The Use of Crystalline Carbon-Based Nanomaterials (CBNs) in Various Biomedical Applications
Abstract
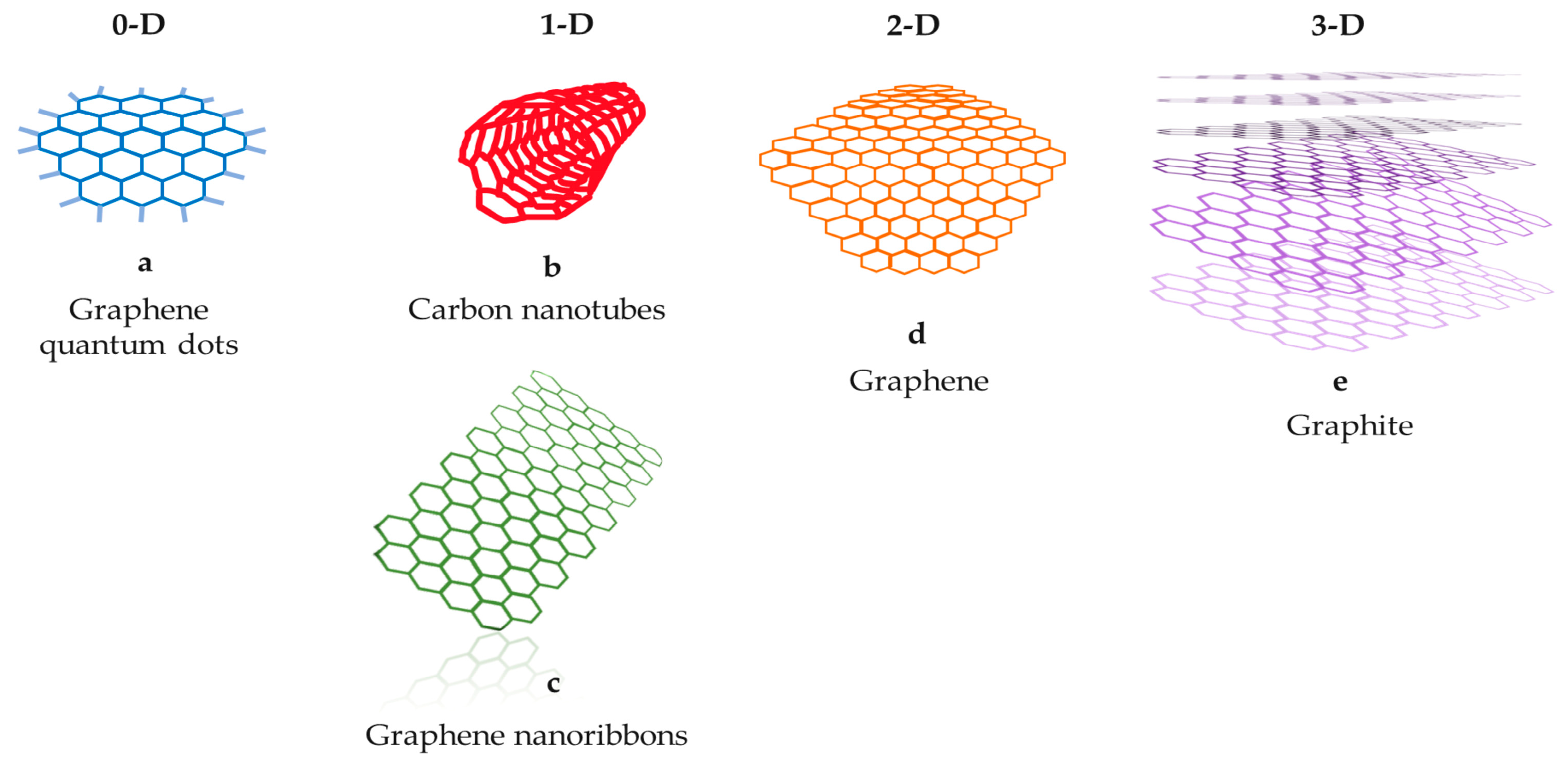
1. Introduction
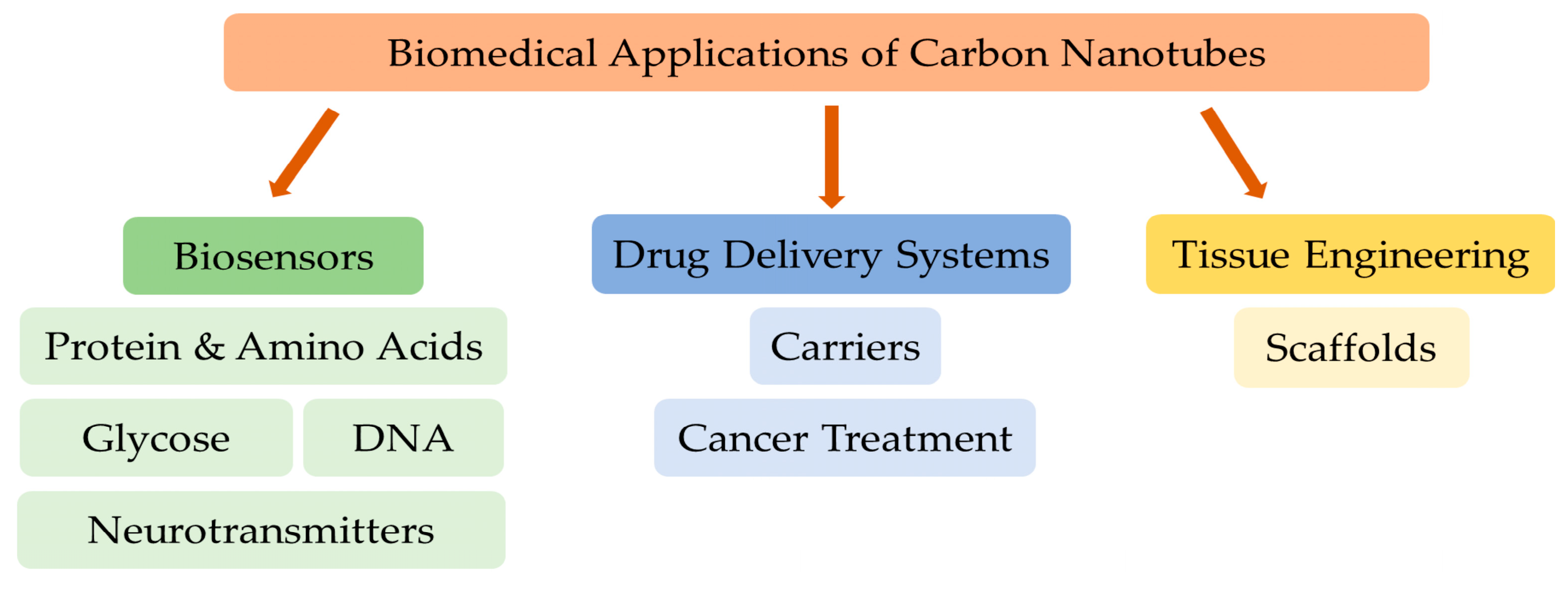
2. Carbon Nanotubes (CNTs)
2.1. CNTs as Biosensors
2.1.1. Sensing of Glucose
2.1.2. Sensing of DNA
2.1.3. Sensing of Proteins and Amino Acids
2.1.4. Sensing of Neurotransmitters
2.2. CNTs for Drug Delivery
2.3. CNTs for Cancer Treatment
2.4. CNTs for Reinforcing Tissue Engineering Scaffolds
2.5. Cytotoxicity of CNTs
3. Graphene and Graphene Oxide
3.1. Graphene and Graphene Oxide as Biosensors
3.2. Graphene and Graphene Oxide for Drug Delivery
3.3. Graphene and Graphene Oxide for Cancer Treatment
3.4. Cytotoxicity of Graphene and Graphene Oxide
4. Graphene Quantum Dots (GQDs)
4.1. GQDs as Biosensors
4.2. GQDs for Drug Delivery
4.3. GQDs for Cancer Treatment
4.4. GQDs in Theragnostic Systems
4.5. Cytotoxicity of GQDs
5. Conclusions and Future Remarks
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pierson, H.O. Handbook of Carbon, Graphite, Diamond, and Fullerenes: Properties, Processing, and Applications; Noyes Publications: Park Ridge, NJ, USA, 1993. [Google Scholar]
- Krüger, A. Carbon Materials and Nanotechnology; Wiley-CH: Weinheim, Germany, 2010. [Google Scholar]
- Maiti, D.; Tong, X.; Mou, X.; Yang, K. Carbon-Based Nanomaterials for Biomedical Applications: A Recent Study. Front. Pharmacol. 2019, 9, 1401. [Google Scholar] [CrossRef]
- Patel, K.D.; Singh, R.K.; Kim, H.-W. Carbon-based nanomaterials as an emerging platform for theranostics. Mater. Horiz. 2019, 6, 434–469. [Google Scholar] [CrossRef]
- Shi, X.; Meng, H.; Sun, Y.; Qu, L.; Lin, Y.; Li, Z.; Du, D. Far-red to near-infrared carbon dots: Preparation and applications in biotechnology. Small 2019, 15, e1901507. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, H. Emerging carbons. Adv. Mater. 2019, 31, e1808208. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.T.; Mi, X.Y.; Li, Y.; Zhan, S.Z. 3D graphene-based macrostructures for water treatment. Adv. Mater. 2020, 32, e1806843. [Google Scholar] [CrossRef]
- Lin, H.; Sturmberg, B.C.P.; Lin, K.T.; Yang, Y.; Zheng, X.; Chong, T.K.; de Sterke, C.M.; Jia, B. A 90-nm-thick graphene metamaterial for strong and extremely broadband absorption of unpolarized light. Nat. Photonics 2019, 13, 270–276. [Google Scholar] [CrossRef]
- Liu, H.; Qiu, H. Recent advances of 3D graphene-based adsorbents for sample preparation of water pollutants: A review. Chem. Eng. J. 2020, 393, 124691. [Google Scholar] [CrossRef]
- Yang, X.; Wan, Y.; Zheng, Y.; He, F.; Yu, Z.; Huang, J.; Wang, H.; Ok, H.S.; Jiang, Y.; Gao, B. Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: A critical review. Chem. Eng. J. 2019, 366, 608–621. [Google Scholar] [CrossRef]
- Song, P.; Liu, B.; Liang, C.; Ruan, K.; Qiu, H.; Ma, Z.; Guo, Y.; Gu, J. Lightweight, flexible cellulose-derived carbon Aerogel@Reduced graphene oxide/PDMS composites with outstanding EMI shielding performances and excellent thermal conductivities. Nanomicro Lett. 2021, 13, 91. [Google Scholar] [CrossRef]
- Tang, T.; Liu, D.; Wang, Y.J.; Cui, L.; Ignaszak, A.; Yu, Y.; Zhang, J. Research advances in biomass-derived nanostructured carbons and their composite materials for electrochemical energy technologies. Prog. Mater. Sci. 2021, 118, 100770. [Google Scholar] [CrossRef]
- Auffan, M.; Rose, J.; Bottero, J.-Y.; Lowry, G.V.; Jolivet, J.-P.; Wiesner, M.R. Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective. Nat. Nanotechnol. 2009, 4, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Tremi, I.; Havaki, S.; Georgitsopoulou, S.; Lagopati, N.; Georgakilas, V.; Gorgoulis, V.G.; Georgakilas, A.G. A Guide for Using Transmission Electron Microscopy for Studying the Radiosensitizing Effects of Gold Nanoparticles In Vitro. Nanomaterials 2021, 11, 859. [Google Scholar] [CrossRef] [PubMed]
- Katifelis, H.; Nikou, M.-P.; Mukha, I.; Vityuk, N.; Lagopati, N.; Piperi, C.; Farooqi, A.A.; Pippa, N.; Efstathopoulos, E.P.; Gazouli, M. Ag/Au Bimetallic Nanoparticles Trigger Different Cell Death Pathways and Affect Damage Associated Molecular Pattern Release in Human Cell Lines. Cancers 2022, 14, 1546. [Google Scholar] [CrossRef] [PubMed]
- Gatou, M.-A.; Lagopati, N.; Vagena, I.-A.; Gazouli, M.; Pavlatou, E.A. ZnO Nanoparticles from Different Precursors and Their Photocatalytic Potential for Biomedical Use. Nanomaterials 2023, 13, 122. [Google Scholar] [CrossRef] [PubMed]
- Lagopati, N.; Kotsinas, A.; Veroutis, D.; Evangelou, K.; Papaspyropoulos, A.; Arfanis, M.; Falaras, P.; Kitsiou, P.V.; Pateras, I.; Bergonzini, A.; et al. Biological Effect of Silver-modified Nanostructured Titanium Dioxide in Cancer. Cancer Genom. Proteom. 2021, 18 (Suppl. S3), 425–439. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou-Fermeli, N.; Lagopati, N.; Pippa, N.; Sakellis, E.; Boukos, N.; Gorgoulis, V.G.; Gazouli, M.; Pavlatou, E.A. Composite Nanoarchitectonics of Photoactivated Titania-Based Materials with Anticancer Properties. Pharmaceutics 2023, 15, 135. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-Y.; Zheng, Y.; Tan, C.-P.; Sun, J.-H.; Zhang, W.; Ji, L.-N.; Mao, Z.-W. Graphene oxide decorated with Ru(II)–polyethylene glycol complex for lysosome-targeted imaging and photodynamic/photothermal therapy. ACS Appl. Mater. Interfaces 2017, 9, 6761–6771. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Zhao, Y.L.; Li, Y.L. Graphdiyne: The fundamentals and application of an emerging carbon material. Adv. Mater. 2019, 31, e1904885. [Google Scholar] [CrossRef]
- Lopez Barreiro, D.; Martin-Moldes, Z.; Yeo, J.; Shen, S.; Hawker, M.J.; Martin-Martinez, F.J.; Kaplan, D.L.; Buehler, M.J. Conductive silk-based composites using biobased carbon materials. Adv. Mater. 2019, 31, e1904720. [Google Scholar] [CrossRef] [PubMed]
- Panwar, N.; Soehartono, A.M.; Chan, K.K.; Zeng, S.; Xu, G.; Qu, J.; Coquet, P.; Yong, K.T.; Chen, X. Nanocarbons for biology and medicine: Sensing, imaging, and drug delivery. Chem. Rev. 2019, 119, 9559–9656. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, S.; Wang, Y.; Wang, H.; Shen, X.; Zhang, M.; Lu, H.; He, M.; Zhang, Y. Bioinspired fluffy fabric with in situ grown carbon nanotubes for ultrasensitive wearable airflow sensor. Adv. Mater. 2020, 32, e1908214. [Google Scholar] [CrossRef] [PubMed]
- Juliette, S.; Emmanuel, F.; Muriel, G. Overview of carbon nanotubes for biomedical applications. Materials 2019, 12, 624. [Google Scholar] [CrossRef]
- Lagopati, N.; Efstathopoulos, E.P.; Veroutis, D.; Katifelis, H.; Theocharous, G.; Pantelis, P.; Evangelou, K.; Gorgoulis, V.G.; Gazouli, M. Hybrid Multifunctional Nanomaterials for Diagnostic and Therapeutic Applications. In Pharmaceutical Nanobiotechnology for Targeted Therapy, 1st ed.; Barabadi, H., Mostafavi, E., Saravanan, M., Eds.; Nanotechnology in the Life Sciences; Springer: Cham, Switzerland, 2022; pp. 489–519. [Google Scholar] [CrossRef]
