Development of Binder Free Interconnected 3D Flower of NiZn2O4 as an Advanced Electrode Materials for Supercapacitor Applications
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
2.3. Electrochemical Measurements
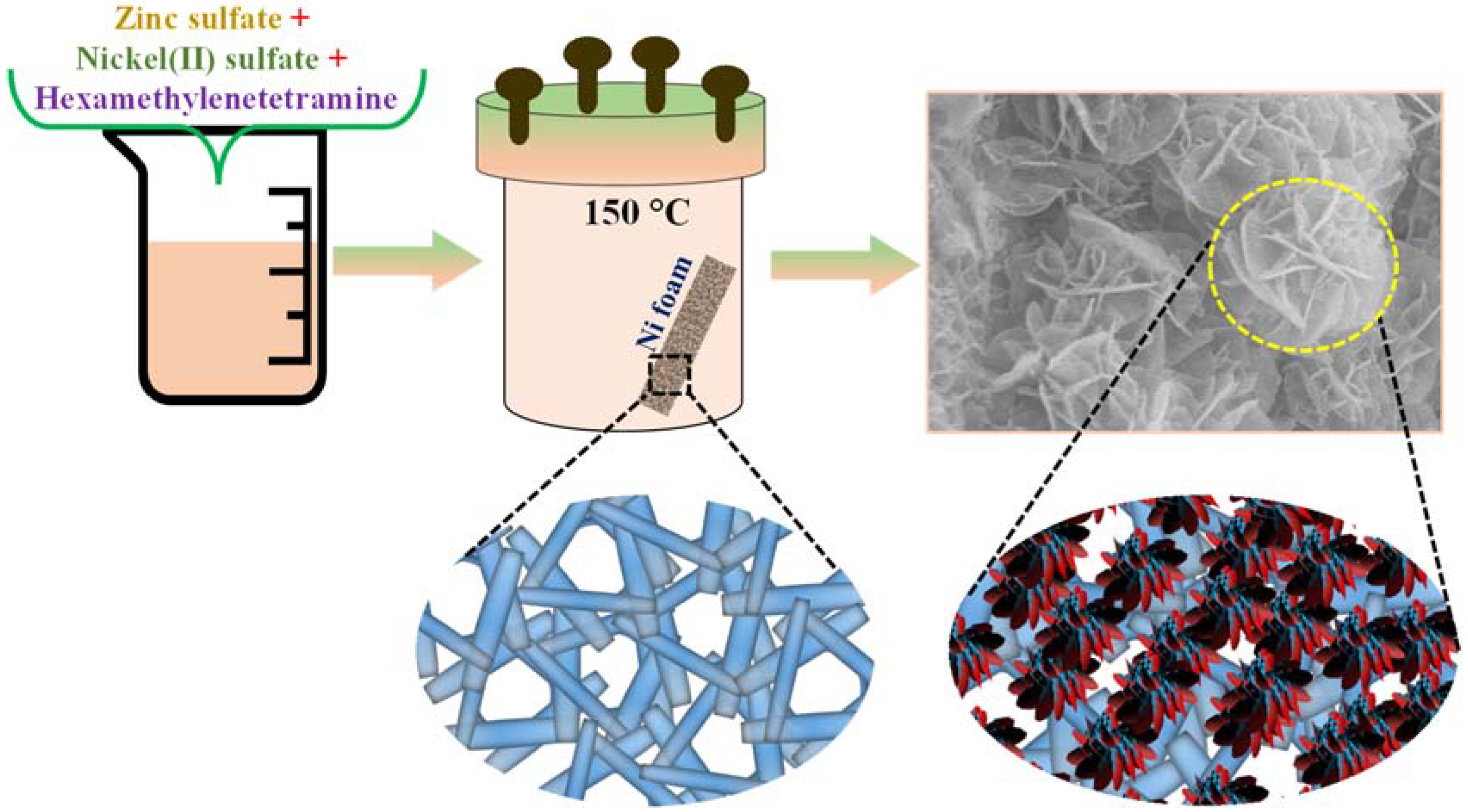
2.4. Synthesis of Binder-Free Bimetallic NiZn2O4@3D-NF Active Material
3. Results and Discussion
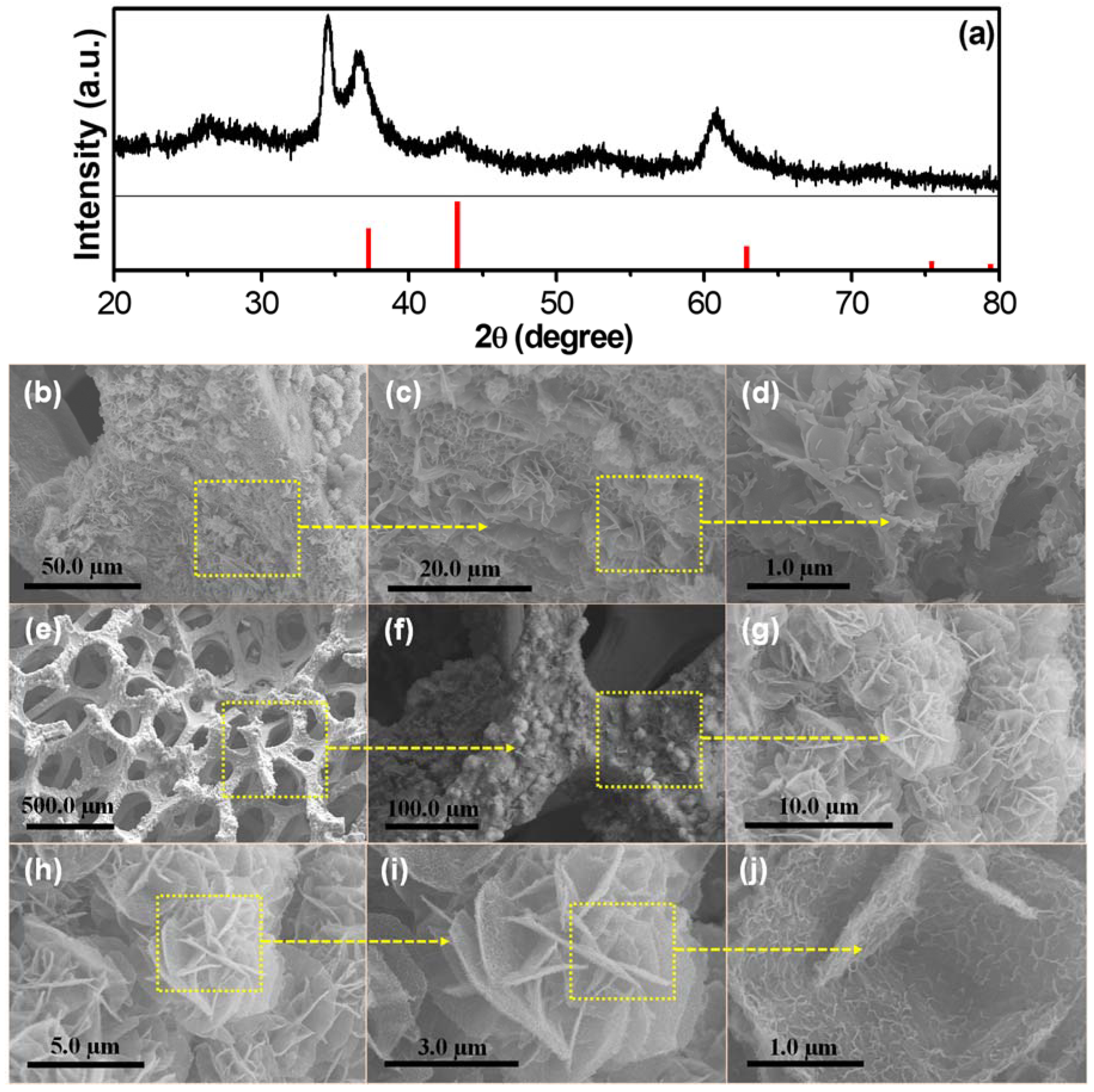
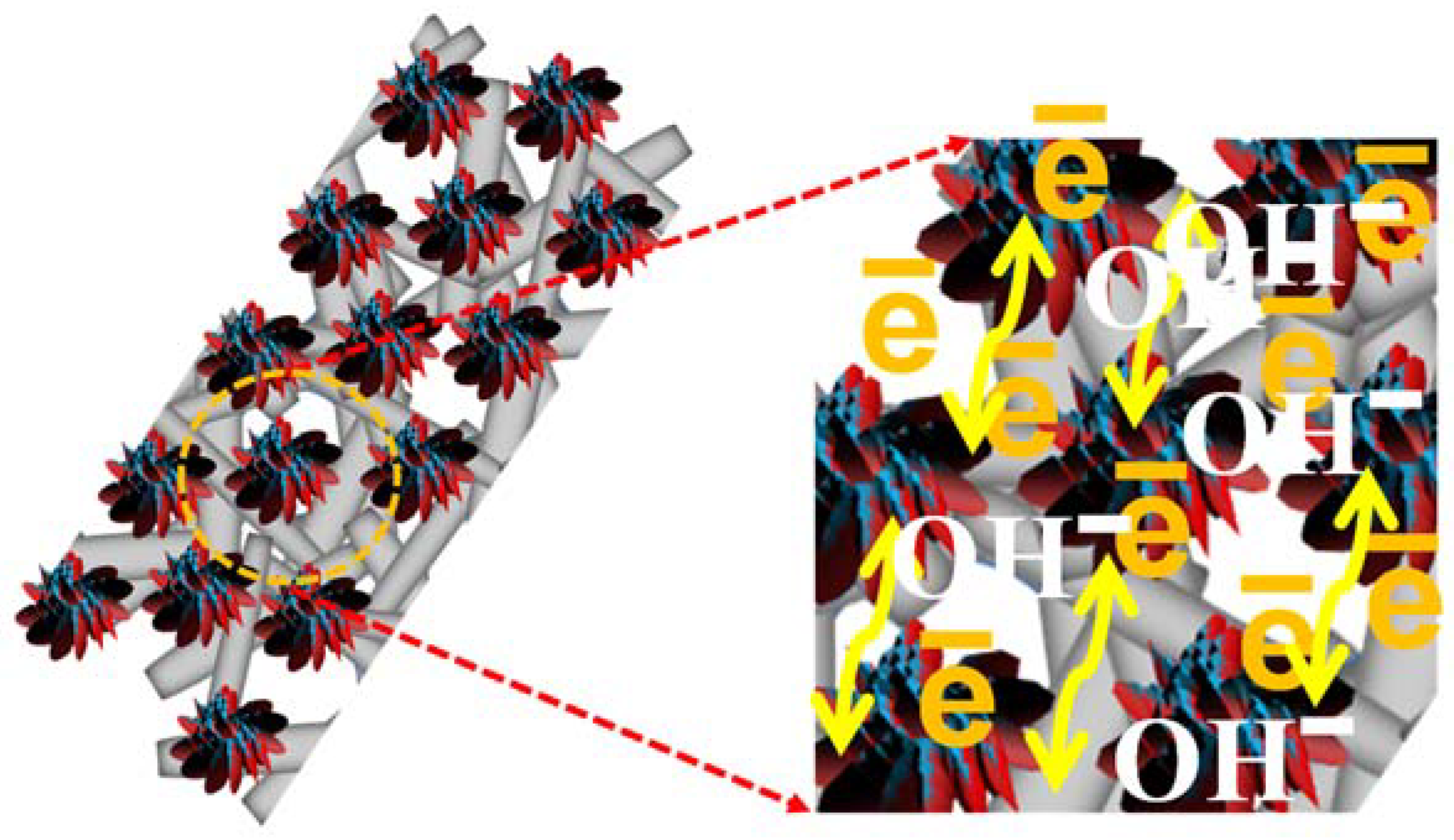
3.1. XRD and FESEM Analysis
3.2. TEM and HRTEM Analysis
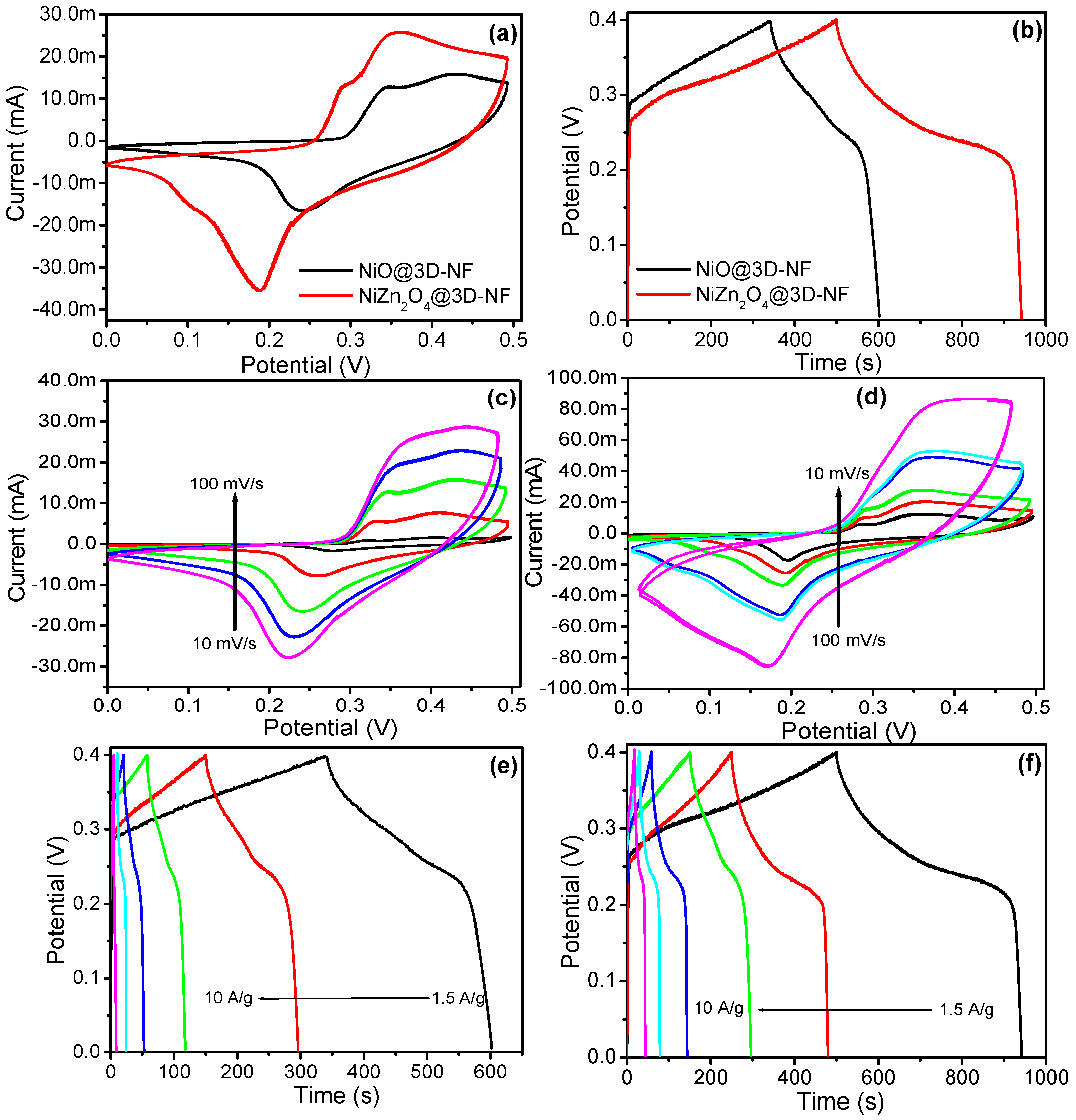
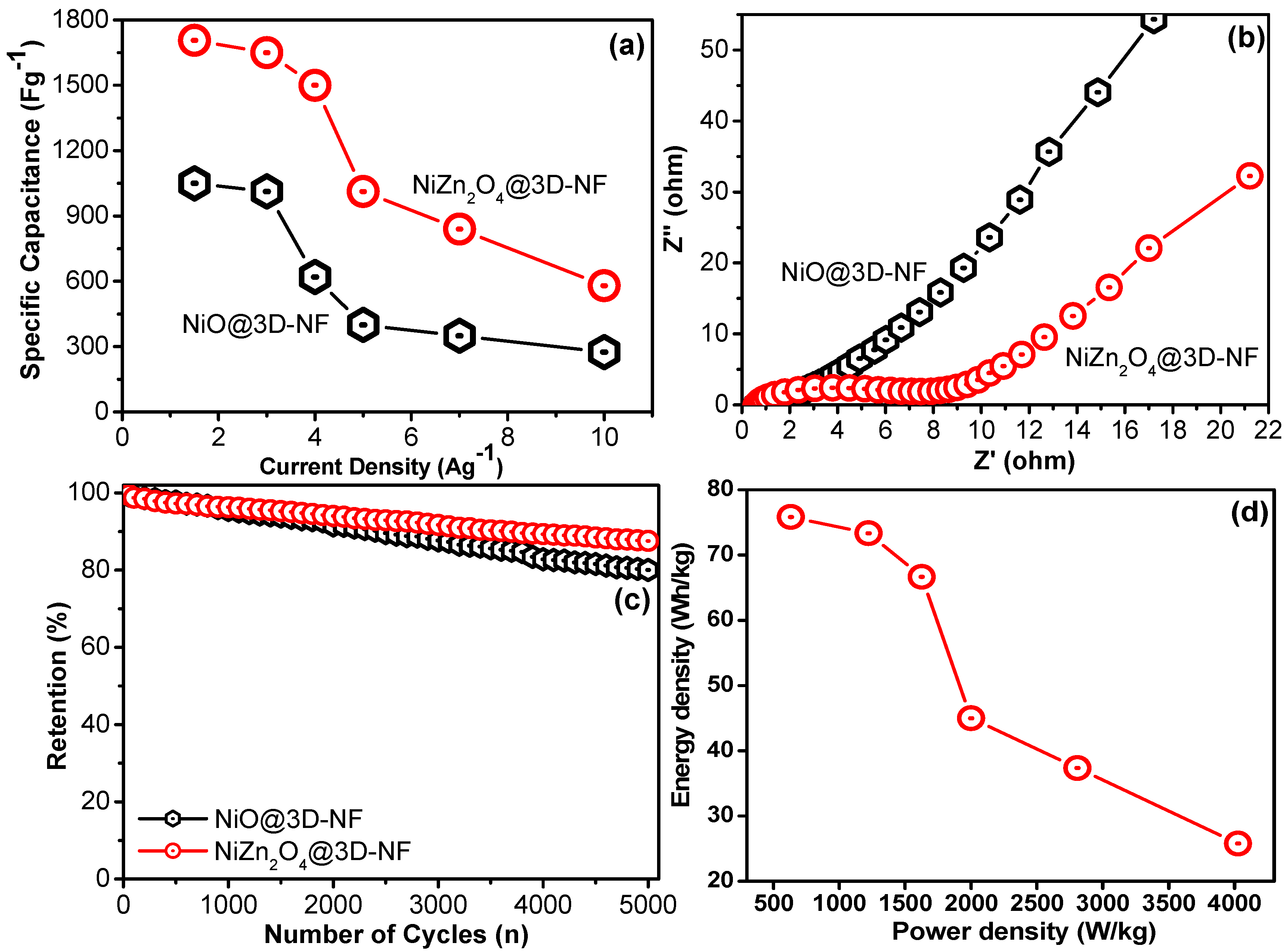
3.3. Electrochemical Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Javed, M.S.; Shaheen, N.; Hussain, S.; Li, J.; Shah, S.S.A.; Abbas, Y.; Ahmad, M.A.; Raza, R.; Mai, W. An ultra-high energy density flexible asymmetric supercapacitor based on hierarchical fabric decorated with 2D bimetallic oxide nanosheets and MOF-derived porous carbon polyhedral. J. Mater. Chem. A 2019, 7, 946–957. [Google Scholar] [CrossRef]
- Parveen, N.; Ansari, S.A.; Ansari, M.Z.; Ansari, M.O. Manganese oxide as an effective electrode material for energy storage: A review. Environ. Chem. Lett. 2021, 1–27. [Google Scholar] [CrossRef]
- Ansari, S.A.; Goumri-Said, S.; Yadav, H.M.; Belarbi, M.; Aljaafari, A.; Kanoun, M.B. Directly grown of NiCo2S4 nanoparticles on a conducting substrate towards the high-performance counter electrode in dye-sensitized solar cell: A combined theoretical and experimental study. Sol. Energy Mater. Sol. Cells 2021, 225, 111064. [Google Scholar] [CrossRef]
- Ansari, M.Z.; Parveen, N.; Nandi, D.K.; Ramesh, R.; Ansari, S.A.; Cheon, T.; Kim, S.H. Enhanced activity of highly conformal and layered tin sulfide (SnSx) prepared by atomic layer deposition (ALD) on 3D metal scaffold towards high performance supercapacitor electrode. Sci. Rep. 2019, 9, 10225. [Google Scholar] [CrossRef]
