1. Introduction
In crystals used as scintillating materials, ionizing radiation creates regions of extremely high densities of electronic excitations. These are the regions in the vicinity of the axis of tracks created by heavy ionizing particles (protons, alpha-particles et al.), and at the end of electron tracks, energy deposition there approaches 50 to 100 eV per nanometer of particle trajectory [
1,
2,
3,
4,
5]. The concentration of excitations created within femtosecond time in these regions reaches 10
20–10
21 elementary excitations (excitons and electron–hole pairs) per cubic centimeter. Such concentrations of excitations result in efficient interaction between them up to e-h plasma effects. Relaxation of electronic excitations and their Auger-type annihilation leads to a fast decrease in their concentration down to the values when the interaction becomes weak. If we deal with fast nanosecond and subnanosecond scintillators [
6,
7] their radiative relaxation is to a large extent defined by the initial relaxation phase when the concentration of the charge carriers is high. A high concentration of interacting excitations created simultaneously can be achieved by excitation of the matter by femtosecond radiation of high intensity if its photon energy falls into the range of exciton or electron–hole pair creation. The method of Z-scan designed for the measurements of non-linear absorption coefficients in the transparency region, when applied in the fundamental absorption range, allows for the investigation of luminescence yield and kinetics as a function of controlled excitation density. Femtosecond laser radiation focused by a lens creates a substantially non-uniform distribution of electronic excitations both across the spot at the sample surface and along the depth profile [
8,
9]. Thus the method yields emission parameters averaged over the concentration of excitations in the laser spot. The application of this technique for the investigation of scintillating single crystals demonstrates that non-proportionality of the scintillation yield (yield dependence on the ionizing particle energy) is caused by the interaction of excitations in the regions of their high concentration, which affects scintillation kinetics as well. It was demonstrated that in some crystals (e.g., crystals with oxy-anions), high excitation density results in luminescence quenching accompanied by the acceleration of its kinetics [
10,
11,
12]; in the other ones, the luminescence yield can substantially increase with the excitation intensity. For the latter case, a strong non-proportionality of the scintillation yield is observed: the yield is lower both for low energies of the ionizing particles (e.g., electrons with the energy below 10–50 KeV) and for high energies (above 200 KeV) as compared to the region of intermediate energy. Examples of such materials are ZnO, undoped CsI, and CsPbCl
3 [
13]. These crystals have a low luminescence yield when excited in the fundamental absorption region (below the threshold of photon multiplication); however, their scintillation yield is reasonable when excited by X-rays or ionizing particles. Here, we present the results for one of the similar crystals, namely lead halide perovskite with an organic cation, which, together with high photoconductivity, demonstrates a good yield of subnanosecond luminescence excited by ionizing radiation. Traditional scintillators are not renowned for high photoconductivity, while crystals producing electric current in response to ionizing radiation normally do not have a substantial luminescence yield. The combination of contradictory parameters in the material under investigation is undoubtedly attractive for the study of the physics of processes under high-density excitation as well as for the practical task of the creation of scintillators with time resolution at the level of tens of picoseconds.
2. Materials and Methods
Single crystals of CH
3NH
3PbBr
3 (MAPbBr
3) with perovskite structure were synthesized using well-known anti-solvent vapor-assisted crystallization [
14] but with accurate controlled diffusion of anti-solvent vapor into process solvent. The single-crystal growth was passed according to the chemical reaction:
N,N-dimethylformamide C
3H
7NO (DMF) was chosen as a solvent as it is characterized by the high solubility of MABr и PbBr
2 in it. Dichloromethane CH
2Cl
2 (DCM) was used as an anti-solvent. The crystals were grown by spontaneous crystallization method over a period of 4–5 days with low growth rates.