- Lagopati, N.; Valamvanos, T.-F.; Proutsou, V.; Karachalios, K.; Pippa, N.; Gatou, M.-A.; Vagena, I.-A.; Cela, S.; Pavlatou, E.A.; Gazouli, M.; et al. The Role of Nano-Sensors in Breath Analysis for Early and Non-Invasive Disease Diagnosis. Chemosensors 2023, 11, 317. [Google Scholar] [CrossRef]
- Iijima, S.; Ichihashi, T. Single-shell carbon nanotubes of 1-nm diameter. Nat. Nanotechnol. 1993, 363, 603–605. [Google Scholar] [CrossRef]
- Saito, R.; Dresselhaus, G.; Dresselhaus, M.S. Physical Properties of Carbon Nanotubes; Imperial College Press: London, UK, 1998. [Google Scholar]
- Baughman, R.H.; Zakhidov, A.A.; de Heer, W.A. Carbon nanotubes: The route toward applications. Science 2002, 297, 787–792. [Google Scholar] [CrossRef]
- Yamamoto, T.; Watanabe, K.; Hernandez, E.R. Mechanical properties, thermal stability and heat transport in carbon nanotubes. In Carbon Nanotubes: Advanced Topics in the Synthesis, Structure, Properties and Applications; Jorio, A., Dresselhaus, G., Dresselhaus, M.S., Eds.; Springer: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Choi, W.B.; Bae, E.; Kang, D.; Chae, S.; Cheong, B.-h.; Ko, J.-h.; Lee, E.; Park, W. Aligned Carbon Nanotubes for Nanoelectronics. Nanotechnology 2007, 15, 512–516. [Google Scholar] [CrossRef]
- Endo, M.; Strano, M.S.; Ajayan, P.M. Potential applications of carbon nanotubes. In Carbon Nanotubes: Advanced Topics in the Synthesis, Structure, Properties and Applications; Jorio, A., Dresselhaus, G., Dresselhaus, M.S., Eds.; Springer: Berlin, Germany, 2008; pp. 13–61. [Google Scholar]
- Bonard, J.M.; Salvetat, J.P.; Stöckli, T.; Forró, L.; Chatelain, A. Field emission from carbon nanotubes: Perspectives for applications and clues to the emission mechanism. Appl. Phys. A Mater. Sci. Process. 1999, 69, 245–254. [Google Scholar] [CrossRef]
- Ajayan, P.M.; Zhou, O.Z. Applications of carbon nanotubes. In Carbon Nanotubes: Synthesis, Structure, Properties, and Applications; Dresselhaus, M.S., Dresselhaus, G., Avouris, P., Eds.; Springer-Verlag: New York, NY, USA, 2001; pp. 391–425. [Google Scholar]
- Harrison, B.S.; Atala, A. Carbon nanotube applications for tissue engineering. Biomaterials 2007, 28, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Kostarelos, K.; Partidos, C.D.; Prato, M. Biomedical applications of functionalized carbon nanotubes. Chem. Commun. 2005, 5, 571–577. [Google Scholar] [CrossRef]
- Tîlmaciu, C.M.; Morris, M.C. Carbon nanotube biosensor. Front. Chem. 2015, 3, 59. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Gan, Z.; Zhuang, Q. Electrochemical sensors based on carbon nanotubes. Electroanalysis 2002, 14, 1609–1613. [Google Scholar] [CrossRef]
- Lin, Y.; Lu, F.; Tu, Y.; Ren, Z. Glucose biosensors based on carbon nanotube nanoelectrode ensembles. Nano Lett. 2004, 4, 191–195. [Google Scholar] [CrossRef]
- Zribi, B.; Roy, E.; Pallandre, A.; Chebil, S.; Koubaa, M.; Mejri, N.; Magdinier Gomez, H.; Sola, C.; Korri-Youssoufi, H.; Haghiri-Gosnet, A.-M. A microfluidic electrochemical biosensor based on multiwall carbon nanotube/ferrocene for genomic DNA detection of Mycobacterium tuberculosis in clinical isolates. Biomicrofluidics 2016, 2, 014115. [Google Scholar] [CrossRef] [PubMed]
- Kaloumenou, M.; Skotadis, E.; Lagopati, N.; Efstathopoulos, E.; Tsoukalas, D. Breath Analysis: A Promising Tool for Disease Diagnosis—The Role of Sensors. Sensors 2022, 22, 1238. [Google Scholar] [CrossRef] [PubMed]
- Otero, F.; Magner, E. Biosensors-recent advances and future challenges in electrode materials. Sensors 2020, 20, 3561. [Google Scholar] [CrossRef] [PubMed]
- Wang, J. Electrochemical glucose biosensors. Chem. Rev. 2008, 108, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Meena, J.; Gupta, A.; Ahuja, R.; Singh, M.; Kumar Panda, A. Recent advances in nano-engineered approaches used for enzyme immobilization with enhanced activity. J. Mol. Liq. 2021, 338, 116602. [Google Scholar] [CrossRef]
- Azimi, S.; Farahani, A.; Sereshti, H. Plasma-functionalized highly aligned CNT-based biosensor for point of care determination of glucose in human blood plasma. Electroanalysis 2019, 32, 394–403. [Google Scholar] [CrossRef]
- Wang, C.; Li, J.; Tan, R.; Wang, Q.; Zhang, Z. Colorimetric method for, glucose detection with enhanced signal intensity using ZnFe2O4-carbon nanotube glucose oxidase composite material. Analyst 2019, 144, 1831–1839. [Google Scholar] [CrossRef] [PubMed]
- Akogulları, S.; Çinar, S.; Ozdokur, K.V.; Aydemir, T.; Ertas, F.N.; Koçak, S. Pulsed deposited manganese and vanadium oxide film modified with carbon nanotube and gold nanoparticle: Chitosan and ionic liquid-based biosensor. Electroanalysis 2019, 32, 445–453. [Google Scholar] [CrossRef]
- Hossain, M.F.; Slaughter, G. Pt NPs decorated chemically derived graphene and carbon nanotubes for sensitive and selective glucose biosensing. J. Electroanal. Chem. 2020, 861, 113990. [Google Scholar] [CrossRef]
- Plocienniczak, P.; Rebis, T.; Leda, A.; Milczarek, G. Lignosulfonate-assisted synthesis of platinum nanoparticles deposited on multi-walled carbon nanotubes for biosensing of glucose. Colloids Surf. B Biointerfaces 2021, 210, 112222. [Google Scholar] [CrossRef] [PubMed]
- Zou, B.; Wang, P.; Xia, J.; Qian, J.; Yan, Y.; Mutombo, J. Enzyme biosensors systems based on Co-modification of carbon nanotubes and enzyme for detection of glucose in food. J. Electrochem. Soc. 2021, 168, 065501. [Google Scholar] [CrossRef]
- Jayakumar, K.; Bennett, R.; Leech, D. Electrochemical glucose biosensor based on an osmium redox polymer and glucose oxidase grafted to carbon nanotubes: A design-of-experiments optimization of current density and stability. Electrochim. Acta 2021, 371, 137845. [Google Scholar] [CrossRef]
- Dhanjai, X.L.; Wu, L.; Chen, J.; Lu, Y. Robust single-molecule enzyme nanocapsules for biosensing with significantly improved biosensor stability. Anal. Chem. 2020, 92, 5830–5837. [Google Scholar] [CrossRef]
- Huang, H.; Li, T.; Jiang, M.; Wei, C.; Ma, S.; Chen, D.; Tong, W.; Huang, X. Construction of flexible enzymatic electrode based on gradient hollow fiber membrane and multi-wall carbon tubes meshes. Biosens. Bioelectron. 2020, 152, 112001. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Liu, H.; Wang, X. Thermal self-regulatory intelligent biosensor based on carbon-nanotubes-decorated phase-change microcapsules for enhancement of glucose detection. Biosens. Bioelectron. 2021, 195, 113586. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-B.; Kim, H.-S.; Jeon, W.-Y.; Lee, B.-H.; Shin, U.S.; Kim, H.-H. The electrochemical glucose sensing based on the chitosan-carbon nanotube hybrid. Biochem. Eng. J. 2019, 144, 227–234. [Google Scholar] [CrossRef]
- Zheng, Y.; Tang, N.; Omar, R.; Hu, Z.; Duong, T.; Wang, J.; Wu, W.; Haick, H. Smart materials enabled with artificial intelligence for healthcare wearables. Adv. Funct. Mater. 2021, 31, 2105482. [Google Scholar] [CrossRef]
- Xia, H.-q.; Tang, H.; Zhou, B.; Li, Y.; Zhang, X.; Shi, Z.; Deng, L.; Song, R.; Li, L.; Zhang, Z.; et al. Mediator-free electron-transfer on patternable hierarchical meso/macro porous bienzyme interface for highly-sensitive sweat glucose and surface electromyography monitoring. Sens. Actuators B Chem. 2020, 312, 127962. [Google Scholar] [CrossRef]
