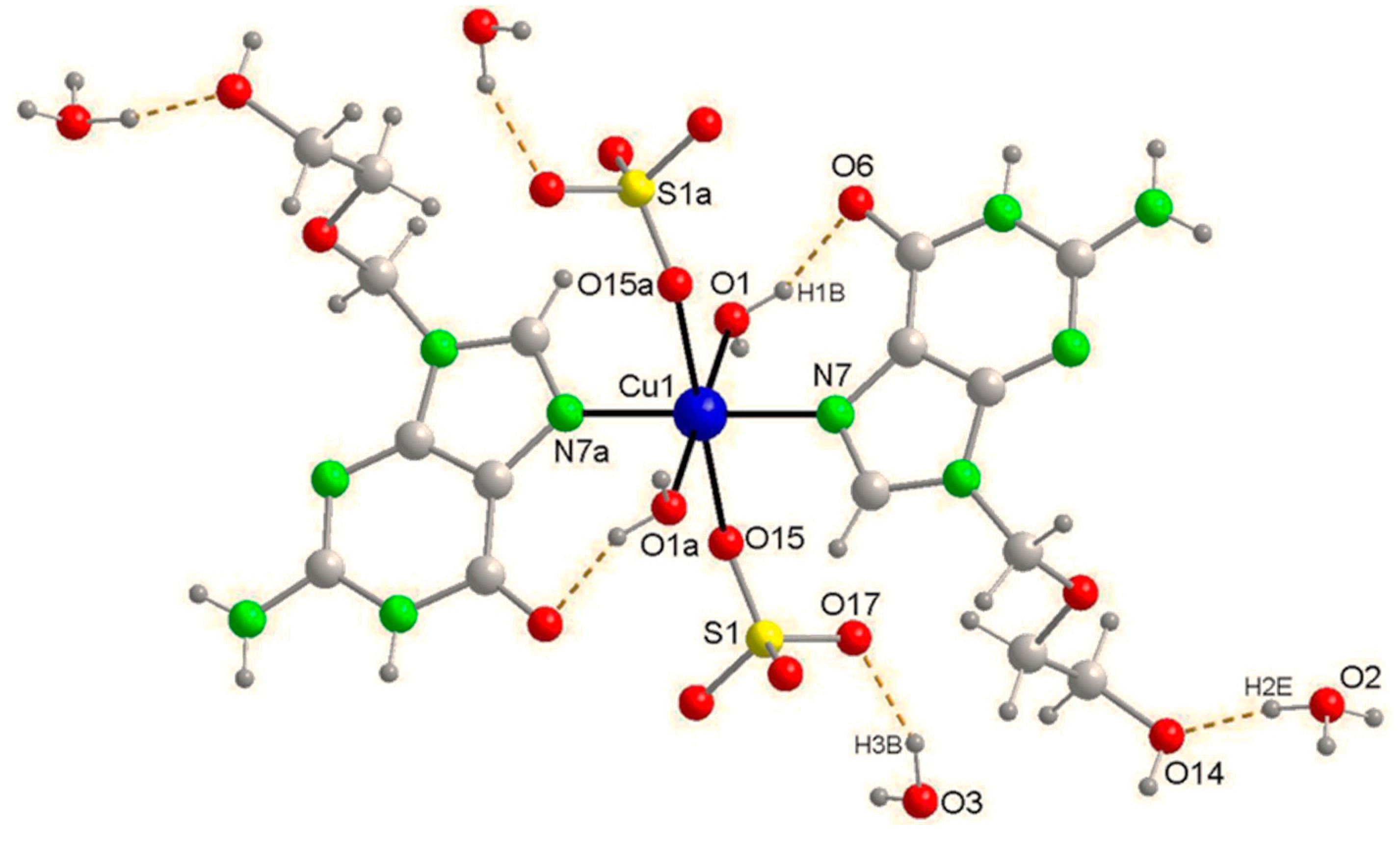
Appendix A1. Structural Data
| Identification code | 14jnac876 |
|---|---|
| Empirical formula | C16H36CuN10O20S2 |
| Formula weight | 816.21 |
| Crystal system, space group | Monoclinic, P21/c |
| Unit cell dimensions | a = 12.1489(4)Å, α = 90° |
| b = 18.2712(5)Å, β = 102.755(1)° | |
| c = 6.8294(2) Å, γ = 90° | |
| Volume | 1478.55(8) Å3 |
| Z, Calculated density | 2, 1.833 Mg/m3 |
| Absorption coefficient | 0.987 mm−1 |
| F(000) | 846 |
| Crystal size | 0.100 × 0.080 × 0.040 mm |
| Theta range for data collection (°) | 2.229 to 29.204 |
| Limiting indices | −15 ≤ h ≤ 16, −24 ≤ k ≤ 24, −9 ≤ l ≤ 9 |
| Reflections collected/unique | 19,513/3985 [Rint = 0.0392] |
| Completeness to θ = 25.242 | 99.8% |
| Absorption correction | Semi-empirical from equivalents |
| Max. and min. transmission | 1.0000 and 0.9069 |
| Refinement method | Full-matrix least-squares on F2 |
| Data/parameters | 3985/223 |
| Goodness-of-fit on F2 | 1.092 |
| Final R indices [I > 2σ(I)] | R1 = 0.0449, wR2 = 0.1042 |
| R indices (all data) | R1 = 0.0559, wR2 = 0.1099 |
| Largest diff. peak and hole | 1.260 and −0.907 e.Å−3 |
| Bond Lengths (Å) of Compound 1 | |
| Cu(1)–O(1) a | 1.9630(18) |
| Cu(1)–O(1) | 1.9630(18) |
| Cu(1)–N(7) a | 2.018(2) |
| Cu(1)–N(7) | 2.018(2) |
| Cu(1)–O(15) | 2.4271(17) |
| Cu(1)–O(15) a | 2.4271(17) |
| Angles (°) for Compound 1 | |
| O(1) a–Cu(1)–O(1) | 180.0 |
| O(1) a–Cu(1)–N(7) a | 90.48(8) |
| O(1)–Cu(1)–N(7) a | 89.52(8) |
| O(1) a–Cu(1)–N(7) | 89.52(8) |
| O(1)–Cu(1)–N(7) | 90.48(8) |
| N(7) a–Cu(1)–N(7) | 180.0 |
| O(1) a–Cu(1)–O(15) | 88.41(7) |
| O(1)–Cu(1)–O(15) | 91.59(7) |
| N(7) a–Cu(1)–O(15) | 86.78(7) |
| N(7)–Cu(1)–O(15) | 93.22(7) |
| O(1) a–Cu(1)–O(15) a | 91.59(7) |
| O(1)–Cu(1)–O(15) a | 88.41(7) |
| N(7) a–Cu(1)–O(15) a | 93.22(7) |
| N(7)–Cu(1)–O(15) a | 86.78(7) |
| O(15)–Cu(1)–O(15) a | 180.00(8) |
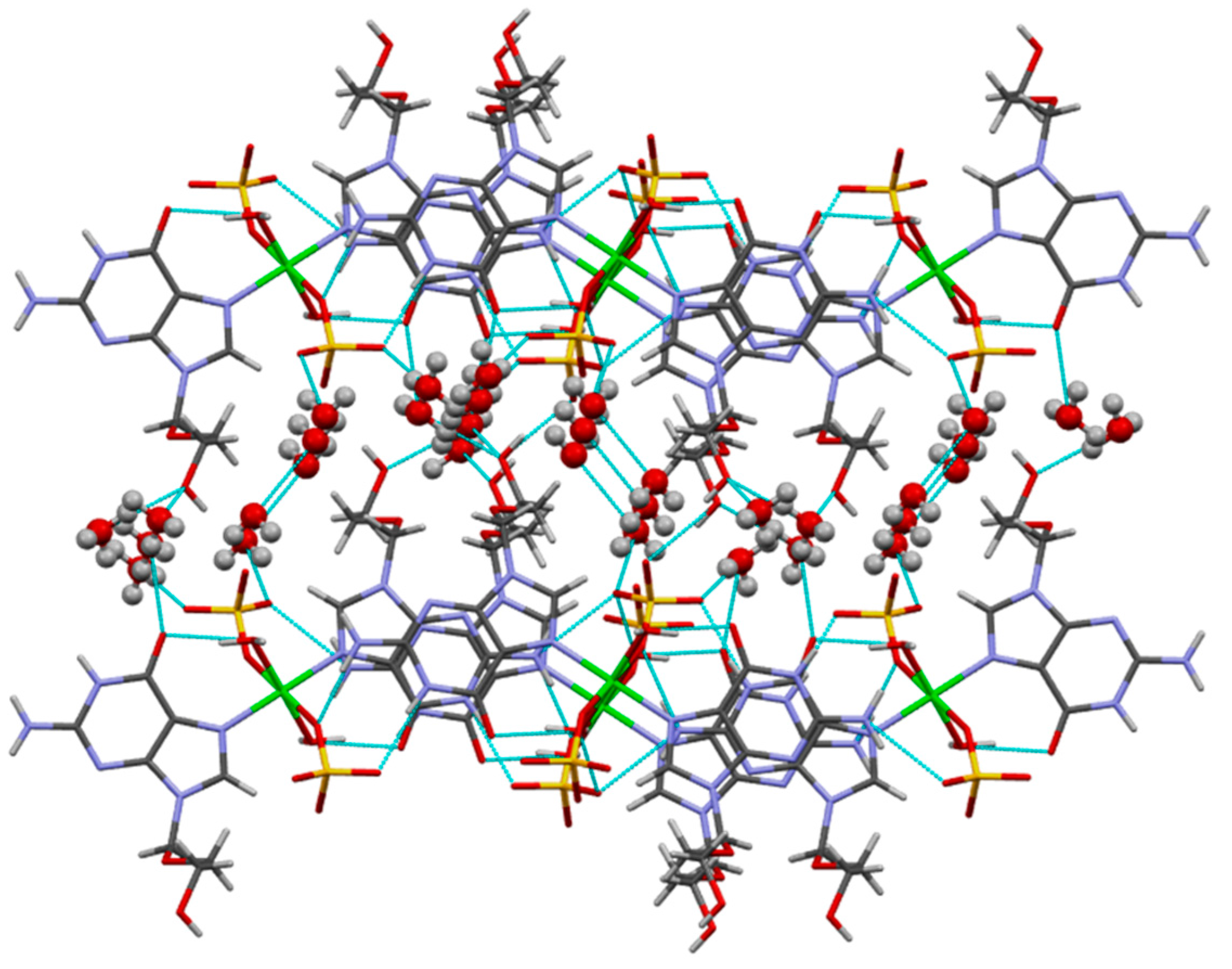
| D–H···A | d(D–H) | d(H···A) | d(D···A) | Z(DHA) |
|---|---|---|---|---|
| O(1)–H(1A)···O(17) | 0.87 | 2.17 | 2.727(3) | 121.3 |
| O(1)–H(1B)···O(6) | 0.87 | 1.79 | 2.615(3) | 157.3 |
| O(14)–H(14)···O(16) c | 0.84 | 1.82 | 2.647(3) | 166.2 |
| N(1)–H(1)···O(18) d | 0.88 | 1.96 | 2.832(3) | 170.2 |
| N(2)–H(2A)···O(17) e | 0.88 | 2.11 | 2.905(3) | 149.4 |
| N(2)–H(2B)···O(15) d | 0.88 | 2.08 | 2.859(3) | 147.3 |
| O(2)–H(2C)···O(6) f | 0.87 | 2.06 | 2.825(3) | 146.2 |
| O(2)–H(2D)···O(18) g | 0.87 | 1.96 | 2.808(3) | 165.5 |
| O(2)–H(2E)···O(14) | 0.98 | 1.82 | 2.786(3) | 170.4 |
| O(3)–H(3A)···O(14) h | 0.84 | 2.61 | 3.124(3) | 121.0 |
| O(3)–H(3A)···O(16) i | 0.84 | 2.59 | 3.101(3) | 120.2 |
| O(3)–H(3A)···O(2) j | 0.84 | 2.48 | 3.053(4) | 125.8 |
| O(3)–H(3B)···O(17) | 0.98 | 1.83 | 2.751(3) | 153.9 |
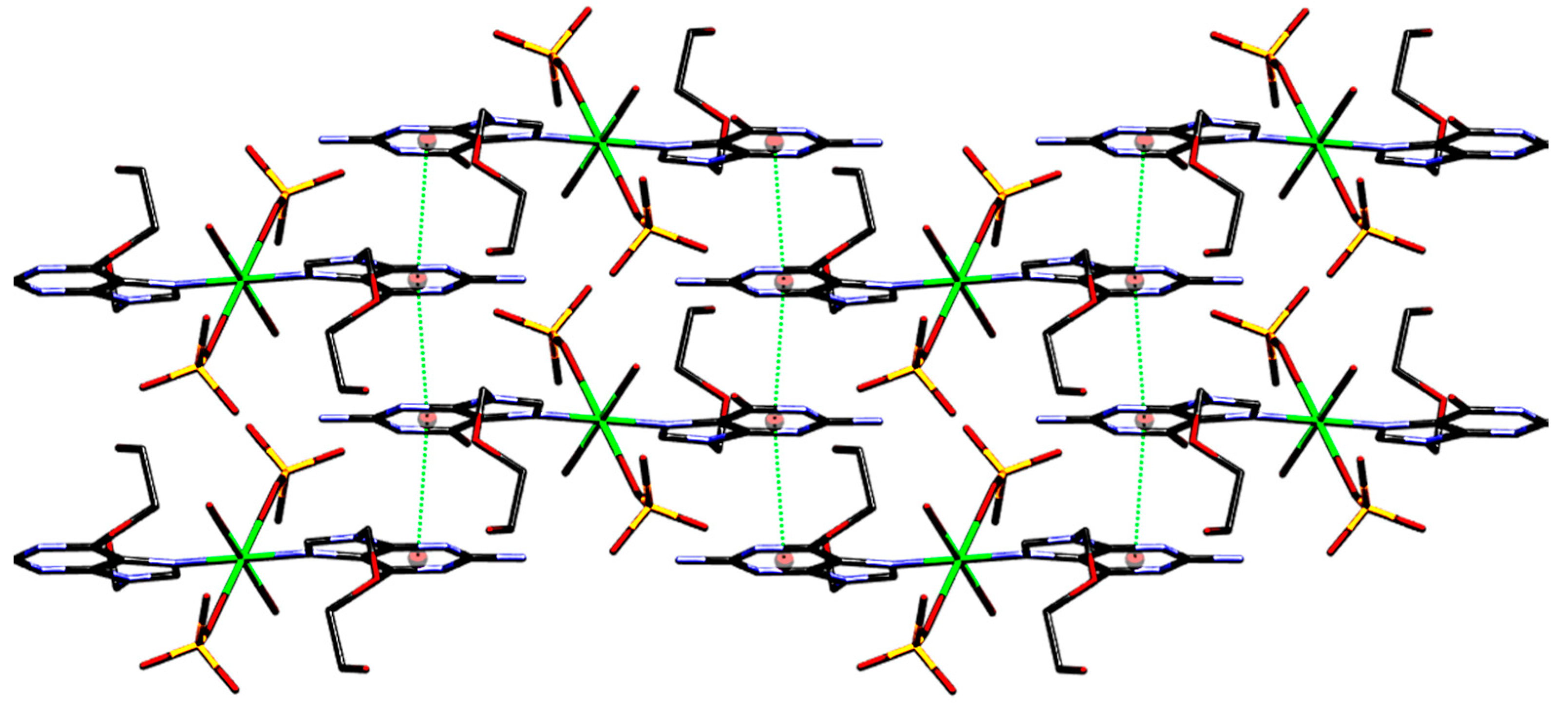
| π···π interactions | Cg(I)···Cg(J) | α |
|---|---|---|
| Cg(1)···Cg(1) e | 3.4235 | 3.00 |
| Cg(1)···Cg(1) k | 3.4235 | 3.00 |
Appendix A2. FT-IR Spectrum
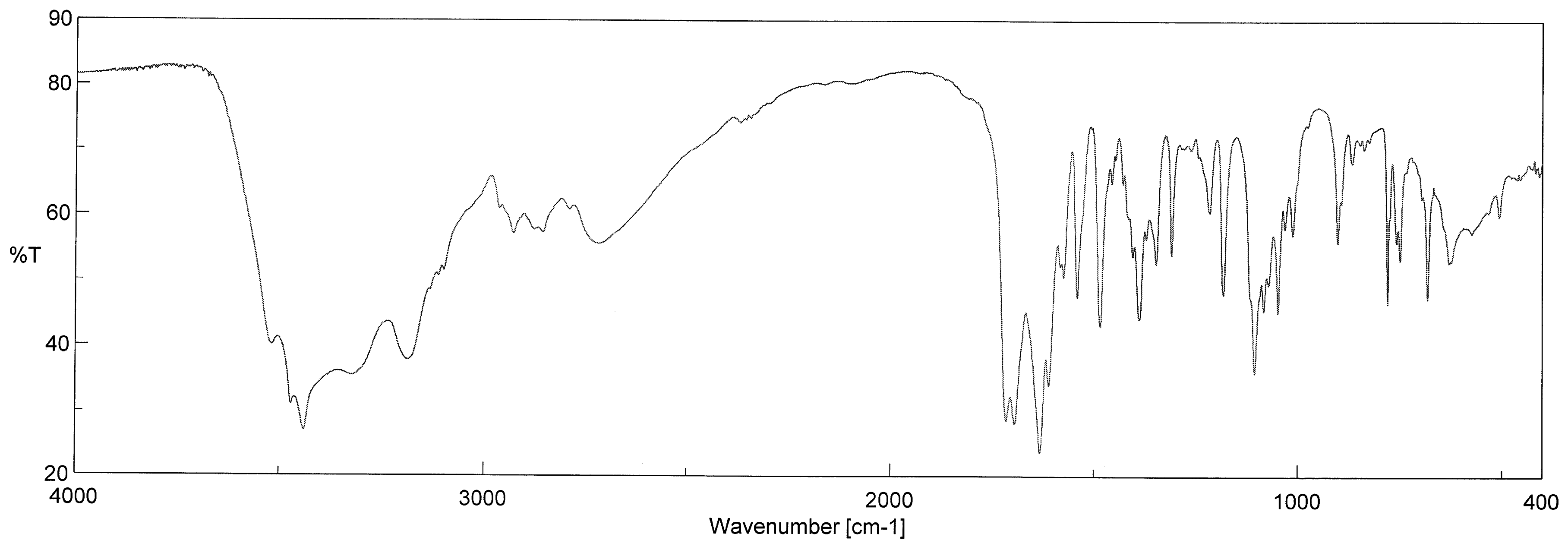
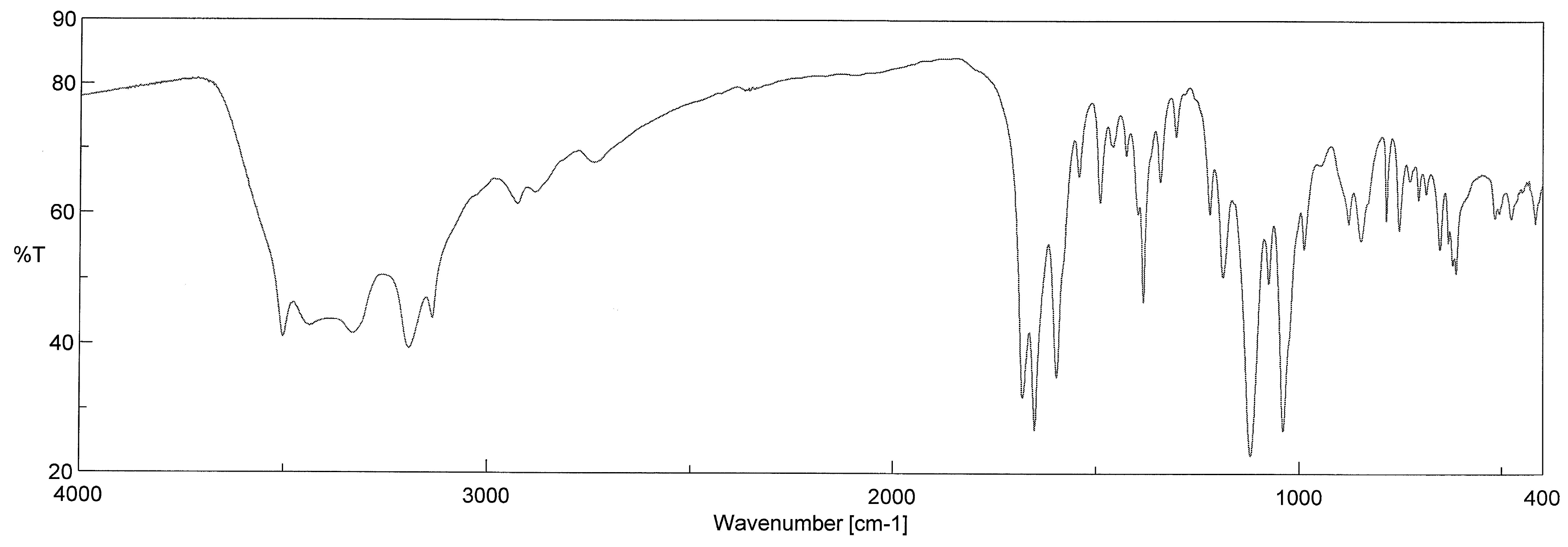
| Ligand or Solvent | Chromophore | Mode | Wavenumber (cm−1) | Band Number in the Read Spectrum |
