Obtaining Niobium Nitride on n-GaN by Surface Mediated Nitridation Technique
Abstract
1. Introduction
2. Experimental Procedures
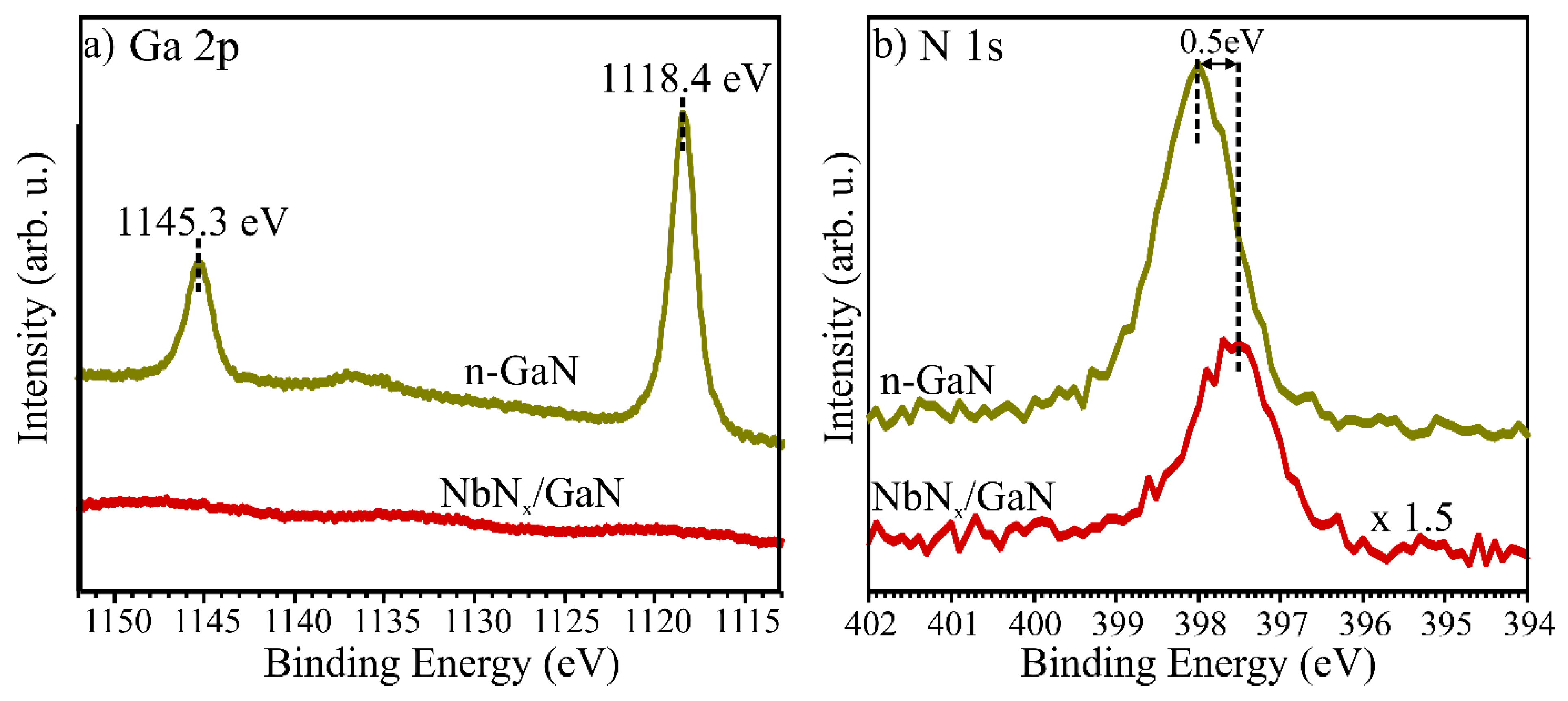
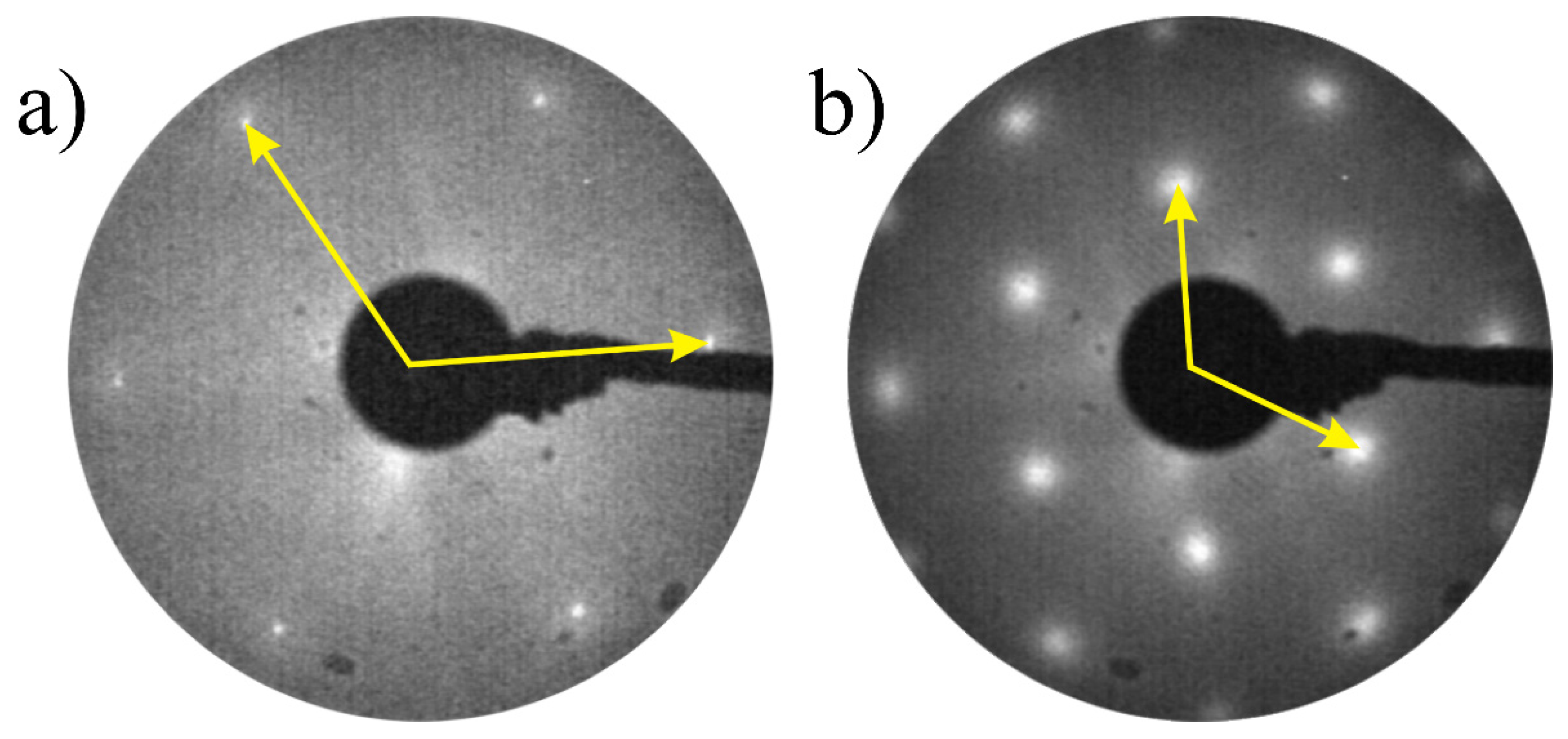
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zou, Y.; Qi, X.; Zhang, C.; Ma, S.; Zhang, W.; Li, Y.; Chen, T.; Wang, X.; Chen, Z.; Welch, D.; et al. Discovery of Superconductivity in Hard Hexagonal ε-NbN. Sci. Rep. 2016, 6, 22330. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Zhu, G.; Liu, X.; Liu, F.; Xie, Y.; Yang, C.; Lin, T.; Gu, H.; Huang, F. Niobium Nitride Nb 4 N 5 as a New High-Performance Electrode Material for Supercapacitors. Adv. Sci. 2015, 2, 1500126. [Google Scholar] [CrossRef] [PubMed]
- Jouve, G.; Séverac, C.; Cantacuzène, S. XPS Study of NbN and (NbTi)N Superconducting Coatings. Thin Solid Films 1996, 287, 146–153. [Google Scholar] [CrossRef]
- Idrees, M.; Mukhtar, A.; Ata-ur-Rehman; Abbas, S. M.; Zhang, Q.; Li, X. Transition Metal Nitride Electrodes as Future Energy Storage Devices: A Review. Mater. Today Commun. 2021, 27, 102363. [Google Scholar] [CrossRef]
- Cucciniello, N.; Lee, D.; Feng, H.Y.; Yang, Z.; Zeng, H.; Patibandla, N.; Zhu, M.; Jia, Q. Superconducting Niobium Nitride: A Perspective from Processing, Microstructure, and Superconducting Property for Single Photon Detectors. J. Phys. Condens. Matter. 2022, 34, 374003. [Google Scholar] [CrossRef]
- Roch, T.; Gregor, M.; Volkov, S.; Čaplovičová, M.; Satrapinskyy, L.; Plecenik, A. Substrate Dependent Epitaxy of Superconducting Niobium Nitride Thin Films Grown by Pulsed Laser Deposition. Appl. Surf. Sci. 2021, 551, 149333. [Google Scholar] [CrossRef]
- Wen, M.; Hu, C.Q.; Meng, Q.N.; Zhao, Z.D.; An, T.; Su, Y.D.; Yu, W.X.; Zheng, W.T. Effects of Nitrogen Flow Rate on the Preferred Orientation and Phase Transition for Niobium Nitride Films Grown by Direct Current Reactive Magnetron Sputtering. J. Phys. D Appl. Phys. 2009, 42, 035304. [Google Scholar] [CrossRef]