- Yao, Y.; Chen, J.; Guo, Y.; Lv, T.; Chen, Z.; Li, N.; Cao, S.; Chen, B.; Chen, T. Integration of interstitial fluid extraction and glucose detection in one device for wearable non-invasive blood glucose sensors. Biosens. Bioelectron. 2021, 179, 113078. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Deng, C.; Chen, P.; You, X.-D.; Su, H.-B.; Yuan, Y.-H.; Zhu, M.-F. Glucose oxidase biosensors based on carbon nanotube non-woven fabrics. Carbon 2014, 67, 795–796. [Google Scholar] [CrossRef]
- Gaitán, C.G.; Rosas, R.R.; Morallón, E.; Amorós, D.C. Effects of the surface chemistry and structure of carbon nanotubes on the coating of glucose oxidase and electrochemical biosensors performance. RSC Adv. 2017, 7, 26867–26878. [Google Scholar] [CrossRef]
- Liu, Q.; Zhong, H.; Chen, M.; Zhao, C.; Liu, Y.; Xi, F.; Luo, T. Functional nanostructure-loaded three-dimensional graphene foam as a non-enzymatic electrochemical sensor for reagentless glucose detection. RSC Adv. 2020, 10, 33739–33746. [Google Scholar] [CrossRef]
- Aun, T.T.; Salleh, N.M.; Ali, U.F.M.; Manan, N.S.A. Non-enzymatic glucose sensors involving copper: An electrochemical perspective. Crit. Rev. Anal. Chem. 2021, 53, 1967720. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; Khan, M.; Tripathy, N.; Khan, M.I.R.; Khosla, A. Hydrothermally synthesized nickel oxide nanosheets for non-enzymatic electrochemical glucose detection. J. Electrochem. Soc. 2020, 167, 107504. [Google Scholar] [CrossRef]
- Abrori, S.A.; Septiani, N.L.W.; Nugraha; Anshori, I.; Suyatman; Suendo, V.; Yuliarto, B. Metal-organic-framework FeBDC-derived Fe3O4 for nonenzymatic electrochemical detection of glucose. Sensors 2020, 20, 4891. [Google Scholar] [CrossRef]
- Viswanathan, P.; Wang, K.; Li, J.; Hong, J.-D. Multicore-shell Ag-CuO networked with CuO nanorods for enhanced non-enzymatic glucose detection. Colloids Surf. Physicochem. Eng. Asp. 2020, 598, 124816. [Google Scholar] [CrossRef]
- Movlaee, K.; Raza, H.; Pinna, N.; Leonardi, S.G.; Neri, G. Ultrasensitive nonenzymatic electrochemical glucose sensor based on NiO/CNT composite. Sensors 2019, 539, 135–140. [Google Scholar] [CrossRef]
- Naqvi, S.T.R.; Shirinfar, B.; Hussain, D.; Majeed, S.; Ashiq, M.N.; Aslam, Y.; Ahmed, N. Electrochemical sensing of ascorbic acid, hydrogen peroxide and glucose by bimetallic (Fe, Ni)-CNTs composite modified electrode. Electroanalysis 2019, 31, 851–857. [Google Scholar] [CrossRef]
- Silva-Carrillo, C.; Felix-Navarro, R.M.; Perez Sicairos, S.; Trujillo-Navarrete, B.; Paraguay-Delgado, F.; Lin-Ho, S.W.; Reynoso-Soto, E.A. Electrokinetic carbohydrates sensor by streaming potential with 3MPBA-PtNPs-CNTs composite material. J. Electrochem. Soc. 2020, 167, 046516. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Cellat, K.; Arikan, K.; Savk, A.; Karimi, F.; Sen, F. Palladium nickel nanoparticles decorated on functionalized-MWCNT for high precision non-enzymatic glucose sensing. Mater. Chem. Phys. 2020, 250, 123042. [Google Scholar] [CrossRef]
- Qian, P.; Qin, Y.; Lyu, Y.; Li, Y.; Wang, L.; Wang, S.; Liu, Y. A hierarchical cobalt/carbon nanotube hybrid nanocomplex-based ratiometric fluorescent nanosensor for ultrasensitive detection of hydrogen peroxide and glucose in human serum. Anal. Bioanal. Chem. 2019, 411, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Miao, L.; Song, Y. Preparation of co-Co3O4/carbon nanotube/carbon foam for glucose sensor. J. Mol. Recogn. 2019, 33, e2820. [Google Scholar] [CrossRef]
- Arikan, K.; Burhan, H.; Bayat, R.; Sen, F. Glucose nano biosensor with nonenzymatic excellent sensitivity prepared with nickel-cobalt nanocomposites on f-MWCNT. Chemosphere 2021, 291, 132720. [Google Scholar] [CrossRef]
- Adeniyi, O.; Nwahara, N.; Mwanza, D.; Nyokong, T.; Mashazi, P. Nanohybrid electrocatalyst based on cobalt phthalocyanine-carbon nanotube-reduced graphene oxide for ultrasensitive detection of glucose in human saliva. Sens. Actuators B Chem. 2021, 348, 130723. [Google Scholar] [CrossRef]
- Scognamiglio, V.; Arduini, F. The technology tree in the design of glucose biosensors. Trends Anal. Chem. 2019, 120, 115642. [Google Scholar] [CrossRef]
- Qin, Y.; Sun, Y.; Li, Y.; Li, C.; Wang, L.; Guo, S. MOF derived Co3O4/N-doped carbon nanotubes hybrids as efficient catalysts for sensitive detection of H2O2 and glucose. Chin. Chem. Lett. 2020, 31, 774–778. [Google Scholar] [CrossRef]
- Kim, S.e.; Muthurasu, A. Highly oriented nitrogen-doped carbon nanotube integrated bimetallic cobalt copper organic framework for non-enzymatic electrochemical glucose and hydrogen peroxide sensor. Electroanalysis 2021, 33, 1333–1345. [Google Scholar] [CrossRef]
- Palve, Y.P.; Jha, N. A novel bilayer of copper nanowire and carbon nanotube electrode for highly sensitive enzyme free glucose detection. Mater. Chem. Phys. 2020, 240, 122086. [Google Scholar] [CrossRef]
- Waqas, M.; Wu, L.; Tang, H.; Liu, C.; Fan, Y.; Jiang, Z.; Wang, X.; Zhong, J.; Chen, W. Cu2O microspheres supported on sulfur-doped carbon nanotubes for glucose sensing. ACS Appl. Nano Mater. 2020, 3, 4788–4798. [Google Scholar] [CrossRef]
- Najmi, A.; Saidi, M.S.; Shahrokhian, S.; Hosseini, H.; Kazemzadeh Hannani, S. Fabrication of a microdialysis-based nonenzymatic microfluidic sensor for regular glucose measurement. Sens. Actuators B Chem. 2021, 333, 129569. [Google Scholar] [CrossRef]
- Mao, X.; Zhang, C. A microfluidic cloth-based photoelectrochemical analytical device for the detection of glucose in saliva. Talanta 2022, 238, 123052. [Google Scholar] [CrossRef]
- Kaiser, J. DNA test to predict odds of severe COVID-19 draws scrutiny. Science 2021, 372, 1139. [Google Scholar] [CrossRef]
- Williams, R.M.; Lee, C.; Heller, D.A. A fluorescent carbon nanotube sensor detects the metastatic prostate cancer biomarker uPA. ACS Sens. 2018, 3, 1838–1845. [Google Scholar] [CrossRef] [PubMed]
- Ganbat, K.; Pan, D.; Chen, K.; Ning, Z.; Xing, L.; Zhang, Y.; Shen, Y. One-pot electrografting preparation of bifunctionalized carbon nanotubes for sensitive electrochemical immunosensing. J. Electroanal. Chem. 2020, 860, 113906. [Google Scholar] [CrossRef]
- Gulati, P.; Mishra, P.; Khanuja, M.; Narang, J.; Islam, S.S. Nano-moles detection of tumor specific biomarker DNA for colorectal cancer detection using vertically aligned multi-wall carbon nanotubes based flexible electrodes. Process. Biochem. 2020, 90, 184–192. [Google Scholar] [CrossRef]
- Grushevskaya, H.V.; Krylova, N.G.; Lipnevich, I.V.; Egorova, V.P.; Babenka, A.S. Single nucleotide polymorphism genotyping using DNA sequencing on multiwalled carbon nanotubes monolayer by CNT-plasmon resonance. Int. J. Mod. Phys. B 2018, 32, 1840033. [Google Scholar] [CrossRef]
- Ye, Q.; Xu, X.; Paghi, A.; Bamford, T.; Horrocks, B.R.; Houlton, A.; Barillaro, G.; Dimitrov, S.; Palma, M. Solution-processable carbon nanotube nanohybrids for multiplexed photoresponsive devices. Adv. Funct. Mater. 2021, 31, 2105719. [Google Scholar] [CrossRef]