|---|---|---|---|---|
| H3O+ ion | H3O+ | ν1(A1) and ν3(E) | 2743 (broad) | 9 |
| ν2(A1) | 1190 | 24 | ||
| H2O | H2O | νas | 3430 | 2 |
| νs | ~3450 | N/M * | ||
| δ | 1652 | 11 | ||
| acv | O(ol)–H | ν | 3502 | 1 |
| δ | 1385 | 20 | ||
| –N(2)H2 | νas | 3327 | 3 | |
| νs | 3195 | 4 | ||
| δ | 1598 | 12 | ||
| –N(1)–H | ν | 3139 | 5 | |
| δ | 1541 | 14 | ||
| –C=O(6) | ν | 1683 | 10 ** | |
| C–O(e)–C | νas | 1178 | 26 | |
| sulfate | SO42− | ν3 | 1122 | 25 |
| 1041 | 27 | |||
| ν1 | 989 | 28 | ||
| ν4 | 652 | 36 | ||
| 611 | 39 | |||
| ν2 | 448 | 44 |
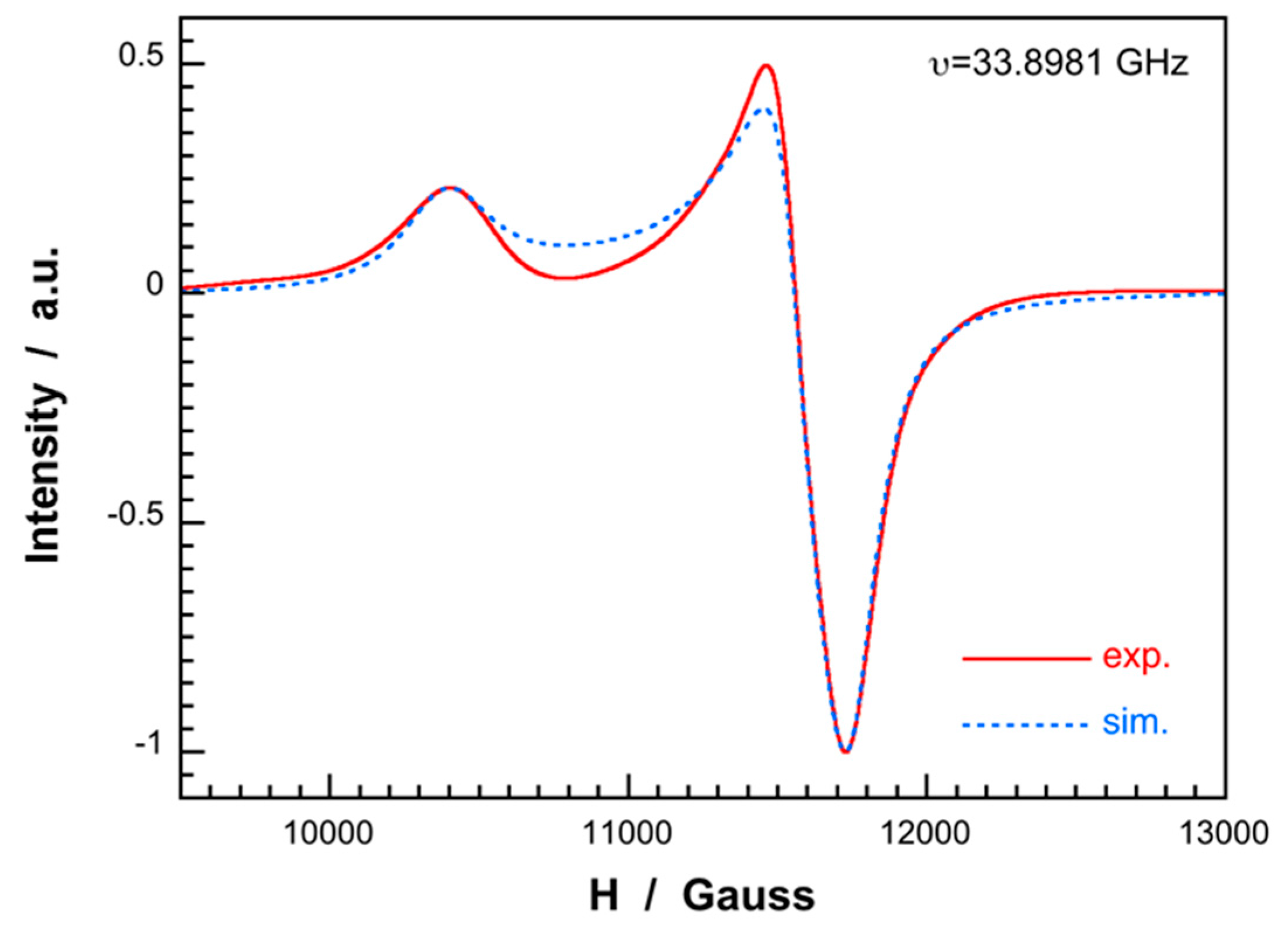
Appendix A3. ESR Spectrum and Magnetic Properties of Compound 1
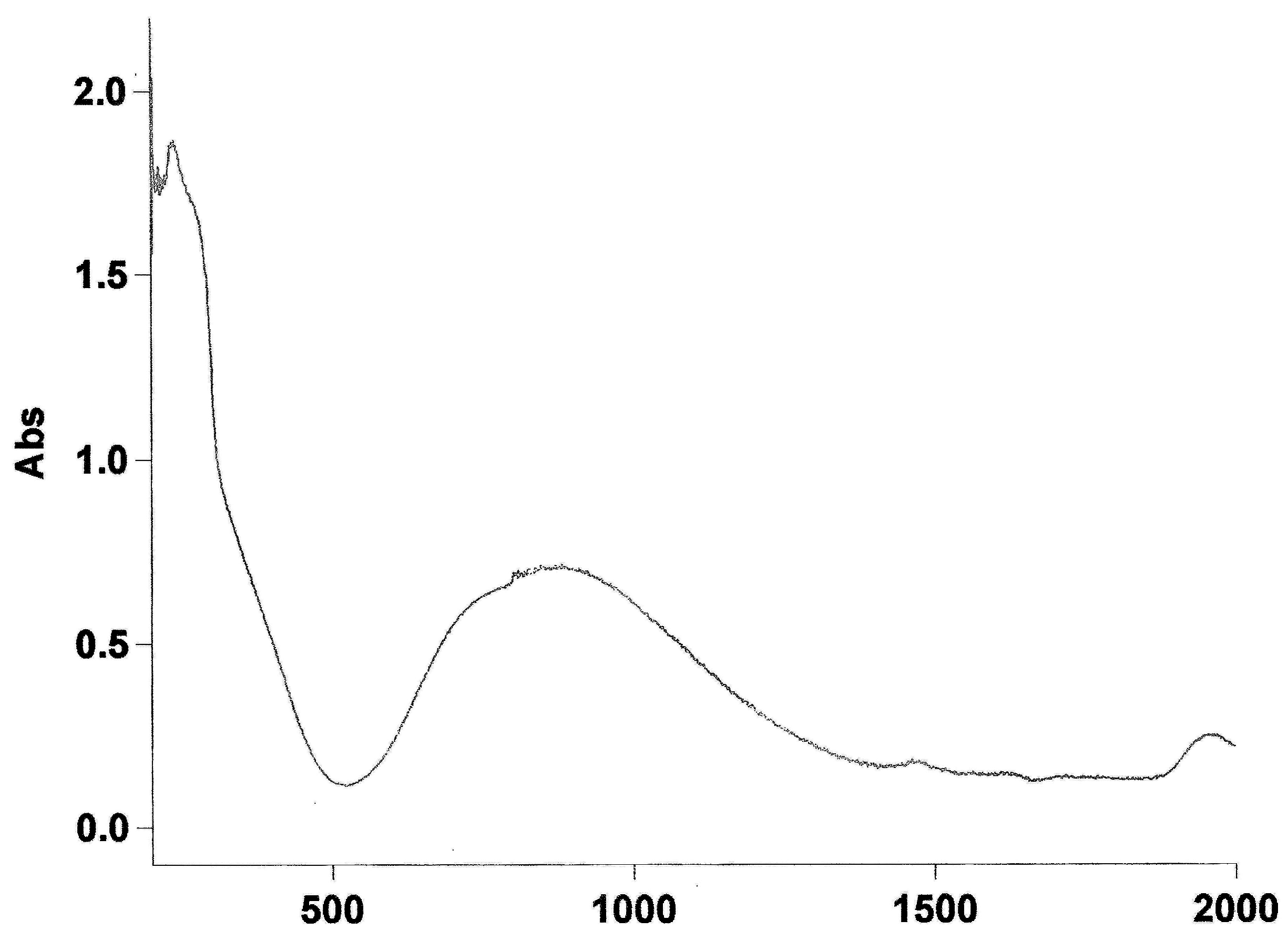
Appendix A4. Electronic Spectrum of Compound 1
A5. Cartesian Coordinates
| Cu | 0.00000055 | 9.13598209 | −0.00000659 |
| S | 1.95000055 | 10.31898209 | 2.88599341 |
| O | −1.55199945 | 8.26798209 | 0.83299341 |
| H | −1.60699945 | 8.50198209 | 1.67299341 |
| H | −1.46399945 | 7.39998209 | 0.78199341 |
| O | −1.59299945 | 5.76898209 | 0.06499341 |
| O | 4.64200055 | 5.81798209 | 2.09099341 |
| O | 5.76400055 | 6.14698209 | 4.77399341 |
| H | 6.38900055 | 6.69498209 | 4.64399341 |
| O | 0.71900055 | 10.04798209 | 2.13099341 |
| O | 3.14500055 | 10.12898209 | 2.04599341 |
| O | 2.01700055 | 9.38598209 | 4.04799341 |
| O | 1.90700000 | 11.71500000 | 3.40000000 |
| N | −0.21299945 | 3.97698209 | 0.19599341 |
| H | −0.92299945 | 3.45898209 | 0.14899341 |
| N | 1.00800055 | 2.02198209 | 0.31099341 |
| H | 1.76200055 | 1.57498209 | 0.37999341 |
| H | 0.24400055 | 1.58798209 | 0.24399341 |
| N | 2.16200055 | 4.02698209 | 0.40799341 |
| N | 1.08500055 | 7.44998209 | 0.22599341 |
| N | 2.97700055 | 6.29598209 | 0.45999341 |
| C | 1.02000055 | 3.35598209 | 0.30799341 |
| C | 1.97600055 | 5.35998209 | 0.36699341 |
| C | 0.79600055 | 6.08098209 | 0.22499341 |
| C | −0.42199945 | 5.34098209 | 0.15099341 |
| C | 2.38500055 | 7.52198209 | 0.37599341 |
| H | 2.86300055 | 8.34098209 | 0.42199341 |
| C | 4.39300055 | 6.03698209 | 0.72299341 |
| H | 4.68000055 | 5.24198209 | 0.20799341 |
| H | 4.92900055 | 6.80998209 | 0.41299341 |
| C | 4.54000055 | 7.03898209 | 2.85799341 |
| H | 3.70400055 | 7.51498209 | 2.61799341 |
| −H | 5.30400055 | 7.63198209 | 2.64799341 |
| C | 4.53600055 | 6.71398209 | 4.32899341 |
| H | 4.35800055 | 7.54198209 | 4.84099341 |
| H | 3.80100055 | 6.07898209 | 4.51699341 |
| S | −1.94999945 | 7.95198209 | −2.88600659 |
| O | 1.55200055 | 10.00298209 | −0.83300659 |
| H | 1.60700055 | 9.76998209 | −1.67300659 |
| H | 1.46400055 | 10.87098209 | −0.78200659 |
| O | 1.59300055 | 12.50198209 | −0.06500659 |
| O | −4.64199945 | 12.45298209 | −2.09100659 |
| O | −5.76399945 | 12.12398209 | −4.77400659 |
| H | −6.38899945 | 11.57698209 | −4.64400659 |
| O | −0.71899945 | 8.22298209 | −2.13100659 |
| O | −3.14499945 | 8.14198209 | −2.04600659 |
| O | −2.01699945 | 8.88498209 | −4.04800659 |
| O | −1.90699945 | 6.55698209 | −3.40000659 |
| N | 0.21300055 | 14.29398209 | −0.19600659 |
| H | 0.92300055 | 14.81198209 | −0.14900659 |