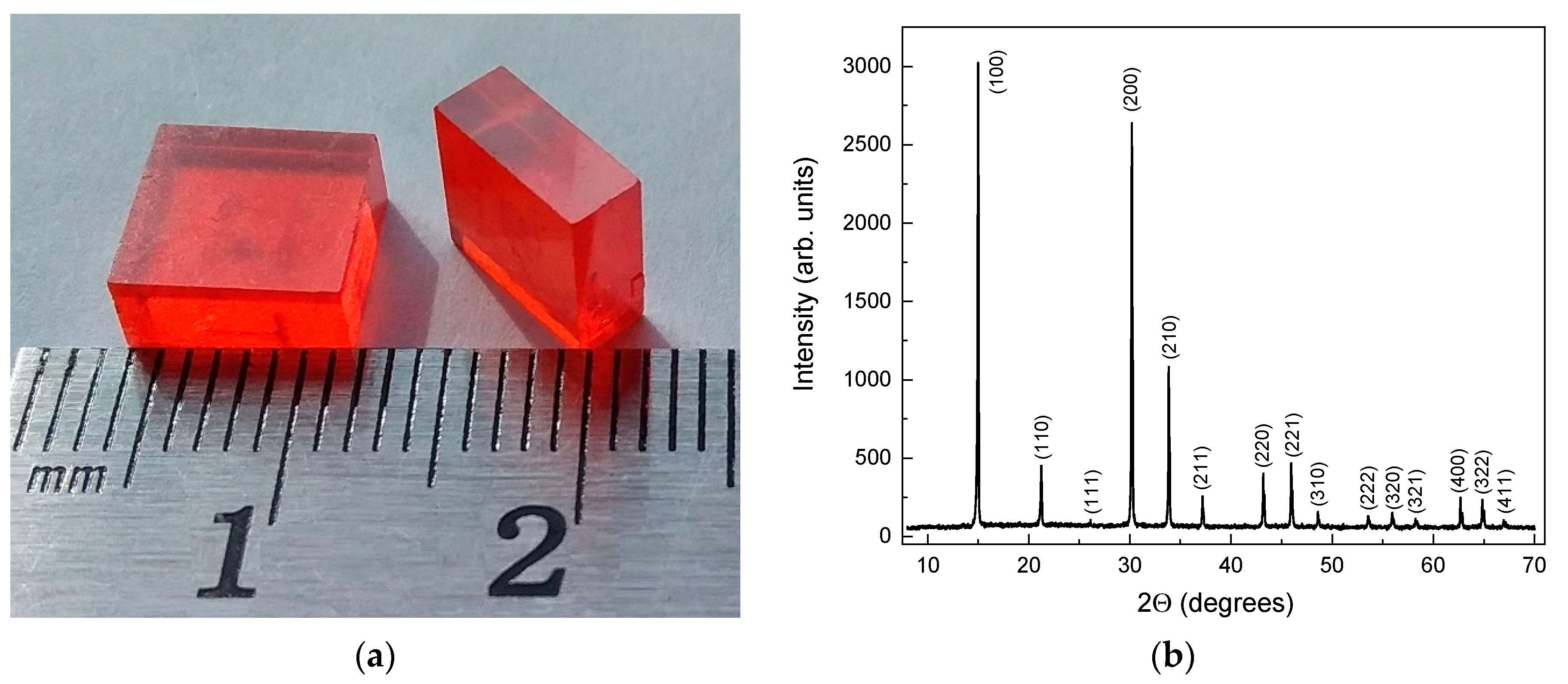
The resulting MAPbBr3 single crystals are shown in
Figure 1a. The crystals had a flat-cubic shape with dimensions of 8 × 8 × 3 mm
3, orange color, mirror-smooth crystal faces with indexes [100], and did not contain internal inclusions. The XRD patterns of crystals (
Figure 1b) contain only peaks that belong to a high-temperature cubic phase of MAPbBr
3 with
spatial group. The value of the lattice parameter was calculated as 5.913 (7) Å, which corresponds well to known estimates. The high sharpness of the XRD peaks and the absence of extraneous one indicates the high crystallinity and phase purity of the grown crystals.
