Relation between Electronic Structure and Thermoelectric Properties of Heusler-Type Ru2VAl Compounds
Abstract
1. Introduction
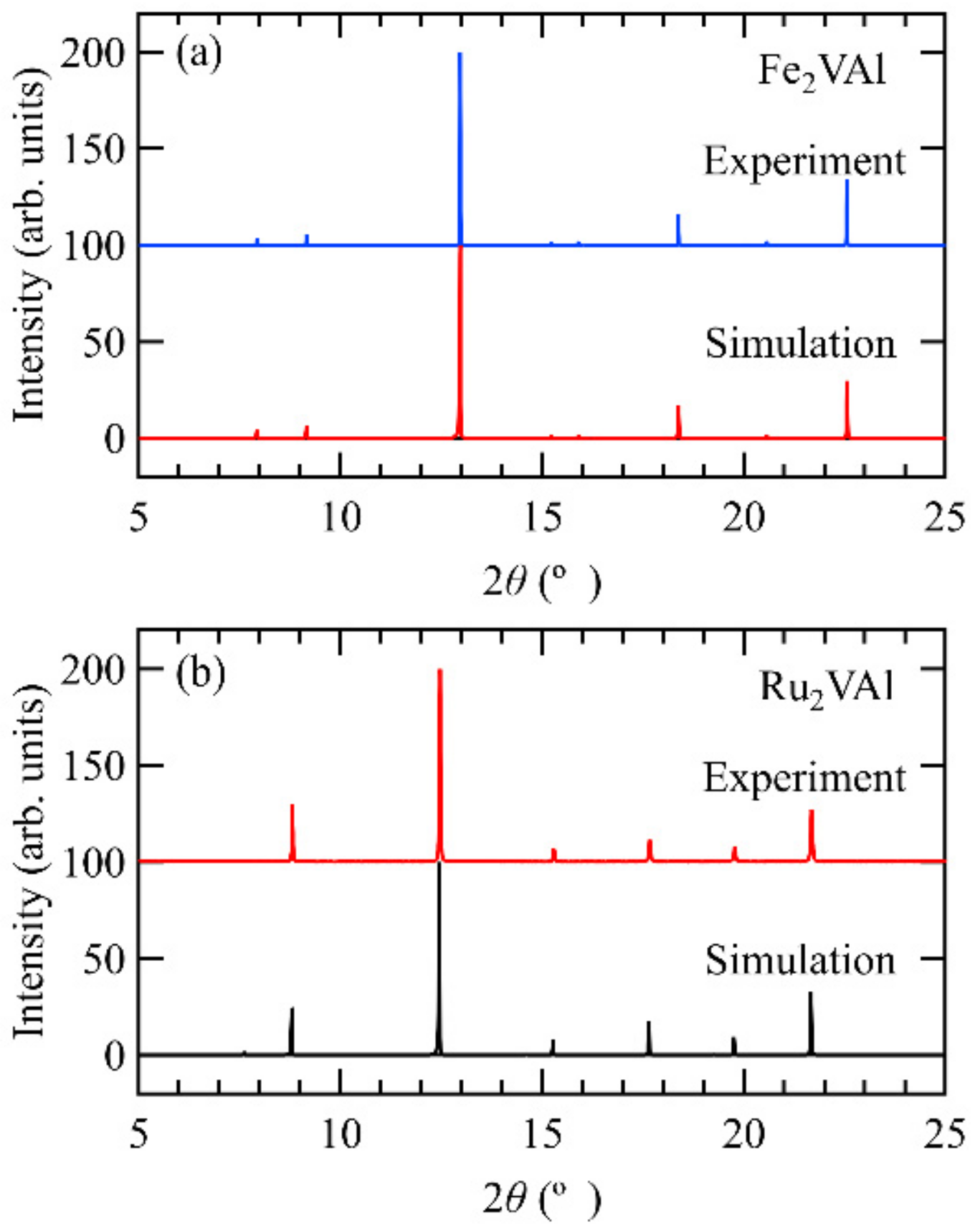
2. Theoretical and Experimental Methods
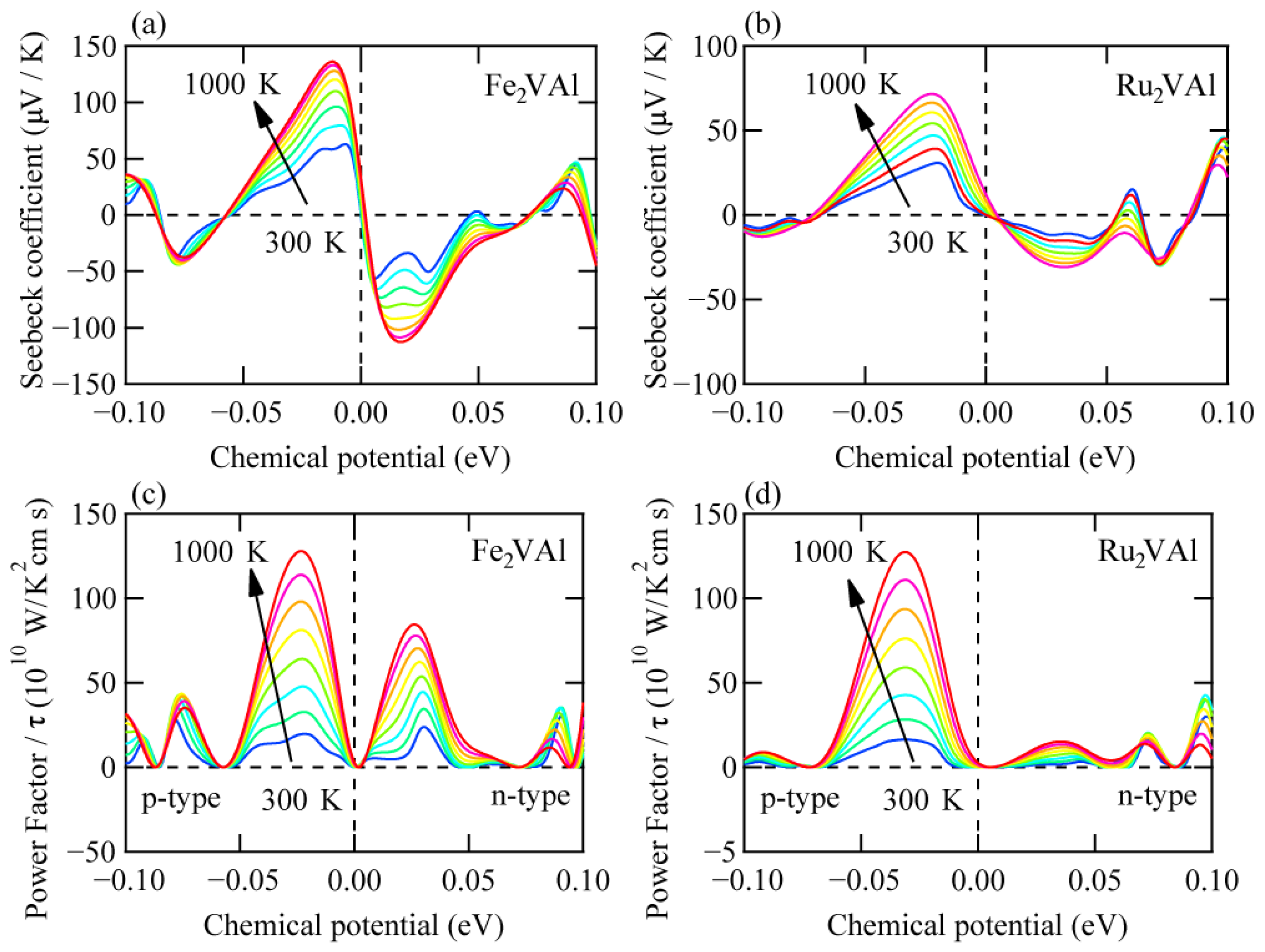
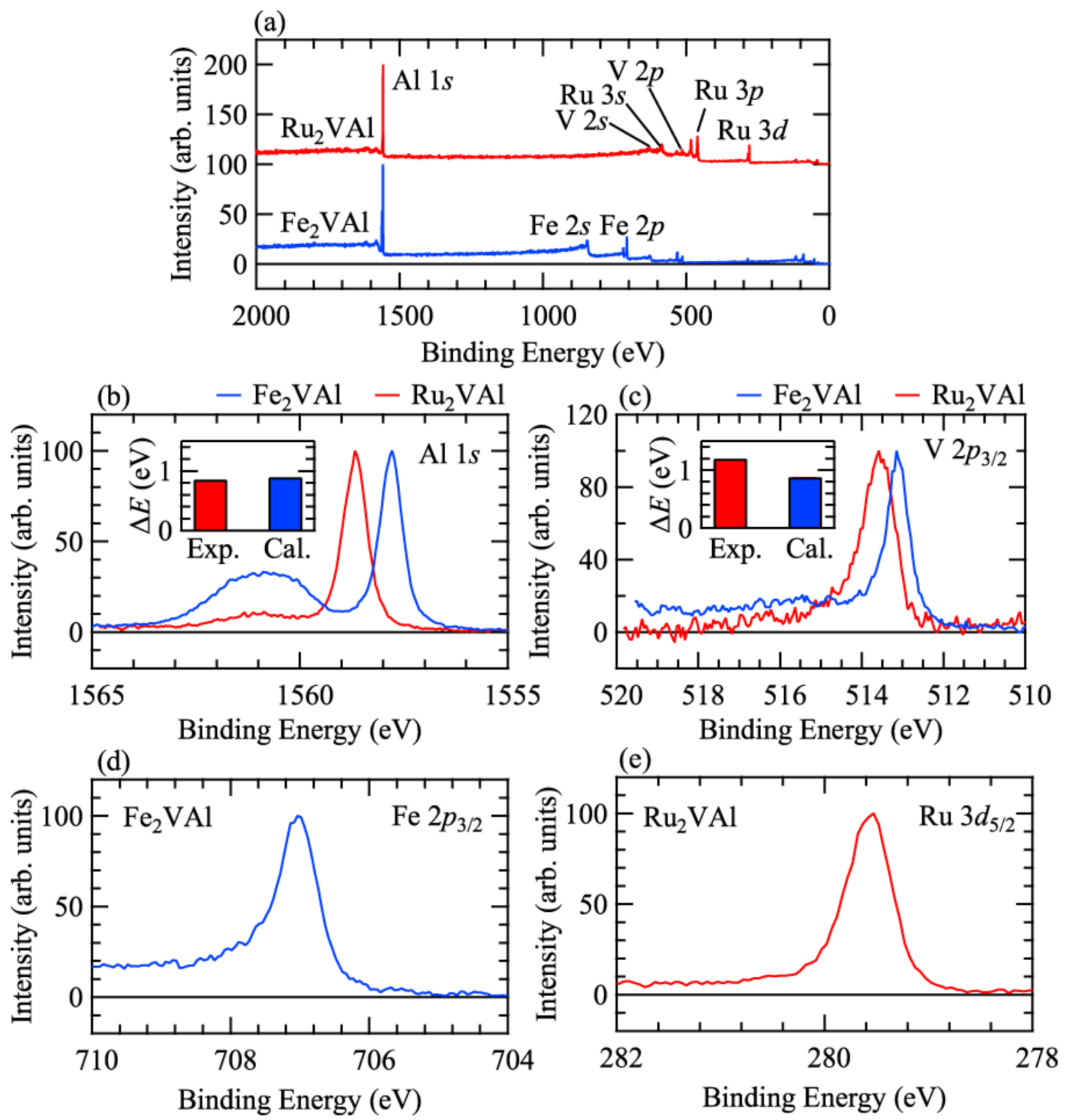
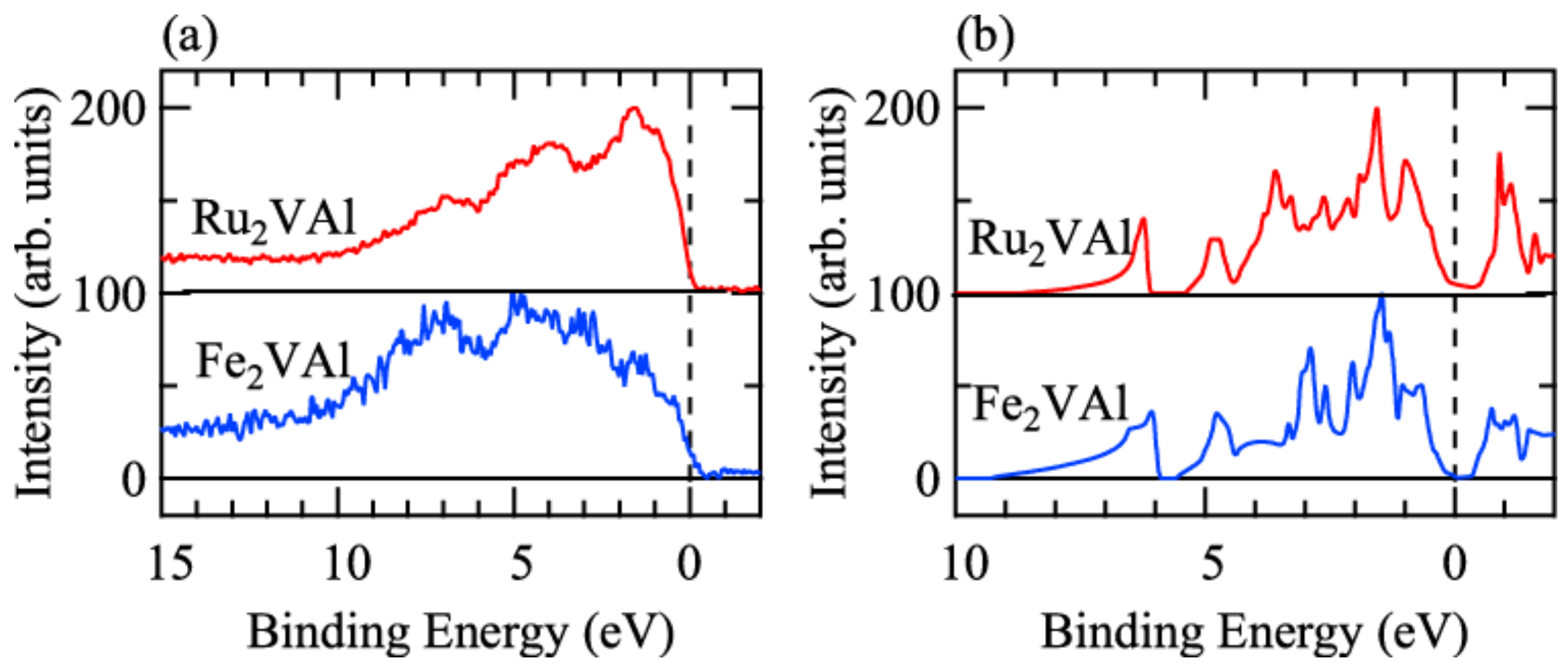
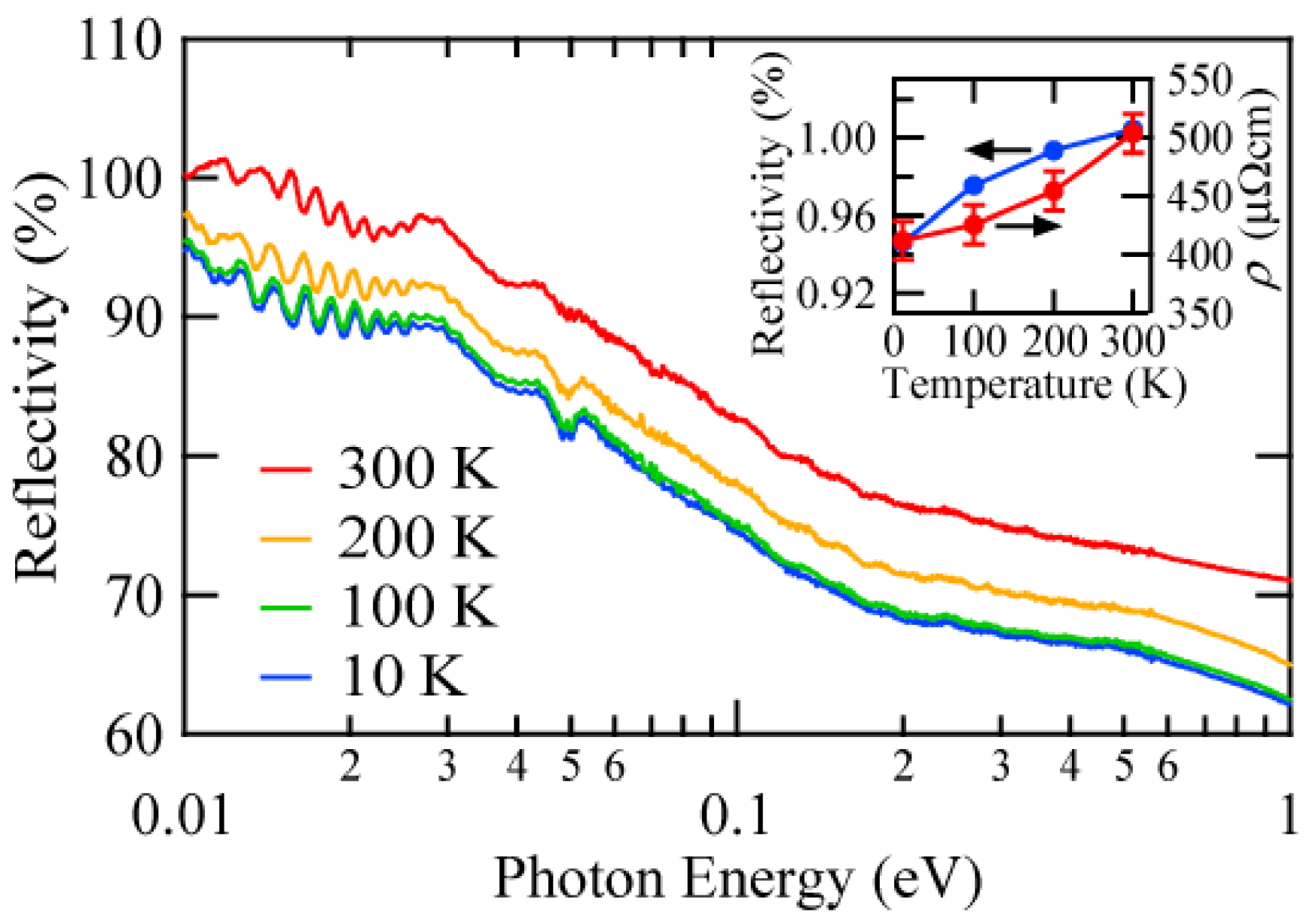
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amin Bhuiyan, M.R.; Mamur, H.; Dilmaç, Ö.F. A Review on Performance Evaluation of Bi2Te3-based and some other Thermoelectric Nanostructured Materials. Curr. Nanosci. 2021, 17, 423–446. [Google Scholar] [CrossRef]
- Chen, Y.; Hou, X.; Ma, C.; Dou, Y.; Wu, W. Review of Development Status of Bi2Te3-Based Semiconductor Thermoelectric Power Generation. Adv. Mater. Sci. Eng. 2018, 2018, 1210562. [Google Scholar] [CrossRef]
- Mamur, H.; Bhuiyan, M.R.A.; Korkmaz, F.; Nil, M. A review on bismuth telluride (Bi2Te3) nanostructure for thermoelectric applications. Renew. Sust. Energ. Rev. 2018, 82, 4159. [Google Scholar] [CrossRef]
- Su, C.-H. Design, growth and characterization of PbTe-based thermoelectric materials. Prog. Cryst. Growth Charact. Mater. 2019, 65, 47–94. [Google Scholar] [CrossRef]
- Nozariasbmarz, A.; Agarwal, A.; Coutant, Z.A.; Hall, M.J.; Liu, J.; Liu, R.; Malhotra, A.; Norouzzadeh, P.; Öztürk, M.C.; Ramesh, V.P.; et al. Thermoelectric silicides: A review. Jpn. J. Appl. Phys. 2017, 56, 05DA04. [Google Scholar] [CrossRef]