- Lambert, B.P.; Gillen, A.J.; Boghossian, A.A. Synthetic biology: A solution for tackling nanomaterial challenges. J. Phys. Chem. Lett. 2020, 11, 4791–4802. [Google Scholar] [CrossRef]
- Prakash, J.; Dey, A.; Uppal, S.; Alexander, R.; Kaushal, A.; Misra, H.S.; Dasgupta, K. Label-free rapid electrochemical detection of DNA hybridization using ultrasensitive standalone CNT aerogel biosensor. Biosens. Bioelectron. 2021, 191, 113480. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Su, T.; Shang, Z.; Jin, D.; Shu, Y.; Xu, Q.; Hu, X. Flexible paper-based Ni-MOF composite/AuNPs/CNTs film electrode for HIV DNA detection. Biosens. Bioelectron. 2021, 184, 113229. [Google Scholar] [CrossRef]
- Belogiannis, K.; Florou, V.A.; Fragkou, P.C.; Ferous, S.; Chatzis, L.; Polyzou, A.; Lagopati, N.; Vassilakos, D.; Kittas, C.; Tzioufas, A.G.; et al. SARS-CoV-2 Antigenemia as a Confounding Factor in Immunodiagnostic Assays: A Case Study. Viruses 2021, 13, 1143. [Google Scholar] [CrossRef]
- Jeong, S.; Gonzalez-Grandio, E.; Navarro, N.; Pinals, R.L.; Ledesma, F.; Yang, D.; Landry, M.P. Extraction of viral nucleic acids with carbon nanotubes increases SARS-CoV-2 quantitative reverse transcription polymerase chain reaction detection sensitivity. ACS Nano 2021, 15, 10309–10317. [Google Scholar] [CrossRef]
- Grushevskaya, H.V.; Krylova, N.G.; Lipnevich, I.V.; Babenka, A.S.; Egorova, V.P.; Chakukov, R.F. CNT-based label-free electrochemical sensing of native DNA with allele single nucleotide polymorphism. Semiconductors 2018, 52, 1836–1838. [Google Scholar] [CrossRef]
- Lee, J.; Morita, M.; Takemura, K.; Park, E.Y. A multi-functional gold/iron-oxide nanoparticle-CNT hybrid nanomaterial as virus DNA sensing platform. Biosens. Bioelectron. 2018, 102, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Liu, W.; Zheng, M.; Wang, R. Label-free and ultrasensitive electrochemical DNA biosensor based on urchinlike carbon nanotube-gold nanoparticle nanoclusters. Anal. Chem. 2020, 92, 4780–4787. [Google Scholar] [CrossRef]
- Huang, R.; Wang, L.; Gai, Q.; Wang, D.; Qian, L. DNA-mediated assembly of carbon nanotubes for enhancing electrochemiluminescence and its application. Sens. Actuators B Chem. 2018, 256, 953–961. [Google Scholar] [CrossRef]
- Heydarzadeh, S.; Roshanfekr, H.; Peyman, H.; Kashanian, S. Modeling of ultrasensitive DNA hybridization detection based on gold nanoparticles/carbon-nanotubes/chitosan-modified electrodes. Colloids Surf. Physicochem. Eng. Asp. 2020, 587, 124219. [Google Scholar] [CrossRef]
- Kuralay, F.; Dükar, N. Polypyrrole-based nanohybrid electrodes: Their preparation and potential use for DNA recognition and paclitaxel quantification. ChemistrySelect 2020, 5, 4708–4714. [Google Scholar] [CrossRef]
- Tamersit, K.; Djeffal, F. Carbon nanotube field-effect transistor with vacuum gate dielectric for label-free detection of DNA molecules: A computational investigation. IEEE Sens. J. 2019, 19, 9263–9270. [Google Scholar] [CrossRef]
- Sun, Y.; Peng, Z.; Li, H.; Wang, Z.; Mu, Y.; Zhang, G.; Chen, S.; Liu, S.; Wang, G.; Liu, C.; et al. Suspended CNT-based FET sensor for ultrasensitive and label-free detection of DNA hybridization. Biosens. Bioelectron. 2019, 137, 255–262. [Google Scholar] [CrossRef]
- Liang, Y.; Xiao, M.; Wu, D.; Lin, Y.; Liu, L.; He, J.; Zhang, G.; Peng, L.M.; Zhang, Z. Wafer-scale uniform carbon nanotube transistors for ultrasensitive and label-free detection of disease biomarkers. ACS Nano 2020, 8, 8866–8874. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Zhao, Q.; Zhang, Q.; Fu, M.; Liu, Y.; Fan, X.; Wang, H.; Zhang, Y.; Wang, H. Nitrogen doped chiral carbonaceous nanotube for ultrasensitive DNA direct electrochemistry, DNA hybridization and damage study. Anal. Chim. Acta 2018, 1038, 41–51. [Google Scholar] [CrossRef]
- Zhao, H.; Liu, M.; Jiang, T.; Xu, J.; Zhang, H.; Yu, C.; Liu, Z.; Wang, Y.; Tang, L. Ultrasensitive monitoring of DNA damage associated with free radicals’ exposure using dynamic carbon nanotubes bridged interdigitated electrode array. Environ. Int. 2020, 139, 105672. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Gao, Y.; Zhang, J.; Wang, X.; Yin, F.; Li, Z.; Zhang, M. Universal DNA detection realized by peptide-based carbon nanotube biosensors. Nanoscale Adv. 2020, 2, 717–723. [Google Scholar] [CrossRef] [PubMed]
- Balchin, D.; Hayer-Hartl, M.; Hartl, F.U. In vivo aspects of protein folding and quality control. Science 2016, 353, 43–54. [Google Scholar] [CrossRef]
- Rafiq, H.S.; Fatima, B.; Hussain, D.; Mohyuddin, A.; Majeed, S.; Manzoor, S.; Imran, M.; Nawaz, R.; Shabbir, S.; Mukhtar, S.; et al. Selective electrochemical sensing of hemoglobin from blood of 8-thalassemia major patients by tellurium nanowires-graphene oxide modified electrode. Chem. Eng. J. 2021, 419, 129706. [Google Scholar] [CrossRef]
- Khan, N.I.; Maddaus, A.G.; Song, E. A low-cost inkjet-printed aptamer-based electrochemical biosensor for the selective detection of lysozyme. Biosensors 2018, 8, 7. [Google Scholar] [CrossRef]
- Janssen, J.; Lambeta, M.; White, P.; Byagowi, A. Carbon nanotube-based electrochemical biosensor for label-free protein detection. Biosensors 2019, 9, 144. [Google Scholar] [CrossRef]
- Huang, Y.; Wen, Y.Q.; Baryeh, K.; Takalkar, S.; Lund, M.; Zhang, X.J.; Liu, G.D. Magnetized carbon nanotubes for visual detection of proteins directly in whole blood. Anal. Chim. Acta 2017, 993, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Moreira, F.T.C.; Rodriguez, B.A.G.; Dutra, R.A.F.; Sales, M.G.F. Redox probe-free readings of a beta-amyloid-42 plastic antibody sensory material assembled on copper@carbon nanotubes. Sens. Actuators B Chem. 2018, 264, 1–9. [Google Scholar] [CrossRef]
- Lee, C.S.; Kim, J.S.; Kim, T.H. A chemodosimeter-modified carbon nanotube field effect transistor: Toward a highly selective and sensitive electrical sensing platform. RSC Adv. 2019, 9, 28414–28420. [Google Scholar] [CrossRef] [PubMed]
- Naveh, Z.M.H.; Khajehdad, M.M.; Sarmazdeh, M.M. A theoretical study on the chirality detection of serine amino acid based on carbon nanotubes with and without stone-wales defects. Struct. Chem. 2020, 31, 455–464. [Google Scholar] [CrossRef]
- Ning, G.; Wang, H.; Fu, M.; Liu, J.; Sun, Y.; Lu, H.; Fan, X.; Zhang, Y.; Wang, H. Dual signals electrochemical biosensor for point-of-care testing of amino acids enantiomers. Electroanalysis 2021, 34, 316–325. [Google Scholar] [CrossRef]
- Xiong, M.; Tao, Y.Z.; Gao, Q.Q.; Feng, B.; Yan, W.; Zhou, Y.Y.; Kotsonis, T.A.; Yuan, T.L.; You, Z.W.; Wu, Z.Y.; et al. Human stem cell-derived neurons repair circuits and restore neural function. Cell Stem Cell 2021, 28, 112–126. [Google Scholar] [CrossRef]
- Vu, T.T.; Dau, T.N.N.; Ly, C.T.; Pham, D.C.; Nguyen, T.T.N.; Pham, V.T. Aqueous electrodeposition of (AuNPs/MWCNT-PEDOT) composite for high-affinity acetylcholinesterase electrochemical sensors. J. Mater. Sci. 2020, 55, 9070–9081. [Google Scholar] [CrossRef]