The characterization of the crystal structure of MAPbBr3 crystals was carried out by the X-ray powder diffraction method on a Rigaku Ultima IV diffractometer with Bragg–Brentano geometry, using CuKα radiation.
Luminescence emission and excitation spectra were measured using a 150 W xenon lamp (Oriel Instruments, Stratford, CT, USA), MDR-206 primary monochromator (Lomo, Saint-Petersburg, Russia), and LOT-Oriel MS-257 spectrograph (Oriel Instruments, Stratford, CT, USA) equipped with Marconi CCD detector (Marconi Applied Technologies Limited, Chelmsford, UK). Samples were mounted into Cryotrade LN-120 vacuum optical cryostat (Cryotrade Engineering, Moscow, Russia).
Luminescence emission spectra and kinetics under X-ray excitation (hν = 19 keV) were measured at the P23 station of the PETRA III storage ring at DESY (Hamburg, Germany). The experimental setup included a nitrogen cryostat, monochromator Andor Kymera 328i, equipped with an Andor iCCD detector and MCP-PMT Hamamatsu R3809U-50.
The effect of excitation density was studied using the Z-scan technique in the CELIA laboratory (Bordeaux, France). The 3rd harmonic (λ = 266 нм) of Ti:Al
2O
3 laser with pulses of 26 fs and 9 mJ/pulse at 1 kHz frequency was used for luminescence excitation [
15]. The samples were in a vacuum helium cryostat with a temperature range of 13 to 300 K. The laser beam was focused onto the sample surface using a lens fixed on a platform moving along the beam axis Z. Photoluminescence signal was transferred to the entrance of TRIA X Jobin-Yvon 190 spectrometer through an optical fiber. ANDOR iCCD was used to record luminescence spectra and MCP Hamamatsu R3809U-58 was used to record luminescence kinetics with a time resolution of ~50 ps.
The excitation density variation is achieved by moving the focusing lens along the laser beam, its 120 mm travel changes the size of the laser spot on the sample surface from 20 to 400 μm in diameter. In the assumption of constant laser pulse output J, it allows for changing the excitation density by 400 times. The relation between the lens position Z and excitation fluence at the beam axis I
0 (in the maximum of Gaussian distribution) is given by the following equations [
13]:
Here, J is the energy of laser pulses (in our experiments J had the values of 1, 2, 5, 10, 20, 50, and 100 nJ), M
2 is the beam quality factor (for the third harmonic M~1),
w0 is the size of laser beam waist (~20 μm), λ—exciting wavelength (266 nm),
f—lens focusing distance.
3. Results
We shall start with the luminescence spectra of MAPbBr
3 measured with laser excitation to introduce some effects specific to hybrid perovskites.
