3.1. Fe-DNA
Fe-DNA is a characteristic complex in
M-DNAs [
8,
13,
14,
15,
16,
17,
19]. The source materials of Fe-DNA are a transparent aqueous solution of DNA and light greenish FeCl
2, whereas Fe-DNA films show characteristic ocher color of FeCl
3, as shown in
Figure 2, which suggests the presence of Fe
3+ in Fe-DNA as a result of a charge transfer from Fe
2+ to DNA bases. XAFS (X-ray absorption fine structure) study also supports the valence state of Fe
3+ in Fe-DNA [
19]. The formation rate of Fe-DNA with Fe
3+ depends on the species of base pairs—adenine–thymine or guanine–cytosine—as demonstrated by the UV/Vis absorption study [
8]. Since Fe
3+ with five
d electrons like Mn
2+ has
(high-spin) or
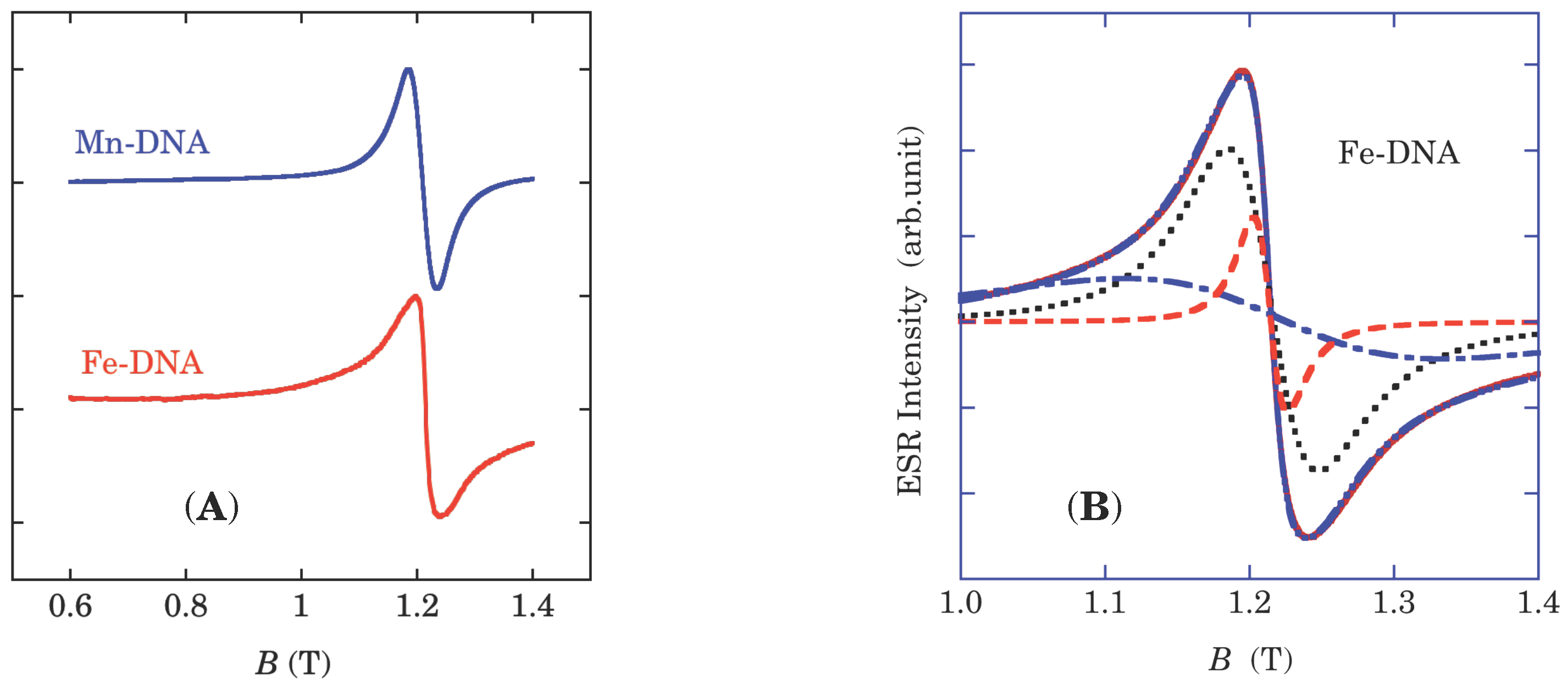
(low-spin) without the electron orbital moment, ESR signals of both Fe-DNA and B-form Mn-DNA are expected to appear around