- Gopal, P.; Narasimha, G.; Reddy, T.M. Development, validation and enzyme kinetic evaluation of multi walled carbon nano tubes mediated tyrosinase based electrochemical biosensing platform for the voltammetric monitoring of epinephrine. Process. Biochem. 2020, 92, 476–485. [Google Scholar] [CrossRef]
- Li, Y.; Ye, W.; Cui, Y.; Li, B.; Yang, Y.; Qian, G. A metal-organic frameworks@carbon nanotubes based electrochemical sensor for highly sensitive and selective determination of ascorbic acid. J. Mol. Struct. 2020, 1209, 127986. [Google Scholar] [CrossRef]
- Shruthi, C.D.; Venkataramanappa, Y.; Suresh, G.S. Reduced MWCNTs/palladium nanotubes hybrid fabricated on graphite electrode for simultaneous detection of ascorbic acid, dopamine and uric acid. J. Electrochem. Soc. 2018, 165, 458–465. [Google Scholar] [CrossRef]
- Guan, J.-F.; Zou, J.; Liu, Y.-P.; Jiang, X.-Y.; Yu, J.-G. Hybrid carbon nanotubes modified glassy carbon electrode for selective, sensitive and simultaneous detection of dopamine and uric acid. Ecotoxicol. Environ. Saf. 2020, 201, 110872. [Google Scholar] [CrossRef]
- Manivel, P.; Thamilselvan, A.; Rajagopal, V.; Nesakumar, N.; Suryanarayanan, V. Enhanced electrocatalytic activity of Ni-CNT nanocomposites for simultaneous determination of epinephrine and dopamine. Electroanalysis 2019, 31, 2387–2396. [Google Scholar] [CrossRef]
- Eom, G.; Oh, C.; Moon, J.; Kim, H.; Kim, M.K.; Kim, K.; Seo, J.-W.; Kang, T.; Lee, H.J. Highly sensitive and selective detection of dopamine using overoxidized polypyrrole/sodium dodecyl sulfate-modified carbon nanotube electrodes. J. Electroanal. Chem. 2019, 848, 113295. [Google Scholar] [CrossRef]
- Ma, X.; Gao, F.; Dai, R.; Liu, G.; Zhang, Y.; Lu, L.; Yu, Y. Novel electrochemical sensing platform based on a molecularly imprinted polymer-decorated 3Dmulti-walled carbon nanotube intercalated graphene aerogel for selective and sensitive detection of dopamine. Anal. Methods 2020, 12, 1845–1851. [Google Scholar] [CrossRef]
- Arumugasamy, S.K.; Govindaraju, S.; Yun, K. Electrochemical sensor for detecting dopamine using graphene quantum dots incorporated with multiwall carbon nanotubes. Appl. Surf. Sci. 2020, 508, 145294. [Google Scholar] [CrossRef]
- Gupta, P.; Tsai, K.; Ruhunage, C.K.; Gupta, V.K.; Rahm, C.E.; Jiang, D.; Alvarez, N.T. True picomolar neurotransmitter sensor based on open-ended carbon nanotubes. Anal. Chem. 2020, 92, 8536–8545. [Google Scholar] [CrossRef]
- Shukla, R.P.; Aroosh, M.; Matzafi, A.; Ben-Yoav, H. Partially functional electrode modifications for rapid detection of dopamine in urine. Adv. Funct. Mater. 2021, 31, 2004146. [Google Scholar] [CrossRef]
- Posha, B.; Kuttoth, H.; Sandhyarani, N. 1-Pyrene carboxylic acid functionalized carbon nanotube-gold nanoparticle nanocomposite for electrochemical sensing of dopamine and uric acid. Microchim. Acta 2019, 186, 672. [Google Scholar] [CrossRef]
- Ashraf, G.; Asif, M.; Aziz, A.; Iftikhar, T.; Liu, H. Rice-spikelet-like copper oxide decorated with platinum stranded in the CNT network for electrochemical in vitro detection of serotonin. ACS Appl. Mater. Interfaces 2021, 13, 6023–6033. [Google Scholar] [CrossRef]
- Da Silva, L.V.; Dos Santos, N.D.; De Almeida, A.K.A.; Dos Santos, D.D.E.R.; Ferreira Santos, A.C.; Franca, M.C.; Lima, D.J.P.; Lima, P.R.; Goulart, M.O.F. A new electrochemical sensor based on oxidized capsaicin/multi-walled carbon nanotubes/glassy carbon electrode for the quantification of dopamine, epinephrine, and xanthurenic, ascorbic and uric acids. J. Electroanal. Chem. 2021, 881, 114919. [Google Scholar] [CrossRef]
- Ganesan, M.; Ramadhass, K.D.; Chuang, H.-C.; Gopalakrishnan, G. Synthesis of nitrogen-doped carbon quantum dots@Fe2O3/multiwall carbon nanotubes ternary nanocomposite for the simultaneous electrochemical detection of 5-fluorouracil, uric acid, and xanthine. J. Mol. Liq. 2021, 331, 115768. [Google Scholar] [CrossRef]
- Panczyk, T.; Wolski, P.; Lajtar, L. Coadsorption of doxorubicin and selected dyes on carbon nanotubes. Theoretical investigation of potential application as a pH-controlled drug delivery system. Langmuir 2016, 32, 4719–4728. [Google Scholar] [CrossRef]
- Singh, B.; Lohan, S.; Sandhu, P.S.; Jain, A.; Mehta, S.K. Functionalized carbon nanotubes and their promising applications in therapeutics and diagnostics. Nanobiomaterials Med. Imaging 2016, 2016, 455–478. [Google Scholar] [CrossRef]
- Zare, H.; Ahmadi, S.; Ghasemi, A.; Ghanbari, M.; Rabiee, N.; Bagherzadeh, M.; Karimi, M.; Webster, T.J.; Hamblin, M.R.; Mostafavi, E. Carbon Nanotubes: Smart Drug/Gene Delivery Carriers Int. J. Nanomed. 2021, 16, 1681–1706. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Kima, H.S.; Shin, U.S. Thermo conductive carbon nanotube-framed membranes for skin heat signal-responsive transdermal drug delivery. Polym. Chem. 2017, 8, 3154–3163. [Google Scholar] [CrossRef]
- Shi, X.; Zheng, Y.; Wang, C.; Yue, L.; Qiao, K.; Wang, G.; Wang, L.; Quan, H. Dual stimulus responsive drug release under the interaction of pH value and pulsatile electric field for a bacterial cellulose/sodium alginate/multi-walled carbon nanotube hybrid hydrogel. RSC Adv. 2015, 5, 41820–41829. [Google Scholar] [CrossRef]
- Estrada, A.C.; Silva, A.L.D.; Trindade, T. Photothermally enhanced drug release by κ-carrageenan hydrogels reinforced with multi-walled carbon nanotubes. RSC Adv. 2013, 3, 10828–10836. [Google Scholar] [CrossRef]
- Dhar, S.; Liu, Z.; Thomale, J.; Dai, H.; Lippard, S.J. Targeted single-wall carbon nanotube-mediated Pt (IV) prodrug delivery using folate as a homing device. J. Am. Chem. Soc. 2008, 130, 11467–111476. [Google Scholar] [CrossRef] [PubMed]
- Mehra, N.K.; Jain, N.K. Optimization of a pretargeted strategy for the PET imaging of colorectal carcinoma via the modulation of radioligand pharmacokinetics. Mol. Pharm. 2015, 12, 630–643. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.S.; Todkar, K.J.; Khutale, G.V.; Chate, G.P.; Biradar, A.V.; Gawande, M.B.; Zboril, R.; Khandare, J.J. Calcium phosphate nanocapsule crowned multiwalled carbon nanotubes for pH triggered intracellular anticancer drug release. J. Mater. Chem. B 2015, 3, 3931–3939. [Google Scholar] [CrossRef]
- Xu, H.; Liu, M.; Lan, M.; Yuan, H.; Yu, W.; Tian, J.; Wan, Q.; Zhang, X.; Wei, Y. Mussel-inspired PEGylated carbon nanotubes: Biocompatibility evaluation and drug delivery applications. Toxicol. Res. 2016, 5, 1371–1379. [Google Scholar] [CrossRef]
- Mejri, A.; Vardanega, D.; Tangour, B.; Gharbi, T.; Picaud, F. Substrate temperature to control moduli and water uptake in thin films of vapor deposited N,N’-Di(1-naphthyl)-N,N’-diphenyl-(1,1′-biphenyl)-4,4′-diamine (n.d.). J. Phys. Chem. B 2015, 119, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Lagopati, N.; Belogiannis, K.; Angelopoulou, A.; Papaspyropoulos, A.; Gorgoulis, V. Non-Canonical Functions of the ARF Tumor Suppressor in Development and Tumorigenesis. Biomolecules 2021, 11, 86. [Google Scholar] [CrossRef] [PubMed]
- Pantelis, P.; Theocharous, G.; Lagopati, N.; Veroutis, D.; Thanos, D.-F.; Lampoglou, G.-P.; Pippa, N.; Gatou, M.-A.; Tremi, I.; Papaspyropoulos, A.; et al. The Dual Role of Oxidative-Stress-Induced Autophagy in Cellular Senescence: Comprehension and Therapeutic Approaches. Antioxidants 2023, 12, 169. [Google Scholar] [CrossRef] [PubMed]