| N | −1.00799945 | 16.24898209 | −0.31100659 |
| H | −1.76199945 | 16.69598209 | −0.38000659 |
| H | −0.24399945 | 16.68298209 | −0.24400659 |
| N | −2.16199945 | 14.24398209 | −0.40800659 |
| N | −1.08499945 | 10.82198209 | −0.22600659 |
| N | −2.97699945 | 11.97498209 | −0.46000659 |
| C | −1.01999945 | 14.91498209 | −0.30800659 |
| C | −1.97599945 | 12.91198209 | −0.36700659 |
| C | −0.79599945 | 12.18998209 | −0.22500659 |
| C | 0.42200055 | 12.92998209 | −0.15100659 |
| C | −2.38499945 | 10.74898209 | −0.37600659 |
| H | −2.86299945 | 9.92998209 | −0.42200659 |
| C | −4.39299945 | 12.23398209 | −0.72300659 |
| H | −4.67999945 | 13.02898209 | −0.20800659 |
| H | −4.92899945 | 11.46198209 | −0.41300659 |
| C | −4.53999945 | 11.23198209 | −2.85800659 |
| H | −3.70399945 | 10.75598209 | −2.61800659 |
| H | −5.30399945 | 10.63898209 | −2.64800659 |
| C | −4.53599945 | 11.55698209 | −4.32900659 |
| H | −4.35799945 | 10.72898209 | −4.84100659 |
| H | −3.80099945 | 12.19198209 | −4.51700659 |
| O | 3.53500000 | 10.00600000 | 6.25700000 |
| H | 2.95000000 | 10.55300000 | 6.51100000 |
| H | 3.16700000 | 9.55200000 | 5.46800000 |
| O | −3.53500000 | 8.26500000 | −6.25700000 |
| H | −2.95000000 | 7.71800000 | −6.51100000 |
| H | −3.16700000 | 8.71900000 | −5.46800000 |
| (a) | |||
| Cu | 0.000 | 9.136 | 0.000 |
| S | 1.950 | 10.319 | 2.886 |
| O | −1.552 | 8.268 | 0.833 |
| H | −1.607 | 8.502 | 1.673 |
| H | −1.464 | 7.400 | 0.782 |
| O | −1.593 | 5.769 | 0.065 |
| O | 4.642 | 5.818 | 2.091 |
| O | 5.764 | 6.147 | 4.774 |
| H | 6.389 | 6.695 | 4.644 |
| O | 0.719 | 10.048 | 2.131 |
| O | 3.145 | 10.129 | 2.046 |
| O | 2.017 | 9.386 | 4.048 |
| O | 1.907 | 11.715 | 3.400 |
| N | −0.213 | 3.977 | 0.196 |
| H | −0.923 | 3.459 | 0.149 |
| N | 1.008 | 2.022 | 0.311 |
| H | 1.762 | 1.575 | 0.380 |
| H | 0.244 | 1.588 | 0.244 |
| N | 2.162 | 4.027 | 0.408 |
| N | 1.085 | 7.450 | 0.226 |
| N | 2.977 | 6.296 | 0.460 |
| C | 1.020 | 3.356 | 0.308 |
| C | 1.976 | 5.360 | 0.367 |
| C | 0.796 | 6.081 | 0.225 |
| C | −0.422 | 5.341 | 0.151 |
| C | 2.385 | 7.522 | 0.376 |
| H | 2.863 | 8.341 | 0.422 |
| C | 4.393 | 6.037 | 0.723 |
| H | 4.680 | 5.242 | 0.208 |
| H | 4.929 | 6.810 | 0.413 |
| C | 4.540 | 7.039 | 2.858 |
| H | 3.704 | 7.515 | 2.618 |
| H | 5.304 | 7.632 | 2.648 |
| C | 4.536 | 6.714 | 4.329 |
| H | 4.358 | 7.542 | 4.841 |
| H | 3.801 | 6.079 | 4.517 |
| S | −1.950 | 7.952 | −2.886 |
| O | 1.552 | 10.003 | −0.833 |
| H | 1.607 | 9.770 | −1.673 |
| H | 1.464 | 10.871 | −0.782 |
| O | 1.593 | 12.502 | −0.065 |
| O | −4.642 | 12.453 | −2.091 |
| O | −5.764 | 12.124 | −4.774 |
| H | −6.389 | 11.577 | −4.644 |
| O | −0.719 | 8.223 | −2.131 |
| O | −3.145 | 8.142 | −2.046 |
| O | −2.017 | 8.885 | −4.048 |
| O | −1.906 | 6.556 | −3.400 |
| N | 0.213 | 14.294 | −0.196 |
| H | 0.923 | 14.812 | −0.149 |
| N | −1.008 | 16.249 | −0.311 |
| H | −1.762 | 16.696 | −0.380 |
| H | −0.244 | 16.683 | −0.244 |
| N | −2.162 | 14.244 | −0.408 |
| N | −1.085 | 10.822 | −0.226 |
| N | −2.977 | 11.975 | −0.460 |
| C | −1.020 | 14.915 | −0.308 |
| C | −1.976 | 12.912 | −0.367 |