g = 2, as actually found in
Figure 3A [
13,
14,
16,
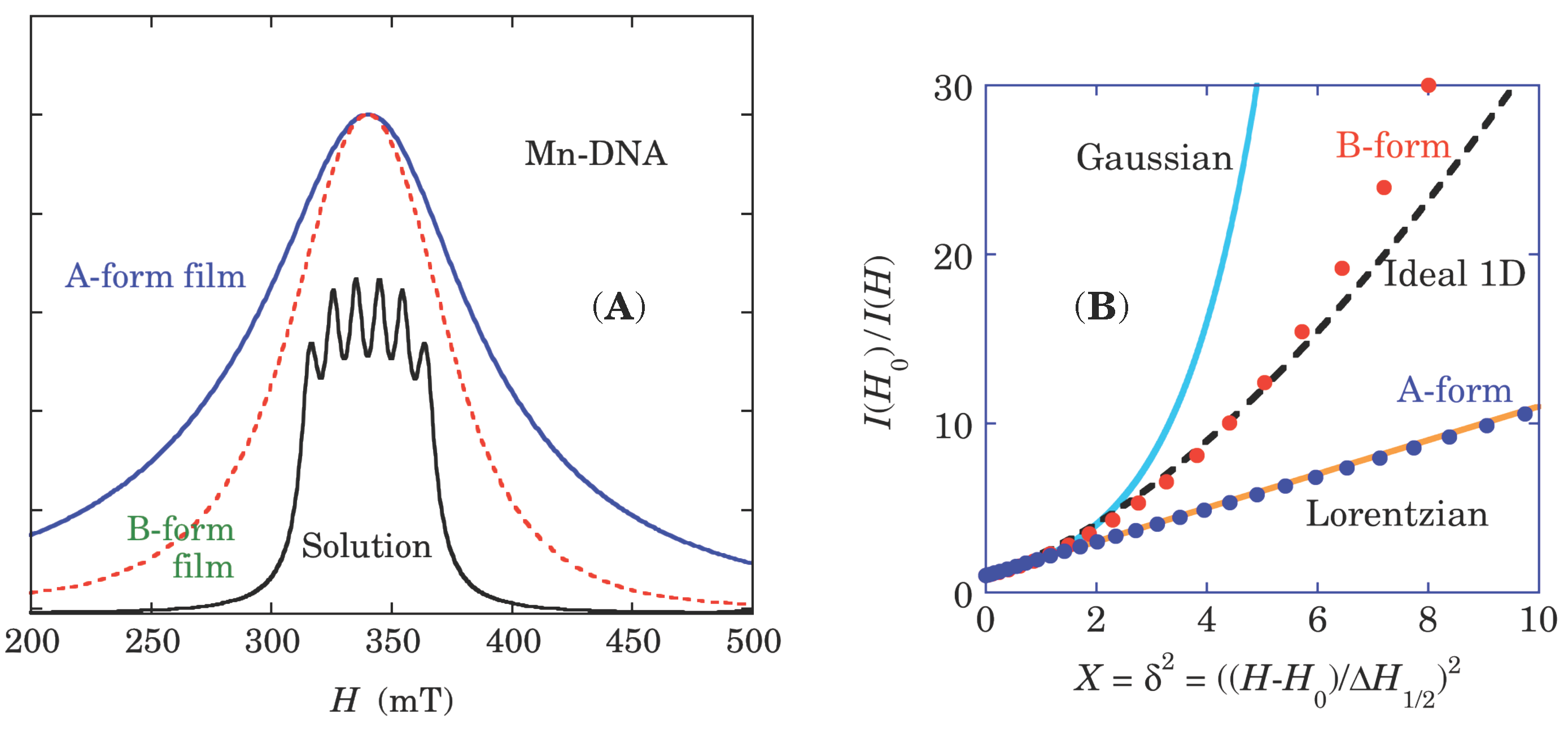
19]. The B-form Mn-DNA has an ESR lineshape characteristic of 1D spin correlation of the exchange interaction between Lorentzian and Gaussian [
17]. In contrast, Fe-DNA shows long tails more than ±0.1 T, which is attributable to the presence of three ESR components, as demonstrated in
Figure 3B [
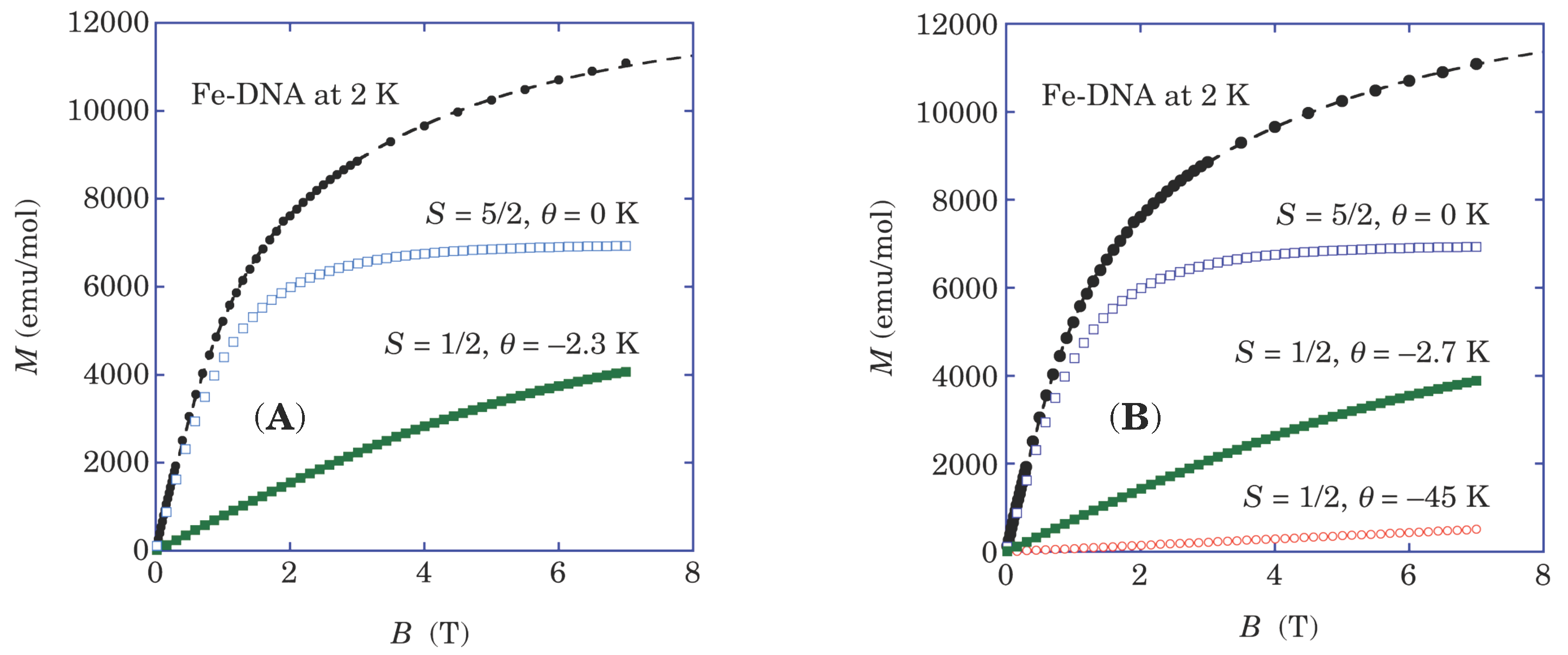
19]. The corresponding multiplicity of the spin species also observed in the magnetization curve of Fe-DNA at 2 K, as shown in
Figure 4. These observations suggest that both of the high-spin and the low-spin states of Fe
3+ coexist in Fe-DNA. The ratio of the spin number
N of the high-spin–the low-spin states is 1–3.35 on average. On the other hand, since the magnetization is proportional to
N ×
S, the ratio of the saturation magnetization is 1–0.67.