- Basu, R.; Singh, A. High temperature Si–Ge alloy towards thermoelectric applications: A comprehensive review. Mater. Today Phys. 2021, 21, 100468. [Google Scholar] [CrossRef]
- Li, Y.; Wang, G.; Akbari-Saatlu, M.; Procek, M.; Radamson, H.H. Si and SiGe Nanowire for Micro-Thermoelectric Generator: A Review of the Current State of the Art. Front. Mater. 2021, 8, 611078. [Google Scholar] [CrossRef]
- Chen, Z.; lin, M.Y.; Xu, G.D.; Chen, S.; Zhang, J.H.; Wang, M.M. Hydrothermal synthesized nanostructure Bi–Sb–Te thermoelectric materials. J. Alloys Compd. 2014, 588, 384–387. [Google Scholar] [CrossRef]
- Tan, M.; Deng, Y.; Hao, Y. Enhancement of thermoelectric properties induced by oriented nanolayer in Bi2Te2.7Se0.3 columnar films. Mater. Chem. Phys. 2014, 146, 153–158. [Google Scholar] [CrossRef]
- Tan, M.; Hao, Y.; Wang, G. Improvement of thermoelectric properties induced by uniquely ordered lattice field in Bi2Se0.5Te2.5 pillar array. J. Solid State Chem. 2014, 215, 219–224. [Google Scholar] [CrossRef]
- Ahmad, K.; Wan, C. Enhanced thermoelectric performance of Bi2Te3 through uniform dispersion of single wall carbon nanotubes. Nanotechnology 2017, 28, 415402. [Google Scholar] [CrossRef] [PubMed]
- Nishino, Y.; Kato, M.; Asano, S.; Soda, K.; Hayasaki, M.; Mizutani, U. Semiconductorlike Behavior of Electrical Resistivity in Heusler-type Fe2VAl Compound. Phys. Rev. Lett. 1997, 79, 1909. [Google Scholar] [CrossRef]
- Nishino, Y. The Science of Complex Alloy Phases; TMS: Warrendale, PA, USA, 2005; pp. 325–344. [Google Scholar]