| C | −0.796 | 12.190 | −0.225 |
| C | 0.422 | 12.930 | −0.151 |
| C | −2.385 | 10.749 | −0.376 |
| H | −2.863 | 9.930 | −0.422 |
| C | −4.393 | 12.234 | −0.723 |
| H | −4.680 | 13.029 | −0.208 |
| H | −4.929 | 11.462 | −0.413 |
| C | −4.540 | 11.232 | −2.858 |
| H | −3.704 | 10.756 | −2.618 |
| H | −5.304 | 10.639 | −2.648 |
| C | −4.536 | 11.557 | −4.329 |
| H | −4.358 | 10.729 | −4.841 |
| H | −3.801 | 12.192 | −4.517 |
| H | 7.901 | 4.003 | 3.867 |
| H | 7.245 | 3.409 | 4.980 |
| H | 6.560 | 4.557 | 4.397 |
| O | −7.129 | 14.485 | −4.204 |
| H | −7.901 | 14.268 | −3.867 |
| H | −7.245 | 14.862 | −4.980 |
| H | −6.560 | 13.714 | −4.397 |
| (b) | |||
| Cu | 0.00000000 | 9.13600000 | 0.00000000 |
| S | 1.95000000 | 10.31900000 | 2.88600000 |
| O | −1.55200000 | 8.26800000 | 0.83300000 |
| H | −1.60700000 | 8.50200000 | 1.67300000 |
| H | −1.46400000 | 7.40000000 | 0.78200000 |
| O | −1.59300000 | 5.76900000 | 0.06500000 |
| O | 4.64200000 | 5.81800000 | 2.09100000 |
| O | 5.76400000 | 6.14700000 | 4.77400000 |
| H | 6.38900000 | 6.69500000 | 4.64400000 |
| O | 0.71900000 | 10.04800000 | 2.13100000 |
| O | 3.14500000 | 10.12900000 | 2.04600000 |
| O | 2.01700000 | 9.38600000 | 4.04800000 |
| O | 1.90699945 | 11.71501791 | 3.40000659 |
| N | −0.21300000 | 3.97700000 | 0.19600000 |
| H | −0.92300000 | 3.45900000 | 0.14900000 |
| N | 1.00800000 | 2.02200000 | 0.31100000 |
| H | 1.76200000 | 1.57500000 | 0.38000000 |
| H | 0.24400000 | 1.58800000 | 0.24400000 |
| N | 2.16200000 | 4.02700000 | 0.40800000 |
| N | 1.08500000 | 7.45000000 | 0.22600000 |
| N | 2.97700000 | 6.29600000 | 0.46000000 |
| C | 1.02000000 | 3.35600000 | 0.30800000 |
| C | 1.97600000 | 5.36000000 | 0.36700000 |
| C | 0.79600000 | 6.08100000 | 0.22500000 |
| C | −0.42200000 | 5.34100000 | 0.15100000 |
| C | 2.38500000 | 7.52200000 | 0.37600000 |
| H | 2.86300000 | 8.34100000 | 0.42200000 |
| C | 4.39300000 | 6.03700000 | 0.72300000 |
| H | 4.68000000 | 5.24200000 | 0.20800000 |
| H | 4.92900000 | 6.81000000 | 0.41300000 |
| C | 4.54000000 | 7.03900000 | 2.85800000 |
| H | 3.70400000 | 7.51500000 | 2.61800000 |
| H | 5.30400000 | 7.63200000 | 2.64800000 |
| C | 4.53600000 | 6.71400000 | 4.32900000 |
| H | 4.35800000 | 7.54200000 | 4.84100000 |
| H | 3.80100000 | 6.07900000 | 4.51700000 |
| S | −1.95000000 | 7.95200000 | −2.88600000 |
| O | 1.55200000 | 10.00300000 | −0.83300000 |
| H | 1.60700000 | 9.77000000 | −1.67300000 |
| H | 1.46400000 | 10.87100000 | −0.78200000 |
| O | 1.59300000 | 12.50200000 | −0.06500000 |
| O | −4.64200000 | 12.45300000 | −2.09100000 |
| O | −5.76400000 | 12.12400000 | −4.77400000 |
| H | −6.38900000 | 11.57700000 | −4.64400000 |
| O | −0.71900000 | 8.22300000 | −2.13100000 |
| O | −3.14500000 | 8.14200000 | −2.04600000 |
| O | −2.01700000 | 8.88500000 | −4.04800000 |
| O | −1.90700000 | 6.55700000 | −3.40000000 |
| N | 0.21300000 | 14.29400000 | −0.19600000 |
| H | 0.92300000 | 14.81200000 | −0.14900000 |
| N | −1.00800000 | 16.24900000 | −0.31100000 |
| H | −1.76200000 | 16.69600000 | −0.38000000 |
| H | −0.24400000 | 16.68300000 | −0.24400000 |