The spin state of Fe
3+ depends on local symmetry and magnitude of the crystal field around the Fe ion in Fe-DNA, in which water molecules surely play some role. As shown in
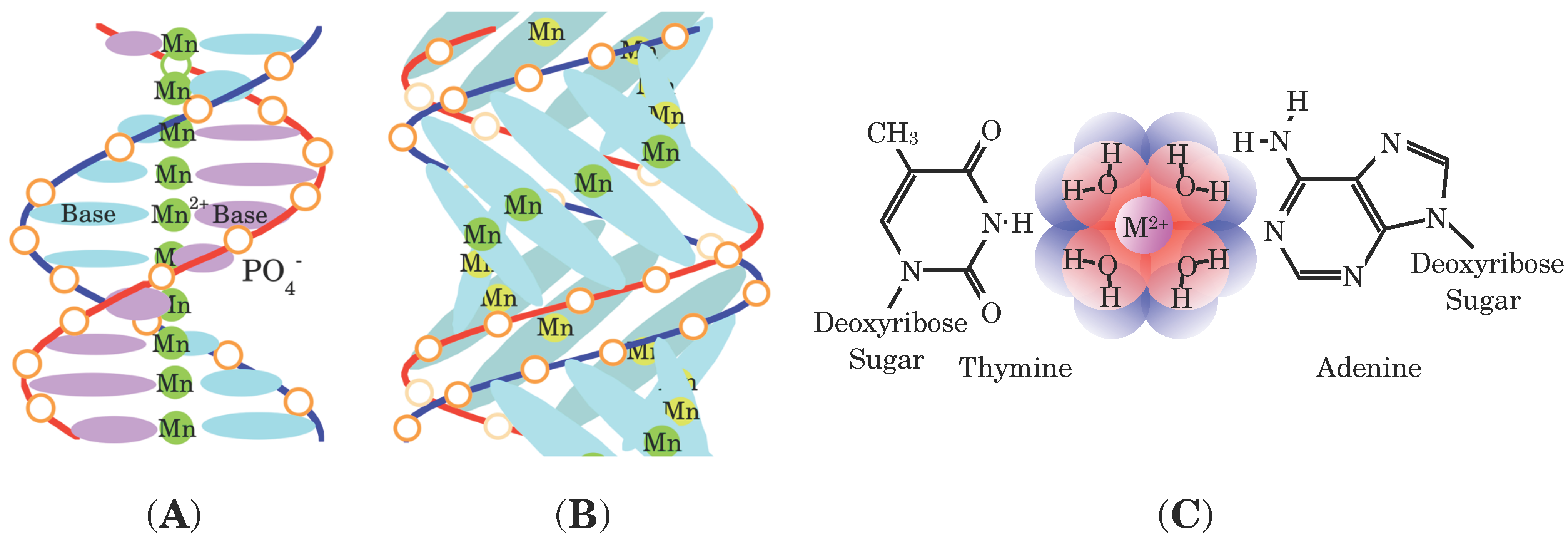
Figure 1C, it was confirmed that the metal ions in
M-DNA are hydrated by several water molecules and located between the bases of a base pair with ionic bonds to the negatively charged DNA [
8,
17]. An octahedral ligand field with six water molecules around the Fe
3+ ions in Fe-DNA provides the high-spin state and a square planer ligand field with four water molecules does the low-spin state. Actually, the number of water molecules in Fe-DNA was controlled by evacuation. Complete evacuation at 300 K reduces the number ratio of the high-spin state–the low-spin state down to 1–5.67 from 1–3.35 [
19], consistent with the above consideration.
Figure 2.
(
A) FeCl
3; (
B) FeCl
2; and (
C) Fe-DNA. (Reused from Ref. [
15]).
Figure 2.
(
A) FeCl
3; (
B) FeCl
2; and (
C) Fe-DNA. (Reused from Ref. [
15]).
Figure 3.
(A) An ESR spectrum of Fe-DNA taken at Q-band (≈ 35 GHz, 1.21 T corresponds to g = 2), together with a Mn-DNA spectrum; (B) A simulation of the ESR spectrum of Fe-DNA at room temperature with three Lorentzians—linewidths of 189 mT (dash-dotted line), 54 mT (dotted line) and 20 mT (dashed line)—. The broad and the medium signals are assigned to the high-spin state, and the low-spin one, of Fe3+, respectively. Since the π electrons are delocalized along the base stacking of the double helix, the ESR spectrum should be narrower than those of Fe3+.
Figure 3.
(A) An ESR spectrum of Fe-DNA taken at Q-band (≈ 35 GHz, 1.21 T corresponds to g = 2), together with a Mn-DNA spectrum; (B) A simulation of the ESR spectrum of Fe-DNA at room temperature with three Lorentzians—linewidths of 189 mT (dash-dotted line), 54 mT (dotted line) and 20 mT (dashed line)—. The broad and the medium signals are assigned to the high-spin state, and the low-spin one, of Fe3+, respectively. Since the π electrons are delocalized along the base stacking of the double helix, the ESR spectrum should be narrower than those of Fe3+.
Figure 4.