- Ashraf, M.; Shah, S.S.; Khan, I.; Aziz, M.A.; Ullah, N.; Khan, M.; Adil, S.F.; Liaqat, Z.; Usman, M.; Tremel, W.; et al. A high-performance asymmetric supercapacitor based on tungsten oxide nanoplates and highly reduced graphene oxide electrodes. Chem.-A Eur. J. 2021, 27, 6973–6984. [Google Scholar] [CrossRef]
- Alam, M.W.; Kumar, V.G.D.; Ravikumar, C.R.; Prashantha, S.C.; Murthy, H.C.A.; Kumar, M.R.A. Chromium (III) doped polycrystalline MgAl2O4 nanoparticles for photocatalytic and supercapacitor applications. J. Phys. Chem. Solids 2022, 161, 110491. [Google Scholar] [CrossRef]
- Balaji, T.E.; Das, H.T.; Maiyalagan, T. Recent trends in bimetallic oxides and their composites as electrode materials for supercapacitor applications. ChemElectroChem 2021, 8, 1723–1746. [Google Scholar] [CrossRef]
- Kumar, Y.A.; Kumar, K.D.; Kim, H.-J. Facile preparation of a highly efficient NiZn2O4-NiO nanoflower composite grown on Ni foam as an advanced battery-type electrode material for high-performance electrochemical supercapacitors. Dalton Trans. 2020, 49, 3622–3629. [Google Scholar] [CrossRef]
- Li, M.; Meng, Z.; Feng, R.; Zhu, K.; Zhao, F.; Wang, C.; Wang, J.; Wang, L.; Chu, P.K. Fabrication of Bimetallic Oxides (MCo2O4: M=Cu, Mn) on Ordered Microchannel Electro-Conductive Plate for High-Performance Hybrid Supercapacitors. Sustainability 2021, 13, 9896. [Google Scholar] [CrossRef]
- Wu, C.; Chen, L.; Lou, X.; Ding, M.; Jia, C. Fabrication of cobalt-nickel-zinc ternary oxide nanosheet and applications for supercapacitor electrode. Front. Chem. 2018, 6, 597. [Google Scholar] [CrossRef] [PubMed]