| N | −2.16200000 | 14.24400000 | −0.40800000 |
| N | −1.08500000 | 10.82200000 | −0.22600000 |
| N | −2.97700000 | 11.97500000 | −0.46000000 |
| C | −1.02000000 | 14.91500000 | −0.30800000 |
| C | −1.97600000 | 12.91200000 | −0.36700000 |
| C | −0.79600000 | 12.19000000 | −0.22500000 |
| C | 0.42200000 | 12.93000000 | −0.15100000 |
| C | −2.38500000 | 10.74900000 | −0.37600000 |
| H | −2.86300000 | 9.93000000 | −0.42200000 |
| C | −4.39300000 | 12.23400000 | −0.72300000 |
| H | −4.68000000 | 13.02900000 | −0.20800000 |
| H | −4.92900000 | 11.46200000 | −0.41300000 |
| C | −4.54000000 | 11.23200000 | −2.85800000 |
| H | −3.70400000 | 10.75600000 | −2.61800000 |
| H | −5.30400000 | 10.63900000 | −2.64800000 |
| C | −4.53600000 | 11.55700000 | −4.32900000 |
| H | −4.35800000 | 10.72900000 | −4.84100000 |
| H | −3.80100000 | 12.19200000 | −4.51700000 |
| O | 7.12900000 | 3.78600000 | 4.20400000 |
| H | 7.90100000 | 4.00300000 | 3.86700000 |
| H | 7.24500000 | 3.40900000 | 4.98000000 |
| H | 6.56000000 | 4.55700000 | 4.39700000 |
| O | −4.65114130 | 15.47000153 | −0.72450806 |
| H | −4.64493571 | 15.98253054 | −1.42730071 |
| H | −4.75966993 | 15.95560247 | −0.01024322 |
| H | −3.80193352 | 15.01038869 | −0.57267532 |
| (c) | |||
| Cu | 0.000 | 9.136 | 0.000 |
| S | 1.950 | 10.319 | 2.886 |
| O | −1.552 | 8.268 | 0.833 |
| H | −1.607 | 8.502 | 1.673 |
| H | −1.464 | 7.400 | 0.782 |
| O | −1.593 | 5.769 | 0.065 |
| O | 4.642 | 5.818 | 2.091 |
| O | 5.764 | 6.147 | 4.774 |
| H | 6.389 | 6.695 | 4.644 |
| O | 0.719 | 10.048 | 2.131 |
| O | 3.145 | 10.129 | 2.046 |
| O | 2.017 | 9.386 | 4.048 |
| O | 1.907 | 11.715 | 3.400 |
| N | −0.213 | 3.977 | 0.196 |
| H | −0.923 | 3.459 | 0.149 |
| N | 1.008 | 2.022 | 0.311 |
| H | 1.762 | 1.575 | 0.380 |
| H | 0.244 | 1.588 | 0.244 |
| N | 2.162 | 4.027 | 0.408 |
| N | 1.085 | 7.450 | 0.226 |
| N | 2.977 | 6.296 | 0.460 |
| C | 1.020 | 3.356 | 0.308 |
| C | 1.976 | 5.360 | 0.367 |
| C | 0.796 | 6.081 | 0.225 |
| C | −0.422 | 5.341 | 0.151 |
| C | 2.385 | 7.522 | 0.376 |
| H | 2.863 | 8.341 | 0.422 |
| C | 4.393 | 6.037 | 0.723 |
| H | 4.680 | 5.242 | 0.208 |
| H | 4.929 | 6.810 | 0.413 |
| C | 4.540 | 7.039 | 2.858 |
| H | 3.704 | 7.515 | 2.618 |
| H | 5.304 | 7.632 | 2.648 |
| C | 4.536 | 6.714 | 4.329 |
| H | 4.358 | 7.542 | 4.841 |
| H | 3.801 | 6.079 | 4.517 |
| S | −1.950 | 7.952 | −2.886 |
| O | 1.552 | 10.003 | −0.833 |
| H | 1.607 | 9.770 | −1.673 |
| H | 1.464 | 10.871 | −0.782 |
| O | 1.593 | 12.502 | −0.065 |
| O | −4.642 | 12.453 | −2.091 |
| O | −5.764 | 12.124 | −4.774 |
| H | −6.389 | 11.577 | −4.644 |
| O | −0.719 | 8.223 | −2.131 |
| O | −3.145 | 8.142 | −2.046 |
| O | −2.017 | 8.885 | −4.048 |
| O | −1.906 | 6.556 | −3.400 |
| N | 0.213 | 14.294 | −0.196 |
| H | 0.923 | 14.812 | −0.149 |
| N | −1.008 | 16.249 | −0.311 |
| H | −1.762 | 16.696 | −0.380 |
| H | −0.244 | 16.683 | −0.244 |
| N | −2.162 | 14.244 | −0.408 |
| N | −1.085 | 10.822 | −0.226 |
| N | −2.977 | 11.975 | −0.460 |
| C | −1.020 | 14.915 | −0.308 |
| C | −1.976 | 12.912 | −0.367 |
| C | −0.796 | 12.190 | −0.225 |
| C | 0.422 | 12.930 | −0.151 |