Magnetization curves of Fe-DNA at 2 K (the black dots). The dashed curves represent trial fittings, as a sum of the following supposed contributions. (A) Double Brillouin functions fitting of the high-spin state of with θ = 0 K and the low-spin state of with θ = −2.3 K of Fe3+ ions, and (B) Three Brillouin functions of the π electrons with θ = −45 K transferred from Fe2+ into the bases, in addition to with θ = 0 K and with θ = −2.7 K of Fe3+ ions.
Figure 4.
Magnetization curves of Fe-DNA at 2 K (the black dots). The dashed curves represent trial fittings, as a sum of the following supposed contributions. (A) Double Brillouin functions fitting of the high-spin state of with θ = 0 K and the low-spin state of with θ = −2.3 K of Fe3+ ions, and (B) Three Brillouin functions of the π electrons with θ = −45 K transferred from Fe2+ into the bases, in addition to with θ = 0 K and with θ = −2.7 K of Fe3+ ions.
In summary, the spin state of Fe3+ in Fe-DNA is determined by the coordination number of water molecules. Fe3+ has the high-spin state with six coordinated water molecules and the low-spin state with four water molecules.
3.3. Zn-DNA
It has been reported that the electronic states of Zn-DNA prepared at pHs above 8 are metallic with Pauli like paramagnetic susceptibility [
11,
26]. However, Zn-DNA prepared with the ethanol precipitation method from aqueous solutions of DNA and
MCl
2 described in
Section 2 did not necessarily agrees with such conclusion [
19]. A systematic study of Zn-DNA under the control of water content in the system has revealed important roles of the water molecules in a Zn-DNA duplex [
18,
19]. The roles of the water molecules are arranged below.
- (1)
In a step of synthesis with aqueous solutions, the metal ions in M-DNA are hydrated by water molecules, which prevent the metal ions from forming covalent bonds with the nitrogens of the bases of a base pair, resulting in the ionic bonds of the metal ions with the phosphate anions of DNA backbones.
- (2)
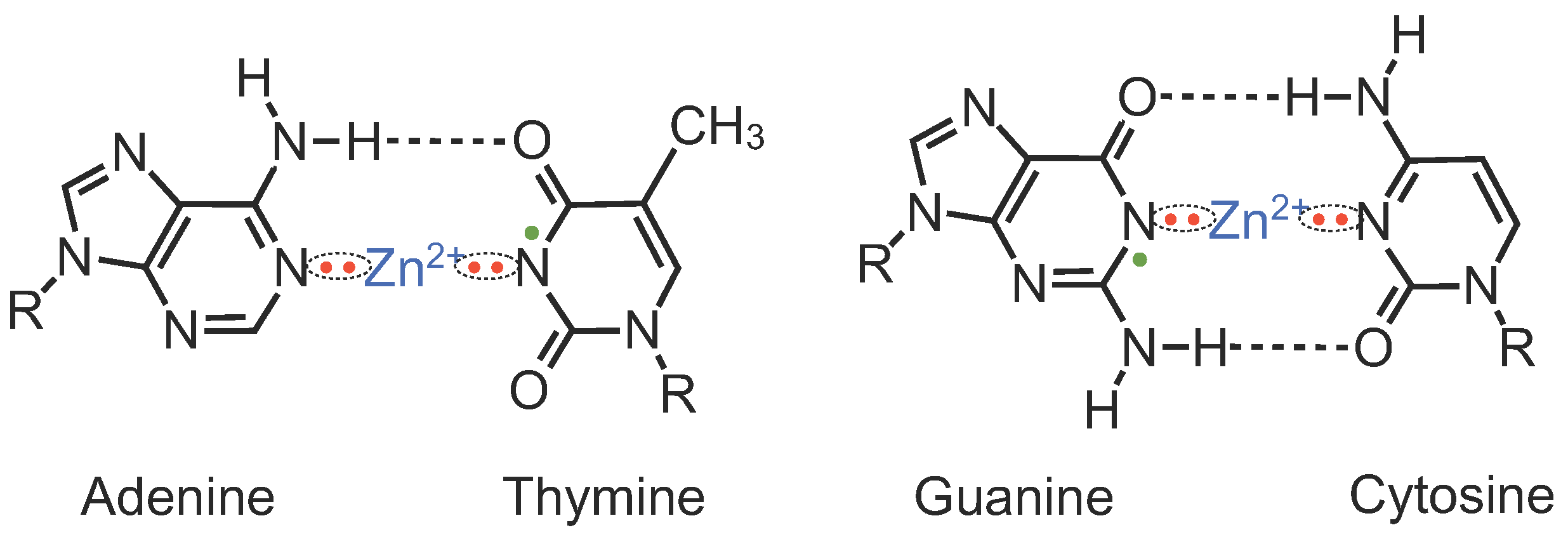
In drying procedure of Zn-DNA after ethanol precipitation, complete evacuation of the water molecules from Zn-DNA by freeze-drying procedure promotes covalent bonding of the metal ions with the nitrogens of the bases in place of the imino hydrogen atoms of guanine and thymine bases, which generates a π spin system in the DNA bases, as shown in
Figure 8.
- (3)
The π electron system of the freeze-dried Zn-DNA (FD-Zn-DNA) has a band width of ≈ 0.24 eV and the electronic states of the π electrons are governed by the water molecules in the FD-Zn-DNA duplex.
- (4)
FD-Zn-DNA without water molecules is nonmagnetic (Only the Zn-DNA diamagnetism is observed.) because of the charge ordering (CO) ground state as a result of the off-site Coulomb energy V between the neighboring base–M–base molecules, which is larger than the on-site Coulomb energy U. This is based on the size of A–Zn–T and G–Zn–C molecules of ≈ 1.5 nm, which is much larger than the distance of ≈ 0.34 nm between the molecules.
- (5)
FD-Zn-DNA with water molecules shows Pauli paramagnetism because of a suppression of CO ground state with the decreased off-site Coulomb energy V, which is caused by the screening effect of the hydrating water molecules on the π electron charges of A–Zn–T and G–Zn–C molecules.