- Mott, N.F.; Jones, H. The Theory of the Properties of Metals; Clarendon Press: Oxford, UK, 1936. [Google Scholar]
- Kato, H.; Kato, M.; Nishino, Y.; Mizutani, U.; Asano, S. Effect of Silicon Substitution on Thermoelectric Properties of Heusler-type Fe2VAl Alloy. J Jpn. Inst. Metal. 2001, 65, 652. [Google Scholar] [CrossRef]
- Matsuura, H.; Nishino, Y.; Mizutani, U.; Asano, S. Doping Effects on Thermoelectric Properties of the Pseudogap Fe2VAl System. J Jpn. Inst. Metal. 2002, 66, 767. [Google Scholar] [CrossRef]
- Lue, C.S.; Chen, C.F.; Lin, J.Y.; Yu, Y.T.; Kuo, Y.K. Thermoelectric properties of quaternary Heusler alloys Fe2VAl1−xSix. Phys. Rev. B 2007, 75, 064204. [Google Scholar] [CrossRef]
- Vasundhara, M.; Srinivas, V.; Rao, V.V. Electronic transport in Heusler-type Fe2VAl1−xMx alloys (M = B,In,Si). Phys. Rev. B 2008, 77, 224415. [Google Scholar] [CrossRef]
- Mikami, M.; Tanaka, S.; Kobayashi, K. Thermoelectric properties of Sb-doped Heusler Fe2VAl alloy. J. Alloys Compd. 2009, 484, 444. [Google Scholar] [CrossRef]
- Terazawa, Y.; Mikami, M.; Itoh, T.; Takeuchi, T. Effects of Heavy Element Substitution on Electronic Structure and Lattice Thermal Conductivity of Fe2VAl Thermoelectric Material. J. Electron. Mater. 2012, 41, 1348. [Google Scholar] [CrossRef]
- Miyazaki, H.; Renard, K.; Inukai, M.; Soda, K.; Nishino, Y. Electronic structure of Heusler-type Fe2V1+xAl1−x thermoelectric materials. J. Electron Spectros. Relat. Rhenomena 2014, 195, 185–188. [Google Scholar] [CrossRef]