- Ziegler, M.; Fritzsch, L.; Day, J.; Linzen, S.; Anders, S.; Toussaint, J.; Meyer, H.-G. Superconducting Niobium Nitride Thin Films Deposited by Metal Organic Plasma-Enhanced Atomic Layer Deposition. Supercond. Sci. Technol. 2013, 26, 025008. [Google Scholar] [CrossRef]
- Hazra, D.; Tsavdaris, N.; Jebari, S.; Grimm, A.; Blanchet, F.; Mercier, F.; Blanquet, E.; Chapelier, C.; Hofheinz, M. Superconducting Properties of Very High Quality NbN Thin Films Grown by High Temperature Chemical Vapor Deposition. Supercond. Sci. Technol. 2016, 29, 105011. [Google Scholar] [CrossRef]
- Kafizas, A.; Carmalt, C.J.; Parkin, I.P. CVD and Precursor Chemistry of Transition Metal Nitrides. Coord. Chem. Rev. 2013, 257, 2073–2119. [Google Scholar] [CrossRef]
- Katzer, D.S.; Nepal, N.; Meyer, D.J.; Downey, B.P.; Wheeler, V.D.; Storm, D.F.; Hardy, M.T. Epitaxial Metallic β-Nb 2 N Films Grown by MBE on Hexagonal SiC Substrates. Appl. Phys. Express 2015, 8, 085501. [Google Scholar] [CrossRef]
- Wang, G.-Y.; Zhu, Z.; Yang, X.-Y.; Dong, L.; Ma, H.-Y.; Sun, H.-H.; Li, A.-M.; Guan, D.-D.; Qian, D.; Liu, C.; et al. Atomically Flat Superconducting NbN Thin Films Grown on SrTiO3 (111) by Plasma-Assisted MBE. APL Mater. 2017, 5, 126107. [Google Scholar] [CrossRef]
- Katzer, D.S.; Nepal, N.; Hardy, M.T.; Downey, B.P.; Storm, D.F.; Jin, E.N.; Yan, R.; Khalsa, G.; Wright, J.; Lang, A.C.; et al. Molecular Beam Epitaxy of Transition Metal Nitrides for Superconducting Device Applications. Phys. Status Solidi A 2020, 217, 1900675. [Google Scholar] [CrossRef]
- Wright, J.G.; Chang, C.S.; Muller, D.A.; Xing, H.G.; Jena, D. Structural and Electronic Properties of NbN/GaN Junctions Grown by Molecular Beam Epitaxy. APL Mater. 2022, 10, 051103. [Google Scholar] [CrossRef]
- Gajar, B.; Yadav, S.; Sawle, D.; Maurya, K.K.; Gupta, A.; Aloysius, R.P.; Sahoo, S. Substrate Mediated Nitridation of Niobium into Superconducting Nb2N Thin Films for Phase Slip Study. Sci. Rep. 2019, 9, 8811. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, C.; Wang, K. Progress of GaN-Based Optoelectronic Devices Integrated with Optical Resonances. Small 2022, 18, 2106757. [Google Scholar] [CrossRef]