Figure 8.
A model of the electronic states of A–Zn–T and G–Zn–C molecules of FD-Zn-DNA. All the hydrated water molecules are removed by the freeze-drying procedure from the structure shown in
Figure 1C. Zn ions prefer to form configurational bonds in place of the imino hydrogen atoms of the guanine and thymine bases. Note that the adenine and cytosine bases have no imino protons. As a result, one unpaired electron on the nitrogen atom provides
spin per base pair (bp) (Reused from Ref. [
18]).
Figure 8.
A model of the electronic states of A–Zn–T and G–Zn–C molecules of FD-Zn-DNA. All the hydrated water molecules are removed by the freeze-drying procedure from the structure shown in
Figure 1C. Zn ions prefer to form configurational bonds in place of the imino hydrogen atoms of the guanine and thymine bases. Note that the adenine and cytosine bases have no imino protons. As a result, one unpaired electron on the nitrogen atom provides
spin per base pair (bp) (Reused from Ref. [
18]).
3.3.1. FD-Zn-DNA
Samples are prepared by the ethanol precipitation method from aqueous solutions of DNA and ZnCl
2. The characteristic aspects of the samples are (1) high purity Zn-DNA without any residual salts, (2) ionic bonding of hydrated Zn
2+ ions with phosphate anions of DNA backbones. The physical properties of the samples are almost the same as those of B-DNA, such as diamagnetism + nonlinear paramagnetism of magnetic impurities and UV/Vis absorption spectra [
8,
18,
19]. With elimination of almost all the water molecules from Zn-DNA by the freeze-drying procedure, FD-Zn-DNA is obtained with the Zn
2+ ions covalently bonded to the nitrogen atoms of bases. The electronic states of FD-Zn-DNA are sensitive to water molecules, which is quite different from Zn-DNA.
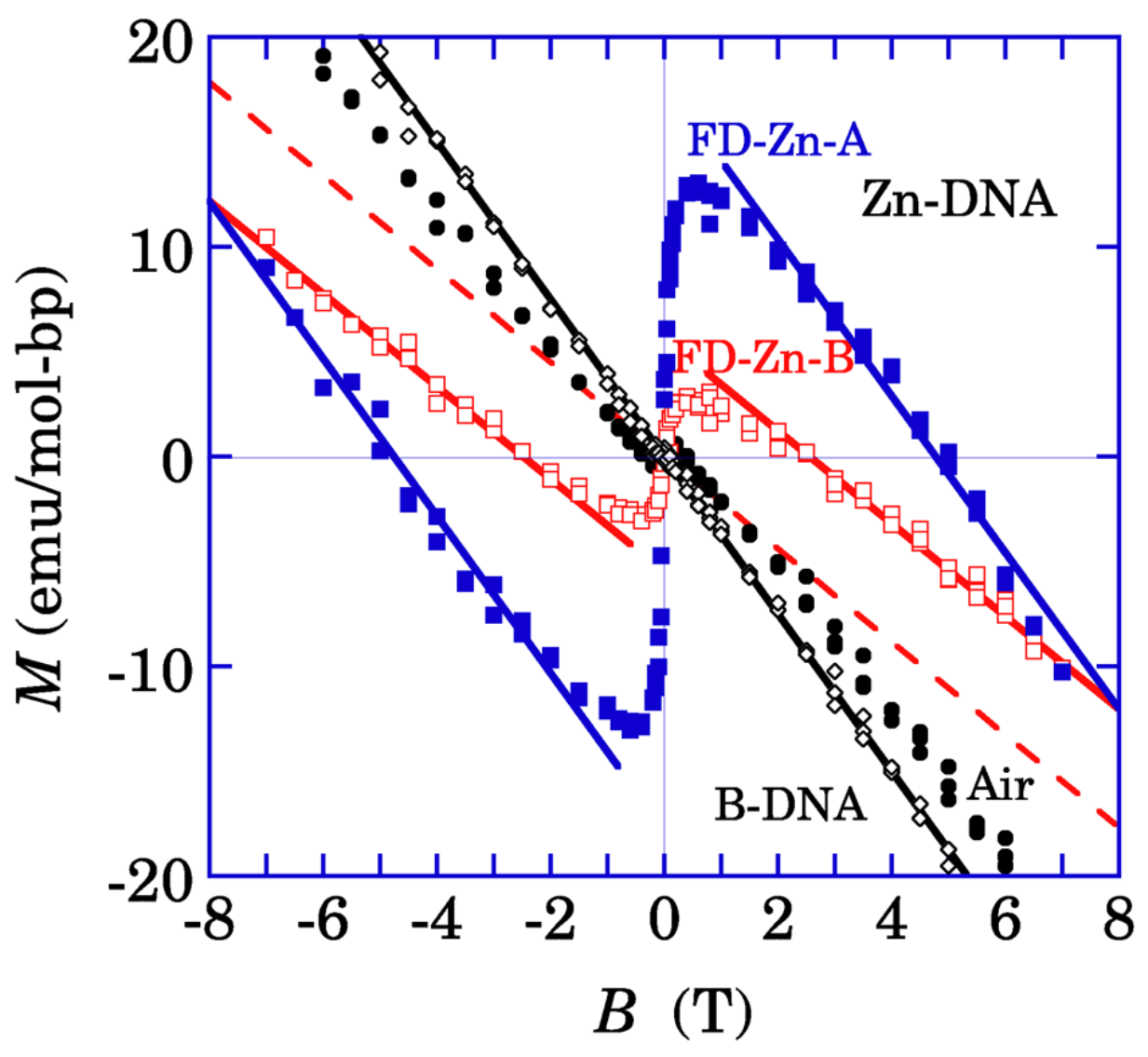
Figure 9 shows the magnetization curves of B-DNA, Zn-DNA and FD-Zn-DNAs. On the basis of a systematic study with the freeze-drying procedure in B-DNA and Zn-DNAs, it has been concluded that the paramagnetic contributions with saturation in all the samples of this figure are caused by the impurities in B-DNA and Zn-DNAs [
18,
19], which behave like ferromagnetism or superparamagnetism under the dried conditions. Strong ESR signals at X-band were also observed in the freeze-dried systems of B-DNA and Zn-DNA with the intensity independent of temperature down to 10 K, which is consistent with the ferromagnetic systems.
