Diffusion Bonding of Al7075 to Ti-6Al-4V by Spark Plasma Sintering and Using a Copper Interlayer
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
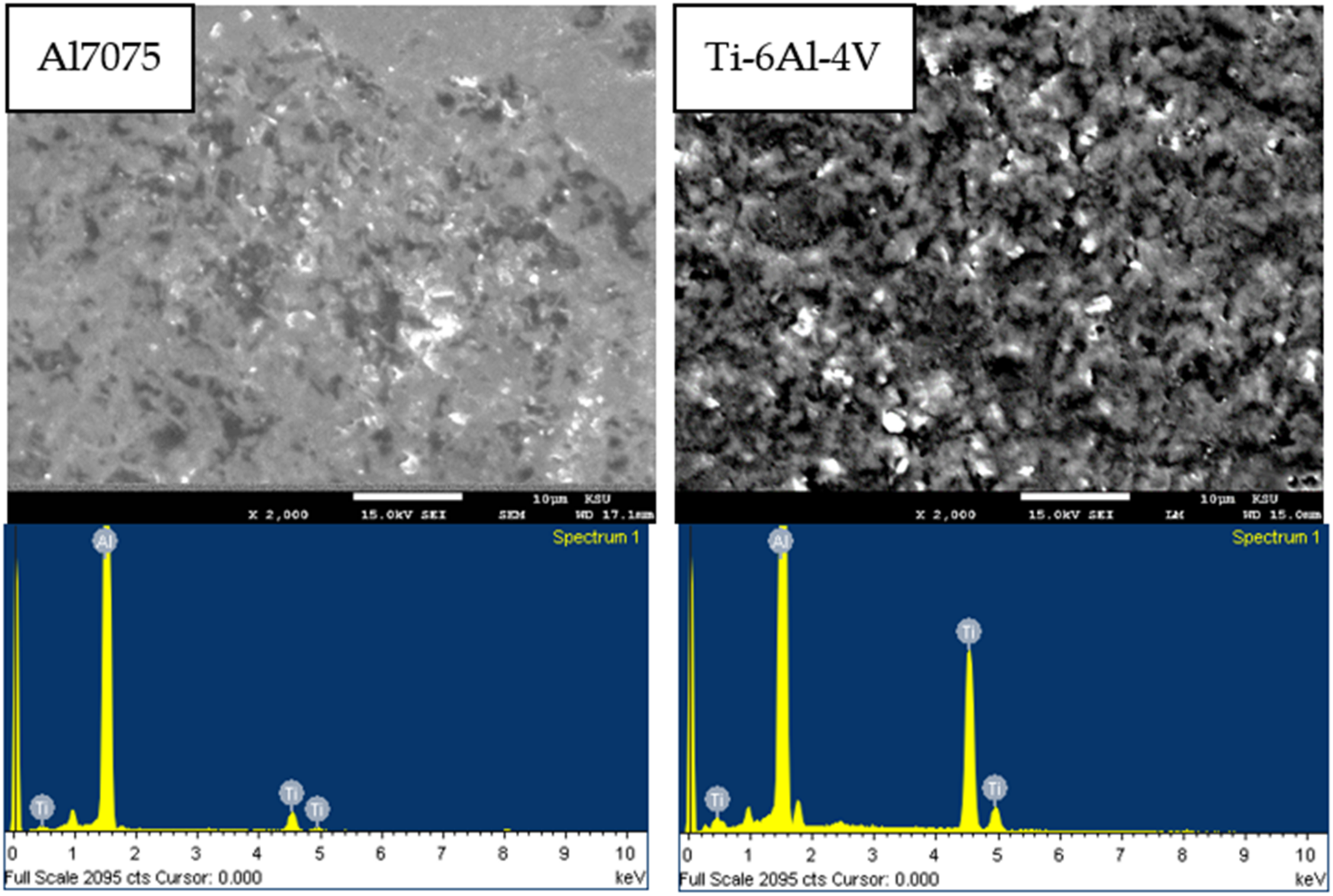
3.1. SEM and EPMA Analysis
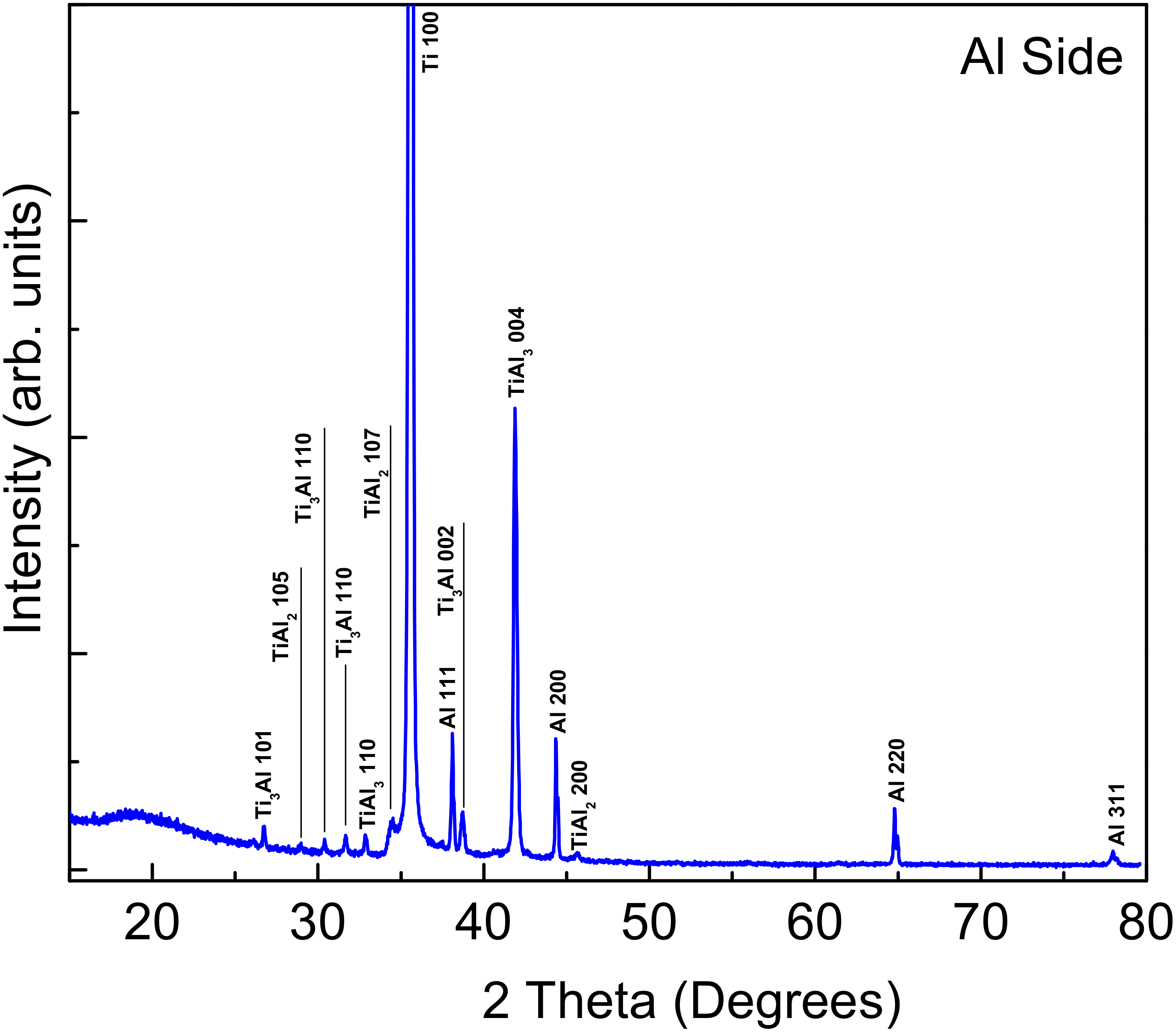
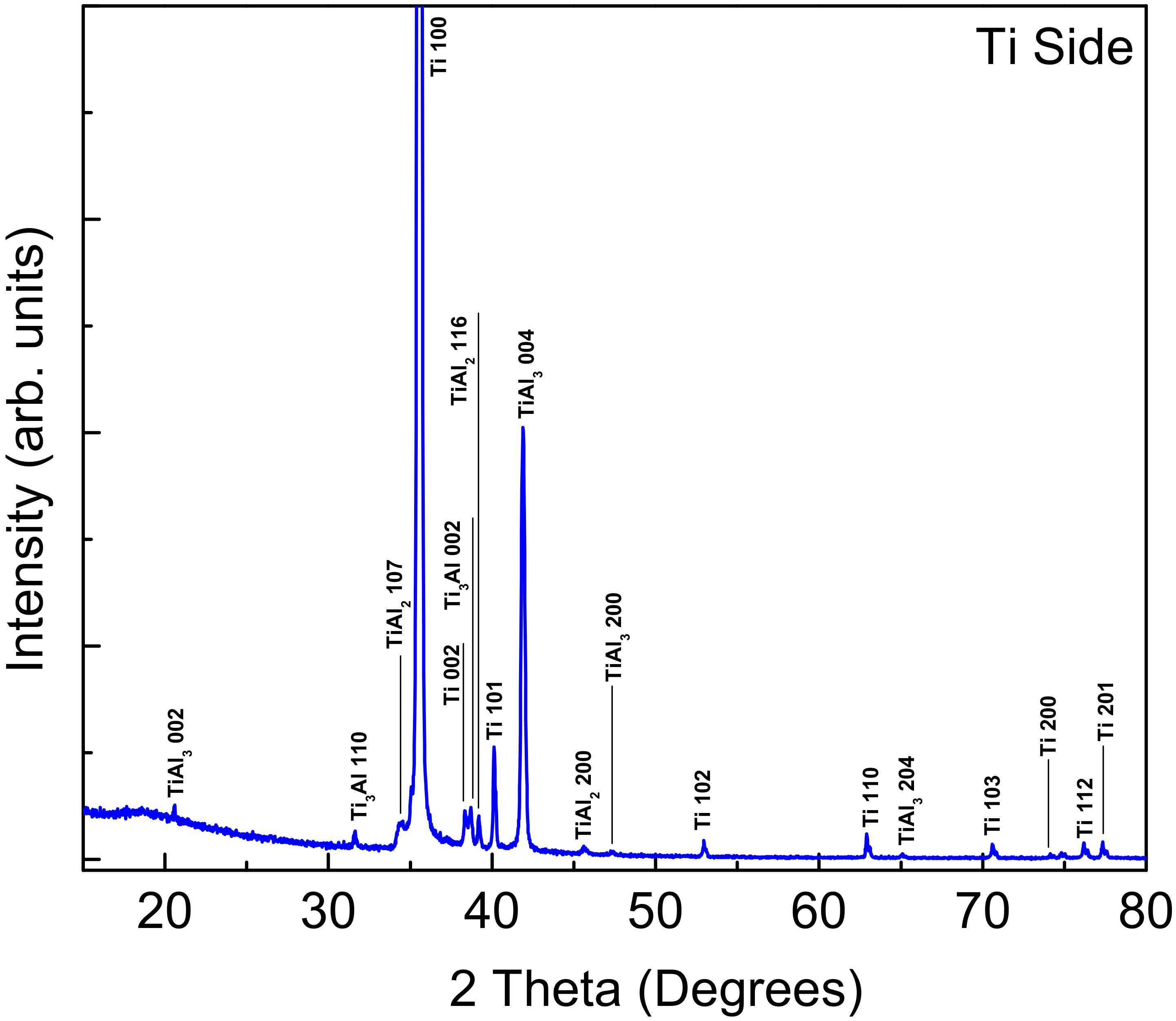
3.2. X-ray Diffraction and EDS of the Fractured Surfaces
3.3. Bond Strength Evaluation
3.4. Effect of Cu on the Joint Formation
3.5. Effect of IMC Morphology and SPS Processing Parameters on the Bond Strength
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prescott, R.; Graham, M.J. The formation of aluminum oxide scales on high-temperature alloys. Oxid. Met. 1992, 38, 233–254. [Google Scholar] [CrossRef]
- AlHazaa, A.; Khan, T.I.; Haq, I. Transient liquid phase (TLP) bonding of Al7075 to Ti–6Al–4V alloy. Mater. Charact. 2010, 61, 312–317. [Google Scholar] [CrossRef]
- Alhazaa, A.N.; Khan, T.I. Diffusion bonding of Al7075 to Ti–6Al–4V using Cu coatings and Sn–3.6 Ag–1Cu interlayers. J. Alloys Compd. 2010, 494, 351–358. [Google Scholar] [CrossRef]
- Omori, M. Sintering, consolidation, reaction and crystal growth by the spark plasma system (SPS). Mater. Sci. Eng. A 2000, 287, 183–188. [Google Scholar] [CrossRef]