- Nishino, Y.; Tamada, Y. Doping effects on thermoelectric properties of the off-stoichiometric Heusler compounds Fe2−xV1+xAl. J. Appl. Phys. 2014, 115, 123707. [Google Scholar] [CrossRef]
- Renard, K.; Mori, A.; Yamada, Y.; Tanaka, S.; Miyazaki, H.; Nishino, Y. Thermoelectric properties of the Heusler-type Fe2VTaxAl1−x alloys. J. Appl. Phys. 2014, 115, 033707. [Google Scholar] [CrossRef]
- Nishino, Y.; Kamizono, S.; Miyazaki, H.; Kimura, K. Effects of off-stoichiometry and Ti doping on thermoelectric performance of Fe2VAl Heusler compound. AIP Adv. 2019, 9, 125003. [Google Scholar] [CrossRef]
- Miyazaki, H.; Tanaka, S.; Ide, N.; Soda, K.; Nishino, Y. Thermoelectric properties of Heusler-type off-stoichiometric Fe2V1+xAl1−x alloys. Mater. Res. Express 2014, 1, 015901. [Google Scholar] [CrossRef]
- Guezmir, A.; Rached, H.; Bentouaf, A.; Caid, M.; Benkhettou, N.; Rached, D.; Sidoumou, M. Theoretical insight of stabilities and optoelectronic features of Ru-based Heusler alloys: Ab-initio calculations. Comput. Condens. Matter. 2021, 28, e00573. [Google Scholar] [CrossRef]
- Abbassa, H.; Hadjri-Mebarki, S.; Amrani, B.; Belaroussi, T.; Driss Khodja, K.; Aubert, P. Theoretical investigation of new Heusler alloys Ru2VGa1−xAlx. J. Alloys Compd. 2015, 637, 557–563. [Google Scholar] [CrossRef]
- Ramachandran, B.; Lin, Y.H.; Kuo, Y.K.; Kuo, C.N.; Gippius, A.A.; Lue, C.S. Thermoelectric properties of Heusler-type Ru2VAl1−xGax alloys. Intermetallics 2018, 92, 36–41. [Google Scholar] [CrossRef]