Figure 9.
Magnetization per unit containing one base pair of B-DNA and various Zn-DNAs at 100 K [
18]. Open diamonds represent the diamagnetism of as-received salmon DNA (B-DNA). Closed circles show the magnetization of Zn-DNA dried in air (“Air”) with diamagnetism of Zn-DNA and small paramagnetism. FD-Zn-A and FD-Zn-B represent the magnetization of two different batches of the freeze dried Zn-DNA with much larger nonlinear paramagnetism than that of the Zn-DNA dried in air. The solid straight lines on FD-Zn-A is almost parallel to the diamagnetism of B-DNA. In contrast, the baseline of FD-Zn-B represented by the dashed line clearly contains the additional linearly increasing paramagnetism with the magnetic field
B over the diamagnetism of B-DNA (Reused from Ref. [
18]).
Figure 9.
Magnetization per unit containing one base pair of B-DNA and various Zn-DNAs at 100 K [
18]. Open diamonds represent the diamagnetism of as-received salmon DNA (B-DNA). Closed circles show the magnetization of Zn-DNA dried in air (“Air”) with diamagnetism of Zn-DNA and small paramagnetism. FD-Zn-A and FD-Zn-B represent the magnetization of two different batches of the freeze dried Zn-DNA with much larger nonlinear paramagnetism than that of the Zn-DNA dried in air. The solid straight lines on FD-Zn-A is almost parallel to the diamagnetism of B-DNA. In contrast, the baseline of FD-Zn-B represented by the dashed line clearly contains the additional linearly increasing paramagnetism with the magnetic field
B over the diamagnetism of B-DNA (Reused from Ref. [
18]).
In contrast, the paramagnetism proportional to the applied magnetic field, as observed in the base line of FD-Zn-B, was ascribed to the nature of the π spin system of FD-Zn-DNA with some water molecules.
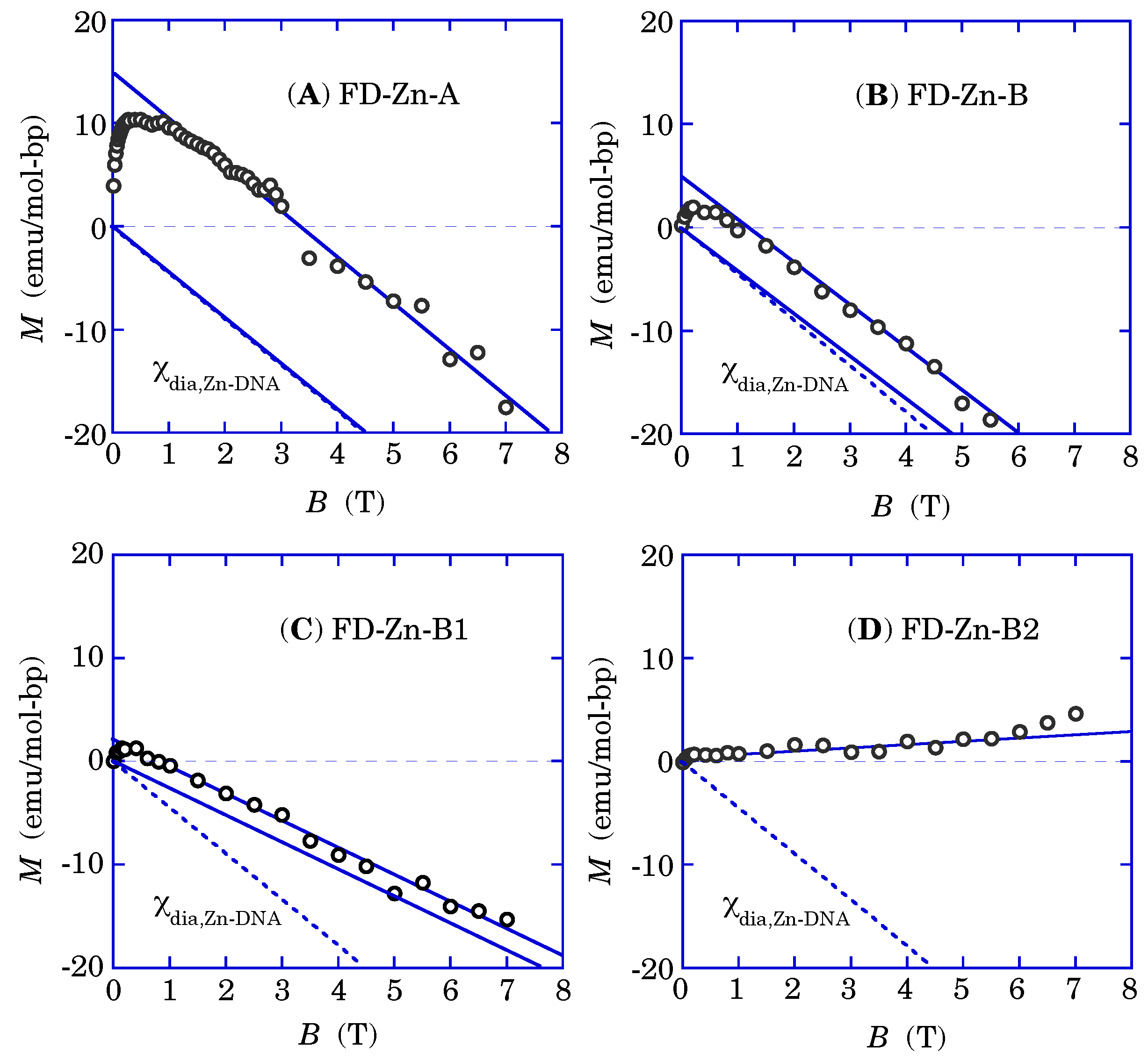
Figure 10 shows the evolution of the experimental steps of FD-Zn-DNAs with the sample names of FD-Zn-A and FD-Zn-B. Details are as follows.