| C | −2.385 | 10.749 | −0.376 |
| H | −2.863 | 9.930 | −0.422 |
| C | −4.393 | 12.234 | −0.723 |
| H | −4.680 | 13.029 | −0.208 |
| H | −4.929 | 11.462 | −0.413 |
| C | −4.540 | 11.232 | −2.858 |
| H | −3.704 | 10.756 | −2.618 |
| H | −5.304 | 10.639 | −2.648 |
| C | −4.536 | 11.557 | −4.329 |
| H | −4.358 | 10.729 | −4.841 |
| H | −3.801 | 12.192 | −4.517 |
| O | 7.129 | 3.786 | 4.204 |
| H | 7.901 | 4.003 | 3.867 |
| H | 7.245 | 3.409 | 4.980 |
| H | 6.560 | 4.557 | 4.397 |
| O | −7.129 | 14.485 | −4.204 |
| H | −7.901 | 14.268 | −3.867 |
| H | −7.245 | 14.862 | −4.980 |
| H | −6.560 | 13.714 | −4.397 |
| C | 9.887 | 0.000 | 9.991 |
| S | 7.937 | 1.183 | 7.106 |
| O | 11.439 | −0.867 | 9.159 |
| H | 11.494 | −0.634 | 8.318 |
| H | 11.351 | −1.736 | 9.209 |
| O | 11.480 | −3.366 | 9.927 |
| O | 5.245 | −3.317 | 7.900 |
| O | 4.123 | −2.988 | 5.217 |
| H | 3.498 | −2.441 | 5.347 |
| O | 9.168 | 0.912 | 7.860 |
| O | 6.742 | 0.993 | 7.946 |
| O | 7.870 | 0.250 | 5.943 |
| O | 7.980 | 2.579 | 6.592 |
| N | 10.100 | −5.158 | 9.795 |
| H | 10.810 | −5.677 | 9.842 |
| N | 8.880 | −7.114 | 9.680 |
| H | 8.125 | −7.561 | 9.612 |
| H | 9.644 | −7.548 | 9.747 |
| N | 7.725 | −5.108 | 9.583 |
| N | 8.802 | −1.686 | 9.765 |
| N | 6.910 | −2.839 | 9.532 |
| C | 8.867 | −5.780 | 9.683 |
| C | 7.911 | −3.776 | 9.624 |
| C | 9.091 | −3.054 | 9.766 |
| C | 10.309 | −3.795 | 9.841 |
| C | 7.503 | −1.613 | 9.615 |
| H | 7.024 | −0.795 | 9.570 |
| C | 5.494 | −3.099 | 9.269 |
| H | 5.207 | −3.894 | 9.783 |
| H | 4.958 | −2.326 | 9.578 |
| C | 5.348 | −2.097 | 7.134 |
| H | 6.183 | −1.621 | 7.374 |
| H | 4.583 | −1.504 | 7.344 |
| C | 5.352 | −2.421 | 5.662 |
| H | 5.529 | −1.593 | 5.150 |
| H | 6.087 | −3.057 | 5.475 |
| S | 11.837 | −1.183 | 12.877 |
| O | 8.335 | 0.867 | 10.824 |
| H | 8.280 | 0.634 | 11.665 |
| H | 8.424 | 1.736 | 10.773 |
| O | 8.295 | 3.366 | 10.056 |
| O | 14.529 | 3.317 | 12.082 |
| O | 15.651 | 2.988 | 14.765 |
| H | 16.276 | 2.441 | 14.635 |
| O | 10.606 | −0.912 | 12.122 |
| O | 13.032 | −0.993 | 12.037 |
| O | 11.904 | −0.250 | 14.040 |
| O | 11.794 | −2.579 | 13.391 |
| N | 9.674 | 5.158 | 10.187 |
| H | 8.964 | 5.677 | 10.141 |
| N | 10.895 | 7.114 | 10.302 |
| H | 11.649 | 7.561 | 10.371 |
| H | 10.131 | 7.548 | 10.236 |
| N | 12.049 | 5.108 | 10.400 |
| N | 10.972 | 1.686 | 10.218 |
| N | 12.865 | 2.839 | 10.451 |
| C | 10.907 | 5.780 | 10.300 |
| C | 11.863 | 3.776 | 10.358 |
| C | 10.683 | 3.054 | 10.216 |
| C | 9.466 | 3.795 | 10.142 |
| C | 12.272 | 1.613 | 10.368 |
| H | 12.750 | 0.795 | 10.413 |
| C | 14.281 | 3.099 | 10.714 |
| H | 14.567 | 3.894 | 10.200 |
| H | 14.816 | 2.326 | 10.404 |
| C | 14.427 | 2.097 | 12.849 |
| H | 13.592 | 1.621 | 12.609 |
| H | 15.191 | 1.504 | 12.639 |
| C | 14.423 | 2.421 | 14.320 |
| H | 14.245 | 1.593 | 14.832 |
| H | 13.688 | 3.057 | 14.508 |
| O | 12.646 | −3.786 | 15.779 |
| H | 11.873 | −4.003 | 16.115 |
| H | 12.530 | −3.409 | 15.003 |
| H | 13.214 | −4.557 | 15.586 |
| O | 2.759 | −5.350 | 5.788 |
| H | 1.986 | −5.132 | 6.124 |
| H | 2.643 | −5.726 | 5.012 |
| H | 3.327 | −4.579 | 5.594 |