Figure 2 shows respective spectra measured at different temperatures and excitation densities. At relatively low excitation density (I
0 = 7.6 μJ/cm
2), the luminescence spectra clearly exhibit an exciton feature, that is centered near the MAPbBr
3 absorption edge (
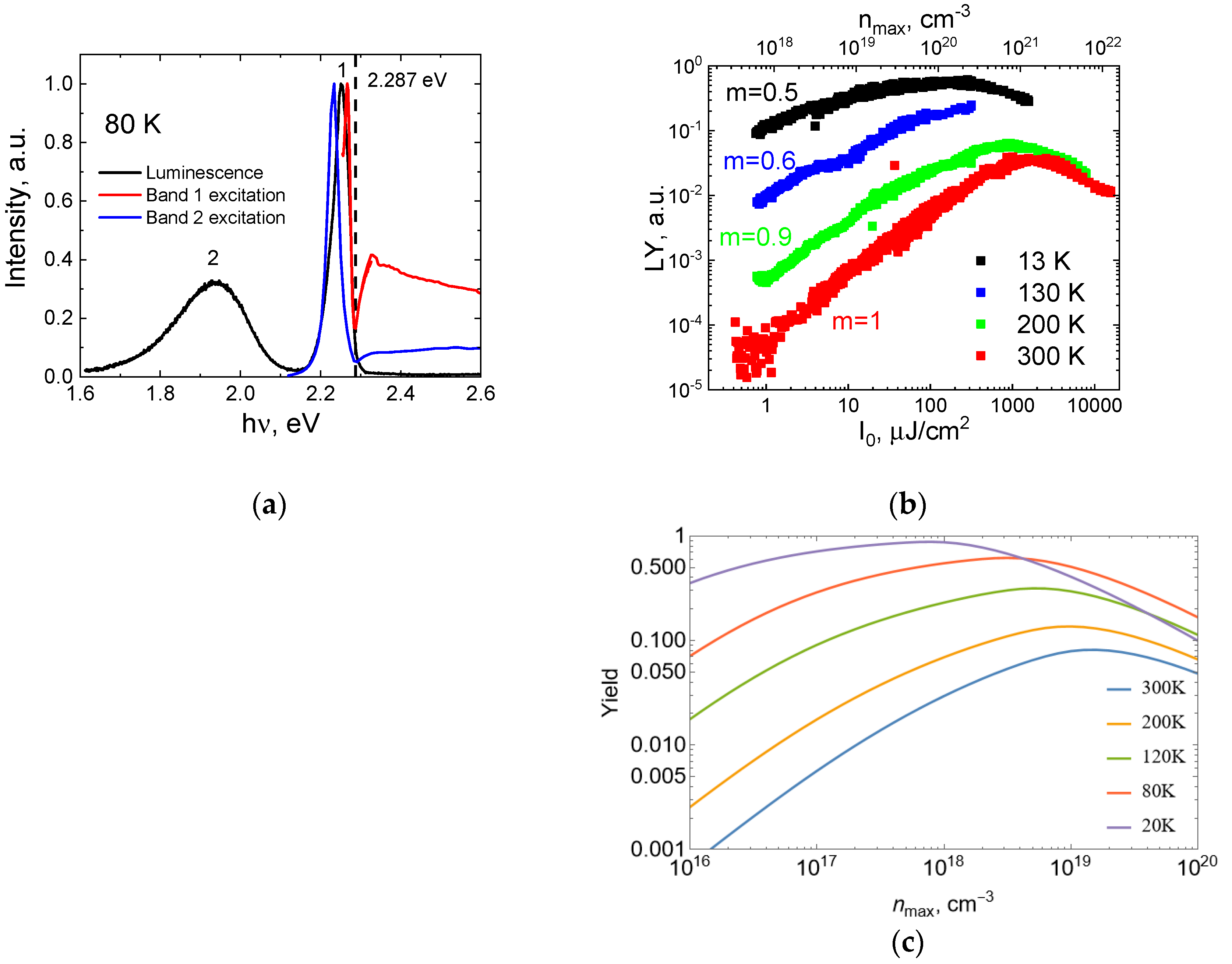
Figure 2a). At 13 K the exciton peak is centered at 2.248 eV and has FWHM of approximately 18 meV. In addition, another peak is present, which is red-shifted compared to the exciton one by approximately 40 meV. This feature can be observed at ~40 K; however, its intensity gradually decreases with temperature and completely disappears by 80 K.
The exciton peak shifts toward higher energies and slightly broadens with temperature. In the temperature range 140 K–160 K, we observe an abrupt change in the luminescence spectrum profile: luminescence maximum red-shifts by approximately 11 meV (from 2.3 to 2.289 eV) and the exciton peak widens by the factor of 1.67 (FWHM increases from 30 to 50 meV). This corresponds to the phase transition of MAPbBr
3 crystal lattice that changes its structure from the othorombic to tetragonal one at 145 K [
16].