- Barbouti, A.; Lagopati, N.; Veroutis, D.; Goulas, V.; Evangelou, K.; Kanavaros, P.; Gorgoulis, V.G.; Galaris, D. Implication of Dietary Iron-Chelating Bioactive Compounds in Molecular Mechanisms of Oxidative Stress-Induced Cell Ageing. Antioxidants 2021, 10, 491. [Google Scholar] [CrossRef]
- Hwang, Y.S.; Park, S.H.; Lee, J.W. Applications of functionalized carbon nanotubes for the therapy and diagnosis of cancer. Polymers 2017, 9, 13. [Google Scholar] [CrossRef]
- Su, Y.; Hu, Y.; Li, M.; Zhou, J.; Liu, Y.; Wang, W.; Wang, Y.; Xu, X.; Yuan, Y.; Li, Y.; et al. A precision-guided MWNT mediated reawakening the sunk synergy in RAS for anti-angiogenesis lung cancer therapy. Biomaterials 2017, 139, 75–90. [Google Scholar] [CrossRef]
- Dong, X.; Sun, Z.; Wang, X.; Leng, X. An innovative MWCNTs/DOX/TC nanosystem for chemo-photothermal combination therapy of cancer. Nanomed. Nanotech. Biol. Med. 2017, 13, 2271–2280. [Google Scholar] [CrossRef]
- Kim, S.W.; Lee, Y.K.; Lee, J.Y.; Hong, J.H.; Khang, D. PEGylated anticancer-carbon nanotubes complex targeting mitochondria of lung cancer cells. Nanotechnology 2017, 28, 465102. [Google Scholar] [CrossRef]
- Song, J.; Wang, F.; Yang, X.; Ning, B.; Harp, M.G.; Culp, S.H.; Hu, S.; Huang, P.; Nie, L.; Chen, J.; et al. Gold nanoparticle coated carbon nanotube ring with enhanced Raman scattering and photothermal conversion Property for theranostic applications. J. Am. Chem. Soc. 2016, 138, 7005–7015. [Google Scholar] [CrossRef]
- Bao, Z.; Liu, X.; Liu, Y.; Liu, H.; Zhao, K. Near-infrared light-responsive inorganic nanomaterials for photothermal therapy. AJPS 2016, 11, 349–364. [Google Scholar] [CrossRef]
- Sahithi, K.; Swetha, M.; Ramasamy, K.; Srinivasan, N.; Selvamurugan, N. Polymeric composites containing carbon nanotubes for bone tissue engineering. Int. J. Biol. Macromol. 2010, 46, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.R.; Bae, H.; Cha, J.M.; Mun, J.Y.; Chen, Y.-C.; Tekin, H.; Shin, H.; Farshchi, S.; Dokmeci, M.R.; Tang, S.; et al. Carbon nanotube reinforced hybrid microgels as scaffold materials for cell encapsulation. ACS Nano 2011, 6, 362–372. [Google Scholar] [CrossRef]
- Sen, R.; Zhao, B.; Perea, D.; Itkis, M.E.; Hu, H.; Love, J.; Bekyarova, E.; Haddon, R.C. Preparation of single-walled carbon nanotube reinforced polystyrene and polyurethane nanofibers and membranes by electrospinning. Nano Lett. 2004, 4, 459–464. [Google Scholar] [CrossRef]
- Lau, C.; Cooney, M.J.; Atanassov, P. Conductive macroporous composite chitosan carbon nanotube scaffolds. Langmuir 2008, 24, 7004–7010. [Google Scholar] [CrossRef]
- Kam, N.W.S.; Jan, E.; Kotov, N.A. Electrical stimulation of neural stem cells mediated by humanized carbon nanotube composite made with extracellular matrix protein. Nano Lett. 2009, 9, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Worsley, M.A.; Kucheyev, S.O.; Kuntz, J.D.; Hamza, A.V.; Satcher, J.J.H.; Baumann, T.F. Stiff and electrically conductive composites of carbon nanotube aerogels and polymers. J. Mater. Chem. 2009, 19, 3370–3372. [Google Scholar] [CrossRef][Green Version]
- Shin, S.R.; Jung, S.M.; Zalabany, M.; Kim, K.; Zorlutuna, P.; Kim, S.; Nikkhah, M.; Khabiry, M.; Azize, M.; Kong, J.; et al. Carbon-nanotube-embedded hydrogel sheets for engineering cardiac constructs and bioactuators. ACS Nano 2013, 7, 2369–2380. [Google Scholar] [CrossRef]
- Yang, S.-T.; Luo, J.; Zhou, Q.; Wang, H. Pharmacokinetics, Metabolism and Toxicity of Carbon Nanotubes for Biomedical Purposes. Theranostics 2012, 2, 271–282. [Google Scholar] [CrossRef]
- Lam, C.-W.; James, J.T.; McCluskey, R.; Arepalli, S.; Hunter, R.L. A Review of carbon nanotube toxicity and assessment of potential occupational and environmental health risks. Crit. Rev. Toxicol. 2006, 36, 189–217. [Google Scholar] [CrossRef]
- Firme, C.P.; Bandaru, P.R. Toxicity issues in the application of carbon nanotubes to biological systems. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 245–256. [Google Scholar] [CrossRef]
- Sato, Y.; Yokoyama, A.; Shibata, K.-I.; Akimoto, Y.; Ogino, S.-I.; Nodasaka, Y.; Kohgo, T.; Tamura, K.; Akasaka, T.; Uo, M.; et al. Influence of length on cytotoxicity of multi-walled carbon nanotubes against human acute monocytic leukemia cell line THP-1 in vitro and subcutaneous tissue of rats in vivo. Mol. BioSyst. 2005, 1, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Wick, P.; Manser, P.; Limbach, L.K.; Dettlaff-Weglikowska, U.; Krumeich, F.; Roth, S.; Stark, W.J.; Bruinink, A. The degree and kind of agglomeration affect carbon nanotube cytotoxicity. Toxicol. Lett. 2007, 168, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Duch, M.C.; Budinger, G.R.S.; Liang, Y.T.; Soberanes, S.; Urich, D.; Chiarella, S.E.; Campochiaro, L.A.; Gonzalez, A.; Chandel, N.S.; Hersam, M.C.; et al. Minimizing oxidation and stable nanoscale dispersion improves the biocompatibility of graphene in the lung. Nano Lett. 2011, 11, 5201–5207. [Google Scholar] [CrossRef]
- Yang, K.; Wan, J.; Zhang, S.; Zhang, Y.; Lee, S.-T.; Liu, Z. In vivo pharmacokinetics, long-term biodistribution, and toxicology of pegylated graphene in mice. ACS Nano 2011, 5, 516–522. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.-H.; Kim, P.; Choi, J.-Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Eda, G.; Fanchini, G.; Chhowalla, M. Large-area ultrathin films of reduced graphene oxide as a transparent and flexible electronic material. Nat. Nanotechnol. 2008, 3, 270–274. [Google Scholar] [CrossRef]
- Tang, L.; Wang, Y.; Li, J. The graphene/nucleic acid nanobiointerface. Chem. Soc. Rev. 2015, 44, 6954–6980. [Google Scholar] [CrossRef] [PubMed]
- Akhavan, O.; Ghaderi, E.; Rahighi, R. Toward single-DNA electrochemical biosensing by graphene nanowalls. ACS Nano 2012, 6, 2904–2916. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.; Yang, L.; Li, X.; Zhou, J.; Chen, Q.; Yan, S.; Li, N.; Chu, M.; Dong, Y.; Xie, Z.; et al. An electrochemical DNA sensing platform using carboxyl functionalized graphene as the electrode modified material. J. Electrochem. Soc. 2017, 164, 345–351. [Google Scholar] [CrossRef]
- Ping, J.; Vishnubhotla, J.R.; Vrudhula, A.; Johnson, A.T.C. Scalable production of high-sensitivity, label-free DNA biosensors based on backgated graphene field Effect transistors. ACS Nano 2016, 10, 8700–8704. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Kang, H.; Naylor, C.H.; Streller, F.; Ducos, P.; Serrano, M.D.; Ping, J.; Zauberman, J.; Rajesh; Carpick, R.W.; et al. Scalable production of sensor arrays based on high-mobility hybrid graphene field effect transistors. ACS Appl. Mater. Interfaces 2016, 8, 27546–27552. [Google Scholar] [CrossRef]
- Zheng, X.T.; Ananthanarayanan, A.; Luo, K.Q.; Chen, P. Glowing graphene quantum dots and carbon dots: Properties, syntheses, and biological applications. Small 2015, 11, 1620–1636. [Google Scholar] [CrossRef]