- Soda, K.; Mizutani, T.; Yoshimoto, O.; Yagi, S.; Mizutani, U.; Sumi, H.; Nishino, Y.; Yamada, Y.; Yokoya, T.; Shin, A.; et al. High-resolution photoelectron spectroscopyof Heusler-type Fe2VAl alloy. J. Synchrotron Radiat. 2002, 9, 233. [Google Scholar] [CrossRef]
- Miyazaki, H.; Soda, K.; Yagi, S.; Kato, M.; Takeuchi, T.; Mizutani, U.; Nishino, Y. Surface and bulk electronic structures of Heusler-type Fe2VAl. J. Vac. Sci. Technol. A 2006, 24, 1464. [Google Scholar] [CrossRef]
- Miyazaki, H.; Soda, K.; Kato, M.; Yagi, S.; Takeuchi, T.; Nishino, Y. Soft X-ray photoemission study of the Heusler-type Fe2VAl1−zGez alloys. J. Electron Spectros. Relat. Rhenomena 2007, 156-158, 347. [Google Scholar] [CrossRef]
- Soda, K.; Harada, S.; Kato, M.; Yagi, S.; Inukai, M.; Miyazaki, H.; Sandaiji, Y.; Tamada, Y.; Tanaka, S.; Sugiura, T.; et al. Soft X-ray photoemission study of thermoelectric alloys Fe2−x−yIryV1+xAl and Fe2−xV1+x−yTiyAl. J. Electron Spectros. Relat. Rhenomena 2011, 184, 236. [Google Scholar] [CrossRef]
- Miyazaki, H.; Inukai, M.; Nishino, Y. Effect of Ta substitution on the electronic structure of Heusler-type Fe2VAl-based alloy. J. Appl. Phys. 2016, 120, 125106. [Google Scholar] [CrossRef]
- Miyazaki, H.; Tateishi, S.; Matsunami, M.; Soda, K.; Yamada, S.; Hamaya, K.; Nishino, Y. Direct observation of pseudo-gap electronic structure in the Heusler-type Fe2VAl thin film. J. Electron Spectros. Relat. Rhenomena 2019, 232, 1–4. [Google Scholar] [CrossRef]