- Parveen, N.; Ansari, S.A.; Cho, M.H. Intercalated reduced graphene oxide and its content effect on the supercapacitance performance of the three dimensional flower-like β-Ni(OH)2 architecture. New J. Chem. 2017, 41, 10467–10475. [Google Scholar] [CrossRef]
- Ansari, S.A.; Khan, N.A.; Hasan, Z.; Shaikh, A.A.; Ferdousi, F.K.; Barai, H.R.; Lopa, N.S.; Rahman, M.M. Electrochemical synthesis of titanium nitride nanoparticles onto titanium foil for electrochemical supercapacitors with ultrafast charge/discharge. Sustain. Energy Fuels 2020, 4, 2480–2490. [Google Scholar] [CrossRef]
- Parveen, N.; Ansari, S.A.; Ansari, S.G.; Fouad, H.; Salam, N.M.; Cho, M.H. Solid-state symmetrical supercapacitor based on hierarchical flower-like nickel sulfide with shape-controlled morphological evolution. Electrochim. Acta 2018, 268, 82–93. [Google Scholar] [CrossRef]
- Ansari, S.A.; Parveen, N.; Kotb, H.M.; Alshoaibi, A. Hydrothermally derived three-dimensional porous hollow double-walled Mn2O3 nanocubes as superior electrode materials for supercapacitor applications. Electrochim. Acta 2020, 355, 136783. [Google Scholar] [CrossRef]
- An, C.; Zhang, Y.; Guo, H.; Wang, Y. Metal oxide-based supercapacitors: Progress and prospectives. Nanoscale Adv. 2019, 1, 4644. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Zhang, B.; Lui, Y.H.; Hu, S. Ni-Mn bimetallic oxide nanosheets as high-performance electrode materials for asymmetric supercapacitors. J. Energy Storage 2019, 25, 100897. [Google Scholar] [CrossRef]
- Wang, X.; Hu, J.; Liu, W.; Wang, G.; An, J.; Lian, J. Ni–Zn binary system hydroxide, oxide and sulfide materials: Synthesis and high supercapacitor performance. J. Mater. Chem. A 2015, 3, 23333. [Google Scholar] [CrossRef]