- Kim, J.; Park, S.-Y.; Kim, S.; Lee, D.H.; Kim, J.H.; Kim, J.M.; Kang, H.; Han, J.-S.; Park, J.W.; Lee, H.; et al. Precise and selective sensing of DNA-DNA hybridization by graphene/Si-nanowires diode-type biosensors. Sci. Rep. 2016, 6, 31984. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Goo, N.I.; Kim, D.E. Mechanism of DNA adsorption and desorption on graphene oxide. Langmuir 2014, 30, 12587–12595. [Google Scholar] [CrossRef]
- Huang, Z.; Liu, J. Length-dependent diblock DNA with poly-cytosine (Poly-C) as high-affinity anchors on graphene oxide. Langmuir 2018, 34, 1171–1177. [Google Scholar] [CrossRef]
- Prabowo, B.A.; Alom, A.; Secario, M.K.; Masim, F.C.P.; Lai, H.-C.; Hatanaka, K.; Liu, K.-C. Graphene-based portable SPR Sensor for the detection of Mycobacterium tuberculosis DNA strain. Procedia Eng. 2016, 168, 541–545. [Google Scholar] [CrossRef]
- Kumar, S.; Amala, G.; Gowtham, S.M. Graphene based sensors in the detection of glucose in saliva—A promising emerging modality to diagnose diabetes mellitus. RSC Adv. 2017, 7, 36949–36976. [Google Scholar] [CrossRef]
- Zhang, M.; Liao, C.; Mak, C.H.; You, P.; Mak, C.L.; Yan, F. Highly sensitive glucose sensors based on enzyme-modified whole-graphene solution gated transistors. Sci. Rep. 2015, 5, 8311. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Park, H.G.; Ji, J.; Cho, J.; Jun, S.C. A Reduced graphene oxide based radio frequency glucose sensing device using multi-dimensional parameters. Micromachines 2016, 7, 136. [Google Scholar] [CrossRef] [PubMed]
- Mani, V.; Devasenathipathy, R.; Chen, S.M.; Subramani, B.; Govindasamy, M. A novel glucose biosensor at glucose oxidase immobilized graphene and bismuth nanocomposite film modified electrode. Int. J. Electrochem. Sci. 2015, 10, 691–700. [Google Scholar] [CrossRef]
- Thirumalraj, B.; Palanisamy, S.; Chen, S.M.; Yang, C.Y.; Periakaruppan, P.; Lou, B.S. Direct electrochemistry of glucose oxidase and sensing of glucose at a glassy carbon electrode modified with a reduced graphene oxide/fullerene-C60 composite. RSC Adv. 2015, 5, 77651–77657. [Google Scholar] [CrossRef]
- Bharath, G.; Madhu, R.; Chen, S.M.; Veeramani, V.; Balamurugan, A.; Mangalaraj, D. Enzymatic electrochemical glucose biosensors by mesoporous 1D hydroxyapatite-on-2D reduced graphene oxide. J. Mater. Chem. B 2015, 3, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, C.; Cheng, L.; Lee, S.T.; Liu, Z. Noble metal coated single-walled carbon nanotubes for applications in surface enhanced Raman scattering imaging and photothermal therapy. J. Am. Chem. Soc. 2012, 134, 7414–7422. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Dong, X.; Chen, P. Biological and chemical sensors based on graphene materials. Chem. Soc. Rev. 2012, 41, 2283–2307. [Google Scholar] [CrossRef]
- Zhang, H.; Grüner, G.; Zhao, Y. Recent advancements of graphene in biomedicine. J. Mater. Chem. B 2013, 1, 2542–2567. [Google Scholar] [CrossRef]
- Kundu, A.; Nandi, S.; Das, P.; Nandi, A.K. Fluorescent graphene oxide via polymer grafting: An efficient nanocarrier for both hydrophilic and hydrophobic drugs. ACS Appl. Mater. Interfaces 2015, 7, 3512–3523. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, S.; Li, Y.; Wang, M.; Shi, P.; Huang, X. Covalent functionalization of graphene oxide with biocompatible poly (ethylene glycol) for delivery of paclitaxel. ACS Appl. Mater. Interfaces 2014, 6, 17268–17276. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, L.; Li, X.; Jia, X.; Liu, L.; Zeng, J.; Guo, J.; Liu, P. Functionalized graphene oxide nanoparticles for cancer cell specific delivery of antitumor drug. Bioconjugate Chem. 2015, 26, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Lerra, L.; Farfalla, A.; Sanz, B.; Cirillo, G.; Vittorio, O.; Voli, F.; Le Grand, M.; Curcio, M.; Nicoletta, F.P.; Dubrovska, A.; et al. Graphene Oxide Functional Nanohybrids with Magnetic Nanoparticles for Improved Vectorization of Doxorubicin to Neuroblastoma Cells. Pharmaceutics 2018, 11, 3. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, M.; Rahmani, F.; Nouranian, S. Molecular simulation of pH-dependent diffusion, loading, and release of doxorubicin in graphene and graphene oxide drug delivery systems. J. Mater. Chem. B 2016, 4, 7441–7451. [Google Scholar] [CrossRef]
- Wei, G.; Dong, R.; Wang, D.; Feng, L.; Dong, S.; Song, A.; Hao, J. Functional materials from the covalent modification of reduced graphene oxide and βcyclodextrin as a drug delivery carrier. New J. Chem. 2014, 38, 140–145. [Google Scholar] [CrossRef]
- Yu, X.; Gao, D.; Gao, L.; Lai, J.; Zhang, C.; Zhao, Y.; Zhong, L.; Jia, B.; Wang, F.; Chen, X.; et al. Inhibiting metastasis and preventing tumor relapse by triggering host immunity with tumor-targeted photodynamic therapy using photosensitizer-loaded functional nanographenes. ACS Nano 2017, 11, 10147–10158. [Google Scholar] [CrossRef]
- Song, L.; Shi, J.; Lu, J.; Lu, C. Structure observation of graphene quantum dots by single-layered formation in layered confinement space. Chem. Sci. 2015, 6, 4846–4850. [Google Scholar] [CrossRef]
- Cheon, Y.A.; Bae, J.H.; Chung, B.G. Reduced graphene oxide nanosheet for chemo-photothermal therapy. Langmuir 2016, 32, 2731–2736. [Google Scholar] [CrossRef]
- Su, S.; Wang, J.; Vargas, E.; Wei, J.; Martínez-Zaguilán, R.; Sennoune, S.R.; Pantoya, M.L.; Wang, S.; Chaudhuri, J.; Qiu, J. Porphyrin immobilized nanographene oxide for enhanced and targeted photothermal therapy of brain cancer. ACS Biomater. Sci. Eng. 2016, 2, 1357–1366. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Zhang, R.; Lu, J.; Zhao, C.; Deng, X.; Wu, Y. Mesoporous silica coated polydopamine functionalized reduced graphene oxide for synergistic targeted chemo-photothermal therapy. ACS Appl. Mater. Interfaces 2017, 9, 1226–1236. [Google Scholar] [CrossRef]
- Dai, C.; Zhang, S.; Liu, Z.; Wu, R.; Chen, Y. Two-dimensional graphene augments nanosonosensitized sonocatalytic tumor eradication. ACS Nano 2017, 11, 9467–9480. [Google Scholar] [CrossRef]
- Ou, L.; Song, B.; Liang, H.; Liu, J.; Feng, X.; Deng, B.; Sun, T.; Shao, L. Toxicity of graphene-family nanoparticles: A general review of the origins and mechanisms. Part. Fibre Toxicol. 2016, 13, 57. [Google Scholar] [CrossRef] [PubMed]
- Shareena, T.P.; McShan, D.; Dasmahapatra, A.K.; Tchounwou, P.B. A review on graphene-based nanomaterials in biomedical applications and risks in environment and health. Nanomicro Lett. 2018, 10, 53. [Google Scholar] [CrossRef]
- Alshehri, R.; Ilyas, A.M.; Hasan, A.; Arnaout, A.; Ahmed, F.; Memic, A. Carbon nanotubes in biomedical applications: Factors, mechanisms, and remedies of toxicity. J. Med. Chem. 2016, 59, 8149–8167. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, Q.; Zhao, Y.; Bai, Y.; Chen, P.; Xia, T.; Wang, D. Response of MicroRNAs to in vitro treatment with graphene oxide. ACS Nano 2014, 8, 2100–2110. [Google Scholar] [CrossRef]
- Chong, Y.; Ge, C.; Yang, Z.; Gárate, J.A.; Gu, Z.; Weber, J.K.; Liu, J.; Zhou, R. Reduced cytotoxicity of graphene nanosheets mediated by blood-protein coating. ACS Nano 2015, 9, 5713–5724. [Google Scholar] [CrossRef]