- Asai, M.; Miyazaki, H.; Watanabe, K.; Yasui, A.; Takagi, Y.; Nishino, Y. Hard X-Ray Photoemission Study of Heusler-Type Fe2−xRexVAl Thermoelectric Compounds. Phys. Stat. Sol. (b) 2022, 259, 2100567. [Google Scholar] [CrossRef]
- Soda, K.; Ikedo, W.; Hayashi, T.; Shirakawa, T.; Miyazaki, H.; Nishino, Y. Hard X-Ray Photoemission Study on Bulk Electronic Structure of Heusler-Type Fe2−xV1+xAl Alloys. J. Phys. Soc. Jpn. 2022, 91, 064713. [Google Scholar] [CrossRef]
- Okamura, H.; Kawahara, J.; Nanba, T.; Kimura, S.; Soda, K.; Mizutani, U.; Nishino, Y.; Kato, M.; Shimoyama, I.; Miura, H.; et al. Pseudogap Formation in the Intermetallic Compounds (Fe1-xVx)3Al. Phys. Rev. Lett. 2000, 84, 3674. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Rhee, J.Y.; Wiener, T.A.; Lynch, D.W.; Hubbard, B.E.; Sievers, A.J.; Schlagel, D.L.; Lograsso, T.A.; Miller, L.L. Physical properties of Heusler-like Fe2VAl. Phys. Rev. B 2001, 63, 165109. [Google Scholar] [CrossRef]
- Blaha, P.; Schwarz, K.; Tran, F.; Laskowski, R.; Madsen, G.K.H.; Marks, L.D. WIEN2k: An APW+lo program for calculating the properties of solids. J. Chem. Phys. 2020, 152, 074101. [Google Scholar] [CrossRef]
- Madsen, G.K.H.; Singh, D.J. BoltzTraP. A code for calculating band-structure dependent quantities. Comput. Phys. Commun. 2006, 175, 67–71. [Google Scholar] [CrossRef]