At temperatures above 160 K, only the exciton peak is left in the luminescence spectra. The second phase transition (from the tetragonal to cubic phase) is known to occur at 220 K [
16]; however, it does not introduce substantial changes in the MAPbBr
3 luminescence spectrum. Exciton peak continues to gradually blue-shift and broaden with temperature until we finally arrive at room temperature, where the maximum position is 2.322 eV and FWHM is approximately 94 meV.
It should be noted that the position of the exciton luminescence peak is provided by different authors with substantial discrepancy, e.g., 2.175 eV in [
14], 2.3 eV in [
17], and 2.35 eV in [
18]. Our experimental results allow us to somewhat account for this discrepancy, suggesting that the luminescence spectra profiles of MAPbBr
3 depend largely on excitation conditions.
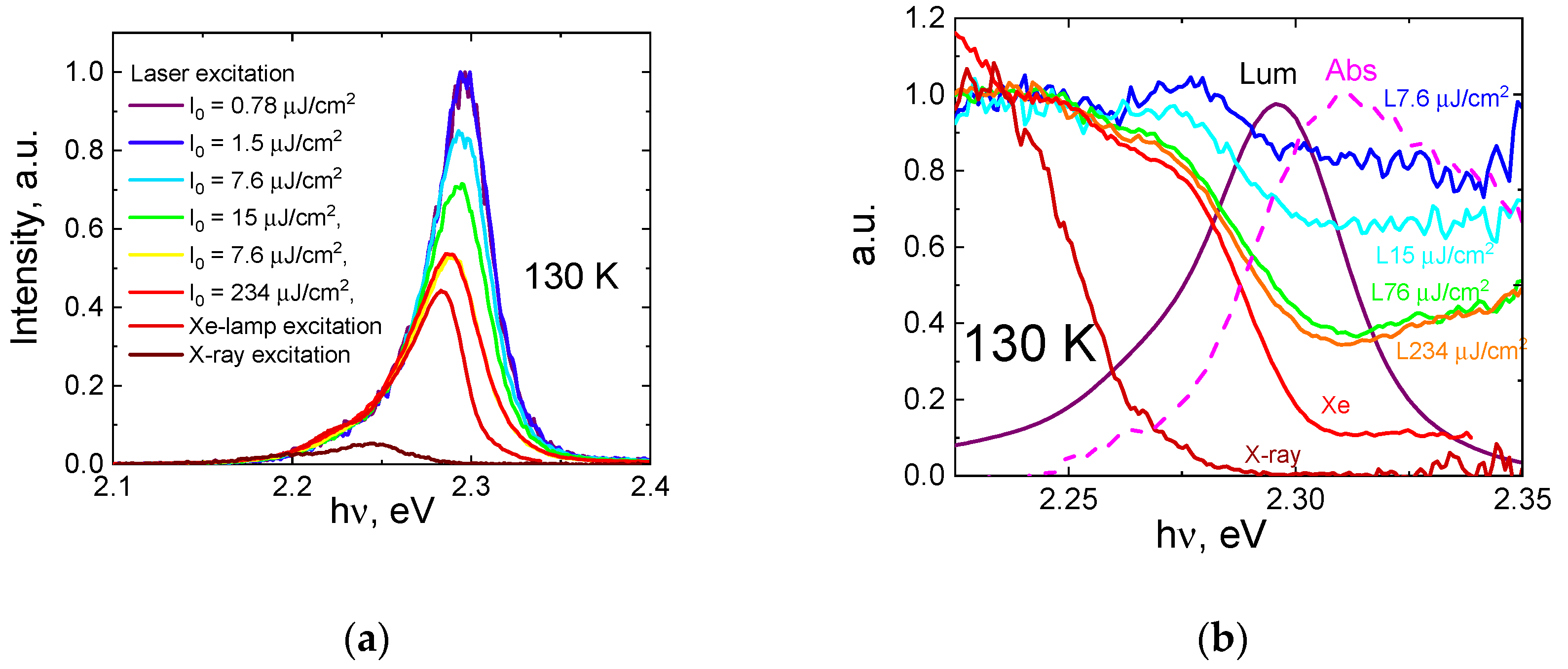
Figure 2b shows luminescence spectra measured at 300 K using the Z-scan technique. At higher excitation densities the maximum of luminescence spectra red-shifts (from 2.334 eV at I
0 = 1 μJ/cm
2 to 2.304 eV at 10 mJ/cm
2) and the exciton line broadens (from 94 meV at I
0 = 1 μJ/cm
2 to 124 meV at 10 mJ/cm
2). A similar tendency was observed for other investigated temperatures. While this example does not cover all the variety of luminescence parameters reported in the literature, it clearly illustrates the influence of excitation conditions on the observed luminescence spectra. This issue is addressed further in the text.