- Cavaliere, P. Spark Plasma Sintering of Materials: Advances in Processing and Applications; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Tokita, M. Development of large-size ceramic/metal bulk FGM fabricated by spark plasma sintering. In Proceedings of the Materials Science Forum, Leuven, Belgium, 11–14 July 2004; Volume 308, pp. 83–88. [Google Scholar]
- Munir, Z.A.; Anselmi-Tamburini, U.; Ohyanagi, M. The effect of electric field and pressure on the synthesis and consolidation of materials: A review of the spark plasma sintering method. J. Mater. Sci. 2006, 41, 763–777. [Google Scholar] [CrossRef]
- Groza, J.R. Nanosintering. Nanostruct. Mater. 1999, 12, 987–992. [Google Scholar] [CrossRef]
- Manière, C.; Nigito, E.; Durand, L.; Weibel, A.; Beynet, Y.; Estournes, C. Spark plasma sintering and complex shapes: The deformed interfaces approach. Powder Technol. 2017, 320, 340–345. [Google Scholar] [CrossRef]
- Nisar, A.; Zhang, C.; Boesl, B.; Agarwal, A. Unconventional materials processing using spark plasma sintering. Ceramics 2021, 4, 20–39. [Google Scholar] [CrossRef]
- Tokita, M. Progress of Spark Plasma Sintering (SPS) Method, Systems, Ceramics Applications and Industrialization. Ceramics 2021, 4, 160–198. [Google Scholar] [CrossRef]