- Kawaguchi, S.; Takemoto, M.; Osaka, K.; Nishibori, E.; Moriyoshi, C.; Kubota, Y.; Kuroiwa, Y.; Sugimoto, K. High-throughput powder diffraction measurement system consisting of multiple MYTHEN detectors at beamline BL02B2 of SPring-8. Rev. Sci. Instrum. 2017, 88, 085111. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Xu, B.; Li, X.; Yu, G.; Zhang, J.; Ma, S.; Wang, Y.; Yi, L. The structural, elastic and thermoelectric properties of Fe2VAl at pressures. J. Alloys Compd. 2013, 565, 22. [Google Scholar] [CrossRef]
- Khalfa, M.; Khachai, H.; Chiker, F.; Baki, N.; Bougherara, K.; Yakoubi, A.; Murtaza, G.; Harmel, M.; Abu-Jafar, M.S.; Omran, S.B.; et al. Mechanical, electronic and thermodynamic properties of full Heusler compounds Fe2VX(X = Al, Ga). International J. Mod. Phys. B 2015, 29, 1550229. [Google Scholar] [CrossRef]
- Weiland, C.; Lysaght, P.; Price, J.; Huang, J.; Woicik, J.C. Hard x-ray photoelectron spectroscopy study of As and Ga out-diffusion in In0.53Ga0.47As/Al2O3 film systems. Appl. Phys. Lett. 2012, 101, 061602. [Google Scholar] [CrossRef]
- Song, T.T.; Yang, M.; Chai, J.W.; Callsen, M.; Zhou, J.; Yang, T.; Zhang, Z.; Pan, J.S.; Chi, D.Z.; Feng, Y.P.; et al. The stability of aluminium oxide monolayer and its interface with two-dimensional materials. Sci. Rep. 2016, 6, 29221. [Google Scholar] [CrossRef]
- Yeh, J.J.; Lindau, I. Atomic subshell photoionization cross sections and asymmetry parameters: 1 ≤ Z ≤ 103. At. Data Nucl. Data Tables 1985, 32, 1. [Google Scholar] [CrossRef]
- Kimura, S.-I.; Im, H.; Mizuno, T.; Narazu, S.; Matsuoka, E.; Takabatake, T. Infrared study on the electronic structure of the alkaline-earth-filled skutterudites AM4Sb12 (A = Sr, Ba; M = Fe, Ru, Os). Phys. Rev. B 2007, 75, 245106. [Google Scholar] [CrossRef]