References
- Caceres, R.A.; Timmers, L.F.S.M.; Ducati, R.G.; da Silva, D.O.N.; Basso, L.A.; de Azevedo, W.F., Jr.; Santos, D.S. Crystal structure and molecular dynamics studies of purine nucleoside phosphorylase from Mycobacterium tuberculosis associated with acyclovir. Biochimie 2011, 94, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Grabner, S.; Plavec, J.; Bukovec, N.; Di Leo, D.; Cini, R.; Natile, G. Synthesis and structural characterization of platinum(II)-acyclovir complexes. J. Chem. Soc. Dalton Trans. 1998, 1447–1452. [Google Scholar] [CrossRef]
- Garcia-Raso, A.; Fiol, J.J.; Badenas, F.; Cons, R.; Terron, A.; Quiros, M. Synthesis and structural characteristics of metal–acyclovir (ACV) complexes: [Ni(or Co)(ACV)2(H2O)4]Cl2·2ACV, [Zn(ACV)Cl2(H2O)], [Cd(ACV)Cl2]·H2O and [{Hg(ACV)Cl2}x]. Recognition of acyclovir by Ni–ACV. J. Chem. Soc. Dalton Trans. 1999, 167–174. [Google Scholar] [CrossRef]
- Barceló-Oliver, M.; Terrón, A.; García-Raso, A.; Fiol, J.J.; Molins, E.; Miravitlles, C. Ternary complexes metal [Co(II), Ni(II), Cu(II) and Zn(II)]–ortho-iodohippurate (I-hip)–acyclovir. X-ray characterization of isostructural [(Co, Ni or Zn)(I−hip)2(ACV)(H2O)3] with stacking as a recognition factor. J. Inorg. Biochem. 2004, 98, 1703–1711. [Google Scholar] [CrossRef] [PubMed]
- Blažič, B.; Turel, I.; Bukovec, N.; Bukovec, P.; Lazarini, F. Synthesis and structure of diaquadichlorobis {9−[(2−hydroxyethoxy)methyl]guanine} copper(II). J. Inorg. Biochem. 1993, 51, 737–744. [Google Scholar] [CrossRef]
- Turel, I.; Pečanac, M.; Golobič, A.; Alessio, E.; Serli, B. Novel RuIII-DMSO Complexes of the Antiherpes Drug Acyclovir. Eur. J. Inorg. Chem. 2002, 1928–1931. [Google Scholar] [CrossRef]
- Turel, I.; Pečanac, M.; Golobič, A.; Alessio, E.; Serli, B.; Bergamo, A.; Sava, G. Solution, solid state and biological characterization of ruthenium(III)-DMSO complexes with purine base derivatives. J. Inorg. Biochem. 2004, 98, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Brandi-Blanco, M.P.; Choquesillo-Lazarte, D.; Domínguez-Martín, A.; González-Pérez, J.M.; Castiñeiras, A.; Niclós-Gutiérrez, J. Metal ion binding patterns of acyclovir: Molecular recognition between this antiviral agent and copper(II) chelates with iminodiacetate or glycylglycinate. J. Inorg. Biochem. 2011, 105, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Toro, I.; Domínguez-Martín, A.; Choquesillo-Lazarte, D.; Vílchez-Rodríguez, E.; González-Pérez, J.M.; Castiñeiras, A.; Niclós-Gutiérrez, J. Lights and shadows in the challenge of binding acyclovir, a synthetic purine-like nucleoside with antiviral activity, at an apical–distal coordination site in copper(II)-polyamine chelates. J. Inorg. Biochem. 2015, 148, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Sinur, A.; Grabner, S. A Platinum(II) Diammine Complex: Cis-[Pt(C8H11N5O3)2(NH3)2]Cl2·2H2O. Acta Crystallogr. 1995, C51, 1769–1772. [Google Scholar] [CrossRef]
- Turel, I.; Anderson, B.; Sletten, E.; White, A.J.P.; Williams, D.J. New studies in the copper(II) acyclovir (acv) system. NMR relaxation studies and the X-ray crystal structure of [Cu(acv)2(H2O)2](NO3)2. Polyhedron 1998, 17, 4195–4201. [Google Scholar] [CrossRef]