- Yadav, H.M.; Ramesh, S.; Kumar, K.A.; Shinde, S.; Sandhu, S.; Sivasamy, A.; Shrestha, N.K.; Kim, H.S.; Kim, H.S.; Bathula, C. Impact of polypyrrole incorporation on nickel oxide@multi walled carbon nanotube composite for application in supercapacitors. Polym. Test. 2020, 89, 106727. [Google Scholar] [CrossRef]
- Sahoo, S.; Nguyen, T.T.; Shim, J.-J. Mesoporous Fe-N-Co ternary oxide nanoflake arrays on Ni foam for high-performance supercapacitor applications. J. Ind. Eng. Chem. 2018, 63, 181–190. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, F.; Cheng, J.P.; Zhang, X.B. Binary nickel-cobalt oxides electrode materials for high-performance supercapacitors: Influence of its composition and porous nature. ACS Appl. Mater. Interfaces 2015, 7, 17630–17640. [Google Scholar] [CrossRef]
- Rui, X.; Tan, H.; Yan, Q. Nanostructured metal sulfides for energy storage. Nanoscale 2014, 6, 9889. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Kang, H.; Jin, T.; Jiao, L. Design, synthesis, and energy-related applications of metal sulfides. Mater. Horiz. 2016, 3, 402. [Google Scholar] [CrossRef]
- Wang, P.; Zhou, H.; Meng, C.; Wang, Z.; Akhtar, K.; Yuan, A. Cyanometallic framework-derived hierarchical Co3O4-NiO/graphene foam as high-performance binder-free electrodes for supercapacitors. Chem. Eng. J. 2019, 369, 57–63. [Google Scholar] [CrossRef]
- Xu, X.; Gao, J.; Hong, W. Ni-based chromite spinel for high-performance supercapacitors. RSC Adv. 2016, 6, 29646–29653. [Google Scholar] [CrossRef]
- Xu, L.; Zhao, Y.; Lian, J.; Xu, Y.; Bao, J.; Qiu, J.; Xu, L.; Xu, H.; Hua, M.; Li, H. Morphology controlled preparation of ZnCo2O4 nanostructures for asymmetric supercapacitor with ultrahigh energy density. Energy 2017, 123, 296–304. [Google Scholar] [CrossRef]