- Paygin, V.; Dvilis, E.; Alishin, T.; Stepanov, S.; Khasanov, O.; Valiev, D.; Ferrari, M. Application of collector pressing method to manufacture various optically transparent oxide ceramics using SPS technique. Opt. Mater. 2022, 128, 112332. [Google Scholar] [CrossRef]
- Armstrong, J.T. Citzaf-a package of correction programs for the quantitative Electron Microbeam X-Ray-Analysis of thick polished materials, thin-films, and particles. Microbeam Anal. 1995, 4, 177–200. [Google Scholar]
- Miriyev, A.; Levy, A.; Kalabukhov, S.; Frage, N. Interface evolution and shear strength of Al/Ti bi-metals processed by a spark plasma sintering (SPS) apparatus. J. Alloys Compd. 2016, 678, 329–336. [Google Scholar] [CrossRef]
- Askill, J. Tracer Diffusion Data for Metals, Alloys, and Simple Oxides; Springer Science & Business Media: Berlin, Germany, 1970; Volume 31, pp. 87–91. [Google Scholar]
- Wöhlbier, F. Diffusion and Defect Data; Trans Tech Publications: Wollerau, Switzerland, 1971; Volume 5, pp. 121–128. [Google Scholar]
- Kenevisi, M.S.; Khoie, M. A study on the effect of bonding time on the properties of Al7075 to Ti–6Al–4V diffusion bonded joint. Mater. Lett. 2012, 76, 144–146. [Google Scholar] [CrossRef]