- Coffie, R.L. High Power High Frequency Transistors: A Material’s Perspective. In High-Frequency GaN Electronic Devices; Fay, P., Jena, D., Maki, P., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 5–41. ISBN 978-3-030-20207-1. [Google Scholar]
- Smith, A.R. Reconstructions of GaN(0001) and (0001) Surfaces: Ga-Rich Metallic Structures. J. Vac. Sci. Technol. B 1998, 16, 2242. [Google Scholar] [CrossRef]
- Bermudez, V.M. The Fundamental Surface Science of Wurtzite Gallium Nitride. Surf. Sci. Rep. 2017, 72, 147–315. [Google Scholar] [CrossRef]
- Grodzicki, M. Properties of Bare and Thin-Film-Covered GaN(0001) Surfaces. Coatings 2021, 11, 145. [Google Scholar] [CrossRef]
- Grodzicki, M.; Mazur, P.; Zuber, S.; Pers, J.; Brona, J.; Ciszewski, A. Effect of Annealing on Ni/GaN(0001) Contact Morphology. Appl. Surf. Sci. 2014, 304, 24–28. [Google Scholar] [CrossRef]
- Grodzicki, M.; Mazur, P.; Pers, J.; Brona, J.; Zuber, S.; Ciszewski, A. Formation of GaPd2 and GaPd Intermetallic Compounds on GaN(0001). Appl. Phys. A 2015, 120, 1443–1451. [Google Scholar] [CrossRef]
- Grodzicki, M.; Mazur, P.; Brona, J.; Ciszewski, A. MnGa and (Mn,Ga)N-like Alloy Formation during Annealing of Mn/GaN(0001) Interface. Appl. Surf. Sci. 2019, 481, 790–794. [Google Scholar] [CrossRef]
- Lewandków, R.; Wasielewski, R.; Mazur, P. Hafnium and nitrogen interaction at Hf/GaN(0001) interface. Surf. Rev. Lett. 2020, 27, 2050013. [Google Scholar] [CrossRef]
- Yan, R.; Khalsa, G.; Vishwanath, S.; Han, Y.; Wright, J.; Rouvimov, S.; Katzer, D.S.; Nepal, N.; Downey, B.P.; Muller, D.A.; et al. GaN/NbN Epitaxial Semiconductor/Superconductor Heterostructures. Nature 2018, 555, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Horcas, I.; Fernández, R.; Gómez-Rodríguez, J.M.; Colchero, J.; Gómez-Herrero, J.; Baro, A.M. WSXM: A Software for Scanning Probe Microscopy and a Tool for Nanotechnology. Rev. Sci. Instrum. 2007, 78, 013705. [Google Scholar] [CrossRef]