- Sahoo, S.; Naik, K.K.; Rout, C.S. Electrodeposition of spinel MnCo2O4 nanosheets for supercapacitor applications. Nanotechnology 2015, 26, 455401. [Google Scholar] [CrossRef]
- Xu, J.; Liu, F.; Peng, X.; Li, J.; Yang, Y.; Jin, D.; Jin, H.; Wang, X.; Hong, B. Hydrothermal synthesis of NiCo2O4/activated carbon composites for supercapacitor with enhanced cycle performance. ChemistrySelect 2017, 2, 5189–5195. [Google Scholar] [CrossRef]
- Yan, D.; Wang, W.; Luo, X.; Chen, C.; Zeng, Y.; Zhu, Z. NiCo2O4 with oxygen vacancies as better performance electrode material for supercapacitor. Chem. Eng. J. 2018, 334, 864–872. [Google Scholar] [CrossRef]
- Sahoo, S.; Shim, J.-J. Facile Synthesis of Three-Dimensional Ternary ZnCo2O4/Reduced Graphene Oxide/NiO Composite Film on Nickel Foam for Next Generation Supercapacitor Electrodes. ACS Sustain. Chem. Eng. 2017, 5, 241–251. [Google Scholar] [CrossRef]
- Wang, S.; Pu, J.; Tong, Y.; Cheng, Y.; Gao, Y.; Wang, Z. ZnCo2O4 nanowire arrays grown on nickel foam for high-performance pseudocapacitors. J. Mater. Chem. A 2014, 2, 5434–5440. [Google Scholar] [CrossRef]
- Guan, B.; Guo, D.; Hu, L.; Zhang, G.; Fu, T.; Ren, W.; Li, J.; Li, Q. Facile synthesis of ZnCo2O4 nanowire cluster arrays on Ni foam for high-performance asymmetric supercapacitors. J. Mater. Chem. A 2014, 2, 16116–16123. [Google Scholar] [CrossRef]
- Shi, X.; Zhou, G. Preparation of zinc-nickel-cobalt ternary oxide nanosheets as electrodes in supercapacitors. Chem. Res. Chin. Univ. 2017, 33, 939–945. [Google Scholar] [CrossRef]