- (1)
Figure 10A shows the magnetization of FD-Zn-A under the evacuated condition, in which the diamagnetism of Zn-DNA and the saturating magnetization of the impurities are observed.
- (2)
Figure 10B describes the magnetization of FD-Zn-B, a different batch from FD-Zn-A, with a little more water molecules than FD-Zn-A, in which the small paramagnetism of the π spin system proportional to the magnetic field and the saturating one of impurity with a weaker amplitude than that of FD-Zn-A case, in addition to the diamagnetism of Zn-DNA, are found.
- (3)
With additional water molecules in FD-Zn-B in
Figure 10C, the increased paramagnetism of the π spin system and further suppression of the impurity magnetization are observed.
- (4)
Figure 10D shows that much more water molecules suppress almost of the saturating impurity magnetization. Only the π spin paramagnetism and the Zn-DNA diamagnetism remains.
Figure 10.
Magnetization curves of FD-Zn-DNA at 300 K. (
A) FD-Zn-A and (
B) FD-Zn-B are prepared from the same source materials but belong to different freeze-drying batches. (
C) FD-Zn-B1 was FD-Zn-B exposed to an “air+humidity” condition to introduce water molecules into the sample. (
D) FD-Zn-B2 was further exposed to the same condition. The solid straight lines in the above panels represent the baselines and the saturation lines of nonlinear magnetization. The dotted lines describe the diamagnetism of Zn-DNA. (Reused from Ref. [
18].)
Figure 10.
Magnetization curves of FD-Zn-DNA at 300 K. (
A) FD-Zn-A and (
B) FD-Zn-B are prepared from the same source materials but belong to different freeze-drying batches. (
C) FD-Zn-B1 was FD-Zn-B exposed to an “air+humidity” condition to introduce water molecules into the sample. (
D) FD-Zn-B2 was further exposed to the same condition. The solid straight lines in the above panels represent the baselines and the saturation lines of nonlinear magnetization. The dotted lines describe the diamagnetism of Zn-DNA. (Reused from Ref. [
18].)
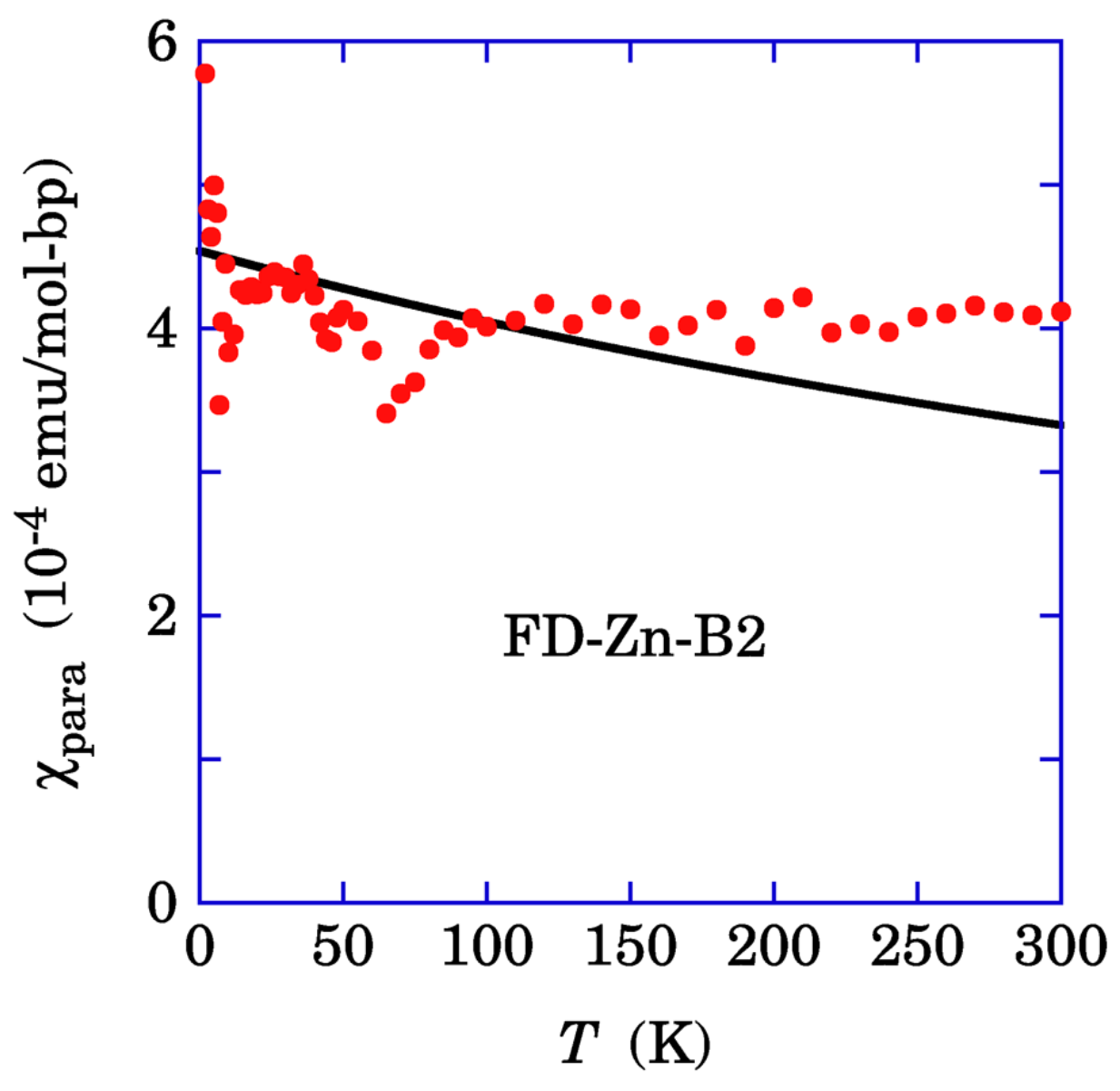
Figure 11 shows the temperature dependence of the paramagnetism of the π spin system. The solid curve represents the Curie–Weiss law with the Weiss temperature of 820 K, which fails to reproduce the data. The possible interpretation is the Pauli paramagnetism
of the highly correlated π spin system with a narrow electron band width, where
N(
EF) = 12.4 states/eV is the density of states at the Fermi energy with
χpara = 4.0 × 10
−4 emu/mol-bp. Since FD-Zn-DNA has A-form with 3D band, the π band width is estimated to be 0.24 eV with the 3D free electron model. The mechanism of the transformation from the nonmagnetic state to the Pauli paramagnetism is discussed in the next section.