- Buabthong, P.; Becerra Stasiewicz, N.; Mitrovic, S.; Lewis, N.S. Vanadium, Niobium and Tantalum by XPS. Surf. Sci. Spectra 2017, 24, 024001. [Google Scholar] [CrossRef]
- Ufuktepe, Y.; Farha, A.H.; Kimura, S.; Hajiri, T.; Karadağ, F.; Al Mamun, M.A.; Elmustafa, A.A.; Myneni, G.; Elsayed-Ali, H.E. Structural, Electronic, and Mechanical Properties of Niobium Nitride Prepared by Thermal Diffusion in Nitrogen. Mater. Chem. Phys. 2013, 141, 393–400. [Google Scholar] [CrossRef]
- Sanjinés, R.; Benkahoul, M.; Papagno, M.; Lévy, F.; Music, D. Electronic Structure of Nb2N and NbN Thin Films. J. Appl. Phys. 2006, 99, 044911. [Google Scholar] [CrossRef]
- Hadamek, T.; Posadas, A.B.; Dhamdhere, A.; Smith, D.J.; Demkov, A.A. Spectral Identification Scheme for Epitaxially Grown Single-Phase Niobium Dioxide. J. Appl. Phys. 2016, 119, 095308. [Google Scholar] [CrossRef]
- Lewandków, R.; Mazur, P.; Grodzicki, M. Niobium Oxides Films on GaN: Photoelectron Spectroscopy Study. Thin Solid Films 2022, 763, 139573. [Google Scholar] [CrossRef]
- Yadav, S.; Sahoo, S. Interface Study of Thermally Driven Chemical Kinetics Involved in Ti/Si3N4 Based Metal-Substrate Assembly by X-ray Photoelectron Spectroscopy. Appl. Surf. Sci. 2021, 541, 148465. [Google Scholar] [CrossRef]
- Fujii, R.; Gotoh, Y.; Liao, M.Y.; Tsuji, H.; Ishikawa, J. Work Function Measurement of Transition Metal Nitride and Carbide Thin Films. Vacuum 2006, 80, 832–835. [Google Scholar] [CrossRef]
- Gotoh, Y.; Tsuji, H.; Ishikawa, J. Measurement of Work Function of Transition Metal Nitride and Carbide Thin Films. J. Vac. Sci. Technol. B 2003, 21, 1607. [Google Scholar] [CrossRef]
- Sanjinés, R.; Benkahoul, M.; Sandu, C.S.; Schmid, P.E.; Lévy, F. Electronic States and Physical Properties of Hexagonal β-Nb2N and Δ′-NbN Nitrides. Thin Solid Films 2006, 494, 190–195. [Google Scholar] [CrossRef]