- Fu, W.; Wang, Y.; Han, W.; Zhang, Z.; Zha, H.; Xie, E. Construction of hierarchical ZnCo2O4@NixCo2x(OH)6x core/shell nanowire arrays for high-performance supercapacitors. J. Mater. Chem. A 2016, 4, 173–182. [Google Scholar] [CrossRef]
- Xing, Z.; Chu, Q.; Ren, X.; Ge, C.; Qusti, A.H.; Asiri, A.M.; Al-Youbi, A.O.; Sun, X. Ni3S2 coated ZnO array for high-performance supercapacitors. J. Power Sources 2014, 245, 463–467. [Google Scholar] [CrossRef]
- Yan, H.; Zhang, D.; Xu, J.; Lu, Y.; Liu, Y.; Qiu, K.; Zhang, Y.; Luo, Y. Solution growth of NiO nanosheets supported on Ni foam as high-performance electrodes for supercapacitors. Nanoscale Res. Lett. 2014, 9, 424. [Google Scholar] [CrossRef] [Green Version]
- Kong, L.; Li, X.; Liu, M.; Ma, X.; Luo, Y.; Kang, L. A hydrothermal process for the fabrication of nickel foam based NiO and Co3O4 nanostructures with excellent properties for electrochemical capacitors. Appl. Mech. Mater. 2013, 291–294, 786–790. [Google Scholar] [CrossRef]
- Dhas, S.D.; Maldar, P.S.; Patil, M.D.; Waikar, M.R.; Sonkawade, R.G.; Moholkar, A.V. Sol-gel synthesized nickel oxide nanostructures on nickel foam and nickel mesh for a targeted energy storage application. J. Energy Storage 2021, 103658. [Google Scholar] [CrossRef]
- Hassan, K.; Farzana, R.; Sahajwalla, V. In-situ fabrication of ZnO thin film electrode using spent Zn-C battery and its electrochemical performance for supercapacitance. SN Appl. Sci. 2019, 1, 302. [Google Scholar] [CrossRef] [Green Version]
- Sami, S.K.; Siddiqui, S.; Shrivastava, S.; Lee, N.; Chung, C. The pine-needle-inspired structure of Zinc Oxide nanorods grown on electrospun nanofibers for high-performance flexible supercapacitors. Small 2017, 13, 1702142. [Google Scholar] [CrossRef]