For comparison,
Figure 3a shows the luminescence spectrum measured with a different excitation source—a xenon lamp (80 K). Under these conditions, the exciton luminescence peak (marked as 1 in
Figure 3a) is centered at 2.254 eV. However, with laser excitation at the same temperature, it is centered at 2.281 eV. The reason for that is in different penetration depths of 3 eV photons (xenon lamp excitation) and 4.65 eV photons (laser excitation), in the latter case, the absorption coefficient is several times higher. The effect of reabsorption of luminescence light increases with the penetration depth of the exciting radiation, strong reabsorption is due to small Stokes shift [
19]. The value of the shift is illustrated by the luminescence excitation spectrum shown in the same graph.
Both the excitation spectrum of the excitonic luminescence and that of a defect emission (band 2 in
Figure 3) have a local minimum at 2.287 eV. This minimum corresponds to the maximum of the absorption spectra manifested via surface losses. Quite often, the features of the excitation spectra in the fundamental absorption region are anti-correlated with the features of the absorption, meaning that the minima of the excitation spectra coincide in position with the maxima of the absorption spectra and vice versa, which is usually explained by the surface losses (see [
20,
21,
22,
23] and
Appendix A). So far, we did not find luminescence excitation spectra for MAPbBr
3 in the literature. In [
24], such excitation spectrum for MAPbCl
3 was measured and it has quite a usual profile: the peak of the excitation spectrum coincides with that of the absorption. Diffusion of the charge carriers to the surface defects, where they recombine non-radiatively, is the origin of the observed effect. Renowned mobility of charge carriers in hybrid perovskites [
25] supports this hypothesis and the strength of the effect in our case (though the sample was freshly cleaved prior to the measurements) indicates that the number of surface defects was large. An additional luminescence band peaking at 1.93 eV in the same figure can be attributed to defects in the bulk. It is efficiently excited in the region of exciton absorption; however, its highest yield is achieved at the low-energy tail of the exciton absorption, where the absorption coefficient is smaller and surface losses are less pronounced. To summarize, in our specific crystal, we must take into account losses due to the surface defects and defects in the bulk.