- Properties of Advanced Semiconductor Materials: GaN, AlN, InN, BN, SiC, SiGe; Levinshteĭn, M.E., Rumyantsev, S.L., Shur, M., Eds.; Wiley: New York, NY, USA, 2001; ISBN 978-0-471-35827-5. [Google Scholar]
- Terao, N. New Phases of Niobium Nitride. J. Less Common Met. 1971, 23, 159–169. [Google Scholar] [CrossRef]
- Kim, S.-J.; Franzen, H.F. A Powder X-ray Diffraction and Landau Theory Investigation of the Cubic to Tetragonal Phase Transition in NbN1 − x. J. Less Common Met. 1988, 143, 339–343. [Google Scholar] [CrossRef]
- Brauer, G. Nitrides, Carbonitrides and Oxynitrides of Niobium. J. Less Common Met. 1960, 2, 131–137. [Google Scholar] [CrossRef]
- Sam-Giao, D.; Pouget, S.; Bougerol, C.; Monroy, E.; Grimm, A.; Jebari, S.; Hofheinz, M.; Gérard, J.-M.; Zwiller, V. High-Quality NbN Nanofilms on a GaN/AlN Heterostructure. AIP Adv. 2014, 4, 107123. [Google Scholar] [CrossRef]
- Wright, J.; Chang, C.; Waters, D.; Lüpke, F.; Feenstra, R.; Raymond, L.; Koscica, R.; Khalsa, G.; Muller, D.; Xing, H.G.; et al. Unexplored MBE Growth Mode Reveals New Properties of Superconducting NbN. Phys. Rev. Mater. 2021, 5, 024802. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazur, P.; Sabik, A.; Lewandków, R.; Trembułowicz, A.; Grodzicki, M. Obtaining Niobium Nitride on n-GaN by Surface Mediated Nitridation Technique. Crystals 2022, 12, 1847. https://doi.org/10.3390/cryst12121847
Mazur P, Sabik A, Lewandków R, Trembułowicz A, Grodzicki M. Obtaining Niobium Nitride on n-GaN by Surface Mediated Nitridation Technique. Crystals. 2022; 12(12):1847. https://doi.org/10.3390/cryst12121847
Chicago/Turabian StyleMazur, Piotr, Agata Sabik, Rafał Lewandków, Artur Trembułowicz, and Miłosz Grodzicki. 2022. "Obtaining Niobium Nitride on n-GaN by Surface Mediated Nitridation Technique" Crystals 12, no. 12: 1847. https://doi.org/10.3390/cryst12121847
APA StyleMazur, P., Sabik, A., Lewandków, R., Trembułowicz, A., & Grodzicki, M. (2022). Obtaining Niobium Nitride on n-GaN by Surface Mediated Nitridation Technique. Crystals, 12(12), 1847. https://doi.org/10.3390/cryst12121847