Figure 11.
Temperature dependence of the paramagnetic susceptibility
χpara of FD-Zn-B2, which was obtained by subtracting the diamagnetism of Zn-DNA, the oxygen contamination, and the small nonlinear paramagnetism shown in
Figure 10D from the SQUID susceptibility data. The solid curve represents the least-squares fit to the Curie–Weiss law, which is in poor agreement with the data (Reused from Ref. [
18].).
Figure 11.
Temperature dependence of the paramagnetic susceptibility
χpara of FD-Zn-B2, which was obtained by subtracting the diamagnetism of Zn-DNA, the oxygen contamination, and the small nonlinear paramagnetism shown in
Figure 10D from the SQUID susceptibility data. The solid curve represents the least-squares fit to the Curie–Weiss law, which is in poor agreement with the data (Reused from Ref. [
18].).
3.3.2. Nature of FD-Zn-DNA
As discussed in the previous section, the new paramagnetic species are generated by the freeze-drying procedure applied to Zn-DNA. Anomalous magnetic behavior of the new spin system is found that the dehydrated FD-Zn-DNA is nonmagnetic, whereas Pauli paramagnetism is carried by the spin system of FD-Zn-DNA with water molecules. Since it is not a rational assumption that the newly appeared π spin system is generated simply by the addition of the water molecules to FD-Zn-DNA, we proposed a model, as shown in
Figure 8, that the freeze-dried Zn-DNA carries the π electron system, which is just generated by the freeze-drying procedure. The water molecules between the metal ion and the nitrogen atoms of the bases of a base pair prevent the metal ion from forming covalent bonds with the nitrogen atoms. The freeze-drying removes such water molecules and helps for the metal ion to form configurational covalent bonds in place of the imino hydrogen atom with one electron in guanine or thymine, which makes the number of electrons of a base–Zn–base molecule odd, that is, the π spin system is generated. Then, the freeze-drying to achieve complete evacuation of the water molecules from Zn-DNA would be a critical condition to generate the π electron spin system. Simple evacuation of Zn-DNA at 300 K did not help to attain the π spin system generation, but the freeze-drying made it possible. One reason is that the freezing invalidates molecular motion and rearrangement of the water molecules in the Zn-DNA duplex, which provides spatial path ways for the water molecules to escape from the duplex.
Thus, it is suggested that the possible mechanisms of the nonmagnetic behavior in the dehydrated FD-Zn-DNA are some singlet and nonmagnetic ground states like a special configuration of antiferromagnetism (AF) or spin density wave (SDW), spin Peierls (SP), charge density wave (CDW), or charge ordering (CO). Since the present sample is in powder form, there is no possibility of the special configuration of AF or SDW with the magnetic field parallel to the spontaneous magnetization. The Peierls instability of the DNA systems concerning on SP or CDW seems to be implausible because of the sizable structural disorder inherent in DNA. The last candidate is the CO state, in which the off-site Coulomb energy
V is a crucial parameter other than the on-site Coulomb energy
U. From the value of the estimated
U and
V by Starikov [
27],
V is comparable to or larger than
U in AT-AT and GC-GC pairs, which suggests the strong possibility of the CO state in the dehydrated FD-Zn-DNA. Since the π electrons would delocalize over each base–Zn–base molecule to reduce their kinetic energy, the fact that the molecular size more than 1.5 nm of base–Zn–base is much larger than the separation of ≈ 0.34 nm between the neighboring base–Zn–base molecules would cause the condition
V>
U. Thus, the π electrons prefer the alternate double occupancy instead of the single occupancy of every A–Zn–T or G–Zn–C molecule, by paying a cost of
U in place of
V, resulting in the insulating CO state with the repetition (... 0 : −2
e : 0 : −2
e : 0 ...) along the direction of the DNA duplex.
The present model for the nonmagnetic state of the dehydrated FD-Zn-DNA is consistent with the Pauli paramagnetism of the hydrated FD-Zn-DNA. The water molecules play an important role in reducing the off-site Coulomb energy V between the π electrons of the neighboring base–Zn–base molecules. The electronic charges of the π electrons of base–Zn–base molecules are strongly screened by the surrounding water molecules with the large dielectric constant of ≈ 80. As a result, the off-site Coulomb energy V is suppressed by the water molecules, but the on-site Coulomb energy U is not affected. Then, the new condition V<U is realized in the hydrated FD-Zn-DNA, which revives the metallic π electron band with the Pauli paramagnetism from the nonmagnetic and insulating CO ground state with the alternate double occupancy of the π electrons. Finally, the present model for FD-Zn-DNA describes successfully the intrinsic part of the anomalous magnetic behavior, which demonstrates the important role of the water molecules with the screening effect as a determining factor of the magnetic property in the metal ion-DNA complexes.