- Abdah, M.A.A.M.; Azman, N.H.N.; Kulandaivalu, S.; Sulaiman, Y. Review of the use of transition-metal-oxide and conducting polymer-based fibres for high-performance supercapacitors. Mater. Des. 2020, 186, 108199. [Google Scholar] [CrossRef]

| No. | Electrode Material | Morphology | Synthesis Method | Synthesis Condition | Current Collector | Electrolyte | Specific Capacitance | Cyclic Stability and Retention | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Cobalt–nickel–zinc ternary oxide | Mesoporous nanosheet | Hydrothermal | 130 °C for 5 h | Ni foam | 6M KOH | 1172.2 C/g (1 A/g) | 81.6% @2000 | [10] |
| 2 | Zinc–nickel–cobalt ternary oxide | Nanosheets | Solvothermal | 120 °C for 7 h | Powder | 6M KOH | 257.5 F/g (0.5 A/g) | 97.18% @2000 | [32] |
| 3 | ZnCo2O4–NixCo2x(OH)6x | Core–shell nanowire | Hydrothermal | 120 °C for 5 h | Ni foam | 2 M KOH | 419.1 F/g (1 A/g) | 81.4% @2000 | [33] |
| 4 | ZnCo2O4/rGO/NiO | Nanowires | Hydrothermal | 210 °C for 24 h | Ni foam | 6 M KOH | 1256 F/g (3 A/g) | 80.2% @3000 | [29] |
| 5 | NiZn2O4–NiO | Nanoleaves with nanoparticles | Hydrothermal | 140 °C for 4 h | Ni foam | 2 M KOH | 1284.2 F/g (1 A/g) | 98.17% @5000 | [8] |
| 6 | ZnO@Ni3S2 | Core–shell nanorods | Electrodeposition | 1.2–0.2 V, 5 mV/s | Ni foam | 2 M KOH | 1529 F/g (2 A/g) | 42% @2000 | [34] |
| 7 | NixZn1xS | Porous spheroid nanoparticles | Modified interface method and hydrothermal | 393 K for 8 h | Powder | 3 M KOH | 1867 F/g (1 A/g) | 77.4% @1000 | [17] |
| 8 | NiO@Ni foam | Nanosheets | Hydrothermal | 200 °C for 24 h | Ni foam | 2 M KOH | 943.5 F/g (5 A/g) | 91.1% @1200 | [35] |
| 9 | NiO coated NF | Nanowires | Hydrothermal | 120 °C for 4 h | Ni Foam | 2 M KOH | 231 F/g (0.5 A/g) | - | [36] |