- AlHazaa, A.; Alhoweml, I.; Shar, M.A.; Hezam, M.; Abdo, H.S.; AlBrithen, H. Transient liquid phase bonding of Ti-6Al-4V and Mg-AZ31 alloys using Zn coatings. Materials 2019, 12, 769. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Acoff, V.L.; Liu, C.; Silberschmidt, V.V.; Chen, Z. Facilitating intermetallic formation in wire bonding by applying a pre-ultrasonic energy. Microelectron. Eng. 2011, 88, 3155–3157. [Google Scholar] [CrossRef]
- Xu, H.; Liu, C.; Silberschmidt, V.V.; Pramana, S.S.; White, T.J.; Chen, Z.; Sivakumar, M.; Acoff, V.L. A micromechanism study of thermosonic gold wire bonding on aluminum pad. J. Appl. Phys. 2010, 108, 113517. [Google Scholar] [CrossRef]
- Zhang, D.; Qin, G.; Ma, H.; Geng, P. Non-uniformity of intermetallic compounds and properties in inertia friction welded joints of 2A14 Al alloy to 304 stainless steel. J. Manuf. Process. 2021, 68, 834–842. [Google Scholar] [CrossRef]
- Sequeira, C.A.C.; Amaral, L. Role of Kirkendall effect in diffusion processes in solids. Trans. Nonferrous Met. Soc. China 2014, 24, 1–11. [Google Scholar] [CrossRef]
- Wang, Q.; Leng, X.S.; Yang, T.H.; Yan, J.C. Effects of FeAl intermetallic compounds on interfacial bonding of clad materials. Trans. Nonferrous Met. Soc. China 2014, 24, 279–284. [Google Scholar] [CrossRef]



| Bonding Parameters | Strength (MPa) |