- Vílchez-Rodríguez, E.; Choquesillo-Lazarte, D.; Domínguez-Martín, A.; Pérez-Toro, I.; Matilla-Hernández, A.; González-Pérez, J.M.; Castiñeiras, A.; Niclós-Gutiérrez, J. Synthetic purine-nucleoside analogs as useful ligands: Looking at the coordination chemistry and metal binding patterns of acyclovir. J. Coord. Chem. Rev. 2016. in Press. [Google Scholar]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination CompoundsPart A, 6th ed.; Wiley: New York, NY, USA, 2009; pp. 173–176. [Google Scholar]
- APEX2 Software, v2010.3–0; Bruker AXS Inc.: Madison, WI, USA, 2010.
- Sheldrick, G.M. SADABS—Program for Empirical Absorption Correction of Area Detector Data; University of Goettingen: Göttingen, Germany, 1997. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.J.C. International Tables for Crystallography, Vol. C; Kluwer Academic Publishers: Dordrecht, Netherlands, 1995. [Google Scholar]
- Putz, H.; Brandenburg, K. DIAMOND—Crystal and Molecular Structure Visualization. Crystal Impact GbR: Bonn, Germany. Available online: http://www.crystalimpact.com/diamond (accessed on 22 October 2016).
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Perdew, J.P. Density-functional approximation for the correlation energy of the inhomogeneous electron gas. Phys. Rev. B 1986, 33, 8822–8824. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Ahlrichs, R.; Bär, M.; Häser, M.; Horn, H.; Kölmel, C. Electronic structure calculations on workstation computers: The program system turbomole. Chem. Phys. Lett. 1989, 162, 165–169. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parameterization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [PubMed]
- Spartan’10, v. 1.1.0; Wavefunction Inc.: Irvin, CA, USA, 2013.
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Rassolov, V.A.; Pople, J.A.; Ratner, M.A.; Windus, T.L. 6–31G* basis set for atoms K through Zn. J. Chem. Phys. 1998, 109, 1223–1229. [Google Scholar] [CrossRef]


© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vílchez-Rodríguez, E.; Pérez-Toro, I.; Bauzá, A.; Matilla-Hernández, A. Structural and Theoretical Evidence of the Depleted Proton Affinity of the N3-Atom in Acyclovir. Crystals 2016, 6, 139. https://doi.org/10.3390/cryst6110139
Vílchez-Rodríguez E, Pérez-Toro I, Bauzá A, Matilla-Hernández A. Structural and Theoretical Evidence of the Depleted Proton Affinity of the N3-Atom in Acyclovir. Crystals. 2016; 6(11):139. https://doi.org/10.3390/cryst6110139
Chicago/Turabian StyleVílchez-Rodríguez, Esther, Inmaculada Pérez-Toro, Antonio Bauzá, and Antonio Matilla-Hernández. 2016. "Structural and Theoretical Evidence of the Depleted Proton Affinity of the N3-Atom in Acyclovir" Crystals 6, no. 11: 139. https://doi.org/10.3390/cryst6110139
APA StyleVílchez-Rodríguez, E., Pérez-Toro, I., Bauzá, A., & Matilla-Hernández, A. (2016). Structural and Theoretical Evidence of the Depleted Proton Affinity of the N3-Atom in Acyclovir. Crystals, 6(11), 139. https://doi.org/10.3390/cryst6110139