| 10 | NiO-NF | Nanoparticles | Sol-gel | - | Ni Foam | 4 M KOH | 871 F/g 5 mV/s | 86.5% @10,000 | [37] |
| 11 | ZnO film | Nanoparticles | Spent Zn–C battery | 60 °C for 24 h | Porous silicon substrate | 0.6 M KOH | 547 F/g 5 mV/s | - | [38] |
| 12 | Zinc Oxide Nanorods | Pine-needle nanorod | Electrospinning | 95 °C for 1 h | Nanofiber | 1 M H2SO4 | 56.99 F/g (0.1 mA/cm2) | 78% @5000 | [39] |
| 13 | NiZn2O4@3D-NF | 3D nanosheet assembled flower with several-burr morphology | Solvothermal | 150 °C for 5 h | Ni foam | 2 M KOH | 1706.25 F/g (1.5 A/g) | 88% @5000 | Present work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ansari, S.A.; Parveen, N.; Al-Othoum, M.A.S.; Ansari, M.O. Development of Binder Free Interconnected 3D Flower of NiZn2O4 as an Advanced Electrode Materials for Supercapacitor Applications. Crystals 2022, 12, 14. https://doi.org/10.3390/cryst12010014
Ansari SA, Parveen N, Al-Othoum MAS, Ansari MO. Development of Binder Free Interconnected 3D Flower of NiZn2O4 as an Advanced Electrode Materials for Supercapacitor Applications. Crystals. 2022; 12(1):14. https://doi.org/10.3390/cryst12010014
Chicago/Turabian StyleAnsari, Sajid Ali, Nazish Parveen, Mohd Al Saleh Al-Othoum, and Mohammad Omaish Ansari. 2022. "Development of Binder Free Interconnected 3D Flower of NiZn2O4 as an Advanced Electrode Materials for Supercapacitor Applications" Crystals 12, no. 1: 14. https://doi.org/10.3390/cryst12010014
APA StyleAnsari, S. A., Parveen, N., Al-Othoum, M. A. S., & Ansari, M. O. (2022). Development of Binder Free Interconnected 3D Flower of NiZn2O4 as an Advanced Electrode Materials for Supercapacitor Applications. Crystals, 12(1), 14. https://doi.org/10.3390/cryst12010014