Figure 3b shows the dependence of luminescence light yield (LY) from the excitation density at different temperatures. By LY we mean the number of luminescence photons divided by the number of incident photons. There are two X-axes in the Figure, one with habitual I
0 μJ/cm
2, and the other one, namely n
max, needs additional comments. The intensity of the laser light on the sample surface has a Gaussian distribution relative to the beam axis. The distribution of absorbed light into the sample depth obeys Beer’s law. Thus, the distribution of carriers created by the light absorption will be strongly inhomogeneous both along the radial coordinate ρ and depth x. To characterize the density of created charge carriers we introduce n
max standing for the maximum concentration of carriers in the center of the laser spot near the surface:
Here,
w(Z) and I
0(Z) are described by formulae (2) and (3), k is the absorption coefficient of MAPbBr
3 taken for hν = 4.65 eV equal to 6 × 10
5 cm
−1 [
26]. The carrier distribution then will be written as:
Such effects as absorption saturation and charge carrier diffusion would affect this distribution resulting in the reduction of the actual maximum density. At RT the LY(n
max) or LY (I
0) dependence can be subdivided into three areas: (i) an almost linear dependence of LY(n
max), which corresponds to a quadratic increase in the luminescence intensity with increasing photon density; (ii) for n
max > 4 × 10
20 cm
−3 (I
0~600 μJ/cm
2) slowing down of LY(n
max) growth with the saturation at n
max~10
21 cm
−3; (iii) decrease in the LY(n
max) with further increase in the excitation density. These results are in good agreement with the works of [
27]. Temperature dependence of the excitation density effects was not investigated previously. At room temperature, we see three orders of magnitude increase in the light yield with excitation density, while at 13 K it is less than one order. Moreover, the saturation of the yield is observed at different densities at different temperatures corresponding to the lowest one at 13 K.
It is natural to explain the observed effect by the conditions of the co-existence of excitons and electron–hole pairs changing with excitation density and temperature. The excitons decay radiatively and charge carriers can be trapped by defects. The ratio of the exciton concentration to the product of electrons and hole concentration in the stationary case depends on temperature and exciton binding energy and is described by kinetic equations in
Appendix B. In our case of excitation by femtosecond laser pulses, we have to account for the following dynamic processes: carrier capture by the defects, radiative decay of excitons, binding of electrons and holes into excitons, and a reverse process using rate equations. We also take account of the concentration-dependent exciton binding energy due to the screening effect [
28], as well as the absence of excitonic states at concentrations above Mott density [
29,
30]. This approach gives qualitative agreement with experimental results (
Figure 3c). The reproduced different density dependencies of luminescence yield at different temperatures, temperature-dependent positions of the maximum of respective curves, as well as the temperature-dependent slopes of the observed yield increase, cannot be attributed to a single process, a whole list of considered competing processes contributes to the observed features. The simulation gave the best agreement for exciton Rydberg energy of 40 meV.
The luminescence kinetics of MAPbBr
3 is also strongly affected by the excitation conditions as is demonstrated by
Figure 4a with the temperature dependence of the kinetics. The instrument response function (IRF) is presented as well. All luminescence decay curves are strongly nonexponential: in the region of picosecond times, the kinetics is affected by the interaction of electronic excitations in the regions of their high density and/or competing channels such as precursor electron or hole capture by defects. In the nanosecond region, the origin of kinetics departure from the true radiative one is due to the processes of re-emission following re-absorption. The increase in the decay time depends on the number of reabsorption events, see, e.g., [
31]. The region of larger times (not presented in the figure) is sensitive to electrons and holes released from shallow traps and binding into excitons (tens and hundreds of nanoseconds). To trace the trends in kinetics changing with temperature and excitation density they were parameterized using the following formula:
which allows the introduction of average decay time τ. An example of fitting parameters for decay kinetics at 200 K is shown in
Table 1.
At 13 K, the decay is very close to IRF, the average decay time can be evaluated as ~55 ps. This value is substantially smaller than the nanoseconds presented, e.g., in [
32]. The difference is due to quenching by defects in agreement with concentrations used in
Appendix B for simulation. With temperature increases up to 130 K, the decays slow down, and the average decay constant is ~120 ps. Further increase in the temperature results in the opposite effect: at 200 K decay is faster, than at 130 K with an average time of ~100 ps. This effect can be attributed to the phase transition.
It should be noted that luminescence kinetics observed with laser excitation at temperatures above 60 K agree rather well both with our measurements under X-ray excitation and with literature data [
32]; however, at 13 K, our decay is much faster than measured in [
32]. The reason for that is discussed in the next section.