| Fe2VAl | Ru2VAl | |
|---|---|---|
| Lattice parameter (nm) | 0.5709 | 0.6011 |
| Experimental Lattice parameter (nm) | 0.5761 | 0.5994 |
| Young module (GPa) | 223.05 | 256.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miyazaki, H.; Kimura, S.-i.; Onishi, K.; Hihara, T.; Yoshimura, M.; Ishii, H.; Mikami, M.; Nishino, Y. Relation between Electronic Structure and Thermoelectric Properties of Heusler-Type Ru2VAl Compounds. Crystals 2022, 12, 1403. https://doi.org/10.3390/cryst12101403
Miyazaki H, Kimura S-i, Onishi K, Hihara T, Yoshimura M, Ishii H, Mikami M, Nishino Y. Relation between Electronic Structure and Thermoelectric Properties of Heusler-Type Ru2VAl Compounds. Crystals. 2022; 12(10):1403. https://doi.org/10.3390/cryst12101403
Chicago/Turabian StyleMiyazaki, Hidetoshi, Shin-ichi Kimura, Kensuke Onishi, Takehiko Hihara, Masato Yoshimura, Hirofumi Ishii, Masashi Mikami, and Yoichi Nishino. 2022. "Relation between Electronic Structure and Thermoelectric Properties of Heusler-Type Ru2VAl Compounds" Crystals 12, no. 10: 1403. https://doi.org/10.3390/cryst12101403
APA StyleMiyazaki, H., Kimura, S.-i., Onishi, K., Hihara, T., Yoshimura, M., Ishii, H., Mikami, M., & Nishino, Y. (2022). Relation between Electronic Structure and Thermoelectric Properties of Heusler-Type Ru2VAl Compounds. Crystals, 12(10), 1403. https://doi.org/10.3390/cryst12101403