|---|---|
| 480 °C/10 MPa | 8.1 |
| 500 °C/10 MPa | 15.5 |
| 520 °C/10 MPa | 19.3 |
| 520 °C/5 MPa | 16.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhazaa, A.; Albrithen, H.; Hezam, M.; Ali Shar, M.; Alhwaimel, I.; Alharbi, Y.; Estournes, C. Diffusion Bonding of Al7075 to Ti-6Al-4V by Spark Plasma Sintering and Using a Copper Interlayer. Crystals 2022, 12, 1293. https://doi.org/10.3390/cryst12091293
Alhazaa A, Albrithen H, Hezam M, Ali Shar M, Alhwaimel I, Alharbi Y, Estournes C. Diffusion Bonding of Al7075 to Ti-6Al-4V by Spark Plasma Sintering and Using a Copper Interlayer. Crystals. 2022; 12(9):1293. https://doi.org/10.3390/cryst12091293
Chicago/Turabian StyleAlhazaa, Abdulaziz, Hamad Albrithen, Mahmoud Hezam, Muhammad Ali Shar, Ibrahim Alhwaimel, Yasser Alharbi, and Claude Estournes. 2022. "Diffusion Bonding of Al7075 to Ti-6Al-4V by Spark Plasma Sintering and Using a Copper Interlayer" Crystals 12, no. 9: 1293. https://doi.org/10.3390/cryst12091293
APA StyleAlhazaa, A., Albrithen, H., Hezam, M., Ali Shar, M., Alhwaimel, I., Alharbi, Y., & Estournes, C. (2022). Diffusion Bonding of Al7075 to Ti-6Al-4V by Spark Plasma Sintering and Using a Copper Interlayer. Crystals, 12(9), 1293. https://doi.org/10.3390/cryst12091293