- Perini, G.; Palmieri, V.; Ciasca, G.; De Spirito, M.; Papi, M. Unravelling the Potential of Graphene Quantum Dots in Biomedicine and Neuroscience. Int. J. Mol. Sci. 2020, 21, 3712. [Google Scholar] [CrossRef]
- Kumawat, M.K.; Thakur, M.; Gurung, R.B.; Srivastava, R. Graphene quantum dots for cell proliferation, nucleus imaging, and photoluminescent sensing applications. Sci. Rep. 2017, 7, 15858. [Google Scholar] [CrossRef]
- Qian, Z.S.; Shan, X.Y.; Chai, L.J.; Ma, J.J.; Chen, J.R.; Feng, H. A universal fluorescence sensing strategy based on biocompatible graphene quantum dots and graphene oxide for the detection of DNA. Nanoscale 2014, 6, 5671–5674. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, L.; Liang, R.P.; Bai, J.M.; Qiu, J.D. Using graphene quantum dots as photoluminescent probes for protein kinase sensing. Anal. Chem. 2013, 85, 9148–9155. [Google Scholar] [CrossRef]
- Xi, J.; Xie, C.; Zhang, Y.; Wang, L.; Xiao, J.; Duan, X.; Ren, J.; Xiao, F.; Wang, S. Ultrafine Pd nanoparticles encapsulated in microporous Co3O4 hollow nanospheres for in situ molecular detection of living cells. ACS Appl. Mater. Interfaces 2016, 8, 22563–22573. [Google Scholar] [CrossRef]
- Srivastava, A.; Yadav, T.; Sharma, S.; Nayak, A.; Kumari, A.; Mishra, N. Polymers in drug delivery. J. Biosci. Med. 2016, 4, 69–84. [Google Scholar] [CrossRef]
- Tian, Z.; Yao, X.; Ma, K.; Niu, X.; Grothe, J.; Xu, Q.; Liu, L.; Kaskel, S.; Zhu, Y. Metal-organic framework/graphene quantum dot nanoparticles used for synergistic chemoand photothermal therapy. ACS Omega 2017, 2, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Zhang, F.; Zhao, C.; Lv, Y.; Ma, G.; Wei, W.; Tian, Z. Beyond a Carrier: Graphene quantum dots as a probe for programmatically monitoring anti-cancer drug delivery, release, and response. ACS Appl. Mater. Interfaces 2017, 9, 27396–27401. [Google Scholar] [CrossRef] [PubMed]
- Nigam, P.; Waghmode, S.; Louis, M.; Wangnoo, S.; Chavan, P.; Sarkar, D. Graphene quantum dots conjugated albumin nanoparticles for targeted drug delivery and imaging of pancreatic cancer. J. Mater. Chem. B 2014, 2, 3190–3195. [Google Scholar] [CrossRef]
- Iannazzo, D.; Pistone, A.; Salamò, M.; Galvagno, S.; Romeo, R.; Giofré, S.V.; Branca, C.; Visalli, G.; Di Pietro, A. Graphene quantum dots for cancer targeted drug delivery. Int. J. Pharm. 2017, 518, 185–192. [Google Scholar] [CrossRef]
- Sui, X.; Luo, C.; Wang, C.; Zhang, F.; Zhang, J.; Guo, S. Graphene quantum dots enhance anticancer activity of cisplatin via increasing its cellular and nuclear uptake. Nanomedicine 2016, 12, 1997–2006. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, X.; Bhirde, A.; Cao, J.; Zeng, Y.; Huang, X.; Sun, Y.; Liu, G.; Chen, X. Carbondot-based two-photon visible nanocarriers for safe and highly efficient delivery of siRNA and DNA. Adv. Healthc. Mater. 2014, 3, 1203–1209. [Google Scholar] [CrossRef]
- Some, S.; Gwon, A.-R.; Hwang, E.; Bahn, G.-H.; Yoon, Y.; Kim, Y.; Kim, S.-H.; Bak, S.; Yang, J.; Jo, D.-G.; et al. Cancer therapy using ultrahigh hydrophobic drug-loaded graphene derivatives. Sci. Rep. 2014, 4, 6314. [Google Scholar] [CrossRef]
- Ge, J.; Lan, M.; Zhou, B.; Liu, W.; Guo, L.; Wang, H.; Jia, Q.; Niu, G.; Huang, X.; Zhou, H.; et al. A graphene quantum dot photodynamic therapy agent with high singlet oxygen generation. Nat. Commun. 2014, 5, 4596. [Google Scholar] [CrossRef]
- Yao, X.; Niu, X.; Ma, K.; Huang, P.; Grothe, J.; Kaskel, S.; Zhu, Y. Graphene quantum dots-capped magnetic mesoporous silica nanoparticles as a multifunctional platform for controlled drug delivery, magnetic hyperthermia, and photothermal therapy. Small 2017, 13, 1602225. [Google Scholar] [CrossRef]
- Li, K.; Liu, W.; Ni, Y.; Li, D.; Lin, D.; Su, Z.; Wei, G. Technical synthesis and biomedical applications of graphene quantum dots. J. Mater. Chem. B 2017, 5, 4811–4826. [Google Scholar] [CrossRef]
- Ailuno, G.; Balboni, A.; Caviglioli, G.; Lai, F.; Barbieri, F.; Dellacasagrande, I.; Florio, T.; Baldassari, S. Boron Vehiculating Nanosystems for Neutron Capture Therapy in Cancer Treatment. Cells 2022, 11, 4029. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, K.L.; Goreham, R.V.; Nann, T. Graphene Quantum Dots for Theranostics and Bioimaging. Pharm. Res. 2016, 33, 2337–2357. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Shen, H.; Wang, H.; Wang, J.; Li, J.; Nienhaus, G.U.; Shang, L.; Wei, G. Motif-designed peptide nanofibers decorated with graphene quantum dots for simultaneous targeting and imaging of tumor cells. Adv. Funct. Mater. 2015, 25, 5472–5478. [Google Scholar] [CrossRef]
- Zheng, X.T.; He, H.L.; Li, C.M. Multifunctional graphene quantum dots-conjugated titanate nanoflowers for fluorescence-trackable targeted drug delivery. RSC Adv. 2013, 3, 24853–24857. [Google Scholar] [CrossRef]
- Huang, C.-L.; Huang, C.-C.; Mai, F.-D.; Yen, C.-L.; Tzing, S.-H.; Hsieh, H.-T.; Ling, Y.-C.; Chang, J.-Y. Application of paramagnetic graphene quantum dots as a platform for simultaneous dual-modality bioimaging and tumor-targeted drug delivery. J. Mater. Chem. B. 2015, 3, 651–664. [Google Scholar] [CrossRef]
- Wang, J.; Cao, S.; Ding, Y.; Ma, F.; Lu, W.; Sun, M. Theoretical investigations of optical origins of fluorescent graphene quantum dots. Sci. Rep. 2016, 6, 24850. [Google Scholar] [CrossRef]
- Liang, L.; Kong, Z.; Kang, Z.; Wang, H.; Zhang, L.; Shen, J.W. Theoretical evaluation on potential cytotoxicity of graphene quantum dots. ACS Biomater. Sci. Eng. 2016, 2, 1983–1991. [Google Scholar] [CrossRef]
- Tian, X.; Xiao, B.-B.; Wu, A.; Yu, L.; Zhou, J.; Wang, Y.; Wang, N.; Guan, H.; Shang, Z.-F. Hydroxylated graphene quantum dots induce cells senescence in both p53-dependent and -independent manner. Toxicol. Res. 2016, 5, 1639–1648. [Google Scholar] [CrossRef]
- Nurunnabi, M.; Khatun, Z.; Huh, K.M.; Park, S.Y.; Lee, D.Y.; Cho, K.J.; Lee, Y.-K. In Vivo biodistribution and toxicology of carboxylated graphene quantum dots. ACS Nano 2013, 7, 6858–6867. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gatou, M.-A.; Vagena, I.-A.; Pippa, N.; Gazouli, M.; Pavlatou, E.A.; Lagopati, N. The Use of Crystalline Carbon-Based Nanomaterials (CBNs) in Various Biomedical Applications. Crystals 2023, 13, 1236. https://doi.org/10.3390/cryst13081236
Gatou M-A, Vagena I-A, Pippa N, Gazouli M, Pavlatou EA, Lagopati N. The Use of Crystalline Carbon-Based Nanomaterials (CBNs) in Various Biomedical Applications. Crystals. 2023; 13(8):1236. https://doi.org/10.3390/cryst13081236
Chicago/Turabian StyleGatou, Maria-Anna, Ioanna-Aglaia Vagena, Natassa Pippa, Maria Gazouli, Evangelia A. Pavlatou, and Nefeli Lagopati. 2023. "The Use of Crystalline Carbon-Based Nanomaterials (CBNs) in Various Biomedical Applications" Crystals 13, no. 8: 1236. https://doi.org/10.3390/cryst13081236
APA StyleGatou, M.-A., Vagena, I.-A., Pippa, N., Gazouli, M., Pavlatou, E. A., & Lagopati, N. (2023). The Use of Crystalline Carbon-Based Nanomaterials (CBNs) in Various Biomedical Applications. Crystals, 13(8), 1236. https://doi.org/10.3390/cryst13081236