Figure 4b shows the kinetics of exciton luminescence at different excitation densities (at 200 K). In the range of I
0 from 0.8 to 25 μJ/cm
2 (n
max ~5 × 10
17–2 × 10
19 cm
−3), the subnanosecond part of decay kinetics with τ < 1 ns remains the same, on the contrary, the slower nanosecond component decreases and this tendency is confirmed by the decrease in the average decay time to 93 ps. With a further increase in I
0~25 −800 μJ/cm
2, both picosecond and nanosecond decay components slow down and the average time increases from 100 to 220 ps.
4. Discussion
In the previous section, we have shown that the luminescence spectra profile depends on the excitation conditions. It is important to note that most changes occur in the higher-energy part of the spectra, while the lower-energy wing of the luminescence band stays almost unchanged. This is illustrated by
Figure 5a, where luminescence spectra measured at different excitation conditions (laser excitation with different excitation density, Xe-lamp excitation, X-ray excitation) are normalized to the low-energy wing (at an arbitrary temperature of 130 K).
One of the effects modulating the profile of the emission spectra depending on the excitation density, energy, and temperature is the re-absorption, which in hybrid perovskites is known to be quite pronounced. It is problematic to measure absorption in this spectral range from a single crystal in a wide dynamic range from the Urbach tail (10
2 cm
−1) to the excitonic absorption (10
5 cm
−1). Absorption spectra found in the literature are quite different and depend on many factors such as the quality of the samples, surface conditions, method of the measurements, etc. [
18,
26,
33,
34,
35]. The depth of luminescence escape, defined in a simple case by Beer’s law only (neglecting surface losses and diffusion, which accounts for the actual charge carrier distribution) can be described as:
where
is the true profile of the luminescence spectrum (in the bulk),
is the absorption coefficient, and
is an averaged coordinate of the emitting exciton. Assuming that, for various excitation conditions, we can introduce respective
, dividing two luminescence spectra differently according to the excitation energy or excitation density yields
, which can be re-written as
. Thus we tried to evaluate the re-absorption by dividing the luminescence spectra measured at different excitation densities and normalized to their long wavelength side by the spectrum measured at the lowest density (the resulting curves are shown in
Figure 5b). The absorption spectrum multiplied by an unknown constant
is shown by a magenta dashed line in
Figure 5b. It represents a spectral distribution of normalized absorption, its absolute value can be obtained from the comparison with experimental values from the literature, e.g., measured by ellipsometry [
26]. Now, we can estimate the dependence of the shift
of the emitting point on the measurement conditions. As a denominator, we used the luminescence spectrum measured at a low density of 0.78 μJ/cm
2 laser radiation at 4.65 eV with the shortest penetration depth of ~17 nm [
26] least affected by the re-absorption. The excitation density of 15 μJ/cm
2 results in the shift of the emitting point by 65 nm, for 230 μJ/cm
2 this value is 140 nm. An increase in
value with the excitation density is related to the absorption saturation. Excitation with a xenon lamp at 3 eV gives about a 300 nm increase in the escape depth while the penetration depth for 3 eV photons is 150 nm. This discrepancy is well explained by the diffusion of the charge carriers. The difference 300 nm–150 nm = 150 nm can be used for the evaluation of the diffusion length for our specific crystal. This value falls within the range provided in the literature but from its low side, it is much less than the tens of microns suggested in [
36]. The important role of defects in our crystals (defining short diffusion length) is confirmed by decay kinetics of the excitons with a very short characteristic decay time of the order of tens of picoseconds even at low temperature (
). X-ray excitation (19 keV) penetrates to a depth of ~100 μm, hence luminescence light has to travel a long distance before the escape and the observer sees only a part of the spectrum corresponding to the region, where the absorption is low. More accurately, re-absorption is considered in
Appendix A.
Re-absorption inherent to the studied perovskite limits its application as a scintillator for spectrometer measurements and in cases that require high energy resolution. However, its fast decay, which can be controlled by material engineering, can be quite attractive for applications demanding high coincidence time resolution.